Abstract
Carcinoma of the urinary bladder exhibits a wide spectrum of morphologies that is characteristic of this disease. Urothelial carcinoma (UCa) represents the most common tumor that develops in the urinary bladder and it is well established that there is remarkable propensity for divergent differentiation in this disease. A number of well-documented variant histologies are commonly encountered in approximately one third of invasive UCa (Amin, Mod Pathol 22: S96–S118, 2009; Linder, et al., J Urol 190: 1692–6, 2013; Shah, et al., Urol Oncol 31: 1650–5, 2013; Moch, et al., World health organization classification of tumours, 2016). These include squamous, glandular, micropapillary, sarcomatoid, small cell/neuroendocrine, clear cell, lymphoepithelioma-like, and plasmacytoid types among others. Pure squamous cell carcinoma and adenocarcinoma can also occur in the bladder but are generally rare. In this chapter we will provide an update on the recent molecular developments with particular focus on those related to the variant histologies of bladder cancer.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Urothelial carcinoma
- Divergent differentiation
- Variant histology
- Squamous
- Glandular
- Small cell
- Neuroendocrine
- Plasmacytoid
- Micropapillary
- Nested
- Microcystic
- Sarcomatoid
- Adenocarcinoma
Introduction
Urothelial carcinoma (UCa) is the most common type of bladder cancer but other rare forms of cancer can rarely develop in the bladder including pure squamous cell carcinoma, adenocarcinoma, and small cell carcinoma. UCa is further subdivided into the conventional subtype (usual form) or one of numerous variant histologies. Historically, bladder cancer subtypes and variants of UCa were primarily subdivided based on morphological features. However, recent developments in our understanding of the genomic profiles of these entities have led to a better understanding of the molecular features associated with a subset of these lesions. This chapter will focus specifically on the diagnosis and molecular features associated with the major subtypes of bladder cancer and a subset of UCa variants that are not addressed in other chapters in this text.
Variants of Urothelial Carcinoma
Urothelial Carcinoma with Divergent Differentiation
The most common divergent differentiation in UCa is squamous and glandular differentiation. These two components are typically identified in association with components of the usual urothelial carcinoma.
Squamous differentiation (SqD) is the most common variant histology identified in UCa occurring in up to 40% of cases [1, 2]. SqD in this setting requires the presence of intercellular bridges and/or keratinization (Fig. 4.1). SqD may also be associated with other divergent histologies within an otherwise “usual” UCa, especially in high-grade and high-stage tumors. The term squamous cell carcinoma (SCC) of the bladder should be reserved for tumors that exhibit pure or nearly pure squamous features [2, 3]. SqD is not limited to UCa of the bladder as such morphology can also be seen in UCa of the upper tract [4].
Thorough and careful light microscopic evaluation is the best way to identify squamous lesions but sometimes such distinction may be difficult. There have been a number of markers proposed to aid in this situation but in most times, such markers work best in areas where the light microscopic features are straightforward and may be less helpful in difficult or less straightforward cases.
Both urothelial and squamous areas express many of the same proteins such as p63 and the high molecular weight cytokeratin (HMWK) at high rates [5,6,7,8,9]. Some markers have a tendency to preferentially stain squamous areas such as CK5/6 and CK14 [10, 11]. A recent study reported a novel panel of markers specific for squamous differentiation in a series of primary bladder squamous cell carcinoma and urothelial carcinoma with squamous differentiation that included MAC387, desmoglein-3, and TRIM29 [12]. These markers preferentially stained squamous cell carcinoma and squamous areas in urothelial carcinoma with squamous differentiation compared to the urothelial areas. Markers that are more likely to stain urothelial than squamous areas include uroplakins, GATA3, S100P, and CK20 [10, 11, 13,14,15,16,17,18,19,20]. It is important to keep in mind, however, that there remains to be some overlap in the expression of these markers in areas of urothelial and squamous features.
The association of human papillomavirus (HPV) and bladder cancer with squamous phenotype has been explored but most evidence points to lack of such association in the vast majority of cases. A few exceptions include patients with neurogenic bladders or those who required repeated catheterization, in which p16 and HPV in situ hybridization was detected in the majority of tumor cells [21, 22]. It is important to note that p16 expression may be seen in conventional urothelial carcinoma with or without squamous differentiation without association with HPV [23]. Expression of this marker is thus insufficient to establish the diagnosis of HPV-associated disease in the absence of HPV genomic integration in the tumor.
A number of studies on the molecular aspects of bladder cancer included cases of UCa with squamous differentiation [24,25,26,27]. These studies have revealed robust molecular subtypes of UCa with interesting patterns of gene expression. They all identified a subtype that is enriched with squamous histology. Tumors in this group showed overexpression of high molecular weight keratins (CK5, CK6, and CK14) and epidermal growth factor receptor (EFGR) as well as underexpression of markers of urothelial differentiation such as uroplakins, GATA3, FOXA1, and thrombomodulin. These studies, however, included samples with mixed squamous and urothelial components and as such did not provide a clear evidence to the exact mechanisms involved in the development of the squamous morphology in this setting.
In a separate study comparing the expression profiles of urothelial carcinoma and squamous cell carcinoma of the bladder, Hansel et al. [28] reported the presence of many similarly dysregulated genes and pathways between the two tumor types but there were also many genes that were preferentially dysregulated in the squamous cell carcinoma group particularly those related to squamous-specific morphology regardless of the site of origin (desmosomal complex, squamous epithelium related intermediate filaments, and squamous cornifying proteins).
Glandular differentiation is less common in urothelial carcinoma and the reported incidence is variable in different studies, which is likely related to the subjectivity and familiarity with identifying this variant histology or to selection or referral bias from the reporting institutions. The reported incidence ranges from 8 to 18% [1, 29,30,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,31]. The morphology of the glandular component in this setting resembles adenocarcinomas of other organs such as enteric/colonic adenocarcinoma, mucinous or a variety of mixed types (Fig. 4.2). There is limited literature on the molecular characteristics of glandular differentiation in UCa and they are likely to be overlapping with those of urothelial carcinoma as there is evidence that such tumors similarly harbor hotspot mutation in the TERT promoter region [32].
Nested and Microcystic Urothelial Carcinoma
These UCa variants are characterized by the presence of deceptively bland nests of invasive carcinoma that lack significant atypia or stromal reaction (Fig. 4.3). The original description of nested UCa included cases with small nests of invasive tumor but following recent reports, it has been expanded to include the recently described large nested variant and urothelial carcinoma with small tubules [33,34,35,36]. Another variant of urothelial carcinoma with bland morphologic features is microcystic UCa which is characterized by the presence of invasive medium-sized cystic structures with bland cytologic features that may show overlapping features with nested UCa [37, 38] (Fig. 4.3). The main challenge in diagnosing these entities is to distinguish them from benign proliferative urothelial conditions including von Brunn nest proliferation, nephrogenic adenoma, cystitis cystica, or inverted papilloma [39, 40]. These variants appear to show similar immunohistochemical features to conventional UCa. As of yet, there is no definitive molecular features associated with these entities to distinguish them from conventional UCa but there seems to be high rate of TERT promoter mutations in nested variant of urothelial carcinoma (including large nested variant) compared to the absence of such an alteration in benign mimickers [41], which may aid in establishing the diagnosis in challenging cases.
Plasmacytoid Urothelial Carcinoma
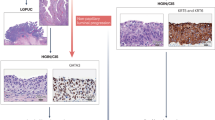
Plasmacytoid UCa is a rare but aggressive variant of UCa characterized by the presence of discohesive, individual cells with fair amount of cytoplasm and eccentrically located nuclei that resemble plasma cells [42,43,44]. In nearly all cases, there is a variable amount of tumor cells with intracytoplasmic vacuoles that give the cells a signet ring cell appearance (Fig. 4.4). This tumor typically follows an aggressive clinical course marked by advanced stage at presentation and association with a high relapse and mortality rate, and frequent peritoneal carcinomatosis despite the apparent initial response to chemotherapy [42,43,44,45,46]. The urothelial nature of this tumor type is supported by immunostains commonly used for urothelial differentiation such as CK7, p63, and uroplakins.
Plasmacytoid urothelial carcinoma with characteristic diffuse and discohesive growth pattern (a). Occasional signet ring cells also present. There is complete loss of E-cadherin expression in the invasive tumor (b, note E-cadherin retention in the overlying non-neoplastic urothelial mucosa). This tumor harbored a truncating CDH1mutation (L729 fs), the gene encoding for E-cadherin. The urothelial carcinoma in-situ component retains membranous E-cadherin expression (c, d)
Unlike other variants of urothelial carcinoma (including NOS), it has been recently shown that the presence of truncating mutations or promoter hypermethylation of CDH1 is the defining feature of plasmacytoid variant of bladder cancer [42]. Using whole exome and targeted sequencing, truncating somatic alterations in the CDH1 gene were identified in 84% of plasmacytoid carcinomas and were specific to this histologic variant (Fig. 4.4). Furthermore, all but one CDH1 wild-type plasmacytoid carcinoma exhibited CDH1 promoter hypermethylation and loss of E-cadherin expression. With the exception of CDH1 mutation, the genomic landscape of plasmacytoid carcinoma was similar to that of UCa, NOS with frequent mutations in chromatin modifying genes, cell cycle regulators, and PI3 kinase pathway alterations [42]. These results suggest that plasmacytoid and UCa-NOS bladder cancers likely evolve from a shared cell of origin. This was further supported by performing exon capture and deep sequencing of two adjacent portions of a bladder tumor which contained distinct regions of plasmacytoid and classic UCa. Both histologic regions shared mutations in CDKN1A (A45fs) and PIK3C2G (S48R), implying that these were early truncal alterations occurring within a common precursor cell. A CDH1 Y68fs mutation along with mutations in PTEN, NOTCH2, FAT4, and other genes were, however, unique to the plasmacytoid component [42].
Functional cell lines studies supported a significant role of CDH1 loss in promoting cell discohesion and stromal invasion, which could explain the higher incidence of both local recurrence and cancer-specific mortality as well as the higher rate of peritoneal spread than those with pure urothelial carcinoma. By performing Clustered Regularly Interspersed Palindromic Repeat (CRISPR)/Cas9-mediated knockout of CDH1 in two CDH1 wild-type urothelial carcinoma cell lines (RT4 and MGHU4), loss of E-cadherin expression resulted in increased migratory capability of MGHU4 cells. Additionally, both RT4 and MGHU4 CDH1-knockout cells displayed enhanced invasion across a Boyden chamber membrane. These results indicate that somatic loss-of-function mutations in CDH1, with consequent E-cadherin loss, lead to enhanced cellular migration and invasive properties in plasmacytoid carcinoma, characterized by marked cell discohesion and single cell infiltration. Notably, E-cadherin staining was absent in the invasive component of plasmacytoid variant tumors but was retained within in situ regions (Fig. 4.4). E-cadherin is a fundamental component of epithelial intercellular adhesions, and E-cadherin loss is implicated in tumor invasion and progression [47, 48], and prior studies have reported E-cadherin loss by immunohistochemistry is high percentage of plasmacytoid carcinoma [43, 49]. These observations indicate that E-cadherin loss, typically as a result of CDH1 mutation and less commonly as a result of CDH1 promoter methylation, is the molecular basis for the distinct pattern of local invasion and spread observed in patients with plasmacytoid bladder cancers. Moreover, in contrast to the germline CDH1 mutations that typify diffuse hereditary gastric cancers and a subset of lobular breast cancer, no germline CDH1 alterations were identified in the plasmacytoid variant bladder cancer [42].
Micropapillary Urothelial Carcinoma
This is a rare variant of urothelial carcinoma that is now increasingly appreciated but whose diagnosis still lacks high degree of interobserver concordance. This is even more problematic since many clinicians advise early cystectomy for this disease even in the absence of invasion into the muscularis propria [50]. The prevalence of this variant histology is variable ranging from 0.7 to 2.2% in the initial reports to as high as 8% in more recent studies, which may depend on the diagnostic threshold used to identify this variant [51, 52]. The characteristic morphologic appearance of this tumor is that of small tight clusters of tumor cells lacking true fibrovascular cores and present within lacunar spaces (Fig. 4.5) [53]. The basis behind this appearance is the “reverse orientation or polarization” of the basal and luminal aspects of the cells, as shown by electron microscopy as well as MUC1 expression, which is a glycoprotein normally located in the apical aspect of normal glandular epithelium and that is localized predominantly on the stroma-facing surface of the tumor cells in this entity [54, 55]. The end result is the lack of cohesion between tumor and stroma.
Micropapillary urothelial carcinoma . Clusters of high-grade tumor cells in lacunar spaces (right). This variant histology is commonly associated with HER2 overexpression and ERBB2 amplification as shown by Chromogenic in situ hybridization (CISH) where many copies of ERBB2 are detected (inset, brown signal)
Clinically, some studies suggested that conservative treatment for this disease is ineffective and advocated early cystectomy, even in T1 patients while other studies suggest that a more standard bladder sparing approach is reasonable in carefully selected patients in this setting [56, 57]. The application of chemotherapy for the treatment of micropapillary carcinoma showed mixed results with studies showing no benefit from neoadjuvant chemotherapy while others reported efficacy with aggressive systemic chemotherapy [58,59,60].
At the molecular level, higher rates of ERBB2 alterations occur in micropapillary carcinoma than in classic UCa, particularly HER2 amplification (Fig. 4.5) [61]. Additionally, a recent study demonstrated that ERBB2 amplification is associated with worsened cancer-specific survival in patients with micropapillary UC following radical cystectomy [62, 63]. Mutations in known hotspots in ERBB2 have also been recently reported in micropapillary carcinoma of the bladder [64] but it is not clear whether the frequency of these mutations is higher in this variant histology compared to classic UCa. In another recent study on gene expression profiling of micropapillary bladder cancer, the authors reported the presence of common downregulation of miR-296 and activation of chromatin-remodeling complex RUVBL1 in this disease but did not provide explanation for how these molecular events contribute to the development of micropapillary bladder cancer [65]. Interestingly ERBB2 was one of the genes that were upregulated in the majority of the studies tumors.
Sarcomatoid Urothelial Carcinoma
Sarcomatoid UCa (formerly referred to as “carcinosarcoma ”) is rare and is usually associated with advanced disease and poor outcomes [66]. This tumor is more common than primary sarcoma of the bladder which is the main differential diagnosis for this entity [1, 67]. Recognizable epithelial morphology is usually present in many of the cases and can represent urothelial, glandular, squamous, and/or small cell/neuroendocrine morphologies. The spectrum of morphologies of the sarcomatous elements is quite variable and may include spindle cell (not otherwise specified), myxoid, pseudoangiosarcomatous, and malignant fibrous histiocytoma-like undifferentiated features. In addition, heterologous elements (osseous, chondroid, etc.) may also be identified in a small subset of cases [1, 68]. It has been shown in earlier studies that the sarcomatous component in this tumor shares common clonal origin with the urothelial component [67]. In a recent study on sarcomatoid urothelial carcinoma, the authors report overexpression of markers of epithelial-to-mesenchymal transition in this tumor including vimentin, FoxC2, SNAIL, and ZEB1, as well as concurrent loss of E-cadherin and elevated N-cadherin expression [68]. Another study reported the presence of frequent TERT promoter mutation in sarcomatoid urothelial carcinoma of the upper urinary tract [69]. Similarly, we have encountered cases of sarcomatoid UCa harboring genetic alterations that are similar to those seen in UCa NOS such as mutations in TERT promoter, TP53 and chromatin-remodeling genes (unpublished data, Fig. 4.6).
An example of sarcomatoid urothelial carcinoma from a cystoprostatectomy specimen. The tumor consists of high-grade spindle and epithelioid cells with extension to the perivesical fat (a). The epithelial component was evident in the transurethral resection specimen (b). By targeted next generation sequencing of the sarcomatoid carcinoma the tumor harbored 12 alterations including TERT promoter (1295228C > T) mutation and truncating mutations in TP53 (Q331*) and ARID1A (T1921Kfs*16). Alterations in these genes are generally very common in urothelial carcinoma
Small Cell/Neuroendocrine Carcinoma of the Bladder
This is a rare variant of bladder cancer that is morphologically identical to the small cell carcinoma of the lung, but may be admixed with an epithelial (or rarely sarcomatoid) component of UCa in up to 50% of cases [1]. Epithelial components associated with this tumor are heterogeneous and include urothelial, squamous, glandular morphology or only an in situ component (Fig. 4.7).
The landscape of genomic alterations of small cell bladder cancer is still undefined, yet a few studies have provided intriguing insights into the similarities and differences between small cell and urothelial histology of bladder tumors as well as small cell cancer of the lung. A retrospective sequencing and copy number analysis of 97 carcinomas of the bladder, including ten small cell carcinomas, revealed RB1 alterations predicted to result in loss of function in every tumor [70], similar to findings in small cell lung cancer [71]. In a second study, 87 matched tumor and germline samples were sequenced from 61 patients with small cell carcinoma of the bladder. Tumors were derived from either transurethral resection (TUR) or cystectomy specimens. Macro-dissection was performed to isolate the neuroendocrine component in those tumors exhibiting mixed histology. Genomic analyses included targeted exon capture, whole exome, and whole transcriptome sequencing. Additionally, two samples were subjected to whole genome sequencing [72, 73]. TP53 and RB1 alterations were detected in 90% and 87% of this cohort, respectively, and 80% of tumors displayed co-alterations of both genes, similar to what is observed in small cell lung cancer. Furthermore, loss of expression of RB1 was identified in some tumors without a corresponding loss-of-function mutation, suggesting an alternative mechanism, such as epigenetic silencing, that may contribute to RB1 loss. Moreover, the high frequency of G1/S phase checkpoint disruption indicates that this may be a necessary event in the development of small cell bladder cancer. Interestingly, alterations commonly detected in UCa were also found in the small cell carcinoma cohort, including TERT promoter mutations in 95% and truncating alterations within epigenetic modifier genes such as CREBBP, EP300, ARID1A, KMT2D, and others, in nearly 75% of samples [72, 73]. A notable exception was KDM6A loss-of-function alterations, which were found more frequently in UCa than small cell histology. Activating FGFR3 mutations, a hallmark of low-grade urothelial tumors and present in approximately 20% of high-grade invasive UCa, were by contrast found in a minority of small cell carcinoma of the bladder. CDKN2A deletion and CCND1 amplifications, found commonly in UCa, were not detected within the small cell carcinoma cohort. E2F3 amplification was found in both small cell and urothelial bladder tumors, while this event was rare in small cell lung cancer.
A high level of chromosomal instability was observed in bladder small cell carcinoma, including whole genome duplication in 72% of tumors that correlated with the presence of TP53 missense mutations. The APOBEC mutation signature that was identified within muscle-invasive bladder cancer from the TCGA bladder cancer study [24] was observed in 95% of small cell bladder cancer in this cohort; notably, small cell lung cancers are typically characterized by a mutation signature associated with tobacco exposure distinct from the APOBEC signature.
In a subset of patients, sequencing was performed on the small cell and urothelial components of the same tumor. In two cases, clonal mutations were present that were identified in both the small cell and urothelial histologies, yet RB1 and TP53 mutations were sequestered within the small cell histology component, implying that these mutations represent evolutionary branching from a common precursor into two separate histologies. In a second example, clonal mutations within the TERT promoter and PIK3CA were identified in the small cell and urothelial histologies, while RB1 and TP53 alterations were only detected in the small cell component and an ERBB2 L755S mutation only within the urothelial component. These findings clearly support the concept that small cell carcinoma of the bladder is closely related to, and develops form, a precursor UCa. It still remains unclear; however, what exact molecular mechanisms underlie the development of the small cell histology from UCa as much of the reported alterations in small cell carcinoma are similar to what is reported in UCa including the combined RB1/TP53 which are co-mutated in a subset of UCa that clearly does not display small cell/neuroendocrine differentiation [24, 70, 74].
Due to their rarity, the treatment recommendations for small cell bladder cancers are extrapolated from those for small cell lung cancer, and include systemic cisplatin-based chemotherapy plus radical cystectomy or chemotherapy and radiation therapy. Similar to small cell lung cancer, metastatic spread of small cell bladder cancer occurs early in the disease course and recurrent disease following definitive therapy is typically resistant to additional chemotherapy. Clearly, novel treatments need to be investigated in small cell bladder cancer. Of note, in the cohort described above, 46% of tumors possessed potential therapeutically actionable alterations, including ERBB2 and PIK3CA hotspot activating mutations. The advent of basket trials of small molecular inhibitors, in which patients are enrolled based upon mutation status independent of tumor histology, provides an appealing treatment opportunity for patients with small cell bladder cancer whose tumors harbor such actionable genomic alterations.
Adenocarcinoma of the Bladder
Adenocarcinomas of the bladder as well as urachal adenocarcinomas are rare. While most of these tumors histologically resemble colorectal adenocarcinomas (Fig. 4.8), the genomic alterations that define this rare subset of bladder cancers are not well defined. In one study from a patient with metastatic urachal adenocarcinoma who achieved a long-term (at least 8 months) response to cetuximab (a monoclonal antibody directed against EGFR), targeted exome sequencing of the patient’s primary tumor initially identified an amplification of EGFR in a KRAS wild-type context. Sequencing of nine additional urachal carcinomas revealed MAPK pathway alterations in four tumors and mutations within APC in two specimens [75]. An additional cohort of 16 urachal adenocarcinomas was analyzed using a targeted exon capture sequencing approach which revealed KRAS hotspot alterations in 5 (29%) and ERBB2 activating mutations as well as amplification in 3 (18%) tumors [76]. These results suggest that MAPK pathway activation is a common phenomenon in urachal adenocarcinomas; moreover, this genomic profile of EGFR amplification, APC mutations, and KRAS activating mutations resembles that of colorectal adenocarcinoma. SMAD4, a tumor suppressor gene involved in TGF beta signaling, is commonly inactivated in pancreatic and colorectal adenocarcinomas, resulting in activation of the TGF beta pathway. Alterations in SMAD4, including two truncating mutations, were observed in 18% of urachal adenocarcinomas in this cohort. Additionally, GNAS hotspot alterations and amplification were identified in 18% of tumors. GNAS encodes the alpha subunit of the trimeric G protein coupled receptor complex that can activate the MAPK pathway. In a second patient with metastatic urachal adenocarcinoma that had progressed on chemotherapy, activating mutations were detected in KRAS (Q61L) and GNAS (R201C). Based upon this genomic profile, the patient was initiated on the MEK inhibitor trametinib for compassionate use and achieved over 29 months of stable disease. This response, in combination with that seen with cetuximab therapy, suggests that adenocarcinomas of the bladder and urachus represent a unique opportunity for MAPK pathway inhibition to derive meaningful clinical benefit. These observations also suggest that the genomic landscape of adenocarcinomas of the urinary tract may represent colorectal adenocarcinomas more closely than UCa.
In a separate cohort of nine primary bladder adenocarcinomas, a similarly high rate of KRAS alterations (43%) was observed. One specimen harbored ERBB2 amplification. Interestingly, mutations in ARID1A and SMARCA4, epigenetic modifiers that are commonly altered in UCa, were also seen. In both urachal and primary bladder adenocarcinomas, TP53 was the most commonly mutated gene [76].
References
Amin MB. Histological variants of urothelial carcinoma: diagnostic, therapeutic and prognostic implications. Mod Pathol. 2009;22(Suppl 2):S96–S118.
Moch H, et al. WHO classification of tumours of the urinary system and male genital organs. In: Bosman FT, et al., editors. World health organization classification of tumours. 4th ed. Lyon: International Agency for Research on Cancer; 2016.
Reuter VE. Bladder, risk and prognostic factors--a pathologist's perspective. Urol Clin North Am. 1999;26(3):481–92.
Rink M, et al. Impact of histological variants on clinical outcomes of patients with upper urinary tract urothelial carcinoma. J Urol. 2012;188(2):398–404.
Bassily NH, et al. Coordinate expression of cytokeratins 7 and 20 in prostate adenocarcinoma and bladder urothelial carcinoma. Am J Clin Pathol. 2000;113(3):383–8.
Kunju LP, et al. Prostate-specific antigen, high-molecular-weight cytokeratin (clone 34betaE12), and/or p63: an optimal immunohistochemical panel to distinguish poorly differentiated prostate adenocarcinoma from urothelial carcinoma. Am J Clin Pathol. 2006;125(5):675–81.
Skinnider BF, et al. Distribution of cytokeratins and vimentin in adult renal neoplasms and normal renal tissue: potential utility of a cytokeratin antibody panel in the differential diagnosis of renal tumors. Am J Surg Pathol. 2005;29(6):747–54.
Buza N, et al. Inverse p16 and p63 expression in small cell carcinoma and high-grade urothelial cell carcinoma of the urinary bladder. Int J Surg Pathol. 2010;18(2):94–102.
Pereira TC, et al. Can we tell the site of origin of metastatic squamous cell carcinoma? An immunohistochemical tissue microarray study of 194 cases. Appl Immunohistochem Mol Morphol. 2011;19(1):10–4.
Gaisa NT, et al. Different immunohistochemical and ultrastructural phenotypes of squamous differentiation in bladder cancer. Virchows Arch. 2011;458(3):301–12.
Gulmann C, et al. Immunohistochemical profile to distinguish urothelial from squamous differentiation in carcinomas of urothelial tract. Hum Pathol. 2013;44(2):164–72.
Huang W, et al. Novel markers of squamous differentiation in the urinary bladder. Hum Pathol. 2013;44(10):1989–97.
Chang A, et al. Utility of GATA3 immunohistochemistry in differentiating urothelial carcinoma from prostate adenocarcinoma and squamous cell carcinomas of the uterine cervix, anus, and lung. Am J Surg Pathol. 2012;36(10):1472–6.
Kaufmann O, Volmerig J, Dietel M. Uroplakin III is a highly specific and moderately sensitive immunohistochemical marker for primary and metastatic urothelial carcinomas. Am J Clin Pathol. 2000;113(5):683–7.
Mhawech P, Uchida T, Pelte MF. Immunohistochemical profile of high-grade urothelial bladder carcinoma and prostate adenocarcinoma. Hum Pathol. 2002;33(11):1136–40.
Parker DC, et al. Potential utility of uroplakin III, thrombomodulin, high molecular weight cytokeratin, and cytokeratin 20 in noninvasive, invasive, and metastatic urothelial (transitional cell) carcinomas. Am J Surg Pathol. 2003;27(1):1–10.
Paner GP, et al. Immunohistochemical evaluation of novel and traditional markers associated with urothelial differentiation in a spectrum of variants of urothelial carcinoma of the urinary bladder. Hum Pathol. 2014;45(7):1473–82.
Gruver AM, et al. Selective immunohistochemical markers to distinguish between metastatic high-grade urothelial carcinoma and primary poorly differentiated invasive squamous cell carcinoma of the lung. Arch Pathol Lab Med. 2012;136(11):1339–46.
Higgins JP, et al. Placental S100 (S100P) and GATA3: markers for transitional epithelium and urothelial carcinoma discovered by complementary DNA microarray. Am J Surg Pathol. 2007;31(5):673–80.
Liu H, et al. Immunohistochemical evaluation of GATA3 expression in tumors and normal tissues: a useful immunomarker for breast and urothelial carcinomas. Am J Clin Pathol. 2012;138(1):57–64.
Blochin EB, et al. Urothelial carcinoma with prominent squamous differentiation in the setting of neurogenic bladder: role of human papillomavirus infection. Mod Pathol. 2012;25(11):1534–42.
Chapman-Fredricks JR, et al. High-risk human papillomavirus DNA detected in primary squamous cell carcinoma of urinary bladder. Arch Pathol Lab Med. 2013;137(8):1088–93.
Alexander RE, et al. p16 expression is not associated with human papillomavirus in urinary bladder squamous cell carcinoma. Mod Pathol. 2012;25(11):1526–33.
Cancer Genome Atlas Research, N. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014;507(7492):315–22.
Choi W, et al. Identification of distinct basal and luminal subtypes of muscle-invasive bladder cancer with different sensitivities to frontline chemotherapy. Cancer Cell. 2014;25(2):152–65.
Sjodahl G, et al. A molecular taxonomy for urothelial carcinoma. Clin Cancer Res. 2012;18(12):3377–86.
Damrauer JS, et al. Intrinsic subtypes of high-grade bladder cancer reflect the hallmarks of breast cancer biology. Proc Natl Acad Sci U S A. 2014;111(8):3110–5.
Hansel DE, et al. Gene profiling suggests a common evolution of bladder cancer subtypes. BMC Med Genet. 2013;6:42.
Linder BJ, et al. The impact of histological reclassification during pathology re-review--evidence of a will Rogers effect in bladder cancer? J Urol. 2013;190(5):1692–6.
Shah RB, et al. Variant (divergent) histologic differentiation in urothelial carcinoma is under-recognized in community practice: impact of mandatory central pathology review at a large referral hospital. Urol Oncol. 2013;31(8):1650–5.
Wasco MJ, et al. Urothelial carcinoma with divergent histologic differentiation (mixed histologic features) predicts the presence of locally advanced bladder cancer when detected at transurethral resection. Urology. 2007;70(1):69–74.
Vail E, et al. Telomerase reverse transcriptase promoter mutations in glandular lesions of the urinary bladder. Ann Diagn Pathol. 2015;19(5):301–5.
Beltran AL, et al. Clinicopathological characteristics and outcome of nested carcinoma of the urinary bladder. Virchows Arch. 2014;465(2):199–205.
Linder BJ, et al. Outcomes following radical cystectomy for nested variant of urothelial carcinoma: a matched cohort analysis. J Urol. 2013;189(5):1670–5.
Lin O, et al. Nested variant of urothelial carcinoma: a clinicopathologic and immunohistochemical study of 12 cases. Mod Pathol. 2003;16(12):1289–98.
Cox R, Epstein JI. Large nested variant of urothelial carcinoma: 23 cases mimicking von Brunn nests and inverted growth pattern of noninvasive papillary urothelial carcinoma. Am J Surg Pathol. 2011;35(9):1337–42.
Lopez Beltran A, Montironi R, Cheng L. Microcystic urothelial carcinoma: morphology, immunohistochemistry and clinical behaviour. Histopathology. 2014;64(6):872–9.
Mai KT, et al. Nested and microcystic variants of urothelial carcinoma displaying immunohistochemical features of basal-like urothelial cells: an immunohistochemical and histopathogenetic study. Pathol Int. 2014;64(8):375–81.
Dhall D, Al-Ahmadie H, Olgac S. Nested variant of urothelial carcinoma. Arch Pathol Lab Med. 2007;131(11):1725–7.
Volmar KE, et al. Florid von Brunn nests mimicking urothelial carcinoma: a morphologic and immunohistochemical comparison to the nested variant of urothelial carcinoma. Am J Surg Pathol. 2003;27(9):1243–52.
Zhong M, et al. Distinguishing nested variants of urothelial carcinoma from benign mimickers by TERT promoter mutation. Am J Surg Pathol. 2015;39(1):127–31.
Al-Ahmadie HA, et al. Frequent somatic CDH1 loss-of-function mutations in plasmacytoid variant bladder cancer. Nat Genet. 2016;48(4):356–8.
Keck B, et al. The plasmacytoid carcinoma of the bladder--rare variant of aggressive urothelial carcinoma. Int J Cancer. 2011;129(2):346–54.
Nigwekar P, et al. Plasmacytoid urothelial carcinoma: detailed analysis of morphology with clinicopathologic correlation in 17 cases. Am J Surg Pathol. 2009;33(3):417–24.
Dayyani F, et al. Plasmacytoid urothelial carcinoma, a chemosensitive cancer with poor prognosis, and peritoneal carcinomatosis. J Urol. 2013;189(5):1656–61.
Kaimakliotis HZ, et al. Plasmacytoid bladder cancer: variant histology with aggressive behavior and a new mode of invasion along fascial planes. Urology. 2014;83(5):1112–6.
Berx G, et al. Cloning and characterization of the human invasion suppressor gene E-cadherin (CDH1). Genomics. 1995;26(2):281–9.
Kemler R. From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet. 1993;9(9):317–21.
Lim MG, et al. E-cadherin expression in plasmacytoid, signet ring cell and micropapillary variants of urothelial carcinoma: comparison with usual-type high-grade urothelial carcinoma. Mod Pathol. 2011;24(2):241–7.
Sangoi AR, et al. Interobserver reproducibility in the diagnosis of invasive micropapillary carcinoma of the urinary tract among urologic pathologists. Am J Surg Pathol. 2010;34(9):1367–76.
Johansson SL, Borghede G, Holmang S. Micropapillary bladder carcinoma: a clinicopathological study of 20 cases. J Urol. 1999;161(6):1798–802.
Solomon JP, et al. Challenges in the diagnosis of urothelial carcinoma variants: can emerging molecular data complement pathology review? Urology. 2016;102:7–16.
Amin MB, et al. Micropapillary variant of transitional cell carcinoma of the urinary bladder. Histologic pattern resembling ovarian papillary serous carcinoma. Am J Surg Pathol. 1994;18(12):1224–32.
Nassar H, et al. Pathogenesis of invasive micropapillary carcinoma: role of MUC1 glycoprotein. Mod Pathol. 2004;17(9):1045–50.
Luna-More S, et al. Invasive micropapillary carcinoma of the breast. A new special type of invasive mammary carcinoma. Pathol Res Pract. 1994;190(7):668–74.
Willis DL, et al. Clinical outcomes of cT1 micropapillary bladder cancer. J Urol. 2015;193(4):1129–34.
Spaliviero M, et al. Clinical outcome of patients with T1 micropapillary urothelial carcinoma of the bladder. J Urol. 2014;192(3):702–7.
Siefker-Radtke AO, et al. A phase 2 clinical trial of sequential neoadjuvant chemotherapy with ifosfamide, doxorubicin, and gemcitabine followed by cisplatin, gemcitabine, and ifosfamide in locally advanced urothelial cancer: final results. Cancer. 2013;119(3):540–7.
Meeks JJ, et al. Pathological response to neoadjuvant chemotherapy for muscle-invasive micropapillary bladder cancer. BJU Int. 2013;111(8):E325–30.
Sui W, et al. Micropapillary bladder cancer: insights from the national cancer database. Bladder Cancer. 2016;2(4):415–23.
Ching CB, et al. HER2 gene amplification occurs frequently in the micropapillary variant of urothelial carcinoma: analysis by dual-color in situ hybridization. Mod Pathol. 2011;24(8):1111–9.
Schneider SA, et al. Outcome of patients with micropapillary urothelial carcinoma following radical cystectomy: ERBB2 (HER2) amplification identifies patients with poor outcome. Mod Pathol. 2014;27(5):758–64.
Tschui J, et al. Morphological and molecular characteristics of HER2 amplified urothelial bladder cancer. Virchows Arch. 2015;466(6):703–10.
Ross JS, et al. A high frequency of activating extracellular domain ERBB2 (HER2) mutation in micropapillary urothelial carcinoma. Clin Cancer Res. 2014;20(1):68–75.
Guo CC, et al. Gene expression profile of the clinically aggressive micropapillary variant of bladder cancer. Eur Urol. 2016;70(4):611–20.
Lopez-Beltran A, et al. Carcinosarcoma and sarcomatoid carcinoma of the bladder: clinicopathological study of 41 cases. J Urol. 1998;159(5):1497–503.
Sung MT, et al. Histogenesis of sarcomatoid urothelial carcinoma of the urinary bladder: evidence for a common clonal origin with divergent differentiation. J Pathol. 2007;211(4):420–30.
Sanfrancesco J, et al. Sarcomatoid urothelial carcinoma of the bladder: analysis of 28 cases with emphasis on clinicopathologic features and markers of epithelial-to-mesenchymal transition. Arch Pathol Lab Med. 2016;140(6):543–51.
Wang X, et al. TERT promoter mutation status in sarcomatoid urothelial carcinomas of the upper urinary tract. Future Oncol. 2017;13:705–14.
Iyer G, et al. Prevalence and co-occurrence of actionable genomic alterations in high-grade bladder cancer. J Clin Oncol. 2013;31(25):3133–40.
George J, et al. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524(7563):47–53.
Chang MT, et al. The landscape of actionable genomic lesions in small cell bladder cancer. Cancer Res. 2014;74(19):316.
Hao XL, et al. Comprehensive molecular analysis of small cell carcinoma of the bladder. Mod Pathol. 2016;29:236A.
Kim PH, et al. Genomic predictors of survival in patients with high-grade urothelial carcinoma of the bladder. Eur Urol. 2015;67(2):198–201.
Collazo-Lorduy A, et al. Urachal carcinoma shares genomic alterations with colorectal carcinoma and may respond to epidermal growth factor inhibition. Eur Urol. 2016;70(5):771–5.
Jordan E, et al. Assessment of genomic alterations in bladder adenocarcinoma and urachal adenocarcinoma. Eur J Cancer. 2015;51:S530.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Al-Ahmadie, H., Iyer, G. (2018). Molecular Alterations in the Pathogenesis of Bladder Cancer Subtypes and Urothelial Carcinoma Variants. In: Hansel, D., Lerner, S. (eds) Precision Molecular Pathology of Bladder Cancer. Molecular Pathology Library. Springer, Cham. https://doi.org/10.1007/978-3-319-64769-2_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-64769-2_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-64767-8
Online ISBN: 978-3-319-64769-2
eBook Packages: MedicineMedicine (R0)