Abstract
The metabolic engineering of plants offers the opportunity to change the content of specific phytonutrients in plant – derived foods.
The plant polyphenol trans-resveratrol (3, 5, 4′-trihydroxystilbene), mainly found in grape, peanut, and other few plants, displays a wide range of biological effects. Numerous in vitro studies have described various biological effects of resveratrol. In order to provide more information regarding absorption, metabolism, and bioavailability of resveratrol, different research approaches have been performed. In recent years, the induction of resveratrol synthesis in plants which normally do not accumulate such polyphenol has been successfully achieved by molecular engineering.
In this context, the ectopic production of resveratrol could have positive effects on the enhancement of the nutritional value of several widely consumed fruits and vegetables. This chapter focuses on the latest findings regarding on resveratrol bioproduction in tomato (Solanum lycopersicum) fruits.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
1 Introduction
Because of resveratrol capacity to confer disease resistance in grapevine and given its clinically useful, most interest has centered, in recent years, on STS gene transfer from grapevine to the genome of numerous plants, with the objective of increasing their tolerance to pathogenic microorganisms and/or improving the of food products through the expression of resveratrol in plants incapable of synthesizing this compound. Studies have been published describing STS-encoding genes isolated from grapevine or other plant species and their use for plant genetic transformation. This chapter explores recent research on the gene transfer of grape STS-encoding genes to tomato plants.
1.1 Overview on Genetics, Biochemistry, and Molecular Biology of Stilbene Synthase
Bioactive ingredients of functional foods help to overcome nutritional deficiencies, and the demand of healthy food products will certainly increase in the future. Plant secondary metabolites are nutritional components of our daily diet.
Stilbenes are small naturally occurring phenolic compounds found in a wide range of plant – derived food – among which berries are important sources [1, 2]. Resveratrol is biosynthetically correlated to stilbenes, but its biosynthesis is restricted to only a few plant species commonly used for human consumption [3–9].
The most abundant levels of naturally occurring resveratrol are found in the roots of Japanese Polygonum cuspidatum which has been used in traditional Asian herb medicine, for hundreds of years, in the treatment of inflammation [5]. Grape (Vitis vinifera L.) is probably the most important source of resveratrol, since the compound is also found in wine. The economic importance of grapevine has encouraged many researchers to study the physiological and molecular basis of berry physiology, particularly those processes affecting wine quality [10]. In this species, a large array of STS genes has been identified thus suggesting the importance of stilbene metabolism [11].
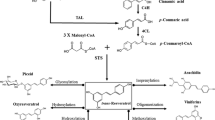
Resveratrol and its 3-O-β-d-glucoside derivative share a chemical structure similar to that of other polyphenol-type stilbenes (Fig. 51.1a) and are controlled by the key enzyme, stilbene synthase (STS), which belongs to a multigene family of the type III group of the polyketide synthase superfamily [3, 4]. STS catalyzes the condensation of three molecules of coumaroyl-CoA to form resveratrol. Synthesis of resveratrol takes place in a single enzymatic step with CoA-esters of cinnamic acid derivatives and 3 malonyl-CoA units as starting blocks (Fig. 51.1b). STS and chalcone synthase (CHS) that are key enzymes of the flavonoid pathway share a high degree of homology and competing for the same substrates.
1.2 Stilbenes Functions in Plants and in Humans
In nature, the most abundant form of resveratrol would appear to be the 5,3,4′-dihydroxystilbene-3-O-β-d-glucopyranoside [12]. Resveratrol exists in two stereoisoforms with cis- or trans-configuration, the latter being the most widely studied, although cis-resveratrol may also possess health-promoting properties. The number as well as the position of moieties play an important role in the biological activity of the compound [13].
Synthesis of resveratrol in plants tissues is either constitutive or inducible being strongly enhanced by fungal attacks, UV irradiation, and other environmental stress conditions [3–5].
Resveratrol possesses numerous important bioactivities including anti-inflammatory, antioxidant, anti-aggregatory functions, and modulation of lipoprotein metabolism [14]. It has also been shown to possess chemopreventive properties against certain forms of cancer and cardiovascular disorders [15]. Subsequent work has shown that resveratrol extends the life spans of lower eukaryotes [16–18]. In mice, long-term administration of resveratrol-induced gene expression patterns that resembled those induced by calorie restriction and delayed aging-related deterioration [19]. Resveratrol also decreased insulin resistance in type 2 diabetic patients [20], suggesting that the pathway targeted by resveratrol might be important for developing therapies for type 2 diabetes.
An important mediator of the metabolic effects of resveratrol [21, 22] is the peroxisome proliferator-activated receptor g coactivator, PGC-1α [23]. Consistent with the known ability of Sirt1 to deacetylate and activate PGC-1α [24, 25], resveratrol increased Sirt1 and PGC-1α activity in mice fed a high-fat diet [21, 22].
AMPK, an emerging key regulator of whole-body metabolism, has been shown to activate Sirt1 and PGC-1α [26–28]. Recently it was shown that AMPK-deficient mice are resistant to the metabolic effects of resveratrol, providing evidence that AMPK is a key mediator of the metabolic benefits produced by resveratrol [27]. Resveratrol directly inhibits cAMP-specific phosphodiesterases and the cAMP effector protein Epac1, which leads to the activation of AMPK and Sirt1 [27].
2 Resveratrol Synthesis in Transgenic Tomato Fruits
Plant metabolic engineering has provided a means to improve polyphenol composition and levels. For example, tomato plants with enhanced flavonols have been recently developed through the overexpression of a transcription factor able to activate flavonol biosynthesis [30–32].
STS structural genes have been transferred to a number of crops, either to improve the resistance to stresses or the nutritional value of the plant (for a review see ref. [33] and Table 51.1).
Few articles reported on the production of stilbenes in transgenic tomato, suggesting that it is indeed possible to introduce new branches of the flavonoid pathway, at least at its first step, by introducing foreign structural genes [34–36]. In order to further explore the possibilities of flavonoid engineering in tomato fruits, some authors [35] have targeted this pathway toward classes of flavonoids, which are normally not present in tomato. Using structural genes from several plant sources and combinations thereof, they were able to produce transgenic tomatoes accumulating stilbenes, deoxychalcones, or flavones. These fruits displayed altered antioxidant profiles and an up to threefold increase in total antioxidant activity of the fruit peel (Table 51.1).
Transgenic tomato plants (Solanum Lycopersicum, cv money maker), expressing stilbene synthase gene under constitutive 35S, and mature fruit-specific promoter (TomLoxB) were obtained by genetic transformation through Agrobacterium infection of cotyledons [34]. The phenotype of all transformed lines was similar to that of the wild-type plants, showing a regular development, flowering, and fruit maturation. However, high resveratrol-producing 35SS tomato fruits were seedless, whereas low resveratrol LoxS fruit showed a normal seed set, comparable to wild type (Fig. 51.2) [38]. Protein analyses revealed that the highest accumulation of stilbene synthase was at green stage in constitutively transformed 35SS tomato line [34] and at the red mature stage in the LoxS line [37]. Resveratrol accumulation showed some quantitative differences since the levels recorded in the LoxS were about 20 times lower than those found in 35SS fruits (Fig. 51.3a and b, respectively).
Stilbenes accumulation in 35SS and LoxS whole mature red fruits (a) and skin (b). The amount of trans-resveratrol, trans-piceid, and their correspondent cis-forms were simultaneously detected on the basis of their retention time and UV spectra of the additional peaks observed after UV irradiation at λ: 366 nm of trans-resveratrol and trans-piceid, respectively
Qualitative and quantitative comparisons between the different transgenic plants synthesizing resveratrol and related stilbenes are difficult, as different analytical methods are used to assay these compounds. These analyses have shown various stilbene levels and spatiotemporal distributions, leading to a considerable variability in terms of relative amounts of different forms.
In particular, the glycosylation of polyphenolic compounds occurs frequently in plant to protect the cell from their potential toxic effects and to prevent their oxidation and enzymatic degradation [13]. In the case of resveratrol, the free compound is first synthesized, before being glycosylated by endogenous glycosyl-transferases. Free resveratrol and its glycosylated forms have been both detected in transgenic plants [13]. Stilbenes content also depends strongly upon plant species, probably on account of different endogenous pools of enzymes or precursors, as well as differences in secondary metabolic pathways. In resveratrol synthesizing tomato lines, the free to glycosylated resveratrol ratio was related to the fruit ripening stage [34]. Furthermore, these related compounds accumulate differentially in different fruit tissues at the mature stage [36].
The comparison of different resveratrol levels in LoxS and 35SS fruits allowed the authors to study the effect of the differential depletion of substrates and, in the meantime, to clarify the effect of a novel metabolite as resveratrol on related and unrelated pathways committed to secondary metabolism.
2.1 Biochemical Analysis of Flavonoids in Fruit
The main flavonoids which had accumulated in tomato fruits were naringenin and rutin (quercetin-3-rutinoside), predominantly detected in the peel [43, 44].
Both these compounds, together with kaempferol-rutinoside (Kae rut), were quantified in the peel extracts from both the resveratrol synthesizing tomato lines (35S and LoxS lines; Fig. 51.4).
Comparison of flavonoid levels in wild type and transgenic skin of fruit at the red stage of ripening. Naringenin levels (λ:.290 nm) in the wild type and transgenic skin (of almost three) red fruits. Outline of flavonols biosynthesis and profiles of accumulation of rutin and kaempferol-rutinoside (λ:.370 nm) in skin of transgenic tomatoes compared with those of wild type
In order to clarify whether the novel biosynthetic pathway introduced in transgenic tomato caused competition for the utilization of the common substrates, levels of main flavonoids in tomato fruit were analyzed. The overall levels of these flavonoids were not impaired in either the transgenic lines. On the contrary, the 35SS line showed an increased level of Kae rut, whereas the LoxS line showed an increased naringenin content. The levels of the other flavonoids did not show any significant differences in comparison with the control tomato line. As far as the other soluble phenolics are concerned, HPLC analysis indicated that significant differences in coumaric and cinnamic acids content, which decreased in 35SS tomato fruit in all the ripening stages [34, 37].
3 Antioxidant Parameters
Fruit ripening has described as an oxidative phenomenon which requires a turnover of ROS, such as H2O2 and superoxide anion. A balance between the production of ROS and their removal by antioxidant systems has been proposed. It is likely therefore that the antioxidant system will play a crucial role in the ripening process.
3.1 Effect of Resveratrol Synthesis on the Level of ASA and GSH in Green and Red Tomato Fruits
The production of resveratrol in transgenic plants increased the levels of ASA and GSH in edible tissues thus improving the nutritional values of tomato fruit (Fig. 51.5a).
Given the importance of ROS in the ripening process, the authors have examined the changes in some oxidative parameters and in the antioxidant system during transgenic tomato fruit ripening. It is worth noting that the highest ASA and GSH contents were detected in fruits at green and red stage of ripening concomitantly to the highest level of resveratrol. In ripening tomato fruits, a transient increase in ROS production occurs during the intermediate phases of maturation. The relevance of antioxidant enzymes and metabolites during ripening has been widely investigated [45]. The acquisition, in tomato fruits, of a new biosynthetic pathway leading to the synthesis of resveratrol, which, as other phenolic compounds, has antioxidant properties, induces an increase in the ASA and GSH pools in the transformed fruits [34, 37]. In the red phase, the transformed plants (both 35S and LoxS) had ascorbate content higher than wild type. Therefore the ex novo synthesis of resveratrol affected the biosynthesis/storage of other antioxidants although these molecules do not have any biosynthetic correlation. Interestingly, only the 35SS fruits, in which stilbene synthase was constitutively expressed, showed increased ascorbate level and enzyme activities altered in the green phase (data not shown). This was in agreement with the fact that stilbene synthase was under the control of spatiotemporal-specific promoter (LoxS), which allowed gene expression preferentially in the late phase of fruit maturation [36, 37].
In transformed fruits, the increase in GSH levels was much higher than that observed in ASA levels at all the ripening stages analyzed, and the GSH redox state was at least twice that of the control [34, (Fig. 51.5b)]. The increase of glutathione biosynthesis could be a consequence of the presence, in the transformed cells, of the foreign molecules. Indeed, besides a role in ROS scavenging, GSH is also the co-substrate of glutathione transferase and is involved in phenol and xenobiotic conjugation and in their segregation into the vacuole [46, 47]. On the other hand, plant transformation with the STS gene does not seem to affect the level of membrane-associated molecules mainly present in the plastids, since their levels do not significantly differ in either transformed or non-transformed fruits, at any stage of ripening [34]. Since ASA and GSH are soluble antioxidants widely distributed in all cellular compartments, the different effect of resveratrol on these two types of metabolites could be the effect of their different cellular localizations.
3.2 Antioxidant Capability
In transgenic tomato, resveratrol synthesis was able to increase the overall antioxidant properties of the fruit, as well as the ascorbate/glutathione content with a consequent two to threefold increase in antioxidant activity of fruits and a correlation was found between resveratrol concentrations and antioxidant capacities in the ripening stages accumulating high resveratrol levels [34].
The higher antioxidant capability of resveratrol synthesizing fruits, compared to wild type, has also been confirmed by the analysis of total antioxidant activity (Fig. 51.6). Resveratrol contributes to such an increase since both hydrosoluble and liposoluble fractions have higher antioxidant activities in the transformed in comparison with wild-type fruits, whereas ASC and GSH only affect the hydrosoluble fractions. Interestingly, the increase in the antioxidant activity of the fruits affects lipid peroxidation, the value of which is significantly lower in transformed fruits in respect to that in wild types [34].
4 Anti-inflammatory Activity of Resveratrol-Producing Tomato
Several studies have reported that resveratrol has cardioprotective effects because of its ability to increase plasmatic antioxidant capacity, inhibit platelet aggregation and coagulation, reduce low-density lipoprotein oxidation, and suppress the pro-inflammatory response [48]. The anti-tumorigenic, anti-inflammatory, and cardioprotective effects of resveratrol seem to be related, at least in part, to its ability to suppress prostaglandin production through its interference with the expression and activity of COX-2, the rate-limiting enzyme in prostaglandin biosynthesis. In particular, macrophages are prominent producers of prostaglandin during inflammatory processes in response to signals that trigger macrophage activation, such as bacterial lipopolysaccharide or phorbol ester [49].
Altered COX-2 levels and consequent abnormally high prostaglandin secretion are thought to be involved in diverse pathological processes, and COX-2-specific inhibitors represent important challenges for cancer treatment, as well as chronic inflammatory diseases such as atherosclerosis [50]. The effects of resveratrol-enriched tomato extracts on COX-2 expression induced by phorbol ester in monocyte–macrophage U937 cells indicated that resveratrol reduces the level of the inducible, but not constitutive, COX isoform, thus confirming and expanding the anti-inflammatory activity of resveratrol (Fig. 51.7), as previously suggested [51–53]. Both 35SS and LoxS fruits, which contained different levels of resveratrol, showed higher antioxidant and anti-inflammatory properties than wild-type fruits [38].
Resveratrol synthesizing tomato fruits inhibited PMA-stimulated COX-2 expression in monocytoid cells. U937 cells were pre-treated with vehicle (ethanol), resveratrol synthesizing tomato fruits (35SS, LoxS) or wild-type fruit, and then stimulated with PMA. Cells extracts, at equal amount of proteins, were immunoblotted with monoclonal antibodies against COX-1 or COX-2. These results are obtained from three independent experiments with similar results. * p < 0.05 and ** p < 0.01, compared with PMA treated cells; # p < 0.05 and ## p < 0.01 compared with wild-type fruit treated cells
Remarkably, the extracts of transgenic resveratrol containing tomato fruits displayed an anti-inflammatory effect greater than that of chemically synthesized resveratrol. In conclusion, these results indicate that the presence of a new biosynthetic route responsible for resveratrol biosynthesis improves the health-giving biological activities of tomatoes.
5 Effects of Stilbene Synthase Expression on Reproductive Development of Tomato
The novel STS activity and the significant changes in the levels and/or redox state of soluble primary antioxidants do not have any apparently adverse effect on the normal vegetative growth and development of the transgenic tomato lines. As far as reproductive development is concerned, 35S transformed plants produced seedless fruits.
The development of tomato fruit in the absence of pollination and/or fertilization (parthenocarpy) was achieved using different approaches [54]. STS-induced male sterility has been demonstrated to be a successful approach to genetically engineer male sterility in a model species (tobacco). The resveratrol synthesis in transgenic tomato plants may provide a new method to obtain parthenocarpic fruits. Indeed, pollen development in 35S STS flowers was hampered, resulting in a strongly reduced seed set and all strong yielded parthenocarpic fruits (Fig. 51.2), prompting the authors to hypothesize that specific polyphenols might be efficiently down-regulated by STS gene expression in tomato flowers.
Flavonoids belonging to the class of flavonols have especially been shown to have strong stimulatory effects on pollen development, germination, pollen tube growth, and seed set [55]. When wild-type pollen was applied on transgenic 35S STS stigmas, pollen tube growth and seed set were fully rescued thus indicating that fertilization was abolished due to the lack of metabolites in the male reproductive organ [56].
Parthenocarpic tomatoes with suppressed chalcone synthase obtained by using RNA interference [55] showed abolished production of flavonoids. Several studies [55, 57] have clearly demonstrated the importance of flavonols for normal pollen tube development and male fertility. Flavonols have been detected in the pollen of all plant species tested so far and clearly play an important role in pollen tube development. In tomato plants overexpressing STS, the analysis of flavonols showed that pollen ablation was independent of the production of detectable amounts of flavonols in flowers [38].
The authors have introduced an alternative hypothesis for STS -induced male sterility, which, if correct, would significantly contribute to the conclusions drawn by other authors [58]. When expressed in tobacco anthers, STS depletes the substrate for CHS which are key molecules in several other pathways, including those linked to sporopollenin or lignin biosynthesis. Therefore, these pathways may be affected as a result of the decreased availability of precursors. Pollen grains are surrounded by a complex cell wall comprising three layers with an outer exine, a multilayered structure primarily made of sporopollenin; an inner intine, made primarily of cellulose; and a lipid- and protein-rich pollen coat. Several studies have indicated that sporopollenin is a complex polymer composed of fatty acids and phenolic compounds [59]. The analyses of coumaric and ferulic acids, precursors of sporopollenin and lignin, levels suggested impairment in their synthesis in STS sterile male tomato flower tissues (Fig. 51.8). Therefore conclude that when constitutively expressed in tomato, STS competes with CHS for common substrates, in particular in flower tissues where leaded to reduced synthesis of naringenin. Similarly, these compounds are key molecules for other important pathways such as the synthesis of exine layer constituents and lignin.
6 Concluding Remarks
Metabolic engineering is generally defined as the redirection of one or more enzymatic reactions to improve the production of existing compounds, produce new compounds, or mediate the degradation of undesirable compounds. It involves the redirection of cellular activities by modifying the enzymes, endocellular localization, and regulatory functions within cell. Even more sophisticated metabolomic tools and analysis systems will offer the possibility to study both the primary and secondary metabolic pathways in an integrated fashion. Some interesting and important developments may be expected from plant transformation with STS gene.
Therefore, further progress in better understanding of the metabolic pathways and our ability to manipulate gene expression, in genetically modified plants, can be envisaged. Success of this approach depends upon the possibility to change the host metabolism and will depend primarily on a far more sophisticated knowledge of plant metabolism, especially the nuances of interconnected cellular networks.
Identifying rate-limiting steps in the synthesis of specific metabolites could provide targets for genetically engineering biochemical pathways to produce increased amounts of compounds as well as new compounds. Together with traditional plant breeding, genetic engineering provides great opportunities to develop plants with the desired levels and/or composition of specific polyphenols. New insights into stilbene synthase expression restricted to flower tissues may provide a novel hybrid seed system for the development of a nutritionally fortified tomato. Moreover, the consequences of the unintended spread of genetic material through pollen and the risk of cross-pollination with non-transformed plants would be avoided using genetically modified sterile male plants.
Metabolite profiling is an essential tool to analyze the effects of intervention on flavonoid composition itself but also on other related or unrelated metabolic pathways. The availability of phenolic precursors toward specific pathways of the phenylpropanoid network is also controlled by biochemical, genetic, environmental, and developmental parameters [60]. The beneficial effect of trans-resveratrol on human health has directed research toward the production of this metabolite in staple plants [61]. The presence of a novel biosynthetic route responsible for resveratrol biosynthesis improved the biological activities of transformed tomatoes [62]. Comparison of the effects of diets containing tomatoes enriched in different polyphenol classes upon onset and progression of diseases in cell assays or animal disease models will allow a quantitative assessment of the efficacy of stilbenes within a common food matrix. The nutritional context could influence the effects of polyphenols, by affecting their bioavailability, or be the result of various dietary phytonutrients acting synergistically, once absorbed [63, 64].
Abbreviations
- ASA:
-
Ascorbate
- CHS:
-
Chalcone synthase
- DHA:
-
Dehydroascorbate
- GSH:
-
Glutathione
- GSSG:
-
Glutathione disulfide
- ROS:
-
Reactive oxygen species
- STS:
-
Stilbene synthase
References
Stervbo U, Vang O, Bonnesen C (2007) A review of the content of the putative chemopreventive phytoalexine resveratrol in red wine. Food Chem 101:449–457
Szajdek A, Borowska EJ (2008) Bioactive compounds and health-promoting properties of berry fruits: a review. Plant Foods Hum Nutr 63:147–156
Sparvoli F, Martin C, Scienza A, Gavazzi G, Tonelli C (1994) Cloning and molecular analysis of structural genes involved in flavonoid and stilbene biosynthesis in grape (Vitis vinifera L.). Plant Mol Biol 24:743–755
Bais AJ, Murphy PJ, Dry IB (2000) The molecular regulation of stilbene phytoalexin biosynthesis in Vitis vinifera during grape berry development. Aust J Plant Physiol 27:425–433
Donnez D, Jeandet P, Clément C, Courot E (2009) Bioproduction of resveratrol and stilbene derivatives by plant cells and microorganisms. Trends Biotechnol 27:706–713
Jandet P, Douillet-Breuil AC, Bessis R, Debord S, Sbaghi M, Adrian M (2002) Phytoalexins from the vitaceae: biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity, and metabolism. J Agric Food Chem 50:2731–2741
Soleas GJ, Diamandis EP, Goldberg DM (2001) The world of resveratrol. Adv Exp Med Biol 492:159–182
Paredes-López O, Cervantes-Ceja ML, Vigna-Pérez M, Hernández-Pérez T (2010) Berries: improving human health and healthy aging, and promoting quality life. Plant Foods Hum Nutr 65:299–308
Castrejón ADR, Eichholz I, Rohn S, Kroh LW, Huyskens-Keil S (2008) Phenolic profile and antioxidant activity of high bush blueberry (Vaccinium corymbosum L.) during fruit maturation and ripening. Food Chem 109:564–572
Conde C, Silva P, Fontes N, Dias ACP, Tavares RM, Sousa MJ, Agasse A, Delrot S, Geros H (2007) Biochemical changes throughout grape berry development and fruit and wine quality. Food 1:1–22
Jaillon O, Aury JM, Noel B, Policriti A et al (2007) The grapevine genome sequence suggests ancestral hexaploidization in major angiosperm phyla. Nature 449:463–467
Iriti M, Faoro F (2009) Bioactivity of grape chemicals for human health. Nat Prod Commun 4:611–634
Hipskind JD, Paiva NL (2000) Constitutive accumulation of a resveratrol-glucoside in transgenic alfaalfa increases resistance to Phoma medicaginis. Mol Plant Microbe Interact 13:551–562
Frei B (2004) Efficacy of dietary antioxidants to prevent oxidative damage and inhibit chronic disease. J Nutr 134:3196–3198
Kundu JK, Surh YJ (2008) Cancer chemopreventive and therapeutic potential of resveratrol: mechanistic perspectives. Cancer Lett 269:243–261
Gruber J, Tang SY, Halliwell B (2007) Evidence for a trade-off between survival and fitness caused by resveratrol treatment of Caenorhabditis elegans. Ann N Y Acad Sci 1100:530–542
Viswanathan M, Kim SK, Berdichevsky A, Guarente L (2005) A role for SIR-2.1 regulation of ER stress response genes in determining C. elegans life span. Dev Cell 9:605–615
Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D (2004) Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 431:107
Pearson KJ, Baur JA, Lewis KN et al (2008) Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab 8:157–168
Brasnyó P, Molnár GA, Mohás M, Markó L, Laczy B, Cseh J, Mikolás E, Szijártó IA, Mérei A, Halmai R et al (2011) Resveratrol improves insulin sensitivity, reduces oxidative stress and activates the Akt pathway in type 2 diabetic patients. Br J Nutr 106:383–389
Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P et al (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 127:1109–1122
Um JH, Park SJ, Kang H, Yang S, Foretz M, McBurney MW, Kim MK, Viollet B, Chung JH (2010) AMP-activated protein kinase-deficient mice are resistant to the metabolic effects of resveratrol. Diabetes 59:554–563
Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM (1998) A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92:829–839
Gerhart-Hines Z, Rodgers JT, Bare O, Lerin C, Kim SH, Mostoslavsky R, Alt FW, Wu Z, Puigserver P (2007) Metabolic control of muscle mitochondrial function and fatty acid oxidation through SIRT1/PGC-1alpha. EMBO J 26:1913–1923
Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P (2005) Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature 434:113–118
Cantó C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, Elliott PJ, Puigserver P, Auwerx J (2009) AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 458:1056–1060
Cantó C, Jiang LQ, Deshmukh AS, Mataki C, Coste A, Lagouge M, Zierath JR, Auwerx J (2010) Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab 11:213–219
Fulco M, Cen Y, Zhao P, Hoffman EP, McBurney MW, Sauve AA, Sartorelli V (2008) Glucose restriction inhibits skeletal myoblast differentiation by activating SIRT1 through AMPK-mediated regulation of Nampt. Dev Cell 14:661–673
Park SJ, Ahmad F, Philp A et al (2012) Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 14:421–433
Niggeweg R, Michael AJ, Martin C (2004) Engineering plants with increased levels of the antioxidant chlorogenic acid. Nat Biotechnol 22:746–754
Butelli E, Titta L, Giorgio M, Mock HP, Matros A, Peterek S, Schijlen EGWM, Hall RD, Bovy AG, Luo J, Martin C (2008) Enrichment of tomato fruit with health-promoting anthocyanins by expression of select transcription factors. Nat Biotechnol 26:1301–1308
Luo J, Butelli E, Hil L, Parr A, Niggeweg R, Bailey P, Weisshaar B, Martin C (2008) AtMYB12 regulates caffeoyl quinic acid and flavonol synthesis in tomato: expression in fruit results in very high levels of both types of polyphenol. Plant J 56:316–326
Giovinazzo G, Ingrosso I, Paradiso A, De Gara L, Santino A (2012) Resveratrol biosynthesis: plant metabolic engineering for nutritional improvement of food. Plant Foods Hum Nutr. doi:10.1007/s11130-012-0299-8 [Epub ahead of print]
Giovinazzo G, D’Amico L, Paradiso A, Bollini R, Sparvoli F, De Gara L (2005) Antioxidant metabolite profiles in tomato fruit constitutively expressing the grapevine stilbene synthase gene. Plant Biotechnol J 3:57–69
Nicoletti I, DeRossi A, Giovinazzo G, Corradini D (2007) Identification an quantification of stilbenes in fruits of transgenic tomato plants (Lycopersicon esculentum Mill.) by reversed phase HPLC with photodiode array and mass detection. J Agric Food Chem 55:3304–3311
Schijlen EGWM, de Vos CHR, Jonker H, van den Broeck H, Molthoff J, van Tunen AJ, Martens S, Bovy A (2006) Pathway engineering for healthy phytochemicals leading to the production of novel flavonoids in tomato fruit. Plant Biotechnol J 4:433–444
D’Introno A, Paradiso A, Scoditti E, D’Amico L, De Paolis A, Carluccio MA, Nicoletti I, De Gara L, Santino A, Giovinazzo G (2009) Antioxidant and anti-inflammatory properties of tomato fruit synthesizing different amount of stilbenes. Plant Biotechnol J 7:422–429
Ingrosso I, Bonsegna S, De domenico S, Laddomada B, Blando F, Santino A, Giovinazzo G (2011) Novel findings into parthenocarpy in tomato: a stilbene synthase approach to induce male sterility. Plant Physiol Biochem 49:1092–1099
Hüsken A, Baumert A, Milkowski C, Becker HC, Strack D, Möllers C (2005) Resveratrol glycoside (piceid) synthesis in seeds of transgenic oilseed rape (Brassica napus L.). Theor Appl Genet 111:1553–1562
Hanhineva K, Kokko H, Siljanen H, Rogachev I, Aharoni A, Kärenlampi SO (2009) Stilbene synthase gene transfer caused alterations in the phenylpropanoid metabolism of transgenic strawberry (Fragaria x ananassa). J Exp Bot 60:2093–2106
Schwekendiek A, Spring O, Heyerick A, Pickel B, Pitsch NT, Peschke F, de Keukeleire D, Weber G (2007) Constitutive expression of a grapevine stilbene synthase gene in transgenic hop (Humulus lupulus L.) yields resveratrol and its derivatives in substantial quantities. J Agric Food Chem 55:7002–7009
Lim JD, Yun SJ, Chung IM, Yu CY (2005) Resveratrol synthase transgene expression and accumulation of resveratrol glycoside in Rehmannia glutinosa. Mol Breed 16:219–233
Muir SR, Collins GJ, Robinson S, Hughes S, Bovy A, de Vos CHR, van Tunen AJ, Verhoeyen ME (2001) Over-expression of petunia chalcone isomerase in tomato results in fruit containing increased levels of flavonols. Nat Biotechnol 19:470–474
Bovy A, de Vos RCH, Kemper M et al (2002) High-flavonol tomatoes resulting from the heterologous expression of the maize transcription factor genes LC and C1. Plant Cell 14:2509–2526
Jimenez A, Creissen G, Kular B, Firmin J, Robinson S, Verhoeyen M, Mullineaux P (2002) Changes in oxidative processes and components of the antioxidant system during tomato fruit ripening. Planta 214:751–758
De Ridder BP, Dixon DP, Edwards R, Goldsbrough PB (2002) Induction of glutathione S-transferases in Arabidopsis by herbicide safeners. Plant Physiol 130:1497–1505
Edwards R, Dixon DP, Walbot V (2000) Plant glutathione S-transferases: enzymes with multiple functions in sickness and in health. Trends Plant Sci 5:193–198
Pace-Asciak CR, Hahn S, Diamandis EP, Soleas G, Goldberg DM (1995) The red wine phenolics trans-resveratrol and quercetin block human platelet aggregation and eicosanoid synthesis: implication for protection against coronary heart disease. Clin Chim Acta 235:207–219
Beloqui O, Páramo JA, Orbe J, Benito A, Colina I, Monasterio A, Díez J (2005) Monocyte cyclooxygenase-2 overactivity: a new marker of subclinical atherosclerosis in asymptomatic subjects with cardiovascular risk factors? Eur Heart J 26:153–158
Warner TD, Mitchell JA (2004) Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J 18:790–804
Subbaramaiah K, Chung WJ, Michaluart P, Telang N, Tanabe T, Inoue H, Jang M, Pezzuto JM, Dannenberg AJ (1998) Resveratrol inhibits cyclooxygenase-2 transcription and activity in phorbol ester-treated human mammary epithelial cells. J Biol Chem 273:21875–21882
Carluccio MA, Ancora MA, Massaro M, Carluccio M, Scoditti E, Distante A, Storelli C, De Caterina R (2007) Homocysteine induces VCAM-1 gene expression through NF-kappaB and NAD(P)H oxidase activation: protective role of Mediterranean diet polyphenolic antioxidants. Am J Physiol Heart Circ Physiol 293:2344–2354
Csiszar A, Smith K, Labinsky N, Orosz Z, Rivera A, Ungvari Z (2006) Resveratrol attenuates TNF-alpha-induced activation of coronary arterial endothelial cells: role of NF-kappaB inhibition. Am J Physiol Heart Circ Physiol 291:1694–1699
Mo Y, Nagel C, Taylor LP (1992) Biochemical complementation of chalcone synthase mutants defines a role for flavonols in functional pollen. Proc Natl Acad Sci USA 89:7213–7217
Fisher R, Budde I, Hain R (1997) Stilbene synthase gene expression causes changes in flower color and male sterility in tobacco. Plant J 11:489–498
Ylstra J, Busscher J, Franken PCH, Hollman JNMM, van Tunen AJ (1994) Flavonols and fertilization in petunia hybrida: localization and mode of action during pollen tube growth. Plant J 6:201–212
Schijlen EGWM, deVos RCH, Martens S, Jonker HH, Rosin FM, Molthoff JW et al (2007) RNA Interference silencing of Chalcone synthase, the first step in the flavonoid biosynthesis pathway leads to parthenocarpic tomato fruits. Plant Physiol 144:520–1530
Höfig KP, Möller R, Donaldso L, Putterill J, Walter C (2006) Towards male sterility in Pinus radiata – a stilbene synthase approach to genetically engineer nuclear male sterility. Plant Biotechnol J 4:333–343
Ylstra B, Touraev A, Benito Moreno RM, Stöger E, van Tunen AJ, Vicente O et al (1992) Flavonols simulate development, germination and tube growth of tobacco pollen. Plant Physiol 100:902–907
Ahlers F, Thom I, Lambert J, Kuckuk R, Wierman R (1999) H1NMR anlysis of sporopollenin from Typha angustifolia. Phytochemistry 5:1095–1098
Burkardt PK, Beyer P, Wunn J, Kloti A, Armstrong GA, Schledz M, von Lintig J, Potrykus I (1997) Transgenic rice (Oryza sativa) endosperm expressing daffodil (Narcissus pseudonarcissus) phythoene synthase accumulates phytoene, a key intermediate of provitamin A biosynthesis. Plant J 11:1071–1078
Frankel EN, Waterhouse AL, Kinsella JE (1993) Inhibition of human LDL oxidation by resveratrol. Lancet 24:1103–1104
Prior RL, Wu XL, Gu LW, Hager TJ, Hager A, Howard LR (2008) Whole berries versus berry anthocyanins: interactions with dietary fat levels in the C57BL/6 J mouse model of obesity. J Agric Food Chem 56:647–653
Titta L, Trinei M, Stendardo M, Berniakovich I et al (2010) Blood orange juice inhibits fat accumulation in mice. Int J Obes (Lond) 34:578–588
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this entry
Cite this entry
Giovinazzo, G., Ingrosso, I., Taurino, M., Santino, A. (2013). Metabolic Engineering for Functional Foods: Tomato Fruits and Stilbenes. In: Ramawat, K., Mérillon, JM. (eds) Natural Products. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-22144-6_193
Download citation
DOI: https://doi.org/10.1007/978-3-642-22144-6_193
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-22143-9
Online ISBN: 978-3-642-22144-6
eBook Packages: Chemistry and Materials ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics