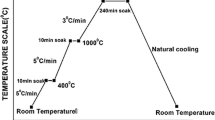
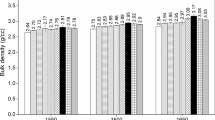
Abstract
Magnesium aluminate spinel is widely used in the metallurgical industry as refractory material, however, natural magnesium aluminate spinel is rare and most of the magnesium aluminate spinel is synthetic. The traditional synthesis of magnesium aluminate spinel often needs high temperature, which leads to more energy-consumption. In this article, solid phase sintering process was employed to synthesize magnesium aluminate spinel, in which, magnesium and aluminum oxide were used as raw materials and magnesium and aluminum powder were used as composite additives. The mass ratio of magnesium and aluminum on the effect of synthesis of magnesium aluminate spinel and the characteristic of the synthesis at different temperature and time was discussed. Experiment found that the synthesis temperature was observably lowered when the addition of magnesium and aluminum was 2.0% and 1.0% respectively.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
References
LR Ping, A.M. Azad, and T.W. Dung,“Magnesium aluminate (MgAl2O4) spinel produced via self-heat-sustained (SHS) technique”, Materials research bulletin, 36(7) (2001), 1417–1430.
E. Ryskhewitch, Oxide Ceramics (New York, NY: Academic Press, 1960), 271.
J. Katanic-Popovic, N. Miljevic, and S. Zec. “Spinal formation from coprecipitated gel”, Ceramics International, 1991, 17(1), 49–52.
J.G. Li, T. Ikegami, J.H. Lee, et al. “A wet-chemical process yielding reactive magnesium aluminate spinel (MgAl2O4) powder”, Ceramics international, 2001, 27(4),481–489.
CR. Bickmore, K.F. Waldner, DR Treadwell, et al. “Ultrafine spinel powders by flame spray pyrolysis of a magnesium aluminum double alkoxide”, Journal of the American Ceramic Society, 1996, 79(5),1419–1423.
J.G.M. Delau. “Preparation of Ceramic Powders from Sulfate Solutions by Spray Drying and Roasting”, Am Ceram Soc Bull, 1970, 49(6), 572–574.
CT. Wang, LS. Lin, S.J. Yang. “Preparation of MgAl2O4 Spinel Powders via Freeze-Drying of Alkoxide Precursors”, Journal of the American Ceramic Society, 1992, 75(8), 2240–2243.
G. Ye, G Oprea, T. Troczynski. “Synthesis of MgAl2O4 spinel powder by combination of sol-gel and precipitation processes”, Journal of the American Ceramic Society, 2005, 88(11), 3241–3244.
D. Domanski, G. Urretavizcaya, F. J. Castro, et al. “Mechanochemical synthesis of magnesium aluminate spinel powder at room temperature”, Journal of the American Ceramic Society, 2004, 87(11), 2020–2024.
W. Kim, F Saito. “Effect of grinding on synthesis of MgAl2O4 spinel from a powder mixture of Mg (OH) 2 and Al (OH) 3”, Powder technology, 2000, 113(1), 109–113.
E. Kostić, S. Bošković, Š. Kiš. “Influence of fluorine ion on the spinel synthesis”, Journal of Materials Science Letters, 1982, 1(12): 507–510.
T.F Baranova, I. Kurskaya, NA. Dabizha “Sintering of high purity fused MgO and MgAl2O4”, Ogneupory, 1981, 46(3): 54–56.
LA. Skomorovskaya “Magnesia spinel ceramics alloyed with rare-earth oxides, Glass and ceramics”, 1993, 50(4): 165–168.
R Sarkar, G Bannerjee. “Effect of addition of TiO2 on reaction sintered MgO-Al2O3 spinels”, Journal of the European Ceramic Society, 2000, 20(12): 2133–2141.
Y.H. Baik. “Sintering of MgAl2O4 spinel and its characteristics”, Yoop Hikoechi, 1985, 22(6): 29–36.
J.F. Pasquier, S. Komarneni, R. Roy. “Synthesis of MgAl2O4 spinel: seeding effects on formation temperature”, Journal of materials science, 1991, 26(14): 3797–3802.
D. Mohapatra, D.Sarkar. “Preparation of MgO-MgAl2O4 composite for refractory application”, Journal of materials processing technology, 2007, 189(1): 279–283.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Xiang, X., Xia, W., Yang, W. (2016). Effect of Compound Additives on Synthetic Magnesium Aluminate Spinel under Low Temperature. In: Hwang, JY., et al. 7th International Symposium on High-Temperature Metallurgical Processing. Springer, Cham. https://doi.org/10.1007/978-3-319-48093-0_55
Download citation
DOI: https://doi.org/10.1007/978-3-319-48093-0_55
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48617-8
Online ISBN: 978-3-319-48093-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)