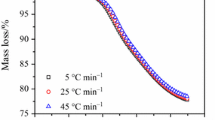
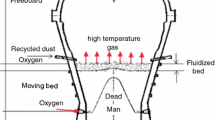
Abstract
To utilize low grade coals to produce high quality semi-coke which can be used in blast furnaces, the pyrolysis of low grade coals was studied in this paper. According to the characteristics of coal pyrolysis process, the temperature range was divided into three parts. The interfacial chemical reaction model, random pore model and the internal diffusion model with the shrinking volume of resultant were established to describe the different ranges of pyrolysis process respectively. The results showed that the experimental data was fitted well, and all the correlation coefficients (r2) exceeded 0.97. Finally, the kinetic parameters for each temperature range have been calculated, which laid a necessary foundation for the simulation and expanded production.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Q. H. Liu, K. Wu, and H. Y. Wang, “Kinetic study of tar’s separation from coals used in COREX,” Journal of China Coal society, 37(10) (2012), 1749–1752.
Y. B. Zhang, G. H. Li, and T. Jiang, “Reduction behavior of tin-bearing iron concentrate psellets using diverse coals as reducers,” International Journal of Mineral Processing, 110 (2012), 109–116.
J. Tomeczek and H. Plaugniok, “Kinetics of mineral matter transformation during coal combustion,” Fuel, 81 (2002), 1251–1258.
S. F. Zhang, F. Zhu, and C. G. Bai, “High temperature pyrolysis behaviour and kinetics of lump coal in COREX melter gasifier,” Ironmaking and steelmaking, 41(3) (2014), 219–228.
A. O. Aboyade, J. F. Görgens and M. Carrier, “Thermogravimetric study of the pyrolysis characteristics and kinetics of coal blends with corn and sugarcane residues,” Fuel processing Technology, 106 (2013), 310–320.
H. M. Jeong, M. W. Seo, and S. M. Jeong, “Pyrolysis kinetics of coking coal mixed with biomass under non-isothermal and isothermal conditions,” Bioresource Technology, 155 (2014), 442–445.
K. Wu, Q. H. Liu, and W. L. Zhan, “Research on Tar Precipitation Kinetics Using Phasewise Analysis,” Journal of Chemical Engineering of Chinese Universities, 28(4) (2014), 738–744.
H. J. Guo, Metallurgy physical chemistry (Beijing: Metallurgical industry press, 2006),114–126.
J. L. Zhang, G. W. Wang, and J. G. Shao, “A modified random pore model for the kinetics of char gasification,” BioResource, 9(2) (2014), 3497–3507.
J. S. Gupta, and S. K. Bhatia, “A modified discrete random pore model allowing for different initial surface reactivity,” Carbon, 38(1) (2000), 47–58.
C. R. Zhang, Y. G. Yang, and G. Zhang, “Kinetic model for solid state reactions controlled by diffusion,” ACTA physico-chimica sinica, 4(5) (1988), 539–544.
C. J. Ping, J. H. Zhou, and J. Cheng, “Research on the pyrolysis kinetics of blended coals,” Proceedings of the CSEE, 27(17) (2007), 6–10.
Z. X. Fu, Z. C. Guo, and Z. F. Yuan, “Swelling and shrinkage behavior of raw and processed coals during pyrolysis,” Fuel, 86 (2007), 418–425.
F. Ferrara, A. Orsini, and A. Plaisant, “Pyrolysis of coal, biomass and their blends: Performance assessment by thermogravimetric analysis,” Bioresource Technology, 171 (2014), 433–441.
M. Ishida, and C. Y. Wen, “Comparison of kinetic and diffusional models for solid-gas reactions,” AIChE J, 14 (2) (1968), 311–317.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Du, R., Wu, K., Yuan, X., Xu, D., Chao, C. (2016). Kinetics Study on the Pyrolysis of Low Grade Coals. In: Hwang, JY., et al. 7th International Symposium on High-Temperature Metallurgical Processing. Springer, Cham. https://doi.org/10.1007/978-3-319-48093-0_53
Download citation
DOI: https://doi.org/10.1007/978-3-319-48093-0_53
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48617-8
Online ISBN: 978-3-319-48093-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)