Abstract
Total hip replacement has become one of the most successful interventions achieved in modern medicine, with excellent technical outcomes and benefits for patients suffering end-stage joint diseases. In-service degradation of prosthetic materials results in the production and accumulation of wear debris particles with devastating effects for the peri-implant tissues. Exposure to wear debris induces a localized inflammatory response and periprosthetic osteolysis, leading to aseptic loosening and failure of the implant. In order to solve this clinical problem, efforts have been focused on improving the tribological properties of biomaterials and elucidating the biological response to wear debris. Unravelling the cellular and molecular mechanisms involved in this pathology will aid to identify diagnostic biomarkers and therapeutic targets.
This chapter provides a general overview of the biological events that govern the etiology of periprosthetic osteolysis, illustrating current knowledge about particles produced by the most commonly used artificial joint articulating surfaces. A brief summary outlines the current and future state of preventive and therapeutic strategies to handle the adverse biological response to wear particles.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Aseptic loosening
- Periprosthetic osteolysis
- Wear debris
- Inflammation
- Bone-resorbing factors
- UHMWPE
- Metal
- Ceramic
- Cement
Total Hip Replacement: Clinical Need and Demand
Total hip arthroplasty (THA) represents the most successful and revolutionary intervention achieved in orthopedic surgery in the last century [1,2,3]. This surgery is performed to restore the injured or degenerated joint function when conservative treatment options have failed and pain, stiffness and other limitations drastically reduce the patient’s quality of life. The clinical settings in which hip arthroplasty is indicated involve acute and chronic underlying joint-related diseases, mostly degenerative osteoarthritis and rheumatoid arthritis but also other arthropathies including avascular necrosis, developmental and congenital disorders, neoplasias, fractures and post-traumatic degenerative arthritis [4]. In 2010, the number of individuals bearing hip implants in USA was estimated in more than 2.5 millions [5]. In Europe, countries with high incidence are Germany, Switzerland and Belgium with ratios, of 296, 287 and 240 THA procedures per 100,000 inhabitants, respectively, while in US and UK the frequencies are 184 and 194 [3].
Early hip failure (within 5 years of implantation) is mainly associated to instability, aseptic loosening (AL), infections, wear and fracture and has a decreasing trend due to improvements in surgery techniques and advances in the biomaterials field. However, an alarming prevalence has been detected in some cohorts, especially in patients with metal-on metal (MoM) bearings [6, 7]. In the long-term, the survival rate (endpoint at revision) of prosthesis after 10–15 years of implantation has been estimated about a 90–95% [8,9,10] although percentages of only 58–62% or even less have been also reported after longer service periods [10,11,12].
Demographic changes and lifestyle habits, with a younger and more active population suffering from disabling joint diseases, have led to a significant raise in the number of THA performed [4, 5, 13]. While THA was initially intended for elderly patients with limited activity, current candidates include young and middle-age patients as well as physically active elderly individuals [3, 13]. As a consequence, the incidence of total hip replacements (THR) increased by a 25% during the period 2000–2009 and is expected to increase annually at a rate of approximately a 5% per year in the coming years, to such an extent that in the period 2005–2030 THA procedures will increase a 174% [4, 14]. Approximately half of the joint replacements currently performed in USA are made in patients younger than 65 years. In Europe, 42% of men and 31% of women who underwent THA in England and Wales were in this age group [15]. By 2030, 52% of THR might be performed in these patients [16, 17]. Given the limited expected time of service of prosthesis in patients younger than 50 years, the revision rates will dramatically increase in coming years [17, 18].
Implant Failure: Aseptic Loosening
Implant failure is still a major complication of joint arthroplasty with severe consequences for the patient that impact the health-care system [8, 16, 19,20,21]. Failure, origined by mechanical and/or biological factors, can arise from multifactorial causes such as implant design, surgical technique, method of fixation or infection but the main factor limiting the longevity of THAs is AL secondary to periprosthetic osteolysis [3, 13, 15, 17]. Specifically, implant loosening due to aseptic osteolysis accounts for over 70% of total hip revisions while infections, recurrent dislocations, periprosthetic fractures and surgical errors contribute to about a 20% of the reported failures [21,22,23]. Instability and infection are complications frequently diagnosed in the early postoperative period, while osteolysis and AL usually appear in the medium to long-term [18]. Revision surgery is a procedure technically more challenging than primary arthroplasty and often associated with poorer prognosis and higher risk of failure [24, 25]. The late development and diagnosis of AL, usually asymptomatic in early stages, and its association to severe periprosthetic bone defects are factors that compromise the revision surgery procedure [18, 26]. Patients for revision surgery are typically in their 70s and 80s, and age-associated issues including morbidity and mortality risks undermine the intervention. The exponential growth in the number of primary total joint replacement procedures is associated to a concomitant increase in the number of revision operations. In this regard it has been estimated that the revision burden (ratio of revision/primary THA) will be about 17% in 2030 [27, 28]. Statistical data have shown that 10 years after of the primary hip arthroplasty, 24% of cases develop osteolysis and about a 15% of recipients require further surgical intervention due to AL [17, 29]. In young patients, under 30 years, the percentage of cases which requires revision surgery can reach rates up to 33% while in older patients range from 7% to 15% [30]. Regarding components, the acetabular cup is revised more frequently than the femoral stem [30, 31]. Currently, 40,000 hip revision procedures due to loosening are performed annually in USA and it is expected to increase about a 137% for 2030 [14, 32].
AL is characterized by areas of osteolysis at the bone-implant interface, identified radiographically as radiolucent zones, which can result in displacement of the prosthetic component [3, 33]. Progression of peri-prosthetic bone loss as assessed by radiology is very slow. Osteolytic lesions often appear many years after the primary surgery and can be associated to mechanically stable implants [34]. Clinical symptoms associated to bone tissue destruction may not be clearly apparent or even remain silenced.
Several factors determine the longevity of hip implants. Apart from the importance of a correct surgical technique (orientation and alignment of prosthetic components, prosthesis stability, anchorage and osteointegration of the implant …) there are significant differences regarding prosthesis- (e.g. type of bearing material, prosthesis design, shape of prosthesis, surface technology, type of fixation …) and patient- related factors (e.g. age, co-morbidities, level of activity and differences in mechanical loading,…) wich influence the host response to the implant and therefore its success or failure [17, 35]. In this regard, Engh et al. found that implant wear and patient-specific propensity equally contribute to the degree of osteolysis and might account for the extent of the area affected by periprosthetic bone loss [36]. Research efforts have attempted to identify clinical risk factors and individual susceptibility to periprosthetic osteolysis in order to predict the outcome and prevent, or at least attenuate, the ensuing complications of THA [37]. Over the last years, the influence of parameters such as gender, body mass index, and age [21, 37, 38], as well as the contribution of genetic factors to the development and progression of AL [15, 37, 38] have been considered. Regarding age and gender, young male patients show high risk of developing osteolysis [38]. Considering other factors, genetic variations affecting molecules involved in inflammation and bone turnover may play a role in the predisposition to AL of patients with THA. Particularly, polimorphisms in genes encoding proteins such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), transforming growth factor-β (TGF-β), interleukin-1 receptor antagonist (IL-1 Ra), matrix metalloproteinase-1 (MMP-1), osteoprotegerin (OPG) or receptor activator for nuclear factor kappa-B ligand (RANKL), have been associated with susceptibility to osteolysis and/or short prosthesis longevity [15, 20, 39,40,41]. These proteins are powerful mediators in the biological response to particulate wear debris and the periprosthetic bone loss around THR.
Other risk factor associated to predisposition to osteolysis is the preoperative diagnosis, which might influence the host response to the biomaterial. While some authors have observed higher rates of loosening in patients who underwent THA due to inflammatory arthritis, post-traumatic arthritis, developmental dysplasia or osteonecrosis as compared to primary osteoarthritis, others have not found significant differences between these groups [15, 42].
The Enemy: Wear Debris Particles
Osteolysis is the long-term consequence of the biological response to wear debris and products derived from corrosion of implants [3, 33, 43]. Particulate wear debris, mainly generated by articulating motion at the bearing surfaces but also by non-articular surfaces, are the primary causes of periprosthetic bone loss and implant loosening [44, 45]. Locally, continous exposure to prosthetic debris combined to repetitive mechanical stresses triggers an inflammatory chronic response which is highly influenced by the intrinsic properties of wear debris particles. The type of prosthesis determines the characteristics of the resultant wear debris particles and therefore the magnitude of the host response [33]. Articulating bearings in THA are hard-on-hard material such as MoM or ceramic-on-ceramic (CoC) and hard-on-soft material (metal-on-polymer) couples, being the most satisfactory the combination of a cobalt chrome femoral head articulating on an ultra high molecular weight polyethylene (UHMWPE) acetabular component [33, 46]. Wear occurs through five major mechanisms: adhesion, abrasion, third body, fatigue, and corrosion. Third body wear damage is considered a relevant source of particles in which metal, polymer, cement or even cortical bone debris are entrapped between the UHMWPE acetabular component and the hard bearing surface contributing to accelerated deterioration of the implant [3, 47]. Research in materials manufacturing and tribology has focused on identifying alternative bearing surfaces that reduce the production of wear particles debris [3, 46]. These new materials might give rise to wear particles with unknown characteristics.
In general, the standing paradigm of AL states that implant-derived-particles stimulate periprosthetic cells to release factors which affect cell functions through autocrine and/or paracrine mechanisms [13, 18, 33, 48]. Potentially, all cell types at the periprosthetic tissue are targets of wear debris but macrophages are critically involved [17, 29, 33, 45]. Macrophages activated by particles produce an array of inflammatory, chemotactic and bone-resorbing factors such as chemokines, growth factors, cytokines, degradative enzymes and reactive oxygen radicals, among others, triggering and perpetuating the periprosthetic osteolytic cascade [3, 24, 43, 48]. As result of the inflammatory response, a granulomatous pseudomembrane generates at the bone-implant interface, which further compromises the interface stability and osseointegration of the device. Altered-load bearing effects contribute to abnormal wear processes and production of particles in the implant bed, leading in advanced stages of periprosthetic osteolysis to further anomalous mechanical loading [45]. Although osteolysis due to wear debris initiates as a localized phenomenon, joint fluid can transport wear debris, cells and molecules to adjacent bone sites extending the affected bone-implant interface. Morever, high fluid production and altered loading increase periprosthetic pressure in the peri-implant region [34, 49]. Moreover, the extent of the inflammatory reaction to wear particles is not confined to joint and adjacent tissues since wear debris, mainly metal particles, have been detected in remote organs including the spleen, liver, kidney and lung [3, 45]. Whether systemic dissemination of wear particles may cause side effects, e.g. toxicity and carcinogenicity, is a matter of debate.
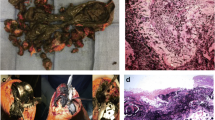
Histological examination of periprosthetic tissues retrieved during revision surgery has revealed significant amounts of prosthesis-derived particles (Fig. 1.1) and a multi-cellular composition characterised by the presence, among other cells, of macrophages, multinucleated foreign body giant cells containing engulfed particles, lymphocytes, fibroblasts and osteoclasts in association to elevated levels of pro-inflammatory cytokines, such as TNF-α, interleukin-1β (IL-1β), prostaglandin-E2 (PGE2) and RANKL [33, 50].
Periprosthetic membrane retrieved from a patient undergoing revision surgery due to hip aseptic loosening. (a) Optical microscopy imaging. Hematoxylin-eosin staining showing characteristic features of the foreign body reaction induced by wear debris particles, with infiltration of inflammatory cells. High amounts of metal particles are found, indicated by white arrows. (b) Transmitted polarized light and confocal microscopy imaging. Nuclei of cells in the tissue were counterstained with DAPI (blue). UHMWPE particles were detected by polarized light as bright areas (indicated by a red arrow) and metal particles were detected by transmitted light (black, white arrows) and by reflection (green false colour labelled). Scale bar: 100 μm
The phagocytosable range size of particles able to induce an in vitro inflammatory response has been established in less than 10 μm [33]. In general, nanometer-sized particles with diameters lower than 150 nm can be internalized through endocytosis or pinocytosis, while particles sized between 150 nm and 10 μm utilize a phagocytosis-mediated process and particles higher than 20 μm induce multinucleated giant cell formation [33, 51]. The greater the particle load of phagocytosable particles, the higher is the local inflammatory response. Regarding shape, elongated particles are more reactive than round particles [23].
Originally coined as “cement disease”, as osteolysis due to AL was thought to be a response to particles of polymethylmethacrylate (PMMA) , later it was observed that osteolysis also occurs with cementless implants and potentially all kind of particles (metals, PE, ceramics, and bone cement) can elicit a biological response in joint tissues [3, 33, 43]. Particle chemistry plays an important role [3, 13, 33]. In general, metal particles are more pro-inflammatory and/or toxic than polymers or ceramics [3, 52,53,54]. An overview of the most relevant and recent findings about the cellular events and molecular pathways modulated by the different types of wear debris is presented in the following sections.
Polyethylene Particles
Analyses of periprosthetic tissues retrieved during revision surgery have shown that UHMWPE wear debris originated from the acetabular liner are the most frequent types of debris, standing for 70–90% of the debris load independently if implants are cemented or not [3, 17]. Probably due to their abundance, UHMWPE particles are considered as key players in the stimulation of periprosthetic bone loss and implant loosening. Traditionally, the most used bearing couple was UHMWPE articulating against a metallic ball [3]. With hundreds of thousands of UHMWPE particles generated in a single step in the periprosthetic space, about 500 billion of particles produced per year, and a total amount of trillions of particles during the lifetime of a prosthesis, osteolysis was associated to a threshold of 2.16 × 109 and polyethylene (PE) particles per gram of interfacial tissue and a wear rate greater than 0.1 mm/year of the UHMWPE acetabular liner [3, 17, 23, 55].
Histological examination of periprosthetic tissues have revealed that UHMWPE debris appears as small particles or large shards, and exhibit birefringence under polarized light [45]. Once isolated from membranes, UHMWPE particles display an irregular surface with a predominant globular shape, although other irregular shapes are also observed especially in those of larger size, being 90% smaller than 1 μm [3, 17]. The different sizes and shapes observed have been related to the specific wear mode as well as to the implantation period [3, 45]. Small particles can be detected within macrophages while large particles are usually included within foreign body multinucleated giant cells [45].
Bacterial lipopolysaccharide (LPS) , possibly derived from subclinical infections or systemic sources, may bind to particles and contribute further to the inflammatory reaction [56, 57]. Some evidence suggests that macrophages might not be able to induce an inflammatory response if adsorbed endotoxins are not present on UHMWPE particles [58]. Proteins present in the physiological fluids, including type I collagen, aggrecan proteoglycans, immunoglobulin, fibronectin and albumin, can also be adsorbed on UHMWPE surfaces [59]. The interaction of adsorbed proteins and cell surface receptors, such as integrins, has a remarkable role in the macrophage interactions with biomaterials. Specifically, integrins participate in receptor-mediated phagocytosis of wear particles of different composition, including UHMWPE. The downstream effects of integrin-mediated interactions result in the production of pro-inflammatory cytokines and osteoclast activation, as shown by Zaveri et al. who have recently reported that Mac-1 integrin and RGD-binding integrins are involved in osteolysis induced by UHMWPE particles [59]. Macrophage activation can occur not only by phagocytosis of UHMWPE particles but also by cell contact through receptor-mediated mechanisms, including Toll-like receptors (TLRs), and the cluster of differentiation molecules CD11b and CD14 [60]. Thus, Maitra et al. reported that UHMWPE particles stimulate in vitro TLR1/2 and activate phagocytosis [61]. In a PE particle-induced osteolysis murine calvarial model, TLR2 and TLR4 were found highly expressed in macrophages [60]. TLRs act primarily through the adapter protein myeloid differentiation primary response protein (MyD88) and induce activation of nuclear factor kappa-B (NF-кB), mitogen-activated protein kinases (MAPK) and interferon-regulatory factors (IRFs), leading to the release of pro-inflammatory cytokines, growth factors and chemokines [22, 24]. TLRs are activated by pathogen- or damage-associated molecular patterns in response to infection or tissue damage. Oxidative stress, e.g. oxygen intermediates and free radicals, may provoke degradation of UHMWPE resulting in products as alkane polymers. These degradation products could influence the “original” inmunogenecity of particles, altering their binding affinity to TLRs or other molecules in the cells surfaces and thereby activating endogenous signaling systems. Moreover, particulate debris affects the host tissue integrity by increasing cell death, which generates danger signals such as heat shock proteins that further increase TLR activation [51].
The inflammasome has been recently involved in the cellular activation by wear particles [17, 61]. Inflammasome activation depends on reactive oxygen species (ROS), enzymes release and other danger signals. Upon phagocytosis of UHMWPE particles, endosomal and lysosomal damage results in release of cathepsins which trigger the inflammasome activation. If particles are larger that 20 μm, multinucleated giant cells may activate also NADPH oxidases and generate ROS, also contributing to its activation [51].
After the initial interaction with macrophages, particles induce an active infiltration of inflammatory cells into the periprosthetic area. Migration is mediated through chemotactic factors such as interleukin-8 (IL-8), macrophage chemotactic protein-1 (MCP-1), and macrophage inflammatory protein-1 (MIP-1) released by inflammatory and mesenchymal cells, among others, upon challenging with particles [62]. Thus, the pro-resorptive environment is created not only by locally activated cells but also by migration of macrophages and osteoclast precursors, i.e. monocytes. In this regard, in vivo studies employing UHMWPE particles have shown the contribution of chemotactic factors released by macrophages and mesenchymal stem cells (MSCs) to cell recruitment [60]. Specifically, the CCR2/MCP-1 and the CCR1/MIP-1α ligand/receptor axes are involved in the systemic recruitment of macrophages and MSCs, respectively, in the presence of UHMWPE particles [60, 63].
The most active field of research during the last decade has been focused in the identification of factors associated to the destruction of the bone tissue when UHMWPE particles are present. For instance, in vitro stimulation of macrophages with these particles increases the expression of genes involved in inflammation and osteoclastogenesis such as MMP9 (coding for matrix metalloproteinase-9), CTSK (coding for cathepsin K protein), CALCR (coding for calcitonin receptor) and TNFRSF11A (coding for receptor activator of nuclear factor kappa-B protein (RANK)). In vivo, calvarial models of PE particle-induced osteolysis have corroborated these findings [16].
Production of factors involved in bone destruction depends on the size and concentration of wear particles. Green et al. showed that, for a given tested concentration, PE particles of 0.24 μm in length induce higher production of TNF-α, IL-1β, IL-6, and PGE2 in macrophages than larger particles. Dose- and size-dependent effects were also reported, as sizes range between 0.45 and 1.71 μm were the most reactive when higher concentrations were tested [51]. A recent study has shown that treatment of peripheral blood mononuclear cells with UHMWPE particles below 50 nm does not induce the release of TNF-α, IL-1β, IL-6 and IL-8 [64]. Regarding concentration, UHMWPE particles showed a dose-dependent induction in the production of TNF-α, IL-1β and IL-6 in macrophages, and in the production of RANKL in osteoblasts [16, 51, 65]. PE particles are able to regulate proliferation and function of osteoblastic or bone-forming cells, through induced expression of pro-osteoclastogenic factors RANKL, IL-6, IL-8, PGE2 and macrophage colony-stimulating factor (M-CSF) and also through the interference in the regulation of matrix synthesis proteins such as collagen or alkaline phosphatase (ALP) [16, 66, 67]. Differentiation of osteoblastic cells into a mature, osteocyte-like phenotype is tighly regulated by several genes including runt-related ranscription factor-2 (Runx2), sclerostin (SOST) and osterix (Osx) whose expression is also altered in the presence of PE particles. In osteoprogenitor cells, UHMWPE particles inhibit in a dose-dependent manner proliferation and osteogenic differentiation [67]. Effects of PE particles on osteoblasts are further influenced by their maturation state [66]. A recent study has detected increased expression of catabolic markers incuding matrix metalloproteinase-13 (MMP-13), carbonic anhydrase 2 (CA2), cathepsin K, and tartrate resistant acid phosphatase (TRAP) in human primary osteocyte-like cultures exposed to PE particles [68]. Moreover, in vitro exposure of macrophages to these particles induces spontaneous differentiation into mature and active osteoclasts [69]. In other coexisting cells in the periprosthetic bone bed, studies are scarce. For instance, in fibroblasts, dominant cell type in the interfacial membrane, PE particles induce the expression of the pro-inflammatory cytokine IL-6 [16]. The role of these cells in wear-induced osteolysis has been mainly attributed to their interaction and cooperation with macrophages to amplify inflammation, fibrosis and osteoclast activation [51]. Macrophages and fibroblasts in the interfacial tissues overexpress macrophage migration inhibitory factor (MIF), which can up-regulate the expression of pro-inflamatory factors and matrix metalloproteinases (MMPs) involved in the periprosthetic bone tissue destruction [70]. New lines of evidence have addressed the role of dendritic cells (DCs) in UHMWPE particles-induced osteolysis, with a similar role than macrophages [51].
Research efforts have recently focused in how to fit the paradigm of polarization of macrophages when exposed to wear particles. Wear particles contribute to the creation of a periprosthetic environment in which macrophages can be polarized to M1 (pro-inflammatory) and M2 (anti-inflammatory) phenotypes. Both phenotypes have been observed and extensively characterized [17, 60]. Polarization of uncommitted M0 or M1 macrophages toward the M2 phenotype, that promotes bone healing, has been proposed as therapeutic strategy to decrease the local inflammation. In a calvaria model employing UHMWPE particles, bone resorption was reduced after administration of the M2 phenotype inducer interleukin-4 (IL-4) [71].
Broad-scale expression profiling of human macrophages challenged to UHMWPE particles revealed changes in the expression of genes related to inflammatory response, cell proliferation, cytokine-mediated signaling, response to stress, cell migration and death [24]. Among others, particles modulated the transcript levels of genes encoding factors with remarkable roles in osteoclastogenesis and bone resorption such as IL-8, MIP-1α, MIP-1β, macrophage inflammatory protein-3 alpha (MIP-3α), interleukin-23 (IL-23), M-CSF, IL-1α and vascular endothelial growth factor (VEGF); and cell surface receptors involved in the recognition of particles such as β-integrins, protein-coupled receptors, intercellular adhesion molecules, TNF receptor superfamily members and TLRs-signaling. Moreover significant up-regulation of the expression of genes encoding MMPs, including MMP-1 and MMP-19, was also found.
Overall, these findings show that inflammation and osteoclastogenesis-related mechanisms activated by UHMWPE particles are main processes involved in the pathogenesis of wear-induced osteolysis. The introduction of highly crosslinked polyethylene (HXLPE) to reduce wear and osteolysis in total joint arthroplasty has attracted great interest. Moreover, as compared to polymeric and metallic particles, HXLPE particles induce a moderate degree of peri-implant osteolysis [72].
Metallic Particles
Metal particles are massively generated from the bearing surfaces of MoM hip replacements, which were conceived as alternative to metal-on-polyethylene (MoP). However, adverse long-term effects are associated to MoM, including a higher rate of failure as compared to other bearing surfaces [46]. Metals are susceptible to degradation upon exposure to extracellular tissue fluids, and therefore the clinical outcome and durability of implants are affiliated to particulate corrosion and wear products. Metal wear particles arise mainly from the bearing surfaces but also from the metal back and fixation screws of the acetabular cup [45, 73]. Under the optical microscope, they appear as black to brownish colored entities, with amorphous or irregular shapes (flakes or needles) and sharp edges, and sizes ranging from 0.1 to 5 μm [3]. Ultrastructural methods showed that the majority of metal particles retrieved from periprosthetic tissues and joint simulators are in the nanometric range [3, 74]. These nanoparticles have a greater relative surface area than microparticles and are potentially more chemically and biologically reactive [45]. About 6–250 × 1012 metal particles are released per year from a MoM articulating surface [3]. As particles with other compositions, metallic particles stimulate inflammation and bone resorption in detriment of bone formation [75]. Resorption areas associate to metal particles and periprosthetic tissues are characterized by express inflammatory mediators including IL-8, IL-1β, macrophage inflammatory protein-2 alpha (MIP-2α), stromal cell-derived factor 1 (SDF-1) and its receptor CXCR4 [9, 76, 77].
In vitro, exposure to Ti particles induces macrophages to release TNF-α, IL-6, IL-1β and PGE2 in a process mediated by tyrosine phosphorylation and MAPK pathway [9]. A recent factor associated to induced inflammation by Ti particles is the cannabinoid receptor type 2 (CB2), inductor of osteoclastogenesis [78]. Similar to described in PE particles, dose- and size-dependent responses have been observed and the release of pro-inflammatory factors can occur independently of phagocytosis-dependent mechanisms. Concentration-dependent effects have been detected for TNF-α, IL-1β and IL-6 [54]. Apart from inflammatory factors, the expression of genes encoding osteoclastogenic markers can be induced by Ti particles, including TRAP, nuclear factor of activated T-cells 1 (NFATC1, also termed NFAT2), cathepsin K and RANK, as well as nitric oxide synthase 2 (NOS2), NF-кB and MMP-9. By regulating the expression of genes involved in the superoxide dismutase pathways, Ti particles also modulate oxidative stress [16]. Regarding chemotactic factors, in vitro exposure of primary human monocytes/macrophages to Ti particles increased the production of MIP-1α, resulting in increased monocyte migration. Other chemokines induced upon exposure to Ti particles in osteoblasts and osteoclasts are CCL17 and CCL22. Interestingly, the expression of the gene encoding CCR4, receptor of both chemokines, is up-regulated in osteoclast precursors exposed to Ti particles which accounts for macrophage recruitment and bone loss [79].
In osteoblastic cells, metal particles modulate the expression of genes encoding pro-inflammatory and bone-resorbing factors. In vitro studies have shown that Ti particles decrease OPG and induce IL-6 production, effects associated to increased nuclear factor IL-6 (NF-IL-6) and NF-кB activation while production of MMP-2 increases through p38 signaling [9, 16, 80]. Ti particles severely impact on the viability and osteogenic potential of osteoblast precursors [16, 81]. Moreover, Ti particle s produce adverse effects in MSCs that include toxicity and increased IL-8 production. Induction of genes encoding pro-apoptotic proteins and down-regulation of those encoding anti-apoptotic and osteogenic factors has been detected in MSCs challenged with Ti particles [16]. Altogether, these findings indicate that Ti particles impair the viability, proliferation and differentiation ability of MSCs. Exposure of fibroblasts to Ti-based particles induce the expression and/or production of RANKL, IL-6 and MCP-1 in a dose-dependent manner and stimulates the release of osteolytic enzymes as stromelysin and collagenase [3, 9, 16]. In human synovial cells, Ti particles increase MMP-2 activity [82].
Bacterial endotoxins are detected frequently and in a significant amount adsorbed to metallic wear particles [57, 75]. In macrophages, exposure to Ti particles adsorbed to LPS results in the regulation of TLRs-mediated responses and the stimulation in the production of the proinflammatory cytokines TNF-α, IL-1β and IL-6 [75]. Nonetheless, endotoxin-free Ti particles activate TLRs-mediated pathways and induce the expression of same genes [83]. However, comparative studies have shown that endotoxin-free Ti particles induce inflammation and osteolysis to a lower extent than those with adsorbed bacterial debris [84]. As observed with UHMWPE particles, NALP3 inflammasome can be activated upon Ti particles internalization and subsequent cathepsin release [17].
In response to implant metal debris challenge, macrophages adopt a pro-inflammatory M1 phenotype [16, 79]. In a study to clarify whether macrophage phenotypes are equally sensitive to Ti particles, these cells were polarized towards phenotype M0, M1 and M2 and then incubated with Ti particles. While no significant effects were observed in M2-macrophages after Ti particle challenge, M1-macrophages experienced drastic changes both at the transcriptome and the proteome levels, with a notable increase in the production of inflammatory chemokines (e.g. MCP-1, MIP-1α, IL-8), cytokines (TNF-α, IL-1β) and growth factors (e.g. granulocyte-macrophage colony-stimulating factor (GM-CSF), granulocyte-colony stimulating factor (G-CSF) and epidermal growth factor (EGF)) [85]. This study supports again the notion that the local microenvironment, which determines the macrophage phenotype, notably influences the response to particles.
Changes in mechanotransduction and adhesive properties of cells treated with metal particles have also been addressed in in vitro studies. For example, Preedy et al. observed that Ti particles increase osteoblast elasticity [86].
In vivo studies employing calvaria or air pouch models have corroborated the in vitro findings regarding Ti particles induction of TNF-α, cyclooxygenase-2 (COX-2), IL-1β, MMP-9, MCP-1, RANK, RANKL, NFATC1, VEGF and CB2 expression and/or production [9, 16]. Similar results were observed when Ti-alloy particles were investigated [16].
Concerns about wear debris derived from CoCr are mainly based on ions release. Co and Cr ions can travel through lymh and blood with harmful consequences for heart, brain and thyroid, spleen or liver [9]. CoCr alloy particles have been histologically identified in necrotic areas with infiltrating macrophages and lymphocytes [87]. In the local tissues, Co-Cr alloy particles and ions impair the expression of proteins related with osteoblastic differentiation (OPG, Osx and osteocalcin (OCN)) and increase those related with inflammation and osteoclastogenesis (IL-6, RANKL, MCP-1 and NFATC1) [16]. Co ions affect osteoblasts and neutrophils functions and stimulate chemokine secretion in both cell types [88]. Ions released from a CoCr alloy (CoCr29Mo6) induced necrosis in osteoblasts and peripheral blood mononuclear cells (PBMCs) and also stimulated IL-6, IL-8, and MMP-1 expression in these cell types [89]. Co and Cr ions are highly cytotoxic for macrophages and lymphocytes, inducing apoptosis [9]. Exposure to CoCr particles, increase expression and/or production of pro-inflammatory factors (IL-1α, IL-6, interleukin-10 (IL-10), IL-8, GM-CSF and PGE2) in monocyte-macrophage lineage cells, reduce viability in histiocytes and fibroblasts and affect osteogenic differentiation in MSCs [9, 16, 90]. Treatment of THP-1 cells with a CoCr alloy (ASTM F75) particles increase the production of TNF-α, IL-1β and IL-8 through TLR4 signaling pathway [91]. In fibroblasts, CoCr nanoparticles increase the production of ROS and induce apoptosis [9, 92]. Regarding size, CoCr nanoparticles release more ions and are more cytotoxic than CoCr microparticles [93].
After being phagocytosed by macrophages metal nanoparticles trigger endoplasmic reticule (ER) stress. Periprosthetic tissues express large amounts of ER stress-associated molecules (Ca2+, IRE1-α, GRP78/Bip, CHOP, cleaved Caspase-4, and JNK). It has been speculated that apoptosis in macrophages challenged to metal particles might be mediated by ER stress pathways, also linked to inflammation and osteoclastogenesis [94]. Similar to UHMWPE particles, chemical changes in metal particles surface altere the recognition of particles by cells and their subsequent effects [9, 95]. Also, release of endogenous alarmins has been proposed to contribute to positive feedback mechanisms in the periprosthetic tissue challenged with metallic debris [84, 18].
Several studies report a scarce number of T-lymphocytes in periprosthetic tissues, concluding that osteolysis occurs independently of these cells [51]. However, lymphocytes play a key role in hypersensitivity reactions leading to early osteolysis [45, 51]. Hypersensitivity or allergy to metallic components is a matter of concern to orthopaedic surgeons. This reaction is usually a cell-mediated (type-IV delayed hypersensitivity) response, characterized by activation of delayed-type hypersensitivity T lymphocytes by haptenic antigen and cytokine release which leads to recruitment of cytotoxic T-cells and macrophages activation. Activated macrophages mediate delayed-type hypersensitivity T lymphocytes activation, self-perpetuating the inflammatory response [9, 79]. Another adverse local tissue reaction is the aseptic lymphocyte-dominated vasculitis-associated lesions (ALVaL). This response is similar to the type IV hypersensitivity response, characterized by inflammation accompanied by lymphocytic infiltration, accumulation of plasma cells and macrophages and soft tissue necrosis [3, 9, 96]. Pseudotumors, mainly affecting soft tissues, are another complication of metal THA. Pseudotumors are identified as a solid or cystic mass-forming tissue characterized by necrosis areas, mononuclear cell infiltration and giant multinucleated cells, with high degree of perivascular lymphocytic aggregation. The prevalence of pseudotumors in hip implanted patients is a controversial issue. While some authors estimated a low frequency, just about a 1% within 5 years of MoM implantation, others have found a much higher incidence, up to 60% [3, 9, 97]. This absence of consensum may be explained due to the asymptomatic nature in some cases and the similarity with other adverse reactions. In this regard, Catelas et al. have stated relevant differences in the proteome of pseudotumors and osteolytic tissues which correlate with predominant adaptive immune responses in patients with pseudotumors and innate immune responses in patients with periprosthetic osteolysis [98]. One molecular mechanism which might explain the local soft tissue growing around the implant (fibro-pseudotumors) is the induction of hypoxia and angiogenesis by metal debris, which increases the levels of transcription factor hypoxia inducible factor 1 alpha (HIF-1α) and VEGF [79]. Finally, some studies suggested an association between MoM implants and risk for developing cancer but epidemiological studies have not been able to prove increased incidence of cancer in patients with these implants [9, 99].
In summary, existing data employing metal and metal-alloys wear debris and their byproducts indicate their involvement in the induction of adverse local tissue reactions, based not only in the activation of inflammation and bone resorption-related mechanisms but also in the induction of the oxidative stress and cytotoxicity. These effects are mainly dose- and concentration-dependents which, in the context of metal particles exhibiting submicrometric and nanometric sizes and being released in high amounts to the periprosthetic space, represent a clear threat to tissue homeostasis.
Ceramic Particles
Ceramics present chemical and mechanical advantageous properties for the manufacturing of orthopaedic devices, especially for young patients, including biochemical inertness, hardness, high-strength and corrosion and wear resistance [3, 100, 101]. Currently, the most used ceramic materials in clinical practice are alumina (AL2O3) and zirconia (ZrO2) [3]. Hip implant revisions of Al2O3-based components have been associated to their brittleness and the subsequent risk of catastrophic failures due to deficiencies in the manufacture [3, 102]. A great variability in the frequency of ceramics fracture rate has been reported but in general, incidence is small [3, 103]. Wear volume of ceramic bearing couples is lower than that of metallic and therefore a lower risk of revision is expected [3, 104]. Moreover, PE component exhibits a lower linear wear in CoP (0.034 mm/year) than in MoP (0.1 mm/year) bearings. On crosslinked PE, ceramics display a wear rate of 0.019 mm/year compared to 0.03 for metals [3]. Relative inertness and low abundance of ceramic wear debris in the periprosthetic space imply limited adverse biologic reactions and risk of osteolysis associated to this type of material.
Ceramic debris appears as fine greyish-brown particles (ZrO2) and brown, brownish-green or black granules (Al2O3) [45, 105, 106]. Regarding size, ceramic particles are similar to metal particles, and ten times smaller than polymeric particles. More in detail, some studies have shown a range from 0.13 to 7.2 μm in tissues around loosened CoC hip implants and others have reported a bimodal size distribution, with most of them in the nanometric-order (5–90 nm) and submicron- to micron-scale for the rest (up to 3.2 μm) [3, 106, 107].
Manufacturing defects, instability or mal position of hip prosthesis components are main factors involved in the generation of ceramic particles, which once produced are “potential” inducers of periprosthetic osteolysis [3, 60, 108]. In fact, production of ceramic debris associated to occasional osteolytic areas has been considered as the consequence of mechanical instability rather than the origin of failure [108]. Typical foreign body reaction was only observed in association to large amounts of wear particles [109]. The cellular mechanisms involved in the biological response to ceramic particles are a controversial issue, not fully elucidated. While some authors state that ceramics are able to activate cells in a similar manner to metallic debris due to their comparable size, others express doubts about the involvement of the same mechanisms. In fact, studies in retrieved tissues have shown a differential cellular response to accumulated ceramic wear debris [110]. Biocompatibility of ZrO2 particles seems to be greater than that of Ti particles since induction of pro-inflammatory gene expression was significantly lower in macrophages challenged with ZrO2. In vivo experiments support these findings, with higher extent of inflammation and bone resorption induced by Ti or PE particles than ceramics [83, 111]. Bylski et al. exposed THP-1 macrophage-like cells to different concentrations of Al2O3 and Ti particles and found that, regardless of particle size and time exposure, ceramic caused only minor up-regulations of RANK, TNF-α and OPG mRNA levels while Ti highly stimulated the expression these genes and led to cytotoxic effects in a dose- and time-dependent manner [107]. Also, minor induction in the production of pro-inflammatory proteins such as IL-1β and MCP-1 has been reported after exposure of primary human macrophages to Al2O3 particles [112]. In a comparative study using murine macrophage cells treated with PE or ceramic particles, the latter led to lower release of TNF-α [113].
The negligible in vitro effects of Al2O3 particles on cell viability and cytokines production may account for the low incidence of osteolysis in patients with CoC prosthesis [60]. Once again, particle size, composition and concentration are relevant factors to consider [114, 115, 116]. Regarding induction of pro-inflammatory cytokines, neither Al2O3 or ZrO2 particles induced IL-1 or IL-6 production in human fibroblast-like synoviocytes isolated from patients with osteoarthritis or rheumatoid arthritis [117]. Al2O3 and ZrO2 are susceptible of internalization in J774 cells, inducing both type of particles the release of TNF-α at same extent [118].
Exposure of human macrophages to ZrO2 or TiO2 nanoparticles upregulated the expression of TLR7 and TLR10 [119]. Moreover, ZrO2 nanoparticles induced IL-1β production and IL-1Ra synthesis while in LPS-treated macrophages IL-1Ra release is reduced, therefore promoting the creation of a pro-inflammatory environment, amplifying the M1 macrophage-effector functions.
A recent study has evaluated the ZrO2 effects on osteoclasts, whose recruitment and activation are directly involved in the periarticular osteolysis [120]. Exposure of osteoclasts to ZrO2 increased cell fusion and expression of osteoclast function- and bone matrix-related proteins including vitronectin receptor (VNR), TRAP, RANK and cathepsin K without stimulating osteoclast bone resorptive capacity. Moreover, higher expression of MMP-1 and an impaired production of the tissue inhibitor of this metalloproteinase were observed.
AL2O3 particles can be internalized by osteoblastic MG-63 cells which resulted in decreased proliferation, ALP activity and TGF-β1 secretion [121]. In contrast, exposure of these cells to ZrO2 stimulated proliferation and ALP activity. Interestingly, both types of particles induced the production of PGE2 in a dose-dependent manner. Treatment of primary human osteoblasts with Al2O3 induced the expression and secretion of IL-6 [122] and affected the osteoblastic function by decreasing the C-terminal type I procollagen (PICP) secretion and ALP activity [123]. The paracrine interactions between macrophages and osteoblasts were also affected by treatment of these cell types with Al2O3 particles which resulted in increased production of IL-6 and GM-CSF as assessed in a a co-culture in vitro model [124]. Another study examined the cross talk between macrophages and osteoblasts in the presence of Al2O3 particles employing media conditioned by osteoblasts exposed to particles in which a slight decrease in OPG-to-RANKL ratio was detected [125]. In this study, secretion of TNF-α, IL-6 and GM-CSF by PBMCs was not induced by culturing these cells with media conditioned by osteoblast exposed to Al2O3. Osteoclasts formation assays showed an increase in TRAP-positive aggregates which suggested the activation of osteoclastogenesis although osteoclast generation was not enhanced in PBMC cultures exposed to conditioned medium of osteoblasts challenged with Al2O3.
In summary, although ceramics present advantages in comparison to other materials, they still seem able to elicit an adverse local reaction. However, the moderate wear rate of ceramic components together with the limited effects of ceramic particles-induced biological responses may significantly account for the low incidence of osteolysis in patients bearing this type of prosthesis.
Cement Particles
PMMA debris arise from the fragmentation of the cement used to fix components of THA [3, 126]. Bone cement is frequently applied to the femoral stem but also the acetabular shell may be fixed to the adjacent bone. PMMA-based bone cement was introduced by Charnley as an effective mean to achieve stable fixation between the bone bed and the implant, and later as matrix for local delivery of antibiotics [126]. Apart from favouring formation of a fibrous interfacial tissue, PMMA experiences brittleness and shrinkage, lack of adherence to the bone and during its application, the in situ exothermic polymerization reaction can damage the adjacent bone tissue. In general, cemented prostheses are not options for young and active patients, with good bone stock quality, as PMMA can cause third body wear and secondary loosening of the hip components [127]. PMMA third-body abrasive particles originated from the failure of the cement mantle can induce surface damage, especially relevant in PE and Ti-based components [3, 128]. ZrO2 and BaSO4 are used as radio-opaque contrast media in bone cement, and therefore disintegration of the cement mantle can produce PMMA and/or ZrO2 or BaSO4 particles which can contribute to direct cell responses and also to third-body wear [3, 126].
Acrylic particles found in the capsular tissue present a size range from 1–2 μm to several hundreds of microns. Several shapes have been observed, being the smallest particles similar to dust granules and the largest ones like pearls clusters or grapes bunch [3]. Under light microscopy, particles present a size between 0.5 and 2 μm with grey or yellow brown colour and under polarized light, they show slight white birefringence [129].
Localized areas of osteolysis exhibiting foreign body response to cement particles are frequently observed [60]. PMMA, ZrO2 and BaSO4 particles of phagocytosable size can be internalized and stored within macrophages while the non-phagocytosable large cement fragments are surrounded by foreign body giant cells. Aggressive granulomatous lesions as well as non-granulomatous AL around cemented total hip prostheses have been observed. PMMA particles are also able to activate the lymphocyte-mediated immune response [3].
Cell surface receptor complement receptor 3 (CR3) is involved in the phagocytosis and activation of signaling pathways of macrophages exposed to PMMA [48]. Production of inflammatory mediators is also mediated by TLRs in a manner partly dependent on MyD88 signaling pathway, since inhibition of this adaptor molecule decreased the induced production of TNF-α in RAW 264.7 murine macrophages exposed to PMMA [22]. As described for polymeric and metallic particles, PMMA particles activate the NALP3 inflammasome [130]. Phagocytosed cement particles induce caspase-1 activation in monocyte/macrophage cells and the release of downstream effectors IL-1β and TNF-α, both involved in the amplification of osteoclastogenesis mediated by RANKL.
In vitro studies suggest that PMMA particles may have more potent osteolytic effects than high density PE particles [131]. In fact, the activation of macrophages by PMMA particles has long been considered as a key mechanism in wear-induced osteolysis of cemented implants [3, 48]. Apart from peri-implant resident cell activation, PMMA particles are also able to recruit peripheral monocyte/macrophages, promoting the systemic trafficking of macrophages to the site of inflammation [3], as observed after PMMA injection in the medullary canal of nude mice and the intravenous administration of RAW264.7 macrophages stably expressing a bioluminescent reporter gene [132]. This finding was corroborated using a severe combined immunodeficiency mouse chimera model, in which fragments of periprosthetic granulomatous tissues and bone chips retrieved during revision surgery in loosened patients were implanted in mice [133]. PBMCs isolated from patients during revision surgery were fluorescently labeled and cultured with cement particles before intraperitoneal injection. Fluorescent-labeled PBMCs challenged with PMMA particles and a high number of TRAP-positive cells accumulated in transplanted periprosthetic tissues.
The chemotactic effects induced by PMMA particles leading to migration of human monocyte and MSCs have been studied in in vitro studies [60, 79]. Macrophages challenged with cement particles increase MCP-1 release. Like MCP-1, MIP-1α is expressed in periprosthetic tissues and produced by cells of the monocytic/macrophagic lineage after priming with PMMA particles [22]. Moreover, neutralizing antibody to MIP-1α lessened the migration of monocytes induced by media conditioned by macrophages exposed to PMMA particles [134]. An independent study confirmed the involvement of MCP-1 and MIP-1α induced by PMMA particles in the homing of monocytes and MSCs, suggesting the involvement of various chemokines in the recruitment of macrophages and MSCs [135]. Fibroblasts, a cell source of chemokines, increase MCP-1 release upon exposure to PMMA particles [136].
Exposure of macrophages to PMMA increases the expression of pro-inflammatory cytokines, via activation of the NF-κΒ pathway [137, 138]. Among other macrophage-releated cell types, dose- and time-dependent induction of the secretion of IL-1 and PGE2 has been observed in peritoneal macrophages [60] while induction in the release of IL-1β and TNF-α was described in bone marrow macrophages [139]. In line with this, Pearle and colleagues performed microarrays profiling of monocytes and unfractionated PBMCs exposed to PMMA and Ti particles to assess, respectively, the activation of the innate immune system and the innate and adaptive immune system [140]. Exposure of monocytes to PMMA induced the transcript levels of TNF-α, IL-1α, IL-1β, IL-6, IL-8 and COX-2, key regulator of PGE2 synthesis, as well as the chemokines MIP-3α and CCL11. Similar data were obtained in PBMC exposed to PMMA. Comparison with data obtained from cells exposed to Ti particles suggested that PMMA-inflammatory effects are mediated by the activation of the innate immune system, i.e., monocytes, in a T cell-independent manner, while lymphocytes are not essential mediators in PMMA-induced osteolysis.
Apart from the induction in the inflammatory response, PMMA particles contribute to mononuclear phagocytes and macrophages differentiation into TRAP-positive cells with resorptive capacity [131, 141]. Other osteoclastic phenotypic markers might be regulated by PMMA particles, as murine RAW264.7 cells exposed to PMMA upregulated cathepsin K and RANK expression [142]. PMMA particles stimulate osteoclastogenesis, at least in part, by induction of RANKL and TNF-α expression and by activation of NF-кB [143]. Signaling pathways that cooperate with RANKL-induced during differentiation of macrophages into osteoclasts, like MAPK pathway, are also activated. Expression and activity of the transcription factor NFAT2, involved critically in osteoclast lineage commitment, increases in osteoclast precursors exposed to PMMA particles [144]. PMMA-derived effects on the osteoclastic lineage seem to be dependent on their stages of development, increasing the number of osteoclasts precursors and enhancing the number and bone resorption capacity of mature cells [145].
As mentioned before, macrophages exhibit functional plasticity in order to adapt to the dynamic periprosthetic microenvironment. Macrophages challenged by PMMA particles exhibit characteristics of M1 phenotype [146]. However, macrophages may evolve from M1 to M2 phenotype as proven in in vitro experiments in which M1 macrophages were treated with IL-4 prior to exposure to PMMA particles or were simultaneously treated with IL-4 and particles [139].
Exposure to PMMA particles impairs viability, proliferation and osteogenic differentiation of osteoprogenitor cells. Transcript levels of the transcription factors Runx2, Osx and Dlx5 that orchestrate osteogenic differentiation, and OCN decrease after exposure of MC3T3-E1 to PMMA particles [138, 147]. Effects on osteoprogenitor proliferation and differentiation have been found to be dose-dependent [148]. The more potent inhibitory effects of PMMA particles have been detected in the early phase of osteoprogenitor differentiation, as shown in cultures of murine bone marrow cells [148, 149]. PMMA-induced inhibition of osteoblastic differentiation is characterized by altered expression of genes encoding bone morphogenetic protein 3 (BMP3) and SOST, which are negative regulators of bone formation [150].
Regarding mature bone-forming cells, proliferation and collagen synthesis of osteoblastic cells exposed to PMMA were reported to be inhibited whereas OCN and IL-6 production were stimulated [151]. Additionally, apoptotic rates and MMP-1 expression were enhanced in osteoblasts exposed to PMMA [152]. As observed with other types of particles, the mechanisms involved in osteoblasts dysfunction after exposure to PMMA particles are not totally understood. Among other participating mechanisms, inhibition of MAPK activity and reduced TGF-β1 production, which play an essential role in differentiation and the response to environmental stress, have been proposed [150]. A recent study has addressed the consequences of the physical stress stimuli that PMMA particles impose to osteoblasts that include an increase in the elastic modulus with time, production of calcium and changes in cytoskeleton organisation which influence cell behaviour and function [86]. Addition of radio-opaque agents favors macrophage-osteoclast differentiation and their resorptive activity, being more resorptive PMMA containing BaSO4 than ZrO2 [153]. Moreover, exposure of osteoblastic cells to cements containing these radio-opaque agents increased the expression ratio RANKL/OPG [154]. Such imbalance might be related to stimulation of osteoclast differentiation and inhibition of osteoclast apoptosis.
Collected evidence indicates that cement-derived wear particles elicit important adverse cellular reactions that lead to to periprosthetic osteolysis. Cement particles are found in high proportion in the periprosthetic bone bed which facilitates their direct action and severe effects on bone and immune cells.
Perspectives
Research efforts in THR have been oriented toward the improvement of the tribology of materials used in prostheses manufacturing as well as to the elucidation of the biological processes triggered by wear particles [26]. Important advances in orthopaedic materials concerning their microstructure, surface characteristics and/or design have been made and multiple biomaterials have been generated as promising alternatives including advanced composites and hybrid materials [155,156,157]. Among others, coatings of the material surfaces with bioceramics or functionalization with extracellular matrix proteins, biological peptides or growth factors are expected to stimulate physiological mechanisms that counterbalance the development of osteolysis [50, 158]. However, novel materials still face issues regarding their long-term performance and biological response to wear and corrosion debris. Moreover, a deeper understanding of the local and systemic biological responses to materials is needed. The identification of signaling pathways and cellular and molecular mediators that contribute to periprosthetic osteolysis will facilitate not only the generation of mimetic, self-diagnosing and multifunctional materials but also the development of targeted and personalized therapeutic strategies. Innovations in genotyping, pharmacogenomics and large-scale molecular phenotyping will facilitate the identification of the mechanisms involved in the pathogenesis of osteolysis that then will be used to design strategies for the diagnosis, prevention and treatment of AL [24, 159]. For instance, recent progresses in the understanding of the signaling pathways that integrate ER stress, apoptosis, inflammation or osteoclastogenesis have been proposed as promising therapeutic targets to mitigate wear particle-induced osteolysis [94, 160]. Acting upstream of the inflammatory cascade activated by wear particles might lead to better therapeutic control of the local process, in opposition to systemic management which might be associated to adverse side-effects. Three main events triggered by the presence of particles can be targeted: cellular chemotaxis, polarization of macrophages and NF-κΒ signaling [60, 85, 161, 162]. Protein phosphatase 2A, a major serine-threonine phosphatase involved in NF-кB and c-Jun N-terminal kinase signaling pathways has been proposed as new target for pharmacological intervention in Ti-induced osteolysis [163]. Inhibition of the glycogen synthetase kinase 3 beta, regulator in the canonical Wnt signaling pathway which is essential in the maintenance of normal bone mass, has been suggested as a target of molecules to treat wear-debris induced osteolysis [164]. Other proposed targets explored are bone morphogenetic proteins [165] and protein kinase C [166]. Therefore, the more we know about intracellular signaling activated by wear particles, the more options we have to develop therapeutic interventions for aseptic implant loosening.
Other therapeutic intervention has focused in controlling the levels of RANKL and OPG [50]. Specific materials, such as biosilica and microstructured Ti surfaces, are able to stimulate the endogenous production of OPG. Treatments with anti-resorptive bisphosphonates (e.g. alendronate and zolendronate), RANKL antibodies or OPG-like molecules have been proposed. Some of these drugs have proven their efficacy in pathologies associated to catabolic bone disorders, but might have an important impact at systemic level. Unwanted side effects have been also associated to anti-inflammatory drugs as corticosteroids or TNF-α antagonists or IL-1 antagonists, which have been suggested to treat wear-induced osteolysis [51]. Other pharmacological agents assayed include the synthetic molecules OA-14 ((N-(3-(dodecylcarbamoyl)phenyl)-1H-indole-2-carboxamide)) [167] and methotrexate [168] or the ursolic acid [169].
An active research has been performed testing agents able to modulate the NF-кB signaling pathway, including natural bioactive compounds such as triptolide, notoginsenoside R1 or sophocarpine [170,171,172], antibiotics as rifampin [173], bioflavonoids such as anthocyanin or amentoflavone, [174, 175], components of the omega-3 fatty acids such as the docosahexanoic acid [176] or the probiotic Lactobacillus casei [177]. In this search of modulators of NF-кB, the hormone melatonin has been reported as candidate for the treatment of wear debris-induced osteolysis [178]. Sirtuin 1, NAD(+)-dependent histone deacetylase which regulates the transcriptional activity of NF-κB, has been considered as a pharmacological target in osteoblast and macrophages challenged to metal particles [179, 180]. Statins such as simvastatin and pitavastatin, employed as lipid-lowering medication, have been also proposed for the prevention and/or treatment in wear particle-induced bone resorption. Our group reported that simvastatin down-regulates IL-6 secretion in osteoblastic cells cultured in isolation or co-cultured with macrophages and exposed to Ti particles [181]. In order to improve the local efficacy of anti-inflammatory and/or anti-osteolytic therapeutic agents different methods to concentrate the drug preferentially into the inflammatory area have been proposed, avoiding or reducing systemic exposure. In this regard, local delivery of dexamethasone conjugated to the copolymer HPMA or covalently conjugated to TiO2 particles has been assayed [182, 183].
Gene therapy has also emerged as a potential therapeutic avenue. For instance, in vitro and in vivo models have shown the effectiveness of viral gene delivery of IL-10, IL-1Ra and OPG in Ti- and UHMWPE-induced osteolysis models [184,185,186,187,188,189]. Other therapeutic experimental approaches employed small interfering RNA to silence TNF-α, the catalytic subunit of phosphoinositide 3-kinase (PI3K) p110β or the chemokine receptor CXCR2 [190,191,192]. Although promising preliminary results have been achieved, the implementation of gene therapy into orthopaedic practice is still a distant possibility.
Other important aspect to consider is the early diagnosis of wear particles-induced osteolysis. Therefore, efforts are focused in the search of biomarkers as diagnostic and prognostic tools to monitor the progression of the disease. Different biomarkers in synovial fluid, urine and serum have been proposed, but still none have proved relevant clinical utility [193, 194]. In this regard, our group has been focused during the last years in the identification of serum proteins with potential to be regarded as biomarkers.
Concluding Remarks
Wear particles-induced osteolysis is one of the major challenges for orthopedic surgeons due to the absence of clinical signs and symptoms until late stages of destruction and failure. The increasing demand of hip arthroplasties and the growing incidence in young patients, with predictions pointing to a substantial increase in revision surgeries, highlights that this issue has to be seriously considered. Currently, strategies are focused on improving the implant behaviour and limiting the biological response to its degradation products. Despite intense research efforts in the materials field, the long-term performance of novel biomaterials is still unknown. In the biomedical field, it is imperative to unravel the cellular and molecular mechanisms triggered by wear debris to establish effective medical interventions. Too many questions that still remain not answered and become major challenges for basic research groups.
References
Learmonth ID, Young C, Rorabeck C. The operation of the century: total hip replacement. Lancet. 2007;370:1508–19.
Apostu D, Lucaciu O, Berce C, Lucaciu D, Cosma D. Current methods of preventing aseptic loosening and improving osseointegration of titanium implants in cementless total hip arthroplasty: a review. J Int Med Res. 2017; https://doi.org/10.1177/0300060517732697.
Bitar D, Parvizi J. Biological response to prosthetic debris. World J Orthop. 2015;6:172–89. https://doi.org/10.5312/wjo.v6.i2.172.
Singh JA. Epidemiology of knee and hip arthroplasty: a systematic review. Open Orthop J. 2011;5:80–5. https://doi.org/10.2174/1874325001105010080.
Kremers HM, Larson DR, Crowson CS, Kremers WK, Washington RE, Steiner CA, et al. Prevalence of total hip and knee replacement in the United States. J Bone Joint Surg Am. 2015;97:1386–97. https://doi.org/10.2106/JBJS.N.01141.
Dobzyniak M, Fehring TK, Odum S. Early failure in total hip arthroplasty. Clin Orthop Relat Res. 2006;447:76–8.
Melvin JS, Karthikeyan T, Cope R, Fehring TK. Early failures in total hip arthroplasty – a changing paradigm. J Arthroplast. 2014;29:1285–8. https://doi.org/10.1016/j.arth.2013.12.024.
Cherian JJ, Jauregui JJ, Banerjee S, Pierce T, Mont MA. What host factors affect aseptic loosening after THA and TKA? Clin Orthop Relat Res. 2015;473:2700–9. https://doi.org/10.1007/s11999-015-4220-2.
Gibon E, Amanatullah DF, Loi F, Pajarinen J, Nabeshima A, Yao Z, et al. The biological response to orthopaedic implants for joint replacement: part I: metals. J Biomed Mater Res B Appl Biomater. 2017;105:2162–73. https://doi.org/10.1002/jbm.b.33734.
Jansen P, Mumme T, Randau T, Gravius S, Hermanns-Sachweh B. Endoglin (CD105) expression differentiates between aseptic loosening and periprosthetic joint infection after total joint arthroplasty. Springerplus. 2014;3:561. https://doi.org/10.1186/2193-1801-3-561.
Mäkelä KT, Eskelinen A, Pulkkinen P, Paavolainen P, Remes V. Results of 3,668 primary total hip replacements for primary osteoarthritis in patients under the age of 55 years. Acta Orthop. 2011;82:521–9. https://doi.org/10.3109/17453674.2011.618908.
Landgraeber S, Jäger M, Jacobs JJ, Hallab NJ. The pathology of orthopedic implant failure is mediated by innate immune system cytokines. Mediat Inflamm. 2014;2014:185150. https://doi.org/10.1155/2014/185150.
Sukur E, Akman YE, Ozturkmen Y, Kucukdurmaz F. Particle disease: a current review of the biological mechanisms in periprosthetic osteolysis after hip arthroplasty. Open Orthop J. 2016;10:241–51. https://doi.org/10.2174/1874325001610010241.
Pajarinen J, Lin TH, Nabeshima A, Jämsen E, Lu L, Nathan K, et al. Mesenchymal stem cells in the aseptic loosening of total joint replacements. J Biomed Mater Res A. 2017;105:1195–207. https://doi.org/10.1002/jbm.a.35978.
MacInnes SJ, Gordon A, Wilkinson JM. Risk factors for aseptic loosening following Total hip arthroplasty. Recent Adv Arthroplast. 2012; https://doi.org/10.5772/26975.
Veronesi F, Tschon M, Fini M. Gene expression in osteolysis: review on the identification of altered molecular pathways in preclinical and clinical studies. Int J Mol Sci. 2017;25:18. https://doi.org/10.3390/ijms18030499.
Nich C, Takakubo Y, Pajarinen J, Ainola M, Salem A, Sillat T, et al. Macrophages-key cells in the response to wear debris from joint replacements. J Biomed Mater Res A. 2013;101:3033–45. https://doi.org/10.1002/jbm.a.34599.
Gallo J, Goodman SB, Konttinen YT, Raska M. Particle disease: biologic mechanisms of periprosthetic osteolysis in total hip arthroplasty. Innate Immun. 2013;19:213–24. https://doi.org/10.1177/1753425912451779.
Camuzard O, Breuil V, Carle GF, Pierrefite-Carle V. Targeting autophagy to inhibit wear debris-induced osteolysis. AME Med J. 2017;2:5.
Yan Y, Hu J, Lu H, Wang W. Genetic susceptibility to total hip arthroplasty failure: a case-control study on the influence of MMP 1 gene polymorphism. Diagn Pathol. 2014;9:177. https://doi.org/10.1186/s13000-014-0177-9.
Towle KM, Monnot AD. An assessment of gender-specific risk of implant revision after primary total hip arthroplasty: a systematic review and meta-analysis. J Arthroplast. 2016;31:2941–8. https://doi.org/10.1016/j.arth.2016.07.047.
Gu Q, Shi Q, Yang H. The role of TLR and chemokine in wear particle-induced aseptic loosening. J Biomed Biotechnol. 2012;2012:596870. https://doi.org/10.1155/2012/596870.
Hallab NJ, Jacobs JJ. Biologic effects of implant debris. Bull NYU Hosp Jt Dis. 2009;67:182–8.
Terkawi MA, Hamasaki M, Takahashi D, Ota M, Kadoya K, Yutani T, et al. Transcriptional profile of human macrophages stimulated by ultra-high molecular weight polyethylene particulate debris of orthopedic implants uncovers a common gene expression signature of rheumatoid arthritis. Acta Biomater. 2018;65:417–25. https://doi.org/10.1016/j.actbio.2017.11.001.
Ulrich SD, Seyler TM, Bennett D, Delanois RE, Saleh KJ, Thongtrangan I, et al. Total hip arthroplasties: what are the reasons for revision? Int Orthop. 2008;32:597–604.
Howie DW, Neale SD, Haynes DR, Holubowycz OT, McGee MA, Solomon LB, et al. Periprosthetic osteolysis after total hip replacement: molecular pathology and clinical management. Inflammopharmacology. 2013;21:389–96. https://doi.org/10.1007/s10787-013-0192-6.
Iorio R, Robb WJ, Healy WL, Berry DJ, Hozack WJ, Kyle RF, et al. Orthopaedic surgeon workforce and volume assessment for total hip and knee replacement in the United States: preparing for an epidemic. J Bone Joint Surg Am. 2008;90:1598–605.
McGrory BJ, Etkin CD, Lewallen DG. Comparing contemporary revision burden among hip and knee joint replacement registries. Arthroplast Today. 2016;2:83–6. https://doi.org/10.1016/j.artd.2016.04.003.
Nich C, Goodman SB. Role of macrophages in the biological reaction to wear debris from joint replacements. J Long-Term Eff Med Implants. 2014;24:259–65.
Girard J, Glorion C, Bonnomet F, Fron D, Migaud H. Risk factors for revision of hip arthroplasties in patients younger than 30 years. Clin Orthop Relat Res. 2011;469:1141–7. https://doi.org/10.1007/s11999-010-1669-x.
McGonagle L, Siney PD, Raut VV. Fate of the unrevised cemented stem following cup only revision: 227 hips at an average of 6 years follow-up. Orthop Traumatol Surg Res. 2015;101:781–4. https://doi.org/10.1016/j.otsr.2015.08.005.
Magone K, Luckenbill D, Goswami T. Metal ions as inflammatory initiators of osteolysis. Arch Orthop Trauma Surg. 2015;135:683–95. https://doi.org/10.1007/s00402-015-2196-8.
Jiang Y, Jia T, Wooley PH, Yang SY. Current research in the pathogenesis of aseptic implant loosening associated with particulate wear debris. Acta Orthop Belg. 2013;79:1–9.
Pajarinen J, Gallo J, Takagi M, Goodman SB, Mjöberg B. Particle disease really does exist. Acta Orthop. 2018;89:133–6. https://doi.org/10.1080/17453674.2017.1402463.
Bordini B, Stea S, De Clerico M, Strazzari S, Sasdelli A, Toni A. Factors affecting aseptic loosening of 4750 total hip arthroplasties: multivariate survival analysis. BMC Musculoskelet Disord. 2007;24(8):69.
Engh CA, Ho H, Powers CC, Huynh C, Beykirch SE, Hopper RH Jr. Osteolysis propensity among bilateral total hip arthroplasty patients. J Arthroplast. 2011;26:555–61. https://doi.org/10.1016/j.arth.2010.05.014.
Stelmach P, Kauther MD, Fuest L, Kurscheid G, Gehrke T, Klenke S, et al. Relationship between GNAS1 T393C polymorphism and aseptic loosening after total hip arthroplasty. Eur J Med Res. 2017;22:29. https://doi.org/10.1186/s40001-017-0271-z.
MacInnes SJ, Del Vescovo E, Kiss-Toth E, Ollier WE, Kay PR, Gordon A, et al. Genetic variation in inflammatory and bone turnover pathways and risk of osteolytic responses to prosthetic materials. J Orthop Res. 2015;33:193–8. https://doi.org/10.1002/jor.22755.
Kolundzić R, Orlić D, Trkulja V, Pavelić K, Troselj KG. Single nucleotide polymorphisms in the interleukin-6 gene promoter, tumor necrosis factor-alpha gene promoter, and transforming growth factor-beta1 gene signal sequence as predictors of time to onset of aseptic loosening after total hip arthroplasty: preliminary study. J Orthop Sci. 2006;11:592–600.
Gallo J, Mrazek F, Petrek M. Variation in cytokine genes can contribute to severity of acetabular osteolysis and risk for revision in patients with ABG 1 total hip arthroplasty: a genetic association study. BMC Med Genet. 2009;10:109. https://doi.org/10.1186/1471-2350-10-109.
Malik MH, Jury F, Bayat A, Ollier WE, Kay PR. Genetic susceptibility to total hip arthroplasty failure: a preliminary study on the influence of matrix metalloproteinase 1, interleukin 6 polymorphisms and vitamin D receptor. Ann Rheum Dis. 2007;66:1116–20.
Kremers HM, Lewallen EA, van Wijnen AJ, Lewallen DG. Clinical factors, disease parameters, and molecular therapies affecting osseointegration of orthopedic implants. Curr Mol Biol Rep. 2016;2:123–32. https://doi.org/10.1007/s40610-016-0042-6.
Ollivere B, Wimhurst JA, Clark IM, Donell ST. Current concepts in osteolysis. J Bone Joint Surg Br. 2012;94:10–5. https://doi.org/10.1302/0301-620X.94B1.28047.
Ries MD, Link TM. Monitoring and risk of progression of osteolysis after total hip arthroplasty. J Bone Joint Surg Am. 2012;94:2097–105.
Revell PA. The combined role of wear particles, macrophages and lymphocytes in the loosening of total joint prostheses. J R Soc Interface. 2008;5:1263–78. https://doi.org/10.1098/rsif.2008.0142.
Kumar N, Arora GN, Datta B. Bearing surfaces in hip replacement – evolution and likely future. Med J Armed Forces India. 2014;70:371–6. https://doi.org/10.1016/j.mjafi.2014.04.015.
Callaghan JJ, Pedersen DR, Johnston RC, Brown TD. Clinical biomechanics of wear in total hip arthroplasty. Iowa Orthop J. 2003;23:1–12.
Noordin S, Masri B. Periprosthetic osteolysis: genetics, mechanisms and potential therapeutic interventions. Can J Surg. 2012;55:408–17. https://doi.org/10.1503/cjs.003711.
Sundfeldt M, Carlsson LV, Johansson CB, Thomsen P, Gretzer C. Aseptic loosening, not only a question of wear: a review of different theories. Acta Orthop. 2006;77:177–97.
Kapasa ER, Giannoudis PV, Jia X, Hatton PV, Yang XB. The effect of RANKL/OPG balance on reducing implant complications. J Funct Biomater. 2017;8(4):E42. https://doi.org/10.3390/jfb8040042.
Kandahari AM, Yang X, Laroche KA, Dighe AS, Pan D, Cui Q. A review of UHMWPE wear-induced osteolysis: the role for early detection of the immune response. Bone Res. 2016;4:16014. https://doi.org/10.1038/boneres.2016.14.
Vallés G, Gil-Garay E, Munuera L, Vilaboa N. Modulation of the cross-talk between macrophages and osteoblasts by titanium-based particles. Biomaterials. 2008;29:2326–35. https://doi.org/10.1016/j.biomaterials.2008.02.011.
Vallés G, González-Melendi P, Saldaña L, Rodriguez M, Munuera L, Vilaboa N. Rutile and titanium particles differentially affect the production of osteoblastic local factors. J Biomed Mater Res A. 2008;84:324–36.
Vallés G, González-Melendi P, González-Carrasco JL, Saldaña L, Sánchez-Sabaté E, Munuera L, et al. Differential inflammatory macrophage response to rutile and titanium particles. Biomaterials. 2006;27:5199–211.
Kobayashi A, Freeman MA, Bonfield W, Kadoya Y, Yamac T, Al-Saffar N, et al. Number of polyethylene particles and osteolysis in total joint replacements. A quantitative study using a tissue-digestion method. J Bone Joint Surg Br. 1997;79:844–8.
Lähdeoja T, Pajarinen J, Kouri VP, Sillat T, Salo J, Konttinen YT. Toll-like receptors and aseptic loosening of hip endoprosthesis-a potential to respond against danger signals? J Orthop Res. 2010;28:184–90. https://doi.org/10.1002/jor.20979.
Xing Z, Pabst MJ, Hasty KA, Smith RA. Accumulation of LPS by polyethylene particles decreases bone attachment to implants. J Orthop Res. 2006;24:959–66.
Alley C, Haggard W, Smith R. Effect of UHMWPE particle size, dose, and endotoxin on in vitro macrophage response. J Long-Term Eff Med Implants. 2014;24:45–56.
Zaveri TD, Dolgova NV, Lewis JS, Hamaker K, Clare-Salzler MJ, Keselowsky BG. Macrophage integrins modulate response to ultra-high molecular weight polyethylene particles and direct particle-induced osteolysis. Biomaterials. 2017;115:128–40. https://doi.org/10.1016/j.biomaterials.2016.10.038.
Gibon E, Córdova LA, Lu L, Lin TH, Yao Z, Hamadouche M, et al. The biological response to orthopedic implants for joint replacement. II: polyethylene, ceramics, PMMA, and the foreign body reaction. J Biomed Mater Res B Appl Biomater. 2017;105:1685–91. https://doi.org/10.1002/jbm.b.33676.
Maitra R, Clement CC, Scharf B, Crisi GM, Chitta S, Paget D, et al. Endosomal damage and TLR2 mediated inflammasome activation by alkane particles in the generation of aseptic osteolysis. Mol Immunol. 2009;47:175–84. https://doi.org/10.1016/j.molimm.2009.09.023.
Goodman SB, Ma T. Cellular chemotaxis induced by wear particles from joint replacements. Biomaterials. 2010;31:5045–50. https://doi.org/10.1016/j.biomaterials.2010.03.046.
Gibon E, Ma T, Ren PG, Fritton K, Biswal S, Yao Z, et al. Selective inhibition of the MCP-1-CCR2 ligand-receptor axis decreases systemic trafficking of macrophages in the presence of UHMWPE particles. J Orthop Res. 2012;30:547–53. https://doi.org/10.1002/jor.21548.
Liu A, Richards L, Bladen CL, Ingham E, Fisher J, Tipper JL. The biological response to nanometre-sized polymer particles. Acta Biomater. 2015;23:38–51. https://doi.org/10.1016/j.actbio.2015.05.016.
Kauther MD, Xu J, Wedemeyer C. Alpha-calcitonin gene-related peptide can reverse the catabolic influence of UHMWPE particles on RANKL expression in primary human osteoblasts. Int J Biol Sci. 2010;6:525–36.
Atkins GJ, Welldon KJ, Holding CA, Haynes DR, Howie DW, Findlay DM. The induction of a catabolic phenotype in human primary osteoblasts and osteocytes by polyethylene particles. Biomaterials. 2009;30:3672–81. https://doi.org/10.1016/j.biomaterials.2009.03.035.
Chiu R, Ma T, Smith RL, Goodman SB. Ultrahigh molecular weight polyethylene wear debris inhibits osteoprogenitor proliferation and differentiation in vitro. J Biomed Mater Res A. 2009;89:242–7. https://doi.org/10.1002/jbm.a.32001.
Ormsby RT, Cantley M, Kogawa M, Solomon LB, Haynes DR, Findlay DM, Atkins GJ. Evidence that osteocyte perilacunar remodelling contributes to polyethylene wear particle induced osteolysis. Acta Biomater. 2016;33:242–51. https://doi.org/10.1016/j.actbio.2016.01.016.
Sartori M, Vincenzi F, Ravani A, Cepollaro S, Martini L, Varani K, et al. RAW 264.7 co-cultured with ultra-high molecular weight polyethylene particles spontaneously differentiate into osteoclasts: an in vitro model of periprosthetic osteolysis. J Biomed Mater Res A. 2017;105:510–20. https://doi.org/10.1002/jbm.a.35912.
Pan X, Mao X, Cheng T, Peng X, Zhang X, Liu Z, et al. Up-regulated expression of MIF by interfacial membrane fibroblasts and macrophages around aseptically loosened implants. J Surg Res. 2012;176:484–9. https://doi.org/10.1016/j.jss.2011.09.047.
Rao AJ, Nich C, Dhulipala LS, Gibon E, Valladares R, Zwingenberger S, et al. Local effect of IL-4 delivery on polyethylene particle induced osteolysis in the murine calvarium. J Biomed Mater Res A. 2013;101:1926–34. https://doi.org/10.1002/jbm.a.34486.
Du Z, Zhu Z, Wang Y. The degree of peri-implant osteolysis induced by PEEK, CoCrMo, and HXLPE wear particles: a study based on a porous Ti6Al4V implant in a rabbit model. J Orthop Surg Res. 2018;13:23. https://doi.org/10.1186/s13018-018-0736-y.
Vallés G, García-Cimbrelo E, Vilaboa N. Involvement of extracellular Hsp72 in wear particle-mediated osteolysis. Acta Biomater. 2012;8:1146–55. https://doi.org/10.1016/j.actbio.2011.12.001.
Vaculova J, Gallo J, Hurnik P, Motyka O, Goodman SB, Dvorackova J. Low intrapatient variability of histomorphological findings in periprosthetic tissues from revised metal/ceramic on polyethylene joint arthroplasties. J Biomed Mater Res B Appl Biomater. 2017; https://doi.org/10.1002/jbm.b.33990.
Hirayama T, Tamaki Y, Takakubo Y, Iwazaki K, Sasaki K, Ogino T, et al. Toll-like receptors and their adaptors are regulated in macrophages after phagocytosis of lipopolysaccharide-coated titanium particles. J Orthop Res. 2011;29:984–92. https://doi.org/10.1002/jor.21369.
Dapunt U, Giese T, Lasitschka F, Reinders J, Lehner B, Kretzer JP, et al. On the inflammatory response in metal-on-metal implants. J Transl Med. 2014;12:74. https://doi.org/10.1186/1479-5876-12-74.
Drynda A, Singh G, Buchhorn GH, Awiszus F, Ruetschi M, Feuerstein B, et al. Metallic wear debris may regulate CXCR4 expression in vitro and in vivo. J Biomed Mater Res A. 2015;103:1940–8. https://doi.org/10.1002/jbm.a.35330.
Geng D, Xu Y, Yang H, Wang J, Zhu X, Zhu G, et al. Protection against titanium particle induced osteolysis by cannabinoid receptor 2 selective antagonist. Biomaterials. 2010;31:1996–2000. https://doi.org/10.1016/j.biomaterials.2009.11.069.
Hallab NJ, Jacobs JJ. Chemokines associated with pathologic responses to orthopedic implant debris. Front Endocrinol (Lausanne). 2017;8:5. https://doi.org/10.3389/fendo.2017.00005.
Chen M, Chen PM, Dong QR, Huang Q, She C, Xu W. p38 signaling in titanium particle-induced MMP-2 secretion and activation in differentiating MC3T3-E1 cells. J Biomed Mater Res A. 2014;102:2824–32. https://doi.org/10.1002/jbm.a.34956.
Qiu S, Zhao F, Tang X, Pei F, Dong H, Zhu L, et al. Type-2 cannabinoid receptor regulates proliferation, apoptosis, differentiation, and OPG/RANKL ratio of MC3T3-E1 cells exposed to titanium particles. Mol Cell Biochem. 2015;399:131–41. https://doi.org/10.1007/s11010-014-2240-y.
Fu C, Xie J, Hu N, Liang X, Chen R, Wang C, et al. Titanium particles up-regulate the activity of matrix metalloproteinase-2 in human synovial cells. Int Orthop. 2014;38:1091–8. https://doi.org/10.1007/s00264-013-2190-0.
Obando-Pereda GA, Fischer L, Stach-Machado DR. Titanium and zirconia particle-induced pro-inflammatory gene expression in cultured macrophages and osteolysis, inflammatory hyperalgesia and edema in vivo. Life Sci. 2014;97:96–106. https://doi.org/10.1016/j.lfs.2013.11.008.
Greenfield EM, Beidelschies MA, Tatro JM, Goldberg VM, Hise AG. Bacterial pathogen-associated molecular patterns stimulate biological activity of orthopaedic wear particles by activating cognate Toll-like receptors. J Biol Chem. 2010;285:32378–84. https://doi.org/10.1074/jbc.M110.136895.
Pajarinen J, Kouri VP, Jämsen E, Li TF, Mandelin J, Konttinen YT. The response of macrophages to titanium particles is determined by macrophage polarization. Acta Biomater. 2013;9:9229–40. https://doi.org/10.1016/j.actbio.2013.06.027.
Preedy EC, Perni S, Prokopovich P. Cobalt, titanium and PMMA bone cement debris influence on mouse osteoblast cell elasticity, spring constant and calcium production activity. RSC Adv. 2015;5:83885–98.
Howie DW, Vernon-Roberts B. Synovial macrophage response to aluminium oxide ceramic and cobalt-chrome alloy wear particles in rats. Biomaterials. 1988;9:442–8.
Devitt BM, Queally JM, Vioreanu M, Butler JS, Murray D, Doran PP, et al. Cobalt ions induce chemokine secretion in a variety of systemic cell lines. Acta Orthop. 2010;81:756–64. https://doi.org/10.3109/17453674.2010.537806.
Jonitz-Heincke A, Tillmann J, Klinder A, Krueger S, Kretzer JP, Høl PJ, et al. The impact of metal ion exposure on the cellular behavior of human osteoblasts and PBMCs: in vitro analyses of osteolytic processes. Materials (Basel). 2017;10:E734. https://doi.org/10.3390/ma10070734.
Rakow A, Schoon J, Dienelt A, John T, Textor M, Duda G, Perka C, Schulze F, Ode A. Influence of particulate and dissociated metal-on-metal hip endoprosthesis wear on mesenchymal stromal cells in vivo and in vitro. Biomaterials. 2016;98:31–40. https://doi.org/10.1016/j.biomaterials.2016.04.023.
Potnis PA, Dutta DK, Wood SC. Toll-like receptor 4 signaling pathway mediates proinflammatory immune response to cobalt-alloy particles. Cell Immunol. 2013;282:53–65. https://doi.org/10.1016/j.cellimm.2013.04.003.
Raghunathan VK, Devey M, Hawkins S, Hails L, Davis SA, Mann S, et al. Influence of particle size and reactive oxygen species on cobalt chrome nanoparticle-mediated genotoxicity. Biomaterials. 2013;34:3559–70. https://doi.org/10.1016/j.biomaterials.2013.01.085.
Papageorgiou I, Brown C, Schins R, Singh S, Newson R, Davis S, Fisher J, Ingham E, Case CP. The effect of nano- and micron-sized particles of cobalt-chromium alloy on human fibroblasts in vitro. Biomaterials. 2007;28:2946–58.
Liu G, Guo T, Zhang Y, Liu N, Chen J, Chen J, Zhang J, Zhao J. Apoptotic pathways of macrophages within osteolytic interface membrane in periprosthestic osteolysis after total hip replacement. APMIS. 2017;125:565–78. https://doi.org/10.1111/apm.12679.
Lewis AC, Ladon D, Heard PJ, Peto L, Learmonth I. The role of the surface chemistry of CoCr alloy particles in the phagocytosis and DNA damage of fibroblast cells. J Biomed Mater Res A. 2007;82:363–72.
Athanasou NA. The pathobiology and pathology of aseptic implant failure. Bone Joint Res. 2016;5:162–8. https://doi.org/10.1302/2046-3758.55.BJR-2016-0086.
Daniel J, Holland J, Quigley L, Sprague S, Bhandari M. Pseudotumors associated with total hip arthroplasty. J Bone Joint Surg Am. 2012;94:86–93. https://doi.org/10.2106/JBJS.J.01612.
Catelas I, Lehoux EA, Ning Z, Figeys D, Baskey SJ, Beaulé PE. Differential proteomic analysis of synovial fluid from hip arthroplasty patients with a pseudotumor vs. periprosthetic osteolysis. J Orthop Res. 2018; https://doi.org/10.1002/jor.23858.
McCarthy CL, Uchihara Y, Vlychou M, Grammatopoulos G, Athanasou NA. Development of malignant lymphoma after metal-on-metal hip replacement: a case report and review of the literature. Skelet Radiol. 2017;46:831–6. https://doi.org/10.1007/s00256-017-2612-y.
Gallo J, Goodman SB, Lostak J, Janout M. Advantages and disadvantages of ceramic on ceramic total hip arthroplasty: a review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2012;156:204–12. https://doi.org/10.5507/bp.2012.063.
Mehmood S, Jinnah RH, Pandit H. Review on ceramic-on-ceramic total hip arthroplasty. J Surg Orthop Adv. 2008;17:45–50.
Tateiwa T, Clarke IC, Williams PA, Garino J, Manaka M, Shishido T, Yamamoto K, Imakiire A. Ceramic total hip arthroplasty in the United States: safety and risk issues revisited. Am J Orthop (Belle Mead NJ). 2008;37:E26–31.
Howard DP, Wall PDH, Fernandez MA, Parsons H, Howard PW. Ceramic-on-ceramic bearing fractures in total hip arthroplasty: an analysis of data from the National Joint Registry. Bone Joint J. 2017;99-B:1012–9. https://doi.org/10.1302/0301-620X.99B8.BJJ-2017-0019.R1.
Lee YK, Yoon BH, Choi YS, Jo WL, Ha YC, Koo KH. Metal on metal or ceramic on ceramic for cementless total hip arthroplasty: a meta-analysis. J Arthroplast. 2016;31:2637–45. https://doi.org/10.1016/j.arth.2016.04.014.
Mochida Y, Boehler M, Salzer M, Bauer TW. Debris from failed ceramic-on-ceramic and ceramic-on-polyethylene hip prostheses. Clin Orthop Relat Res. 2001;389:113–25.
Hatton A, Nevelos JE, Nevelos AA, Banks RE, Fisher J, Ingham E. Alumina-alumina artificial hip joints. Part I: a histological analysis and characterisation of wear debris by laser capture microdissection of tissues retrieved at revision. Biomaterials. 2002;23:3429–40.
Bylski D, Wedemeyer C, Xu J, Sterner T, Löer F, von Knoch M. Alumina ceramic particles, in comparison with titanium particles, hardly affect the expression of RANK-, TNF-alpha-, and OPG-mRNA in the THP-1 human monocytic cell line. J Biomed Mater Res A. 2009;89:707–16. https://doi.org/10.1002/jbm.a.31956.
Savarino L, Baldini N, Ciapetti G, Pellacani A, Giunti A. Is wear debris responsible for failure in alumina-on-alumina implants? Acta Orthop. 2009;80:162–7. https://doi.org/10.3109/17453670902876730.
Man K, Jiang LH, Foster R, Yang XB. Immunological responses to total hip arthroplasty. J Funct Biomater. 2017;8:E33. https://doi.org/10.3390/jfb8030033.
Wooley PH. How has the introduction of new bearing surfaces altered the biological reactions to byproducts of wear and modularity? Clin Orthop Relat Res. 2014;472:3699–708. https://doi.org/10.1007/s11999-014-3725-4.
Warashina H, Sakano S, Kitamura S, Yamauchi KI, Yamaguchi J, Ishiguro N, et al. Biological reaction to alumina, zirconia, titanium and polyethylene particles implanted onto murine calvaria. Biomaterials. 2003;24:3655–61.
Kaufman AM, Alabre CI, Rubash HE, Shanbhag AS. Human macrophage response to UHMWPE, TiAlV, CoCr, and alumina particles: analysis of multiple cytokines using protein arrays. J Biomed Mater Res A. 2008;84:464–74.
Petit A, Catelas I, Antoniou J, Zukor DJ, Huk OL. Differential apoptotic response of J774 macrophages to alumina and ultra-high-molecular-weight polyethylene particles. J Orthop Res. 2002;20:9–15.
Germain MA, Hatton A, Williams S, Matthews JB, Stone MH, Fisher J, et al. Comparison of the cytotoxicity of clinically relevant cobalt-chromium and alumina ceramic wear particles in vitro. Biomaterials. 2003;24:469–79.
Gutwein LG, Webster TJ. Increased viable osteoblast density in the presence of nanophase compared to conventional alumina and titania particles. Biomaterials. 2004;25:4175–83.
Lerouge S, Huk O, Yahia LH, Sedel L. Characterization of in vivo wear debris from ceramic-ceramic total hip arthroplasties. J Biomed Mater Res. 1996;32:627–33.
Liagre B, Moalic S, Vergne P, Charissoux JL, Bernache-Assollant D, Beneytout JL. Effects of alumina and zirconium dioxide particles on arachidonic acid metabolism and proinflammatory interleukin production in osteoarthritis and rheumatoid synovial cells. J Bone Joint Surg Br. 2002;84:920–30.
Catelas I, Huk OL, Petit A, Zukor DJ, Marchand R, Yahia L. Flow cytometric analysis of macrophage response to ceramic and polyethylene particles: effects of size, concentration, and composition. J Biomed Mater Res. 1998;41:600–7.
Lucarelli M, Gatti AM, Savarino G, Quattroni P, Martinelli L, Monari E, et al. Innate defence functions of macrophages can be biased by nano-sized ceramic and metallic particles. Eur Cytokine Netw. 2004;15:339–46.
Pasold J, Markhoff J, Tillmann J, Krogull M, Pisowocki P, Bader R. Direct influence of titanium and zirconia particles on the morphology and functionality of mature human osteoclasts. J Biomed Mater Res A. 2017;105:2608–15. https://doi.org/10.1002/jbm.a.36114.
Lohmann CH, Dean DD, Köster G, Casasola D, Buchhorn GH, Fink U, Schwartz Z, Boyan BD. Ceramic and PMMA particles differentially affect osteoblast phenotype. Biomaterials. 2002;23:1855–63.
Rodrigo AM, Martinez ME, Saldaña L, Vallés G, Martinez P, González-Carrasco JL, et al. Effects of polyethylene and alpha-alumina particles on IL-6 expression and secretion in primary cultures of human osteoblastic cells. Biomaterials. 2002;23:901–8.
Rodrigo AM, Martínez ME, Martínez P, Escudero ML, Ruíz J, Saldaña L, et al. Effects of MA 956 superalloy and alpha-alumina particles on some markers of human osteoblastic cells in primary culture. J Biomed Mater Res. 2001;54:30–6.
Rodrigo A, Vallés G, Saldaña L, Rodríguez M, Martínez ME, Munuera L, Vilaboa N. Alumina particles influence the interactions of cocultured osteoblasts and macrophages. J Orthop Res. 2006;24:46–54.
Granchi D, Ciapetti G, Amato I, Pagani S, Cenni E, Savarino L, Avnet S, Peris JL, Pellacani A, Baldini N, Giunti A. The influence of alumina and ultra-high molecular weight polyethylene particles on osteoblast-osteoclast cooperation. Biomaterials. 2004;25:4037–45.
Vaishya R, Chauhan M, Vaish A. Bone cement. J Clin Orthop Trauma. 2013;4:157–63. https://doi.org/10.1016/j.jcot.2013.11.005.
Saleh KJ, El Othmani MM, Tzeng TH, Mihalko WM, Chambers MC, Grupp TM. Acrylic bone cement in total joint arthroplasty: a review. J Orthop Res. 2016;34:737–44. https://doi.org/10.1002/jor.23184.
Sagbas B, Durakbasa MN. Third-body wear behavior of orthopedic biopolymers. Int J Min Mater Metall Eng (IJMMME). 2016;2:1–7.
Morawietz L, Classen RA, Schröder JH, Dynybil C, Perka C, Skwara A, et al. Proposal for a histopathological consensus classification of the periprosthetic interface membrane. J Clin Pathol. 2006;59:591–7.
Burton L, Paget D, Binder NB, Bohnert K, Nestor BJ, Sculco TP, Santambrogio L, Ross FP, Goldring SR, Purdue PE. Orthopedic wear debris mediated inflammatory osteolysis is mediated in part by NALP3 inflammasome activation. J Orthop Res. 2013;31:73–80. https://doi.org/10.1002/jor.22190.
Pandey R, Quinn J, Joyner C, Murray DW, Triffitt JT, Athanasou NA. Arthroplasty implant biomaterial particle associated macrophages differentiate into lacunar bone resorbing cells. Ann Rheum Dis. 1996;55:388–95.
Ren PG, Lee SW, Biswal S, Goodman SB. Systemic trafficking of macrophages induced by bone cement particles in nude mice. Biomaterials. 2008;29:4760–5. https://doi.org/10.1016/j.biomaterials.2008.09.004.
Yang SY, Zhang K, Bai L, Song Z, Yu H, McQueen DA, et al. Polymethylmethacrylate and titanium alloy particles activate peripheral monocytes during periprosthetic inflammation and osteolysis. J Orthop Res. 2011;29:781–6. https://doi.org/10.1002/jor.21287.
Nakashima Y, Sun DH, Trindade MC, Chun LE, Song Y, Goodman SB, Schurman DJ, Maloney WJ, Smith RL. Induction of macrophage C-C chemokine expression by titanium alloy and bone cement particles. J Bone Joint Surg Br. 1999;81:155–62.
Huang Z, Ma T, Ren PG, Smith RL, Goodman SB. Effects of orthopedic polymer particles on chemotaxis of macrophages and mesenchymal stem cells. J Biomed Mater Res A. 2010;94:1264–9. https://doi.org/10.1002/jbm.a.32803.
Yaszay B, Trindade MC, Lind M, Goodman SB, Smith RL. Fibroblast expression of C-C chemokines in response to orthopaedic biomaterial particle challenge in vitro. J Orthop Res. 2001;19:970–6.
Lin TH, Tamaki Y, Pajarinen J, Waters HA, Woo DK, Yao Z, Goodman SB. Chronic inflammation in biomaterial-induced periprosthetic osteolysis: NF-κB as a therapeutic target. Acta Biomater. 2014;10:1–10. https://doi.org/10.1016/j.actbio.2013.09.034.
Shen Y, Wang W, Li X, Markel DC, Ren W. Mitigative effect of erythromycin on PMMA challenged preosteoblastic MC3T3-E1 cells. Sci World J. 2014;2014:107196. https://doi.org/10.1155/2014/107196.
Antonios JK, Yao Z, Li C, Rao AJ, Goodman SB. Macrophage polarization in response to wear particles in vitro. Cell Mol Immunol. 2013;10:471–82. https://doi.org/10.1038/cmi.2013.39.
Pearle AD, Crow MK, Rakshit DS, Wohlgemuth J, Nestor BJ. Distinct inflammatory gene pathways induced by particles. Clin Orthop Relat Res. 2007;458:194–201.
Sabokbar A, Pandey R, Quinn JM, Athanasou NA. Osteoclastic differentiation by mononuclear phagocytes containing biomaterial particles. Arch Orthop Trauma Surg. 1998;117:136–40.
Li N, Xu Z, Wooley PH, Zhang J, Yang SY. Therapeutic potentials of naringin on polymethylmethacrylate induced osteoclastogenesis and osteolysis, in vitro and in vivo assessments. Drug Des Devel Ther. 2013;8:1–11. https://doi.org/10.2147/DDDT.S52714.
Yamanaka Y, Abu-Amer Y, Faccio R, Clohisy JC. Map kinase c-JUN N-terminal kinase mediates PMMA induction of osteoclasts. J Orthop Res. 2006;24:1349–57.
Yamanaka Y, Abu-Amer W, Foglia D, Otero J, Clohisy JC, Abu-Amer Y. NFAT2 is an essential mediator of orthopedic particle-induced osteoclastogenesis. J Orthop Res. 2008;26:1577–84. https://doi.org/10.1002/jor.20714.
Zhang H, Ricciardi BF, Yang X, Shi Y, Camacho NP, Bostrom MG. Polymethylmethacrylate particles stimulate bone resorption of mature osteoclasts in vitro. Acta Orthop. 2008;79:281–8. https://doi.org/10.1080/17453670710015166.
Rao AJ, Gibon E, Ma T, Yao Z, Smith RL, Goodman SB. Revision joint replacement, wear particles, and macrophage polarization. Acta Biomater. 2012;8:2815–23. https://doi.org/10.1016/j.actbio.2012.03.042.
Chiu R, Smith KE, Ma GK, Ma T, Smith RL, Goodman SB. Polymethylmethacrylate particles impair osteoprogenitor viability and expression of osteogenic transcription factors Runx2, osterix, and Dlx5. J Orthop Res. 2010;28:571–7. https://doi.org/10.1002/jor.21035.
Chiu R, Ma T, Smith RL, Goodman SB. Polymethylmethacrylate particles inhibit osteoblastic differentiation of bone marrow osteoprogenitor cells. J Biomed Mater Res A. 2006;77:850–6.
Chiu R, Ma T, Smith RL, Goodman SB. Kinetics of polymethylmethacrylate particle-induced inhibition of osteoprogenitor differentiation and proliferation. J Orthop Res. 2007;25:450–7.
Ma GK, Chiu R, Huang Z, Pearl J, Ma T, Smith RL, et al. Polymethylmethacrylate particle exposure causes changes in p38 MAPK and TGF-beta signaling in differentiating MC3T3-E1 cells. J Biomed Mater Res A. 2010;94:234–40. https://doi.org/10.1002/jbm.a.32686.
Zambonin G, Colucci S, Cantatore F, Grano M. Response of human osteoblasts to polymethylmetacrylate in vitro. Calcif Tissue Int. 1998;62:362–5.
Lochner K, Fritsche A, Jonitz A, Hansmann D, Mueller P, Mueller-Hilke B, et al. The potential role of human osteoblasts for periprosthetic osteolysis following exposure to wear particles. Int J Mol Med. 2011;28:1055–63. https://doi.org/10.3892/ijmm.2011.778.
Sabokbar A, Fujikawa Y, Murray DW, Athanasou NA. Radio-opaque agents in bone cement increase bone resorption. J Bone Joint Surg Br. 1997;79:129–34.
Granchi D, Cenni E, Savarino L, Ciapetti G, Forbicini G, Vancini M, et al. Bone cement extracts modulate the osteoprotegerin/osteoprotegerin-ligand expression in MG63 osteoblast-like cells. Biomaterials. 2002;23:2359–65.
Pezzotti G, Yamamoto K. Artificial hip joints: the biomaterials challenge. J Mech Behav Biomed Mater. 2014;31:3–20. https://doi.org/10.1016/j.jmbbm.2013.06.001.
Ghalme SG, Mankar A, Bhalerao Y. Biomaterials in hip joint replacement. Int J Mater Sci Eng. 2016;4:113–25. https://doi.org/10.17706/ijmse.2016.4.2.113-125.
Grieco PW, Pascal S, Newman JM, Shah NV, Stroud SG, Sheth NP, Maheshwari AV. New alternate bearing surfaces in total hip arthroplasty: a review of the current literature. J Clin Orthop Trauma. 2018;9:7–16. https://doi.org/10.1016/j.jcot.2017.10.013.
Zhang BG, Myers DE, Wallace GG, Brandt M, Choong PF. Bioactive coatings for orthopaedic implants-recent trends in development of implant coatings. Int J Mol Sci. 2014;15:11878–921. https://doi.org/10.3390/ijms150711878.
Cooper HJ. Emerging applications of proteomics in hip and knee arthroplasty. Expert Rev Proteomics. 2014;11:5–8. https://doi.org/10.1586/14789450.2014.865522.
Zhang L, Tian Z, Li W, Wang X, Man Z, Sun S. Inhibitory effect of quercetin on titanium particle-induced endoplasmic reticulum stress (ERS)-related apoptosis and in vivo osteolysis. Biosci Rep. 2017;37:BSR20170961. https://doi.org/10.1042/BSR20170961.
Goodman SB, Gibon E, Pajarinen J, Lin TH, Keeney M, Ren PG, et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J R Soc Interface. 2014;11:20130962. https://doi.org/10.1098/rsif.2013.0962.
Mahon OR, O'Hanlon S, Cunningham CC, McCarthy GM, Hobbs C, Nicolosi V, et al. Orthopaedic implant materials drive M1 macrophage polarization in a spleen tyrosine kinase- and mitogen-activated protein kinase-dependent manner. Acta Biomater. 2018;65:426–35. https://doi.org/10.1016/j.actbio.2017.10.041.
Wang L, Guo X, Zhou W, Ding Y, Shi J, Wu X, et al. Protein phosphatase 2A as a new target for downregulating osteoclastogenesis and alleviating titanium particle-induced bone resorption. Acta Biomater. 2018; https://doi.org/10.1016/j.actbio.2018.04.013.
Geng D, Wu J, Shao H, Zhu S, Wang Y, Zhang W, et al. Pharmaceutical inhibition of glycogen synthetase kinase 3 beta suppresses wear debris-induced osteolysis. Biomaterials. 2015;69:12–21. https://doi.org/10.1016/j.biomaterials.2015.07.061.
Nam JS, Sharma AR, Jagga S, Lee DH, Sharma G, Nguyen LT, et al. Suppression of osteogenic activity by regulation of WNT and BMP signaling during titanium particle induced osteolysis. J Biomed Mater Res A. 2017;105:912–26. https://doi.org/10.1002/jbm.a.36004.
Drynda A, Ren Q, Buchhorn GH, Lohmann CH. The induction of CXCR4 expression in human osteoblast-like cells (MG63) by CoCr particles is regulated by the PLC-DAG-PKC pathway. J Biomed Mater Res B Appl Biomater. 2017;105:2326–32. https://doi.org/10.1002/jbm.b.33770.
Tian B, Jiang T, Shao Z, Zhai Z, Li H, Fan Q, et al. The prevention of titanium-particle-induced osteolysis by OA-14 through the suppression of the p38 signaling pathway and inhibition of osteoclastogenesis. Biomaterials. 2014;35:8937–50. https://doi.org/10.1016/j.biomaterials.2014.06.055.
Mediero A, Perez-Aso M, Wilder T, Cronstein BN. Brief report: methotrexate prevents wear particle-induced inflammatory osteolysis in mice via activation of adenosine A2A receptor. Arthritis Rheumatol. 2015;67:849–55. https://doi.org/10.1002/art.38971.
Jiang C, Xiao F, Gu X, Zhai Z, Liu X, Wang W, et al. Inhibitory effects of ursolic acid on osteoclastogenesis and titanium particle-induced osteolysis are mediated primarily via suppression of NF-κB signaling. Biochimie. 2015;111:107–18. https://doi.org/10.1016/j.biochi.2015.02.002.
Huang J, Zhou L, Wu H, Pavlos N, Chim SM, Liu Q, et al. Triptolide inhibits osteoclast formation, bone resorption, RANKL-mediated NF-қB activation and titanium particle-induced osteolysis in a mouse model. Mol Cell Endocrinol. 2015;399:346–53. https://doi.org/10.1016/j.mce.2014.10.016.
Zhao S, Yan L, Li X, Zhang Z, Sun Y, Wang J. Notoginsenoside R1 suppresses wear particle-induced osteolysis and RANKL mediated osteoclastogenesis in vivo and in vitro. Int Immunopharmacol. 2017;47:118–25. https://doi.org/10.1016/j.intimp.2017.03.018.
Zhou CH, Shi ZL, Meng JH, Hu B, Zhao CC, Yang YT, et al. Sophocarpine attenuates wear particle-induced implant loosening by inhibiting osteoclastogenesis and bone resorption via suppression of the NF-κB signalling pathway in a rat model. Br J Pharmacol. 2018;175:859–76. https://doi.org/10.1111/bph.14092.
Zhu L, Kang H, Guo CA, Fan WS, Wang YM, Deng LF, et al. Rifampin suppresses osteoclastogenesis and titanium particle-induced osteolysis via modulating RANKL signaling pathways. Biochem Biophys Res Commun. 2017;484:64–70. https://doi.org/10.1016/j.bbrc.2017.01.071.
Li Y, Li J, Li B, Qin H, Peng X, Zhao Y, et al. Anthocyanin suppresses CoCrMo particle-induced osteolysis by inhibiting IKKα/β mediated NF-κB signaling in a mouse calvarial model. Mol Immunol. 2017;85:27–34. https://doi.org/10.1016/j.molimm.2017.02.003.
Zhang Z, Zhao S, Li X, Zhuo X, Zhang W, Nie Q, et al. Amentoflavone inhibits osteoclastogenesis and wear debris-induced osteolysis via suppressing NF-κB and MAPKs signaling pathways. Planta Med. 2018;84:759–67. https://doi.org/10.1055/s-0043-124594.
Kim HJ, Ohk B, Yoon HJ, Kang WY, Seong SJ, Kim SY, et al. Docosahexaenoic acid signaling attenuates the proliferation and differentiation of bone marrow-derived osteoclast precursors and promotes apoptosis in mature osteoclasts. Cell Signal. 2017;29:226–32. https://doi.org/10.1016/j.cellsig.2016.11.007.
Wang Z, Xue K, Bai M, Deng Z, Gan J, Zhou G, et al. Probiotics protect mice from CoCrMo particles-induced osteolysis. Int J Nanomedicine. 2017;12:5387–97. https://doi.org/10.2147/IJN.S130485.
Ping Z, Wang Z, Shi J, Wang L, Guo X, Zhou W, et al. Inhibitory effects of melatonin on titanium particle-induced inflammatory bone resorption and osteoclastogenesis via suppression of NF-κB signaling. Acta Biomater. 2017;62:362–71. https://doi.org/10.1016/j.actbio.2017.08.046.
Deng Z, Wang Z, Jin J, Wang Y, Bao N, Gao Q, et al. SIRT1 protects osteoblasts against particle-induced inflammatory responses and apoptosis in aseptic prosthesis loosening. Acta Biomater. 2017;49:541–54. https://doi.org/10.1016/j.actbio.2016.11.051.
Deng Z, Jin J, Wang Z, Wang Y, Gao Q, Zhao J. The metal nanoparticle-induced inflammatory response is regulated by SIRT1 through NF-κB deacetylation in aseptic loosening. Int J Nanomedicine. 2017;12:3617–36. https://doi.org/10.2147/IJN.S124661.
Vallés G, Pérez C, Boré A, Martín-Saavedra F, Saldaña L, Vilaboa N. Simvastatin prevents the induction of interleukin-6 gene expression by titanium particles in human osteoblastic cells. Acta Biomater. 2013;9:4916–25. https://doi.org/10.1016/j.actbio.2012.08.027.
Ren K, Dusad A, Yuan F, Yuan H, Purdue PE, Fehringer EV, et al. Macromolecular prodrug of dexamethasone prevents particle-induced peri-implant osteolysis with reduced systemic side effects. J Control Release. 2014;175:1–9. https://doi.org/10.1016/j.jconrel.2013.11.024.
Rodrigues M, Perni SD, Sloan A, Prokopovich PD. Dexamethasone-loaded TiO2 nanoparticles to locally target wear-debris induced inflammation. Front Bioeng Biotechnol. Conference Abstract: 10th World Biomaterials Congress. 2016; https://doi.org/10.3389/conf.FBIOE.2016.01.01681.
Carmody EE, Schwarz EM, Puzas JE, Rosier RN, O'Keefe RJ. Viral interleukin-10 gene inhibition of inflammation, osteoclastogenesis, and bone resorption in response to titanium particles. Arthritis Rheum. 2002;46:1298–308.
Yang SY, Wu B, Mayton L, Mukherjee P, Robbins PD, Evans CH, et al. Protective effects of IL-1Ra or vIL-10 gene transfer on a murine model of wear debris-induced osteolysis. Gene Ther. 2004;11:483–91.
Ulrich-Vinther M, Carmody EE, Goater JJ, S balle K, O'Keefe RJ, Schwarz EM. Recombinant adeno-associated virus-mediated osteoprotegerin gene therapy inhibits wear debris-induced osteolysis. J Bone Joint Surg Am. 2002;84-A:1405–12.
Yang SY, Mayton L, Wu B, Goater JJ, Schwarz EM, Wooley PH. Adeno-associated virus-mediated osteoprotegerin gene transfer protects against particulate polyethylene-induced osteolysis in a murine model. Arthritis Rheum. 2002;46:2514–23.
Wang H, Jia TH, Zacharias N, Gong W, Du HX, Wooley PH, et al. Combination gene therapy targeting on interleukin-1β and RANKL for wear debris-induced aseptic loosening. Gene Ther. 2013;20:128–35. https://doi.org/10.1038/gt.2012.1.
Langlois J, Hamadouche M. New animal models of wear-particle osteolysis. Int Orthop. 2011;35:245–51. https://doi.org/10.1007/s00264-010-1143-0.
Qin CQ, Huang DS, Zhang C, Song B, Huang JB, Ding Y. Lentivirus-mediated short hairpin RNA interference targeting TNF-alpha in macrophages inhibits particle-induced inflammation and osteolysis in vitro and in vivo. BMC Musculoskelet Disord. 2016;17:431.
Huang JB, Ding Y, Huang DS, Zeng WK, Guan ZP, Zhang ML. rna interference targeting p110β reduces tumor necrosis factor-alpha production in cellular response to wear particles in vitro and osteolysis in vivo. Inflammation. 2013;36:1041–54. https://doi.org/10.1007/s10753-013-9636-9.
Wang C, Liu Y, Wang Y, Li H, Zhang RX, He MS, et al. Adenovirus-mediated siRNA targeting CXCR2 attenuates titanium particle-induced osteolysis by suppressing osteoclast formation. Med Sci Monit. 2016;22:727–35.
Mertens MT, Singh JA. Biomarkers in arthroplasty: a systematic review. Open Orthop J. 2011;5:92–105. https://doi.org/10.2174/1874325001105010092.
Illingworth KD, Wachter N, Maloney WJ, Paprosky WG, Ries MD, Saleh KJ. Advances in acetabular osteolysis: biomarkers, imaging, and pharmacologic management. Instr Course Lect. 2014;63:177–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Vallés, G., Vilaboa, N. (2019). Osteolysis After Total Hip Arthroplasty: Basic Science. In: García-Rey, E., García-Cimbrelo, E. (eds) Acetabular Revision Surgery in Major Bone Defects. Springer, Cham. https://doi.org/10.1007/978-3-319-98596-1_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-98596-1_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-98595-4
Online ISBN: 978-3-319-98596-1
eBook Packages: MedicineMedicine (R0)