Abstract
In recent years, there has been growing interest in third-generation biofuels, i.e., fuels from algal biomass. Considering microalgae, the production and transformation processes are currently under study by researchers across the world, as microalgae appear to be a promising alternative to meet our sustainability goals in the energy sector. Considering the Life Cycle Assessment (LCA) applied to biofuels from microalgae, a number of studies have been published to date, covering a wide geographical range and analyzing several process configurations. This chapter presents the microalgae-to-biofuel process and a review of the published LCA studies in the field. The findings show that the majority of these studies do not have access to primary data but only to secondary data sources. Most studies do not consider the whole process, but only some of the process stages, thus limiting the relevance of the results to the specific context to which they refer. Only about half of the studies reviewed consider the impacts of water and land use, and only two present a detailed analysis of the economic and social impacts. For this reason, further efforts are still necessary in order to obtain a comprehensive sustainability assessment of this potential solution to the energy problem.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
It is a common belief that the development of green fuel technologies with low CO2 emissions can help meet global energy requirements in a more sustainable fashion, reducing our over-reliance on fossil fuels, which currently meet 80% of the world’s energy demand (Medeiros et al. 2013). In this context, growing evidence has illustrated the high potential for biofuels to improve the sustainability of the energy sector, especially for those countries and regions where fossil fuel availability is limited (Stephens et al. 2010).
For this reason, the exploitation of biomass for energy, and particularly liquid biofuels for use in transport, have been of increasing interest to policymakers, even though first- and second-generation biofuels, based on the use of crops, have received criticism (Crutzen et al. 2008), primarily associated with the use of energy crops and fertile land that generally lead to higher environmental impacts and to an increase in crops prices.
Among the different biomass feedstocks, microalgae has shown great potential as a sustainable feedstock for biofuels (also referred to as third generation biofuels), particularly for biodiesel, especially because microalgae are highly efficient lipid producers (Rickman et al. 2013; Leite et al. 2013). In particular, the lipid content of microalgae may reach up to 70% on an algal biomass dry weight basis mainly depending on species and cultivation conditions (Banerjee et al. 2002).Footnote 1
Microalgal feedstocks have been investigated for different applications and products and several technologies have been proposed and investigated for the commercial production and transformation of microalgae (Grierson et al. 2013; Campbell et al. 2011). Nonetheless, the sustainability of the commercial production of microalgae-based biodiesel has yet to be proven, both from the environmental and economic point of view. The most promising directions that researchers have identified points to year-round cultivation, the ability to use wastewater as a nutrient source, higher solar energy yields and minimal use of arable land (Batan et al. 2011; Dismukes et al. 2008; Williams and Inman 2009). Moreover, it should be noted that microalgae can be cultivated in both salt and fresh water environments, and they are suited to areas where the cultivation of crops could be marginal, challenging, or expensive (Hiibel et al. 2015).
In this chapter, after having introduced the process for the production of biodiesel from microalgae and having analyzed the alternative technological pathways for the different steps of the process, we present a literature review on the environmental performance of microalgae in the production of biodiesel. The review highlights the lack of primary data and high production costs as the main weaknesses, while a promising solution seems to be the use of co-products or by-products from other industrial processes.
2 The Microalgae-to-Biodiesel
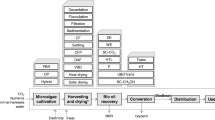
The process for the production and exploitation of biodiesel from microalgae generally follows the scheme outlined in Fig. 10.1 and consists of seven main steps that can employ different technologies/chemicals/processes.
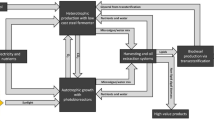
During cultivation, microalgae are grown in water (or wastewater) and supplied with nutrients, such as nitrogen and phosphorus, and a carbon source, mainly coming from inorganic CO2. For microalgal cultivation, two alternative technologies have traditionally been employed: open ponds, i.e., shallow oval ponds exposed to air and light, which are likely to have lower operating costs, despite having higher net energy ratios and lower productivity rates (Collet et al. 2011; Chisti 2007); and photobioreactors, i.e., enclosed chambers for microalgal growth subjected to natural or (in northern climates) artificial light. These generally have higher operating costs and productivity. The use of wastewater throughout the process seems to be a promising manner to improve the environmental and economic sustainability of algae cultivation (Shrestha et al. 2013; Ficara et al. 2014; Ge and Champagne 2016). Similarly, flue gas from industrial sites (e.g., cement plants, power generation plants, etc.) has been evaluated as a potential source of CO2 (Ge and Champagne 2016; Collotta et al. 2016). For this reason, the co-location of microalgal production facilities with wastewater treatment plants (or anaerobic digestion facilities), providing access to nutrients, waste energy and CO2, could maximize the use of waste resources in an integrated resource management approach and increase the techno-economic feasibility of the overall process (Collotta et al. 2016, 2017b, 2018; Davis et al. 2016; Slade and Bauen 2013; Powers and Baliga 2010).
For the harvesting, which brings algae concentration from about 0.2% to about 20%, different pathways are also utilized. The most commonly considered is flocculation (stimulating the formation of solids flocs within the microalgal slurry) and centrifugation, and sometimes in combination (Collotta et al. 2017a; Lardon et al. 2009). The energy required for harvesting could be decreased through process innovations; for example, increasing PO4 concentration in the growth medium can lead to a phenomenon called auto-flocculation in which the microalgae aggregate in flocs and then precipitate from the culture medium (Clarens et al. 2011). Other approaches have explored the harvesting phase, adopting the high pressure CO2, without requiring the addition of coagulants, in order to separate algae from suspension (Lee et al. 2015).
Dewatering is an important stage as it is an energy-intensive process. This stage is often required to increase the percentage of algal biomass from about 20% to 90–95%, depending on the lipid extraction process requirements. A variety of technologies have been explored for this step including belt dryers, solar and steam dryers, natural gas dryers and co-combustion with coal (Powers and Baliga 2010; Clarens et al. 2011; Lardon et al. 2009; Stephenson et al. 2010; Yang et al. 2011).
Different approaches are also used for the lipid extraction phase, the separation of lipids from the remainder of the biomass, which generally employ a solvent or co-solvent system, supercritical CO2, and in some cases a prior or simultaneous cell disruption technique such as drill pressing, (Brentner et al. 2011) or dry de-gumming (Cox et al. 2014), microwave, sonication, freezing, etc. (Harris et al. 2018). More advanced approaches currently under exploration include the use of switchable hydrophilicity solvents (SHS) at room temperature (Boyd et al. 2012), the CO2 expanded methanol approach (Paudel et al. 2015) or liquid CO2, which present better lipid extraction yields (Paudel et al. 2015).
In the transesterification phase, lipids and alcohols are transformed into methyl or ethyl esters and glycerol. This reaction can be driven with esterification, sonication with a direct esterification and the Honeywell UOP™ process, which involves hydrogenation to produce synthetic hydrocarbons followed by selective hydrocracking and distillation (Brentner et al. 2011; Cox et al. 2014). Direct transesterification can also be adopted, which using supercritical conditions combine the lipid extraction and transesterification in a single phase with wet biomass (Brentner et al. 2011).
The transportation phase is the last step before biofuel usage and is generally implemented using trucks or pipelines, depending on the volumes produced and/or location of the plant. Production facilities should be placed at the most convenient location, for instance close to end users, close to the feedstock supply or close to a cement plant (as a source of CO2) or a wastewater treatment plant (as a source of water and nutrients) (Stephenson et al. 2010; Powers and Baliga 2010; Collotta et al. 2016, 2018; Batan et al. 2016).
Finally, the end-use of the energy product is considered. Baseline comparisons between bio-based product (e.g., biodiesel, biojet) and their petroleum-based counterparts suggest that the impact of some substitutions—for instance, replacing coal-fired electricity—may lead to more significant environmental offsets than others (U.S. Energy Information Administration 2016). Understanding the end-use of the microalgae-based energy product is essential to understanding the overall impact of the system.
3 Literature Review of the LCA Studies on Biodiesel from Microalgae
The application of the Life Cycle Assessment (LCA) methodology to the production of biodiesel from microalgae is an ongoing endeavor, and the results obtained are affected by a high level of uncertainty, mainly because of the lack of large-scale production facilities and, consequently, because of the scarcity of primary data.
In this review, 24 LCA studies analyzing microalgae-to-energy systems have been identified and reviewed. Table 10.1 summarizes the main characteristics of these studies. As it can be seen, only 6 of the studies have used primary data for the life cycle inventory, while the majority have used sensitivity analyses to reduce the impact of uncertainty on the results. Nine studies evaluated systems in the European Union, but none are located in Italy.
As it can be seen from Table 10.2, the upstream process stages are included in the system boundaries in almost all of the studies considered (the cultivation phase is always considered), while the downstream stages are more frequently neglected, especially for what concern the use of the residual biomass, the transportation of the biofuel and its use. This result is particularly relevant, since a number of studies have shown the importance of the definition of system boundaries (Tillman et al. 1994).
The impact categories or indicators considered in the LCA studies under review are shown in Table 10.3.
Since the production of biofuels from microalgae is often cited as a solution to the climate change problem, (Medeiros et al. 2013) it is not unexpected that Global Warming Potential (GWP), which is representative of the combined emissions of several greenhouse gases (primarily CO2, N2O, CH4), is quantified in almost all of the studies (22 of 23).
Within the biofuel life cycle, greenhouse gases mainly come from fossil fuel combustion for the generation of electricity and heat; the use of fuels for product transportation; and the manufacturing and use of chemicals in the process. Other GWP-related impact factors have also been noted in the studies under review, including energy use (10 studies), fossil resource depletion (6 studies), and abiotic depletion (consumption of natural but non-renewable resources—4 studies).
Greenhouse gas emissions are primarily related to energy consumption in the harvesting, dewatering/drying, lipid extraction, and transesterification phases. Some studies argue that harvesting and dewatering could contribute up to 20–30% of operational costs (Uduman et al. 2010; Grima et al. 2003), while other studies identified the lipid extraction and transesterification phases as having the highest energy demands (Khoo et al. 2011).
Given the relevance of energy use, it also has to be highlighted that the related impact factors change significantly with a changing energy mix (Itten et al. 2012), and thus depend on the country or region where the specific study is located. Moreover, it is evident that a microalgae-to-energy system that utilizes waste heat, or derives electricity from an onsite anaerobic digestion plant, could substantially reduce fossil greenhouse gases emissions and likely decrease overall operational costs.
With reference to the land and water requirements, also important when evaluating microalgal production systems, it has to be noted that water availability, which is dependent upon geographic location, is often the most critical issue affecting the feasibility of the process and its operational costs. While both fresh and salt water can theoretically be used; however, fresh water allows for a reduction in operational costs since, in the case of seawater, salts have to be extracted via processes such as evaporation, for example (Gendy and El-Temtamy 2013). In addition, sunlight potential and temperature of the location have been shown to influence the productivity of algae cultivation systems in terms of growth rate (Medeiros et al. 2013). The need for sunny days also defines the potential land requirements for algal production in outdoor environments, as in the case of open raceways ponds (Malik et al. 2015). In particular, in countries or regions with high land costs, open pond cultivation may become unfeasible, unless it can be located within existing industrial facilities. Moreover, cultivation on arable land may raise concerns regarding impacts on food supply.
Although water and land use are clearly important impact factors to consider, only 13 of 24 studies under review included land use, eight examined water use and 12 considered eutrophication. This would suggest that water and land use are not monitored as regularly as greenhouse gas emissions, probably because of a paucity of data or a limited understanding of their importance.
3.1 Economic and Social Impact Assessment
One clear issue that emerges from the analysis of the LCA studies is that several process developments are still required for the production of algal biofuels to be economically viable. In fact, while many have speculated that biofuels from microalgae bring to environmental benefits, at the same time they have been presented to have a low economic feasibility due to the high costs associated with dewatering and lipid extraction (Campbell et al. 2011).
However, in any case, most studies have drawn from bench-scale operations, because of the absence of commercial facilities, and few of the analyses have assessed the potential economic impacts process scale-up. The integration of capital and operating costs would represent a key complement to the environmental impact assessment and, it would also be beneficial to consider the effects of specific policy measures, such as renewable fuel mandates, carbon pricing, or excise tax exemptions.
Three the 24 LCA studies reviewed have incorporated some economic considerations, and one in particular presented an innovative hybrid LCA model that integrated economic and social analyses along the supply chain (Malik et al. 2015). The life cycle costing methodology was used in one of the studies (Campbell et al. 2011), which defined a quite comprehensive model for tracking total production cost, including not only plant facility and main operational costs, but also items often neglected, such as the costs associated with research and development, design, failures, contribution margin loss, corrective and preventive maintenance and plant final disposal.
Other studies estimated the impact of increased or decreased water volume or arable land use on the production costs (Li et al. 2008; Borowitzka and Moheimani 2015) or the feasibility of using regional waste streams as resources (CO2, wastewater and waste heat) for the algae cultivation. (Collotta et al. 2016).
With reference to the social impact assessment of biofuels from microalgae, two studies adopted their use as an impact category to track. In particular, one study (Brentner et al. 2011) examines employment through the unemployment index, while the other simply tracked the full-time workers required to operate the designed system (Malik et al. 2015). These studies suggested a higher number of employees for microalgae-to-energy systems compared to comparable food and nutraceutical production (10 employees) as well as conventional crude oil production facilities (29 employees). The implication is that the effect that microalgae-to-energy facilities may have on host communities, given the labor force demand derived by this plant, should be considered for a complete analysis. Although this is a first step towards the assessment of the social sustainability of biofuels from microalgae, to have a comprehensive and more reliable assessment, other factors need to be included, such as human rights, labor conditions and health and safety benefits, as well as corruption and their effects on the legal system (Ekener-Petersen et al. 2014).
4 Conclusions
In recent years, many advances have been achieved through the research and development of microalgae-to-energy systems. The LCA methodology, as an eco-design tool, can provide a relevant contribution to guiding this development towards sustainability direction.
Considering the state of the art regarding the application of LCA to biofuels from microalgae, one of the clearest evidences is the heterogeneity of the system boundaries adopted. In particular, the review highlighted a wide range of process configurations. Few of the LCAs currently published in this field consider the full range of process stages, most of them investigating five or fewer stages, with the most commonly omitted stages involving the transportation of biofuel to end users and end product use. Although such studies can give a relevant contribution in the specific context to which they refer, they are generally limited in contributing to evaluations of the environmental impacts of an integrated microalgae-to-energy scenario.
Another relevant aspect to highlight is that many LCAs have focused primarily on GWP (as measured via greenhouse gas emissions), while water and land use, highly significant in microalgal production systems, were not nearly as well quantified and analyzed in the selected studies. This is likely due to the fact that researchers have focused on the potential for microalgae-to-energy systems to meet global warming challenges. However, an important lack of primary and secondary data have been highlighted in these systems. Moreover water depletion remains an important topic to investigate for future commercial applications.
Finally, it should also be noted that, for the most part, published LCAs do not take into account the economic and social impacts of microalgae-to-energy systems. In fact, only two studies introduced some aspects of the economic and social benefits of biodiesel production. Although the integration of economic and social considerations in the sustainability assessment of microalgae-to-energy systems still presents a high level of uncertainty, due to their early technological development stage, a comprehensive sustainability assessment is crucial both to provide an impetus for the development and deployment of these technologies, and to give reliable profitability assessment.
Notes
- 1.
Chlorella Vulgaris, with standard Nitrogen fraction, has a lipid content of 175 g/kg with a low heating value of 17.5 MJ/kg.
References
Banerjee A, Sharma R, Chisti Y, Banerjee UC (2002) Botryococcus braunii: a renewable source of hydrocarbons and other chemicals. Crit Rev Biotechnol 22:245–279. https://doi.org/10.1080/07388550290789513
Batan L, Quinn J, Bradley T, Willson B (2011) Net energy and greenhouse gas emissions evaluation of biodiesel derived from microalgae. Environ Sci Technol 45:1160. https://doi.org/10.1021/es1038479
Batan LY, Graff GD, Bradley TH (2016) Techno-economic and Monte Carlo probabilistic analysis of microalgae biofuel production system. Bioresour Technol 219:45–52. https://doi.org/10.1016/j.biortech.2016.07.085
Borowitzka MA, Moheimani NR (2015) Algae for biofuels and energy
Boyd AR, Champagne P, McGinn PJ et al (2012) Switchable hydrophilicity solvents for lipid extraction from microalgae for biofuel production. Bioresour Technol 118:628–632. https://doi.org/10.1016/j.biortech.2012.05.084
Brentner LB, Eckelman MJ, Zimmerman JB (2011) Combinatorial life cycle assessment to inform process design of industrial production of algal biodiesel. Environ Sci Technol 45:7060–7067. https://doi.org/10.1021/es2006995
Campbell PK, Beer T, Batten D (2011) Life cycle assessment of biodiesel production from microalgae in ponds. Bioresour Technol 102:50–56. https://doi.org/10.1016/j.biortech.2010.06.048
Chisti Y (2007) Biodiesel from microalgae. In: Biotechnology advances, pp 294–306. Elsevier, Amsterdam
Clarens AF et al (2010) Environmental life cycle comparison of algae to other bioenergy feedstocks. Environ Sci Technol 44(5):1813–1819
Clarens AF, Nassau H, Resurreccion EP et al (2011) Environmental impacts of algae-derived biodiesel and bioelectricity for transportation. Environ Sci Technol 45:7554–7560
Collet P, Hélias A, Lardon L et al (2011) Life-cycle assessment of microalgae culture coupled to biogas production. Bioresour Technol 102:207–214. https://doi.org/10.1016/j.biortech.2010.06.154
Collet P et al (2014) Biodiesel from microalgae - life cycle assessment and recommendations for potential improvements. Renewable Energy 71:525–533. Available at http://linkinghub.elsevier.com/retrieve/pii/S0960148114003498
Collotta M, Champagne P, Mabee W et al (2016) Environmental assessment of co-location alternatives for a microalgae cultivation plant: a case study in the city of Kingston (Canada). Energy Procedia 95:29–36. https://doi.org/10.1016/j.egypro.2016.09.007
Collotta M, Busi L, Champagne P, et al (2017a) Comparative LCA of three alternative technologies for Lipid extraction in biodiesel from microalgae production. In: Energy procedia
Collotta M, Champagne P, Mabee W et al (2017b) Comparative LCA of flocculation for the harvesting of microalgae for biofuels production. Procedia CIRP 61:756–760. https://doi.org/10.1016/j.procir.2016.11.146
Collotta M, Champagne P, Mabee W, Tomasoni G (2017c) Wastewater and waste CO2 for sustainable biofuels from microalgae. Algal Res
Collotta M, Champagne P, Mabee W, Tomasoni G (2018) Wastewater and waste CO2 for sustainable biofuels from microalgae. Algal Res 29. https://doi.org/10.1016/j.algal.2017.11.013
Cox K, Renouf M, Dargan A et al (2014) Environmental life cycle assessment (LCA) of aviation biofuel from microalgae, Pongamia pinnata, and sugarcane molasses. Biofuels Bioprod Biorefin 8:579–593. https://doi.org/10.1002/bbb.1488
Crutzen PJ, Mosier AR, Smith KA, et al (2008) Physics N2O release from agro-biofuel production negates global warming reduction by replacing fossil fuels, pp 389–395
Davis R, Markham J, Kinchin C, et al (2016) Process design and economics for the production of algal biomass: algal biomass production in open pond systems and processing through dewatering for downstream conversion. Natl Renew Energy Lab 128. https://doi.org/10.2172/1239893
Dismukes GC, Carrieri D, Bennette N et al (2008) Aquatic phototrophs: efficient alternatives to land-based crops for biofuels. Curr Opin Biotechnol 19:235–240. https://doi.org/10.1016/j.copbio.2008.05.007
Ekener-Petersen E, Höglund J, Finnveden G (2014) Screening potential social impacts of fossil fuels and biofuels for vehicles. Energy Policy 73:416–426. https://doi.org/10.1016/j.enpol.2014.05.034
Ficara E, Uslenghi A, Basilico D, Mezzanotte V (2014) Growth of microalgal biomass on supernatant from biosolid dewatering. Water Sci Technol 69:896–902
Ge S, Champagne P (2016) Nutrient removal, microalgal biomass growth, harvesting and lipid yield in response to centrate wastewater loadings. Water Res 88:604–612. https://doi.org/10.1016/j.watres.2015.10.054
Gendy TS, El-Temtamy SA (2013) Commercialization potential aspects of microalgae for biofuel production: an overview. Egypt J Pet 22:43–51. https://doi.org/10.1016/j.ejpe.2012.07.001
Gnansounou E, Raman JK (2016) Life cycle assessment of algae biodiesel and its co-products. Appl Energy 161:300–308. Available at http://linkinghub.elsevier.com/retrieve/pii/S0306261915012702
Grierson S, Strezov V, Bengtsson J (2013) Life cycle assessment of a microalgae biomass cultivation, bio-oil extraction and pyrolysis processing regime. Algal Res 2:299–311. https://doi.org/10.1016/j.algal.2013.04.004
Grima EM, Belarbi E-H, Fernández FGA et al (2003) Recovery of microalgal biomass and metabolites: process options and economics. Biotechnol Adv 20(7–8):491–515
Harris J, Viner K, Champagne P, Jessop P (2018) Advances in microalgal lipid extraction for biofuel production: a review. Biofuels Bioprod Biorefin
Hiibel SR, Lemos MS, Kelly BP, Cushman JC (2015) Evaluation of diverse microalgal species as potential biofuel feedstocks grown using municipal wastewater. Front Energy Res 3:20. https://doi.org/10.3389/fenrg.2015.00020
Holma A et al (2013) Current limits of life cycle assessment framework in evaluating environmental sustainability - case of two evolving biofuel technologies. J Cleaner Production 54
Itten R, Frischknecht R, Stucki M, et al (2012) Life cycle inventories of electricity mixes and grid, pp 1–229
Khoo HH, Sharratt PN, Das P et al (2011) Life cycle energy and CO2 analysis of microalgae-to-biodiesel: preliminary results and comparisons. Bioresour Technol 102:5800–5807. https://doi.org/10.1016/j.biortech.2011.02.055
Lardon L, Hélias A, Sialve B et al (2009) Life-cycle assessment of biodiesel production from microalgae. Environ Sci Technol 43:6475–6481. https://doi.org/10.1021/es900705j
Lee R, Jessop PG, Champagne P, et al (2015) Carbon dioxide pressure-induced coagulation of microalgae subject areas. Authors for correspondence : Philos Trans A. https://doi.org/10.1098/rsta.2015.0016
Leite GB, Abdelaziz AEM, Hallenbeck PC (2013) Algal biofuels: challenges and opportunities. Bioresour Technol 145:134–141. https://doi.org/10.1016/j.biortech.2013.02.007
Malik A, Lenzen M, Ralph PJ, Tamburic B (2015) Hybrid life-cycle assessment of algal biofuel production. Bioresour Technol 184:436–443. https://doi.org/10.1016/j.biortech.2014.10.132
Medeiros DL, Sales EA, Kiperstok A (2013) Energy production from microalgae biomass: the carbon footprint and energy balance. 4th Int Work Adv Clean Prod 12
Montazeri M, Soh L, Pérez‐López P, Zimmerman JB, Eckelman MJ (2016) Time‐dependent life cycle assessment of microalgal biorefinery co‐products. Biofuels, Bioprod Bioref 10:409–421. https://doi.org/10.1002/bbb.1649
Paudel A, Jessop MJ, Stubbins SH et al (2015) Extraction of lipids from microalgae using CO2-expanded methanol and liquid CO2. Bioresour Technol 184:286–290. https://doi.org/10.1016/j.biortech.2014.11.111
Powers SE, Baliga R (2010) Sustainable algae biodiesel production in cold climates. Int J Chem Eng 2010:1–13. https://doi.org/10.1155/2010/102179
Rickman M, Pellegrino J, Hock J et al (2013) Life-cycle and techno-economic analysis of utility-connected algae systems. Algal Res 2:59–65. https://doi.org/10.1016/j.algal.2012.11.003
Shrestha RP, Haerizadeh F, Hildebrand M (2013) Handbook of microalgal culture. Handb Microalgal Cult Appl Phycol Biotechnol 146–167
Slade R, Bauen A (2013) Micro-algae cultivation for biofuels: cost, energy balance, environmental impacts and future prospects. Biomass Bioenerg 53:29–38
Soomro RR, Ndikubwimana T, Zeng X, Lu Y, Lin L, Danquah MK (2016) Development of a two-stage microalgae dewatering process – a life cycle assessment approach. Front Plant Sci 7:113. https://doi.org/10.3389/fpls.2016.00113
Stephens E, Ross IL, King Z et al (2010) An economic and technical evaluation of microalgal biofuels. Nat Biotechnol 28:126–128. https://doi.org/10.1038/nbt0210-126
Stephenson A, Kazamia E, Dennis JS et al (2010) Life-cycle assessment of potential algal biodiesel production in the United Kingdom: a comparison of raceways and air-lift tubular bioreactors. Energy Fuels 24:4062–4077. https://doi.org/10.1021/ef1003123
Tillman A-M, Ekvall T, Baumann H, Rydberg T (1994) Choice of system boundaries in life cycle assessment. J Clean Prod 2:21–29. https://doi.org/10.1016/0959-6526(94)90021-3
Tsang M, Fox‐Lent C, Wallace S, Welp T, Bates M, Linkov I (2015) Life‐cycle impacts of soybean and algae biodiesel: case study of US marine vessels. Biofuels, Bioprod Bioref 9:567–580. https://doi.org/10.1002/bbb.1569
Uduman N, Qi Y, Danquah MK et al (2010) Dewatering of microalgal cultures: a major bottleneck to algae-based fuels. J Renew Sustain Energy, 2. https://doi.org/10.1063/1.3294480
U.S. Energy Information Administration (2016) Short-term energy outlook, p 2
Williams P, Inman D (2009) Environmental and sustainability factors associated with next-generation biofuels in the U.S.: what do we really know? Environ Sci Technol 43:4763–4775. https://doi.org/10.1021/es900250d
Li Y, Horsman M, Wu N, et al. (2008) Biofuels from microalgae. In: Handbook of microalgal culture: applied phycology and biotechnology, pp 566–577
Yang J, Xu M, Zhang X et al (2011) Life-cycle analysis on biodiesel production from microalgae: water footprint and nutrients balance. Bioresour Technol 102:159–165. https://doi.org/10.1016/j.biortech.2010.07.017
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Collotta, M., Champagne, P., Mabee, W., Tomasoni, G., Alberti, M. (2019). Life Cycle Analysis of the Production of Biodiesel from Microalgae. In: Basosi, R., Cellura, M., Longo, S., Parisi, M. (eds) Life Cycle Assessment of Energy Systems and Sustainable Energy Technologies. Green Energy and Technology. Springer, Cham. https://doi.org/10.1007/978-3-319-93740-3_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-93740-3_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-93739-7
Online ISBN: 978-3-319-93740-3
eBook Packages: EnergyEnergy (R0)