Abstract
Nova Scotia minkery farms generate huge amounts of wastewater and continuously ignored the public outcry for their environmental impacts. As a consequence of the Fur Industry Act, approved on January 11, 2013, the industry is forced to employ bioenvironmental technologies for reducing the environmental impacts of its operations. Employing microalgae or cyanobacteria could potentially serve a double purpose to generate highly valuable biomass while assisting in the bioremediation of minkery wastewater. This study aimed to investigate the potential of integrating minkery wastewater into Chlorella vulgaris and Anabaena sp. cultivation. The growth characteristics of Chlorella vulgaris in minkery wastewater were significantly better than those in traditional modified BBM under most of the light cycles employed. The traditional BG-11o was superior compared to minkery wastewater for Anabaena sp. cultivation due to the insufficient nitrate concentration of minkery wastewater. This study proved that the minkery wastewater is a better alternative for microalgae cultivation.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Chlorella vulgaris
- Anabaena sp.
- Minkery wastewater
- Wastewater treatment
- Controlled environment
- Growth medium
- Light cycle
1 Introduction
The mink industry in Nova Scotia began in the 1930s and had grown at a high rate over the past 20 years. Today, there are around 100 mink farms, employing more than 1000 workers in Nova Scotia. According to the 2012 Statistics Canada census, Nova Scotian mink farming accounted for 54.3% of Canada’s total mink production. Even further, the estimated value of the mink industry in Nova Scotia in 2012 was $140 million [1]. This number has dropped since then, due to weak demand and a problematic virus called Aleutian mink disease (AMD) virus . Mink products still consistently form the largest agricultural export in Nova Scotia. Amid the success of Nova Scotian mink industry , there are some concerns about the environmental impact of mink production. The main concerns are feces and urine produced by the mink. Rough estimates suggest that there are at least 18,000 tons of mink manure and 16 million liters of urine generated by mink farms in Nova Scotia each year [2]. Many concerned citizens, academics, and journalists suspect that the impacts of mink production on adjacent surface waters are detrimental. One report concluded that multiple instances of eutrophication and algae blooms were related to discharges from mink farms [3]. As with most agricultural wastewaters, mink discharges have high concentrations of pollutants, such as ammonium and phosphorus [4]. The composition of minkery wastewater will invariably have impacts on the local aquatic ecosystems, thereby damaging vital Nova Scotian resource.
As mentioned above, along with solid wastes, Nova Scotia minkery farms generate huge amounts of wastewater and continuously ignored the public outcry for their environmental impacts. As a consequence of the Fur Industry Act , approved on January 11, 2013, the industry is now forced to employ different bioenvironmental technologies for reducing the environmental impacts of its operations. With the new regulations from this act, mink producers are required to identify means to reduce pollutants in effluent waters [5]. This new government regulation of fur industry will have further implications for operational practices in the future. Minkery wastewater integrated into microalgae and cyanobacteria production as a means to reduce effluent water pollutants may provide a service to the mink industry as it tries to adapt to new regulations. Many studies reported microalgae and cyanobacteria assimilate multiple nutrients present in waste streams, which include ammonium, nitrate, nitrite, and phosphorus, and it has even been found to remove heavy metals along with some toxic organic compounds as well [6, 7]. It is the characteristic of microalgae and cyanobacteria that make them obvious candidates for wastewater treatment . Studies on using the microalgae and cyanobacteria cultivation as a tertiary wastewater treatment process started in the early 1970s [8]. The initial purpose was to treat the secondary wastewater even further to mitigate the potential eutrophication of surface water bodies following discharge [9]. One study even concluded that freshwater microalga Chlorella pyrenoidosa removed nutrients from settled domestic sewage more efficiently than activated sludge process did, suggesting that it would be more economical and desirable to employ microalgae cultivation as the secondary rather than tertiary treatment process [10].
At present, the large-scale microalgae and cyanobacteria production faces a number of cost-related bottlenecks [11, 12]. The energy and fertilizer costs of the large-scale microalgae and cyanobacteria production have been too high to be economically feasible. Despite the tremendous opportunity and value of the large-scale microalgae and cyanobacteria production, it is still hampered by the nagging impediment of reaching economic viability [13]. One study analyzing resource implications of microalgae and cyanobacteria cultivation concluded that nutrients, water, carbon, land, and energy were five main factors of reaching microalgae and cyanobacteria cultivation success [14]. Extensive research has been conducted exploring using a variety of agricultural wastewaters to offset the financial burden of nutrient, water, and even carbon supplies. Many researchers have demonstrated that a number of agricultural wastewaters have potential to be a viable substrate for microalgae and cyanobacteria cultivation with even improved yields when compared to other substrates [15,16,17].
As with most agricultural wastewaters, minkery wastewater has extremely high concentrations of nutrients including ammonium, phosphorus, and even organic carbon [4]. While posing some serious environmental problems, minkery wastewater could potentially be an ideal substrate for the large-scale microalgae and cyanobacteria production, especially under heterotrophic and mixotrophic conditions. At present, the most common organic carbon source for the large-scale microalgae and cyanobacteria production is glucose, and it comes at an enormous expense. Minkery wastewater is extremely rich in organic compounds that could potentially be an excellent organic carbon alternative for heterotrophic and mixotrophic cultivation. Further research is essential because many questions remain unanswered regarding the suitability of using minkery wastewater as a substrate for microalgae and cyanobacteria cultivation. Such concerns include investigating the challenge of introducing a biologically diverse substrate into a pure culture, exploring potential sterilization techniques, quantifying biomass and bioproduct productivities, identifying nutrient deficiencies (if any) in wastewaters, and quantifying remediation efficiencies. Effectively answering these questions will provide invaluable technical information to microalgae and cyanobacteria industry.
In building upon existing research, this study aimed to assess the viability of using minkery wastewaters for microalgae and cyanobacteria cultivation, identify the effect of different cultivation techniques on microalgae and cyanobacteria cultivation, and quantify remediation efficiencies of wastewater treatment using microalgae and cyanobacteria. Our findings will provide the framework for future investigation of using minkery wastewater as a potential resource in Nova Scotia.
2 Materials and Methods
2.1 Strain and Culture Maintenance
Two strains used in this study, namely, Chlorella vulgaris and Anabaena sp., were purified and inoculated in 1-L Erlenmeyer flasks at approximately 20 °C and illuminated with a cool-white fluorescent light (32 W, 6500 K) with a light cycle of 16-h light and 8-h dark. Both flasks were sealed with a plug, allowing for aseptic gas exchange, and placed on a mechanical stirrer (HI 190, Hanna Instruments, USA) with 150-rpm rotation.
Both strains and mediums were purchased from the Canadian Phycological Culture Centre (CPCC) , Department of Biology, University of Waterloo, Ontario, Canada. Chlorella vulgaris was cultured in a modified Bold’s basal medium, and Anabaena sp. was cultured in BG-11o medium (modified by J. Acreman). Table 27.1 summarizes the nutrient content of these two mediums.
2.2 Pretreatment of Minkery Wastewater
Minkery wastewater (cage-washing wastewater) was collected from the Dalhousie University’s Fur Animal Research Centre in Bible Hill, Nova Scotia, and all samples were taken on the same day to ensure homogeneity among samples. Samples were frozen and then thawed as needed in order to discourage biological activity.
Prior to experiments, the raw minkery wastewater was thoroughly homogenized and filtered with filter cloth in order to remove all large solid particles. And then, the minkery wastewater was filtered one more time using 1.5-μm glass microfiber filters (691, VWR, UK). In this way, all side effects of solid particles and indigenous bacteria in minkery wastewater on the cultivation of microalgae and cyanobacteria were eliminated. After filtration, the minkery wastewater was autoclaved at a sustained temperature and pressure of 121°C and 15 psi, respectively, for 20 min; after which, the minkery wastewater was stored at 4°C and prepared for the experiment.
2.3 Experimental Equipment
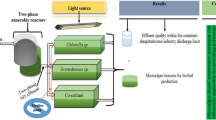
For each experimental unit , cultures were transferred from the flasks to three transparent plastic cylinders, which were 2.25-L vertical column controlled environment photobioreactors (Aqua Medic GmbH, Plankton Reactor, Bissendorf, Germany) (Fig. 27.1). These three photobioreactors essentially served as a holding tank, allowing microalgae or cyanobacteria cultivation and wastewater treatment . Three photobioreactors were employed under three independent cool-white fluorescent lights (8 W, 6700 K) at 20°C. Each fluorescent light can provide enough light for photosynthesis to occur. Three independent ambient air pumps (Fusion Quiet Power, 400, Taiwan) were used to continuously agitate the culture in each photobioreactor in order to keep the microalgae or cyanobacteria culture homogeneous. Moreover, three independent pH meters (Milwaukee, SMS 122, Romania) controlled and continuously monitored the pH of microalgae or cyanobacteria cultures.
Schematic diagram of the photobioreactor . (Adapted from [18], Fig. 30.7)
Once all three photobioreactors were inoculated, the growth period commenced. Upon analyzing the samples at the end of the growth period, all three photobioreactors were taken apart and sanitized carefully and put back together for the proceeding experimental units.
2.4 Experimental Design
In this study, for each photobioreactor, exactly 400-mL purified Chlorella vulgaris or Anabaena sp. inoculum (0.3–0.4 g/L) (20%) and 1600 mL of certain medium (80%) were inoculated. Each experimental unit was limited to 6 days of cultivation. A split-split-plot experimental design was used to arrange each experimental unit accordingly (Table 27.2). The whole plots were strains, the subplots were light cycle s, and the sub-subplots were mediums. In total, the design had 24 different treatments, and three replications for each treatment were used to ensure effective integrity of findings. All statistical analyses were performed using a combination of the Minitab 17 and SAS softwares. As mentioned earlier, strains were Chlorella vulgaris and Anabaena sp.; light cycles were (1) 6-day continuous light, (2) 48-h light and 24-h dark, (3) 24-h light and 48-h dark, and (4) 6-day continuous dark; and mediums were (a) minkery wastewater, (b) traditional mediums (modified Bold’s basal medium and BG-11o medium) (first control), and (c) distilled water (second control).
2.5 Quantification of Growth Kinetics
Four biomass estimation methods are commonly used to determine biomass generated in microalgae and cyanobacteria cultivation, including dry weight, optical density, cell count, and chlorophyll a content. Each method has an ideal sampling condition with regard to strain selection, culture size, growth medium, and desired output accuracy and precision.
Dry weight is a simple and intuitive method for biomass estimation. The major advantages of this method are the useful mass-based units, the simplicity of execution, and the inclusion of constituents based upon minimum particle size. The limiting assumption is that all material collected will be the target organism, which is only applicable in a pure culture. Dry weight of the samples was measured from the triplicate averages of volatile suspended solids (VSS) obtained by means of membrane filtration according to the Standard Methods for the Examination of Water and Wastewater, Method 2540 [19].
Optical density, also known as absorbance or turbidity , is frequently used as a rapid measurement of biomass in cultures. Using absorbance as an indicator offers perhaps the simplest and quickest means for quantifying culture productivity. This method has enormous value because it provides effective noninvasive approximation of biomass. There are however some major disadvantages of this method, including not being able to distinguish dead and live cells, and cellular conglomerates can give faulty readings. The optical density of Chlorella vulgaris was measured by a UV-Vis spectrophotometer (Cole-Parmer, USA) at 684 nm wavelength [20], with a light path of 10 mm, and the optical density of Anabaena sp. was measured with a light path of 10 mm at 683 nm [21], using the same UV-Vis spectrophotometer.
Cell count is another widely used method for estimating biomass through direct enumeration of the cells in the solution. The manual cell count method offers specific advantages in observer control and identification of contaminants. It requires an extensive time commitment and is potentially subjected to observer bias. The Bright-Line™ hemocytometer was used to count the number of cells under the microscope (Motic, BA310). The counting method was taken from Standard Methods for the Examination of Water and Wastewater, Methods 10200E and 10200F [19].
Chlorophyll a is regularly used as an estimator of microalgae and cyanobacteria biomass. The greatest advantage of this method is that it directly targets plant biomass. A substantial margin of error exists in the underlying assumption about the chlorophyll a concentration, because this concentration is highly variable in microalgae and cyanobacteria cells. Chlorophyll a content of the samples was measured according to the Standard Methods for the Examination of Water and Wastewater , Method 150.1 [19].
Overall, methods of optical density and cell count were performed every 12 h, while dry weight and chlorophyll a content methods were carried out at the beginning and end of each growth period. Once these measurements were taken, biomass increase in percentage of each growth period was calculated using Eq. 27.1.
where x0 was the measurement at the beginning of each growth period (day 0) and x6 was the measurement at the end (day 6).
2.6 Evaluation of Wastewater Treatment
The efficiency of wastewater treatment was evaluated by comparing the selected nutrients’ contents of wastewater samples before and after microalgae and cyanobacteria cultivation. All samples were filtered using a Millipore 47 mm vacuum filter assembly with a 0.4-μm glass microfiber filter (GB-140, ADVANTEC, Japan) to remove all microalgae or cyanobacteria cells and other large suspended solids before analysis. Nutrient removal analysis was performed using Hanna Multiparameter Photometer (HI 83200, Hanna Instruments) and corresponding reagents, according to the instruction manual. The tested nutrients were ammonium, nitrate, nitrite, and phosphorus. Once these measurements were taken, efficiencies of wastewater treatment were calculated using Eq. 27.2 below:
where y0 was the nutrient content at the beginning of each growth period (day 0) and y6 was the content at the end of each growth period (day 6).
3 Results and Discussion
When combined with biofuel production, microalgae species from the genus of Chlorella were most commonly used due to its high productivity of fatty acids relevant to transesterification reaction [22]. Anabaena is a genus of filamentous cyanobacteria known for nitrogen-fixing abilities. They are also one of four genera of cyanobacteria that produce neurotoxins, which are harmful to local wildlife. Both Chlorella vulgaris and Anabaena sp. showed potential to simultaneously accumulate biomass and treat minkery wastewater. Thereby, Chlorella vulgaris and Anabaena sp. were selected representing highly valuable and viable microalgae and cyanobacteria, respectively.
3.1 Minkery Wastewater Characteristics
The raw minkery wastewater collected from the Dalhousie Fur Animal Research Centre had an extremely high concentration of some major plant nutrients, such as nitrogen and phosphorus. These nutrient concentrations were much higher than what would normally be needed for microalgae and cyanobacteria cultivation. Therefore, the dilution of raw minkery wastewater should be employed to provide optimum nutrient content for microalgae and cyanobacteria cultivation. In our pre-experimental tests, Chlorella vulgaris and Anabaena sp. were cultivated in various concentrations of diluted minkery wastewaters, and it demonstrated that the most advantageous minkery wastewater concentration for microalgae cultivation ranged from 1% to 3% and the most favorable concentration for cyanobacteria cultivation was between 0.5% and 1%. As a consequence, all pretreated minkery wastewaters in this study were diluted with 99% autoclaved distilled water prior to actual experiment.
The nutrient content of the 1% pretreated minkery wastewater is provided in Table 27.3. Compared to the traditional mediums mentioned earlier, the characteristic of the minkery wastewater is different in terms of nitrogen form. In minkery wastewater, about 99% of total nitrogen was in the form of ammonium, which makes it an excellent growth medium for strains with a high ammonium demand, such as Chlorella vulgaris and Chlorella sp. [23, 24]. It may however not be an ideal growth medium for strains with a high demand in nitrate and nitrite, such as cyanobacteria Anabaena sp. and Anabaena cylindrica [25, 26].
With emerging government regulations [5], the surface and groundwater monitoring are required following recommendations from the designated professionals, and the mink farm owner must sample for total phosphorus, nitrate, and ammonia concentrations of the surface water and groundwater at the farm. If the water sample results come back higher than any of the concentrations listed, the owner of that mink farm may be required to take measures to reduce concentration levels.
Compared to the concentration limits of the surface water and groundwater for mink farms, the 1% pretreated minkery wastewater used in this study already had a much lower nitrate concentration. However, the ammonium and total phosphorus concentrations of the 1% pretreated minkery wastewater were much higher than those concentration limits. It means that a further wastewater treatment would be required to reduce ammonium and total phosphorus concentrations of even 1% pretreated minkery wastewater.
3.2 Biomass Accumulation Comparisons
The means of biomass increase in dry weight of Chlorella vulgaris and Anabaena sp. in various mediums under different light cycle s were provided in Table 27.4.
Based on the findings, Chlorella vulgaris achieved the highest biomass increase in minkery wastewater under the light cycle of 6-day continuous light. This particular treatment yielded a biomass increase of 834% over the 6-day period, which was significantly higher than those under any treatment associated with traditional medium and distilled water.
While Chlorella vulgaris did experience the largest biomass accumulation in minkery wastewater under light cycle of 6-day continuous light, the Tukey’s test (with a confidence coefficient of 95%) determined that there was no significant difference between the mean biomass increase under the light cycles of 6-day continuous light and 48-h light and 24-h dark. This leads to the conclusion that both light cycles, 6-day continuous light and 48-h light and 24-h dark cycles, could be employed for achieving highest biomass cultivation of Chlorella vulgaris in minkery wastewater; and using 48-h light and 24-h dark instead of 6-day continuous light has a potential to reduce energy costs of microalgae and cyanobacteria cultivation.
In contrast, the biomass accumulation of Chlorella vulgaris cultivated in traditional medium and distilled water was observed to be highly dependent on the light cycles. Li et al. [27] reported similar finding in the biomass accumulations of microalgae Chlorella protothecoide and Chlorella kessleri cultured in highly concentrated municipal wastewater correlated to the light cycles .
In comparing these experimental results to the minkery wastewater characteristics, these findings could be easily explained. Both traditional mediums and distilled water used in this study were short of organic carbon (less than 30 mg/L); however, minkery wastewater contained a certain amount of organic carbon (126 mg/L), which makes it a better organic carbon substrate for the heterotrophic cultivation of Chlorella vulgaris. The only deficiency of minkery wastewater used in this study was that its content of organic carbon was too low to support a 6-day heterotrophic cultivation due to the dilution of minkery wastewater before the experiment. As a consequence, although mean biomass increase of Chlorella vulgaris under the light cycle of 6-day continuous dark in minkery wastewater was higher than those in traditional medium and distilled water under the same heterotrophic condition, the Tukey’s test did not determine that there was a statistically significant difference between these findings.
In comparing the mean biomass increase in minkery wastewater between Chlorella vulgaris and Anabaena sp. across each of the light cycles, it was found that there was no significant difference for the 6-day continuous dark. There was, however, a significant difference for the rest of light cycles . Chlorella vulgaris showed a much better adaptation than Anabaena sp. in minkery wastewater because the mean biomass increase of Anabaena sp. was significantly lower than those of Chlorella vulgaris. The growth characteristics of Anabaena sp. in BG-11o medium were significantly better than those in minkery wastewater . Oliveira et al. [25] reported that Anabaena sp. has a high demand in nitrate. Insufficient nitrate of minkery wastewater made the minkery wastewater an inappropriate substrate for Anabaena sp. cultivation.
3.3 Nutrient Removal Comparisons
The means of total nitrogen removal from various mediums via cultivation of Chlorella vulgaris and Anabaena sp. under different light cycles were provided in Table 27.5.
In comparing the total nitrogen removal from minkery wastewater between Chlorella vulgaris and Anabaena sp. across each of the light cycles , it was found that Chlorella vulgaris has higher potential than Anabaena sp. to treat minkery wastewater and accumulate highly valuable biomass simultaneously. The total nitrogen removal from minkery wastewater via cultivation of Chlorella vulgaris was significantly higher than that of Anabaena sp. under three of the four light cycles, including 6-day continuous light, 48-h light and 24-h dark, and 24-h light and 48-h dark.
The highest total nitrogen removal from minkery wastewater in this study was achieved through the cultivation of Chlorella vulgaris under light cycle of 6-day continuous light. This particular treatment achieved a nitrogen removal of 96.2% over the 6-day growth period, and the Tukey’s test (with a confidence coefficient of 95%) determined that there were no significant differences between this and the total nitrogen removal from minkery wastewater by cultivating Chlorella vulgaris under the light cycle of 48-h light and 24-h dark. This leads to the conclusion that Chlorella vulgaris showed the highest total nitrogen removal from minkery wastewater under the light cycle of 6-day continuous light and 48-h light and 24-h dark, and using light cycle of 48-h light and 24-h dark instead of 6-day continuous light reduced energy costs.
In comparison, Li et al. [27] reported that mixotrophic microalgae Chlorella kessleri and Chlorella protothecoide cultivated in highly concentrated municipal wastewater achieved 62.2% and 64.5% total nitrogen removal in only 4 days, respectively. Wang et al. [25] reported that microalgae Chlorella sp. cultivated in municipal wastewater from sludge centrifuge achieved 82.8% total nitrogen removal in 9 days. It should be noted that minkery wastewater used in our study had a higher initial nitrogen concentration than those of municipal wastewaters used in those studies, which makes these numbers even more impressive.
Figure 27.2 provided the ammonium concentrations of minkery wastewater before and after 6-day treatment by Chlorella vulgaris under four light cycles employed. It demonstrates that the minkery wastewater after 6-day treatment by Chlorella vulgaris under light cycles of 6-day continuous light and 48-h light and 24-h dark had a lower ammonium concentration than both surface water and groundwater concentration limits. Ammonium concentration limits of the surface water and groundwater for mink farms are 1 mg/L and 4 mg/L, respectively.
The means of total phosphorus removal from various mediums by culturing Chlorella vulgaris and Anabaena sp. under different light cycles are shown in Table 27.6.
In comparing the total phosphorus removal from minkery wastewater between Chlorella vulgaris and Anabaena sp. across each of the light cycles, it was observed that the total phosphorus removal from minkery wastewater through cultivation of Chlorella vulgaris was significantly higher than that of Anabaena sp. under all four light cycles.
The highest total phosphorus removal from minkery wastewater was performed by cultivating Chlorella vulgaris under 6-day continuous light, and the Tukey’s test (with a confidence coefficient of 95%) did not determine that there was a statistically significant difference between this and the total phosphorus removal from minkery wastewater via culturing Chlorella vulgaris under 48-h light and 24-h dark. This leads to the conclusion that Chlorella vulgaris showed the highest total phosphorus removal from minkery wastewater under the light cycles of 6-day continuous light and 48-h light and 24-h dark, and using light cycle of 48-h light and 24-h dark instead of 6-day continuous light reduced energy costs of the cultivation. These two particular treatments achieved phosphorus removals of 29.7% and 28.8% over the 6-day growth period, respectively.
In comparison with other studies, the phosphorus removal rates observed in this study were relatively low. Li et al. [27] observed that mixotrophic microalgae Chlorella kessleri and Chlorella protothecoide cultivated in highly concentrated municipal wastewater achieved 87.4% and 86.1% total phosphorus removal in only 4 days. Wang et al. [25] reported that microalga Chlorella sp. cultivated in municipal wastewater from sludge centrifuge achieved 85.6% total phosphorus removal in 9 days. It should be noted that minkery wastewater used in our study had a much higher initial phosphorus concentration than those of the municipal wastewaters used in those studies. When it comes to the amount of phosphorus removal (mg/L), the findings were much closer.
The total phosphorus concentrations of minkery wastewater before and after 6-day treatment by Chlorella vulgaris under different light cycles are provided in Fig. 27.3. And total phosphorus concentration limits of the surface water and groundwater for mink farms are 20 μg/L and 0.1 mg/L, respectively. Unfortunately, even after 6-day treatment by Chlorella vulgaris, the total phosphorus concentration of minkery wastewater was still much higher than the required concentration limits for the surface water and groundwater, which means that further treatment would be required to reduce the concentration levels of total phosphorus in minkery wastewater.
4 Conclusions
According to the experimental data, the minkery wastewater was proved to be a superior medium than modified Bold’s basal medium for Chlorella vulgaris cultivation under most of the light cycles, and the growth characteristics of Anabaena sp. in BG-110 were significantly better than those in minkery wastewater under most of the light cycles. Based on the findings, this study proved the potential of using minkery wastewater as an alternative growth medium, even though the cultivation of Anabaena sp. in minkery wastewater remains a challenge due to the insufficient nitrate levels of minkery wastewater. Furthermore, this study also demonstrated the wastewater treatment potential of Chlorella vulgaris in controlled environment minkery wastewater. It is concluded that the best treatment potential for minkery wastewater was achieved using Chlorella vulgaris under the light cycle of 48-h light and 24-h dark. This particular treatment yielded the highest biomass increase, the highest total nitrogen removal, and the highest total phosphorus removal along with a reduced demand in light energy.
- D:
-
Dark
- DW:
-
Distilled water
- L:
-
Light
- MW:
-
Minkery wastewater
- TM:
-
Traditional growth medium
- x :
-
Algae biomass measurement
- y :
-
Nutrient content measurement
- 0:
-
Day 0
- 6:
-
Day 6
References
Statistics Canada (2011) Fur Statistics 2010, Catalogue no. 23-013-X, vol. 8, ISSN 1705-4273, Ottawa, pp. 1–20
Boon J (2014) Poop problems: mink stink leads to formal complaints, The Coast: Halifax’s Website. https://www.thecoast.ca/RealityBites/archives/2014/11/13/poop-problemsLast accessed 10 Oct 2017
Taylor DA (2009) A water quality survey of nine lakes in the Carleton river watershed area Yarmouth county, Nova Scotia. Nova Scotia Environment, pp.1–48, https://novascotia.ca/nse/surface.water/docs/Yarmouth.Area.Lakes.Report.with.Appendices.pdf Last accessed 10 Oct 2017
Ferguson JL (2001) Characterizing the process of composting mink manure and pelted mink carcasses. Master’s Thesis, Nova Scotia. Agricultural College. Truro, NS, Canada
Fur Industry Act. (2010). Fur Industry Act, Chapter 4 of the Acts of 2010 as amended by 2012, c. 58: An act respecting the fur industry. House of Assembly, Halifax, Nova Scotia, pp. 1–18, https://nslegislature.ca/sites/default/files/legc/statutes/fur%20industry.pdf Last accessed 10 Oct 2017
Rai L, Gaur J, Kumar H (1981) Phycology and heavy metal pollution. Biol Rev 56(2):99–151
Redalje DG, Duerr EO, de la Noiie J, Mayzaud P, Nonomura AM, Cassin R (1989) Algae as ideal waste removers: biochemical. In: Biotreatment of agricultural wastewater. CRC press, Boca Raton, p 91
McGriff EC, McKenney RE (1971) Activated algae: a nutrient process. Water Sewage Works 118:377
McGriff EC, McKinney RC (1972) The removal of nutrients and organics by activated algae. Water Res 6(10):1155
Tam NFY, Wong YS (1989) Wastewater nutrient removal by Chlorella pyrenoidosa and Scenedesmus sp. Environ Pollut 58:19–34
Campbell PK, Beer T, Batten D (2011) Life cycle assessment of biodiesel production from microalgae in ponds. Bioresour Technol 102:50–56
Lee YK (2001) Microalgal mass culture systems and methods: their limitation and potential. J Appl Phycol 13:307–315
Lardon L, Helias A, Sialve B, Steyer JP, Bernard O (2009) Life-cycle assessment of biodiesel production from microalgae. Environ Sci Technol 43:6475–6481
Pate R, Klise G, Wu B (2011) Resource demand implications for US algae biofuels production scale-up. Appl Energy 88:3377–3388
Shi J, Podola B, Melkonian M (2007) Removal of nitrogen and phosphorus from wastewater using microalgae immobilized on twin layers: an experimental study. J Appl Phycol 19:417–423
Hammouda O, Gaber A, Abdelraouf N (1995) Microalgae and wastewater treatment. Ecotoxicol Environ Saf 31:205–210
Lowrey J, Yildiz I (2011) Seawater/wastewater production of microalgae-based biofuels in closed loop tubular photobioreactors. Masters Theses Proj. Reports
Yildiz I, Nguyen-Quang T, Mehlitz T, Brooker B (2013) Algae, Biofuels, and Modeling. In: Dincer, I, Colpan, CO, and Kadioglu, F (Eds), Causes, Impacts, and Solutions to Global Warming. Springer, New York, NY, 30:525–607
American Public Health Association (ed) (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington, DC
Griffiths MJ, Garcin C, van Hille RP, Harrison ST (2011) Interference by pigment in the estimation of microalgal biomass concentration by optical density. J Microbiol Methods 85(2):119–123
Yoon J, Sim S, Kim M, Park T (2002) High cell density culture of Anabaena variabilis using repeated injections of carbon dioxide for the production of hydrogen. Int J Hydrog Energy 27:1265–1270
Hempel N, Petrick I, Behrendt F (2012) Biomass productivity and productivity of fatty acids and amino acids of microalgae strains as key characteristics of suitability for biodiesel production. J Appl Phycol 24:1407–1418
Šoštarič M, Golob J, Bricelj M, Klinar D, Pivec A (2009) Studies on the growth of Chlorella vulgaris in culture media with different carbon sources. Chem Biochem Eng Q 23(4):471–477
Wang L, Min M, Li Y, Chen P, Chen Y, Liu Y, Wang Y, Ruan R (2009) Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl Biochem Biotechnol 162:1174–1186
Oliveira P, Martins NM, Santos M, Couto NAS, Wright PC, Tamagnini P (2015) The Anabaena sp. PCC 7120 exoproteome: taking a peek outside the box. Life 5:130–163
Weare NM, Benemann JR (1972) Nitrogen fixation by Anabaena cylindrica: localization of nitrogen fixation in the heterocysts. Arch Mikrobiol 90:323–332
Li Y, Zhou W, Hu B, Min M, Chen P, Ruan RR (2012) Effect of light intensity on algal biomass accumulation and biodiesel production for mixotrophic strains Chlorella kessleri and Chlorella protothecoide cultivated in highly concentrated municipal wastewater. Biotechnol Bioeng 109:2222–2229
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Ji, Y., Yildiz, I. (2018). Investigation of Cultivation and Wastewater Treatment Potential of Microalgae and Cyanobacteria in Controlled Environment Minkery Wastewater. In: Nižetić, S., Papadopoulos, A. (eds) The Role of Exergy in Energy and the Environment. Green Energy and Technology. Springer, Cham. https://doi.org/10.1007/978-3-319-89845-2_27
Download citation
DOI: https://doi.org/10.1007/978-3-319-89845-2_27
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-89844-5
Online ISBN: 978-3-319-89845-2
eBook Packages: EnergyEnergy (R0)