Abstract
Survival of the preterm infant has improved dramatically over the past several decades. Access to neonatal intensive care units (NICU), improved nutrition, exogenous surfactant, advances in respiratory support, and minimally invasive surgical techniques have all contributed to an improvement in survival (Stoll et al. JAMA 314(10):1039–1051, 2015 [1]). Nevertheless, morbidity and mortality remains high, particularly in the extremely low birth weight infant. Worldwide, preterm birth was the leading cause of childhood mortality in 2013, accounting for 965,000 deaths out of the 6.3million deaths among children who died prior to 5 years of age (Liu et al. Lancet 385(9966):430–440, 2015) [2]. In 2014, 9.6 % of live births in the United States were considered preterm (i.e., born at <37 weeks gestation) (Martin et al. NCHS Data Brief 216:1–8, 2015; Hamilton et al. Natl Vital Stat Syst 64(12):1–64, 2015) [3, 4]. Many of these preterm infants developed pulmonary sequelae, ranging from mild respiratory symptoms that resolve over time to chronic respiratory failure requiring tracheostomy placement and mechanical ventilation. Although the lung parenchymal and small airways disease of bronchopulmonary dysplasia is one of the more common complications of prematurity, pulmonary sequelae in preterm infants can encompass virtually every part of the respiratory system. Since all aspects of the respiratory system can be affected, preterm infants may present with a variety of respiratory phenotypes, which in turn can lead to significant variation in care and outcomes (Guaman et al. Am J Perinatol32(10):960–967, 2015; Lapcharoensap et al. JAMA Pediatr 169(2):e143676, 2015) [5, 6]. In this chapter, we describe common pulmonary complications of prematurity and their epidemiology, including prevalence and risk factors, where known.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Lung Parenchymal Disease
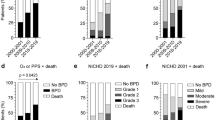
The archetypal lung disease in preterm infants and one of the more common complications of preterm birth is bronchopulmonary dysplasia (BPD), which may include both parenchymal and small airway components. Common clinical manifestations of BPD include hypoxia, hypercarbia, tachypnea, and asthma-like symptoms [7]. “Classic” or “old” BPD was first described in 1967 by Northway et al. [8], and was characterized by inflammation with airway injury and alveolar fibrosis. Over time, BPD has evolved (“new” BPD), particularly with use of newer ventilation strategies and exogenous surfactant, to a phenotype characterized by fewer and larger simplified alveoli with dysnaptic growth of the pulmonary vascular bed [9, 10]. Although several definitions of BPD or chronic lung disease of prematurity have been used since 1967 [11], currently the most widely agreed upon definition of BPD was developed at a NICHD workshop published in 2001 [12]. At this workshop, the diagnosis and severity of BPD in premature infants were based on gestational age (<32 weeks or ≥32 weeks) and need for oxygen and/or respiratory support at specified time points. This definition was subsequently validated in 2005 [11]. It should be recognized that this oxygen-based definition could overestimate or underestimate the incidence of BPD as goal oxygen saturations remain controversial [13, 14] and can vary among clinical centers [15]. Additionally, this oxygen-based definition may overestimate the incidence of BPD in clinical sites at higher altitudes where the partial pressure of oxygen is decreased [16, 17]. Furthermore, many preterm infants are placed on supplemental oxygen for apnea of prematurity who may or may not have moderate or severe BPD. In addition, these criteria do not define the BPD severity with reference to emerging respiratory support technologies, such as high-flow nasal cannulas.
In comparison to many of the other pulmonary manifestations of prematurity, more data exist on the epidemiology of BPD. Approximately 25 % of infants born with a birth weight of less than 1500 g are diagnosed with BPD [18], although the frequency of BPD reported may vary by center [5, 18]. Assuming that 1.4 % births in the United States annually are born with a birth weight less than 1500 g [4], this extrapolates to 14,000 infants who develop BPD in the United States annually. Although rates for preterm births have declined slightly in recent years [4], rates of BPD may not be mirroring this decline [19].
Infants with BPD frequently experience hypoxemia and many require supplemental oxygen at home, usually delivered via nasal cannula. A validation study of the NICHD BPD criteria in 4866 infants born at less than 32 weeks gestation and weighing less than 1000 g at birth observed that 2.8 % of infants with no or mild BPD were discharged on oxygen, while 64.4 % of those with moderate or severe BPD were discharged to home on supplemental oxygen [11]. These data would suggest that 31.6 % of these extremely low birth weight preterm infants are discharged to home on oxygen. Also, in this validation study, infants with no BPD or mild/moderate BPD were discontinued from home-supplemental oxygen between 7–8 months of age on average, whereas infants with severe BPD were discontinued from oxygen at 9.7 months of age [11].
Infants and children with the most severe BPD resulting in chronic respiratory failure may require tracheostomy placement for long-term ventilation with varying published estimates on the incidence. A 2010 study from Riley Children’s Hospital estimated the incidence to be 4.77 per 100,000 live births with a median duration of home ventilator support of 24 months of age [20], which extrapolates to 400 patients with BPD on home ventilation in the United States at any given time. However, a 2011 study from the Ventilator Assisted Children’s Home Program in Pennsylvania estimated that 8000 children in the United States are receiving home-invasive ventilation, and in this program, 36 % of ventilator-dependent children have chronic lung disease, with 77 % of these diagnosed with BPD [21], which extrapolates approximately 2200 patients with BPD on home ventilation in the United States at any given time. One single site study of 102 infants with BPD requiring home ventilation found an 81.4 % survival rate with 83.1 % of survivors being weaned from ventilation at a median age of 24 months, and 87.0 % of those weaned from ventilation being decannulated at a median age of 37.5 months [20].
Contributing to the difficulty of formulating algorithms for the management of BPD is its multifactorial development. Identified risk factors certainly include gestational age at birth, with infants born at earlier gestational ages experiencing a higher incidence of BPD [19]. Fetal growth also plays a role as infants with lower birth weights corrected for gestational age (fetal growth restriction) have higher rates of BPD [22, 23], and a large Swedish cohort found a 2.7 times higher risk of developing BPD with being born small for gestational age (2 standard deviations below the mean) [24]. Some prenatal risk factors for the development of BPD could include preeclampsia [25, 26], chorioamnionitis [26, 27], and the premature rupture of membranes [24, 28]. However, there remains controversy regarding the role of preeclampsia, as other studies have found preeclampsia to be protective [29] or not a risk factor at all [23, 30]. Likewise, studies of chorioamnionitis as a risk factor have also produced conflicting results [31, 32]. Postnatal risk factors may include patent ductus arteriosus [24, 33, 34] (and again, this is not definitively clear) [35], late-onset infections [24, 34, 36], pneumothorax [24], and mechanical ventilation [24, 34, 37]. In terms of postnatal therapies that may alter the incidence of BPD, retrospective data suggest that prophylactic use of caffeine within the first few days of life may reduce the likelihood of developing BPD [38–40]. However, while antenatal corticosteroids administered to women at risk of preterm birth can accelerate fetal lung maturation [41], neither the use of postnatal systemic or inhaled corticosteroids has been shown to reduce the incidence of BPD [42–45].
In addition, genetic factors may predispose individual infants to develop BPD based on heritability studies of twins [46, 47], but genomewide association studies have not confirmed positive findings from candidate gene studies [48]. Three genomewide association studies have been performed among infants with BPD, and one performed in European and African populations identified SPOCK2, which may have a potential role in alveolar development, to be associated with BPD [49], but two North American studies did not identify any SNPs meeting genomewide significance, including SPOCK2 [50, 51]. Pathway analyses in one of the GWAS studies found an association with BPD and known pathways of lung development and repair (CD44, phosphorus oxygen lyase activity) as well as novel pathways (adenosine deaminase, targets of miR-219) [51]. In addition to genetic factors, an emerging risk factor for the development of BPD may be epigenetic changes (nonnucleotide changes to the genome) that alter gene expression [52]. One study of umbilical cord blood demonstrated that preterm infants that developed BPD had distinct patterns of chromatin remodeling and histone acetylation pathways compared to those who did not (n = 54) [53].
Small Airways Disease
Another frequently seen pulmonary sequelae of preterm infants is small airways disease, which is often secondary to BPD, and can manifest as wheezing and coughing among patients of all ages, and obstructive findings on pulmonary function testing in older children. Computed tomography of the chest in preterm children with a prior history of BPD may reveal emphysema, hyperexpansion, and fibrous/interstitial changes [54].
Objective data regarding the incidence of small airways disease in infants are difficult to capture, as infant pulmonary function testing is only available in a small number of centers and carries the risks attendant to sedating an infant with known respiratory disease. In older children, spirometry can routinely be carried out as a marker of obstructive lung function secondary to small airways disease. A meta-analysis of lung function in former preterm and full-term infants published in 2013 demonstrated that the percent predicted forced expiratory volume in 1 s (FEV1), a commonly used marker of obstructive lung disease, in former preterm infants has been steadily improving in later birth cohorts, although still is approximately 90 % that of former full-term infants [55].
Although the small airways disease associated with prematurity is not necessarily equivalent to asthma, the prototypical obstructive lung disorder in children, the diagnosis of asthma can serve as a marker of small airways disease among preterm infants. Although the risk of asthma is highest in preterm infants born at ≤32 weeks gestation (adjusted OR: 3.9) compared to full-term infants born at 39–40 weeks gestation, even late preterm infants born at 33–36 weeks gestation and early term infants born at 37–38 weeks gestation may be at higher risk as well (adjusted ORs: 1.7 and 1.2, respectively) [56]. It should be noted that the association between asthma and late preterm births has not been consistently observed [57], and that the strength of this association may decrease with age [58]. Nevertheless, given that the prevalence of asthma is 9.5 % among children aged 0–17 years within the general population in the United States [59] and there is an increased odds ratio of asthma of ~3.9 among early preterm infants [56, 58], the prevalence of asthma among these infants may approach 40 %.
Large Airways Disease
Large airways disease in preterm infants is most likely to manifest as tracheomalacia and/or bronchomalacia. Typically, infants with large airway malacia present with respiratory distress (often with abrupt cyanosis), or persistent atelectasis or hyperinflation on imaging [60, 61]. A common clinical scenario of infants with severe large airway malacia may be the onset of sudden respiratory distress and profound desaturations in the setting of agitation or vagal stimulation, and may require use of a bag valve mask for resuscitation.
In terms of the epidemiology of large airway disease in preterm infants, the prevalence is less well characterized and may be underdiagnosed, perhaps due to the difficulties in diagnosis. Classically, direct visualization with bronchoscopy is utilized for diagnosis, which may not be readily available at all NICUs, and may include the attendant risks of sedation and intubation. Newer modalities of imaging, such as CT scanning, may be helpful in the diagnosis of evaluating tracheomalacia [62], but may be only available at select tertiary care institutions and may not be reliable in all patients [61].
One prospective study of 117 preterm infants with a history of intubation for >7 days found that 16.2 % had tracheomalacia, which was associated with severe BPD, hypercarbia, and apnea [63]. Of these patients with moderate to severe tracheomalacia, 21 % required tracheostomies for management.
Vascular Disease
Over the past decade, there has been emerging recognition of pulmonary vascular disease in preterm infants in the form of pulmonary hypertension, which is defined as a resting mean pulmonary artery pressure >25 mm Hg after 2–3 months of age [64]. Pulmonary hypertension in preterm infants is almost always associated with severe lung disease and secondary to a reactive or diminished pulmonary vascular bed, a fact reflected in recent international classifications of pulmonary hypertension [65–67]. However, pulmonary hypertension can also be secondary to cardiovascular lesions; for example, pulmonary vein stenosis has also been reported in preterm infants with pulmonary hypertension and can be associated with poorer outcomes [68, 69]. Although the gold standard for diagnosis of pulmonary hypertension is cardiac catheterization [64], many cases are screened or diagnosed via echocardiograms. Among young children with chronic lung disease, including BPD, echocardiography has been shown to diagnose pulmonary hypertension, but may not be reliable in determining severity [70].
Pulmonary hypertension in preterm infants can manifest as persistent BPD symptoms poorly responsive to therapies or evidence of right heart insufficiency or failure [71]. Also, patients with baseline pulmonary hypertension can experience acute hypertensive crises, which present with an acute increase in work of breathing, hypoxemia, and/or, in the most severe instances, cardiorespiratory failure or arrest [71]. These sometimes life-threatening episodes can be triggered by anesthesia induction [72] or respiratory tract infections [73], possibly through changes in gas exchange leading to hypoxemia or hypercarbia [74–76].
Prospective and retrospective studies have demonstrated a range in prevalence of pulmonary hypertension ranging from 14 to 43 % of preterm infants with BPD [77–82]. Unfortunately, the mortality with patients with pulmonary hypertension remains high, ranging from 14 to 38 % [77–82]. In addition, these patients also experience additional morbidity in the form of longer initial hospital stays, with infants with pulmonary hypertension having a 2.2 month longer initial NICU admission compared to infants with BPD without pulmonary hypertension, which may result in at least an additional $198,000 in health care expenditures [83].
Pulmonary hypertension likely arises from a series of complex interactions between multiple factors, including genetic and prenatal/postnatal environmental factors [84]. These factors may include alterations in angiogenic pathways [85–88], possible genetic modifiers [46, 89], intrauterine growth [78, 81, 90–92], oligohydramnios [79, 92], patent ductus arteriosus [77], and placental vascularity [93, 94]. Although most cases of pulmonary hypertension arise as a function of reactive or diminished pulmonary vascular bed, pulmonary hypertension can occur as a function of pulmonary vein stenosis [68, 69], for which umbilical catheters may be a risk factor [95].
Control of Breathing
Infants born prematurely frequently demonstrate issues with immature control of breathing manifesting as central apneas. These apneas, if prolonged, can be accompanied by bradycardia and oxygen desaturations. The oxygen desaturations may be more prominent in infants with poor pulmonary reserve (i.e., parenchymal disease) and can occur with brief central apneas that last only a few seconds [96]. In addition, preterm infants with BPD may have blunted chemoreflex responses to both hypoxia [97] and/or hyperoxia [98]. Limited data suggest that infants with a history of posthemorrhagic hydrocephalus are more likely to have apneic events [99, 100], and certainly untreated or inadequately treated elevated intracranial pressures from hydrocephalus may cause bradypnea or apnea.
Although central apneas may be isolated events, they also may occur in rapid successive sequence with periodic breathing. Periodic breathing is commonly observed in infants, including preterm infants, but may frequently lead to oxygen desaturations in preterm infants [101]. The incidence of disordered control of breathing is likely underestimated, as the use of supplemental oxygen or augmented airflow may also mask control of breathing abnormalities [102]. Nevertheless, the frequency of such events and time to resolution are associated with gestational age at birth with infants born earlier at a higher risk of demonstrating apnea and a later age of resolution [103, 104].
Data from apnea monitor downloads suggest that preterm infants may be at a risk of more extreme events until 43 weeks postconceptional age [105]. Overnight polysomnography data obtained on infants and toddlers born prematurely also demonstrate a decrease in central apneas after 12 months of age [96]. Although there are no published data regarding sleep-disordered breathing in adults born prematurely, alterations in ventilatory control, including both hypoventilation and hyperventilation, in exercising children and adults born prematurely have been observed compared to term controls [106] as well as in adults born prematurely exposed to hypoxic and hyperoxic conditions [107].
Upper Airway Obstruction
Upper airway obstruction leading to obstructive apneas or hypopneas can occur in several anatomic locations in preterm infants. During infancy, obstruction in the palatal or retroglossal regions may be more common, in addition to laryngeal lesions such as vocal cord paresis and subglottic stenosis, whereas in toddlers, anatomic obstruction is more likely to be secondary to adenotonsillar hypertrophy [108]. In addition, a relatively larger tongue size for preterm infants has been postulated to contribute to narrowing of the upper airway based on respiration-timed radiographs [109]. Although the gold standard for diagnosis of upper airway obstruction is overnight polysomnography with specialist evaluation [110], specific diagnoses are often made by direct visualization by an otolaryngologist.
The risk factors and prevalence for upper airway obstruction in preterm infants are dependent on the site of the obstruction. Acquired subglottic stenosis is often associated with intubation, and pertinent risk factors may include duration of intubation, endotracheal tube size, and the occurrence of traumatic intubation [111]. Acquired subglottic cysts may also be more likely to occur with traumatic or prolonged intubation [112, 113]. In at least one prospective study, subglottic cysts were identified in 7.2 % of preterm infants after prolonged intubation (n = 153) [113]. Although it can be postulated that gastroesophageal reflux (which is not uncommon in preterm infants) could be associated with obstructive apnea through upper airway inflammation or laryngeal chemoreflexes [114], the association between reflux and apnea is not well established in infants, regardless of prematurity [115, 116]. Vocal cord paresis may occur as a complication of cardiac surgeries, including PDA ligation. In one meta-analysis, the weighted pooled proportion of unilateral vocal fold paralysis in studies that assessed the vocal folds postoperatively was 29.8 % for any congenital cardiac surgery (11 studies; n = 584) and 39 % for patent ductus arteriosus ligation (6 studies; n = 274) [117].
Although there are limited data regarding the prevalence of sleep-disordered breathing among former preterm infants, a US-based population cohort study of 850 children aged 8–11 years, of whom 46 % were born preterm, found that sleep-disordered breathing was 3–5 times more common among those born prematurely compared to those born at term [118]. Other retrospective chart reviews have also found an association between prematurity and obstructive sleep apnea [119].
Conclusions
Preterm infants can manifest a variety of respiratory complications, which providers who care for preterm infants should be aware of. The diverse elements of the respiratory tract that may be involved and the varying severity of presentations make it difficult to apply common algorithms for prevention and management. Further research is necessary to more clearly delineate the scope of disease as well as best practices for diagnosis and treatment.
References
Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993-2012. JAMA. 2015;314(10):1039–51. PubMed PMID: 26348753.
Liu L, Oza S, Hogan D, Perin J, Rudan I, Lawn JE, et al. Global, regional, and national causes of child mortality in 2000-13, with projections to inform post-2015 priorities: an updated systematic analysis. Lancet. 2015;385(9966):430–40. PubMed PMID: 25280870.
Martin JA, Hamilton BE, Osterman MJ. Births in the United States, 2014. NCHS Data Brief. 2015;216:1–8. PubMed PMID: 26460599.
Hamilton BE, Martin JA, Osterman MJ, Curtin SC, Matthews TJ. Births: final data for 2014. National vital statistics reports: from the Centers for Disease Control and Prevention, National Center for Health Statistics. Natl Vital Stat Syst. 2015;64(12):1–64. PubMed PMID: 26727629.
Guaman MC, Gien J, Baker CD, Zhang H, Austin ED, Collaco JM. Point prevalence, clinical characteristics, and treatment variation for infants with severe bronchopulmonary dysplasia. Am J Perinatol. 2015;32(10):960–7. PubMed PMID: 25738785. Pubmed Central PMCID: 4617756.
Lapcharoensap W, Gage SC, Kan P, Profit J, Shaw GM, Gould JB, et al. Hospital variation and risk factors for bronchopulmonary dysplasia in a population-based cohort. JAMA Pediatr. 2015;169(2):e143676. PubMed PMID: 25642906.
Baraldi E, Filippone M. Chronic lung disease after premature birth. N Engl J Med. 2007;357(19):1946–55. PubMed PMID: 17989387.
Northway Jr WH, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. N Engl J Med. 1967;276(7):357–68. PubMed PMID: 5334613.
Jobe AJ. The new BPD: an arrest of lung development. Pediatr Res. 1999;46(6):641–3. PubMed PMID: 10590017.
Thebaud B, Abman SH. Bronchopulmonary dysplasia: where have all the vessels gone? Roles of angiogenic growth factors in chronic lung disease. Am J Respir Crit Care Med. 2007;175(10):978–85. PubMed PMID: 17272782. Pubmed Central PMCID: 2176086.
Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA, et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116(6):1353–60. PubMed PMID: 16322158.
Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163(7):1723–9. PubMed PMID: 11401896.
Group BIUKC, Group BIAC, Group BINZC, Stenson BJ, Tarnow-Mordi WO, Darlow BA, et al. Oxygen saturation and outcomes in preterm infants. N Engl J Med. 2013;368(22):2094–104. PubMed PMID: 23642047.
Network SSGotEKSNNR, Carlo WA, Finer NN, Walsh MC, Rich W, Gantz MG, et al. Target ranges of oxygen saturation in extremely preterm infants. N Engl J Med. 2010;362(21):1959–69. PubMed PMID: 20472937. Pubmed Central PMCID: 2891970.
Ellsbury DL, Acarregui MJ, McGuinness GA, Klein JM. Variability in the use of supplemental oxygen for bronchopulmonary dysplasia. J Pediatr. 2002;140(2):247–9. PubMed PMID: 11865280.
Britton JR. Altitude, oxygen and the definition of bronchopulmonary dysplasia. J Perinatol Off J California Perinat Assoc. 2012;32(11):880–5. PubMed PMID: 22343395.
Fernandez CL, Fajardo CA, Favareto MV, Hoyos A, Jijon-Letort FX, Carrera MS, et al. Oxygen dependency as equivalent to bronchopulmonary dysplasia at different altitudes in newborns 1500 g at birth from the SIBEN network. J Perinatol Off J California Perinat Assoc. 2014;34(7):538–42. PubMed PMID: 24699220.
Jensen EA, Schmidt B. Epidemiology of bronchopulmonary dysplasia. Birth Defects Res Part A Clin Mol Teratol. 2014;100(3):145–57. PubMed PMID: 24639412.
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010;126(3):443–56. PubMed PMID: 20732945. Pubmed Central PMCID: 2982806.
Cristea AI, Carroll AE, Davis SD, Swigonski NL, Ackerman VL. Outcomes of children with severe bronchopulmonary dysplasia who were ventilator dependent at home. Pediatrics. 2013;132(3):e727–34. PubMed PMID: 23918888. Pubmed Central PMCID: 3876749.
Boroughs D, Dougherty JA. Decreasing accidental mortality of ventilator-dependent children at home: a call to action. Home Healthc Nurse. 2012;30(2):103–11. quiz 12-3. PubMed PMID: 22306756.
Bose C, Van Marter LJ, Laughon M, O’Shea TM, Allred EN, Karna P, et al. Fetal growth restriction and chronic lung disease among infants born before the 28th week of gestation. Pediatrics. 2009;124(3):e450–8. PubMed PMID: 19706590. Pubmed Central PMCID: 2891899.
Torchin H, Ancel PY, Goffinet F, Hascoet JM, Truffert P, Tran D, et al. Placental complications and bronchopulmonary dysplasia: EPIPAGE-2 cohort study. Pediatrics. 2016;137(3):e20152163. PubMed PMID: 26908662.
Eriksson L, Haglund B, Odlind V, Altman M, Ewald U, Kieler H. Perinatal conditions related to growth restriction and inflammation are associated with an increased risk of bronchopulmonary dysplasia. Acta Paediatr. 2015;104(3):259–63. PubMed PMID: 25469645.
Hansen AR, Barnes CM, Folkman J, McElrath TF. Maternal preeclampsia predicts the development of bronchopulmonary dysplasia. J Pediatr. 2010;156(4):532–6. PubMed PMID: 20004912.
Eriksson L, Haglund B, Odlind V, Altman M, Kieler H. Prenatal inflammatory risk factors for development of bronchopulmonary dysplasia. Pediatr Pulmonol. 2014;49(7):665–72. PubMed PMID: 24039136.
Hartling L, Liang Y, Lacaze-Masmonteil T. Chorioamnionitis as a risk factor for bronchopulmonary dysplasia: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2012;97(1):F8–F17. PubMed PMID: 21697236.
Hanke K, Hartz A, Manz M, Bendiks M, Heitmann F, Orlikowsky T, et al. Preterm prelabor rupture of membranes and outcome of very-low-birth-weight infants in the German Neonatal Network. PLoS One. 2015;10(4):e0122564. PubMed PMID: 25856083. Pubmed Central PMCID: 4391753.
Yen TA, Yang HI, Hsieh WS, Chou HC, Chen CY, Tsou KI, et al. Preeclampsia and the risk of bronchopulmonary dysplasia in VLBW infants: a population based study. PLoS One. 2013;8(9):e75168. PubMed PMID: 24073247. Pubmed Central PMCID: 3779258.
O’Shea JE, Davis PG, Doyle LW, Victorian Infant Collaborative Study G. Maternal preeclampsia and risk of bronchopulmonary dysplasia in preterm infants. Pediatr Res. 2012;71(2):210–4. PubMed PMID: 22258134.
Plakkal N, Soraisham AS, Trevenen C, Freiheit EA, Sauve R. Histological chorioamnionitis and bronchopulmonary dysplasia: a retrospective cohort study. J Perinatol Off J California Perinat Assoc. 2013;33(6):441–5. PubMed PMID: 23238570.
Lacaze-Masmonteil T. That chorioamnionitis is a risk factor for bronchopulmonary dysplasia – the case against. Paediatr Respir Rev. 2014;15(1):53–5. PubMed PMID: 24120077.
Schena F, Francescato G, Cappelleri A, Picciolli I, Mayer A, Mosca F, et al. Association between hemodynamically significant patent ductus arteriosus and bronchopulmonary dysplasia. J Pediatr. 2015;166(6):1488–92. PubMed PMID: 25882876.
Tapia JL, Agost D, Alegria A, Standen J, Escobar M, Grandi C, et al. Bronchopulmonary dysplasia: incidence, risk factors and resource utilization in a population of South American very low birth weight infants. J Pediatr. 2006;82(1):15–20. PubMed PMID: 16532142.
Clyman RI. The role of patent ductus arteriosus and its treatments in the development of bronchopulmonary dysplasia. Semin Perinatol. 2013;37(2):102–7. PubMed PMID: 23582964. Pubmed Central PMCID: 3627220.
Shah J, Jefferies AL, Yoon EW, Lee SK, Shah PS, Canadian Neonatal N. Risk factors and outcomes of late-onset bacterial sepsis in preterm neonates born at <32 weeks’ gestation. Am J Perinatol. 2015;32(7):675–82. PubMed PMID: 25486288.
Keszler M, Sant’Anna G. Mechanical ventilation and bronchopulmonary dysplasia. Clin Perinatol. 2015;42(4):781–96. PubMed PMID: 26593078.
Patel RM, Leong T, Carlton DP, Vyas-Read S. Early caffeine therapy and clinical outcomes in extremely preterm infants. J Perinatol Off J California Perinat Assoc. 2013;33(2):134–40. PubMed PMID: 22538326.
Dobson NR, Patel RM, Smith PB, Kuehn DR, Clark J, Vyas-Read S, et al. Trends in caffeine use and association between clinical outcomes and timing of therapy in very low birth weight infants. J Pediatr. 2014;164(5):992–8. e3.PubMed PMID: 24461786. Pubmed Central PMCID: 3992195.
Lodha A, Seshia M, McMillan DD, Barrington K, Yang J, Lee SK, et al. Association of early caffeine administration and neonatal outcomes in very preterm neonates. JAMA Pediatr. 2015;169(1):33–8. PubMed PMID: 25402629.
Roberts D, Dalziel S. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. 2006;3:CD004454. PubMed PMID: 16856047.
Onland W, Offringa M, van Kaam A. Late (>/= 7 days) inhalation corticosteroids to reduce bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2012;4:CD002311. PubMed PMID: 22513906.
Shah VS, Ohlsson A, Halliday HL, Dunn M. Early administration of inhaled corticosteroids for preventing chronic lung disease in ventilated very low birth weight preterm neonates. Cochrane Database Syst Rev. 2012;5:CD001969. PubMed PMID: 22592680.
Shah SS, Ohlsson A, Halliday HL, Shah VS. Inhaled versus systemic corticosteroids for preventing chronic lung disease in ventilated very low birth weight preterm neonates. Cochrane Database Syst Rev. 2012;5:CD002058. PubMed PMID: 22592683.
Shah SS, Ohlsson A, Halliday HL, Shah VS. Inhaled versus systemic corticosteroids for the treatment of chronic lung disease in ventilated very low birth weight preterm infants. Cochrane Database Syst Rev. 2012;5:CD002057. PubMed PMID: 22592682.
Bhandari V, Bizzarro MJ, Shetty A, Zhong X, Page GP, Zhang H, et al. Familial and genetic susceptibility to major neonatal morbidities in preterm twins. Pediatrics. 2006;117(6):1901–6. PubMed PMID: 16740829.
Lavoie PM, Pham C, Jang KL. Heritability of bronchopulmonary dysplasia, defined according to the consensus statement of the national institutes of health. Pediatrics. 2008;122(3):479–85. PubMed PMID: 18762515. Pubmed Central PMCID: 4631604.
Lal CV, Ambalavanan N. Genetic predisposition to bronchopulmonary dysplasia. Semin Perinatol. 2015;39(8):584–91. PubMed PMID: 26471063. Pubmed Central PMCID: 4644695.
Hadchouel A, Durrmeyer X, Bouzigon E, Incitti R, Huusko J, Jarreau PH, et al. Identification of SPOCK2 as a susceptibility gene for bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2011;184(10):1164–70. PubMed PMID: 21836138.
Wang H, St Julien KR, Stevenson DK, Hoffmann TJ, Witte JS, Lazzeroni LC, et al. A genome-wide association study (GWAS) for bronchopulmonary dysplasia. Pediatrics. 2013;132(2):290–7. PubMed PMID: 23897914. Pubmed Central PMCID: 3727675.
Ambalavanan N, Cotten CM, Page GP, Carlo WA, Murray JC, Bhattacharya S, et al. Integrated genomic analyses in bronchopulmonary dysplasia. J Pediatr. 2015;166(3):531–7. e13. PubMed PMID: 25449221. Pubmed Central PMCID: 4344889.
Piersigilli F, Bhandari V. Biomarkers in neonatology: the new “omics” of bronchopulmonary dysplasia. J Matern Fetel Neonatal Med Off J Eur Assoc Perinat Med Feder Asia Oceania Perinat Soc Int Soc Perinat Obstet. 2015;2015:1–7. PubMed PMID: 26135768.
Cohen J, Van Marter LJ, Sun Y, Allred E, Leviton A, Kohane IS. Perturbation of gene expression of the chromatin remodeling pathway in premature newborns at risk for bronchopulmonary dysplasia. Genome Biol. 2007;8(10):R210. PubMed PMID: 17916252. Pubmed Central PMCID: 2246284.
Ochiai M, Hikino S, Yabuuchi H, Nakayama H, Sato K, Ohga S, et al. A new scoring system for computed tomography of the chest for assessing the clinical status of bronchopulmonary dysplasia. J Pediatr. 2008;152(1):90–5. 5 e1-3. PubMed PMID: 18154907.
Kotecha SJ, Edwards MO, Watkins WJ, Henderson AJ, Paranjothy S, Dunstan FD, et al. Effect of preterm birth on later FEV1: a systematic review and meta-analysis. Thorax. 2013;68(8):760–6. PubMed PMID: 23604458.
Harju M, Keski-Nisula L, Georgiadis L, Raisanen S, Gissler M, Heinonen S. The burden of childhood asthma and late preterm and early term births. J Pediatr. 2014;164(2):295–9. e1. PubMed PMID: 24210922.
Crump C, Winkleby MA, Sundquist J, Sundquist K. Risk of asthma in young adults who were born preterm: a Swedish national cohort study. Pediatrics. 2011;127(4):e913–20. PubMed PMID: 21422091. Pubmed Central PMCID: 3387891.
Damgaard AL, Hansen BM, Mathiasen R, Buchvald F, Lange T, Greisen G. Prematurity and prescription asthma medication from childhood to young adulthood: a Danish national cohort study. PLoS One. 2015;10(2):e0117253. PubMed PMID: 25651521. Pubmed Central PMCID: 4317188.
Moorman JE, Akinbami LJ, Bailey CM, Zahran HS, King ME, Johnson CA, et al. National surveillance of asthma: United States, 2001-2010. Series 3, Analytical and epidemiological studies / [US Dept of Health and Human Services, Public Health Service, National Center for Health Statistics]. Vital Health Stat. 2012;2012(35):1–58. PubMed PMID: 24252609.
Miller RW, Woo P, Kellman RK, Slagle TS. Tracheobronchial abnormalities in infants with bronchopulmonary dysplasia. J Pediatr. 1987;111(5):779–82. PubMed PMID: 3668747.
Mok Q, Negus S, McLaren CA, Rajka T, Elliott MJ, Roebuck DJ, et al. Computed tomography versus bronchography in the diagnosis and management of tracheobronchomalacia in ventilator dependent infants. Arch Dis Child Fetal Neonatal Ed. 2005;90(4):F290–3. PubMed PMID: 15857878. Pubmed Central PMCID: 1721907.
Long FR, Castile RG. Technique and clinical applications of full-inflation and end-exhalation controlled-ventilation chest CT in infants and young children. Pediatr Radiol. 2001;31(6):413–22. PubMed PMID: 11436888.
Downing GJ, Kilbride HW. Evaluation of airway complications in high-risk preterm infants: application of flexible fiberoptic airway endoscopy. Pediatrics. 1995;95(4):567–72. PubMed PMID: 7700760.
Abman SH, Hansmann G, Archer SL, Ivy DD, Adatia I, Chung WK, et al. Pediatric pulmonary hypertension: guidelines from the American Heart Association and American Thoracic Society. Circulation. 2015;132(21):2037–99. PubMed PMID: 26534956.
Simonneau G, Gatzoulis MA, Adatia I, Celermajer D, Denton C, Ghofrani A, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2013;62(25 Suppl):D34–41. PubMed PMID: 24355639.
Lammers AE, Adatia I, Cerro MJ, Diaz G, Freudenthal AH, Freudenthal F, et al. Functional classification of pulmonary hypertension in children: report from the PVRI pediatric taskforce, Panama 2011. Pulm Circ. 2011;1(2):280–5. PubMed PMID: 21874157. Pubmed Central PMCID: 3161406.
Cerro MJ, Abman S, Diaz G, Freudenthal AH, Freudenthal F, Harikrishnan S, et al. A consensus approach to the classification of pediatric pulmonary hypertensive vascular disease: report from the PVRI Pediatric Taskforce, Panama 2011. Pulm Circ. 2011;1(2):286–98. PubMed PMID: 21874158. Pubmed Central PMCID: 3161725.
Laux D, Rocchisani MA, Boudjemline Y, Gouton M, Bonnet D, Ovaert C. Pulmonary hypertension in the preterm infant with chronic lung disease can be caused by pulmonary vein stenosis: a must-know entity. Pediatr Cardiol. 2016;37(2):313–21. PubMed PMID: 26573816.
Smith SC, Rabah R. Pulmonary venous stenosis in a premature infant with bronchopulmonary dysplasia: clinical and autopsy findings of these newly associated entities. Pediatr Dev Pathol Off J Soc Pediatr Pathol Paediatr Pathol Soc. 2012;15(2):160–4. PubMed PMID: 22313395.
Mourani PM, Sontag MK, Younoszai A, Ivy DD, Abman SH. Clinical utility of echocardiography for the diagnosis and management of pulmonary vascular disease in young children with chronic lung disease. Pediatrics. 2008;121(2):317–25. PubMed PMID: 18245423. Pubmed Central PMCID: 3121163.
Collaco JM, Romer LH, Stuart BD, Coulson JD, Everett AD, Lawson EE, et al. Frontiers in pulmonary hypertension in infants and children with bronchopulmonary dysplasia. Pediatr Pulmonol. 2012;47(11):1042–53. PubMed PMID: 22777709. Pubmed Central PMCID: 3963167.
Carmosino MJ, Friesen RH, Doran A, Ivy DD. Perioperative complications in children with pulmonary hypertension undergoing noncardiac surgery or cardiac catheterization. Anesth Analg. 2007;104(3):521–7. PubMed PMID: 17312201. Pubmed Central PMCID: 1934984.
Farquhar M, Fitzgerald DA. Pulmonary hypertension in chronic neonatal lung disease. Paediatr Respir Rev. 2010;11(3):149–53. PubMed PMID: 20692628.
Shukla AC, Almodovar MC. Anesthesia considerations for children with pulmonary hypertension. Pediatr Crit Care Med J Soc Crit Care Med World Feder Pediatr Inten Crit Care Soc. 2010;11(2 Suppl):S70–3. PubMed PMID: 20216167.
van der Griend BF, Lister NA, McKenzie IM, Martin N, Ragg PG, Sheppard SJ, et al. Postoperative mortality in children after 101,885 anesthetics at a tertiary pediatric hospital. Anesth Analg. 2011;112(6):1440–7. PubMed PMID: 21543787.
Friesen RH, Williams GD. Anesthetic management of children with pulmonary arterial hypertension. Paediatr Anaesth. 2008;18(3):208–16. PubMed PMID: 18230063.
An HS, Bae EJ, Kim GB, Kwon BS, Beak JS, Kim EK, et al. Pulmonary hypertension in preterm infants with bronchopulmonary dysplasia. Korean Circ J. 2010;40(3):131–6. PubMed PMID: 20339498. Pubmed Central PMCID: 2844979.
Khemani E, McElhinney DB, Rhein L, Andrade O, Lacro RV, Thomas KC, et al. Pulmonary artery hypertension in formerly premature infants with bronchopulmonary dysplasia: clinical features and outcomes in the surfactant era. Pediatrics. 2007;120(6):1260–9. PubMed PMID: 18055675.
Kumar VH, Hutchison AA, Lakshminrusimha S, Morin 3rd FC, Wynn RJ, Ryan RM. Characteristics of pulmonary hypertension in preterm neonates. J Perinatol Off J California Perinat Assoc. 2007;27(4):214–9. PubMed PMID: 17330053.
Slaughter JL, Pakrashi T, Jones DE, South AP, Shah TA. Echocardiographic detection of pulmonary hypertension in extremely low birth weight infants with bronchopulmonary dysplasia requiring prolonged positive pressure ventilation. J Perinatol Off J California Perinat Assoc. 2011;31(10):635–40. PubMed PMID: 21311503.
Bhat R, Salas AA, Foster C, Carlo WA, Ambalavanan N. Prospective analysis of pulmonary hypertension in extremely low birth weight infants. Pediatrics. 2012;129(3):e682–9. PubMed PMID: 22311993. Pubmed Central PMCID: 3289526.
Mourani PM, Sontag MK, Younoszai A, Miller JI, Kinsella JP, Baker CD, et al. Early pulmonary vascular disease in preterm infants at risk for bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2014;191(1):87–95. PubMed PMID: 25389562.
Stuart BD, Sekar P, Coulson JD, Choi SE, McGrath-Morrow SA, Collaco JM. Health-care utilization and respiratory morbidities in preterm infants with pulmonary hypertension. J Perinatol Off J California Perinat Assoc. 2013;33(7):543–7. PubMed PMID: 23328926.
Mourani PM, Abman SH. Pulmonary hypertension and vascular abnormalities in bronchopulmonary dysplasia. Clin Perinatol. 2015;42(4):839–55. PubMed PMID: 26593082.
Lassus P, Turanlahti M, Heikkila P, Andersson LC, Nupponen I, Sarnesto A, et al. Pulmonary vascular endothelial growth factor and Flt-1 in fetuses, in acute and chronic lung disease, and in persistent pulmonary hypertension of the newborn. Am J Respir Crit Care Med. 2001;164(10 Pt 1):1981–7. PubMed PMID: 11734455.
Maniscalco WM, Watkins RH, Pryhuber GS, Bhatt A, Shea C, Huyck H. Angiogenic factors and alveolar vasculature: development and alterations by injury in very premature baboons. Am J Physiol Lung Cell Mol Physiol. 2002;282(4):L811–23. PubMed PMID: 11880308.
Tang JR, Karumanchi SA, Seedorf G, Markham N, Abman SH. Excess soluble vascular endothelial growth factor receptor-1 in amniotic fluid impairs lung growth in rats: linking preeclampsia with bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2012;302(1):L36–46. PubMed PMID: 22003089. Pubmed Central PMCID: 3349373.
Li F, Hagaman JR, Kim HS, Maeda N, Jennette JC, Faber JE, et al. eNOS deficiency acts through endothelin to aggravate sFlt-1-induced pre-eclampsia-like phenotype. J Am Soc Nephrol. 2012;23(4):652–60. PubMed PMID: 22282588. Pubmed Central PMCID: 3312503.
Trittmann JK, Nelin LD, Zmuda EJ, Gastier-Foster JM, Chen B, Backes CH, et al. Arginase I gene single-nucleotide polymorphism is associated with decreased risk of pulmonary hypertension in bronchopulmonary dysplasia. Acta Paediatr. 2014;103(10):e439–43. PubMed PMID: 24919409. Pubmed Central PMCID: 4180790.
Bruno CJ, Meerkov M, Capone C, Vega M, Sutton N, Kim M, et al. CRIB scores as a tool for assessing risk for the development of pulmonary hypertension in extremely preterm infants with bronchopulmonary dysplasia. Am J Perinatol. 2015;32(11):1031–7. PubMed PMID: 26368789.
Check J, Gotteiner N, Liu X, Su E, Porta N, Steinhorn R, et al. Fetal growth restriction and pulmonary hypertension in premature infants with bronchopulmonary dysplasia. J Perinatol Off J California Perinat Assoc. 2013;33(7):553–7. PubMed PMID: 23328924. Pubmed Central PMCID: 3633609.
Kim DH, Kim HS, Choi CW, Kim EK, Kim BI, Choi JH. Risk factors for pulmonary artery hypertension in preterm infants with moderate or severe bronchopulmonary dysplasia. Neonatology. 2012;101(1):40–6. PubMed PMID: 21791938.
Yallapragada S, Mestan KK, Palac HL, Porta NF, Gotteiner NL, Hamvas A, et al. Placental villous vascularity is decreased in premature infants with bronchopulmonary dysplasia-associated pulmonary hypertension. Pediatr Dev Pathol Off J Soc Pediatr Pathol Paediatr Pathol Soc. 2015;19(2):101–7. PubMed PMID: 26366786.
Mestan KK, Check J, Minturn L, Yallapragada S, Farrow KN, Liu X, et al. Placental pathologic changes of maternal vascular underperfusion in bronchopulmonary dysplasia and pulmonary hypertension. Placenta. 2014;35(8):570–4. PubMed PMID: 24906549. Pubmed Central PMCID: 4119480.
Jaillard SM, Godart FR, Rakza T, Chanez A, Lequien P, Wurtz AJ, et al. Acquired pulmonary vein stenosis as a cause of life-threatening pulmonary hypertension. Ann Thorac Surg. 2003;75(1):275–7. PubMed PMID: 12537232.
McGrath-Morrow SA, Ryan T, McGinley BM, Okelo SO, Sterni LM, Collaco JM. Polysomnography in preterm infants and children with chronic lung disease. Pediatr Pulmonol. 2012;47(2):172–9. PubMed PMID: 21815283.
Calder NA, Williams BA, Smyth J, Boon AW, Kumar P, Hanson MA. Absence of ventilatory responses to alternating breaths of mild hypoxia and air in infants who have had bronchopulmonary dysplasia: implications for the risk of sudden infant death. Pediatr Res. 1994;35(6):677–81. PubMed PMID: 7936817.
Katz-Salamon M, Jonsson B, Lagercrantz H. Blunted peripheral chemoreceptor response to hyperoxia in a group of infants with bronchopulmonary dysplasia. Pediatr Pulmonol. 1995;20(2):101–6. PubMed PMID: 8570299.
Mancini MC, Barbosa NE, Banwart D, Silveira S, Guerpelli JL, Leone CR. Intraventricular hemorrhage in very low birth weight infants: associated risk factors and outcome in the neonatal period. Rev Hosp Clin. 1999;54(5):151–4. PubMed PMID: 10788836.
Robles P, Poblano A, Hernandez G, Ibarra J, Guzman I, Sosa J. Cortical, brainstem and autonomic nervous system dysfunction in infants with post-hemorrhagic hydrocephalus. Rev Invest Clin. 2002;54(2):133–8. PubMed PMID: 12053811.
Razi NM, DeLauter M, Pandit PB. Periodic breathing and oxygen saturation in preterm infants at discharge. J Perinatol Off J California Perinat Assoc. 2002;22(6):442–4. PubMed PMID: 12168119.
Coste F, Ferkol T, Hamvas A, Cleveland C, Linneman L, Hoffman J, et al. Ventilatory control and supplemental oxygen in premature infants with apparent chronic lung disease. Arch Dis Child Fetal Neonatal Ed. 2015;100(3):F233–7. PubMed PMID: 25716677. Pubmed Central PMCID: 4732273.
Henderson-Smart DJ. The effect of gestational age on the incidence and duration of recurrent apnoea in newborn babies. Aust Paediatr J. 1981;17(4):273–6. PubMed PMID: 7347216.
Eichenwald EC, Aina A, Stark AR. Apnea frequently persists beyond term gestation in infants delivered at 24 to 28 weeks. Pediatrics. 1997;100(3 Pt 1):354–9. PubMed PMID: 9282705.
Ramanathan R, Corwin MJ, Hunt CE, Lister G, Tinsley LR, Baird T, et al. Cardiorespiratory events recorded on home monitors: comparison of healthy infants with those at increased risk for SIDS. JAMA. 2001;285(17):2199–207. PubMed PMID: 11325321.
Bates ML, Pillers DA, Palta M, Farrell ET, Eldridge MW. Ventilatory control in infants, children, and adults with bronchopulmonary dysplasia. Respir Physiol Neurobiol. 2013;189(2):329–37. PubMed PMID: 23886637. Pubmed Central PMCID: 3812402.
Bates ML, Farrell ET, Eldridge MW. Abnormal ventilatory responses in adults born prematurely. N Engl J Med. 2014;370(6):584–5. PubMed PMID: 24499235. Pubmed Central PMCID: 4769592.
Don GW, Kirjavainen T, Broome C, Seton C, Waters KA. Site and mechanics of spontaneous, sleep-associated obstructive apnea in infants. J Appl Physiol. 2000;89(6):2453–62. PubMed PMID: 11090602.
Tonkin SL, McIntosh C, Gunn AJ. Does tongue size contribute to risk of airway narrowing in preterm infants sitting in a car safety seat? Am J Perinatol. 2014;31(9):741–4. PubMed PMID: 24338121.
Wise MS, Nichols CD, Grigg-Damberger MM, Marcus CL, Witmans MB, Kirk VG, et al. Executive summary of respiratory indications for polysomnography in children: an evidence-based review. Sleep. 2011;34(3):389–98. AW. PubMed PMID: 21359088. Pubmed Central PMCID: 3041716.
Amin RS, Rutter MJ. Airway disease and management in bronchopulmonary dysplasia. Clin Perinatol. 2015;42(4):857–70. PubMed PMID: 26593083.
Watson GJ, Malik TH, Khan NA, Sheehan PZ, Rothera MP. Acquired paediatric subglottic cysts: a series from Manchester. Int J Pediatr Otorhinolaryngol. 2007;71(4):533–8. PubMed PMID: 17239962.
Downing GJ, Hayen LK, Kilbride HW. Acquired subglottic cysts in the low-birth-weight infant. Characteristics, treatment, and outcome. Am J Dis Child. 1993;147(9):971–4. PubMed PMID: 8362815.
Qubty WF, Mrelashvili A, Kotagal S, Lloyd RM. Comorbidities in infants with obstructive sleep apnea. J Clin Sleep Med Off Pub Am Acad Sleep Med. 2014;10(11):1213–6. PubMed PMID: 25325583. Pubmed Central PMCID: 4224722.
Arad-Cohen N, Cohen A, Tirosh E. The relationship between gastroesophageal reflux and apnea in infants. J Pediatr. 2000;137(3):321–6. PubMed PMID: 10969254.
Di Fiore J, Arko M, Herynk B, Martin R, Hibbs AM. Characterization of cardiorespiratory events following gastroesophageal reflux in preterm infants. J Perinatol Off J California Perinat Assoc. 2010;30(10):683–7. PubMed PMID: 20220760. Pubmed Central PMCID: 2891417.
Strychowsky JE, Rukholm G, Gupta MK, Reid D. Unilateral vocal fold paralysis after congenital cardiothoracic surgery: a meta-analysis. Pediatrics. 2014;133(6):e1708–23. PubMed PMID: 24843065.
Rosen CL, Larkin EK, Kirchner HL, Emancipator JL, Bivins SF, Surovec SA, et al. Prevalence and risk factors for sleep-disordered breathing in 8- to 11-year-old children: association with race and prematurity. J Pediatr. 2003;142(4):383–9. PubMed PMID: 12712055.
Cote V, Ruiz AG, Perkins J, Sillau S, Friedman NR. Characteristics of children under 2 years of age undergoing tonsillectomy for upper airway obstruction. Int J Pediatr Otorhinolaryngol. 2015;79(6):903–8. PubMed PMID: 25912628.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Collaco, J.M., McGrath-Morrow, S.A. (2017). The Problem of the Preterm Lung: Definitions, History, and Epidemiology. In: Hibbs, A., Muhlebach , M. (eds) Respiratory Outcomes in Preterm Infants. Respiratory Medicine. Humana Press, Cham. https://doi.org/10.1007/978-3-319-48835-6_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-48835-6_1
Published:
Publisher Name: Humana Press, Cham
Print ISBN: 978-3-319-48834-9
Online ISBN: 978-3-319-48835-6
eBook Packages: MedicineMedicine (R0)