Abstract
Background: Several surgical techniques are used for the management of Chiari malformation type I (CM-I). Bony posterior fossa decompression is considered a good option in children, though with a higher risk of requiring reoperation. However, there is not enough evidence from the series in the literature, which are often limited by inadequate follow-up. The goal of this study was to assess the effectiveness of suboccipital craniectomy alone in children after long-term follow-up.
Methods: Forty-two children (25 female and 17 male; mean age 6.7 years), operated on with bony decompression alone, were retrospectively reviewed. All patients underwent suboccipital craniectomy. Thirty-eight children required C1 laminectomy, and 21 also underwent dural delamination on the basis of intraoperative ultrasound investigations. The outcome was assessed using the traditional measurement and the Chicago Chiari Outcome Scale (CCOS). The mean follow-up period was 11.3 years (range 5–15 years).
Results: Headache was the most frequent preoperative symptom (81%), followed by neck pain (40%), vertigo (40%), ataxia (26%), and upper and lower extremity paraesthesia (26%). Syringomyelia was present in 19 patients (45%). Resolution and significant improvement of preoperative clinical symptoms were observed in 36.5% and 21.5% of cases, respectively. Three children required adjunctive surgery for symptom recurrence (7%). The tonsil position and syringomyelia were normalized or improved in 50% and 79% of cases, respectively. No complications occurred. According to the CCOS scores, 69.5% of children had an excellent outcome, 28.5% had a functional outcome and 2% had an impaired outcome.
Conclusion: Bony decompression alone is an effective, safe and long-lasting treatment for children with CM-I. A certain risk of symptom recurrence requiring new surgery exists, but it is widely counterbalanced by the low risk of complications. Careful patient selection is crucial for a good outcome. Prospective and randomized studies are needed for further validation.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Chiari malformation type I (CM-I) is a heterogeneous condition encompassing a wide spectrum of clinical and radiological presentations. Several surgical techniques are currently used for its correction, ranging from suboccipital craniectomy/C1 laminectomy alone to transoral or transnasal ventral decompression followed by posterior stabilization, and including endoscopic posterior decompression, dural delamination, duraplasty, arachnoid dissection with/without tonsil coagulation, and minimally invasive tonsillectomy [1,2,3,4,5,6,7,8,9]. When one is dealing with CM-I patients, the first limitation is the lack of standardized surgical management. In fact, no guidelines are available in the literature, where no evidence of a ‘best’ treatment can be found. Similarly, no standardized methods for outcome evaluation have been identified so far [10]; a further limitation, partially related to this, is represented by the relatively short follow-up reported in many series. In this paper we present the late outcome of a paediatric series after long follow-up to contribute information on the management of selected cases with bony decompression alone.
Materials and Methods
All consecutive paediatric patients (aged 0–16 years) surgically treated at our institution with suboccipital craniectomy (with/without C1 laminectomy) in the decade of 2001–2011 were considered. This time period was established to ensure a minimum of 5-year follow-up. The patient-tailored selection criteria in use at our institution can be summarized as follows: (1) asymptomatic patients without relevant syringomyelia (<2 mm thickness): clinical and radiological follow-up; (2) symptomatic patients with tonsillar herniation above C2 or asymptomatic patients with 2–5 mm syringomyelia: bony decompression of the posterior cranial fossa; and (3) symptomatic patients (especially those with signs of brainstem dysfunction) with tonsils below C2 and/or >5 mm syringomyelia: bony decompression plus duraplasty (and subpial tonsil coagulation, if needed). Patients with psychomotor delay or behavioural problems but without clear signs/symptoms of CM-I were not considered for surgery but were enrolled for follow-up.
All patients underwent a preoperative workup consisting of physical and neurological examination, craniospinal magnetic resonance imaging (MRI), polysomnography and neuropsychological tests. The same examinations were utilized for the postoperative follow-up with the following timing: physical and neurological examinations every 6 months for the first 2 years, then yearly; neuropsychological tests yearly up to the scholar age and working age, according to the patients’ characteristics; craniospinal MRI yearly for the first 3 years, then every 2 or 3 years; and polysomnography 1 year after surgery in negative cases and yearly until normalization in positive cases. Decompression of the posterior fossa was achieved by suboccipital craniectomy with/without C1 laminectomy and/or delamination of the external layer of the dura mater. The epidural fibrous ring at the occipitocervical junction was resected. The extension of the craniectomy, as well as the laminectomy, were tailored on the basis of the patient’s age and characteristics. In any case, the re-expansion of the cisterna magna and pulsations of the tonsils were assessed by intraoperative ultrasound (IUS), and the extent of the craniectomy was adapted accordingly. The posterior arch of C1 was routinely removed unless the posterior compression at that level was not relevant. Beyond the traditional treatment outcome measurement (‘resolution’, ‘improvement’, ‘stability’ or ‘worsening’), the Chicago Chiari Outcome Scale (CCOS) was used for outcome assessment [11]. According to the CCOS, the outcome is graded as ‘excellent’ with a score of 16 points, ‘functional’ with a score of 12–15 points, ‘impaired’ with a score of 8–11 points and ‘incapacitated’ with a score of 4–7 points.
Results
Patient Characteristics
Among 164 paediatric patients admitted for CM-I during the relevant period, only 42 children were eligible for the study. Indeed, more than one third of all cases were asymptomatic, while the others needed duraplasty at least. Moreover, in six cases where a suboccipital craniectomy was planned, IUS did not show good expansion of the cerebellomedullary cistern, so duraplasty was performed (and they were not included in the present series).
The cohort was composed of 17 boys and 25 girls (male to female ratio 0.68), with a mean age of 6.7 years (range 15 months–16 years) at the time of surgery. The mean follow-up period for the entire cohort was 11.3 years, ranging from 5 to 15 years. In five cases (12%), the diagnosis was incidental (asymptomatic patients), while 15 children (35.5%) presented with specific signs—such as macrocrania and psychomotor delay—other than a suboccipital headache. Finally, 22 patients (52.5%) showed CM-I/syringomyelia–specific symptoms and signs, such as a suboccipital headache, neck pain, vertigo, ataxia, upper and/or lower limb paraesthesia and scoliosis (see Table 1). Tonsillar caudal herniation was detected in all cases: a 5-mm ectopia in eight cases (19%), a 6- to 10-mm ectopia in 18 cases (43%) and a ≥11 mm ectopia in 16 cases (38%). Syringomyelia was present in 19 patients (45%): cervical (eight cases, 43%), dorsal (three cases, 15.5%), cervicodorsal (five cases, 26%) and holocord (three cases, 15.5%) (see Table 2). Neuropsychological tests did not show significant alterations, with normal full-scale IQ (FSIQ), verbal IQ (VIQ) and performance IQ (PIQ) scores in 88% of cases (37 patients). In five children, some working memory, planning and attention disorders were detected, which clearly correlated with sleep disorders. Polysomnography was normal in 35 cases (83.5%) but revealed sleep-disordered breathing in seven cases (16.5%).
Surgical Outcome
All patients underwent suboccipital craniectomy. C1 laminectomy was carried out in 38 cases (90%), while dural delamination was performed in 21 cases (50%). At the end of the surgical procedure, IUS showed a satisfactory fluid film between the dura and the tonsils, and good tonsil pulsation in all cases. Complete resolution of the clinical picture at late follow-up was achieved in 76.5% of cases (32 patients) and an improvement in nine cases (21.5%); only in one case was there no improvement (2%). The evolution of the clinical symptoms is reported in detail in Table 1. These results were obtained after three patients (7%) experienced symptom recurrence ranging from 1 to 7 years after the first operation (all of them had experienced initial clinical improvement). All of them underwent second-look surgery: in the first patient (the youngest in the series), a newly formed suboccipital bone was found and was removed again, with clinical resolution; in the second patient, a thick fibrous scar was found, and clinical normalization was achieved after its excision; the third case required two operations (the first one for fibrous scar removal followed by a second one for tonsil coagulation and duraplasty), resulting in only transient symptom improvement. No surgical complications occurred except for postoperative neck pain in one third of cases, requiring extra analgesic drug therapy. On the basis of CCOS scores and considering the single reoperation as a ‘transient complication’ and the double reoperation as a ‘permanent complication, well controlled’, 29 patients (69.5%) had an excellent outcome, 12 (28.5%) had a functional outcome and one (2%) had an impaired outcome.
The position of the cerebellar tonsils remained stable or improved in 69% of cases (29 patients), while normalization of their position was achieved in the remaining 13 cases (31%). The tonsil ascent was mainly observed in the late phases of follow-up, often seven or more years after surgery (Fig. 1). Syringomyelia disappeared or showed a significant reduction in 31% (six patients) and in 48% of cases (nine patients), respectively; it remained unchanged in the remaining four cases (21%). The radiological results are summarized in Table 2. Polysomnography improved in all cases, with normalization in five of seven patients. A sleep-correlated improvement in the neuropsychological deficits was detected in all five affected cases, with normalization in four of them.
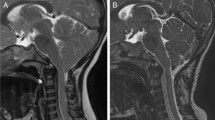
Magnetic resonance imaging of a boy operated on for Chiari malformation type I at the age of 6 years. (a) Preoperative image showing a stenosis of the foramen magnum with 8 mm caudal descent of the cerebellar tonsils. (b) Postoperative image after 3-year follow-up, demonstrating ascent of the cerebellar tonsils but not yet normalization. (c) Normalization of the radiological picture after 6.5-year follow-up
Discussion
According to a recent analysis of a large US nationwide healthcare network, the numbers of surgical procedures performed for CM-I increased by 51% in the paediatric population and by 28% in the adult population in the last 14 years [12]. Despite this large number of surgical procedures, the ‘old’ dilemma regarding decompression of the posterior fossa—posterior fossa bony decompression alone (PFD) versus posterior fossa bony decompression plus duraplasty (PFDD)—has not been resolved yet. Although the matter is still controversial, PFDD is currently suggested in adults—despite the higher rate of complications and the longer operation time—on the basis of the significantly better outcome and the lower recurrence (and reoperation) rates [13,14,15,16]. As far as children are concerned, the current trend seems to be in favour of PFD, though there is no level I or IIa evidence for this [17]. Some authors, however, advocate the use of duraplasty for the same reasons as those given for adults [18, 19], whereas many authors propose to reserve duraplasty for the small number of children who experience recurrence after bony decompression, which shows a significantly lower rate of complications (especially cerebrospinal fluid [CSF] leakage) [20,21,22,23,24,25] and virtually no risk of surgical death [26]. One meta-analysis (which is still the only one available in the literature for children), published by Durham and Fjeld-Olenec in 2008, considered seven papers describing 582 paediatric patients treated by suboccipital craniectomy alone (266 cases) or with duraplasty (316 cases), and found that the outcome was statistically similar with the two techniques in term of clinical improvement and syrinx reduction, while PFDD ensured a significantly lower rate of recurrence (2.1% versus 12.6%) [27]. As expected, the data were significantly in favour of PFD when CSF leakage was considered (1.8% versus 18.5%), with no substantial differences regarding the other complications. The authors noted that the follow-up (ranging from 2 to 120 months) was ‘inconsistently reported across studies’.
The goal of our study was to report a series with a sufficient length of follow-up to present definitive results. After 11.3 years of follow-up, 76.5% of our cases showed clinical resolution and 21.5% of them showed a considerable clinical improvement, with an excellent outcome in nearly 70% of cases and a functional outcome in 28% of cases (according to their CCOS scores). Only one patient did not show any improvement and had an impaired outcome despite two reoperations (the second one involved subpial tonsillectomy). On these grounds, PFD can be definitely considered a valid option for surgical management of CM-I in children. A certain risk of recurrence exists (7% in the present series) but can be successfully managed by second-look surgery. Moreover, permanent and/or significant complications are virtually absent. Indeed, the risk of reoperation is counterbalanced by the low rate of complications, so the final balance between complications and reoperations in PFDD and in PFD seems to be more favourable in PFD [28, 29]. To achieve this outcome, it is necessary for the patients to be carefully selected, and PFD is used in less severe cases [22, 30]. On these bases, the less satisfactorily radiological results do not represent a limitation since they are frequently reported in the literature and do not preclude a good clinical outcome [20, 31, 32].
Such good outcomes of PFD in children result from two main factors. The first one is progressive enlargement of the posterior fossa in children, which is due to physiological growth of the skull in the paediatric age group. In contrast to the scenario in adults, this growth may make PFDD unnecessary simply because the bone opening supports the spontaneous bone enlargement in providing space for the neural structures of the posterior fossa. For these reasons, the extent of bony decompression is crucial and must be tailored according to the patient’s characteristics and age. Indeed, in infants and young children the suboccipital craniectomy should not be too large (because of the risk of a pseudomeningocele resulting from cerebellar pulsations) and should not be too small (because of the risk of bone re-formation, as happened in one patient in our series) [6]. In patients with thicker, more slowly growing bone, such as older children and adolescents, the extent of bone removal should be estimated on the basis of the severity of the clinical and radiological picture. IUS currently is the most widely used tool to assess the correct extension of craniectomy/laminectomy because it allows the neurosurgeon to directly visualize the re-expansion and pulsation of the cisterna magna. In our experience, IUS was effective in achieving this goal: six patients (not included in the present series) were actually managed with PFDD because PFD did not provide good intraoperative results; moreover, among the three patients with recurrence, only one needed PFDD, while the other two patients were managed with newly formed bone and scar removal. Successful experiences with IUS have been reported by other authors [33, 34], even though it is less effective in cases of tonsil herniation caudal to C1 [35]. A criticism of this strategy has been raised by the use of intraoperative MRI in CM-I surgery. Bond et al. recently reported their experience involving 15 adults who underwent pre-incision prone intraoperative MRI: at that time, 93% of them (14 of 15 cases) showed a substantial improvement in the CSF flow dorsal to the cerebellar tonsils [36]. Therefore, one could speculate that the prone position is sufficient to improve CSF dynamics, so bony decompression does not add significant advantages to the operation. However, this experience needs to be replicated in children, where the physiologically ‘crowded’ posterior fossa (in comparison with that in adults) would not respond so well to prone positioning alone.
The second crucial factor is careful patient selection. The main limitation of our study, which is also the main merit of our management policy, is represented by a selection bias due to the adopted criteria (see ‘Materials and Methods’). This is the reason why we did not present a comparative cohort: since the patients treated with PFD and those treated with PFDD have different clinical and radiological characteristics, it is not possible to compare them. At the same time, however, this policy allowed us to select the most suitable candidates for PFD and to obtain good and stable long-term results. These considerations apply also to the literature [23, 25, 37, 38] and are further supported by the very recent experience reported by Pomeraniec and co-workers in their series of 95 children with CM-I [39]. After careful patient selection, 70 children were managed conservatively and had a good outcome without clinical or radiological worsening. Only 25 children underwent surgery, with a significant clinical improvement (75%) and radiological improvement (87.5%), regardless of the surgical technique used (bony decompression with a dural split versus duraplasty).
Taking into account the limitations inherent in a retrospective analysis of a single cohort from a single institution, this analysis of long-term results allows us to conclude that in children, PFD should be considered as a first-choice option in patients with a less severe clinical picture (no signs of brainstem dysfunction, tonsils above C2, thin syringomyelia), who are often the majority among paediatric patients. PFDD, with or without intradural manipulation, should be performed in more severe cases or in rare cases of failure of the previous strategy. The good final outcome of our series, according to the CCOS scores, shows that overall the risk of recurrence after PFD is broadly balanced by the very low risk of complications. For correct outcome assessment, a sufficiently long follow-up period (at least 7–10 years) is mandatory. Further prospective and randomized studies are required to validate these observations.
Compliance with Ethical Standards
No financial support was received for this work.
Competing Interests
The authors declare that they have no competing interests.
References
Albert GW, Menezes AH, Hansen DR, Greenlee JD, Weinstein SL. Chiari malformation type I in children younger than age 6 years: presentation and surgical outcome. J Neurosurg Pediatr. 2010;5:554–61.
Beecher JS, Liu Y, Qi X, Bolognese PA. Minimally invasive subpial tonsillectomy for Chiari I decompression. Acta Neurochir. 2016;158:1807–11.
Chauvet D, Carpentier A, George B. Dura splitting decompression in Chiari type 1 malformation: clinical experience and radiological findings. Neurosurg Rev. 2009;32:465–70.
Di X. Endoscopic suboccipital decompression on pediatric Chiari type I. Minim Invasive Neurosurg. 2009;52:119–25.
Hankinson TC, Grunstein E, Gardner P, Spinks TJ, Anderson RC. Transnasal odontoid resection followed by posterior decompression and occipitocervical fusion in children with Chiari malformation type I and ventral brainstem compression. J Neurosurg Pediatr. 2010;5:549–53.
Massimi L, Caldarelli M, Paternoster G, Novegno F, Tamburrini G, Di Rocco C. Mini-invasive surgery for Chiari type I malformation. Neuroradiol J. 2008;21:65–70.
Parker SR, Harris P, Cummings TJ, George T, Fuchs H, Grant G. Complications following decompression of Chiari malformation type I in children: dural graft or sealant? J Neurosurg Pediatr. 2011;8:177–83.
Sindou M, Gimbert E. Decompression for Chiari type I-malformation (with or without syringomyelia) by extreme lateral foramen magnum opening and expansile duraplasty with arachnoid preservation: comparison with other technical modalities (literature review). Adv Tech Stand Neurosurg. 2009;34:85–110.
Tubbs RS, Beckman J, Naftel RP, Chern JJ, Wellons JC, Rozzelle CJ, Blount JP, Oakes WJ. Institutional experience with 500 cases of surgically treated pediatric Chiari malformation type I. J Neurosurg Pediatr. 2011;7:248–56.
Greenberg JK, Milner E, Yarbrough CK, Lipsey K, Piccirillo JF, Smyth MD, Park TS, Limbrick DD Jr. Outcome methods used in clinical studies of Chiari malformation type I: a systematic review. J Neurosurg. 2015;122:262–72.
Aliaga L, Hekman KE, Yassari R, Straus D, Luther G, Chen J, Sampat A, Frim D. A novel scoring system for assessing Chiari malformation type I treatment outcomes. Neurosurgery. 2012;70:656–65.
Wilkinson DA, Johnson K, Garton HJ, Muraszko KM, Maher CO. Trends in surgical treatment of Chiari malformation type I in the United States. J Neurosurg Pediatr. 2016;11:1–9.
Förander P, Sjåvik K, Solheim O, Riphagen I, Gulati S, Salvesen Ø, Jakola AS. The case for duraplasty in adults undergoing posterior fossa decompression for Chiari I malformation: a systematic review and meta-analysis of observational studies. Clin Neurol Neurosurg. 2014;125:58–64.
Gürbüz MS, Berkman MZ, Ünal E, Akpınar E, Gök Ş, Orakdöğen M, Aydın S. Foramen magnum decompression and duraplasty is superior to only foramen magnum decompression in Chiari malformation type 1 associated with syringomyelia in adults. Asian Spine J. 2015;9:721–7.
Rehman L, Akbar H, Bokhari I, Babar AK, M Hashim AS, Arain SH. Posterior fossa decompression with duraplasty in Chiari-1 malformations. J Coll Physicians Surg Pak. 2015;25:254–8.
Xu H, Chu L, He R, Ge C, Lei T. Posterior fossa decompression with and without duraplasty for the treatment of Chiari malformation type I—a systematic review and meta-analysis. Neurosurg Rev. 2016;40(2):213–21.
Hankinson T, Tubbs RS, Wellons JC. Duraplasty or not? An evidence-based review of the pediatric Chiari I malformation. Childs Nerv Syst. 2011;27:35–40.
Foreman P, Safavi-Abbasi S, Talley MC, Boeckman L, Mapstone TB. Perioperative outcomes and complications associated with allogeneic duraplasty for the management of Chiari malformations type I in 48 pediatric patients. Neurosurg Pediatr. 2012;10:142–9.
Ma J, You C, Chen H, Huang S, Ieong C. Cerebellar tonsillectomy with suboccipital decompression and duraplasty by small incision for Chiari I malformation (with syringomyelia): long term follow-up of 76 surgically treated cases. Turk Neurosurg. 2012;22:274–9.
Caldarelli M, Novegno F, Massimi L, Romani R, Tamburrini G, Di Rocco C. The role of limited posterior fossa craniectomy in the surgical treatment of Chiari malformation type I: experience with a pediatric series. J Neurosurg. 2007;106(3 Suppl):187–95.
Genitori L, Peretta P, Nurisso C, Macinante L, Mussa F. Chiari type I anomalies in children and adolescents: minimally invasive management in a series of 53 cases. Childs Nerv Syst. 2000;16:707–18.
Kennedy BC, Kelly KM, Phan MQ, Bruce SS, McDowell MM, Anderson RC, Feldstein NA. Outcomes after suboccipital decompression without dural opening in children with Chiari malformation type I. J Neurosurg Pediatr. 2015;16:150–8.
Lee A, Yarbrough CK, Greenberg JK, Barber J, Limbrick DD, Smyth MD. Comparison of posterior fossa decompression with or without duraplasty in children with type I Chiari malformation. Childs Nerv Syst. 2014;30:1419–24.
Limonadi FM, Selden NRJ. Dura-splitting decompression of the craniocervical junction: reduced operative time, hospital stay, and cost with equivalent early outcome. J Neurosurg. 2004;101:184–8.
Navarro R, Olavarria G, Seshadri R, Gonzales-Portillo G, McLone DG, Tomita T. Surgical results of posterior fossa decompression for patients with Chiari I malformation. Childs Nerv Syst. 2004;20:349–56.
Korshunov AE, Kushel’ YV. Posterior decompression of the craniovertebral junction in children with Chiari malformation: a surgery extent issue [in Russian]. Zh Vopr Neirokhir Im N N Burdenko. 2016;80:13–20.
Durham SR, Fjeld-Olenec K. Comparison of posterior fossa decompression with and without duraplasty for the surgical treatment of Chiari malformation type I in pediatric patients: a meta-analysis. J Neurosurg Pediatr. 2008;2:42–9.
Mutchnick IS, Janjua RM, Moeller K, Moriarty TM. Decompression of Chiari malformation with and without duraplasty: morbidity versus recurrence. J Neurosurg Pediatr. 2010;5:474–8.
Shweikeh F, Sunjaya D, Nuno M, Drazin D, Adamo MA. National trends, complications, and hospital charges in pediatric patients with Chiari malformation type I treated with posterior fossa decompression with and without duraplasty. Pediatr Neurosurg. 2015;50:31–7.
Chotai S, Medhkour A. Surgical outcomes after posterior fossa decompression with and without duraplasty in Chiari malformation-I. Clin Neurol Neurosurg. 2014;125:182–8.
McGirt MJ, Nimjee SM, Floyd J, Bulsara KR, George TM. Correlation of cerebrospinal fluid flow dynamics and headache in Chiari I malformation. Neurosurgery. 2005;56:716–21.
McGirt MJ, Nimjee SM, Fuchs HE, George TM. Relationship of cine phase-contrast magnetic resonance imaging with outcome after decompression for Chiari I malformations. Neurosurgery. 2006;59:140–6.
Narenthiran G, Parks C, Pettorini B. Management of Chiari I malformation in children: effectiveness of intra-operative ultrasound for tailoring foramen magnum decompression. Childs Nerv Syst. 2015;31:1371–6.
Yeh DD, Koch B, Crone KR. Intraoperative ultrasonography used to determine the extent of surgery necessary during posterior fossa decompression in children with Chiari malformation type I. J Neurosurg. 2006;105(1 Suppl):26–32.
McGirt MJ, Attenello FJ, Datoo G, Gathinji M, Atiba A, Weingart JD, Carson B, Jallo GI. Intraoperative ultrasonography as a guide to patient selection for duraplasty after suboccipital decompression in children with Chiari malformation type I. J Neurosurg Pediatr. 2008;2:52–7.
Bond AE, Jane JA Sr, Liu KC, Oldfield EH. Changes in cerebrospinal fluid flow assessed using intraoperative MRI during posterior fossa decompression for Chiari malformation. J Neurosurg. 2015;122:1068–75.
Munshi I, Frim D, Stine-Reyes R, Weir BK, Hekmatpanah J, Brown F. Effects of posterior fossa decompression with and without duraplasty on Chiari malformation–associated hydromyelia. Neurosurgery. 2000;46:1384–9.
Novegno F, Caldarelli M, Massa A, Chieffo D, Massimi L, Pettorini B, Tamburrini G, Di Rocco C. The natural history of the Chiari type I anomaly. J Neurosurg Pediatr. 2008;2:179–87.
Pomeraniec IJ, Ksendzovsky A, Awad AJ, Fezeu F, Jane JA Jr. Natural and surgical history of Chiari malformation type I in the pediatric population. J Neurosurg Pediatr. 2016;7:343–52.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Massimi, L., Frassanito, P., Chieffo, D., Tamburrini, G., Caldarelli, M. (2019). Bony Decompression for Chiari Malformation Type I: Long-Term Follow-Up. In: Visocchi, M. (eds) New Trends in Craniovertebral Junction Surgery. Acta Neurochirurgica Supplement, vol 125. Springer, Cham. https://doi.org/10.1007/978-3-319-62515-7_17
Download citation
DOI: https://doi.org/10.1007/978-3-319-62515-7_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-62514-0
Online ISBN: 978-3-319-62515-7
eBook Packages: MedicineMedicine (R0)