Abstract
Migratory flight close to the Earth’s surface (within the so-called flight boundary layer) occurs in some insects, but the vast majority of migrants ascend above this layer and harness the power of the wind for transport. The resulting displacements range from dispersive movements over a few tens of metres to seasonal migrations covering thousands of kilometres. In this chapter, we summarize knowledge of the use of the aerosphere by insects, focusing particularly on longer migrations, in relation to: the height and duration of flight, direction and speed of movement, seasonal and diel patterns, and responses to atmospheric conditions and phenomena. The seasonal mass movements have major ecological consequences in the invaded areas, and these are discussed briefly. We also highlight recent comparisons of insect movement strategies with those of flying vertebrates and mention interactions between these groups in the atmosphere. We conclude with some suggestions for the future development of these topics.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
1 Introduction
The discipline of aeroecology is concerned with the interaction of airborne organisms—principally insects, birds, and bats—with the atmosphere and with each other. The important role played by the atmosphere in insect migration and the effects of atmospheric phenomena on insect flight have long been recognized by entomologists (Johnson 1969; Pedgley 1982; Drake and Farrow 1988; Rainey 1989; Isard and Gage 2001). As insects are rather small animals, their self-propelled flight speeds (airspeeds) are comparatively low, and thus their ‘appetitive’ or foraging flights (e.g. those in search of food, a mate, or an oviposition site) take place very largely within their ‘flight boundary layer’ (FBL)—the zone extending up from the ground where the ambient wind speed is lower than the insect’s air speed (Taylor 1974). Consequently, insects high in the air are very likely to be engaged in migration, while birds or bats flying at altitude may be engaged in either foraging (with insects often the prey items) or migratory movement.
In order to achieve generality across the animal kingdom, many migration biologists, particularly entomologists, have concluded that migration is best recognized behaviourally (e.g. Kennedy 1985; Dingle and Drake 2007; Dingle 2014; Reynolds et al. 2014; Chapman et al. 2015a; Chapman and Drake 2017). Migratory organisms exhibit a syndrome of morphological, physiological and behavioural traits that bring about persistent, rectilinear movements accompanied by some inhibition of responsiveness to appetitive stimuli. This allows the migrant to move away from its ‘home range’, and migration is consequently quite distinct from foraging, commuting, or territorial patrolling (Dingle 2014). The distinctive behavioural processes of migration in individuals lead, of course, to a collective outcome—a significant spatial redistribution of the population—and the migration syndrome is understood to have evolved principally in response to spatio-temporal variation in the availability and/or quality of resources, either seasonally or on some shorter time scale (Drake and Gatehouse 1995; Dingle 2014; Chapman and Drake 2017). Aerial movement provides insects with a potentially highly efficient means of transportation, allowing populations to colonize and exploit new habitats, in some cases over considerable distances because the migrants concerned can remain airborne for long periods of time (see below) while they are carried along by the wind.
When conditions are sufficiently warm (see below), insects can generally be found at altitudes up to ~2 km above ground level (a.g.l.) (Fig. 7.1), and occasionally atmospheric temperatures may allow flight up to 4–5 km. Thus, the region of the atmosphere we are concerned with is the ‘atmospheric boundary layer’ (ABL) and the region of the lower troposphere immediately above it. The ABL is typically convective by day, extending to ~1 km, while at night a shallower ‘nocturnal (stable) boundary layer’ often forms with a ‘residual layer’ above it (Fig. 7.2). Our coverage in this chapter will concentrate on migratory movements above the vegetation canopy and will exclude further discussion of:
-
(a)
Almost all foraging or appetitive flights (except a few that extend to considerable height).
-
(b)
Those migrations, typical of many butterflies, that take place near the ground, within the FBL (although we note some butterflies may employ both within- and above-FBL migratory flight; Stefanescu et al. 2007, 2013).
-
(c)
Movements not employing powered flight, i.e. those undertaken with the aid of silk draglines or relying on minute body size alone (Reynolds et al. 2014).
-
(d)
Very short-range migrations and the ‘ranging’ movements (Dingle 2014) associated with dispersal.
Diurnal variability in intensity and vertical extent of a layer of small insect targets—as shown by reflectivity from a 35-GHz cloud radar on a cloud-free day in Oklahoma, USA. The depth of the insect layer follows the diurnal variation of the convective boundary layer and reaches a maximum in the afternoon when it can extend up to 2 km (From Luke et al. 2008. © American Meteorological Society. Used with permission)
Schematic diagram showing the structure of the ABL during fair weather over flat terrain (Modified from http://lidar.ssec.wisc.edu/papers/akp_thes/node6.htm. With permission from Dr Antti Piironen)
Much of the material presented here derives from observations made with meteorological and special-purpose entomological radars (see Chaps. 9 and 12). As radar observation of insect migration has recently been the subject of both a review (Chapman et al. 2011a) and a monograph (Drake and Reynolds 2012), we will focus on the more recent literature.
2 The Composition and Incidence of Insects in the Air
An important early contribution to what is now termed aeroecology was made in England shortly after World War II by C.G. Johnson and L.R. Taylor who sampled airborne insects quantitatively and at a series of heights with traps attached to towers and tethered balloons (Johnson 1969). Distributions of small day-flying migrants, particularly aphids, airborne in convective conditions were found to display a linear inverse relation of log density with log height, with the slope of the line varying with the degree of atmospheric stability; a discontinuity in the profile near the surface was probably associated with the insects’ FBL (Johnson 1969; Taylor 1974). The gradient of (log) density against (log) height also varied with type of insect: a steep regression line (i.e. one nearly parallel with the height axis) was typical of very small day-flying insects like thrips (Order Thysanoptera) or aphids that are easily carried up by convection. Insects with somewhat stronger flight, e.g. Coccinellid beetles, showed gradients with larger coefficients because a greater proportion of fliers remained at low altitudes. Integration of the density profile over height allowed the estimation of the total number of the particular species in the air (per unit area of the surface). The form of the profile is determined by the processes of insects continually taking off, being circulated around in the atmosphere by convective up- and downdrafts, and eventually settling out again. This work has recently been re-visited (Wainwright et al. 2017); small insect flight in updrafts was found to be unenergetic—individuals rise but at a slower speed than the surrounding air, and their ascent is slowed further if updraft strength increases.
Typical distributions of insect masses estimated by aerial trapping and by X-band entomological radar are illustrated in Fig. 7.3, for a site in southern England at night. The aerial netting samples show a peak in the size distribution at 0.5–1 mg, and in Britain these would comprise mainly aphids, small flies (Diptera), and parasitoid wasps (Hymenoptera particularly Ichneumonoidea and Chalcidoidea) (Chapman et al. 2004). It can be seen (Fig. 7.3) that number concentrations of insects >10 mg are at least two orders of magnitude down from those in the 1-mg range, and for insects of 100 mg or larger, aerial densities may be lower by five orders of magnitude. There is little comparable information from other regions of the world, but our expectation is that the general pattern would be similar.
Average densities (numbers per 106 m3) for a series of insect mass categories estimated with an X-band vertical-looking entomological radar (circles) and from aerial netting (squares). Aerial catches were taken at ~200 m a.g.l. on several July (i.e. mid-summer) nights at Cardington, Bedfordshire, southern England, and the radar data are for a night in July at a nearby site (Harpenden, Hertfordshire). Only targets detected by the radar at its lowest observing height, which was close to the height at which the net was operated, are included (From Drake and Reynolds 2012)
It should be noted that the current X-band entomological radars (Drake and Reynolds 2012; and see Chap. 9) can detect individual insects weighing ~2 mg at their shortest range (~150 m), but in practice they are not efficient at registering targets of this size (mainly due to numerous small targets interfering with each other), and it is only for insects of ~10 mg and heavier that the radar and netting densities approximately coincide (Fig. 7.3). Furthermore, only individuals above about 15 mg are detectable over the whole sensed volume of the (vertical-looking) radar, up to ~1 km a.g.l. The vertical-looking X-band systems (VLRs) have, therefore, been used primarily for studies of relatively large insects (e.g. larger moths, grasshoppers, and locusts). For smaller species, a shorter radar wavelength is required (Drake and Reynolds 2012); meteorological research ‘cloud radars’, for example, during cloudless periods in the warm season, will be detecting almost solely insect targets (Luke et al. 2008; Wood et al. 2009; Chandra et al. 2010; Wainwright et al. 2017; and see Fig. 7.1). In the Wood et al. (2009) study, there was good agreement between the target size range detected by a 35 GHz cloud radar and the most commonly occurring insect masses, as identified from aerial netting.
3 Insect Migration-in-Progress 1: Broad-Scale Phenomena
Because aeroecology is the study of organisms while they are in the atmosphere, it is concerned primarily with the process of migration rather than its outcomes or its broader function as part of the organisms’ life histories and strategies. This section considers those aspects of ‘migration-in-progress’, such as synchronized emigration, altitudinal layering and orientation patterns, that are manifested over large areas—perhaps several hundred kilometres across. More local and transient phenomena are discussed in section 4.
The numbers of a particular species found high in the air are, of course, primarily dependent on the numbers of that species on the ground in potential source areas and the proportion of individuals whose migration syndrome has prepared them for take-off (Chap. 5 in Johnson 1969). Environmental factors such as previous weather in the source areas may be significant, particularly in small fast-breeding insects like aphids. The daytime emigration curves of Aphis fabae, for example, are determined by the rate of moulting into the winged adults, the length of the teneral period from moulting to flight, and the environmental factors inhibiting flight; together these processes result in a temporary decrease in flight-ready individuals during the middle of the day, and the daily take-off pattern is typically bimodal (Johnson 1969, p. 279). More generally, when the migrant is flight mature, various thresholds for flight (particularly light intensity, temperature, and perhaps wind speed) have to be exceeded (Johnson 1969). While the intensity of migration is influenced by weather factors, especially temperature, the timing of migratory flight is often determined by diel cycles in illumination levels (Lewis and Taylor 1964; Johnson 1969). This is particularly evident in dusk and dawn flights, which change seasonally with the timing of sunset/sunrise or can change from day to day with respect to cloud cover (or even a solar eclipse) (Chap. 10 in Drake and Reynolds 2012).
Although there are exceptions, radar observations reveal little insect activity when surface temperatures are below ~10 °C (Chaps. 10 and 15 in Drake and Reynolds 2012), so this apparently represents a temperature threshold for high-flying insect migrants of many taxa. In northern temperate areas, like the UK, it would seem that temperatures are nearly always suboptimal as the (log) number of insects recorded by entomological radar increases linearly with surface temperature and shows no sign of tailing off (Smith et al. 2000; Chapman et al. 2002a). Where temperatures are favourable for take-off and ascent, the diel pattern of insect migratory flight activity revealed by studies in various parts of the world is highly consistent (Reynolds et al. 2008; Chapman et al. 2011a; Drake and Reynolds 2012). A typical sequence, with three fairly distinct emigration periods, viz.: dusk, dawn, and daytime, is shown in Fig. 7.4.
Time/height plot of insect numbers recorded by a vertical-looking entomological radar, in 5-min samples taken every 15 min, from midday on 16 June 2000 to 10:00 h the next day. The color-scale bar refers to the number of resolvable insect targets detected in each of the 15 vertical sampling intervals, during each 5-min sampling period (No data was available for heights below ~180 m above the radar). The figure shows some typical features in the vertical profile, including: the extended daytime flight period ending about 18:00 h, the activity peak at dusk, a post-dusk elevated insect layer (here forming in the evening after about 22:15 h and persisting until dawn), and the activity peak at dawn. Additionally, in this case, a narrow elevated layer formed after dawn at an altitude of about 400 m and this merged into the daytime flight activity which started about 07:30 h. Daytime convection generally disrupts dawn layers but on this occasion layering continued (and, in fact, intensified) at the higher altitude of 600 m, until about 10:00 h. Cdk = civil dusk (21:21 h) and Cdn = civil dawn (02:59 h)
3.1 Dusk Emigration
A mass emigration peak at dusk is one of the most consistently observed temporal features in insect migration studies, being found in virtually all investigations that had the capability to detect it (Chap. 10 in Drake and Reynolds 2012). The dusk emigration often consists of two parts: first, a peak composed of mainly small, crepuscular insects that take off while it is still quite light and secondly a take-off of larger insects, usually a little later when it is becoming dark. Many of the small insects fly for relatively short periods of an hour or so (Riley et al. 1987; Chapman et al. 2004), while the larger species may be deferring their flight to avoid attack by daytime predators. On the displays of X-band scanning entomological radars, the early peak appears as a diffuse echo (of the type produced by multiple small targets within each radar pulse volume) while the later-occurring larger insects produce discrete echoes that can be followed individually over several scans (i.e. for ~10–20 s). After ascent to altitudes of a few hundred metres, migrant insects (both small and large) may maintain flight for several hours or sometimes for the whole night and may become concentrated into narrow altitudinal layers (Reynolds et al. 2005; Chap. 10 in Drake and Reynolds 2012; Rennie 2014; and see below).
Often the two phases of dusk emigration overlap, but on the occasion shown in Fig. 7.4 they appear more distinct—the peak of mainly small insect emigration taking place before civil twilight, while the ascent of larger insects occurred after it. The different faunal composition of these phases is reflected in radar-derived mass estimates: the insects recorded between 20.30 and 22.05 h had a peak in the 5–20-mg category, while those in the layer and recorded between 22.15 h and midnight exhibited a rather broad mass distribution with a median of ~150 mg. There was an invasion of noctuid moths (including Autographa gamma) on southerly winds on this night (Chapman et al. 2008b), and these migrants will have contributed to the layer which persisted until dawn. Generally in temperate areas, many of the large nocturnally flying insects will be noctuid moths, but grasshoppers and locusts form an important component of the night-time aerial fauna in the semi-arid subtropics (Chap. 13 in Drake and Reynolds 2012; Drake and Wang 2013).
The development of the vertical profile of aerial density has been quite well studied for larger insects (see Chap. 10 in Drake and Reynolds 2012). Initially, large numbers of targets appear at the lowest altitudes observable with the radar. A little later, when more insects have ascended, the decrease in the insect aerial density with height may assume a semi-logarithmic form (up to some ‘ceiling’ height). Then, after an hour or so, numbers at the lowest altitudes (100–200 m) usually start to thin out as take-off ceases (and perhaps some very short-duration migrants land), and the profile assumes a relatively stable state which often shows evidence of layering (see section 3.5 below). In the absence of atmospheric disturbances, changes then occur only gradually through the night.
3.2 Dawn Emigration
There is generally a discrete take-off of crepuscular species during the dawn twilight period. This flight tends to be short-lived, although occasionally it continues for some time (Riley et al. 1991, 1994; Irwin and Thresh 1988; Reynolds et al. 2008; Rennie 2014) and leads to the formation of layer concentrations that may persist for extended periods in the stable early-morning atmosphere. Dawn layers sometimes ascend as the morning progresses, and if they last long enough they will usually be disrupted by the growth of daytime convection; their constituent insects then merge with the insect activity that has built up later in the morning in association with the convection. Occasionally, one or more insect layers associated with stable zones in the atmosphere above the convective boundary layer are still present at midday (e.g. Campistron 1975; Lothon et al. 2002).
3.3 Daytime Flight Activity
In fair weather, day-flying migrants take off from mid-morning onward as surface heating promotes boundary layer convection; these insects generally descend as convection dies down before nightfall. Insects flying in the convective ABL will tend to be circulated around in the atmosphere by up- and down-drafts, perhaps taking ~1–2 h to complete an individual flight (Johnson 1969). Continuously operating vertical-pointing meteorological or entomological radars are particularly suited to recording the development of daytime insect activity in the convective boundary layer (e.g. Contreras and Frasier 2008; Luke et al. 2008; Wood et al. 2009; Drake and Reynolds 2012; Bell et al. 2013). Detailed observations of insect targets within fair-weather convection cells will be discussed further below.
Occasionally, in northern temperate regions, small numbers of day-flying species such as aphids remain airborne into the dusk period or later in the night (Irwin and Thresh 1988; Chapman et al. 2004). This phenomenon may be much more common in warmer regions of the world (e.g. high densities of aphids were caught at altitude after midnight in northeast India; Reynolds et al. 1999). The mass invasion of aphids into southern Finland evidently required night flights across the Gulf of Finland/Baltic Sea (Nieminen et al. 2000; Leskinen et al. 2011), but here the over-water trajectories may have forced the aphids to continue in flight, and in any case during the midsummer migration periods there would be no astronomical darkness at these latitudes.
3.4 The ‘Transmigration’ Phase
Insects that enter the atmosphere above their FBL may not stay there very long—a huge variety of insects, particularly small ones, seem to ascend for relatively short periods at dusk, and sometimes at dawn (Riley et al. 1987; Drake and Reynolds 2012), leading to movements of a few kilometres or perhaps a few tens of kilometres. On the other hand, some highly migratory species, after the initial ascent phase, may maintain flight at heights well above their FBL for many hours before the final descent and alighting. This main period of horizontal translocation or transmigration is characterized by continuous steady wing-beating and, at least in the stable nocturnal ABL, rather little change in orientation direction or flight altitude. Displacement in near-geostrophic winds means that long distances can be covered in a night’s flight (e.g. up to ~400 km for the Australian plague locust, Chortoicetes terminifera) (Drake et al. 2001; Drake and Reynolds 2012; Drake and Wang 2013). Other well-documented examples of this type of transmigration include solitarious desert locusts, Schistocerca gregaria (Pedgley 1981), migratory grasshoppers (Chap. 13 in Drake and Reynolds 2012), some moths particularly those from the family Noctuidae (Chapman et al. 2010, 2012; Drake and Reynolds 2012) but also including the tiny diamond-back moth, Plutella xylostella (Chapman et al. 2002b), and certain rice planthoppers (Riley et al. 1991; Otuka et al. 2005, 2010).
Other long transmigrations occur when typical day-fliers (e.g. host-alternating aphids) sometimes continue flight after dark (Irwin and Thresh 1988; Reynolds et al. 1999, 2006). If migrants find themselves over the sea at the time of day (or night) when they would normally land, flights may be further extended and result in amazing traverses (e.g. migrations of the wandering glider dragonfly, Pantala flavescens, from India across the Indian Ocean via the Maldives, Chagos, and Seychelles to east Africa; Anderson 2009).
3.5 Insect Layering
The relatively stable insect density profile that typically becomes established an hour or so after the dusk emigration is representative of the ‘transmigration’ phase of migratory activity. Sometimes densities may be relatively constant up to a flight ‘ceiling’, but often the migrants concentrate around particular altitudes forming one or more layers of very broad horizontal extent (Drake 2014; Rennie 2014; Chap. 10 in Drake and Reynolds 2012). The insect layers so formed may become highly pronounced with densities one or two orders of magnitude greater than those just above or below the layer. The layering is likely to be a reflection of the stratified character of the nocturnal ABL, with migrants reacting to physical features of their atmospheric environment.
Many layers form at maxima in the temperature profile, particularly near the top of the nocturnal surface inversion (Drake and Farrow 1988; Wood et al. 2010; Chaps. 10 and 13 in Drake and Reynolds 2012; Rennie 2014; Boulanger et al. 2017), suggesting a response of the migrants to air temperature. In warm conditions where temperatures are less limiting, insects may fly far above the top of the surface inversion, in the residual layer. Elevated layers may form here associated with higher-altitude isothermal zones or inversions (e.g. a capping inversion left from the daytime atmospheric boundary layer) (Reynolds et al. 2005). In other cases, a layer may form at a flight ceiling, or there may be a gradual increase in density up to a distinct upper boundary at the ceiling. An example from China involved the brown planthopper, Nilaparvata lugens, and here the well-defined ceiling corresponded to a known temperature threshold for sustained flight in this species (Riley et al. 1991).
In other situations, the observed insect layers do not correspond to any temperature feature or known threshold, but they are associated with a local maximum in wind speed (see Chap. 10 in Drake and Reynolds 2012; Qi et al. 2014). This raises the question of what wind-related cues are being used by migrants in the selection of height of flight, and this is discussed further in the section on ‘Orientation behaviour’. Migration at the top of the nocturnal surface inversion would frequently locate the migrants at a boundary-layer wind speed maximum (i.e. in the low-level jet), so it may be difficult to separate the effects of temperature and wind-related cues in this case.
Insect layers have also been observed to form in regions of wind shear at the interface between a lower and an upper airflow. This may occur following the passage of a ‘gravity current’ (see Fig. 7.5) if insects are not retained within the head of the current but are swept back over its upper surface, forming a layer that may extend for many tens of kilometres (Schaefer 1976). In this case, the insects in the layer may be merely avoiding colder air in the density current itself, but there may also be responses to turbulence or Kelvin–Helmholtz waves at the current’s upper surface. In some circumstances, the insects seemed to be concentrated at an altitude where wind shear was greatest (Schaefer 1976; Hobbs and Wolf 1989), but in other situations layers were located in regions of reduced shear, e.g. migrants flying preferentially near the centreline of a nocturnal low-level jet (Wolf et al. 1986). Complex behavioural responses by the insects to the shear-zone turbulence, which may contribute to the formation of these layers (Reynolds et al. 2009), are of relevance to possible cues used by migrants to sense wind velocity at altitude and thus take-up wind-related orientation directions (see below); radar observations indicate that well-developed layering and the common orientation of migrants often occur together (e.g. Hobbs and Wolf 1989; Aralimarad et al. 2011).
Schematic view of a gravity current which is advancing towards the right (From Geerts et al. 2006. © American Meteorological Society. Used with permission)
Insect layers may also occur during the day. Apart from layering in the stable early-morning atmosphere (mentioned above in connection with dawn take-off), layers of stable air may occur later in the day above the ABL (Campistron 1975; Chap. 10 in Drake and Reynolds 2012); for example, in warm layered airstreams advected above a cooler surface flow (Browning et al. 2011). It seems that day-flying insects (particularly small species that depend on convective upcurrents to assist in their ascent) will be confined by any neutral or stable layers aloft and accumulate near the base of these features leading to highly stratified density profiles (Isard et al. 1990; Lothon et al. 2002).
There are occasional radar reports of large insects in layers at particularly high altitudes (~3 km a.g.l.) during daytime (Drake and Farrow 1985; Leskinen et al. 2012). These observations were made by experienced observers (so the echoes concerned were not likely to have been confused with those from birds), and they are of interest because flight was occurring at very low temperatures (~5 °C), and because the insects were probably noctuid moths and these might be expected to land at daybreak. We must conclude that these large moths [the case described by Leskinen et al. (2012) involved in mass migration of underwings Catocala spp.] generate enough internal heat by wing-beating that they are able to continue flying in low ambient temperatures.
Lastly, in conditions of high directional wind shear, it is conceivable that layers could arise from differing take-off numbers in different source regions, or from differing flight durations in the two airflows, one of which is closer to a favoured displacement direction (Chapman et al. 2010)—though only if there is little ascent or descent of migrants from one airflow to the other.
3.6 The High-Altitude Orientation Behaviour of Migrants
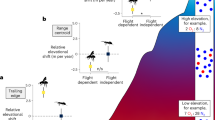
It might be thought that high-flying nocturnal insect migrants would be swept along by strong high-altitude winds, orientating at random in what amounts to a ‘sensory vacuum’, but from the earliest days of radar entomology it has been clear that large insects, at least, often show a high degree of common alignment (e.g. Schaefer 1976). A recent advance has been the revelation of the degree of sophistication of some orientation strategies (see review by Reynolds et al. 2016) and accumulating evidence that these would significantly enhance migration success, i.e. the observed orientations are ecologically adaptive (Chapman et al. 2012). The best documented case is that of A. gamma (silver Y moth) which shows the following elements: (i) migrations are only initiated (or sustained for any length of time) on nights when the winds at altitude will carry the moths approximately in the seasonally favourable direction; (ii) there is a seasonal reversal of the preferred direction between spring and autumn (but the nature of the internal compass sense is not known); (iii) flight is often at the altitude of the fastest winds; and (iv) headings are adopted that partially counteract crosswind drift from the preferred migration direction (Chapman et al. 2008a, b, 2010, 2013, 2015b).
Some smaller migrants use a simpler strategy—i.e. they adopt a more-or-less downwind heading thus adding their airspeed to the wind speed and maximizing the displacement distance in a given time. Current evidence indicates that high-altitude nocturnal orientations are maintained by some intrinsic property of the wind, rather than (or as well as) an optomotor-type visual reaction to relative ground movement (Reynolds et al. 2016). In fact, the night-flying migrants seem to be using turbulent velocity and acceleration cues to determine the mean wind direction, and this suggestion is supported by a consistent tendency for headings to be offset on average by ~20° to the right of the downwind direction—the bias expected (in the Northern Hemisphere) from theory (Reynolds et al. 2010, 2016; Aralimarad et al. 2011). The day-flying windborne migrant fauna has been rather neglected in radar entomological studies, but recent analyses (Hu et al. 2016a) found that adaptive tailwind selectivity and wind-related orientations were extremely common in medium-sized (~10–70 mg) and large (70–500 mg) insects migrating during the daytime, and in the period around sunset. Observations of insect layers with powerful Doppler meteorological radars show that echo patterns due to common orientation extend over very large areas (e.g. Lang et al. 2004; Rennie 2014), and this is further evidence that behavioural mechanisms are dependent on broad-scale, rather than localized, cues.
Lastly, we note that in very small migrant insects, headings at altitude may be random (Riley et al. 1991; Qi et al. 2014), or at least unrelated to facilitating movement in any ecologically adaptive migration direction. This is not surprising as, due to their low airspeeds, these insects will have very little influence on their migration direction when flying above their FBL. In the case of aphid clonal migrants, such large numbers of offspring are produced that a strategy of flight in winds from all directions is quite sustainable.
3.7 Aerial Densities and Migration Rates of Insects Aloft
Aerial densities and other quantitative measures of intensity of insect flight activity may provide information useful in many fields of basic and applied ecology. In the context of aeroecology, they may be of particular interest in connection with the feeding resources of aerial insectivores (McCracken et al. 2008; Kelly et al. 2013; Krauel et al. 2015). To mention one example, large numbers of Brazilian free-tailed bats (Tadarida brasiliensis) are known to forage at high altitude over Texas and perform a valuable ecological service for farmers by preying on migrant pest moths such as Helicoverpa zea (Cleveland et al. 2006; McCracken et al. 2012). Calculations of insect migration intensity from radar-derived data can be used to assess the ecological significance of a migration (Hu et al. 2016b) and to determine the food resources available to the bats (see p. 230 of Drake and Reynolds 2012).
Density values recorded with radar during peak emigration periods, when insect flight activity is likely to be most intense, are shown in Table 7.1; the values span almost six orders of magnitude (1 × 10−7–1 × 10−1 m−3). As might be expected, species in which outbreaks occur (locusts, and some grasshoppers and moths) exhibit high aerial densities on evenings immediately after a population has developed into its adult (i.e. flight-capable) form (e.g. Riley et al. 1983; Rose et al. 1985). Area densities of largish insects are, however, dwarfed by those of small species such as the rice brown planthopper (Table 7.1). To put these values in context, aphids and other small insects near the ground, and swarms of the desert locust, may sometimes attain densities of ~10 m−3 (Johnson 1969), though typical values would be much lower (Table 11.1 in Drake and Reynolds 2012).
One measure of the outcome of a migration is the increase in population in the destination region. Radar observations provide a proxy measure for this in the form of the ‘passage rate’ or the ‘total overflight’ (over a line drawn perpendicular to the direction of motion; see Chap. 9). Once again, published values (see Table 10.3 in Drake and Reynolds 2012) are likely to be associated with significant migration events and thus tend towards the maximal rather than the typical. Migrations of large insects like common noctuid moths or night-flying grasshoppers/locusts have recorded total overflights ranging from a few tens to a few thousands per meter per night, while for small insects like the brown planthopper overflights as high as 1.2 million per meter in just 2.5 h have been estimated. The overflight quantity can be accumulated over extended periods where appropriate, e.g. for the duration of fledging/emergence for a particular subpopulation (Chen et al. 1989), or throughout the migration period for the seasonal redistribution of a species between different climate zones (Chapman et al. 2012; Hu et al. 2016b).
3.8 Insect Movement in Relation to Large-Scale Weather Systems
The relation of long-range insect migrants to synoptic airflows is discussed by Johnson (1969), Pedgley (1982), Drake and Farrow (1988), Johnson (1995) and Pedgley et al. (1995). In middle latitudes, weather conditions and wind directions are predominantly determined by a succession of large-scale pressure systems (depressions and their associated fronts, and anticyclones) which move generally eastwards, each one taking a few days to pass over any particular locality (see Chap. 2). Depressions are associated with cloud, rain, and strong winds while conditions during the passage of an anticyclone are characteristically settled, with usually clear skies. Winds circulate around both types of system (in opposite directions), so that a particular locality experiences changing wind directions as the system passes over. Johnson (1969, pp. 477–481) presents some idealized trajectories that take account of the ambient wind, the movement of a depression, and the orientation and air speed of an insect. In practice, of course, trajectories are likely to be disturbed by weather associated with the depression, particularly if/when the insect encounters cold air or falling rain.
Winds from lower latitudes will generally be warm, and these winds provide transport for insect migrants to move polewards in spring, when cooler regions become seasonally favourable for breeding; numerous examples are mentioned in the references cited in the previous paragraph. In north-west Europe, some mass arrivals are from the east rather than the south, from sources in western Russia or even further afield, on warm south-east winds blowing around large anticyclones centred over north-western Russia (Mikkola 1986; Pedgley et al. 1995). Selective movement on warmer winds could arise without a specific adaptation, if the threshold temperature for flight is only exceeded when these are blowing. However, as temperatures fall in autumn, many migrant species need to return to warmer winter-rainfall regions, and this movement will often require a cued response, with flight in warmer conditions suppressed as it would now take the migrants in exactly the wrong direction (McNeil 1987; Chapman et al. 2012).
Radar observations in southeastern Australia identified warm northerly and northwesterly winds ahead of a cold front as a vehicle for major southward (i.e. poleward) movements of a variety of moth species in spring (Drake et al. 1981; Drake and Farrow 1985). In contrast, a northward movement of locusts that persisted for several days in early summer occurred in southerly winds on the western side of an almost stationary anticyclone (Drake and Farrow 1983). In the southern USA, southward movements of moths in autumn occurred on northerly airflows immediately following the passage of a cold front (Pair et al. 1987; Beerwinkle et al. 1994; Krauel et al. 2015). Similar examples have been observed in East Asia (Ming et al. 1993; Riley et al. 1995; Feng et al. 2009). In other cases, the suitability of winds for equator-ward ‘return’ migrations cannot be assessed from simple meteorological factors such as temperature, and migrants seemingly use an internal compass sense to assess whether the direction of windborne displacement is close enough to an innate seasonally preferred direction (e.g. in A. gamma in northern Europe).
In the tropics, climate and weather are dominated by the seasonal movements of the Intertropical Convergence Zone (ITCZ) and its associated monsoonal winds, which bring alternating wet and dry seasons (see Chap. 2). In the savanna and steppe regions in West Africa, for example, several species of grasshoppers, heteropteran bugs, moths, and flies are able to take advantage of the constancy of the seasonal reversal in the monsoon/trade wind system to move between complementary breeding (or breeding and diapausing) areas (Pedgley et al. 1995). As the ITCZ moves northwards, rain falls at progressively higher latitudes, and windborne migrants move into areas where vast seasonal resources are becoming available (e.g. new growth of semi-arid vegetation or newly flowing waterways). Correspondingly in autumn, equatorward migrations predominate on the re-established trade winds in the wake of the retreating ITCZ. Some populations may move far to the south where the rains are late enough to allow breeding, while others move less far and persist in dry-season refuges. We note that the putative world’s longest insect migration—that of the above-mentioned P. flavescens dragonflies from India to Africa across the western Indian Ocean—takes place on north-easterly winds at altitudes above 1000 m behind the southward-moving ITCZ (Anderson 2009).
Another remarkable migration is the movement of pest planthoppers and moths from central and southern China to Korea and Japan (e.g. Otuka et al. 2005; Tojo et al. 2013), which occurs in June–July every year on low-level jets associated with the quasi-stationary Bai-u front (East Asian subtropical rain-band)—the boundary separating the hot humid tropical air mass to the south from the cooler polar air mass to the north (see Chap. 2 on the transport of biota on Asian monsoonal winds). Because of the economic implications of these pest invasions, the planthopper movements, particularly, have been intensively modelled using numerical weather prediction programmes combined with submodels to take account of atmospheric dispersion and insect flight parameters (e.g. Otuka et al. 2005). In North America, migrations of moths of the fall armyworm Spodoptera frugiperda, a serious crop pest, have been the subject of a comparably comprehensive computer modelling effort (Westbrook et al. 2016).
The subtropics are the locations of extensive deserts, where insect breeding is limited by rainfall. The classic long-distance desert migrant is the desert locust, S. gregaria, whose downwind movements are able to span the Sahara, allowing breeding on monsoon rains associated with the ITCZ in summer, and on cyclonic rains associated with midlatitude westerly airflows in the Mediterranean in winter (Pedgley 1981). It has recently been shown that migrations of the painted lady butterfly Vanessa cardui extend from northern Europe to both the Maghreb region north of the Sahara and the Sahel zone to its south and therefore involve a desert crossing (Stefanescu et al. 2016; Talavera and Vila 2017).
4 Insect Migration-in-Progress 2: Localized Aggregations of Insects Aloft
Aggregations of insects in the air can arise from a variety of causes (Chap. 11 in Drake and Reynolds 2012). In this section, we will be mainly concerned with the effects on flying insects of small-scale wind systems, or of precipitation, but first three other causes of localized concentrations seen on radar displays will be mentioned.
If there is non-uniformity in the spatial distribution of ground populations around the radar site, this may be reflected in patchiness of echoes from emigrating insects displayed on the radar PPI screen. These patches can be plumes of insects streaming away from what is virtually a point source (a single tree, perhaps) or patches of dimensions of several hundred meters such as crop fields (Chap. 11 in Drake and Reynolds 2012) up to extensive areas (thousands of square kilometres) observable with operational weather radars (Westbrook 2008; Rennie 2012; Westbrook et al. 2014; Boulanger et al. 2017; Westbrook and Eyster 2017). Secondly, migrating insects may show an active behavioural response to a topographical feature (hill, coastline, river, etc.). For example, Russell and Wilson (1996, 2001) found radar evidence of a tendency in insects aloft to resist being carried over a coastline by (light) off-shore winds, and this resulted in a temporary accumulation of insects along the coast. Thirdly, there may be behavioural responses to other individuals, of the same or a different species. The classic example here is the locust swarm, where cohesion is maintained through mutual interactions between the gregarious individuals comprising the swarm (pp. 257–258 in Drake and Reynolds 2012). Other examples of ‘swarms’ observed on radar include: honeybee drone congregation areas (pp. 352–354 in Drake and Reynolds 2012) and the nuptial and oviposition swarms of mayflies (Ephemeroptera) above rivers and lakes (pp. 350–352 in Drake and Reynolds 2012). In the latter case, the enormous swarms of mayflies on the Upper Mississippi valley regularly produce spectacular displays on the WSR-88D radars of the US National Weather Service (http://www.crh.noaa.gov/arx/?n=mayflygeneral). It should be noted that these phenomena involve appetitive flights rather than migration. An example of another taxon, namely bats, causing an effect on concentrations of large migrating insects (e.g. grasshoppers and moths) is manifested by distinctive echo-free lacunae that persist for a few tens of seconds on PPI displays of entomological radars (pp. 258–260 in Drake and Reynolds 2012). These ‘blank patches’ are the result of the insects making evasive flight manoeuvres (power diving, stalling, turning away) in response to the ultrasonic emissions from an approaching hunting bat.
4.1 Concentration by Small-Scale Meteorological Disturbances
The relatively steady movement of migrant insects in broad-scale wind flows (described above) may be disrupted by disturbances on horizontal scales of a few tens of meters up to ~200 km (i.e. more or less the micro-α, meso-γ, and meso-β scales in standard meteorological parlance, see Orlanski 1975). These disturbances comprise terrain-induced phenomena (organized daytime convection, land/sea breezes, slope winds, lee waves, and vortices) and those (e.g. convective storms, squall lines) induced by instabilities in moving synoptic-scale systems. Most of these wind perturbations will have associated convergence zones within which airborne insects will tend to become concentrated. The insects are carried horizontally towards the zone of convergence by the low-altitude wind flow; they then resist (to a greater or lesser extent) being carried upward in the rising air at the convergence and thus tend to accumulate there, forming distinctive echo patterns on entomological and meteorological scanning radars. At a certain altitude, air temperatures will drop below the insects’ flight threshold, and the migrants would be physiologically unable to fly; before this point is reached, however, it appears that migrants often respond behaviourally, by ceasing flight or actively flying downward (Achtemeier 1991, 1992; Geerts and Miao 2005; Browning et al. 2011). Some such mechanism has presumably evolved to prevent insects being carried involuntarily to great heights in the violent updrafts associated with thunderstorms (Browning et al. 2011), though this does occasionally happen—as evidenced by small insects forming the core of hailstones (Browning 1981).
In daytime fair weather, mesoscale shallow (1–2 km deep) convection may take the form of polygonal cells or parallel bands in the ABL, and these patterns are visible on radar due to insect echoes. The smallest-scale insect concentrations detected by radar include portions of the walls of individual convective cells (Schaefer 1976), and a series of studies with different types of radar confirm that the regions of high reflectivity are located in updrafts (Chandra et al. 2010, pp. 261–264 in Drake and Reynolds 2012); the emptier centres of the cells are broader regions of downdraft, which are relatively free of insects. Occasionally, large areas can be covered by patterns of these 5–10 km diameter ‘Bénard-like’ cells (Hardy and Ottersten 1969; Rennie 2012). Under certain conditions of surface-layer heat flux and wind shear, the cellular pattern changes to ‘streets’ of counter-rotating convective rolls aligned downwind (Weckwerth et al. 1999; Koch 2006). In fine weather, day-flying insects ranging from aphids to swarming locusts can take advantage of updrafts produced in unstable convective atmospheres in order to reduce the energy needed to remain aloft, so staying within a region of uplift in a convection cell is clearly adaptive. In fact, on days (or at times of day) when atmospheric convection is absent, insects do not show this ‘cumuliform’ vertical distribution; instead, aerial density profiles are stratified with aphids, for example, being restricted to within a few tens of metres of the surface (Johnson 1969), or confined by stable or neutral layers at higher altitude (Isard et al. 1990). However, as far as we know, even large insects like swarming desert locusts do not appear to actively seek out thermals (pp. 37–38 in Rainey 1989) in the way that soaring birds like vultures and storks do (see Chaps. 8 and 9).
Gravity Currents
Another common insect-concentrating mechanism is the atmospheric gravity current (also known as a ‘density current’ or ‘buoyancy current’), which occurs when a higher density (cooler) airflow intrudes into and displaces lower density (warmer) air. The leading edge of the advancing gravity current forms a distinct ‘front’ where it undercuts the ambient warmer air and both airstreams (mostly) ascend. The gravity current is often slightly deeper immediately behind the front—in other words, it forms a ‘head’ on the top surface of which there may be a zone of intense mixing, with breaking waves (Kelvin–Helmholtz waves) streaming back over the upper surface of the current (Simpson 1997; and see Fig. 7.5). The passage of the leading edge of a gravity current is typically marked by a gust line with a wind-shift, turbulence, a pressure jump and a temperature fall. Insects carried into the head from the rear tend to resist being taken up by the rising air and thus accumulate there. In addition, any insects that are carried upwards in the rising ambient air just ahead of the front may be entrained by the zone of strong turbulent mixing around the head and be incorporated into the concentration there.
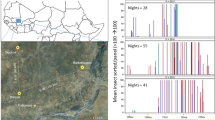
Various manifestations of the atmospheric gravity current revealed by radar returns from insects include the following: storm outflows (Lothon et al. 2011), sea or lake breezes (Simpson 1994; Sauvageot and Despaux 1996; Russell and Wilson 1997; Isard et al. 2001), katabatic flows (Schaefer 1976), drylines (sharp boundaries separating moist and dry air masses) (Weiss et al. 2006), mesoscale cold fronts (Geerts et al. 2006), and prefrontal wind-shift lines (Bluestein et al. 2014); see Chap. 11 of Drake and Reynolds (2012) for a unified treatment of these phenomena. One striking example is shown in Fig. 7.6—a radially spreading storm outflow observed in Niger in the West African Sahel (Lothon et al. 2011).
Reflectivity pattern in a horizontal cross section at 600 m height, using data from a C-band Doppler radar at Niamey, Niger, in late afternoon (17.20 UTC) of 10 July 2006. A westward-moving storm system (denoted S0) has produced the circular and radially expanding gust front made visible by insect scatterers. A second, smaller, convective cell (denoted C1) has formed at the east side of the circular outflow apparently caused by the interaction of the outflow with a low-altitude wave train previously generated by S0. Further observation indicated the circular gust front dissipated shortly after 2100 UTC, about 60 or 70 km away from the radar (From Lothon et al. 2011. © American Meteorological Society. Used with permission)
The insect density in the concentrations may be 1–2 orders of magnitude greater than in surrounding areas, and volume densities may approach 10−1 m−3 for small day-flying insects. These concentrations may form a valuable feeding resource for aerial insectivores, and, indeed, sea-breeze fronts were delineated on early air-traffic control radars by swifts feeding along the front (p.135 in Simpson 1994). The aerial concentration of pest insects, if these land while in a convergence or soon after encountering one, may lead to high ground densities and serious localized outbreaks. In the case of migratory grasshoppers such as Oedaleus senegalensis or Aiolopus simulatrix in the Sahel, the flight-capable (i.e. adult) stage can cause immediate damage to crops, and Joyce (1983) considered that A. simulatrix is a problem only when grasshoppers have been concentrated in convergence zones. In other cases, such as with the African armyworm moth (Spodoptera exempta), the damage is caused by caterpillars developing after mass oviposition by the initial migrants. There is a well-attested case in this insect for the initial low-density moth populations being brought together by wind convergence associated with rainstorms, so that the resulting caterpillars occur as very dense infestations of the ‘gregarious’ phase which can cause widespread and serious damage to crops (Rose et al. 2000; Chap. 12 in Drake and Reynolds 2012). In fact, satellite-derived information on rainstorm distribution is useful in predicting potential armyworm outbreak sites (Tucker 1997).
There can be little doubt that gravity currents perturb the flight trajectories of migrants flying at lower altitudes, and the flight orientations of insects trapped in the head of the current are certainly disrupted (Greenbank et al. 1980). In the case of storm outflows, modifications to migration flight paths would appear to be limited in duration and usually rather random in direction and timing, but gravity currents associated with large-scale topographic features, such as sea breezes and katabatic flows, may produce more systematic effects. For example, the regular penetration of sea breezes deep inland has significant effects on population distribution of the spruce budworm moth (Choristoneura fumiferana) in New Brunswick, Canada (Neumann and Mukammal 1981).
A dramatic detrimental effect of the coastal circulation on small insects was highlighted in a study made with a polarimetric Ka-band Doppler radar on the Atlantic coast of France (Sauvageot and Despaux 1996). On days when the land/sea breeze cell became well established, small insects were seen to be carried out to sea on the ‘return flow’ (between ~500 and 1000 m altitude) but were brought back towards land when they descended into the sea breeze layer (below 500 m). By the early evening, however, the diurnal veering of the low-altitude wind meant that the insects were no longer being returned by the onshore flow; instead they accumulated in a band located between a few kilometres and ~50 km out to sea. Eventually, the bands dissipated in situ, as the insects were apparently deposited in the ocean. This phenomenon has seemingly not been reported from elsewhere, so it is not clear how widespread its occurrence is; indeed, there is radar evidence from elsewhere indicating that insects sometimes resist being taken over coastlines (see above). Experiments with tethered individuals suggest that desert locusts, at least when flying at low altitudes, can detect the polarized reflections from large bodies of water and thus avoid moving over them (Shashar et al. 2005).
Lake breezes are atmospheric circulations similar to (but generally weaker than) sea breezes that form along the shorelines of large bodies of water such as the Great Lakes of North America. Isard et al. (2001) review information on a variety of insect taxa with particular reference to the mechanisms causing the movement between the Great Lakes’ coastlines and the inland, and the deposition of high concentrations of (living) insects along the lake shore. They suggest that the insects are initially collected and lifted up at a lake breeze front and are transported in the counter-flow out over the lake. If they then descend to near the water surface, they will be carried in the surface flow back to the coastline, where retardation of the air flow by dunes and trees slows the wind and allows the insects to land (Isard et al. 2001). Clearly, other outcomes are possible: if the lake breeze is strong, insects at the coastline may be merely blown inland. Interpretation of insect movement over land–water boundaries may also be complicated by interactions between the wind flows of the lake/sea breeze circulation, and the insects’ optomotor reactions to the water or land surface, which may lead to so-called ‘rectification’— i.e. the boundary facilitates transport in one direction, but hinders it in another (for details, see pp. 134–136 in Johnson 1969).
Bores and Waves
Another group of phenomena that may perturb the steady horizontal migration of insects comprises atmospheric bores (propagating hydraulic jumps indicated by an abrupt change in the height of an atmospheric layer) and waves (e.g. solitary waves, Kelvin–Helmholtz waves, lee waves) (Chap. 11 in Drake and Reynolds 2012). A bore may take the form of a smooth wave front followed by a train of waves (an undular bore). These atmospheric internal bores can be initiated by colliding gravity currents or by an advancing gravity current intruding into an existing stable layer. Once generated, a bore can propagate far ahead of the initiating gravity current, and its passage is typically attended by a wind shift and a small but sustained increase in surface pressure, but (unlike a gravity current) there is no rapid drop in surface temperature. The bore may then evolve further into a solitary wave—a stable ‘wave of elevation’ (occurring either individually or as an amplitude-ordered short wave train) which can propagate for long distances with little loss of form (Knupp 2006). Bore and wave disturbances generally seem to displace migrants upwards and downwards, rather than increase aerial densities, but concentration may occur in closed horizontal vortices which develop in some large amplitude waves. Insects need to remain entrained in the closed circulations for some time, however, before there will be any significant effects on the migrants’ flight trajectories (Drake 1985). Bore-type disturbances can be quite common in certain locations /seasons, most famously as the ‘Morning Glory’ cloud formation over the Gulf of Carpentaria in northern Australia, but radar observations have recently documented examples in other parts of Australia (Rennie 2012) and in the USA (Knupp 2006). Figure 7.7 shows an example observed on a Doppler C-band weather radar in northern Victoria, Australia (Rennie 2012). Rennie commented that waves often preceded or sometimes followed precipitation, suggesting their origin in a storm outflow or gust front; insect migration often coincides with unsettled convective weather in this part of inland Australia, rendering these disturbances detectable with the weather radar.
Insect echo revealing linear disturbance, probably an atmospheric solitary wave train, shown in a reflectivity scan from a Doppler C-band weather radar at Yarrawonga, N.E. Victoria, Australia (00.12 h LST, 25 Nov 2010; elev. ~1°). The wave train—three lines are visible, the first being the most prominent—is approaching from the west, ahead of rain echo. In this example, the line echo was not associated with any substantial change in the insect velocities measured by the radar (not shown), indicating a wave disturbance rather than a gravity current (From Rennie 2012. © 2012 CSIRO and the Bureau of Meteorology)
Lee waves are stationary waves on the downwind side of a topographic barrier such as a mountain ridge, and if the waves are of large amplitude they can contain stationary closed horizontal vortices or ‘rotors’. The effect of rotors and other topographic eddies on flying African armyworm moths has been documented in the Rift Valley of Kenya (Chap. 11 in Drake and Reynolds 2012). Rotor circulations may occur repeatedly at particular locations and so potentially have more significant effects than travelling waves on migrant insect populations transiting or developing in the vicinity.
Tornadic Thunderstorms and Hurricane Eyes
‘Hook echoes’ seen on Doppler weather radars are typical of supercell thunderstorms that give rise to tornadoes, and these echoes are likely to be regions of very strong inflow. The Differential Doppler velocity (DDV) field can exhibit very large values of both signs at the ends of ‘hooks’ and this, as well as other polarimetric radar variables, indicates these areas are filled with highly non-spherical (i.e. biological) scatterers—most likely trapped insects and birds (Melnikov et al. 2014a, b).
Insects and birds can also become trapped in the eyes of well-developed tropical cyclones, e.g. large numbers of black witch moths (Ascalapha odorata) were present in the eye of a hurricane on the Texas coast (Freeman 2003). Early reports of this phenomenon are reviewed by Van den Broeke (2013) who also documents recent examples observed by polarimetric radar where the areas of bioscatter were revealed by high differential reflectivity combined with a very low copolar correlation coefficient.
4.2 Effects of Rain on Migrating Insects
In many areas of the world, outside of the arid/semi-arid zones, one of the most tangible agents disrupting steady long-distance insect migration is rainfall. The extensive nature of some synoptic rain-bearing systems makes them difficult to avoid, and these systems may have significant general effects on migratory activity in insects. However, radar studies of the precise effects of rain on migration are hindered by the strong attenuation and masking effects of raindrops at X-band frequencies, thus making it difficult to detect insect targets in anything other than the lightest precipitation. Consequently, the effects of rain are less certain compared to the influences of various fine-weather phenomena. Complications also arise because there are several mechanisms associated with rainy weather, other than direct impaction by raindrops, that may cause migrants to descend or land. These include: descent due to ambient temperatures falling below the flight threshold; descent due to cessation of convection (which many day-flying migrants require to remain aloft); and insects being forced down by strong downdraughts associated with convective rainstorms.
A recent laboratory study has shown that a relatively small flying insect (an Anopheles mosquito) flying in heavy rain is likely to be struck by raindrops but that its small mass, and the fact that it can be displaced downwards (unlike an insect on a solid surface), allows it to survive these high-speed impacts (Dickerson et al. 2012). Nevertheless, the insect experiences enormous accelerative forces (100–300 g) and its trajectory is greatly disrupted, so the rain may be best avoided.
Spruce budworm moths (C. fumiferana) have been observed to continue flying in very light rain associated with a synoptic warm sector (Greenbank et al. 1980). In cases like this, the air temperature evidently remained above the flight threshold, and the probability of raindrop impact may be so low that migrants are unaware of the rain. In heavier rain, most radar evidence points to a termination of migratory flight (see Chap. 11 in Drake and Reynolds 2012). In the case of scattered convective rainfall cells, radar observations have shown that either the heavy rain itself or associated cold-air downdrafts locally interrupted migratory flight (Greenbank et al. 1980). The rain shafts falling from higher altitudes were vividly envisaged as puncturing a layer of migrating moths producing a high-density strip of deposited moths on the ground; however, flight continued outside the actual rain cells. In the case of synoptic systems (e.g. midlatitude warm fronts) that produce widespread moderate-intensity rainfall over an extended period, one can envisage that migration may well be terminated for the night over an extensive area.
In arid and semi-arid areas of the world, where rainfall is erratic or highly seasonal, deposition (whether active or passive) in the vicinity of rainstorms may be highly adaptive. Here migrants such as locusts and African armyworm moths (see above) need to arrive in locations where rain has recently fallen, as this will provide the flushes of new vegetation and other resources needed for survival and breeding (Pedgley 1981; Rose et al. 2000).
5 Ecosystem Consequences of Long-Range Mass Movements of Insects
While aeroecology is concerned primarily with the processes of animal movement within the atmosphere, much of the work described here has been inspired and driven by concerns about its consequences, i.e. with the outcomes of the migrations. Quantitative measures of intensity and directional transport of migrant insects may provide information useful in the fields of population ecology and ecological genetics, the ecology of vectored microbes, pest and disease management, and conservation biology (Chapman et al. 2015a; Bauer et al. 2017). Clearly, the immense seasonal shifts of insect biomass to and from higher latitudes in response to seasonal temperature changes (Hu et al. 2016b), or tracking erratic or seasonal rainfall in arid lands (Drake et al. 2001), will have huge ecological impact as a result of trophic and transport effects (see reviews by Holdo et al. 2011; Bauer and Hoye 2014; Landry and Parrott 2016). The effects of insect migration on ecosystem functioning include direct ‘trophic’ effects of a mass arrival of consumers, competitors, predators or prey—for example, competition with concomitant migrant or resident species for shared resources, or interactions mediated by shared predators (apparent competition). For example, an investigation of a dual-migrant (noctuid moth and bat) system in Texas found that spikes in local abundances of moths were associated with synoptic weather (e.g. passage of cold fronts), and this in turn influenced behaviour and resource acquisition of Brazilian free-tailed bats (Krauel et al. 2015) (Chap. 15). The indirect ‘transport’ effects include: the transport and recycling of nutrients and energy (via feeding, excretion, reproduction, and decomposition), the ‘vectoring’ of parasites, pathogenic agents and symbionts, and the spread of genetic material (by means of the migrants themselves or by the carriage of seeds and pollen).
Many effects of insect migrants will be benign, e.g. delivery of ecosystem services such as pollination (Ssymank et al. 2008), biological control of pests (Chapman et al. 2006; Jeffries et al. 2013), maintaining susceptibility to insecticides by diluting the pool of resistant genes (Brévault et al. 2008), and provision of food to higher trophic levels (e.g. the above-mentioned aerial insectivores, grizzly bears feeding on the army cutworm (Euxoa auxiliaris) (Bauer and Hoye 2014), or the entire suite of predators sustained by bogong moths (Agrotis infusa) (Green 2011; Warrant et al. 2016)). Other effects will be harmful, however, e.g. infesting crops with pests that cause major yield losses (Chen et al. 1989), spreading infectious plant and animal diseases (Reynolds et al. 2006; Otuka 2013), and the spreading of genotypes associated with resistance to insecticides (Scott et al. 2005) and overcoming insect-resistance in crop varieties (Tanaka and Matsumura 2000). Elucidating the diverse impacts of a mass arrival of insect migrants on ecosystem function remains an extremely challenging prospect, but the first stage is to accurately quantify it, both in terms of the numbers of insects involved and their biomass. For some classic invasive agricultural pests (locusts, aphids, armyworm moths), a major objective of determining the population trajectory of the population (or its various subpopulations) through space and time has been largely achieved [e.g. for swarming populations of the desert locust (Schistocerca gregaria) (Pedgley 1981) and Australian plague locust (Deveson and Walker 2005), or for aphid crop pests in the UK (Taylor 1985)], although there may be current concerns as to whether these trajectories will alter with climate change and whether current operational forecasting capabilities can be sustained into the future. Operational applications of radar in insect pest forecasting and management have already been implemented in a few instances (e.g. Leskinen et al. 2011; Drake and Wang 2013), and there is current interest in developing further systems, e.g. in China and the USA.
6 Contrasting the Airborne Migration Strategies in Large Insects and Vertebrates
Here, we draw attention to the opportunities provided by modern observing techniques for making comparisons of migration physiology, behaviour and ecology in distantly related taxa (Hein et al. 2012); migration in a fluid medium (air or water) where movement is affected, to a greater or lesser extent, by the flow of the medium itself appears of particular interest (Chapman et al. 2011b). Highlighting similarities and differences among diverse taxa in orientation and navigation mechanisms, the energetics of migratory flight, and mortality rates during migration (Dudley 2000; Dingle 2014; Chapman et al. 2012, 2015a, b; Reynolds et al. 2016) will surely lead to insights into the evolutionary drivers of animal migration strategies.
For example, a comparative radar study of passerine songbirds (Old World warblers, flycatchers and chats) and noctuid moths (A. gamma) migrating over north-western Europe showed that the moths, despite having much slower self-propelled airspeeds, achieved virtually the same ground speeds and track directions as the birds (Alerstam et al. 2011; Chapman et al. 2015b, 2016a). The primary reason for this surprising result was that the moths took greater advantage of wind assistance (but only flew when the wind direction was favourable), whereas the birds were able to migrate on winds from a variety of directions but consequently received less assistance. There were also interesting differences in the types of orientation behavior exhibited by the two groups of migrants (Chapman et al. 2015b, 2016a). Overall, the birds’ strategies allowed more temporal and spatial control over their flight paths, and they were able to be more risk-averse than were the moths. The high fecundity of many migratory noctuids (Chapman et al. 2012) would, of course, enable them to bear large migration losses.
Quantification of mortality during migration is particularly problematic—there is very little empirical data on survival during regular seasonal migrations of insects, such as are becoming available for birds (Sillett and Holmes 2002; Menu et al. 2005; Klaassen et al. 2014) (but note Ward et al. 1998; Chapman et al. 2012), and it is difficult to assess the effect of enormous losses at sea which are occasionally reported (e.g. Farrow 1975; Sauvageot and Despaux 1996). The advent of continuous monitoring of insect movement over large areas by weather surveillance radar networks (see below) should help in this respect.
Incidentally, the recent comparison of migration distances among taxa (Hein et al. 2012) showed how truly spectacular insect migrations are, when scaled for body size; the number of body lengths covered by moths and dragonflies, for example, was roughly 25-times that travelled by geese.
7 Prospects and Future Challenges
There is no shortage of specific unresolved aeroecological questions that can be investigated by radar and complementary techniques (see Chaps. 9 and 12). For example, tracking radar could be used to identify individual high-altitude ‘hawking’ targets, i.e. types of bats or birds, while at the same time, information could be obtained on their insect prey by means of vertical-looking entomological radars (VLRs). In the case of bats such as the Brazilian free-tailed bat (Tadarida brasiliensis) that prey on quite large insects, the prey items (e.g. noctuid moths) could be identified with current X-band VLRs, but if the aerial predators consume small insects, a higher frequency VLR system would be needed, in order to reliably detect micro-insects (mass ~1 mg) over all their flight heights.
Future prospects for the remote sensing of airborne migrants over extensive spatial scales is entering an exciting new phase due to (a) the expansion of networks of specialized biological radars and (b) their integration with weather surveillance radars in order to monitor continent-wide movement of birds, bats, and insects (Dokter et al. 2011; Chilson et al. 2012a, b; Kelly et al. 2012; Shamoun-Baranes et al. 2014). In Europe, this initiative is being implemented under the ENRAM (European Network for the Radar surveillance of Animal Movement) research network (Shamoun-Baranes et al. 2014), and the entomological aspects of the program include pilot studies to create a platform for intensive cross-calibration measurements, involving the simultaneous deployment of multiple remote sensing devices (e.g. air traffic surveillance radar, dedicated ornithological and entomological radars, thermal imaging equipment, and opto-electronic systems) all within the sensed volume of one or more operational weather radars. The ultimate objective is to combine real-time insect altitude profiles into a multi-radar mosaic so as to produce visualizations of insect migration on a regional- or continental scale. Such radar-based monitoring networks will provide essential data on seasonal numbers, timings, and routes of insect mass migrations—there is, for example, a pressing need for more information on the overwintering distributions for a range of insects that invade northern temperate areas each summer. We note that case studies exist for quite a few long-range migratory insects, particularly pest species (Drake and Reynolds 2012), and the challenge is now to extend these to ecosystem-wide monitoring over regional scales (Kelly and Horton 2016). Wide-area monitoring networks will also help the operational detection (and perhaps ‘nowcasting’) of irregular incursions due to irruptive outbreaks of particular pest species (c.f. Nieminen et al. 2000; Leskinen et al. 2011); similarly, fully automated vertical-beam entomological radars located in a region prone to locust outbreaks have been envisaged as fulfilling a ‘sentinel’ role (Drake and Wang 2013).
In conclusion, we note that in over four decades of radar entomological research, much of the ‘phenomenology’ of high-altitude insect interactions with wind and weather are reasonably well understood. The outstanding uncertainties concern the mechanisms for detection of wind velocity at attitude, and the maintenance of seasonal orientation directions, in nocturnal insect migrants. There is also a need for more radar-derived information on the termination stages of migration: there have been very few observations of migrants in the process of descent and landing (Chap. 10 in Drake and Reynolds 2012). Progress is currently being made in quantifying the interaction of insects with other animals (particularly insectivores) high in the air, as indicated above. Finally, just as the first deployment of scanning radars for entomological purposes nearly 50 years ago revealed previously unsuspected phenomena, new perspectives on insect movement can be expected to emerge through the application of novel radar, optical, and data-logging technologies that are now becoming available (see Chap. 9).
References
Achtemeier GL (1991) The use of insects as tracers for ‘clear-air’ boundary-layer studies by Doppler radar. J Atmos Ocean Technol 8:746–765
Achtemeier GL (1992) Grasshopper response to rapid vertical displacements within a ‘clear air’ boundary layer as observed by Doppler radar. Environ Entomol 21:921–938
Alerstam T, Chapman JW, Bäckman J et al (2011) Convergent patterns of long-distance nocturnal migration in noctuid moths and passerine birds. Proc R Soc B Biol Sci 278:3074–3080
Anderson RC (2009) Do dragonflies migrate across the western Indian Ocean? J Trop Ecol 25:347–348
Aralimarad P, Reynolds AM, Lim KS, Reynolds DR, Chapman JW (2011) Flight altitude selection increases orientation performance in high-flying nocturnal insect migrants. Anim Behav 82:1221–1225
Bauer S, Hoye BJ (2014) Migratory animals couple biodiversity and ecosystem functioning worldwide. Science 344:1242552. https://doi.org/10.1126/science.1242552
Bauer S, Chapman JW, Reynolds DR et al (2017) From agricultural benefits to aviation safety: realizing the potential of continent-wide radar networks. BioScience 67(10):912–918
Beerwinkle KR, Lopez JD, Witz JA, Schleider PG, Eyster RS, Lingren PD (1994) Seasonal radar and meteorological observations associated with nocturnal insect flight at altitudes to 900 meters. Environ Entomol 23:676–683
Bell JR, Aralimarad P, Lim K-S, Chapman JW (2013) Predicting insect migration density and speed in the daytime convective boundary layer. PLoS One 8(1):e54202
Bluestein HB, Snyder JC, Thiem KJ, Wienhoff ZB, Reif D, Turner D (2014) Doppler-radar observations of a prefrontal wind-shift line in the Southern Plains of the U.S. Poster MES.P01 presented at ERAD2014: Eighth European Conference on Radar in Meteorology and Hydrology, 1–5 Sept 2014, Garmisch-Partenkirchen, Germany. DLR- Institut für Physik der Atmosphäre, Oberpfaffenhofen, Germany (Extended abstract)
Boulanger Y, Fabry F, Kilambi A et al (2017) The use of weather surveillance radar and high-resolution three dimensional weather data to monitor a spruce budworm mass exodus flight. Agric For Meteorol 234:127–135
Brévault T, Achaleke J, Sougnabé SP, Vaissayre M (2008) Tracking pyrethroid resistance in the polyphagous bollworm, Helicoverpa armigera (Lepidoptera: Noctuidae) in the shifting landscape of a cotton growing area. Bull Entomol Res 98:565–573
Browning KA (1981) Ingestion of insects by intense convective updraughts. Antenna 5:14–17
Browning KA, Nicol JC, Marsham JH, Rogberg P, Norton EG (2011) Layers of insect echoes near a thunderstorm and implications for the interpretation of radar data in terms of airflow. Q J R Meteorol Soc 137:723–735
Campistron B (1975) Characteristic distributions of angel echoes in the lower atmosphere and their meteorological implications. Bound Layer Meteorol 9:411–426
Chandra AS, Kollias P, Giangrande SE, Klein SA (2010) Long-term observations of the convective boundary layer using insect radar returns at the SGP ARM climate research facility. J Climate 23:5699–5714
Chapman JW, Reynolds DR, Smith AD, Riley JR, Pedgley DE, Woiwod IP (2002a) High-altitude migration of the diamondback moth Plutella xylostella to the U.K.: a study using radar, aerial netting and ground trapping. Ecol Entomol 27:641–650
Chapman JW, Smith AD, Woiwod IP, Reynolds DR, Riley JR (2002b) Development of vertical-looking radar technology for monitoring insect migration. Comput Electron Agric 35:95–110
Chapman JW, Reynolds DR, Smith AD, Smith ET, Woiwod IP (2004) An aerial netting study of insects migrating at high-altitude over England. Bull Entomol Res 94:123–136
Chapman JW, Reynolds DR, Brooks SJ, Smith AD, Woiwod IP (2006) Seasonal variation in the migration strategies of the green lacewing Chrysoperla carnea species complex. Ecol Entomol 31:378–388
Chapman JW, Reynolds DR, Hill JK, Sivell D, Smith AD, Woiwod IP (2008a) A seasonal switch in compass orientation in a high-flying migrant moth. Curr Biol 18:R908–R909
Chapman JW, Reynolds DR, Mouritsen H et al (2008b) Wind selection and drift compensation optimize migratory pathways in a high-flying moth. Curr Biol 18:514–518
Chapman JW, Nesbit RL, Burgin LE, Reynolds DR, Smith AD, Middleton DR, Hill JK (2010) Flight orientation behaviors promote optimal migration trajectories in high-flying insects. Science 327:682–685
Chapman JW, Drake VA (2017) Insect migration. Elsevier, In Reference Module in Life Sciences. Oxford. http://www.sciencedirect.com/science/article/pii/B9780128096338012486
Chapman JW, Drake VA, Reynolds DR (2011a) Recent insights from radar studies of insect flight. Annu Rev Entomol 56:337–356
Chapman JW, Klaassen RHG, Drake VA et al (2011b) Animal orientation strategies for movement in flows. Curr Biol 21:R861–R870
Chapman JW, Bell JR, Burgin LE, Reynolds DR, Pettersson LB, Hill JK, Bonsall MB, Thomas JA (2012) Seasonal migration to high latitudes results in major reproductive benefits in an insect. Proc Natl Acad Sci USA 109(37):14924–14929
Chapman JW, Lim KS, Reynolds DR (2013) The significance of midsummer movements of Autographa gamma: Implications for a mechanistic understanding of orientation behavior in a migrant moth. Curr Zool 59:360–370
Chapman JW, Nilsson C, Lim KS, Bäckman J, Reynolds DR, Alerstam T, Reynolds AM (2015a) Detection of flow direction in high-flying insect and songbird migrants. Curr Biol 25(17):R733–R752
Chapman JW, Reynolds DR, Wilson K (2015b) Long-range seasonal migration in insects: mechanisms, evolutionary drivers and ecological consequences. Ecol Lett 18:287–302
Chapman JW, Nilsson C, Lim KS, Bäckman J, Reynolds DR, Alerstam T (2016) Adaptive strategies in nocturnally migrating insects and songbirds: contrasting responses to wind. J Anim Ecol 85:115–124
Chen R-L, Bao X-Z, Drake VA, Farrow RA, Wang S-Y, Sun Y-J, Zhai B-P (1989) Radar observations of the spring migration into northeastern China of the oriental armyworm moth, Mythimna separata and other insects. Ecol Entomol 14:149–162
Chilson PB, Bridge E, Frick WF, Chapman JW, Kelly JF (2012a) Radar aeroecology: exploring the movements of aerial fauna through radio-wave remote sensing. Biol Lett 8:698–701
Chilson PB, Frick WF, Kelly JF, Howard KW, Larkin RP, Diehl RH, Westbrook JK, Kelly TA, Kunz TH (2012b) Partly cloudy with a chance of migration: weather, radars, and aeroecology. Bull Am Meteorol Soc 93:669–686
Cleveland CJ, Betke M, Federico P et al (2006) Economic value of the pest control service provided by Brazilian free-tailed bats in south-central Texas. Front Ecol Environ 4:238–243
Contreras RF, Frasier SJ (2008) High-resolution observations of insects in the atmospheric boundary layer. J Atmos Ocean Technol 25:2176–2187
Deveson ED, Walker PW (2005) Not a one-way trip: historical distribution data for Australian plague locusts supports frequent seasonal exchange migrations. J Orthop Res 14:95–109
Dickerson AK, Shankles PG, Madhavan NM, Hu DL (2012) Mosquitoes survive raindrop collisions by virtue of their low mass. Proc Natl Acad Sci USA 109(25):9822–9827
Dingle H (2014) Migration: the biology of life on the move, 2nd edn. Oxford University Press, Oxford
Dingle H, Drake VA (2007) What is migration? BioScience 57:113–121
Dokter AM, Liechti F, Stark H, Delobbe L, Tabary P, Holleman I (2011) Bird migration flight altitudes studied by a network of operational weather radars. J R Soc Interface 8:30–43
Drake VA (1985) Solitary wave disturbances of the nocturnal boundary layer revealed by radar observations of migrating insects. Bound Layer Meteorol (3):269–286
Drake VA (2014) Estimation of unbiased insect densities and density profiles with vertically pointing entomological radars. Int J Remote Sens 35(13):4630–4654
Drake VA, Farrow RA (1983) The nocturnal migration of the Australian plague locust, Chortoicetes terminifera (Walker) (Orthoptera: Acrididae): quantitative radar observations of a series of northward flights. Bull Entomol Res 73:567–585
Drake VA, Farrow RA (1985) A radar and aerial-trapping study of an early spring migration of moths (Lepidoptera) in inland New South Wales. Aust J Ecol:10223–10235
Drake VA, Farrow RA (1988) The influence of atmospheric structure and motions on insect migration. Annu Rev Entomol 33:183–210
Drake VA, Gatehouse AG (eds) (1995) Insect migration: tracking resources through space and time. Cambridge University Press, Cambridge
Drake VA, Reynolds DR (2012) Radar entomology: observing insect flight and migration. CABI, Wallingford
Drake VA, Wang HK (2013) Recognition and characterization of migratory movements of Australian plague locusts, Chortoicetes terminifera, with an insect monitoring radar. J Appl Remote Sens 7(1):075095, 17pp
Drake VA, Helm KF, Readshaw JL, Reid DG (1981) Insect migration across Bass Strait during spring: a radar study. Bull. Entomol. Res.71:449–466
Drake VA, Gregg PC, Harman IT, Wang H-K, Deveson ED, Hunter, DM, Rochester WA (2001) Characterizing insect migration systems in inland Australia with novel and traditional methodologies. In: Woiwod IP, Reynolds DR, Thomas CD (eds) Insect movement: mechanisms and consequences. CAB International, Wallingford, pp 207–233
Dudley R (2000) The biomechanics of insect flight: form, function, evolution. Princeton University Press, Princeton, NJ
Farrow RA (1975) Offshore migration and the collapse of outbreaks of the Australian plague locust Chortoicetes terminifera Walk. in south-east Australia. Aust J Zool 234:569–595
Feng H-Q, Wu X-F, Wu B, Wu K-M (2009) Seasonal migration of Helicoverpa armigera (Lepidoptera: Noctuidae) over the Bohai Sea. J Econ Entomol 102:95–104
Freeman B (2003) A fallout of black witches (Ascalapha odorata) associated with Hurricane Claudette. News Lepid Soc 43(3):71
Geerts B, Miao Q (2005) The use of millimeter Doppler radar echoes to estimate vertical air velocities in the fair-weather convective boundary layer. J Atmos Ocean Technol 22:225–246
Geerts B, Damiani R, Haimov S (2006) Finescale vertical structure of a cold front as revealed by an airborne Doppler radar. Mon Weather Rev 134:251–271
Green K (2011) The transport of nutrients and energy into the Australian Snowy Mountains by migrating Bogong moths Agrotis infusa. Austral Ecol 36:25–34
Greenbank DO, Schaefer GW, Rainey RC (1980) Spruce budworm (Lepidoptera: Tortricidae) moth flight and dispersal: new understanding from canopy observations, radar, and aircraft. Mem Entomol Soc Can 110:1–49
Hardy KR, Ottersten H (1969) Radar investigations of convective patterns in the clear atmosphere. J Atmos Sci 26:666–672
Hein AM, Hou C, Gillooly JF (2012) Energetic and biomechanical constraints on animal migration distance. Ecol Lett 15:104–110
Hobbs SE, Wolf WW (1989) An airborne radar technique for studying insect migration. Bull Entomol Res 79:693–704
Holdo RM, Holt RD, Sinclair ARE, Godley BJ, Thirgood S (2011) Migration impacts on communities and ecosystems: empirical evidence and theoretical insights. In: Milner-Gulland EJ, Fryxell JM, Sinclair ARE (eds) Animal migrations: a synthesis. Oxford University Press, Oxford, pp 131–143
Hu G, Lim KS, Reynolds DR, Reynolds AM, Chapman JW (2016a) Wind-related orientation patterns in diurnal, crepuscular and nocturnal high-altitude insect migrants. Front Behav Neurosci 10: article 32, 8pp
Hu G, Lim KS, Horvitz N, Clark SJ, Reynolds DR, Sapir N and Chapman JW (2016b) Mass seasonal bioflows of high-flying insect migrants. Science 354: 1584–1587
Isard SA, Gage SH (2001) Flow of life in the atmosphere: an airscape approach to understanding invasive organisms. Michigan State University Press, East Lansing
Irwin ME, Thresh JM (1988) Long range aerial dispersal of cereal aphids as virus vectors in North America. Philos Trans R Soc Lond B 321:421–446
Isard SA, Irwin ME, Hollinger SE (1990) Vertical distribution of aphids (Homoptera: Aphididae) in the planetary boundary layer. Environ Entomol 19:1473–1484
Isard SA, Kristovich DAR, Gage SH et al (2001) Atmospheric motion systems that influence the redistribution and accumulation of insects on the beaches of the Great Lakes in North America. Aerobiologia 17:275–291
Jeffries DL, Chapman J, Roy HE, Humphries S, Harrington R et al (2013) Characteristics and drivers of high-altitude ladybird flight: insights from vertical-looking entomological radar. PLoS One 8(12):e82278
Johnson CG (1969) Migration and dispersal of insects by flight. Methuen, London
Johnson SJ (1995) Insect migration in North America: synoptic-scale transport in a highly seasonal environment. In: Drake VA, Gatehouse AG (eds) Insect migration: tracking resources through space and time. Cambridge University Press, Cambridge, pp 31–66
Joyce RJV (1983) Aerial transport of pests and pest outbreaks. EPPO Bull 13:111–119
Kelly JF, Horton KG (2016) Toward a predictive macrosystems framework for migration ecology. Glob Ecol Biogeogr 25(10):1159–1165. https://doi.org/10.1111/geb.12473
Kelly JF, Shipley JR, Chilson PB, Howard KW, Frick WF, Kunz TH (2012) Quantifying animal phenology in the continental scale using NEXRAD weather radars. Ecosphere 3:article 16. https://doi.org/10.1890/ES11-00257.1
Kelly JF, Bridge ES, Frick WF, Chilson PB (2013) Ecological energetics of an abundant aerial insectivore, the purple martin. PLoS One 8(9):e76616
Kennedy JS (1985) Migration: behavioral and ecological. In: Rankin MA (ed) Migration: mechanisms and adaptive significance. Contributions in Marine Science 27 (Supplement). Marine Science Institute, University of Texas at Austin, Port Aransas, TX, pp 5–26
Klaassen RH, Hake M, Strandberg R et al (2014) When and where does mortality occur in migratory birds? Direct evidence from long-term satellite tracking of raptors. J Anim Ecol 83(1):176–184
Knupp KR (2006) Observational analysis of a gust front to bore to solitary wave transition within an evolving nocturnal boundary layer. J Atmos Sci 63:2016–2035
Koch GJ (2006) Using a Doppler light detection and ranging (lidar) system to characterize an atmospheric thermal providing lift for soaring raptors. J Field Ornithol 77:315–318
Krauel JJ, Westbrook JK, McCracken GF (2015) Weather-driven dynamics in a dual-migrant system: moths and bats. J Anim Ecol 84:604–614
Landry JS, Parrott L (2016) Could the lateral transfer of nutrients by outbreaking insects lead to consequential landscape-scale effects? Ecosphere 7:e01265
Lang TJ, Rutledge SA, Stith JL (2004) Observations of quasi-symmetric echo patterns in clear air with the CSU-CHILL polarimetric radar. J Atmos Ocean Technol 21:1182–1189
Leskinen M, Markkula I, Koistinen J, Pylkkö P, Ooperi S, Siljamo P, Ojanen H, Raiskio S, Tiilikkala K (2011) Pest insect immigration warning by an atmospheric dispersion model, weather radars and traps. J Appl Entomol 135:55–67
Leskinen M, Rojas L, Mikkola K (2012) Weather radar observations of Underwing moth immigrations to Finland. Poster at ERAD 2012—the Seventh European Conference on Radar in Meteorology and Hydrology, Toulouse, France, 24–29 June 2012. Météo France, Toulouse. http://www.meteo.fr/cic/meetings/2012/ERAD/extended_abs/NMUR_215_ext_abs.pdf
Lewis T, Taylor LR (1964) Diurnal periodicity of flight by insects. Trans R Entomol Soc Lond 116:393–479
Lothon M, Campistron B, Jacoby-Koaly S, Bénech B, Lohou F, Girard-Ardhuin F (2002) Comparison of radar reflectivity and vertical velocity observed with a scannable C-band radar and two UHF profilers in the lower troposphere. J Atmos Ocean Technol 19:899–910
Lothon M, Campistron B, Chong M, Couvreux F, Guichard F, Rio C, Williams E (2011) Life cycle of a mesoscale circular gust front observed by a C-Band Doppler radar in West Africa. Mon Weather Rev 139:1370–1388
Luke EP, Kollias P, Johnson KL, Clothiaux EE (2008) A technique for the automatic detection of insect clutter in cloud radar returns. J Atmos Ocean Technol 25:1498–1513
McCracken GF, Gillam EH, Westbrook JK, Lee Y, Jensen M, Balsley B (2008) Brazilian free-tailed bats (Tadarida brasiliensis: Molossidae, Chiroptera) at high altitude: links to migratory insect populations. Integr Comp Biol 48:107–118
McCracken GF, Westbrook JK, Brown VA et al (2012) Bats track and exploit changes in insect pest populations. PLoS One 7(8):e43839
McNeil JN (1987) The true armyworm, Pseudaletia unipuncta—a victim of the pied piper or a seasonal migrant. Insect Sci Appl 8:591–597
Melnikov V, Leskinen M, Koistinen J (2014a) Doppler velocities at orthogonal polarizations in radar echoes from insects and birds. IEEE Geosci Remote Sens Lett 11(3):592–596
Melnikov V, Zrnic D, Burgess D, Mansell E (2014b) Observations of hail cores of tornadic thunderstorms with three polarimetric radars. Presentation at 94th American Meteorological Society Annual Meeting, 2–6 Feb, 2014, Atlanta, 16pp. https://ams.confex.com/ams/94Annual/webprogram/Paper233038.html
Menu S, Gauthier G, Reed A (2005) Survival of young greater snow geese (Chen caerulescens atlantica) during fall migration. Auk 122:479–496
Mikkola K (1986) Direction of insect migrations in relation to the wind. In: Danthanarayana W (ed) Insect flight: dispersal and migration. Springer-Verlag, Berlin, pp 152–171
Ming J-G, Jin H, Riley JR et al (1993) Autumn southward ‘return’ migration of the mosquito Culex tritaeniorhynchus in China. Med Vet Entomol 7:323–327
Neumann HH, Mukammal EI (1981) Incidence of mesoscale convergence lines as input to spruce budworm control strategies. Int J Biometeorol 25:175–187
Nieminen M, Leskinen M, Helenius J (2000) Doppler radar detection of exceptional mass migration of aphids into Finland. Int J Biometeorol 44:172–181
Orlanski I (1975) A rational subdivision of scales for atmospheric processes. Bull Am Meteorol Soc 56:527–530
Otuka A (2013) Migration of rice planthoppers and their vectored re-emerging and novel rice viruses in East Asia. Front Microbiol 4:article 309. https://doi.org/10.3389/fmicb.2013.00309.
Otuka A, Dudhia J, Watanabe T, Furuno A (2005) A new trajectory analysis method for migratory planthoppers, Sogatella furcifera (Horváth) (Homoptera: Delphacidae) and Nilaparvata lugens (Stål), using an advanced weather forecast model. Agric For Entomol 7:1–9
Otuka A, Matsumura M, Sanada-Morimura S, Takeuchi H, Watanabe T, Ohtsu R, Inoue H (2010) The 2008 overseas mass migration of the small brown planthopper, Laodelphax striatellus, and subsequent outbreak of rice stripe disease in western Japan. Appl Entomol Zool 45:259–266
Pair SD, Raulston JR, Westbrook JR, Wolf WW, Sparks AN, Schuster MF (1987) Development and production of corn earworm and fall armyworm in the Texas High Plains: evidence for the reverse fall migration. Southwestern Entomologist 12:89–99
Pedgley DE (ed) (1981) Desert Locust forecasting manual, vols 1 and 2. Centre for Overseas Pest Research, London
Pedgley DE (1982) Windborne pests and diseases: Meteorology of airborne organisms. Ellis Horwood, Chichester
Pedgley DE, Reynolds DR, Tatchell GM (1995) Long-range insect migration in relation to climate and weather: Africa and Europe. In: Drake VA, Gatehouse AG (eds) Insect migration: tracking resources through space and time. Cambridge University Press, Cambridge, pp 3–29
Qi H, Jiang C, Zhang Y, Yang X, Cheng D (2014) Radar observations of the seasonal migration of brown planthopper (Nilaparvata lugens Stal) in Southern China. Bull Entomol Res 104:731–741
Rainey RC (1989) Migration and meteorology: flight behaviour and the atmospheric environment of locusts and other migrant pests. Oxford University Press, Oxford
Rennie SJ (2012) Doppler weather radar in Australia. CAWCR (Centre for Australian Weather and Climate Research) Technical Report 055. Bureau of Meteorology, Melbourne
Rennie SJ (2014) Common orientation and layering of migrating insects in southeastern Australia observed with a Doppler weather radar. Meteorol Appl 21:218–229
Reynolds DR, Mukhopadhyay S, Riley JR, Das BK, Nath PS, Mandal SK (1999) Seasonal variation in the windborne movement of insect pests over northeast India. Int J Pest Manag 45:195–205
Reynolds DR, Chapman JW, Edwards AS, Smith AD, Wood CR, Barlow JF et al (2005) Radar studies of the vertical distribution of insects migrating over southern Britain: the influence of temperature inversions on nocturnal layer concentrations. Bull Entomol Res 95:259–274
Reynolds DR, Chapman JW, Harrington R (2006) The migration of insect vectors of plant and animal viruses. Adv Virus Res 67:453–517
Reynolds DR, Smith AD, Chapman JW (2008) A radar study of emigratory flight and layer formation by insects at dawn over southern Britain. Bull Entomol Res 98:35–52
Reynolds AM, Reynolds DR, Riley JR (2009) Does a ‘turbophoretic’ effect account for layer concentrations of insects migrating in the stable night-time atmosphere? J R Soc Interface 6:87–95
Reynolds AM, Reynolds DR, Smith AD, Chapman JW (2010) A single wind-mediated mechanism explains high-altitude ‘non-goal oriented’ headings and layering of nocturnally migrating insects. Proc R Soc B Biol Sci 277:765–772
Reynolds DR, Reynolds AM, Chapman JW (2014) Non-volant modes of migration in terrestrial arthropods. Anim Migr 2:8–28
Reynolds AM, Reynolds DR, Sane SP, Hu G, Chapman JW (2016) Orientation in high-flying migrant insects in relation to flows: mechanisms and strategies. Philos Trans R Soc B: 371(1704):20150392
Riley JR, Reynolds DR, Farmery MJ (1983) Observations of the flight behaviour of the armyworm moth, Spodoptera exempta, at an emergence site using radar and infra-red optical techniques. Ecol Entomol 8:395–418
Riley JR, Reynolds DR, Farrow RA (1987) The migration of Nilaparvata lugens (Stal) (Delphacidae) and other Hemiptera associated with rice during the dry season in the Philippines: a study using radar, visual observations, aerial netting and ground trapping. Bull Entomol Res 77:145–169
Riley JR, Cheng X-N, Zhang X-X, Reynolds DR, Xu G-M, Smith AD, Cheng J-Y, Bao A-D, Zhai B-P (1991) The long-distance migration of Nilaparvata lugens (Stal) (Delphacidae) in China: radar observations of mass return flight in the autumn. Ecol Entomol 16:471–489
Riley JR, Reynolds DR, Smith AD, Rosenberg LJ, Cheng X-N, Zhang X-X, Xu G-M, Cheng J-Y, Bao A-D, Zhai B-P, Wang H-K (1994) Observations on the autumn migration of Nilaparvata lugens (Homoptera: Delphacidae) and other pests in east central China. Bull Entomol Res 84:389–402
Riley JR, Reynolds DR, Smith AD, Edwards AS, Zhang X-X, Cheng X-N, Wang H-K, Cheng J-Y, Zhai B-P (1995) Observations of the autumn migration of the rice leaf roller Cnaphalocrocis medinalis (Lepidoptera: Pyralidae) and other moths in eastern China. Bull Entomol Res 85:397–414
Rose DJW, Page WW, Dewhurst CF et al (1985) Downwind migration of the African armyworm moth, Spodoptera exempta, studied by mark-and-recapture and by radar. Ecol Entomol 10:299–313
Rose DJW, Dewhurst CF, Page WW (2000) The African armyworm handbook: the status, biology, ecology, epidemiology and management of Spodoptera exempta (Lepidotera: Noctuidae), 2nd edn. Natural Resources Institute, Chatham
Russell RW, Wilson JW (1996) Aerial plankton detected by radar. Nature 381:200–201
Russell RW, Wilson JW (1997) Radar-observed ‘fine lines’ in the optically clear boundary layer: reflectivity contribution from aerial plankton and its predators. Bound Layer Meteorol 82:235–262
Russell RW, Wilson JW (2001) Spatial dispersion of aerial plankton over east-central Florida: aeolian transport and coastline concentrations. Int J Remote Sens 22:2071–2082
Sauvageot H, Despaux G (1996) The clear-air coastal vespertine radar bands. Bull Am Meteorol Soc 77:673–681
Schaefer GW (1976) Radar observations of insect flight. In: Rainey RC (ed) Insect flight (Symposia of the Royal Entomological Society no. 7). Blackwell Scientific, Oxford, pp 157–197
Scott KD, Lawrence N, Lange CL, Scott LJ, Wilkinson KS, Merritt MA, Miles M, Murray D, Graham GC (2005) Assessing moth migration and population structuring in Helicoverpa armigera (Lepidoptera: Noctuidae) at the regional scale: example from the Darling Downs, Australia. J Econ Entomol 98:2210–2219
Shamoun-Baranes J, Alves JA, Bauer S et al. (2014) Continental-scale radar monitoring of the aerial movements of animals. Mov Ecol 2: article 9, 6pp
Shashar N, Sabbah S, Aharoni N (2005) Migrating locusts can detect polarized reflections to avoid flying over the sea. Biol Lett 1:472–475
Sillett TS, Holmes RT (2002) Variation in survivorship of a migratory songbird throughout its annual cycle. J Anim Ecol 71:296–308
Simpson JE (1994) Sea breeze and local winds. Cambridge University Press, Cambridge
Simpson JE (1997) Gravity currents in the environment and the laboratory, 2nd edn. Cambridge University Press, Cambridge
Smith AD, Reynolds DR, Riley JR (2000) The use of vertical-looking radar to continuously monitor the insect fauna flying at altitude over southern England. Bull Entomol Res 90:265–277
Ssymank A, Kearns CA, Pape T, Thompson FC (2008) Pollinating flies (Diptera): a major contribution to plant diversity and agricultural production. Biodiversity 9(1–2):86–89
Stefanescu C, Alarcón M, Àvila A (2007) Migration of the painted lady butterfly, Vanessa cardui, to north-eastern Spain is aided by African wind currents. J Anim Ecol 76:888–898
Stefanescu C, Páramo F, Åkesson S et al (2013) Multi-generational long-distance migration of insects: studying the painted lady butterfly in the Western Palaearctic. Ecography 36:474–486
Stefanescu C, Soto DX, Talavera G, Vila R, Hobson KA (2016) Long-distance autumn migration across the Sahara by painted lady butterflies: exploiting resources in the tropical savannah. Biol Lett 12:20160561
Talavera G, Vila R (2017) Discovery of mass migration and breeding of the painted lady butterfly Vanessa cardui in the Sub-Sahara: the Europe–Africa migration revisited. Biol J Linn Soc 120:274–285
Tanaka K, Matsumura M (2000) Development of virulence to resistant rice varieties in the brown planthopper, Nilaparvata lugens (Homoptera: Delphacidae), immigrating into Japan. Appl Entomol Zool 35:529–533
Taylor LR (1974) Insect migration, flight periodicity and the boundary layer. J Anim Ecol 43:225–238
Taylor LR (1985) An international standard for the synoptic monitoring and dynamic mapping of migrant pest aphid populations. In: MacKenzie DR, Barfield CS, Kennedy GC, Berger RD, Taranto DJ (eds) The movement and dispersal of agriculturally important biotic agents. Claitor’s Publishing Division, Baton Rouge, pp 337–380
Tojo S, Ryuda M, Fukuda T, Matsunaga T, Choi D-R, Otuka A (2013) Overseas migration of the common cutworm, Spodoptera litura (Lepidoptera: Noctuidae), from May to mid-July in East Asia. Appl Entomol Zool 48:131–140
Tucker MR (1997) Satellite-derived rainstorm distribution as an aid to forecasting African armyworm outbreaks. Weather 52:204–212
Van Den Broeke MS (2013) Polarimetric Radar observations of biological scatterers in Hurricanes Irene (2011) and Sandy (2012). J Atmos Ocean Technol 30:2754–2767
Wainwright CE, Stepanian PM, Reynolds DR, Reynolds AM (2017) The movement of small insects in the convective boundary layer: linking patterns to processes. Sci Rep 7(1):article 5438
Ward SA, Leather SR, Pickup J, Harrington R (1998) Mortality during dispersal and the cost of host-specificity in parasites: how many aphids find hosts? J Anim Ecol 67:763–773
Warrant E, Frost B, Green K, Mouritsen H, Dreyer D, Adden A, Brauburger K, Heinze S (2016) The Australian bogong moth Agrotis infusa: a long-distance nocturnal navigator. Front Behav Neurosci 10:77
Weckwerth T, Horst TW, Wilson JW (1999) An observational study of the evolution of horizontal convective rolls. Mon Weather Rev 127:2160–2179
Weiss CC, Bluestein HB, Pazmany AL (2006) Finescale radar observations of the 22 May 2002 dryline during the International H2O Project (IHOP). Mon Weather Rev 134:273–293
Westbrook JK (2008) Noctuid migration in Texas within the nocturnal aeroecological boundary layer. Integr Comp Biol 48:99–106
Westbrook JK, Eyster RS (2017) Doppler weather radar detects emigratory flights of noctuids during a major pest outbreak. Remote Sensing Applications: Society and Environment 8:64–70
Westbrook JK, Eyster RS, Wolf WW (2014) WSR-88D Doppler radar detection of corn earworm moth migration. Int J Biometeorol 58:931–940
Westbrook JK, Nagoshi RN, Meagher RL, Fleischer SJ, Jairam S (2016) Modeling seasonal migration of fall armyworm moths. Int J Biometeorol 60:255–267
Wolf WW, Westbrook JK, Sparks AN (1986) Relationship between radar entomological measurements and atmospheric structure in south Texas during March and April 1982. In: Sparks AN (ed) Long-range migration of moths of agronomic importance to the United States and Canada: Specific examples of occurrence and synoptic weather patterns conducive to migration. ARS-43. United States Department of Agriculture, Agricultural Research Service, Washington, DC, pp 84–97
Wood CR, O’Connor EJ, Hurley RA, Reynolds DR, Illingworth AJ (2009) Cloud-radar observations of insects in the UK convective boundary layer. Meteorol Appl 16:491–500
Wood CR, Clark SJ, Barlow JF, Chapman JW (2010) Layers of nocturnal insect migrants at high-altitude: the influence of atmospheric conditions on their formation. Agric For Entomol 12:113–121
Acknowledgements
We acknowledge the support provided by COST—European Cooperation in Science and Technology through the Action ES1305 ‘European Network for the Radar Surveillance of Animal Movement’ (ENRAM). JWC acknowledges support of Rothamsted Research, where he was employed during much of the drafting process; Rothamsted Research is a national institute of bioscience strategically funded by the UK Biotechnology and Biological Sciences Research Council (BBSRC).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Reynolds, D.R., Chapman, J.W., Drake, V.A. (2017). Riders on the Wind: The Aeroecology of Insect Migrants. In: Chilson, P., Frick, W., Kelly, J., Liechti, F. (eds) Aeroecology. Springer, Cham. https://doi.org/10.1007/978-3-319-68576-2_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-68576-2_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-68574-8
Online ISBN: 978-3-319-68576-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)