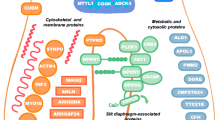
Abstract
Steroid-resistant nephrotic syndrome (SRNS) is a paradigmatic disease to illustrate the major clinical consequences that can arise from genetic testing. Patients with pathogenic variants should be spared inefficient and potentially toxic immunosuppressive therapy; although a partial reduction of proteinuria has been reported with calcineurin inhibitors and/or renin-angiotensin-aldosterone inhibitors in a small number of cases. A better understanding of disease mechanisms may help identifying targeted therapeutic agents, and guide specific management of extra-renal features. Moreover, these patients have a very low risk of recurrence after kidney transplantation. Finally, such diagnosis is crucial for genetic counseling with respect to family planning but also to assess the suitability of relatives as organ donors. Next-generation sequencing approaches are increasingly used as the first step of genetic analyses. Causative pathogenic variants are identified in approximately 30% of children with SRNS and at least 66% in congenital and infantile cases. Among these genes, NPHS1 and NPHS2 are by far the two main autosomal recessive genes implicated in SRNS, while INF2 is the leading cause of autosomal dominant SRNS. Most novel gene variants are rare and involve few families. Moreover, NGS has revealed causative variants in unexpected genes such as ciliary or tubular genes and has widened the phenotypes associated with SRNS. More complex inheritance involving susceptibility variants, oligogenism or interallelic influence of variants have also been described as a cause of SRNS.
Similar content being viewed by others
References
Kestila M, et al. Positionally cloned gene for a novel glomerular protein – nephrin – is mutated in congenital nephrotic syndrome. Mol Cell. 1998;1:575–82.
Boute N, et al. NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resistant nephrotic syndrome. Nat Genet. 2000;24:349–54.
Kaplan JM, et al. Mutations in ACTN4, encoding alpha-actinin-4, cause familial focal segmental glomerulosclerosis. Nat Genet. 2000;24:251–6.
Hinkes B, et al. Specific podocin mutations correlate with age of onset in steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2008;19:365–71.
Philippe A, et al. Nephrin mutations can cause childhood-onset steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2008;19:1871–8.
Sadowski CE, et al. A single-gene cause in 29.5% of cases of steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2015;26:1279–89.
Trautmann A, et al. IPNA clinical practice recommendations for the diagnosis and management of children with steroid-resistant nephrotic syndrome. Pediatr Nephrol. 2020;35:1529–61.
Lovric S, et al. Rapid detection of monogenic causes of childhood-onset steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol. 2014;9:1109–16.
Bullich G, et al. Targeted next-generation sequencing in steroid-resistant nephrotic syndrome: mutations in multiple glomerular genes may influence disease severity. Eur J Hum Genet. 2015;23:1192–9.
McCarthy HJ, et al. Simultaneous sequencing of 24 genes associated with steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol. 2013;8:637–48.
Hinkes BG, et al. Nephrotic syndrome in the first year of life: two thirds of cases are caused by mutations in 4 genes (NPHS1, NPHS2, WT1, and LAMB2). Pediatrics. 2007;119:e907–19.
Caridi G, et al. Broadening the spectrum of diseases related to podocin mutations. J Am Soc Nephrol. 2003;14:1278–86.
Weber S, et al. NPHS2 mutation analysis shows genetic heterogeneity of steroid-resistant nephrotic syndrome and low post-transplant recurrence. Kidney Int. 2004;66:571–9.
Ruf RG, et al. Patients with mutations in NPHS2 (podocin) do not respond to standard steroid treatment of nephrotic syndrome. J Am Soc Nephrol. 2004;15:722–32.
Trautmann A, et al. Spectrum of steroid-resistant and congenital nephrotic syndrome in children: the PodoNet registry cohort. Clin J Am Soc Nephrol. 2015;10:592–600.
Bouchireb K, et al. NPHS2 mutations in steroid-resistant nephrotic syndrome: a mutation update and the associated phenotypic spectrum. Hum Mutat. 2014;35:178–86.
Machuca E, et al. Genotype-phenotype correlations in non-Finnish congenital nephrotic syndrome. J Am Soc Nephrol. 2010;21:1209–17.
Frishberg Y, et al. The heart of children with steroid-resistant nephrotic syndrome: is it all podocin? J Am Soc Nephrol. 2006;17:227–31.
Caridi G, et al. Lack of cardiac anomalies in children with NPHS2 mutations. Nephrol Dial Transplant. 2007;22:1477–9.
Roselli S, Moutkine I, Gribouval O, Benmerah A, Antignac C. Plasma membrane targeting of podocin through the classical exocytic pathway: effect of NPHS2 mutations. Traffic. 2004;5:37–44.
Nishibori Y, et al. Disease-causing missense mutations in NPHS2 gene alter normal nephrin trafficking to the plasma membrane. Kidney Int. 2004;66:1755–65.
Huber TB, et al. Molecular basis of the functional podocin-nephrin complex: mutations in the NPHS2 gene disrupt nephrin targeting to lipid raft microdomains. Hum Mol Genet. 2003;12:3397–405.
Ohashi T, Uchida K, Uchida S, Sasaki S, Nihei H. Intracellular mislocalization of mutant podocin and correction by chemical chaperones. Histochem Cell Biol. 2003;119:257–64.
Koziell A, et al. Genotype/phenotype correlations of NPHS1 and NPHS2 mutations in nephrotic syndrome advocate a functional inter-relationship in glomerular filtration. Hum Mol Genet. 2002;11:379–88.
Pereira AC, et al. NPHS2 R229Q functional variant is associated with microalbuminuria in the general population. Kidney Int. 2004;65:1026–30.
Tsukaguchi H, et al. NPHS2 mutations in late-onset focal segmental glomerulosclerosis: R229Q is a common disease-associated allele. J Clin Invest. 2002;110:1659–66.
Machuca E, et al. Clinical and epidemiological assessment of steroid-resistant nephrotic syndrome associated with the NPHS2 R229Q variant. Kidney Int. 2009;75:727–35.
Berdeli A, et al. NPHS2 (podicin) mutations in Turkish children with idiopathic nephrotic syndrome. Pediatr Nephrol. 2007;22:2031–40.
Karle SM, et al. Novel mutations in NPHS2 detected in both familial and sporadic steroid-resistant nephrotic syndrome. J Am Soc Nephrol. 2002;13:388–93.
Lipska BS, et al. Genetic screening in adolescents with steroid-resistant nephrotic syndrome. Kidney Int. 2013;84:206–13.
Gribouval O, et al. Identification of genetic causes for sporadic steroid-resistant nephrotic syndrome in adults. Kidney Int. 2018;94:1013–22.
Tory K, et al. Mutation-dependent recessive inheritance of NPHS2-associated steroid-resistant nephrotic syndrome. Nat Genet. 2014;46:299–304.
Straner P, et al. C-terminal oligomerization of podocin mediates interallelic interactions. Biochim Biophys Acta Mol basis Dis. 2018;1864:2448–57.
Rood IM, Deegens JK, Wetzels JF. Genetic causes of focal segmental glomerulosclerosis: implications for clinical practice. Nephrol Dial Transplant. 2012;27:882–90.
Santín S, et al. Nephrin mutations cause childhood- and adult-onset focal segmental glomerulosclerosis. Kidney Int. 2009;76:1268–76.
Hinkes B, et al. Positional cloning uncovers mutations in PLCE1 responsible for a nephrotic syndrome variant that may be reversible. Nat Genet. 2006;38:1397–405.
Gbadegesin R, et al. Mutations in PLCE1 are a major cause of isolated diffuse mesangial sclerosis (IDMS). Nephrol Dial Transplant. 2008;23:1291–7.
Boyer O, et al. Mutational analysis of the PLCE1 gene in steroid resistant nephrotic syndrome. J Med Genet. 2010;47:445–52.
Mele C, et al. MYO1E mutations and childhood familial focal segmental glomerulosclerosis. N Engl J Med. 2011;365:295–306.
Ozaltin F, et al. Disruption of PTPRO causes childhood-onset nephrotic syndrome. Am J Hum Genet. 2011;89:139–47.
Ebarasi L, et al. Defects of CRB2 cause steroid-resistant nephrotic syndrome. Am J Hum Genet. 2015;96:153–61.
Slavotinek A, et al. CRB2 mutations produce a phenotype resembling congenital nephrosis, Finnish type, with cerebral ventriculomegaly and raised alpha-fetoprotein. Am J Hum Genet. 2015;96:162–9.
Kim JM, et al. CD2-associated protein haploinsufficiency is linked to glomerular disease susceptibility. Science. 2003;300:1298–300.
Takano T, et al. Recessive mutation in CD2AP causes focal segmental glomerulosclerosis in humans and mice. Kidney Int. 2019;95:57–61.
Löwik MM, et al. Focal segmental glomerulosclerosis in a patient homozygous for a CD2AP mutation. Kidney Int. 2007;72:1198–203.
Brown EJ, et al. Mutations in the formin gene INF2 cause focal segmental glomerulosclerosis. Nat Genet. 2010;42:72–6.
Boyer O, et al. Mutations in INF2 are a major cause of autosomal dominant focal segmental glomerulosclerosis. J Am Soc Nephrol. 2011;22:239–45.
Barua M, et al. Mutations in the INF2 gene account for a significant proportion of familial but not sporadic focal and segmental glomerulosclerosis. Kidney Int. 2013;83:316–22.
Gbadegesin RA, et al. Inverted formin 2 mutations with variable expression in patients with sporadic and hereditary focal and segmental glomerulosclerosis. Kidney Int. 2012;81:94–9.
Boyer O, et al. INF2 mutations in Charcot-Marie-Tooth disease with glomerulopathy. N Engl J Med. 2011;365:2377–88.
Büscher AK, et al. Mutations in INF2 may be associated with renal histology other than focal segmental glomerulosclerosis. Pediatr Nephrol. 2018;33:433–7.
Challis RC, et al. Thrombotic microangiopathy in inverted formin 2-mediated renal disease. J Am Soc Nephrol. 2017;28:1084–91.
Winn MP, et al. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science. 2005;308:1801–4.
Heeringa SF, et al. A novel TRPC6 mutation that causes childhood FSGS. PLoS One. 2009;4:e7771.
Santin S, et al. TRPC6 mutational analysis in a large cohort of patients with focal segmental glomerulosclerosis. Nephrol Dial Transplant. 2009;24:3089–96.
Reiser J, et al. TRPC6 is a glomerular slit diaphragm-associated channel required for normal renal function. Nat Genet. 2005;37:739–44.
Gigante M, et al. TRPC6 mutations in children with steroid-resistant nephrotic syndrome and atypical phenotype. Clin J Am Soc Nephrol. 2011;6:1626–34.
Lion M, et al. Mutation de TRPC6: a rare cause of steroid-resistant nephrotic syndrome in children. Pediatr Nephrol. 2012;27:1702.
Wang M, et al. Two children with novel TRPC6 spontaneous missense mutations and atypical phenotype: a case report and literature review. Front Pediatr. 2020;8:269.
Nagano C, et al. Comprehensive genetic diagnosis of Japanese patients with severe proteinuria. Sci Rep. 2020;10:270.
Lipska BS, et al. Genotype-phenotype associations in WT1 glomerulopathy. Kidney Int. 2014;85:1169–78.
Lipska-Ziętkiewicz BS. WT1 disorder. In: Adam MP et al., editors. GeneReviews®. Seattle: University of Washington; 1993–2020.
Chernin G, et al. Genotype/phenotype correlation in nephrotic syndrome caused by WT1 mutations. Clin J Am Soc Nephrol. 2010;5:1655–62.
Lipska-Ziętkiewicz BS, et al. Genetic aspects of congenital nephrotic syndrome: a consensus statement from the ERKNet-ESPN inherited glomerulopathy working group. Eur J Hum Genet. 2020; https://doi.org/10.1038/s41431-020-0642-8.
Lehnhardt A, et al. Clinical and molecular characterization of patients with heterozygous mutations in Wilms tumor suppressor gene 1. Clin J Am Soc Nephrol. 2015;10:825–31.
Boyer O, Dorval G, Servais A. Hereditary podocytopathies in adults: the next generation. Kidney Dis (Basel). 2017;3:50–6.
Mathis BJ, et al. A locus for inherited focal segmental glomerulosclerosis maps to chromosome 19q13. Kidney Int. 1998;53:282–6.
Pollak MR, Alexander MP, Henderson JM. A case of familial kidney disease. Clin J Am Soc Nephrol. 2007;2:1367–74.
Choi HJ, et al. Familial focal segmental glomerulosclerosis associated with an ACTN4 mutation and paternal germline mosaicism. Am J Kidney Dis. 2008;51:834–8.
Henderson JM, Alexander MP, Pollak MR. Patients with ACTN4 mutations demonstrate distinctive features of glomerular injury. J Am Soc Nephrol. 2009;20:961–8.
Akilesh S, et al. Arhgap24 inactivates Rac1 in mouse podocytes, and a mutant form is associated with familial focal segmental glomerulosclerosis. J Clin Invest. 2011;121:4127–37.
Gbadegesin RA, et al. Mutations in the gene that encodes the F-actin binding protein anillin cause FSGS. J Am Soc Nephrol. 2014;25:1991–2002.
Dorval G, et al. TBC1D8B loss-of-function mutations lead to x-linked nephrotic syndrome via defective trafficking pathways. Am J Hum Genet. 2019;104:348–55.
Drash A, Sherman F, Hartmann WH, Blizzard RM. A syndrome of pseudohermaphroditism, Wilms’ tumor, hypertension, and degenerative renal disease. J Pediatr. 1970;76:585–93.
Roca N, et al. Long-term outcome in a case series of Denys-Drash syndrome. Clin Kidney J. 2019;12:836–9.
Frasier SD, Bashore RA, Mosier HD. Gonadoblastoma associated with pure gonadal dysgenesis in monozygous twins. J Pediatr. 1964;64:740–5.
Niaudet P, Gubler M-C. WT1 and glomerular diseases. Pediatr Nephrol. 2006;21:1653–60.
Ahn YH, et al. Genotype-phenotype analysis of pediatric patients with WT1 glomerulopathy. Pediatr Nephrol. 2017;32:81–9.
Sinha A, et al. Frasier syndrome: early gonadoblastoma and cyclosporine responsiveness. Pediatr Nephrol. 2010;25:2171–4.
Melo KFS, et al. An unusual phenotype of Frasier syndrome due to IVS9 +4C>T mutation in the WT1 gene: predominantly male ambiguous genitalia and absence of gonadal dysgenesis. J Clin Endocrinol Metab. 2002;87:2500–5.
Zenker M, et al. Human laminin beta2 deficiency causes congenital nephrosis with mesangial sclerosis and distinct eye abnormalities. Hum Mol Genet. 2004;13:2625–32.
Pierson M, Cordier J, Hervouuet F, Rauber G. An unusual congenital and familial congenital malformative combination involving the eye and kidney. J Genet Hum. 1963;12:184–213.
Choi HJ, et al. Variable phenotype of Pierson syndrome. Pediatr Nephrol. 2008;23:995–1000.
Zemrani B, et al. A novel LAMB2 gene mutation associated with a severe phenotype in a neonate with Pierson syndrome. Eur J Med Res. 2016;21:19.
Hasselbacher K, et al. Recessive missense mutations in LAMB2 expand the clinical spectrum of LAMB2-associated disorders. Kidney Int. 2006;70:1008–12.
Matejas V, et al. Mutations in the human laminin beta2 (LAMB2) gene and the associated phenotypic spectrum. Hum Mutat. 2010;31:992–1002.
Tahoun M, et al. Mutations in LAMB2 are associated with albuminuria and optic nerve hypoplasia with hypopituitarism. J Clin Endocrinol Metab. 2020;105:595.
Bredrup C, et al. Ophthalmological aspects of Pierson syndrome. Am J Ophthalmol. 2008;146:602–11.
Mohney BG, et al. A novel mutation of LAMB2 in a multigenerational mennonite family reveals a new phenotypic variant of Pierson syndrome. Ophthalmology. 2011;118:1137–44.
Wühl E, et al. Neurodevelopmental deficits in Pierson (microcoria-congenital nephrosis) syndrome. Am J Med Genet A. 2007;143:311–9.
Galloway WH, Mowat AP. Congenital microcephaly with hiatus hernia and nephrotic syndrome in two sibs. J Med Genet. 1968;5:319–21.
Colin E, et al. Loss-of-function mutations in WDR73 are responsible for microcephaly and steroid-resistant nephrotic syndrome: Galloway-Mowat syndrome. Am J Hum Genet. 2014;95:637–48.
Steiss JO, Gross S, Neubauer BA, Hahn A. Late-onset nephrotic syndrome and severe cerebellar atrophy in Galloway-Mowat syndrome. Neuropediatrics. 2005;36:332–5.
Sartelet H, et al. Collapsing glomerulopathy in Galloway-Mowat syndrome: a case report and review of the literature. Pathol Res Pract. 2008;204:401–6.
Vodopiutz J, et al. WDR73 mutations cause infantile neurodegeneration and variable glomerular kidney disease. Hum Mutat. 2015;36:1021–8.
Rosti RO, et al. Homozygous mutation in NUP107 leads to microcephaly with steroid-resistant nephrotic condition similar to Galloway-Mowat syndrome. J Med Genet. 2017;54:399–403.
Ben-Omran T, et al. Nonsense mutation in the WDR73 gene is associated with Galloway-Mowat syndrome. J Med Genet. 2015;52:381–90.
Braun D, et al. Mutations in KEOPS-complex genes cause nephrotic syndrome with primary microcephaly. Nat Genet. 2017;49:1529–38.
Arrondel C, et al. Defects in t(6)A tRNA modification due to GON7 and YRDC mutations lead to Galloway-Mowat syndrome. Nat Commun. 2019;10:3967.
Braun DA, et al. Mutations in WDR4 as a new cause of Galloway-Mowat syndrome: mutations in WDR4 as a new cause of Galloway-Mowat syndrome. Am J Med Genet A. 2018;176:2460–5.
Gee HY, et al. ARHGDIA mutations cause nephrotic syndrome via defective RHO GTPase signaling. J Clin Invest. 2013;123:3243–53.
Gee HY, et al. KANK deficiency leads to podocyte dysfunction and nephrotic syndrome. J Clin Invest. 2015;125:2375–84.
Skre H. Genetic and clinical aspects of Charcot-Marie-Tooth’s disease. Clin Genet. 1974;6:98–118.
Schimke RN, Horton WA, King CR, Martin NL. Chondroitin-6-sulfate mucopoly-saccharidosis in conjunction with lymphopenia, defective cellular immunity and the nephrotic syndrome. Birth Defects Orig Artic Ser. 1974;10:258–66.
Boerkoel CF, et al. Mutant chromatin remodeling protein SMARCAL1 causes Schimke immuno-osseous dysplasia. Nat Genet. 2002;30:215–20.
Lipska-Ziętkiewicz B, et al. Low renal but high extrarenal phenotype variability in Schimke immuno-osseous dysplasia. PLoS One. 2017;12:e0180926.
Baradaran-Heravi A, et al. Bone marrow transplantation in Schimke immuno-osseous dysplasia. Am J Med Genet A. 2013;161A:2609–13.
Zuber J, et al. Donor-targeted serotherapy as a rescue therapy for steroid-resistant acute GVHD after HLA-mismatched kidney transplantation. Am J Transplant. 2020;20:2243–53.
Hawkins CF, Smith OE. Renal dysplasia in a family with multiple hereditary abnormalities including iliac horns. Lancet. 1950;1:803–8.
Dreyer SD, et al. Mutations in LMX1B cause abnormal skeletal patterning and renal dysplasia in nail patella syndrome. Nat Genet. 1998;19:47–50.
Boyer O, et al. LMX1B mutations cause hereditary FSGS without extrarenal involvement. J Am Soc Nephrol. 2013;24:1216–22.
Has C, et al. Integrin α3 mutations with kidney, lung, and skin disease. N Engl J Med. 2012;366:1508–14.
Prasad R, et al. Sphingosine-1-phosphate lyase mutations cause primary adrenal insufficiency and steroid-resistant nephrotic syndrome. J Clin Invest. 2017;127:942–53.
Lovric S, et al. Mutations in sphingosine-1-phosphate lyase cause nephrosis with ichthyosis and adrenal insufficiency. J Clin Invest. 2017;127:912–28.
Najafian B, et al. Accumulation of globotriaosylceramide in podocytes in fabry nephropathy is associated with progressive podocyte loss. J Am Soc Nephrol. 2020;31:865–75.
Bonten EJ, et al. Novel mutations in lysosomal neuraminidase identify functional domains and determine clinical severity in sialidosis. Hum Mol Genet. 2000;9:2715–25.
Maroofian R, et al. Parental whole-exome sequencing enables sialidosis type ii diagnosis due to an NEU1 missense mutation as an underlying cause of nephrotic syndrome in the child. Kidney Int Rep. 2018;3:1454–63.
Chen W, et al. Histological studies of renal biopsy in a boy with nephrosialidosis. Ultrastruct Pathol. 2011;35:168–71.
Berkovic SF, et al. Array-based gene discovery with three unrelated subjects shows SCARB2/LIMP-2 deficiency causes myoclonus epilepsy and glomerulosclerosis. Am J Hum Genet. 2008;82:673–84.
Reczek D, et al. LIMP-2 is a receptor for lysosomal mannose-6-phosphate-independent targeting of beta-glucocerebrosidase. Cell. 2007;131:770–83.
Badhwar A, et al. Action myoclonus-renal failure syndrome: characterization of a unique cerebro-renal disorder. Brain. 2004;127:2173–82.
National Cancer Institute. https://www.cancer.gov/publications/dictionaries/genetics-dictionary/def/phenocopy. Accessed 9/7/2020.
Malone AF, et al. Rare hereditary COL4A3/COL4A4 variants may be mistaken for familial focal segmental glomerulosclerosis. Kidney Int. 2014;86:1253–9.
Barua M, et al. Mutations in PAX2 associate with adult-onset FSGS. J Am Soc Nephrol. 2014;25:1942–53.
Huynh Cong E, et al. A homozygous missense mutation in the ciliary gene TTC21B causes familial FSGS. J Am Soc Nephrol. 2014;25:2435–43.
Ovunc B, et al. Exome sequencing reveals cubilin mutation as a single-gene cause of proteinuria. J Am Soc Nephrol. 2011;22:1815–20.
Bedin M, et al. Human C-terminal CUBN variants associate with chronic proteinuria and normal renal function. J Clin Invest. 2019; https://doi.org/10.1172/JCI129937.
Warejko JK, et al. Whole exome sequencing of patients with steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol. 2018;13:53–62.
Landini S, et al. Reverse phenotyping after whole-exome sequencing in steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol. 2020;15:89–100.
Genovese G, et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science. 2010;329:841–5.
Kopp JB, et al. Clinical features and histology of apolipoprotein L1-associated nephropathy in the FSGS clinical trial. J Am Soc Nephrol. 2015;26:1443–8.
Adeyemo A, et al. HLA-DQA1 and APOL1 as risk loci for childhood-onset steroid-sensitive and steroid-resistant nephrotic syndrome. Am J Kidney Dis. 2018;71:399–406.
Kopp JB, et al. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol. 2011;22:2129–37.
Gribouval O, et al. APOL1 risk genotype in European steroid-resistant nephrotic syndrome and/or focal segmental glomerulosclerosis patients of different African ancestries. Nephrol Dial Transplant. 2018; https://doi.org/10.1093/ndt/gfy176.
Ekulu PM, et al. A focus on the association of Apol1 with kidney disease in children. Pediatr Nephrol. 2020; https://doi.org/10.1007/s00467-020-04553-z.
Löwik M, et al. Bigenic heterozygosity and the development of steroid-resistant focal segmental glomerulosclerosis. Nephrol Dial Transplant. 2008;23:3146–51.
Voskarides K, et al. Evidence that NPHS2-R229Q predisposes to proteinuria and renal failure in familial hematuria. Pediatr Nephrol. 2012;27:675–9.
Goncalves S, et al. A homozygous KAT2B variant modulates the clinical phenotype of ADD3 deficiency in humans and flies. PLoS Genet. 2018;14:e1007386.
Gee HY, et al. Mutations in EMP2 cause childhood-onset nephrotic syndrome. Am J Hum Genet. 2014;94:884–90.
Karp AM, Gbadegesin RA. Genetics of childhood steroid-sensitive nephrotic syndrome. Pediatr Nephrol. 2017;32:1481–8.
Lane BM, Cason R, Esezobor CI, Gbadegesin RA. Genetics of childhood steroid sensitive nephrotic syndrome: an update. Front Pediatr. 2019;7:8.
Gbadegesin RA, et al. HLA-DQA1 and PLCG2 are candidate risk loci for childhood-onset steroid-sensitive nephrotic syndrome. J Am Soc Nephrol. 2015;26:1701–10.
Jia X, et al. Common risk variants in NPHS1 and TNFSF15 are associated with childhood steroid-sensitive nephrotic syndrome. Kidney Int. 2020;98:1308–22.
Jia X, et al. Strong association of the HLA-DR/DQ locus with childhood steroid-sensitive nephrotic syndrome in the Japanese population. J Am Soc Nephrol. 2018;29:2189–99.
Dufek S, et al. Genetic identification of two novel loci associated with steroid-sensitive nephrotic syndrome. J Am Soc Nephrol. 2019;30:1375–84.
Debiec H, et al. Transethnic, genome-wide analysis reveals immune-related risk alleles and phenotypic correlates in pediatric steroid-sensitive nephrotic syndrome. J Am Soc Nephrol. 2018;29:2000–13.
Kari JA, Sinnott P, Khan H, Trompeter RS, Snodgrass GJ. Familial steroid-responsive nephrotic syndrome and HLA antigens in Bengali children. Pediatr Nephrol. 2001;16:346–9.
Landau D, et al. Familial steroid-sensitive nephrotic syndrome in Southern Israel: clinical and genetic observations. Pediatr Nephrol. 2007;22:661–9.
Moncrieff MW, et al. The familial nephrotic syndrome. II. A clinicopathological study. Clin Nephrol. 1973;1:220–9.
Motoyama O, Sugawara H, Hatano M, Fujisawa T, Iitaka K. Steroid-sensitive nephrotic syndrome in two families. Clin Exp Nephrol. 2009;13:170–3.
Xia Y, et al. Familial steroid-sensitive idiopathic nephrotic syndrome: seven cases from three families in China. Clinics (Sao Paulo). 2013;68:628–31.
Dorval G, et al. Clinical and genetic heterogeneity in familial steroid-sensitive nephrotic syndrome. Pediatr Nephrol. 2018;33:473–83.
Park E, et al. Familial IPEX syndrome: different glomerulopathy in two siblings. Pediatr Int. 2015;57:e59–61.
Lahdenkari A-T, Kestilä M, Holmberg C, Koskimies O, Jalanko H. Nephrin gene (NPHS1) in patients with minimal change nephrotic syndrome (MCNS). Kidney Int. 2004;65:1856–63.
Roberts ISD, Gleadle JM. Familial nephropathy and multiple exostoses with exostosin-1 (EXT1) gene mutation. J Am Soc Nephrol. 2008;19:450–3.
Kitamura A, et al. A familial childhood-onset relapsing nephrotic syndrome. Kidney Int. 2007;71:946–51.
Faul C, et al. The actin cytoskeleton of kidney podocytes is a direct target of the antiproteinuric effect of cyclosporine A. Nat Med. 2008;14:931–8.
Li X, et al. Cyclosporine A protects podocytes via stabilization of cofilin-1 expression in the unphosphorylated state. Exp Biol Med (Maywood). 2014;239:922–36.
Xing C-Y, et al. Direct effects of dexamethasone on human podocytes. Kidney Int. 2006;70:1038–45.
Ashraf S, et al. Mutations in six nephrosis genes delineate a pathogenic pathway amenable to treatment. Nat Commun. 2018;9:1960.
Buscher AK, et al. Rapid response to cyclosporin A and favorable renal outcome in nongenetic versus genetic steroid-resistant nephrotic syndrome. Clin J Am Soc Nephrol. 2016;11:245–53.
Bensimhon AR, Williams AE, Gbadegesin RA. Treatment of steroid-resistant nephrotic syndrome in the genomic era. Pediatr Nephrol. 2019;34:2279–93.
Hall G, Gbadegesin RA. Translating genetic findings in hereditary nephrotic syndrome: the missing loops. Am J Physiol Renal Physiol. 2015;309:F24–8.
Montini G, Malaventura C, Salviati L. Early coenzyme Q10 supplementation in primary coenzyme Q10 deficiency. N Engl J Med. 2008;358:2849–50.
Ashraf S, et al. ADCK4 mutations promote steroid-resistant nephrotic syndrome through CoQ10 biosynthesis disruption. J Clin Invest. 2013;123:5179–89.
Vivante A, Hildebrandt F. Exploring the genetic basis of early-onset chronic kidney disease. Nat Rev Nephrol. 2016;12:133–46.
Gupta IR, et al. ARHGDIA: a novel gene implicated in nephrotic syndrome. J Med Genet. 2013;50:330–8.
Hölttä T, et al. Good long-term renal graft survival and low incidence of cardiac pathology in adults after short dialysis period and renal transplantation in early childhood – a cohort study. Transpl Int. 2020;33:89–97.
Holtta T, et al. Timing of renal replacement therapy does not influence survival and growth in children with congenital nephrotic syndrome caused by mutations in NPHS1: data from the ESPN/ERA-EDTA Registry. Pediatr Nephrol. 2016;31:2317–25.
Gross O, Weber M, Fries JWU, Müller G-A. Living donor kidney transplantation from relatives with mild urinary abnormalities in Alport syndrome: long-term risk, benefit and outcome. Nephrol Dial Transplant. 2009;24:1626–30.
Ding WY, et al. Initial steroid sensitivity in children with steroid-resistant nephrotic syndrome predicts post-transplant recurrence. J Am Soc Nephrol. 2014;25:1342–8.
Bierzynska A, Saleem MA. Deriving and understanding the risk of post-transplant recurrence of nephrotic syndrome in the light of current molecular and genetic advances. Pediatr Nephrol. 2018;33:2027–35.
Morello W, et al. Post-transplant recurrence of steroid resistant nephrotic syndrome in children: the Italian experience. J Nephrol. 2020;33:849–57.
Patrakka J, et al. Recurrence of nephrotic syndrome in kidney grafts of patients with congenital nephrotic syndrome of the Finnish type: role of nephrin. Transplantation. 2002;73:394–403.
Battelino N, Arnol M, Kandus A, Ponikvar R, Novljan G. Post-transplant recurrence of focal segmental glomerulosclerosis in a child with heterozygous mutations in NPHS1 and NPHS2. Ther Apher Dial. 2016;20:312–7.
Holmberg C, Jalanko H. Congenital nephrotic syndrome and recurrence of proteinuria after renal transplantation. Pediatr Nephrol. 2014;29:2309–17.
Srivastava T, et al. Recurrence of proteinuria following renal transplantation in congenital nephrotic syndrome of the Finnish type. Pediatr Nephrol. 2006;21:711–8.
Ghiggeri GM, et al. Posttransplant recurrence of proteinuria in a case of focal segmental glomerulosclerosis associated with WT1 mutation. Am J Transplant. 2006;6:2208–11.
Seeman T, Vondrak K. First report of recurrent nephrotic syndrome after kidney transplantation in a patient with NUP93 gene mutations: a case report. Transplant Proc. 2018;50:3954–6.
Bertelli R, et al. Recurrence of focal segmental glomerulosclerosis after renal transplantation in patients with mutations of podocin. Am J Kidney Dis. 2003;41:1314–21.
Becker-Cohen R, et al. Recurrent nephrotic syndrome in homozygous truncating NPHS2 mutation is not due to anti-podocin antibodies. Am J Transplant. 2007;7:256–60.
Rossanti R, et al. Molecular assay for an intronic variant in NUP93 that causes steroid resistant nephrotic syndrome. J Hum Genet. 2019;64:673–9.
Bierzynska A, et al. MAGI2 mutations cause congenital nephrotic syndrome. J Am Soc Nephrol. 2017;28:1614–21.
Braun DA, et al. Mutations in nuclear pore genes NUP93, NUP205 and XPO5 cause steroid-resistant nephrotic syndrome. Nat Genet. 2016;48:457–65.
Jeanpierre C, et al. Identification of constitutional WT1 mutations, in patients with isolated diffuse mesangial sclerosis, and analysis of genotype/phenotype correlations by use of a computerized mutation database. Am J Hum Genet. 1998;62:824–33.
Goto Y, Nonaka I, Horai S. A mutation in the tRNA(Leu)(UUR) gene associated with the MELAS subgroup of mitochondrial encephalomyopathies. Nature. 1990;348(6302):651–3.
Quinzii C, Naini A, Salviati L, Trevisson E, Navas P, Dimauro S, et al. A mutation in para-hydroxybenzoate-polyprenyl transferase (COQ2) causes primary coenzyme Q10 deficiency. Am J Hum Genet. 2006;78(2):345–9.
Lopez LC, Schuelke M, Quinzii CM, Kanki T, Rodenburg RJ, Naini A, et al. Leigh syndrome with nephropathy and CoQ10 deficiency due to decaprenyl diphosphate synthase subunit 2 (PDSS2) mutations. Am J Hum Genet. 2006;79(6):1125–9.
Heeringa SF, Chernin G, Chaki M, Zhou W, Sloan AJ, Ji Z, et al. COQ6 mutations in human patients producae nephrotic syndrome with sensorineural deafness. J Clin Invest. 2011;121(5):2013–24.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer-Verlag GmbH Germany, part of Springer Nature
About this entry
Cite this entry
Boyer, O., Gbadegesin, R., Waters, A. (2021). Clinical Aspects of Genetic Forms of Nephrotic Syndrome. In: Emma, F., Goldstein, S., Bagga, A., Bates, C.M., Shroff, R. (eds) Pediatric Nephrology. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-27843-3_91-1
Download citation
DOI: https://doi.org/10.1007/978-3-642-27843-3_91-1
Received:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-27843-3
Online ISBN: 978-3-642-27843-3
eBook Packages: Springer Reference MedicineReference Module Medicine