Abstract
All progressive muscle contractile impairments (including age-related muscle weakness) need permanent management. We discuss evidence that subclinical denervation contributes to atrophy and slowness of aged muscle, and we provide stimulation protocols for aged and denervated skeletal muscles. We describe in Sect. 11.1 the effects of home-based neuromuscular FES in elderly persons and then in Sect. 11.2 the diagnostics, training protocols, and clinical results of home-based functional electrical stimulation (h-b FES) for denervated–degenerated skeletal muscle tissue. Using molecular analyses, histological morphometry, and Muscle Color Computed Tomography, we evaluated muscles of elderly persons, showing that increased levels of muscle contractions induced by h-b FES reverse muscle weakness and atrophy both in sedentary seniors and in conus and cauda equina complete syndrome.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
1 Structural and Molecular Markers of Muscle Weakness and of Recovery Induced by h-b FES
Aging is an unavoidable process, if lethal diseases do not prematurely end survival. A final evidence of this obvious concept has been recently provided by Gava et al. [1], describing the power decay of the series of world records obtained by master athletes (from 30 to 100 years). Skeletal muscle power begins to decline at the age of 30 years and continues, almost linearly, to zero at the age of 110 years. During aging, many factors contribute to the decline of the skeletal muscle, including subclinical and reversible muscle denervation/reinnervation [2]. Recently, we added evidence that denervation contributes to atrophy and slowness of aged muscles by comparing muscles of lifelong active seniors to those of sedentary elderly people. We found that the sportsmen have more muscle mass and slow fiber groupings, providing in this way evidence that physical activity maintains slow motoneurons, which reinnervate muscle fibers [3,4,5]. A premature muscle atrophy/degeneration occurs in irreversible and complete conus and cauda equina syndrome consequent to a lumbar/sacral spinal cord injury, determining a complete loss of muscle fibers within 5–8 years [6,7,8].
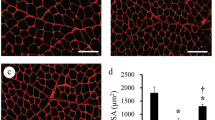
The impact of muscle denervation on aging skeletal muscle fibers is a relatively orphan topic [9, 10]. This is related in part to the difficulties in determining by molecular approaches whether motoneurons release chemical neurotrophic agents to the muscle fibers of the motor units. It is well known that such mechanism contributes to neuromuscular junction development and maintenance. However, if and which chemical trophic factors influence the synchronized expression of the hundreds of nuclei belonging to a single muscle fiber remains a subject of hypotheses. The synchronized spread of muscle action potential seems to be, on the contrary, a more rational mechanism [11]. On the other hands, the conclusions of recent reviews [8, 9, 12] are that FES is poorly used in rehabilitation of elderly patients. Aging skeletal muscle presents both a loss of muscle mass and a conspicuous reduction in myofiber plasticity [9, 12,13,14] and alterations in muscle-specific transcriptional mechanisms. During the aging process, protein synthetic rates decrease, and an increase in protein degradation follows [15, 16], affecting muscle fibers [17]. It is generally recognized that an ineffective damage repair is a contributory cause of functional impairment during aging [18,19,20] and promotes the detrimental replacement of functional contractile muscle with fibrous tissue [17]. Volitional physical exercise can reverse these damaging processes. Interestingly, it has been shown that both acute and prolonged resistance exercises stimulate the proliferation of satellite cells in healthy sedentary elderly subjects [21]. This fact may be explained by the increased levels of myostatin, a negative regulator of muscle mass. An increase in autophagy in the muscle of athletic people has been reported, suggesting that exercise may activate an important system that detoxifies muscle cells [22]. Another important factor that is associated with physical exercise is insulin-like growth factor 1 (IGF-1) [23]. The evidence suggests that training and regular exercise can increase muscle strength and attenuate sarcopenia by modulating expression of autophagy, myokines (IL-6), and IGF-1 [24].
Unfortunately, elderly people may be unable or reluctant to adequately participate in physical exercise. We therefore suggested that FES could be an alternative approach. A stimulator for neuromuscular electrical stimulation (ES), which especially suits elderly people requirements, was designed. As detailed in Kern et al. [15], older persons were exposed to regular neuromuscular ES training for a period of 9 weeks, starting two times a week for the first 3 weeks and then switching to three times a week for the next 6 weeks, for a total amount of 24 training sessions (3 × 10 min for each session). After detailed instructions, ES training was performed by the subjects themselves at home, using a two-channel custom-built battery-powered stimulator [25]. The subjects applied two conductive rubber electrodes (9 × 14 cm, 126 cm2) which were attached to the skin by wet sponge on the anterior thigh on both sides (left/right). The electrode pairs for the left and right thigh were connected to the two channels of the stimulator. This allowed independent activation of the left and right thigh muscles, which were alternatively stimulated. Each repetition (muscle contraction) was evoked by a 3.5 s train (60 Hz) of electrical pulses (rectangular, biphasic, width 0.6 ms). Consecutive contractions of the same thigh were separated by 4.5 s off intervals. In this study, constant voltage stimulation devices were applied. The subjects were also instructed to increase the stimulation intensity until their maximal tolerance was reached. Using this intensity, a full knee extension was achieved in all subjects. Nevertheless, the applied current and voltage were recorded by the stimulation device for each training session (the mean stimulation current was 128 ± 16 mA and voltage of 39 ± 14 V). The outcome was an increase in muscle strength, associated with an increase of fast fibers, which are the first to respond to ES and are related to the power of the skeletal muscle [14, 15]. We found that ES increases expression of IGF-1, the markers of satellite cell proliferation and extracellular matrix remodeling, downregulating the expression of proteases [14, 58]. There is also a collagen remodeling during both volitional physical exercise and ES. Three different forms of collagen are upregulated in electrically stimulated muscle [14, 15], but the increase in collagen does not likely stimulate fibrosis, as is shown by both morphological evidence and the increase in expression of miR29 [26]. The INTERREG IVa project recruited sedentary seniors with a normal lifestyle who were trained for 9 weeks with ES. Functional tests of trained subjects showed that ES induced improvements of leg muscle strength and mobility [27, 28]. ES significantly increased the size of fast-type muscle fibers and the number of Pax7- and NCAM-positive satellite cells. Moreover, no signs of muscle damage and/or inflammation were observed in muscle biopsies [14, 15]. Altogether, the results here summarized demonstrate that physical exercise, either voluntary or induced by ES, improves the functional performance of aging muscles. Of course, physical training can’t stop the aging process, but ES is a safe home-based method that is able to counteract atrophy of fast-type muscle fibers.
Age-related muscle power decline is partially attributable to a loss of innervation, and it can be delayed by a lifelong high-level activity [3,4,5]. Diseases involving permanent denervation show a similar, but premature, aging process and a much more severe muscle deterioration. Despite doubts and criticisms of related literature [29, 30], we have shown that, with appropriate protocols, h-b FES can even inhibit degeneration of denervated muscle and even reverse it [31,32,33]. Therefore, the use of h-b FES should be extended in critical care units, rehabilitation centers, and nursing facilities and at home of elderly persons in presence of peripheral nerve lesions to improve hopefully muscle reinnervation [34,35,36,37].
2 FES for Partially Denervated Muscle and FES Protocols for Training of Denervated–Degenerated Skeletal Muscle
Functional electrical stimulation (FES) was introduced more than 50 years ago to identify those applications of electrical currents to organs and tissues of humans and other large mammals to obtain responses mimicking and/or restoring lost functions [38]. Spinal cord injury causes loss of functions and muscle wasting, which are especially severe after lower motor neuron (LMN) lesions. During the last decades, a great attention has been paid to denervated muscles, numerous studies focusing on the changes in gross anatomy, mechanical properties, and potential for reinnervation of skeletal muscle when the lesions involve the LMN. Results in literature are at best contradictory, and claims suggest that electrical stimulation of partially denervated muscles may even interfere with nerve regeneration [7]. During the last decade, we studied the effects of physical exercise induced by FES in spinal cord injury (SCI) with complete conus and cauda equina syndrome, in which denervated leg muscles are permanently and completely disconnected from the nervous system. Denervated human muscles become refractory to the electrical stimulation developed by the common electrical devices and undergo ultrastructural disorganization within few months, while severe atrophy with nuclear clumping and fibro-fatty degeneration appears within 3 and 6 years [8, 9]. To counteract these progressive changes, a novel therapy concept for paraplegic patients with complete LMN denervation was developed in Vienna: the home-based FES (h-b FES) for denervated–degenerated muscles. New electrodes and a safe stimulator have been designed to reverse severe atrophy by delivering high-intensity (up to 2.4 J) and long-duration impulses (up to 150 ms) to stimulate contractions in long-term denervated skeletal muscle fibers. A specific clinical assessment and training, based on sound evidence from animal experiments, was developed at the Wilhelminenspital in Vienna, Austria. After 2-year training using h-b FES, the patient showed (1) significant increase of muscle mass and myofiber size with striking improvements of the muscle ultrastructural organization; (2) recovery of tetanic contractility with significant increase in muscle force output, during electrical stimulation; and (3) capacity to perform FES-assisted stand-up and stepping-in-place exercise. In conclusion h-b FES of permanent denervated muscle is an effective home therapy that results in rescue of muscle perfusion, mass, and function. Additional benefits are reduced leg edema, prophylaxis of thromboembolism, and enhanced cushioning effect for seating [32]. In our clinical experiences, nerve regrowth is not inhibited by FES of partially denervated muscles. On the contrary, we observed sensory or motor reinnervation (or both) during the application of high intensive stimulation training even 2–5 years after injury [39]. The complex response of muscle tissue to denervation is one of the most studied processes in muscle physiology and pathology. One of the important conclusions of pioneering studies is that, after a certain period of time (6 months in the rodents), denervated muscle undergoes some irreversible changes that inhibit its full restoration even after reinnervation. Findings from different experimental models, including free autotransplant, led to the same conclusion: after 2–7 months of denervation the restorative capacity of the muscle declines progressively in rat [40]. These observations were translated to the macroscopic behavior of the denervated human muscle and its potential for reinnervation, establishing a dogma that continues to negatively influence clinical management of denervated muscle. Despite countless published results [41], our recent studies in animal models and humans are enlightening several unrecognized characteristics and behaviors of the complex processes that occur during permanent LMN denervation of the human muscle tissue. These results are strengthening the rational basis of FES to maintain/recover permanently LMN denervated muscles. Some of the effects of long-lasting (in terms of years) LMN denervation of the human muscle we recently described were unexpected. In contrast to the well-known rodent model, the LMN denervated human muscle presents simple atrophy up to 1 year after SCI [7]. On the other hand, the characteristic denervation-induced muscle fiber disorganization in the atrophying muscle, documented by electron microscopy, occurs much earlier [7]. The ultrastructural disorganization of the muscle fibers that appears much earlier than severe atrophy in both animal models [42, 43] and humans [6, 7, 32, 44] explains the early functional impairments of the LMN denervated muscle. Until today, muscles of the extremities in conus and cauda equina syndrome were commonly not treated with FES, because it was widely recognized that long-term and completely denervated muscles cannot be effectively stimulated. On the other hand, our studies in animal models and humans indicate that (1) severe atrophy does not occur in rats for at least 3–4 months [40, 41]; (2) in rabbit, the degeneration of muscle tissue does not appear in the first year after denervation [43]; and (3) muscle tissue degeneration starts from the third year onward, in humans [32, 33, 42, 44]. Our findings that the long-term denervated rat muscle maintains L-type Ca2+ current and gene expression of the related proteins longer than functional contractile machineries [45] provide the molecular, structural, and functional rationale of a rehabilitation training for human permanently denervated muscles, which was developed as a result of empirical clinical observations [32]. The FES device stimulates muscle fibers in the absence of nerve endings and after prolonged denervation, determining (1) restoration of muscle fiber ultrastructure, (2) recovery of conduction velocity of the excitation–contraction apparatus up to a level that allows tetanic contraction, and (3) the maintenance/recovery of muscle mass and fiber size. Before this, electrical stimulation was believed to be effective only when started very early after LMN lesion, and FES was used only to delay muscle atrophy, since long-term denervated muscles (6 months onward) do not respond, when stimulated by surface electrodes and standard commercial electrical stimulators developed for innervated muscles. The results of the EU RISE Project [32, 33, 42, 44], and of the related animal research [43, 45], provide different perspectives.
2.1 Stimulation Devices and Electrodes
During the last 20 years, clinicians and engineers developed in Vienna novel rehabilitation concepts for paraplegic patients with complete LMN denervation of the lower extremity due to complete lesion of the conus and cauda equina. To counteract the progressive changes that transform muscle into an unexcitable tissue, these new rehabilitation managements became possible due to the development and optimization of new stimulation devices for h-b FES. They have been specifically designed to reverse longstanding and severe atrophy of LMN denervated muscles by delivering high-intensity and long-duration impulses that can directly elicit contraction of denervated skeletal muscle fibers in absence of functional neuromuscular endplates. These new stimulators and the large surface electrodes needed to cover large denervated muscles were developed by the Center of Medical Physics and Biomedical Engineering at the Medical University of Vienna, Austria [46,47,48]. Based on these prototypes, an electrical stimulator for denervated muscle is now commercially available, the “Stimulette den2x” of the Schuhfried Medizintechnik GmbH, Vienna, Austria [49]. Patients were provided with stimulators and electrodes in order to perform stimulation at home for 5 days per week. Large (180 cm2) electrodes (Schuhfried Medizintechnik GmbH, Mödling, Austria) made of conductive polyurethane were placed on the skin surface using a wet sponge cloth (early training) and fixed via elastic textile cuffs. As soon as the skin was accustomed to the necessary high current density, gel was used under the polyurethane electrodes to achieve minimal transition impedance. The electrodes were flexible enough to maintain evenly distributed pressure to the uneven and moving skin, thus providing homogeneous current distribution throughout the entire contact area. In parallel, specific clinical assessments and training settings were developed at the Wilhelminenspital Wien, Austria [50,51,52,53,54].
2.2 Training Protocol
Details of the four phases of this rehabilitation strategy are summarized in Table 11.1 and Fig. 11.1. The rehabilitation training for complete denervated muscles was validated by the clinical outcome, strongly supported by the results obtained from light and electron microscopy muscle biopsies’ analyses performed in Padua and Chieti Universities (Italy), respectively, and described by Kern et al. in his longitudinal prospective study [32]. The training strategy consisted of two progressive stimulation programs (Fig. 11.1), and positive changes and effects of FES on structural, functional, histological, and ultrastructural parameters were described in detail by Kern et al. in 2010 [32]. FES treatment can be integrated into daily life of patients without too much interference on their daily activities. The reduction of leg edema [55], the improved cosmetic appearance of lower extremities, the enhanced cushioning effect for seating, and the ability to perform home exercises are additional effects of FES. We are thus confident that it will be possible to translate into a wider population of diseased subjects and to aged people the knowledge of our interdisciplinary group of European scientists and clinicians. In fact, current studies suggest that electrical stimulation of the LMN denervated muscles could be extended to patients with incomplete lesions or elderly persons and could reduce secondary complications related to disuse and impaired blood perfusion (reduction in bone density, risk of bone fracture, decubitus ulcers, and thromboembolism) [55,56,57].
Parameters for progressive home-based functional electrical stimulation (h-b FES) training of long-term fully denervated human muscles. Figure shows a sample of a progressive FES training according to the described training in Table 11.1. It starts with bursts of a stimulation duration (SD) of 4 s and a stimulation pause (SP) of 2 s containing impulses with an impulse duration (ID) of 150 ms and an impulse pause (IP) of 400 ms for 4 months (it can be reduced if the time of denervation is under 6 months) to activate poor denervated muscle fibers. According to the enhanced activation, the ID can be reduced to 80 ms and SD increased to 5 s for another 4 months approximately. The next training phase implements tetanic bursts of 3 s SD and 3 s SP with impulses of 36 ms ID and 10 ms IP to increase muscle fiber diameter, muscle mass, density, and force, with leg extensions with and without additional weights on the ankles of the patients. If a good condition is achieved, depending not only to the training but also to the time span of denervation, standing, stepping-in-place, and walking exercises can be performed with continuous stimulation (controlled by an external switch) with 36 ms ID and 10 ms IP
Key Points
-
Elderly and mobility-impaired patients may develop severe limitations to their independence after arm and leg immobilization or being bedridden.
-
Education and support of patients to perform daily exercises in the hospital and then at home is an effective and low-cost measure to limit disability and improve physical and mental being of older patients, but when persons are reluctant or hampered to perform volitional exercise, functional electrical stimulation in the hospital and then at home is an alternative worth to be tested in many patients.
-
Section 11.1 discusses structural and molecular markers of muscle weakness and protocols of FES-induced recovery in aging and neuromuscular impairments.
-
Section 11.2 presents detailed protocols of h-b FES for denervated–degenerated muscles.
References
Gava P, Kern H, Carraro U (2015) Age-associated power decline from running, jumping, and throwing male masters world records. Exp Aging Res 41:115–135
McKinnon NB, Connelly DM, Rice CL et al (2016) Neuromuscular contributions to the age-related reduction in muscle power: mechanisms and potential role of high velocity power training. Ageing Res Rev 35:147–154
Mosole S, Rossini K, Kern H et al (2013) Significant increase of vastus lateralis reinnervation in 70-year sportsmen with a lifelong history of high-level exercise. Eur J Transl Myol Basic Appl Myol 23:117–122
Mosole S, Carraro U, Kern H et al (2014) Long-term high-level exercise promotes muscle reinnervation with age. J Neuropathol Exp Neurol 73:284–294
Mosole S, Carraro U, Kern H et al (2016) Use it or lose it: tonic activity of slow motoneurons promotes their survival and preferentially increases slow fiber-type groupings in muscles of old lifelong recreational sportsmen. Eur J Transl Myol Basic Appl Myol 26:339–346
Kern H, Carraro U, Adami N, Hofer C, Loefler S, Vogelauer M, Mayr W, Rupp R, Zampieri S (2010) One year of home-based Functional Electrical Stimulation (FES) in complete lower motor neuron paraplegia: recovery of tetanic contractility drives the structural improvements of denervated muscle. Neurol Res 32:5–12
Kern H, Boncompagni S, Rossini K, Mayr W et al (2004) Long-term denervation in humans causes degeneration of both contractile and excitation-contraction coupling apparatus that can be reversed by functional electrical stimulation (FES). A role for myofiber regeneration? J Neuropathol Exp Neurol 63:919–931
Hepple RT, Rice CL (2015) Innervation and neuromuscular control in ageing skeletal muscle. J Physiol 594(8):1965–1978
Carraro U, Kern H, Gava P et al (2016) Recovery from muscle weakness by exercise and FES: lessons from masters, active or sedentary seniors and SCI patients. Aging Clin Exp Res:1–12
Carraro U, Kern H (2016) Severely atrophic human muscle fibers with nuclear misplacement survive many years of permanent denervation. Eur J Transl Myol 26(2):5894
Lømo T (2014) The response of denervated muscle to long-term stimulation (1985, revisited here in 2014). Eur J Transl Myol Basic Appl Myol 24:13–19
Mitchell WK, Williams J, Atherton P et al (2012) Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front Physiol 3:260
Zampieri S, Mosole S, Löfler S et al (2015) Physical exercise in aging: nine weeks of leg press or electrical stimulation training in 70 years old sedentary elderly people. Eur J Transl Myol Basic Appl Myol 25:237–242
Zampieri S, Pietrangelo L, Loefler S et al (2015) Lifelong physical exercise delays age-associated skeletal muscle decline. J Gerontol A Biol Sci Med Sci 70:163–173
Kern H, Barberi L, Löfler S et al (2014) Electrical stimulation counteracts muscle decline in seniors. Front Aging Neurosci 6:189
Carnio S, LoVerso F, Baraibar MA et al (2014) Autophagy impairment in muscle induces neuromuscular junction degeneration and precocious aging. Cell Rep 8:1509–1521
Barberi L, Scicchitano BM, Musarò A (2015) Molecular and cellular mechanisms of muscle aging and sarcopenia and effects of electrical stimulation in seniors. Eur J Transl Myol Basic Appl Myol 25:231–236
Scicchitano BM, Rizzuto E, Musarò A (2009) Counteracting muscle wasting in aging and neuromuscular diseases: the critical role of IGF-1. Aging 1(5):451–457
Vinciguerra M, Musaro A, Rosenthal N (2010) Regulation of muscle atrophy in aging and disease. Adv Exp Med Biol 694:211–233
Carosio S, Berardinelli MG, Aucello M, Musarò A (2011) Impact of ageing on muscle cell regeneration. Ageing Res Rev 10:35–42
Musarò A (2014) The basis of muscle regeneration. Adv Biol 2014:1–16
Sandri M, Barberi L, Bijlsma AY et al (2013) Signalling pathways regulating muscle mass in ageing skeletal muscle: the role of the IGF1-Akt-mTOR-FoxO pathway. Biogerontology 14:303–323
Adamo ML, Farrar RP (2006) Resistance training, and IGF involvement in the maintenance of muscle mass during the aging process. Ageing Res Rev 5:310–331
Pelosi L, Berardinelli MG, De Pasquale L (2015) Functional and morphological improvement of dystrophic muscle by Interleukin 6 receptor blockade. EBioMedicine 2:274–275
Krenn M, Haller M, Bijak M et al (2011) Safe neuromuscular electrical stimulator designed for the elderly. Artif Organs 35:253–256
He Y, Huang C, Lin X, Li J (2013) MicroRNA-29 family, a crucial therapeutic target for fibrosis diseases. Biochimie 95:1355–1359
Cvecka J, Tirpakova V, Sedliak M et al (2015) Physical activity in elderly. Eur J Transl Myol Basic Appl Myol 25:249–252
Sarabon N, Löfler S, Hosszu G, Hofer C (2015) Mobility test protocols for the elderly: a methodological note. Eur J Transl Myol Basic Appl Myol 25:253–256
Hughes AM, Burridge JH, Demain SH et al (2014) Translation of evidence-based assistive technologies into stroke rehabilitation: users’ perceptions of the barriers and opportunities. BMC Health Serv Res 14:124
Bersch I, Tesini S, Bersch U, Frotzler A (2015) Functional electrical stimulation in spinal cord injury: clinical evidence versus daily practice. Artif Organs 39:849–854
Kern H, Hofer C, Moedlin M et al (2002) Denervated muscle in humans: limitations and problems of currently used functional electrical stimulation training protocols. Artif Organs 26:216–218
Kern H, Carraro U, Adami N, Biral D, Hofer C, Forstner C, Mödlin M, Vogelauer M, Pond A, Boncompagni S, Paolini C, Mayr W, Protasi F, Zampieri S (2010) Home-based functional electrical stimulation rescues permanently denervated muscles in paraplegic patients with complete lower motor neuron lesion. Neurorehabil Neural Repair 24:709–721. doi:10.1177/1545968310366129
Kern H, Carraro U (2014) Home-based Functional Electrical Stimulation (h-b FES) for long-term denervated human muscle: history, basics, results and perspectives of the Vienna Rehabilitation Strategy. Eur J Transl Myol Basic Appl Myol 24:27–40
Willand MP (2015) Electrical stimulation enhances reinnervation after nerve injury. Eur J Transl Myol 25(4):243–248. doi:10.4081/ejtm.2015.5243. Review
Catapano J, Willand MP, Zhang JJ et al (2016) Retrograde labeling of regenerating motor and sensory neurons using silicone caps. J Neurosci Methods 259:122–128. doi:10.1016/j.jneumeth.2015.11.020.
Willand MP, Nguyen MA, Borschel GH, Gordon T (2016) Electrical stimulation to promote peripheral nerve regeneration. Neurorehabil Neural Repair 30(5):490–496
Willand MP, Rosa E, Michalski B et al (2016) Electrical muscle stimulation elevates intramuscular BDNF and GDNF mRNA following peripheral nerve injury and repair in rats. Neuroscience 334:93–104
Coste CA, Mayr W, Bijak M, Musarò A, Carraro U (2016) FES in Europe and beyond: current translational research. Eur J Transl Myol Basic Appl Myol 26(4):6369. http://www.pagepressjournals.org/index.php/bam/article/view/6369/6187
Rossato E, Marziali A, Carraro U et al (2009) Rise2-Italy Project: muscle FES after peripheral nerve lesions. Basic Appl Myol 19:169–172
Carlson BM (2014) The biology of long-term denervated skeletal muscle. Eur J Transl Myol 24:3293
Midrio M (2006) The denervated muscle: facts and hypotheses. A historical review. Eur J Appl Physiol 98:1–21
Boncompagni S, Kern H, Rossini K et al (2007) Structural differentiation of skeletal muscle fibers in the absence of innervation in humans. Proc Natl Acad Sci U S A 104:19339–19344
Ashley Z, Sutherland H, Lanmüller H et al (2007) Atrophy, but not necrosis, in rabbit skeletal muscle denervated for periods up to one year. Am J Physiol Cell Physiol 292:440–451
Rossini K, Zanin ME, Carraro U (2002) To stage and quantify regenerative myogenesis in human long-term permanent denervated muscle. Basic Appl Myol 12:277–286
Squecco R, Carraro U, Kern H et al (2009) Despite lost contractility, a sub-population of rat muscle fibers maintains an assessable excitation-contraction coupling mechanism after long-standing denervation. J Neuropathol Exp Neurol 68:1256–1268
Kern H, Hofer C, Mayr W, Carraro U (2009) European Project RISE: partners, protocols, demography. Basic Appl Myol/Eur J Transl Myol 19:211–216
Hofer C, Forstener C, Moedlin M et al (2005) In vivo assessment of conduction velocity and refractory period of denervatd muscle fibers. Artif Organs 29:436–439
Hofer C, Mayr W, Stöhr H et al (2002) A stimulator for functional activation of denervated muscles. Artif Organs 26:276–279
Dr. Schuhfried Medizintechnik GMBH (2012) The “stimulette den2x” is a compact, high performance two-channel electrotherapy device that was developed to be used exclusively for stimulating denervated muscles. http://www.schuhfriedmed.at/en/stimulette-en/stimulette-den2x-2/
Lehmkühl LD, Dimitrijevic MR, Zidar J (1988) Lumbosacral evoked potential (LSEPs) and cortical somatosensory evoked potentials (SEPs) in patients with lesions of the conus medullaris and cauda equina. Electroencephalogr Clin Neurophysiol 71:161–169
Gutrecht JA (1994) Sympathetic skin response. J Clin Neurophysiol 11:519–524
Kern H, Ch H, Mayr W et al (2012) Elektrostimulation komplett denervierter Muskulatur. In: Fialka-Moser V (ed) Kompendium der Physikalischen Medizin und Rehabilitation. Springer, Vienna, pp 445–456
Kern H, Hofer C, Mayr W (2008) Protocols for clinical work package of the European Project RISE. Basic Appl Myol 18:39–44
Pieber K, Herceg M, Paternostro-Sluga T, Schuhfried O (2015) Optimizing stimulation parameters in functional electrical stimulation of denervated muscles: a cross-sectional study. J Neuroeng Rehabil 12:51. doi:10.1186/s12984-015-0046-0
Bizzarini E, Zampa A, Malisan C et al (2009) Epidemiology and FES of Conus-Cauda Syndrome in Friuli Venezia Giulia: data of the spinal unit of udine and report of a difficult-to-stimulate case. Basic Appl Myol 19:163–168
Czyrny JJ, Kaplan RE, Wilding GE et al (2016) Electrical foot stimulation: a potential new method of deep venous thrombosis prophylaxis. ERJ Open Res 2(1):20–27
Carraro U, Sandri M, Zampieri S (2015) Patologia Generale del Tessuto Muscolare Scheletrico. In: Pontieri GM, Russo MA, Frati L (eds) Patologia Generale. Piccin Nuova Libraria spa, Padova, pp 1523–42. ISBN 978-88-299-2036-5
Barberi L, Scicchitano BM, De Rossi M et al (2013) Age-dependent alteration in muscle regeneration: the critical role of tissue niche. Biogerontology 14:273–292
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Kern, H. et al. (2018). Functional Electrical Stimulation of Skeletal Muscles in Aging and Premature Aging. In: Masiero, S., Carraro, U. (eds) Rehabilitation Medicine for Elderly Patients. Practical Issues in Geriatrics. Springer, Cham. https://doi.org/10.1007/978-3-319-57406-6_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-57406-6_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-57405-9
Online ISBN: 978-3-319-57406-6
eBook Packages: MedicineMedicine (R0)