Abstract
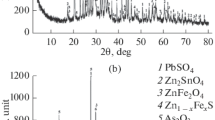
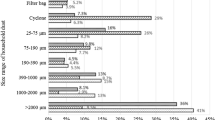
The lead smelting dusts contain complex composition such as Pb, Zn, Cd, S and As, which pose serious environment problem. This research used several physical and chemical methods to study the basic properties of different lead smelting dusts including blast furnace dust, reduction furnace dust, reverberatory furnace dust and bottom blowing furnace dust. The results of characterization showed that these dusts have different phases, element composition and surface morphology. In addition, the distribution of elements and compounds presented a certain trend, which will impact the leachability of their major elements. At the same time, leaching experiments were carried out on thefour types lead smelting dusts to provide leachability information for metal recovery.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
B.H. Jia, “The lead market review and outlook in July 2013,” Nonferrous Metals Engineering, 3 (4) (2013), 9–10.
W. Ye, “The lead market review and outlook in March 2013,” Nonferrous Metals Engineering, 3 (2) (2013), 9–10.
H.Y. Sohn and M.O. Martinez, “Chapter 2.3 — Lead and Zinc Production, Treatise on Process Metallurgy,” (Beijing: Metallurgical Industry Press, 2014), 29–31.
C.Y. Wang, W. Gao and F. Yin, “Present situation of lead smelting technology and the performance of trial production of the first lead flash smelting furnace in China,” Nonferrous Metal:Smelting, 1 (2010), 9–13.
W.F. Li, X.G. Zhang and X.Y. Guo, “Status and progress of lead smelting technology in China,” China Nonferrous Metallurgy, 2 (2010), 29–33.
Z.D. Wang, T. Lei and Z. Shi, “Experimentation on treatment of slag from lead smelting furnace by fuming process,” Yunnan Metallurgy, 6 (1) (2007), 45–47.
J.B. Wang, J. Wang and X.M. Wand, “Renovation of ESP in the process of rich oxygen under-gate blast reduction,” Industrial Safety and Environmental Protection, 29 (7) (2003), 8–9.
L.M. Liu, “A new indium extration process from lead dross reverberatory furnace dust,” Rare Metal, 27 (1) (2003), 112–115.
A.H. Jiang, S.H. Yand and C. Mei, “Exergy analysis of oxygen bottom blown furnace in SKS lead smelting system,” Journal of Central South University (Science and Technology), 41 (3) (2010), 1191–1193.
M. Guo, “Technical analysis and energy-saving and emission reduction effects of lead metallurgy by advanced oxygen bottom-blowing process,” Nonferrous Metallurgy and Energy Saving, 5 (2008), 15–19.
Mushak, “Chapter3-Lead in the Human Environment: Production, Uses, Trends,” Trace Metals and other Contaminants in the Environment, 10 (25) (2011), 31.
M.F. Gomes, T.F. Mendes, and K. Wada, “Reduction in toxicity and generation of slag in secondary lead process,” Journal of Cleaner Production, 19 (9) (2011), 1096–1103.
L. Jiang, G.F. Fu and D.Q. Wang, “Removal off and Cl from zinc oxide dust using selective chlorination roasting process,” Nonferrous Metals, 53 (3) (2001), 28–31.
M.T. Tang, P. Zhang and J. He, “Leaching zinc dust in system of Zn (II)-(NH4) 2SO4-H2O,” J. Cent. South Univ. (Science and Technology), 38 (5) (2007), 867–872.
M.D. Turan, H.S. Altundoðan and F. Tümen, “Recovery of zinc and lead from zinc plant residue,” Hydrometallurgy, 2004 (75), 169–176.
O. Alguacil, “Recycling of an electric arc furnace flue dust to obtain high grade ZnO,” Journal of Hazardous Material, 2007 (141), 33–36.
F.M. Martins, J.M. Neto and C.J. Cunha, “Mineral phases of weathered and recent electric arc furnace dust,” Journal of hazardous materials, 2008 (154), 417–425.
C.L. Zhang, “Mineralogical characteristics of unusual black talc ores in Guangfeng County,” Jiangxi Province, China, Applied Clay Science, 2013 (74), 37–46.
T. Sofiliæ et al., “Characterization of steel mill electric-arc furnace dust, Journal of hazardous materials,” 2004 (109), 59–70.
J.G. Machado et al., “Chemical, physical, structural and morphological characterization of the electric arc furnace dust,” Journal of hazardous materials, 2006 (136), 953–960.
S. Kelebek, S. Yörük and B. Davis, “Characterization of basic oxygen furnace dust and zinc removal by acid leaching,” Minerals Engineering, 2004 (17), 285–291.
K. Nakamoto, “Infrared and Raman Spectra of Inorganic and Coordination Compounds, Chapter II. third ed.” Wiley Interscience, New York, 1978, 132–148.
Patrick N L et al., “Influence of the chemical and mineralogical composition on the reactivity of volcanic ashes during alkali activation,” Ceramics International, 2014 (40), 811–820.
M. Paul and G.C. Allen, “Chemical characterization of transition metal spinel type oxides by infrared spectroscopy,” Appl. Spectrosc, 1995 (49), 451–458.
Medina A ea al., “Fly ash from a Mexican mineral coal I: Mineralogical and chemical characterization,” Journal of hazardous materials, 2010 (181), 82–90.
D. Zingg and D.M. Hercules, “Electron spectroscopy for chemical analysis studies of lead sulfide oxidation,” The Journal of Physical Chemistry, 1978 (82), 1992–1995.
A.P. Davis and C. Huang, “The removal of substituted phenols by a photocatalytic oxidation process with cadmium sulfide,” Water Research, 1990 (24), 543–550.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 TMS (The Minerals, Metals & Materials Society)
About this paper
Cite this paper
Li, H., Xie, Y., Li, C., Liu, Z., Huang, M. (2016). The Mineral Constitution and Leachability Characteristics of Dusts from Different Lead Smelting Furnace. In: Reddy, R.G., Chaubal, P., Pistorius, P.C., Pal, U. (eds) Advances in Molten Slags, Fluxes, and Salts: Proceedings of the 10th International Conference on Molten Slags, Fluxes and Salts 2016. Springer, Cham. https://doi.org/10.1007/978-3-319-48769-4_148
Download citation
DOI: https://doi.org/10.1007/978-3-319-48769-4_148
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48625-3
Online ISBN: 978-3-319-48769-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)