Abstract
Today, a variety of surgical procedures can be used to successfully treat gingival recession defects. Among them, the autogenous connective tissue graft in conjunction with a coronally advanced flap is commonly considered the gold standard procedure. However, the most significant disadvantages of this procedure are the potential morbidity associated with autogenous tissue harvesting and the limited availability of donor tissue. For these reasons, alternative surgical procedures using membranes, enamel matrix derivative, and soft tissue graft substitutes have been proposed and tested. The aim of the present chapter is to provide an overview on the use of soft tissue substitutes as a possible alternative to connective tissue grafts in the surgical management of gingival recession defects.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Coronally Advanced Flap (CAF)
- Enamel Matrix Derivative (EMD)
- Connective Tissue Graft (CTG)
- Gingival Recession Defects
- Root Coverage
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
9.1 Introduction
Various surgical techniques have been proposed in the past few decades to achieve successful and predictable coverage of gingival recession defects [1, 2]. Although all these surgical therapeutic approaches provide significant reduction in recession depth, connective tissue graft (CTG) procedures have shown to offer the best outcomes for root coverage and gain of keratinized tissue [2]. The clinical efficacy of the CTGs has been mainly attributed to the double blood supply at the recipient site and thus enhanced graft revascularization and wound healing [3,4,5]. More recently, it has also been suggested that the CTG may act as a biological filler with the ability to reduce soft tissue contraction in the healing phase [5, 6]. Therefore, the CTG procedures are rightly considered to be the gold standard and reference therapy for root coverage. However, the CTG is also associated with a number of disadvantages, including a second surgical procedure to harvest the graft, potential donor site morbidity, increased operative time, and limited availability of donor tissue. Moreover, it has been reported that healing following a CTG procedure is mainly characterized by a long junctional epithelium and connective tissue attachment with only limited capacity for periodontal regeneration [7,8,9]. To avoid the drawbacks mentioned above, several alternative surgical procedures have been advocated. Among these procedures, the coronally advanced flap (CAF) alone is considered easy to perform and effective in obtaining root coverage without the need for a second surgical site [10]. On the other hand, the CAF alone has been reported to be associated with an apical relapse of the gingival margin in the long term [11]. This observation has been attributed to the inadequate thickness and amount of keratinized tissue obtained with the CAF alone [11, 12]. Other approaches aimed at enhancing the outcome of the CAF procedure and substituting the CTG include the use of barrier membranes, enamel matrix derivatives, and soft tissue graft substitutes (acellular dermal matrices and xenogeneic collagen matrices).
9.1.1 Barrier Membranes (Guided Tissue Regeneration)
Guided tissue regeneration (GTR) with the use of resorbable and nonresorbable barrier membranes was proposed as an alternative approach in the treatment of gingival recession defects [13]. The rationale behind this technique was to avoid a second surgical site and to promote regeneration of periodontal tissues on the previously exposed root surface [14,15,16]. Indeed, from a histological point of view, the use of barrier membranes in conjunction with a CAF procedure has proven to promote periodontal regeneration with the formation of new cementum, periodontal ligament, and alveolar bone [17, 18]. Accordingly, clinical studies demonstrated good predictability of the GTR technique in terms of root coverage and gain of clinical attachment [15, 19, 20]. This technique was reported to produce a mean root coverage of 75% and complete coverage of the recession defects in 42% of the cases [13]. However, more recent evidence shows that GTR-based root coverage appears ineffective in improving clinical outcomes of the CAF in terms of complete root coverage and recession reduction [5]. Less favorable clinical outcomes were also reported when GTR-based root coverage was compared to the CTG [5, 21]. Moreover, the use of GTR for root coverage has been associated with several complications and drawbacks. Thus, membrane exposure was reported as a common complication that may result in site contamination, infection, and failure of the procedure [22, 23]. Furthermore, when nonresorbable membranes are used, a second surgical procedure is required for membrane retrieval, causing an additional trauma to the regenerating tissue [19, 23]. This may also account for the observation that the use of nonresorbable membranes has been associated with a lower percentage of complete root coverage when compared with resorbable membranes [13]. Another shortcoming of the GTR-based root coverage technique is that it is not suitable for the management of multiple recession defects at the same time. It has also been demonstrated that the use of barrier membranes for root coverage has only limited ability to increase gingival tissue thickness [16]. The various disadvantages may also explain the limited clinical benefits of this technique when compared to other root coverage procedures.
Taken together, GTR-based root coverage has various limitations, and so its routine use cannot be recommended at present.
9.1.2 Enamel Matrix Derivative
The use of enamel matrix derivative (EMD) has been proposed as another approach to substitute the connective tissue graft in root coverage procedures and to promote periodontal regeneration on the previously exposed root surface. Indeed, histology from a human biopsy study showed enhanced formation of new cementum, periodontal ligament, and alveolar bone 9 months after application of EMD in conjunction with a CAF [9]. From a clinical standpoint, a recent systematic review concluded that the adjunctive use of EMD significantly improved recession reduction, complete root coverage, and keratinized tissue gain compared to CAF alone [5]. Hence, the additional use of EMD to CAF appears as a safe approach superior to the use of CAF alone [5, 21]. Studies have also demonstrated that EMD provides clinical benefits comparable to connective tissue grafting, with stable clinical results in the long term [24, 25]. A recent systematic review concluded that the use of EMD in conjunction with a CAF leads to clinical outcomes close to those reported for CTGs and thus may be considered as a viable alternative to autogenous donor tissue [21]. The main advantages of using EMD combined with CAF are the simplicity of the procedure and avoidance of a second surgical site. Thus, the combination of EMD + CAF has been associated with improved early healing and less postoperative discomfort compared to CTG + CAF [24]. On the other hand, current literature suggests that the combination of EMD + CAF is not as effective as CTG + CAF in augmenting the width of keratinized tissue [26, 27]. More recently, Rebele et al. [28] compared the use of EMD combined with CAF to a CTG with the tunnel technique for the treatment of Miller class I and II recession defects. The results from that study demonstrated that the CTG procedure was able to create a significantly thicker marginal tissue than EMD. Thus, the use of EMD appears to be less effective than the CTG in increasing the width and thickness of keratinized tissue. A possible added value of combining EMD with the CTG for the treatment of Miller class I and II recession defects was evaluated more recently by Roman et al. [29]. At 1 year after surgery, the combination of EMD and CTG + CAF had no beneficial effect on root coverage compared with the CTG alone. Conversely, Henriques et al. [30] demonstrated significantly better clinical outcomes using EMD + CTG compared to CTG alone in the treatment of Miller class III recession defects. However, at present there is not enough evidence to support the combined use of EMD and CTG in recession coverage procedures.
Based on the current evidence, EMD + CAF has the ability to promote periodontal regeneration and improve clinical outcomes comparable to CTG procedures and thus may be considered a safe substitute for autogenous grafts in recession coverage procedures.
9.1.3 Soft Tissue Graft Substitutes (Acellular Dermal Matrices and Xenogeneic Collagen Matrices)
The use of acellular dermal matrix (ADM) grafts in periodontal plastic surgery has been proposed as a substitute for palatal donor tissue in order to eliminate the disadvantages associated with autogenous grafts. This ADM allograft is obtained from donated human skin and further treated to remove all viable cells that could cause an inflammatory or immunogenic response. The remaining connective tissue matrix provides a collagen structure that functions as a scaffold to allow ingrowth and subsequent replacement by host tissues. Advantages of using ADM grafts in root coverage procedures include the avoidance of palatal tissue harvesting, reduced morbidity compared to autogenous grafts, unlimited tissue availability, reduction in surgery time, and increased patient treatment acceptance [31]. A number of commercial products are available and include AlloDerm® (BioHorizons), Puros® Dermis (Zimmer Biomet), PerioDerm™ (Dentsply), and Epiflex® (DIZG).
From a histological point of view, there is only limited information available regarding the type of healing following recession coverage with ADM. Cummings et al. [8] reported that root coverage utilizing ADM resulted in a healing characterized by the formation of a long junctional epithelium and connective tissue adhesion. A human case report by Richardson and Maynard [32] evaluated histologically the type of attachment after an ADM augmentation procedure and observed a fibrous tissue apposition with no attachment to the root surface. When tested clinically, several studies demonstrated that the use of ADM in conjunction with CAF significantly improved root coverage outcomes compared to CAF alone [33,34,35,36]. Moreover, a number of studies showed that the combination of ADM + CAF can achieve clinical results comparable to those obtained with CTG procedures [37,38,39,40]. A recent meta-analysis by Guan et al. [41] reported no significant differences between ADM and CTG in terms of recession coverage, gain in clinical attachment, and amount of keratinized tissue (KT). Similarly, a systematic review conducted by Chambrone and Tatakis [21] concluded that there is strong evidence to support the use of ADM + CAF as an alternative to autogenous donor tissue in root coverage procedures. Furthermore, the use of ADM was found to enhance marginal tissue thickness similar to CTG grafts [39]. Conversely, a recent systematic review evaluating the efficacy of root coverage procedures showed that ADM + CAF provides inferior outcomes compared to CTG + CAF and no additional benefit over CAF alone [5]. These inconsistent clinical outcomes can be explained, at least in part, by the technique sensitivity and healing characteristics of ADM. Indeed, while ADM has some benefits for the clinician and the patient, its use in root coverage procedures is less forgiving than autogenous material. Since ADM is an avascular and acellular material, it depends on the revascularization and nutrition coming from the recipient site. Therefore, it is mandatory to ensure full coverage of the ADM by the overlying flap in a tension-free manner, especially in areas with high muscle activity. Consequently, shrinkage of the flap with exposure of ADM during the healing phase may compromise revascularization of the graft material and lead to its disintegration [42, 43]. Moreover, it has been demonstrated that ADM is associated with a prolonged healing period when compared to the CTG [39]. This observation was attributed to the fact that ADM is a non-vital material requiring an additional period of time to be resorbed and substituted by the host tissue. Because of its specific healing properties, the overall effectiveness and predictability of ADM are closely related to the surgical technique employed. As yet, various surgical techniques have been proposed for the use of ADM in root coverage procedures [43,44,45]. In this context, Barros et al. [44] proposed an extended flap with the releasing incisions displaced to the adjacent teeth and demonstrated that the combination of ADM with this technique improved recession coverage in comparison with the use of ADM with the conventional technique described by Langer and Langer [3]. The extended flap approach showed also superior clinical results compared to a flap approach without vertical releasing incisions when using ADM [43]. More recently, Ayub et al. [46] introduced a modification of the extended flap technique in which the ADM graft is positioned 1 mm apical to the CEJ and the flap 1 mm coronal to the CEJ, with the intention to prevent ADM exposure and to compensate primary soft tissue shrinkage. The authors demonstrated with the proposed technique improved clinical outcomes when compared to the conventional extended flap technique. Ozenci et al. [45] compared root coverage outcomes of ADM in conjunction with the tunnel technique or a CAF. Although both techniques were effective in obtaining root coverage, the CAF technique resulted in significantly improved clinical outcomes when compared with the tunnel technique. Hence, as yet there is no consensus on the most appropriate surgical technique associated with the use of ADM. However, the use of a flap technique with vertical releasing incisions provides a good control over the procedure owing to better visibility and easier flap repositioning in comparison with more technique-sensitive surgical approaches, e.g., the tunnel technique [47]. In general, the selection of a surgical technique for the use of ADM should be based on the goal of preserving the vascular supply of the flap, in order to obtain proper nutrition and revascularization of the graft material.
Another common observation associated with the use of ADM in root coverage procedures is the trend toward less keratinized tissue formation when compared to autogenous grafts [39, 42, 48]. Although the exact mechanism by which ADM increases the amount of keratinized tissue is still unknown, it is commonly thought that the non-vital ADM itself has only little influence on the cytodifferentiation of the covering epithelium and that the type of epithelium that covers the ADM seems to be determined by the surrounding tissues [49]. In this context, Shin et al. [50] demonstrated that the additional use of EMD with ADM in root coverage procedures resulted in a significant increase of keratinized tissue when compared to ADM alone.
A further important factor to be considered is the long-term stability of clinical outcomes obtained with ADM. Thus, Harris [51] reported that the root coverage obtained with ADM tended to break down in the long term, whereas sites treated with CTGs remained stable. In contrast, Moslemi et al. [40] observed in a 5-year follow-up study a significant relapse of root coverage outcomes in ADM- and CTG-treated sites, with no statistically significant difference between the two procedures.
In sum, the use of ADM in root coverage procedures can be considered a safe and patient-friendly alternative to autogenous donor tissue.
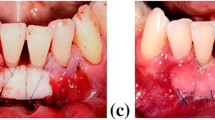
Recently, xenogeneic collagen matrix (CM) materials have been introduced as an alternative to ADM and autogenous tissue in root coverage procedures (Figs. 9.1, 9.2, 9.3, and 9.4). These collagen matrices are derived from porcine tissue and further processed to remove antigenic cellular components, while preserving the structure of the source tissue. Porcine dermal tissue as a source of CM appears favorable because it is structurally and immunologically similar to its human counterpart [52, 53]. Moreover, the use of porcine-derived CM might alleviate some of the shortcomings associated with ADM derived from human cadavers. Indeed, ethical concerns and the possible risk of disease transmission may have limited the more widespread use of human ADM in root coverage procedures [54]. When used in a clinical setting, the porcine CM is intended to act as a temporary 3D scaffold to support host cell infiltration and tissue ingrowth without eliciting a foreign body or immunogenic response [47, 55, 56]. Examples of commercially available porcine-derived CMs include Mucoderm® (Botiss biomaterials), Mucograft® (Geistlich), Osteobiol® Derma (Tecnoss), MucoMatrixX® (Dentegris), and DynaMatrix® (Keystone).
(a) Multiple gingival recessions affecting the maxillary right quadrant. (b) Flap elevation using a split-full-split approach without vertical releasing incisions. (c) CM sutured in place. (d) Flap coronally advanced and sutured to cover the entire CM. (e) Clinical outcome 3 months after surgery. (f) Clinical outcome 18 months after surgery
(a) Preoperative gingival recession on a mandibular left lateral incisor. (b) Tunnel flap preparation and adjustment of CM dimensions. (c) Application of EMD on the root surface. (d) Placement of the CM into the prepared tunnel. (e) Flap coronally advanced and sutured. (f) Clinical outcome 3 months after surgery
(a) Multiple gingival recessions affecting the maxillary left anterior area. (b) Flap elevation using a split-full-split approach with a short vertical incision distal to the canine. (c) Application of EMD on the root surface. (d) CM sutured in place. (e) Flap coronally advanced and sutured to cover the entire CM. (f) Clinical outcome 3 months after surgery. (g) Clinical outcome 9 months after surgery. (h) Clinical outcome 2 years after surgery (Reproduced from Kasaj A, Quintessence Int 2016;47:775–783, courtesy Quintessence Publishing)
(a) Gingival recession affecting the maxillary right canine. (b) Flap elevation using a split-full-split approach and deepithelialization of the anatomical papillae. (c) Application of EMD on the root surface. (d) CM sutured in place. (e) Flap coronally advanced and sutured to cover the entire CM. (f) Clinical outcome 6 months after surgery. (g) Clinical outcome 2 years after surgery
With respect to human histologic outcomes of recession defects treated with CM, Camelo et al. [57] showed the formation of a long junctional epithelium and connective tissue adhesion without evidence of periodontal regeneration. Clinical studies demonstrated that the use of CM in the treatment of recession defects significantly improved clinical outcomes in terms of root coverage, gingival thickness, and gain of KT when compared to CAF alone [58,59,60]. These findings were further confirmed in recent systematic reviews [5, 61]. Conversely, Moreira et al. [62] found in a recent clinical study with a 6-month follow-up that CM in conjunction with CAF was not able to improve recession reduction compared to CAF alone. Similarly, Jepsen et al. [63] evaluated CM + CAF versus CAF alone and found at 6 months no significant difference between the groups in terms of root coverage. However, the addition of CM significantly increased the width of KT and gingival thickness compared to CAF alone. Moreover, the authors demonstrated that CM was able to improve root coverage of CAF alone in large recession defects (≥ 3 mm). When CM was compared with CTG, one study reported similar clinical outcomes for both procedures [64], whereas other studies found outcomes to be inferior for CM [65, 66]. Moreover, some studies demonstrated less than 50% of complete root coverage with the use of CM, despite satisfactory results achieved for mean root coverage [66, 67]. In the most recent systematic review on this topic, Atieh et al. [61] showed that the CTG in conjunction with CAF was more effective than CM + CAF in terms of root coverage and recession reduction. On the other hand, the use of CM was associated with a shorter surgery time and reduced postoperative morbidity when compared with CTG. With respect to patient-reported aesthetic satisfaction and gain of KT, no significant difference was found between the two procedures. McGuire and Scheyer [68] reported 5-year clinical outcomes of patients treated either with CM + CAF or CTG + CAF. The results demonstrated a mean root coverage of 78% for CM + CAF compared with 95.5% for CTG + CAF. Despite these differences, the authors concluded that CM provides a viable and long-term alternative for the CTG, when balanced with patient-reported outcomes for aesthetics and compared with historical root coverage results reported by other investigators. Likewise, Chambrone and Tatakis [21] reported in their systematic review that the use of CM in conjunction with CAF provides clinical outcomes close to those of CTGs (difference in mean root coverage 8.9%) and thus may be considered a viable alternative to autogenous donor tissue. A possible approach to further improve the clinical outcomes of CM is the combination with EMD. However, a recent study failed to demonstrate enhanced clinical outcomes of CM + EMD + CAF compared to CM + CAF [60].
Taken together, the currently available evidence suggests that CM may be used as a safe and adequate substitute for autogenous grafts in patients with a limited amount of donor tissue or patients who want to avoid the palatal donor site surgery.
Conclusions
Various biomaterials have been proposed as a substitute for autogenous grafts in root coverage procedures. Current evidence suggests that enamel matrix derivative, acellular dermal matrix grafts, and xenogeneic collagen matrices combined with coronally advanced flaps can be considered as safe and effective treatment procedures for obtaining aesthetic root coverage. Moreover, the use of these soft tissue substitutes as an alternative to autogenous donor tissue offers certain advantages such as increased surgical efficiency and reduced patient morbidity. This approach also appears to be particularly useful in patients with limited donor tissue availability or patients who wish to avoid a second site surgery. On the other hand, soft tissue substitutes are still associated with inferior clinical results when compared with those of the connective tissue graft procedures. There is also only limited data available focusing on long-term outcomes following treatment with these biomaterials. The clinician’s decision to use soft tissue substitutes as an alternative to autogenous donor tissue should be based upon consideration of the clinical situation, availability of palatal donor tissue, and patient preferences.
References
Richardson CR, Allen EP, Chambrone L, Langer B, McGuire MK, Zabalegui I, Zadeh HH, Tatakis DN. Periodontal soft tissue root coverage procedures: practical applications from the AAP Regeneration Workshop. Clin Adv Periodontics. 2015;5:2–10.
Tatakis DN, Chambrone L, Allen EP, Langer B, McGuire MK, Richardson CR, Zabalegui I, Zadeh HH. Periodontal soft tissue root coverage procedures: a consensus report from the AAP Regeneration Workshop. J Periodontol. 2015;86:S52–5.
Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol. 1985;56:715–20.
Guiha R, el Khodeiry S, Mota L, Caffesse R. Histological evaluation of healing and revascularization of the subepithelial connective tissue graft. J Periodontol. 2001;72:470–8.
Cairo F, Nieri M, Pagliaro U. Efficacy of periodontal plastic surgery procedures in the treatment of localized facial gingival recessions. A systematic review. J Clin Periodontol. 2014;41:S44–62.
Cortellini P, Tonetti M, Baldi C, Francetti L, Rasperini G, Rotundo R, Nieri M, Franceschi D, Labriola A, Prato GP. Does placement of a connective tissue graft improve the outcomes of coronally advanced flap for coverage of single gingival recessions in upper anterior teeth? A multi-centre, randomized, double-blind, clinical trial. J Clin Periodontol. 2009;36:68–79.
Harris RJ. Human histologic evaluation of root coverage obtained with a connective tissue with partial thickness double pedicle graft. A case report. J Periodontol. 1999;70:813–21.
Cummings LC, Kaldahl WB, Allen EP. Histologic evaluation of autogenous tissue and acellular dermal matrix grafts in humans. J Periodontol. 2005;76:178–86.
McGuire MK, Scheyer ET, Schupbach P. A prospective, case-controlled study evaluating the use of enamel matrix derivative on human buccal recession defects: a human histologic examination. J Periodontol. 2016;87:645–53.
Zucchelli G, Mounssif I, Mazzotti C, Stefanini M, Marzadori M, Petracci E, Montebugnoli L. Coronally advanced flap with and without connective tissue graft for the treatment of multiple gingival recessions: a comparative short- and long-term controlled randomized clinical trial. J Clin Periodontol. 2014;41:396–403.
Pini-Prato GP, Cairo F, Nieri M, Franceschi D, Rotundo R, Cortellini P. Coronally advanced flap versus connective tissue graft in the treatment of multiple gingival recessions: a split-mouth study with a 5-year follow-up. J Clin Periodontol. 2010;37:644–50.
Cairo F, Pagliaro U, Nieri M. Treatment of gingival recession with coronally advanced flap procedures: a systematic review. J Clin Periodontol. 2008;35:136–62.
Al-Hamdan K, Eber R, Sarment D, Kowalski C, Wang HL. Guided tissue regeneration-based root coverage: meta-analysis. J Periodontol. 2003;74:1520–33.
Tinti C, Vincenzi G, Cortellini P, Pini Prato G, Clauser C. Guided tissue regeneration in the treatment of human facial recession. A 12-case report. J Periodontol. 1992;63:554–60.
Pini Prato G, Clauser C, Cortellini P, Tinti C, Vincenzi G, Pagliaro U. Guided tissue regeneration versus mucogingival surgery in the treatment of human buccal recessions. A 4-year follow-up study. J Periodontol. 1996;67:1216–23.
Paolantonio M. Treatment of gingival recessions by combined periodontal regenerative technique, guided tissue regeneration, and subpedicle connective tissue graft. A comparative clinical study. J Periodontol. 2002;73:53–62.
Cortellini P, Clauser C, Prato GP. Histologic assessment of new attachment following the treatment of a human buccal recession by means of a guided tissue regeneration procedure. J Periodontol. 1993;64:387–91.
Parma-Benfenati S, Tinti C. Histologic evaluation of new attachment utilizing a titanium-reinforced barrier membrane in a mucogingival recession defect. A case report. J Periodontol. 1998;69:834–9.
Borghetti A, Glise JM, Monnet-Corti V, Dejou J. Comparative clinical study of a bioabsorbable membrane and subepithelial connective tissue graft in the treatment of human gingival recession. J Periodontol. 1999;70:123–30.
Tatakis DN, Trombelli L. Gingival recession treatment: guided tissue regeneration with bioabsorbable membrane versus connective tissue graft. J Periodontol. 2000;71:299–307.
Chambrone L, Tatakis DN. Periodontal soft tissue root coverage procedures: a systematic review from the AAP Regeneration Workshop. J Periodontol. 2015;86:S8–51.
Lins LH, de Lima AF, Sallum AW. Root coverage: comparison of coronally positioned flap with and without titanium-reinforced barrier membrane. J Periodontol. 2003;74:168–74.
Wang HL, Modarressi M, Fu JH. Utilizing collagen membranes for guided tissue regeneration-based root coverage. Periodontol 2000. 2012;59:140–57.
McGuire MK, Nunn M. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue. Part 1: comparison of clinical parameters. J Periodontol. 2003;74:1110–25.
McGuire MK, Scheyer ET, Nunn M. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue: comparison of clinical parameters at 10 years. J Periodontol. 2012;83:1353–62.
Koop R, Merheb J, Quirynen M. Periodontal regeneration with enamel matrix derivative in reconstructive periodontal therapy: a systematic review. J Periodontol. 2012;83:707–20.
Cheng GL, Fu E, Tu YK, Shen EC, Chiu HC, Huang RY, Yuh DY, Chiang CY. Root coverage by coronally advanced flap with connective tissue graft and/or enamel matrix derivative: a meta-analysis. J Periodontal Res. 2015;50:220–30.
Rebele SF, Zuhr O, Schneider D, Jung RE, Hürzeler MB. Tunnel technique with connective tissue graft versus coronally advanced flap with enamel matrix derivative for root coverage: a RCT using 3D digital measuring methods. Part II. Volumetric studies on healing dynamics and gingival dimensions. J Clin Periodontol. 2014;41:593–603.
Roman A, Soancă A, Kasaj A, Stratul SI. Subepithelial connective tissue graft with or without enamel matrix derivative for the treatment of Miller class I and II gingival recessions: a controlled randomized clinical trial. J Periodontal Res. 2013;48:563–72.
Henriques PS, Pelegrine AA, Nogueira AA, Borghi MM. Application of subepithelial connective tissue graft with or without enamel matrix derivative for root coverage: a split-mouth randomized study. J Oral Sci. 2010;52:463–71.
Allen EP. AlloDerm: an effective alternative to palatal donor tissue for treatment of gingival recession. Dent Today. 2006;25:48,50–2.
Richardson CR, Maynard JG. Acellular dermal graft: a human histologic case report. Int J Periodontics Restorative Dent. 2002;22:21–9.
Woodyard JG, Greenwell H, Hill M, Drisko C, Iasella JM, Scheetz J. The clinical effect of acellular dermal matrix on gingival thickness and root coverage compared to coronally positioned flap alone. J Periodontol. 2004;75:44–56.
Mahajan A, Dixit J, Verma UP. A patient-centered clinical evaluation of acellular dermal matrix graft in the treatment of gingival recession defects. J Periodontol. 2007;78:2348–55.
de Queiroz Cȏrtes A, Sallum AW, Casati MZ, Nociti FH Jr, Sallum EA. A two-year prospective study of coronally positioned flap with or without acellular dermal matrix graft. J Clin Periodontol. 2006;33:683–9.
Ahmedbeyli C, Ipçi ŞD, Cakar G, Kuru BE, Yilmaz S. Clinical evaluation of coronally advanced flap with or without acellular dermal matrix graft on complete defect coverage for the treatment of multiple gingival recessions with thin tissue biotype. J Clin Periodontol. 2014;41:303–10.
Aichelmann-Reidy ME, Yukna RA, Evans GH, Nasr HF, Mayer ET. Clinical evaluation of acellular allograft dermis for the treatment of human gingival recession. J Periodontol. 2001;72:998–1005.
Novaes AB Jr, Grisi DC, Molina GO, Souza SL, Taba M Jr, Grisi MF. Comparative 6-month clinical study of a subepithelial connective tissue graft and acellular dermal matrix graft for the treatment of gingival recession. J Periodontol. 2001;72:1477–84.
Paolantonio M, Dolci M, Esposito P, D’Archivio D, Lisanti L, Di Luccio A, Perinetti G. Subpedicle acellular dermal matrix graft and autogenous connective tissue graft in the treatment of gingival recessions: a comparative 1-year clinical study. J Periodontol. 2002;73:1299–307.
Moslemi N, Mousavi Jazi M, Haghighati F, Morovati SP, Jamali R. Acellular dermal matrix allograft versus subepithelial connective tissue graft in treatment of gingival recessions: a 5-year randomized clinical study. J Clin Periodontol. 2011;38:1122–9.
Guan W, Liao H, Guo L, Wang C, Cao Z. Root coverage using a coronally advanced flap with or without acellular dermal matrix: a meta-analysis. J Periodontal Implant Sci. 2016;46:22–34.
Tal H, Moses O, Zohar R, Meir H, Nemcovsky C. Root coverage of advanced gingival recession: a comparative study between acellular dermal matrix allograft and subepithelial connective tissue grafts. J Periodontol. 2002;73:1405–11.
Felipe ME, Andrade PF, Grisi MF, Souza SL, Taba M, Palioto DB, Novaes AB. Comparison of two surgical procedures for use of the acellular dermal matrix graft in the treatment of gingival recessions: a randomized controlled clinical study. J Periodontol. 2007;78:1209–17.
Barros RR, Novaes AB, Grisi MF, Souza SL, Taba MJ, Palioto DB. A 6-month comparative clinical study of a conventional and a new surgical approach for root coverage with acellular dermal matrix. J Periodontol. 2004;75:1350–6.
Ozenci I, Ipci SD, Cakar G, Yilmaz S. Tunnel technique versus coronally advanced flap with acellular dermal matrix graft in the treatment of multiple gingival recessions. J Clin Periodontol. 2015;42:1135–42.
Ayub LG, Ramos UD, Reino DM, Grisi MF, Taba M Jr, Souza SL, Palioto DB, Novaes AB Jr. A randomized comparative clinical study of two surgical procedures to improve root coverage with the acellular dermal matrix graft. J Clin Periodontol. 2012;39:871–8.
Kasaj A. Gingival recession coverage: Do we still need autogenous grafts? Quintessence Int. 2016;47:775–83.
Harris RJ. A comparative study of root coverage obtained with an acellular dermal matrix versus a connective tissue graft: results of 107 recession defects in 50 consecutively treated patients. Int J Periodontics Restorative Dent. 2000;20:51–9.
Scarano A, Barros RR, Iezzi G, Piattelli A, Novaes AB Jr. Acellular dermal matrix graft for gingival augmentation: a preliminary clinical, histologic, and ultrastructural evaluation. J Periodontol. 2009;80:253–9.
Shin SH, Cueva MA, Kerns DG, Hallmon WW, Rivera-Hidalgo F, Nunn ME. A comparative study of root coverage using acellular dermal matrix with and without enamel matrix derivative. J Periodontol. 2007;78:411–21.
Harris RJ. A short-term and long-term comparison of root coverage with an acellular dermal matrix and a subepithelial graft. J Periodontol. 2004;75:734–43.
Lavker RM, Dong G, Zheng PS, Murphy GF. Hairless micropig skin. A novel model for studies of cutaneous biology. Am J Pathol. 1991;138:687–97.
Hoyama E, Schellini SA, Marques ME, Rossa R, Padovani CR. A comparison of human and porcine acellular dermal tissues in the subcutaneous space of a rat model. Orbit. 2005;24:249–55.
Sanz M, Lorenzo R, Aranda JJ, Martin C, Orsini M. Clinical evaluation of a new collagen matrix (Mucograft prototype) to enhance the width of keratinized tissue in patients with fixed prosthetic restorations: a randomized prospective clinical trial. J Clin Periodontol. 2009;36:868–76.
Núñez J, Caffesse R, Vignoletti F, Guerra F, San Roman F, Sanz M. Clinical and histological evaluation of an acellular dermal matrix allograft in combination with the coronally advanced flap in the treatment of Miller class I recession defects: an experimental study in the mini-pig. J Clin Periodontol. 2009;36:523–31.
Rennert RC, Sorkin M, Garg RK, Januszyk M, Gurtner GC. Cellular response to a novel fetal acellular collagen matrix: implications for tissue regeneration. Int J Biomater. 2013;2013:527957.
Camelo M, Nevins M, Nevins ML, Schupbach P, Kim DM. Treatment of gingival recession defects with xenogenic collagen matrix: a histologic report. Int J Periodontics Restorative Dent. 2012;32:167–73.
Cardaropoli D, Tamagnone L, Roffredo A, Gaveglio L. Coronally advanced flap with and without a xenogenic collagen matrix in the treatment of multiple recessions: a randomized controlled clinical study. Int J Periodontics Restorative Dent. 2014;34:s97–102.
Jepsen K, Stefanini M, Sanz M, Zucchelli G, Jepsen S. Log-term stability of root coverage by coronally advanced flap procedures. J Periodontol. 2017;88:626–33.
Sangiorgio JPM, Neves FLDS, Rocha Dos Santos M, França-Grohmann IL, Casarin RCV, Casati MZ, Santamaria MP, Sallum EA. Xenogenous collagen matrix and/or enamel matrix derivative for treatment of localized gingival recessions—a randomized clinical trial. Part I: clinical outcomes. J Periodontol. 2017;88:1309–18.
Atieh MA, Alsabeeha N, Tawse-Smith A, Payne AG. Xenogeneic collagen matrix for periodontal plastic surgery procedures: a systematic review and meta-analysis. J Periodontal Res. 2016;51:438–52.
Moreira ARO, Santamaria MP, Silvério KG, Casati MZ, Nociti Junior FH, Sculean A, Sallum EA. Coronally advanced flap with or without porcine collagen matrix for root coverage: a randomized clinical trial. Clin Oral Investig. 2016;20:2539–49.
Jepsen K, Jepsen S, Zucchelli G, Stefanini M, de Sanctis M, Baldini N, Greven B, Heinz B, Wennström J, Cassel B, Vignoletti F, Sanz M. Treatment of gingival recession defects with a coronally advanced flap and a xenogeneic collagen matrix: a multicenter randomized clinical trial. J Clin Periodontol. 2013;40:82–9.
Cardaropoli D, Tamagnone L, Roffredo A, Gaveglio L. Treatment of gingival recession defects using coronally advanced flap with a porcine collagen matrix compared to coronally advanced flap with connective tissue graft: a randomized controlled clinical trial. J Periodontol. 2012;83:321–8.
McGuire MK, Scheyer ET. Xenogeneic collagen matrix with coronally advanced flap compared to connective tissue with coronally advanced flap for the treatment of dehiscence-type recession defects. J Periodontol. 2010;81:1108–17.
Aroca S, Molnár B, Windisch P, Gera I, Salvi GE, Nikolidakis D, Sculean A. Treatment of multiple adjacent Miller class I and II gingival recessions with a modified coronally advanced tunnel (MCAT) technique and a collagen matrix or palatal connective tissue graft: a randomized, controlled clinical trial. J Clin Periodontol. 2013;40:713–20.
Cieślik-Wegemund M, Wierucka-Mlynarczyk B, Tanasiewicz M, Gilowski L. Tunnel technique with collagen matrix compared with connective tissue graft for treatment of periodontal recession: a randomized clinical trial. J Periodontol. 2016;87:1436–43.
McGuire MK, Scheyer ET. Long-term results comparing xenogeneic collagen matrix and autogenous connective tissue grafts with coronally advanced flaps for treatment of dehiscence-type recession defects. J Periodontol. 2016;87:221–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Kasaj, A. (2018). Recession Coverage Using Soft Tissue Substitutes. In: Kasaj, A. (eds) Gingival Recession Management. Springer, Cham. https://doi.org/10.1007/978-3-319-70719-8_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-70719-8_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-70717-4
Online ISBN: 978-3-319-70719-8
eBook Packages: MedicineMedicine (R0)