Abstract
Extensive resective surgery plays a major role in prolonging survival of glioma patients. However, patient’s will is not to survive as long as possible, but rather to enjoy the longest possible «normal» life. Because glioma infiltrates functional tissue, it is necessary to define resection boundaries by functional maps rather than by tumoral imaging. In this perspective, advances in brain mapping have deeply improved the surgical management of glioma patients. In this chapter, we summarize the preoperative and intraoperative methods that allows to map the brain and we review the cortical and axonal mapping of the main cognitive functions.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The goal of surgery in primary brain tumors is to optimize the extent of resection, in order to significantly increase the survival, while preserving or even improving quality of life (for example by controlling intractable epilepsy). In other words, the aim is to find the best “onco-functional balance”, that is, to give the opportunity to the patients to enjoy a normal life as long as possible. To this end, due to a considerable inter-individual anatomo-functional variability, structural landmarks are important but not enough. Furthermore, in essence, “tumoral limits” do not exist in diffuse gliomas, because these tumors migrate within brain parenchyma, especially along the white matter tracts. As a consequence, in the past decade, it has been proposed to switch from “image-guided surgery” to “functional mapping-guided surgery”, i.e., to achieve resection up to eloquent structures, both at cortical and subcortical levels. Indeed, advances in brain mapping have deeply improved the surgical management of glioma patients, regarding functional as well as oncological outcomes. In this chapter, we summarize the preoperative and intraoperative methods that allows to map the brain and we review the cortical and axonal mapping of the main cognitive functions.
Preoperative Mapping
The preoperative planning is likely to be the most important step in functional brain tumor resection. Apart the non-invasive techniques of brain mapping that will be discussed in this section, the preoperative time is essential for selecting the intraoperative tasks.
The choice of those tasks rely on three parameters:
-
location of the tumor, in relation to the knowledge we have about the functional anatomy and neural networks [1],
-
the deficit evidenced on extensive neuropsychological assessment. Indeed, any slight deficit in a cognitive domain testifies that plasticity limitations of the lesioned networks have been reached, meaning that this specific domain should be tested if it should be preserved (see for e.g. Chapter 19 in [2]),
-
in-depth discussion with the patient. The choice of functions to be preserved depends on the patient’s way of life (profession, hobbies …) [3].
Over the last twenty years, several methods have been developed to map preoperatively brain functions. Although none of these methods is reliable enough to get rid of awake brain mapping by direct electrical stimulation, their combined use can be of help when there is a contra-indication to awake surgery. We rapidly review these methods and refer the reader to more specific review papers on this topic.
Preoperative Cortical Methods
Task-Based fMRI (T-fMRI)
The link between non-invasive T-fMRI mapping and intraoperative DES mapping remains poorly understood. For primary motor areas, the degree of correlation is quite high [4], although not perfect (for e.g., sensitivity of 71% reported in [5]). The problem is far more complex for higher cognitive functions, like complex motor task or language. Previous studies have approached the problem through simple comparison between activated areas on task-based fMRI and DES eloquent sites. It has been concluded that sensitivity and specificity of T-fMRI (with respect to DES areas) are much too low to rely solely on T-fMRI for determining functional boundaries. Depending on the T-fMRI paradigm, some studies concluded to a high sensitivity and low specificity, and some others to the reverse [6]. Of note, the advent of 3T MRI did not improve the reliability of fMRI (for e.g., sensitivity of 37.1% and specificity of 83.4% in [7]). At least two caveats might explain the discrepancy between T-fMRI and DES. First, as mentioned in [8], the two methods are intrinsically different: DES will jam some networks preventing their functionality. But, through instantaneous dynamic reorganization of the undisturbed networks, the function can eventually still be implemented by compensatory networks. This would explain some false positive of fMRI (i.e. an activated area on T-fMRI is not found eloquent by DES). To explain false negatives of fMRI, there are two possibilities: either they correspond to “false positives of DES” (i.e. removal of an eloquent DES areas would not cause any deficit) or it is a real false negative of fMRI (the site is eloquent and not detected by fMRI). This last situation is grounded by our recent understanding of the link between neuronal activity and BOLD signal. Indeed, it has been shown that BOLD contrast indicate areas with input and local computations rather than output spiking activity [9].
In the same vein, a recent study has shown that in temporal regions, high-gamma band power (which is believed to be the best surrogate of BOLD signal) on electrocorticography during picture naming and word reading vanishes after a short duration of 10 s [10]. On the contrary, in frontal areas, this activity lasted all along the 60 s of analysis. Such dissociation in neuronal activity between frontal and temporal areas during a language task likely explains that temporal areas are more difficult to detect on T-fMRI than frontal areas (see [11] and references therein).
Rest-Based fMRI (R-fMRI)
Although the first observation of low-frequency (0.1 Hz) correlations within distinct brain networks in a resting subject dates back to 1995 [12], it is only 10 years later that a seminal paper offered to use this technique to perform a segregation of brain areas in different functional networks [13]. Among others, have been recognized motor, language, attention (dorsal and ventral), and default mode networks. According to preliminary reports, R-fMRI could be a promising tool, with better correlations with DES [14, 15]. Moreover, a very important study revealed a high degree of correlations between R-fMRI and cortico-cortical evoked potentials (CCEPs) [16]. Last but not least, positive and negative correlations exhibited by R-fMRI have been found to exist also electrophysiologically (by measuring the correlations in the high-gamma band) [17]. All these datas support the idea that the partitioning of the brain based on low-frequency correlations in R-fMRI could be a powerful method to achieve in a very short time a global brain mapping for each patient.
rTMS: Motor/Language
The interest of rTMS regarding motor function mapping has been established. The motor maps obtained through neuronavigated rTMS are highly correlated with the intraoperative maps obtained by DES [18]. Moreover, for high-grade tumors, due to edema and vascular redistribution, T-fMRI might be unable to locate primary motor areas, whereas rTMS is still effective. For that reason, rTMS appears to be the gold standard for preoperative mapping of primary motor areas. Nonetheless, as it will be discussed in the paragraph about intraoperative methods, there is currently no reports testing by rTMS higher motor functions (like grasping, fine movements, bimanual coordination …). Moreover, there are conflicting results regarding the interest of rTMS for mapping language functions [19, 20]. Its specificity for language mapping is only 23.8%, with a positive predictive value of 35.6% according to [21]. A recent study investigated the relationship between T-fMRI, rTMS and DES maps [22]. It was found that rTMS was more sensitive compared to DES. On the contrary, T-fMRI was not enough sensitive with regards to DES. More specifically, T-fMRI failed to detect temporal language areas, as previously discussed.
Preoperative Axonal Mapping
Preoperative tractography is gaining interest among neurooncological surgeons. This MRI modality enables to locate the main white matter fiber tracts of the brain. In the past few years, several new algorithmic methods have been developed to infer fibers directions from diffusion-weighted images. Diffusion tensor estimation is the most widely used. It allows to draw RGB maps superimposed on a 3D-T1 anatomy, which is the simplest way to visualize white matter anisotropy. But this diffusion tensor estimation was unable to resolve the problem of crossing fibers. Two different new categories of algorithm attempted to overcome this limitation: the constrained spherical deconvolution and q-ball. Finally, it should be reminded that there is another layer of algorithms to determine the continuous line of a pathway: tracking algorithms. These algorithm are classified in two broad categories: deterministic or probabilistic. Tractograms can then be uploaded in the neuronavigation systems, allowing to correlated images with intraoperative stimulation, both for motor functions of the pyramidal tract and language functions of the dorsal and ventral streams. However, clinical interest of tractography is quite limited for two reasons:
-
First, the variability of the trackings with the different methods. CSD and q-ball are supposed to resolve the problem of crossing fibers, however, there is currently no way to validate the obtained tractograms. As a consequence, a recent study by the DTI challenge concluded that there are still limitations for clinical use of DTI in neurosurgery [23]. Moreover, the problem of kissing fibers is even more challenging.
-
Second, even if tractography would be perfect, this imaging method cannot inform us about the functional deficit that would be encountered after resection of a tracked fasciculus.
All in all, preoperative mapping methods are becoming more informative about individual brain functional and structural anatomy. It can be anticipated that datas coming from all these methods (T-fMRI, R-fMRI, tractography, rTMS) could be integrated by means of biocomputationals models of the brain, allowing better correlations with DES and surgical outcome.
Intraoperative Mapping
Cortical Mapping Under General Anesthesia
Motor: DES Versus Train of Five
The primary motor areas can be identified under GA by direct electrical stimulation. This seminal technique, introduced by Penfield, consists to apply a 60 Hz current for a few seconds (what we call Ojemann stimulation, OS), until a movement is elicited. About 20 years ago, the train of five (To5) technique has been introduced [24]. Since then, very few papers have studied the pro and cons of the two techniques. In a large series of glioma patients, Bello et al. very recently compared the two techniques and concluded that whereas To5 is always applicable, OS is not recommended for cases with increased excitability [25]. This excitability can be predicted from preoperative parameters, including long seizure history, diffuse margins on FLAIR, infiltration of CST, preoperative deficits.
However, it should be kept in mind that theTo5 technique is more challenging in terms of equipments and that interpretation of motor-evoked potentials has to be made by a qualified neurophysiologist. For this reason, many teams prefer to awake the patient also for motor functions.
Non-motor Functions: Cortico-Cortical Evoked Potentials
Up to now, awake surgery was the only way to identify cortical areas eloquent for non-motor functions. However, the technique of CCEP, initiated in 2004 by Matusmoto et al. (although there are some sporadic earlier reports cited in the paper of Matsumoto et al.) shows promising results regarding the possibility to map language functions under GA. In their initial report, these authors have shown, in an extraoperative recordings of grids put all over the perisylvian language areas, that 1 Hz stimulation of anterior frontal language areas (as detected by functional disturbances at 60 Hz stimulation) generates CCEPs in a wide posterior temporal region (including the 60 Hz temporal language site). And reciprocally, the posterior temporal language site also elicited CCEPs in the frontal operculum. Importantly, it has been recently shown the shape of these CCEPs is quite similar in awake and asleep patients [26]. More studies are needed to evaluate the clinical value of this new tool.
Cortical Mapping in Awake Condition
Several techniques can be used for intraoperative mapping in an awake patient. The first kind of methods is just to record electrical activity (by electrocortiograms, ECoG) while the patient is performing a task. Although high-gamma activity (HGA) of ECoG was the focus of several previous studies [27–31], it is only recently that this activity was analyzed on-line for language mapping [10]. It was shown to be highly correlated with direct electrical stimulation. This method is appealing, because all the brain surface can be mapped in a very short period of time. However, this technique is quite sophisticated, limiting currently its spread in daily practice [32].
Moreover, it should be noted that, similarly to T-fMRI, HGA mapping is in essence inadequate for distinguishing participating areas from essential ones. Hence, only methods interfering with neuronal networks can make the distinction. There are two different ways to interfere with brain networks: cooling and electrical stimulation. Cooling as been reported once in humans, and showed somewhat different results compared to electrical stimulation [33]. For some reason, this method has not been used by any other team to our knowledge, and consequently, we will focus, in the next sections, on brain mapping by direct electrical stimulation.
Sensori-Motor Functions
In comparison to the motor mapping that can be done asleep, awake motor mapping offers the possibility to test not only the ability to move, but also to perform complex movements or motor behavior. Although there exists a variety of tasks (repetitive movement of flexion and extension of the different segments of the superior limb, grasping, using a screw-driver, in-phase and out-of-phase bimanual movements), their use remains rare during awake surgery. In fact, the first cases have been reported during extraoperative mapping for epilepsy surgery [34, 35]. A recent review [36] discussed these so called negative motor areas (negative, because they stop the on-going motor behavior). Two clusters have been found on each hemisphere: the posterior end of inferior frontal gyrus (pars opercularis) together with the lower end of precentral gyrus on the lateral surface, and the SMA on the mesial surface. In a somehow contradictory study about the exact location of these sites on the superior frontal gyrus, it has been shown recently that, in fact, 95% of sites blocking repetitive complex movements or bilateral coordination are located in the convexity of the superior frontal gyrus (see also Fig. 5.1), while only 5% are located in the mesial part of the superior frontal gyrus [37]. Last but not least, awake condition allows the patient to report about sensory functions: for example, he can warn the surgeon by telling during stimulation (usually of parietal areas) «I do not feel my leg anymore» and the preservation of these sites is crucial to keep such a fundamental function as walking.
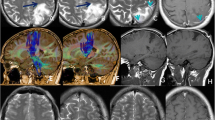
Illustrative case of a 50 years-old frustrated left-handed patient diagnosed with a left frontal glioblastoma revealed by headaches and language disturbances. a Preoperative T1-gadolinium enhanced MRI, showing a large enhancing mass of the superior frontal gyrus, extending towards the ventricle and the head of the caudate nucleus. b Intraoperative mapping, cortical mapping evidenced a complete anarthria in the parietal operculum (tag 1), primary motor area of the thumb (tag 2), negative motor areas, stopping repetitive movement of upper limb (tag 3 and 4), and disturbances of PPTT with preservation of picture naming (tag 5). Axonally, resection was stopped in the lateral vicinity of the head of the caudate nucleus, where stimulation at the cross-road between aslant tract and IFOF generated perseverations and semantic paraphasias. c Postoperative FLAIR MRI showing a small residue in the posterior wall (the enhancement was completely removed). Patient was then treated with radiotherapy with concomitant Temozolomide, followed by 6 cycles of Temozolomide. He was able to resume his professional activity of sales representative full time. He remained recurrence-free for 18 months
Primary Visual Areas
The preservation of the primary visual areas on both sides of the calcarine sulcus is in some cases of utmost importance. Indeed, it should be reminded to the patient that the driving license is no more valid in case of hemianopsia. As initially reported in 1968 [38], electrical stimulation generates mainly positive visual phenomena, like phosphene, flash of light, … Of note, as for any primary motor or sensory areas, there is almost no plasticity, meaning that anatomical landmarks allow to locate primary visual areas reliably.
Language
Language is the most widely mapped function during awake surgery. Since the very first reports by Penfield, the technology did not change that much: applying electrical stimulation for a period of 3–4 s disturbs the language task. However, the classification of the different types of observed errors has been only recently clarified: it is of utmost importance to distinguish motor arrest, speech arrest, and anomia. A simple algorithm has been proposed, in order to optimize the initial testing for differentiating the areas to these different types of error [39]. Most commonly, the language assessment is performed through a picture naming task. This raises a still debated question: is it enough to assess a function as complex as language just by naming pictures? In a first response, it is important to notice that picture naming allows to investigate the two main components of language: phonology and semantics [40]. Indeed, the speech therapist can make the on-line distinction between phonological paraphasia (e.g. log instead of dog) versus semantic paraphasia (for e.g. cat instead of dog) [41]. The current anatomo-functional model maps these two components on the dorsal pathway for phonology and ventral stream for semantics [42]. However, on the cortical surface, there is no clear-cut spatial segregation between phonological and semantic sites [43]. There is for each domain, three clusters in the left hemisphere: one in the pars triangularis, one at the posterior end of the middle frontal gyrus, and one in the posterior part of the superior temporal gyrus. The great well known inter-patients variability [44] proves once again the necessity of intraoperative mapping in an awake patient. Of note, there is a striking resemblance between this map and the clusters evidenced by a meta-analysis of T-fmRI studies [45]. This overlapping suggests that the phonological and semantic systems are not anatomically separable at the 5 mm scale at the cortical level. Of note, when the patient’s response is an anomia, one cannot conclude whether this is a disturbance of the phonological or semantic system, or both.
Hence, the ability to name a picture insures that both phonological and lexico-semantic abilities are preserved. However, it should be kept in mind that naming does not warrant full semantic judgment. Indeed, a dissociation has been shown between picture naming and pyramid-palm-tree test (PPTT), a supramodal semantic association task: in some fronto-opercular and superior temporal sites (see also Fig. 5.1), patients were able to name pictures, without being able to make semantic association [46, 47].
Moreover, language requires more than phonology and basic semantics: syntax, that is the ability to find the meaning of a sentence from rules and links between words, is also an essential part of language. Interestingly, although some patients exhibit difficulties to understand syntactically complex sentences in the immediate postoperative course, permanent syntactic deficit after picture-naming based awake surgery are rarely observed [48]. Similarly, there are no reported case of patients with a long lasting deficit of repetition (i.e. a language task with an auditory rather than visual input). Still, many authors have proposed numerous tasks for better evaluating language intraoperatively (see following review papers [49–51]), and some new protocols are still under investigation [52, 53].
Nevertheless, it should be mentioned that proper name deficits have been reported after left anterior temporal lobectomy [54, 55]. Whereas this deficit can be prevented by adding a task of naming famous faces, this should be balanced on a case-by-case basis with the oncological benefit: indeed, the impact in daily life of an inability to name proper names is highly variable for each patient.
Similarly, one should not forget to test patients in mother-tongue and secondary learned languages for bilingual patients [56–60].
Last but not least, reading is an essential part of language abilities. Removal of visual word form area (VWFA) has been shown to result in long lasting reading deficit [61], while its identification by DES allows to preserve reading abilities [62, 63]. Apart in the basal temporo-occipital areas, different studies reported specific reading disturbances when stimulating posterior superior left temporal gyrus and left supramarginal gyrus [64], but also posterior part of inferior and middle left frontal gyrus [65]. These latter sites were generally superimposed or close to naming sites, meaning that they would have been preserved even if lecture would not have been tested. Finally, stimulation of FEF areas also induced reading troubles, by generating involuntary ocular movements (but this area can be detected just by looking at eyes movements [66]). On a practical point of view, these results show that reading per se needs to be tested only in (left) dominant posterior temporo-occipital regions. Of note, disturbances were very infrequently observed in the anterior temporal lobe. This is somehow in disagreement with the triangle model, in which it has been shown recently that the semantico-phonological conversion in reading is thought to be supported by the anterior temporal lobe [67]. Moreover, in this model, the role of posterior inferior and middle frontal regions was not assigned. Hence, electrostimulation datas should be better integrated in the currents neurocomputational models of reading.
Spatial Consciousness
It has been well known from stroke studies that right parietal lobe lesions can result in spatial neglect of the opposite hemi-space. Because such deficit can be as debilitating in daily life, the importance of preventing this syndrome cannot be overemphasized. In a seminal report in 2005, Thiebaut de Schotten et al. described two cases of right parietal tumor resection with spatial consciousness monitoring [68]. The bisection test is very simple: the patient is asked to draw the middle of a 20 cm line. In case of unilateral spatial neglect (transiently generated by the electrical stimulation), patient will deviate on the right of the line. Two cortical deviations sites were identified, in the caudal superior temporal gyrus and in the supramarginal gyrus. Since then, two studies reported larger series of spatial consciousness mapping [69, 70]. Roux et al. found several cortical regions with rightward but also leftward deviations. Those regions were centered around the temporo-parietal junction and deviations in the superior parietal lobule were uncommon. On the contrary, Vallar et al. observed rightward deviations mainly in Broadman’s area 7b. Like for language, it is likely that spatial consciousness is supported by a distributed set of interconnected cortical areas.
Calculation
The neural correlates of mental calculation have been intensively studied by T-fMRI studies. It involves a large set of areas, grouped in three different networks [71]: the bilateral horizontal segment of intraparietal sulcus for quantity processing, the left angular gyrus for numbers manipulation in verbal modality, and the bilateral superior parietal regions for focusing attention (and this network is not specific to number manipulations). Accordingly, several authors reported disruption of calculation in various cortical areas, such as left angular gyrus [72–74], right angular gyrus [75, 76], left superior parietal lobule [77], horizontal portion of left intraparietal sulcus and left supramarginal gyrus [74]. In all these studies, some sites were specific to calculation and even to a subtype of computations (susbtraction vs. multiplication), while some others were also related to language disturbances. It should be noted that, in comparison with language mapping, there are very few reports about mental calculation mapping. This might be in relation to the relative rarity of parietal diffuse low-grade glioma. Moreover, the functional benefit of preserving mental calculation should be balanced with the oncological benefit to remove these areas: indeed, except for some very specific professions requiring mathematical abilities, patients usually do not care to keep their mental calculation abilities (probably because we are more and more relying on computers for daily arithmetic).
Double Task
The importance in efficient cognition of being able to perform simultaneously two tasks cannot be overemphasized. Fortunately, this can be easily tested intraoperatively: a motor task can be added to any other cognitive task. Hence, patients can be asked to do picture naming or PPTT or mental calculation while simultaneously performing a repetitive movement of the upper limb. This double task greatly enhances the sensitivity to electrical stimulation, that is, some sites will respond only to the double task, while each task separately would be efficiently performed [42]. Again, depending on the preoperative discussion with the patient, one can decide to preserve or not such areas requiring a high-level of cognitive functioning.
Mentalizing
Emotions have a major influence in daily life, particularly for decision making. Assessing the subjective experience of others in terms of mental states is a brain function referred to as mentalizing. Recent theories hypothesizes a two levels hierarchical network: a low-level network for emotion recognition or motor intention (theory of mind), and a higher-level network of mentalizing per se for complex inferences about other’s state of mind and intentions. The low-level network is supposed to be linked to the mirror neuron system, whereas the higher-level is related to the default-mode network, in particular the ability to attribute the intentions of others [78]. Interestingly, the low-level network can be monitored intraoperatively by the Read the Mind in the Eyes test. A first study reported responses in the right superior temporal gyrus, middle temporal gyrus and supramarginal gyrus [79]. In another study, responses were found in the pars opercularis and triangularis of the right frontal operculum [80]. If it is safer to preserve these sites, it should be noted that this function is likely to be distributed over redundant areas, ensuring a high plastic potential: indeed, in a series without intraoperative testing, only two patients out of ten kept an impairment on the assessment 3 months after surgery [81]. Of note, whereas mentalizing per se can be objectively evaluated with the comic strips task, there is currently no way to test the higher-level network in an intraoperative setting. It could be anticipated that this higher-level function is even more prone to plasticity than its lower-level counterpart. This would explain the very low risk of permanent deficit (10%).
Other Self-reported Effects of Stimulations
A major advantage of on-line monitoring during awake surgery is the possibility for the patient to continuously report any inner conscious and subjective feeling induced by electrical stimulation. For example, intention to move (by stimulation in the posterior parietal cortex [82]) or out-of-body experiments (interpreted as a disruption of multisensory information by stimulating the (right) temporo-parietal junction [83]) have been described by patients.
In the same vein, disruption of consciousness of the external environment (induced by stimulating the ventral part of the posterior cingulate cortex) has been reported [84]. After recovering from the stimulation, the patients could describe their state as if «in a dream».
Axonal Mapping Under General Anesthesia
Motor: DES Versus Train of Five
The To5 technique is becoming a popular tool for axonal mapping of the cortico-spinal tract.
Continuous monitoring can be achieved by stimulating with the resective surgical tool, either the ultrasonic aspirator [85] or a suction device [86].
All in all, To5 appears to be a powerful method, for both cortical and axonal identification of cortico-spinal pathway. However, it should be kept in mind that with this technique, it is currently not possible to assess higher order motor function (like grasping, fine movements, bimanual coordination …) and the only way to monitor these functions is intraoperative electrical mapping in an awake patient.
Non-motor Functions: ACEPs
Until very recently, there was no technique to map non-motor function axonally under GA. A very recent study reported the identification of the arcuate fasciculus by axono-cortical evoked potentials [26]. The basic idea is to stimulate at 1 Hz during white matter removal and to record in anterior and posterior language areas identified by CCEPs. The reliability of this technique needs to be assessed further, but seems promising.
Axonal Mapping in Awake Patients
Motor Functions
The possibility to test repetitive movements and bimanual coordination opened new avenues in the axonal mapping of motor functions. In particular, the stimulation of the fibers located in the depth of the precentral sulcus generates impairments of repetitive movements of unilateral or bilateral limbs. It has been argued that the stimulated pathway has direct projection to the spinal cord [87]. In addition, the network for motor control might also involve the frontal aslant tract (linking the SMA to the pars opercularis/vPMC) and the fronto-striatal tract [88, 89]. This is not surprising, considering that the aslant tract makes the link between the two clusters of «negative motor areas». From past experience in surgery with motor mapping under GA, it is known that resection of these pathways will lead to the so called «SMA syndrome», with a transient akinesia (and mutism on the left dominant side). Again, the oncological benefit should be carefully balance for each patient: it is certainly important to preserve a high level of motor coordination in a tennis player or a pianist, while it might be not necessary for a sales manager.
Language Function
Since the seminal paper in 2002 [90], great advances have been made in our understanding of error patterns elicited by white matter pathways stimulation. These advances were concomitant with the development of new neuropsychological models of language (identifying phonological and semantic as the two main subsystems [40], and hypothesizing that they are sustained anatomically by two parallel pathways, the dorsal and ventral streams respectively [91]) and with the (re)-discovery of white matter anatomy by diffusion tensor imaging and cadaveric fiber dissections. For example, the inferior fronto-occipital fasciculus has been rediscovered by DTI [92] and detailed by fiber dissections [93, 94], and new pathways (aslant [95–97], middle longitudinal fasciculus [98–100]) were discovered by virtual dissections and then confirmed by fiber dissections [96, 97, 101]. On the other side, as explained above, tracking algorithms can lead to unrealistic pathways: for example, the trajectory of the SLF I as described by DTI [102] is the subject of debate among fiber dissection experts (see [103] for initial controversy, and [104] for recent discussion). In the same vein, a long standing debate about the putative existence of a superior occipital-frontal fasciculus seems to be resolved [105].
The datas gathered from axonal mapping can be summarized in two ways:
-
Probabilistic maps, either of functional tumor remnants [106] or of eloquent sites [107].
-
Neuropsychological models with anatomical substrate [41, 42].
In brief, phonological errors are more frequently encountered by stimulation of the arcuate fasciculus [108, 109], while perseverations and semantic errors are more frequently encountered by stimulation of IFOF (see Fig. 5.1) [110, 111]. This is in line with the dual stream model of language [91, 112]. Articulatory aspects are supported by the SLF III [113], while disturbances of speech fluency are observed in relation to the aslant and fronto-striatal tracts [114, 115], linking the SMA area to the pars opercularis/vPMC and the head of caudate nucleus respectively. The connectivity of the reading system has also been investigated [116]. The results show that: stimulation of inputs to the VWFA (i.e. the posterior part of the inferior longitudinal fasciculus) induces complete alexia; stimulation of the white matter anteriorly adjacent to the VWFA induces difficulties for irregular words reading (i.e. for the semantic/addressed pathway in the triangle model); while stimulation of the white matter located superiorly to the VWFA generates difficulties for both irregular and pseudo-words, but not for regular words. This intriguing observation suggest that the posterior part of the arcuate fasciculus is involved in the executive control that normally regulates the balance between the semantic/addressed and phonological/assembled pathways.
Visual Functions
Whenever the patients cannot accept to live with a hemianopsia, the visual pathways should be preserved by DES mapping. The anatomical complexity of the visuals pathways has recently been revisited, thanks to DTI and fiber dissection studies [117–121]. It should be mentioned that intraoperative testing of vision in a hemi-field remains a challenge, because axonal stimulation can generate visual defect rather than positive phenomena. Hence, it is necessary to use for example picture naming with images distributed in the different quadrants. However, because the patient can compensate with ocular movements, this is not a 100% reliable methodology [122] and this should be taken into account in the onco-functional balance [3].
Spatial Consciousness
In a seminal study [68], the 2nd branch of the superior longitudinal fasciculus was identified as the tract whose stimulation generated rightward deviations in line bisection test. Since this first description, two other teams reported their observations of intraoperative line bisection [69, 70]. Results are not easy to compare, as methodology were slightly different (line on paper versus tablet, use of right hand or left hand to mark the midpoint). Interestingly, leftward as well as rightward deviations were observed. Remarkably, both studies agreed that it is the stimulation of the second branch of the superior longitudinal fasciculus that generates massive deviations. How these observations (and especially the leftward deviations) could be integrated in the general model of spatial neglect as an interaction between right dominant stimulus-driven ventral attention network (sustained by SLF III) and goal-directed bilateral dorsal attention network (sustained by SLF II) [123] remains an open question.
Mentalizing
A lesion study suggested that disconnection of the right arcuate fasciculus and/or SLF III was associated with poorer low-level mentalizing, while disconnection of the right cingulum was associated with poorer high-level mentalizing [78]. Accordingly, stimulation of the white matter of the right frontal operculum generated error in the Read the Mind in the Eyes test, at a location that could correspond to the terminations of the the arcuate fasciculus/SLF III [80]. More studies are needed to make the link with the more posterior sites observed cortically, that is to stimulate the arcuate fasciculus/SLF III in the depth of a tumor located to the right supra-marginal gyrus.
Mental Calculation
While a first study did not report any disturbances of mental calculation when stimulating white matter of the left angular gyrus [73], such effects were observed when stimulating the white matter in the right parietal lobe [124].
Plasticity and Remapping
Whenever the resection has been pushed until encountering functional responses in an awake patient, an immediate postoperative decline is usually observed in one or several cognitive domains. The onset of this deterioration is not always immediate, and usually takes place between postoperative day 1 and 3 [125]. The classical explanation of this delay is that deterioration is concomitant to the peak of edema. In addition, it can be hypothesized that in the first postoperative days, the brain is massively reorganizing its connection weights, resulting in a transient abnormal functioning. Recovery usually occurs in two steps: a rapid spontaneous postoperative recovery in the first week after deterioration, and a slow rehabilitation-guided long-term recovery (lasting about 3 months). The importance of intensive rehabilitation cannot be overemphasized. It should be started as soon as possible, right after the surgery [126, 127], and it should be under the supervision of a trained speech therapist or neuropsychologist. On a patho-physiological point of view, this long-term recovery is directly related to the patient’s potential of plasticity, and it has been observed that it took longer time in older patients.
All in all, for low-grade glioma, postoperative plasticity is driven first by rehabilitation and then by the slow regrowth of the tumor. As a consequence, it should be kept in mind that, thanks to this plasticity, responsive sites identified during a first surgery might be unresponsive some years later. This opens the possibility to reoperate on low-grade glioma and to remove the second time some areas found eloquent at first surgery—thus to increase the extent of resection without eliciting permanent functional deficits [128, 129].
Conclusions
Advances in brain mapping techniques have allowed a better understanding of the dynamic organization of human brain, i.e. in large-scale, parallel, delocalized and interactive sub-networks. Therefore, anatomical landmarks are not enough to preserve an optimal quality of life in brain tumor patients undergoing maximal resective surgery: individual functional mapping is mandatory to tailor the resection according to cortical and subcortical eloquent structures for each patient. Because non-invasive preoperative functional neuroimaging is currently not reliable enough to identify the cortices and white matter tracts crucial for brain processing, in particular with regard to high order cognitive functions, intraoperative mapping using direct electrical stimulation is the goal standard to remove diffuse gliomas. In awake patients, it is now possible to achieve an extensive mapping, not only of sensorimotor and language functions, but also of cognitive and emotional functions. Cortical and axonal stimulation enables a precise investigation of the neural circuits of glioma patients, to detect a possible remapping elicited by the tumor itself, in addition to the interindividual variability, and to define in real-time the boundaries of surgical resection in order to improve both the oncological results (e.g. by performing a supratotal resection, extended beyond the enhancement in high-grade gliomas and beyond the FLAIR abnormalities in low-grade glioma) as well as the functional outcomes. The ultimate goal is to increase the quantity of quality of life, based upon a personalized surgical strategy taking into account the wishes of the patient. Therefore, a comprehensive explanation of the natural history of the disease, but also the determination of the individual quality of life (according notably to the job and hobby of the patient) is essential before the surgical act, with the aim to adapt the selection of cognitive tasks during resection, and then to optimize the onco-functional balance of surgery.
Abbreviations
- ACEPS:
-
Axonal-cortical evoked potentials
- CCEPs:
-
Cortico-cortical evoked potentials
- CST:
-
Cortico-spinal tract
- DES:
-
Direct electrical stimulation
- DTI:
-
Diffusion tensor imaging
- ECoG:
-
Electrocortiograms
- HGA:
-
High-gamma activity
- IFOF:
-
Inferior fronto-occipital fasciculus
- PPTT:
-
Pyramid-palm-tree test
- R-fMRI :
-
Rest-based fMRI
- rTMS:
-
Repetitive transcranial magnetic stimulation
- SLF:
-
Superior longitudinal fasciculus
- SMA:
-
Supplementary motor area
- T-fMRI:
-
Task-based fMRI
- vPMC:
-
Ventral premotor cortex
- VWFA:
-
Visual word from area
References
Fernández Coello A, Moritz-Gasser S, Martino J, Martinoni M, Matsuda R, Duffau H. Selection of intraoperative tasks for awake mapping based on relationships between tumor location and functional networks. J Neurosurg. 2013;119(6):1380–94.
Diffuse Low-Grade Gliomas in Adults—Natural History, Interaction with the Brain, and New Individualized [Internet]. Springer. Hugues Duffau; [cited 2013 Dec 8]. Available from: http://www.springer.com/medicine/oncology/book/978-1-4471-2212-8.
Duffau H, Mandonnet E. The, “onco-functional balance” in surgery for diffuse low-grade glioma: integrating the extent of resection with quality of life. Acta Neurochir (Wien). 2013;155(6):951–7.
Gabriel M, Brennan NP, Peck KK, Holodny AI. Blood oxygen level dependent functional magnetic resonance imaging for presurgical planning. Neuroimaging Clin N Am. 2014;24(4):557–71.
Bartos R, Jech R, Vymazal J, Petrovický P, Vachata P, Hejcl A, et al. Validity of primary motor area localization with fMRI versus electric cortical stimulation: a comparative study. Acta Neurochir (Wien). 2009;151(9):1071–80.
Giussani C, Roux F-E, Ojemann J, Sganzerla EP, Pirillo D, Papagno C. Is preoperative functional magnetic resonance imaging reliable for language areas mapping in brain tumor surgery? Review of language functional magnetic resonance imaging and direct cortical stimulation correlation studies. Neurosurgery. 2010;66(1):113–20.
Kuchcinski G, Mellerio C, Pallud J, Dezamis E, Turc G, Rigaux-Viodé O, et al. Three-tesla functional MR language mapping: comparison with direct cortical stimulation in gliomas. Neurology. 2015;84(6):560–8.
Mandonnet E. Intraoperative electrical mapping: advances, limitations and perspectives. In: MD PHD, editor. Brain mapping [Internet]. Springer Vienna; 2011 [cited 2016 Feb 6]. p. 101–8. Available from: http://springerlink.bibliotecabuap.elogim.com/chapter/10.1007/978-3-7091-0723-2_8.
Logothetis NK. The neural basis of the blood-oxygen-level-dependent functional magnetic resonance imaging signal. Philos Trans R Soc Lond B Biol Sci. 2002;357(1424):1003–37.
Ogawa H, Kamada K, Kapeller C, Hiroshima S, Prueckl R, Guger C. Rapid and minimum invasive functional brain mapping by real-time visualization of high gamma activity during awake craniotomy. World Neurosurg. 2014;82(5):912.e1–10.
Kunii N, Kamada K, Ota T, Kawai K, Saito N. Characteristic profiles of high gamma activity and blood oxygenation level-dependent responses in various language areas. Neuroimage. 2013;15(65):242–9.
Biswal B, Yetkin FZ, Haughton VM, Hyde JS. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med. 1995;34(4):537–41.
Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci USA. 2005;102(27):9673–8.
Mitchell TJ, Hacker CD, Breshears JD, Szrama NP, Sharma M, Bundy DT, et al. A novel data-driven approach to preoperative mapping of functional cortex using resting-state functional magnetic resonance imaging. Neurosurgery. 2013;73(6):969–982; discussion 982–983.
Qiu T, Yan C, Tang W, Wu J, Zhuang D, Yao C, et al. Localizing hand motor area using resting-state fMRI: validated with direct cortical stimulation. Acta Neurochir (Wien). 2014;156(12):2295–302.
Keller CJ, Bickel S, Entz L, Ulbert I, Milham MP, Kelly C, et al. Intrinsic functional architecture predicts electrically evoked responses in the human brain. Proc Natl Acad Sci USA. 2011;108(25):10308–13.
Keller CJ, Bickel S, Honey CJ, Groppe DM, Entz L, Craddock RC, et al. Neurophysiological investigation of spontaneous correlated and anticorrelated fluctuations of the BOLD signal. J Neurosci. 2013;33(15):6333–42.
Picht T, Schmidt S, Brandt S, Frey D, Hannula H, Neuvonen T, et al. Preoperative functional mapping for rolandic brain tumor surgery: comparison of navigated transcranial magnetic stimulation to direct cortical stimulation. Neurosurgery. 2011;69(3):581–588; discussion 588.
Krieg SM, Shiban E, Buchmann N, Gempt J, Foerschler A, Meyer B, et al. Utility of presurgical navigated transcranial magnetic brain stimulation for the resection of tumors in eloquent motor areas. J Neurosurg. 2012;116(5):994–1001.
Krieg SM, Tarapore PE, Picht T, Tanigawa N, Houde J, Sollmann N, et al. Optimal timing of pulse onset for language mapping with navigated repetitive transcranial magnetic stimulation. Neuroimage. 2014;15(100):219–36.
Picht T, Krieg SM, Sollmann N, Rösler J, Niraula B, Neuvonen T, et al. A comparison of language mapping by preoperative navigated transcranial magnetic stimulation and direct cortical stimulation during awake surgery. Neurosurgery. 2013;72(5):808–19.
Ille S, Sollmann N, Hauck T, Maurer S, Tanigawa N, Obermueller T, et al. Impairment of preoperative language mapping by lesion location: a functional magnetic resonance imaging, navigated transcranial magnetic stimulation, and direct cortical stimulation study. J Neurosurg. 2015;123(2):314–24.
Pujol S, Wells W, Pierpaoli C, Brun C, Gee J, Cheng G, et al. The DTI challenge: toward standardized evaluation of diffusion tensor imaging tractography for neurosurgery. J Neuroimaging. 2015;25(6):875–82.
Taniguchi M, Cedzich C, Schramm J. Modification of cortical stimulation for motor evoked potentials under general anesthesia: technical description. Neurosurgery. 1993;32(2):219–26.
Bello L, Riva M, Fava E, Ferpozzi V, Castellano A, Raneri F, et al. Tailoring neurophysiological strategies with clinical context enhances resection and safety and expands indications in gliomas involving motor pathways. Neuro-oncology. 2014;16(8):1110–28.
Yamao Y, Matsumoto R, Kunieda T, Arakawa Y, Kobayashi K, Usami K, et al. Intraoperative dorsal language network mapping by using single-pulse electrical stimulation. Hum Brain Mapp. 2014;35(9):4345–61.
Canolty RT, Edwards E, Dalal SS, Soltani M, Nagarajan SS, Kirsch HE, et al. High gamma power is phase-locked to theta oscillations in human neocortex. Science. 2006;313(5793):1626–8.
Canolty RT, Soltani M, Dalal SS, Edwards E, Dronkers NF, Nagarajan SS, et al. Spatiotemporal dynamics of word processing in the human brain. Front Neurosci. 2007;1(1):185–96.
Edwards E, Nagarajan SS, Dalal SS, Canolty RT, Kirsch HE, Barbaro NM, et al. Spatiotemporal imaging of cortical activation during verb generation and picture naming. Neuroimage. 2010;50(1):291–301.
Mainy N, Jung J, Baciu M, Kahane P, Schoendorff B, Minotti L, et al. Cortical dynamics of word recognition. Hum Brain Mapp. 2008;29(11):1215–30.
Pei X, Leuthardt EC, Gaona CM, Brunner P, Wolpaw JR, Schalk G. Spatiotemporal dynamics of electrocorticographic high gamma activity during overt and covert word repetition. Neuroimage. 2011;54(4):2960–72.
Pallud J. A concept car or an all-road car to drive all along glioma resection? World Neurosurg. 2015;84(1):187.
Bakken HE, Kawasaki H, Oya H, Greenlee JDW, Howard MA. A device for cooling localized regions of human cerebral cortex. Technical note. J Neurosurg. 2003;99(3):604–8.
Lüders HO, Dinner DS, Morris HH, Wyllie E, Comair YG. Cortical electrical stimulation in humans. The negative motor areas. Adv Neurol. 1995;67:115–29.
Mikuni N, Ohara S, Ikeda A, Hayashi N, Nishida N, Taki J, et al. Evidence for a wide distribution of negative motor areas in the perirolandic cortex. Clin Neurophysiol. 2006;117(1):33–40.
Filevich E, Kühn S, Haggard P. Negative motor phenomena in cortical stimulation: implications for inhibitory control of human action. Cortex. 2012;48(10):1251–61.
Martino J, Gabarrós A, Deus J, Juncadella M, Acebes JJ, Torres A, et al. Intrasurgical mapping of complex motor function in the superior frontal gyrus. Neuroscience. 2011;14(179):131–42.
Brindley GS, Lewin WS. The visual sensations produced by electrical stimulation of the medial occipital cortex. J Physiol (Lond). 1968;194(2):54–5.
Mandonnet E, Sarubbo S, Duffau H. Proposal of an optimized strategy for intraoperative testing of speech and language during awake mapping. Neurosurgical Review. In Press.
Indefrey P, Levelt WJM. The spatial and temporal signatures of word production components. Cognition. 2004;92(1–2):101–44.
Duffau H, Moritz-Gasser S, Mandonnet E. A re-examination of neural basis of language processing: Proposal of a dynamic hodotopical model from data provided by brain stimulation mapping during picture naming. Brain Lang. 2013.
Duffau H. Stimulation mapping of white matter tracts to study brain functional connectivity. Nat Rev Neurol. 2015;11(5):255–65.
Tate MC, Herbet G, Moritz-Gasser S, Tate JE, Duffau H. Probabilistic map of critical functional regions of the human cerebral cortex: Broca’s area revisited. Brain. 2014.
Ojemann GA. Individual variability in cortical localization of language. J Neurosurg. 1979;50(2):164–9.
Vigneau M, Beaucousin V, Hervé PY, Duffau H, Crivello F, Houdé O, et al. Meta-analyzing left hemisphere language areas: phonology, semantics, and sentence processing. Neuroimage. 2006;30(4):1414–32.
Gatignol P, Capelle L, Le Bihan R, Duffau H. Double dissociation between picture naming and comprehension: an electrostimulation study. NeuroReport. 2004;15(1):191–5.
Bello L, Gallucci M, Fava M, Carrabba G, Giussani C, Acerbi F, et al. Intraoperative subcortical language tract mapping guides surgical removal of gliomas involving speech areas. Neurosurgery. 2007;60(1):67–80; discussion 80–82.
Teichmann M, Rosso C, Martini J-B, Bloch I, Brugières P, Duffau H, et al. A cortical-subcortical syntax pathway linking Broca’s area and the striatum. Hum Brain Mapp. 2015;36(6):2270–83.
De Witte E, Mariën P. The neurolinguistic approach to awake surgery reviewed. Clin Neurol Neurosurg. 2013;115(2):127–45.
Rofes A, Miceli G. Language mapping with verbs and sentences in awake surgery: a review. Neuropsychol Rev. 2014;24(2):185–99.
Talacchi A, Santini B, Casartelli M, Monti A, Capasso R, Miceli G. Awake surgery between art and science. Part II: language and cognitive mapping. Funct Neurol. 2013;28(3):223–39.
De Witte E, Satoer D, Robert E, Colle H, Verheyen S, Visch-Brink E, et al. The Dutch Linguistic Intraoperative Protocol: a valid linguistic approach to awake brain surgery. Brain Lang. 2015;140:35–48.
Rofes A, de Aguiar V, Miceli G. A minimal standardization setting for language mapping tests: an Italian example. Neurol Sci. 2015;36(7):1113–9.
Papagno C, Casarotti A, Comi A, Pisoni A, Lucchelli F, Bizzi A, et al. Long-term proper name anomia after removal of the uncinate fasciculus. Brain Struct Funct. 2014.
Papagno C, Miracapillo C, Casarotti A, Romero Lauro LJ, Castellano A, Falini A, et al. What is the role of the uncinate fasciculus? Surgical removal and proper name retrieval. Brain. 2011;134(Pt 2):405–14.
Roux F-E, Trémoulet M. Organization of language areas in bilingual patients: a cortical stimulation study. J Neurosurg. 2002;97(4):857–64.
Giussani C, Roux F-E, Lubrano V, Gaini SM, Bello L. Review of language organisation in bilingual patients: what can we learn from direct brain mapping? Acta Neurochir (Wien). 2007;149(11):1109–1116; discussion 1116.
Lucas TH, McKhann GM, Ojemann GA. Functional separation of languages in the bilingual brain: a comparison of electrical stimulation language mapping in 25 bilingual patients and 117 monolingual control patients. J Neurosurg. 2004;101(3):449–57.
Walker JA, Quiñones-Hinojosa A, Berger MS. Intraoperative speech mapping in 17 bilingual patients undergoing resection of a mass lesion. Neurosurgery. 2004;54(1):113–117; discussion 118.
Bello L, Acerbi F, Giussani C, Baratta P, Taccone P, Songa V, et al. Intraoperative language localization in multilingual patients with gliomas. Neurosurgery. 2006;59(1):115–125; discussion 115–125.
Gaillard R, Naccache L, Pinel P, Clémenceau S, Volle E, Hasboun D, et al. Direct intracranial, FMRI, and lesion evidence for the causal role of left inferotemporal cortex in reading. Neuron. 2006;50(2):191–204.
Gil-Robles S, Carvallo A, Jimenez MDM, Gomez Caicoya A, Martinez R, Ruiz-Ocaña C, et al. Double dissociation between visual recognition and picture naming: a study of the visual language connectivity using tractography and brain stimulation. Neurosurgery. 2013;72(4):678–86.
Mandonnet E, Gatignol P, Duffau H. Evidence for an occipito-temporal tract underlying visual recognition in picture naming. Clin Neurol Neurosurg. 2009;111(7):601–5.
Roux F-E, Durand J-B, Jucla M, Réhault E, Reddy M, Démonet J-F. Segregation of lexical and sub-lexical reading processes in the left perisylvian cortex. PLoS ONE. 2012;7(11):e50665.
Roux F-E, Lubrano V, Lauwers-Cances V, Trémoulet M, Mascott CR, Démonet J-F. Intra-operative mapping of cortical areas involved in reading in mono- and bilingual patients. Brain. 2004;127(Pt 8):1796–810.
Milea D, Lobel E, Lehéricy S, Duffau H, Rivaud-Péchoux S, Berthoz A, et al. Intraoperative frontal eye field stimulation elicits ocular deviation and saccade suppression. NeuroReport. 2002;13(10):1359–64.
Hoffman P, Lambon Ralph MA, Woollams AM. Triangulation of the neurocomputational architecture underpinning reading aloud. Proc Natl Acad Sci USA. 2015;112(28):E3719–28.
Thiebaut de Schotten M, Urbanski M, Duffau H, Volle E, Lévy R, Dubois B, et al. Direct evidence for a parietal-frontal pathway subserving spatial awareness in humans. Science. 2005;309(5744):2226–8.
Roux F-E, Dufor O, Lauwers-Cances V, Boukhatem L, Brauge D, Draper L, et al. Electrostimulation mapping of spatial neglect. Neurosurgery. 2011;69(6):1218–31.
Vallar G, Bello L, Bricolo E, Castellano A, Casarotti A, Falini A, et al. Cerebral correlates of visuospatial neglect: a direct cerebral stimulation study. Hum Brain Mapp. 2014;35(4):1334–50.
Dehaene S, Piazza M, Pinel P, Cohen L. Three parietal circuits for number processing. Cogn Neuropsychol. 2003;20(3):487–506.
Whalen J, McCloskey M, Lesser RP, Gordon B. Localizing arithmetic processes in the brain: evidence from a transient deficit during cortical stimulation. J Cogn Neurosci. 1997;9(3):409–17.
Duffau H, Denvil D, Lopes M, Gasparini F, Cohen L, Capelle L, et al. Intraoperative mapping of the cortical areas involved in multiplication and subtraction: an electrostimulation study in a patient with a left parietal glioma. J Neurol Neurosurg Psychiatr. 2002;73(6):733–8.
Pu S, Li Y, Wu C, Wang Y, Zhou X, Jiang T. Cortical areas involved in numerical processing: an intraoperative electrostimulation study. Stereotact Funct Neurosurg. 2011;89(1):42–7.
Yu X, Chen C, Pu S, Wu C, Li Y, Jiang T, et al. Dissociation of subtraction and multiplication in the right parietal cortex: evidence from intraoperative cortical electrostimulation. Neuropsychologia. 2011;49(10):2889–95.
Roux F-E, Boetto S, Sacko O, Chollet F, Trémoulet M. Writing, calculating, and finger recognition in the region of the angular gyrus: a cortical stimulation study of Gerstmann syndrome. J Neurosurg. 2003;99(4):716–27.
Roux F-E, Boukhatem L, Draper L, Sacko O, Démonet J-F. Cortical calculation localization using electrostimulation. J Neurosurg. 2009;110(6):1291–9.
Herbet G, Lafargue G, Bonnetblanc F, Moritz-Gasser S. Menjot de Champfleur N, Duffau H. Inferring a dual-stream model of mentalizing from associative white matter fibres disconnection. Brain. 2014;137(Pt 3):944–59.
Giussani C, Pirillo D, Roux F-E. Mirror of the soul: a cortical stimulation study on recognition of facial emotions. J Neurosurg. 2010;112(3):520–7.
Herbet G, Lafargue G, Moritz-Gasser S, Bonnetblanc F, Duffau H. Interfering with the neural activity of mirror-related frontal areas impairs mentalistic inferences. Brain Struct Funct. 2015;220(4):2159–69.
Herbet G, Lafargue G, Bonnetblanc F, Moritz-Gasser S, Duffau H. Is the right frontal cortex really crucial in the mentalizing network? A longitudinal study in patients with a slow-growing lesion. Cortex. 2013;49(10):2711–27.
Desmurget M, Reilly KT, Richard N, Szathmari A, Mottolese C, Sirigu A. Movement intention after parietal cortex stimulation in humans. Science. 2009;324(5928):811–3.
Knight J. An out of body experience. Nature. 2002;419(6903):106–7.
Herbet G, Lafargue G, de Champfleur NM, Moritz-Gasser S, le Bars E, Bonnetblanc F, et al. Disrupting posterior cingulate connectivity disconnects consciousness from the external environment. Neuropsychologia. 2014;56:239–44.
Shiban E, Krieg SM, Obermueller T, Wostrack M, Meyer B, Ringel F. Continuous subcortical motor evoked potential stimulation using the tip of an ultrasonic aspirator for the resection of motor eloquent lesions. J Neurosurg. 2015;123(2):301–6.
Raabe A, Beck J, Schucht P, Seidel K. Continuous dynamic mapping of the corticospinal tract during surgery of motor eloquent brain tumors: evaluation of a new method. J Neurosurg. 2014;120(5):1015–24.
Schucht P, Moritz-Gasser S, Herbet G, Raabe A, Duffau H. Subcortical electrostimulation to identify network subserving motor control. Hum Brain Mapp. 2013;34(11):3023–30.
Rech F, Herbet G, Moritz-Gasser S, Duffau H. Disruption of bimanual movement by unilateral subcortical electrostimulation. Hum Brain Mapp. 2013.
Rech F, Herbet G, Moritz-Gasser S, Duffau H. Somatotopic organization of the white matter tracts underpinning motor control in humans: an electrical stimulation study. Brain Struct Funct. 2015.
Duffau H, Capelle L, Sichez N, Denvil D, Lopes M, Sichez J-P, et al. Intraoperative mapping of the subcortical language pathways using direct stimulations. An anatomo-functional study. Brain. 2002;125(Pt 1):199–214.
Hickok G, Poeppel D. Dorsal and ventral streams: a framework for understanding aspects of the functional anatomy of language. Cognition. 2004;92(1–2):67–99.
Catani M, Howard RJ, Pajevic S, Jones DK. Virtual in vivo interactive dissection of white matter fasciculi in the human brain. Neuroimage. 2002;17(1):77–94.
Martino J, Brogna C, Robles SG, Vergani F, Duffau H. Anatomic dissection of the inferior fronto-occipital fasciculus revisited in the lights of brain stimulation data. Cortex. 2010;46(5):691–9.
Sarubbo S, De Benedictis A, Maldonado IL, Basso G, Duffau H. Frontal terminations for the inferior fronto-occipital fascicle: anatomical dissection, DTI study and functional considerations on a multi-component bundle. Brain Struct Funct. 2013;218(1):21–37.
Catani M, Dell’acqua F, Vergani F, Malik F, Hodge H, Roy P, et al. Short frontal lobe connections of the human brain. Cortex. 2012;48(2):273–91.
Ford A, McGregor KM, Case K, Crosson B, White KD. Structural connectivity of Broca’s area and medial frontal cortex. Neuroimage. 2010;52(4):1230–7.
Kinoshita M, Shinohara H, Hori O, Ozaki N, Ueda F, Nakada M, et al. Association fibers connecting the Broca center and the lateral superior frontal gyrus: a microsurgical and tractographic anatomy. J Neurosurg. 2012;116(2):323–30.
Makris N, Preti MG, Asami T, Pelavin P, Campbell B, Papadimitriou GM, et al. Human middle longitudinal fascicle: variations in patterns of anatomical connections. Brain Struct Funct. 2013;218(4):951–68.
Makris N, Preti MG, Wassermann D, Rathi Y, Papadimitriou GM, Yergatian C, et al. Human middle longitudinal fascicle: segregation and behavioral-clinical implications of two distinct fiber connections linking temporal pole and superior temporal gyrus with the angular gyrus or superior parietal lobule using multi-tensor tractography. Brain Imaging Behav. 2013;7(3):335–52.
Makris N, Papadimitriou GM, Kaiser JR, Sorg S, Kennedy DN, Pandya DN. Delineation of the middle longitudinal fascicle in humans: a quantitative, in vivo, DT-MRI study. Cereb Cortex. 2009;19(4):777–85.
Maldonado IL, de Champfleur NM, Velut S, Destrieux C, Zemmoura I, Duffau H. Evidence of a middle longitudinal fasciculus in the human brain from fiber dissection. J Anat. 2013;223(1):38–45.
Makris N, Kennedy DN, McInerney S, Sorensen AG, Wang R, Caviness VS, et al. Segmentation of subcomponents within the superior longitudinal fascicle in humans: a quantitative, in vivo, DT-MRI study. Cereb Cortex. 2005;15(6):854–69.
Maldonado IL, Mandonnet E, Duffau H. Dorsal fronto-parietal connections of the human brain: a fiber dissection study of their composition and anatomical relationships. Anat Rec (Hoboken). 2012;295(2):187–95.
Yagmurlu K, Middlebrooks EH, Tanriover N, Rhoton AL. Fiber tracts of the dorsal language stream in the human brain. J Neurosurg. 2015;20:1–10.
Forkel SJ, Thiebaut de Schotten M, Kawadler JM, Dell’Acqua F, Danek A, Catani M. The anatomy of fronto-occipital connections from early blunt dissections to contemporary tractography. Cortex. 2014;56:73–84.
Ius T, Angelini E. Thiebaut de Schotten M, Mandonnet E, Duffau H. Evidence for potentials and limitations of brain plasticity using an atlas of functional resectability of WHO grade II gliomas: towards a “minimal common brain”. Neuroimage. 2011;56(3):992–1000.
Sarubbo S, De Benedictis A, Merler S, Mandonnet E, Balbi S, Granieri E, et al. Towards a functional atlas of human white matter. Hum Brain Mapp. 2015;36(8):3117–36.
Maldonado IL, Moritz-Gasser S, Duffau H. Does the left superior longitudinal fascicle subserve language semantics? A brain electrostimulation study. Brain Struct Funct. 2011;216(3):263–74.
Maldonado IL, Moritz-Gasser S, de Champfleur NM, Bertram L, Moulinié G, Duffau H. Surgery for gliomas involving the left inferior parietal lobule: new insights into the functional anatomy provided by stimulation mapping in awake patients. J Neurosurg. 2011;115(4):770–9.
Duffau H, Gatignol P, Mandonnet E, Peruzzi P, Tzourio-Mazoyer N, Capelle L. New insights into the anatomo-functional connectivity of the semantic system: a study using cortico-subcortical electrostimulations. Brain. 2005;128(Pt 4):797–810.
Khan OH, Herbet G, Moritz-Gasser S, Duffau H. The role of left inferior fronto-occipital fascicle in verbal perseveration: a brain electrostimulation mapping study. Brain Topogr. 2013.
Hickok G, Poeppel D. The cortical organization of speech processing. Nat Rev Neurosci. 2007;8(5):393–402.
Duffau H, Gatignol P, Denvil D, Lopes M, Capelle L. The articulatory loop: study of the subcortical connectivity by electrostimulation. NeuroReport. 2003;14(15):2005–8.
Kemerdere R, de Champfleur NM, Deverdun J, Cochereau J, Moritz-Gasser S, Herbet G, et al. Role of the left frontal aslant tract in stuttering: a brain stimulation and tractographic study. J Neurol. 2015.
Kinoshita M, de Champfleur NM, Deverdun J, Moritz-Gasser S, Herbet G, Duffau H. Role of fronto-striatal tract and frontal aslant tract in movement and speech: an axonal mapping study. Brain Struct Funct. 2014.
Zemmoura I, Herbet G, Moritz-Gasser S, Duffau H. New insights into the neural network mediating reading processes provided by cortico-subcortical electrical mapping. Hum Brain Mapp. 2015;36(6):2215–30.
Goga C, Türe U. The anatomy of Meyer’s loop revisited: changing the anatomical paradigm of the temporal loop based on evidence from fiber microdissection. J Neurosurg. 2015;122(6):1253–62.
Sarubbo S, De Benedictis A, Milani P, Paradiso B, Barbareschi M, Rozzanigo U, et al. The course and the anatomo-functional relationships of the optic radiation: a combined study with “post mortem” dissections and “in vivo” direct electrical mapping. J Anat. 2015;226(1):47–59.
Sincoff EH, Tan Y, Abdulrauf SI. White matter fiber dissection of the optic radiations of the temporal lobe and implications for surgical approaches to the temporal horn. J Neurosurg. 2004;101(5):739–46.
Rubino PA, Rhoton AL, Tong X, Oliveira E de. Three-dimensional relationships of the optic radiation. Neurosurgery. 2005;57(4 Suppl.):219–27; discussion 219–227.
Ebeling U, Reulen HJ. Neurosurgical topography of the optic radiation in the temporal lobe. Acta Neurochir (Wien). 1988;92(1–4):29–36.
Gras-Combe G, Moritz-Gasser S, Herbet G, Duffau H. Intraoperative subcortical electrical mapping of optic radiations in awake surgery for glioma involving visual pathways. J Neurosurg. 2012;117(3):466–73.
Corbetta M, Kincade MJ, Lewis C, Snyder AZ, Sapir A. Neural basis and recovery of spatial attention deficits in spatial neglect. Nat Neurosci. 2005;8(11):1603–10.
Della Puppa A, De Pellegrin S, Lazzarini A, Gioffrè G, Rustemi O, Cagnin A, et al. Subcortical mapping of calculation processing in the right parietal lobe. J Neurosurg. 2015;122(5):1038–41.
Wilson SM, Lam D, Babiak MC, Perry DW, Shih T, Hess CP, et al. Transient aphasias after left hemisphere resective surgery. J Neurosurg. 2015;123(3):581–93.
Bartolo M, Zucchella C, Pace A, Lanzetta G, Vecchione C, Bartolo M, et al. Early rehabilitation after surgery improves functional outcome in inpatients with brain tumours. J Neurooncol. 2012;107(3):537–44.
Zucchella C, Capone A, Codella V, De Nunzio AM, Vecchione C, Sandrini G, et al. Cognitive rehabilitation for early post-surgery inpatients affected by primary brain tumor: a randomized, controlled trial. J Neurooncol. 2013;114(1):93–100.
Martino J, Taillandier L, Moritz-Gasser S, Gatignol P, Duffau H. Re-operation is a safe and effective therapeutic strategy in recurrent WHO grade II gliomas within eloquent areas. Acta Neurochir (Wien). 2009;151(5):427–36; discussion 436.
Southwell DG, Hervey-Jumper SL, Perry DW, Berger MS. Intraoperative mapping during repeat awake craniotomy reveals the functional plasticity of adult cortex. J Neurosurg. 2015;6:1–10.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Mandonnet, E., Duffau, H. (2017). Mapping the Brain for Primary Brain Tumor Surgery. In: Moliterno Gunel, J., Piepmeier, J., Baehring, J. (eds) Malignant Brain Tumors . Springer, Cham. https://doi.org/10.1007/978-3-319-49864-5_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-49864-5_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-49863-8
Online ISBN: 978-3-319-49864-5
eBook Packages: MedicineMedicine (R0)