Abstract
Several environmental and endogenous factors create a variety of lesions in the genome of an organism. These lesions could potentially be genotoxic and might lead to mutations, which could be lethal. All organisms exhibit a prompt response against DNA damage, which is referred to as the DNA damage response. The existence of elaborate, evolutionarily conserved systems to repair the damage, mostly at the expense of huge amount of energy, points to the importance of safeguarding the integrity of DNA. Most of the current understanding about DNA damage response and repair pathways has been distilled through decades of research on prokaryotes, yeast, and mammalian systems. The response to genotoxic stresses and the repair mechanisms involved in plants has only recently begun to be investigated. Herein, we present a comprehensive account of the types of DNA damage, the DNA damage response, and the repair pathways with reference to the recent insights gained from the plants. Although, the underlying common theme runs through to the plants, the mechanisms in plants demonstrate some unique features. Also, there are several missing links that need to be unravelled for inferring a complete picture. A thorough understanding of the mechanisms involved would aid in devising strategies to help plants avoid irreparable damages to their genome upon exposure to genotoxic agents.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The genome contains the blueprint of life. Protecting the sanctity of the information in DNA is a cellular imperative for maintaining and perpetuating life. Though intrinsically more stable than other macromolecules, DNA is nonetheless prone to damages that can compromise its function and the eventual fate of an organism. A variety of processes, some endogenous and other catalyzed by environmental agents , can undermine the integrity of DNA. The endogenous sources of damage include metabolic by-products and stalled replication forks while environmental agents like ultraviolet (UV) and ionizing (IR) radiation and chemical mutagens are the exogenous ones. To combat the DNA damage, organisms contain elaborate cellular networks termed the DNA damage response (DDR) that are critical to maintain the integrity of the genome (Ciccia and Elledge 2010). The components of this response include processes to detect the damage, to relay the signal to specific regulators in the cell, and to elicit the production of effector molecules (Vespa et al. 2005; Culligan et al. 2006). The orchestration of these processes ultimately leads to the repair of DNA lesions or the induction of cellular death, if the damages are beyond repair. The DDR pathways are fundamental to life and most of them have been conserved through the evolution. Much of the current understanding of these mechanisms derives from the research done in prokaryotic, yeast, and mammalian systems. The delineation of DDR in plants has only been attempted lately.
Plants are continuously exposed to environmental, edaphic, and other anthropogenic stresses. These include extremes of temperature, drought, UV-B, IR, as well as air and soil pollutants. In addition to severely impacting the structural, enzymatic, and nonenzymatic components of plants, these stresses potentially threaten the plant genomes (Wasi et al. 2013). Unrepaired DNA damages can lead to mutations, which can impact the stability of a plant’s genomes, its growth and productivity, and might threaten the survival of the plant (Singh et al. 2010; Biedermann et al. 2011). Being sessile, plants cannot evade these stresses and combat is the only alternative for their survival and growth. It is, thus, imperative that an efficient and specific DDR system be in place in plants to cope with DNA damage (Yoshiyama et al. 2013). The sequencing of several plant genomes has given huge impetus to the study of DDR in plants, with many components of the system having been discovered in plant genomes (Mannuss et al. 2012). Although the basic mechanism remains conserved in eukaryotes, several plant-specific regulators have been reported, alluding to the existence of some unique DDR systems in plants.
In this chapter, we have endeavored to summarize the current understanding of DDR in plants. The following sections in the chapter provide details on the DNA damaging agents, types of DNA damage, mechanism of genotoxic stress perception, transduction of the signal, and repair of the damage.
DNA Damaging Agents
Different intrinsic and extrinsic factors, classified either as physical or chemical, are capable of damaging the genetic material.
Physical Agents
Ultraviolet and ionizing radiation are the most common physical factors contributing to DNA damage. The innocuous sunlight, essential for photosynthesis in plants, also contains radiations that are potentially genotoxic. These energy-rich radiations include UV-C (100–280 nm), UV-B (290–320 nm), and UV-A (320–400 nm). UV-C is filtered out by absorption in the upper stratospheric and ozone layers but UV-B and UV-A enter the atmosphere, their amount reaching the earth’s surface dependent on latitude and elevation, as well as the cloud cover and canopy density. UV-B, on account of its higher energy, is potentially more deleterious to living organisms. Besides damaging proteins and biomembranes, UV-B is strongly mutagenic (Pang and Hays 1991; Britt 1999). It is absorbed by the DNA and may lead to the generation of cyclobutane pyrimidine dimers (CPDs) and to a lesser extent pyrimidine (6-4) pyrimidone dimers (6-4 PPs; Friedberg et al. 2006). These lesions affect the transcription and also result in error-prone replication. Besides, UV light is strongly absorbed by many environmental organic contaminants, thereby, increasing their toxicity (Huang et al. 1993). The UV-A and visible spectrum of light cannot be absorbed by the native DNA. However, they can still damage DNA through reactive oxygen species (ROS) generation via photosensitizing reactions (Alscher et al. 1997; Iovine et al. 2009). ROS can induce a variety of DNA damages like base and nucleotide modifications, especially in guanine-rich sequences and may even cause strand breaks (Wiseman and Halliwell 1996; Tuteja et al. 2001; Tuteja and Tuteja 2001).
IR is another major mutagen that damages DNA , either directly or indirectly through the production of free radicals (Ward 1975). Chromosomal breaks, inversions, duplications, and translocations can result from double-strand (DSBs) and single-strand breaks (SSBs) caused by the absorption of IR by the sugar–phosphate backbone in the DNA. The indirect effect of IR is mediated through the radiolysis of water which generates OH radicals, the most damaging of all the ROS that attack the DNA along with proteins, lipids, and other cellular constituents.
Chemical Agents
Chemically, genotoxic agents could either be inorganic or organic. Heavy metals like Cd2+, Cu2+, Ni2+, and Pb2+ are the main inorganic genotoxic compounds which cause the generation of free radicals that damage the DNA (Imlay and Linn 1988). Metal ions can also influence the efficiency and fidelity of DNA replication. Ions like Ni2+ and As3+ change the stability of DNA by altering their methylation pattern (Davis et al. 2000).
Genotoxic organic compounds can affect the DNA in different ways. Alkylating agents like ethylmethane sulfonate (EMS) and methylmethane sulfonate (MMS) cause DSBs while aromatic compounds such as polycyclic aromatic hydrocarbons (PAHs) and polychlorinated biphenyls (PCBs) are mutagenic because they intercalate between the DNA strands.
Types of DNA Damage
DNA damage caused by physical or chemical agents can be broadly classified into three types of lesions: mismatched bases, double-strand breaks, and chemically modified bases. The damages induced by the different agents are discussed below.
UV-Induced DNA Damage
A major type of DNA damage induced by UV-B is the formation of dimers between adjacent pyrimidines—the CPDs and 6-4 PPs (Mitchell and Nairn 1989; McGregor 1999; Fig. 7.1). CPDs can occur in any of the diastereoisomeric forms, i.e., cis/trans (with respect to the relative position of pyrimidine rings) and syn/anti (with reference to relative orientation of C5–C6 bonds). Whereas cis-syn is the predominant CPD form in DNA, the trans-syn occurs exclusively in single-stranded DNA. The 6-4 PPs are formed between adjacent TT, TC, and CC nucleotides depending upon the UV wavelength and adjacent sequence (Mouret et al. 2006). CPDs and 6-4 PPs make up approximately 70–80 % and 20–30 %, respectively, of the total UV photoproducts (Mitchell and Nairn 1989). The pyrimidine dimers inhibit DNA replication and transcription by inducing structural distortions within the DNA, produced as a consequence of bending and unwinding of the DNA helix (Demple and Harrison 1994).
UV-induced ROS can damage DNA mostly by the formation of 8-hydroxydeoxyguanosine (8-OHdG) at the 5′-site of –GG– sequence in the double-stranded DNA. This modified guanine pairs with adenine at 50 % probability causing G:C to T:A transversions (Ito et al. 1993).
IR-Induced DNA Damage
Like UV, IR, too, can induce DNA damage both by being directly absorbed and by production of ROS as a consequence of radiolysis of water. The most common DNA lesions generated by IR include sugar and base damage, single- and double-strand breaks, and DNA–DNA and DNA–protein cross-links (Belli et al. 2002). Direct absorption of IR by the sugar–phosphate backbone of DNA generates nicks and DSBs. IR-induced SSBs are characterized by the presence of a one nucleotide gap containing a 5′-phosphate and either a 3′-phosphoglycolate or a 3′-phosphate (Henner et al. 1983).
Hydrolysis-Induced DNA Damage
Hydrolysis of glycosidic bonds between bases and sugar–phosphate backbone in DNA results in the formation of apurinic/apyrimidinic (AP) sites. These sites prevent normal DNA replication and transcription and are the most common locations for the creation of DSBs because of the collision of replication forks. These lesions are generally recognized and repaired under normal conditions, but their occasional bypass during replication can be mutagenic (Gentil et al. 1984).
Cytosine and methyl-cytosine can undergo hydrolytic deamination to uracil and thymine, respectively. During replication, this can lead to C:G to T:A transition because of mispairing with adenine. Though, the unusual presence of uracil in the DNA is promptly recognized and the lesion is repaired, the transition to thymine is difficult to detect and frequently leads to point mutation.
Alkylation-Induced DNA Damage
7-Methylguanine, 3-methyladenine, and O6-methylguanine are the most common lesions produced by alkylating agents like methyl methane sulfonate (MMS) and ethyl methane sulfonate. While 7-methylguanine is a neutral mutation, 3-methyladenine blocks DNA synthesis creating DSBs and O6-methylguanine pairs with thymine to produce G:C to A:T transition.
Oxidation-Induced DNA Damage
Increased ROS production is a common manifestation of many abiotic stresses in plants with chloroplasts and mitochondria being the major sites of production. ROS-induced oxidative damage to DNA includes modified bases and sugar, abasic sites, strand breaks, DNA–protein cross-links. The most common oxidation product of purines is 7,8-dihydro-8-oxoguanine, also known as 8-oxoguanine, which can base pair equally well with adenine and cytosine (Maki and Sekiguchi 1992). Thymine glycol is the main oxidatively modified form of pyrimidines and can block DNA replication. Furthermore, ROS can react with sugar of the sugar–phosphate backbone leading to SSBs and DSBs.
DDR Pathway: Perspectives from Animals and Plants
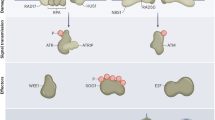
The DDR pathway has mostly been investigated in animals and yeast . At the molecular level, it consists of a number of components including DNA damage sensors, signal transducers, mediators, and effectors. In mammals , ataxia telangiectasia mutated (ATM), ATM, and Rad3-related (ATR), and DNA-dependent protein kinase subunit (DNA-PKcs) are the major regulators of DDR and are rapidly activated in response to DNA damage (Sirbu and Cortez 2013). DSBs are mostly sensed by ATM which initiates a cascade of steps that comprise the ATM pathway. ATR, on the other hand, can respond to a variety of DNA lesions, especially those associated with DNA replication to commence the ATR pathway (Cimprich and Cortez 2008).
Sensing the Damage
The different types of DNA damages are sensed by either the ATM or ATR pathway and culminate into a suitable response depending upon the severity of the damage. In mammals, the DSBs are recognized in the ATM pathway by a ternary MRN complex that comprises the proteins MRE11, RAD50, and NBS1 (Rupnik et al. 2010). This complex assembles at the site of DSBs and recruits ATM whereby the interaction with NBS1 activates the kinase activity of ATM resulting in the phosphorylation of target proteins, especially the histone variant H2AX. γH2AX, the phosphorylated form of H2AX, further recruits other DDR proteins (Dickey et al. 2009). Homologs of MRE11, RAD50, and NBS1 have been reported in Arabidopsis thaliana (Hartung 1999; Gallego and White 2001; Akutsu et al. 2007). Phosphorylation of H2AX by AtATM and AtATR, the A. thaliana homologs of ATM and ATR, has also been reported in response to DSBs (Friesner et al. 2005). That the induction of γH2AX was observed to be impaired in the rad50 and mre11 mutant plants suggested the involvement of the MRN complex in the phosphorylation of H2AX via the ATM and ATR homologs in plants (Amiard et al. 2010).
In mammals, replication protein A (RPA), a single-strand DNA (ssDNA)-binding protein acts as a sensor in the ATR pathway. The RPA-ssDNA complex formed at the site of stalled replication fork (near a DNA lesion) is bound by an ATR-interacting protein (ATRIP) which recruits ATR, eventually activating the check point kinase 1 (CHK1; Zou and Elledge 2003). Alternatively, the ATR pathway can be initiated by recognition of the damage by the RAD9/RAD1/HUS1 (9-1-1) complex. This complex is a ring-like clamp that is loaded onto ssDNA at the damage site with the help of a clamp loader, RAD17 (Bermudez et al. 2003). The homologs of RPA, 9-1-1 complex, and RAD17 have been identified in the A. thaliana genome (Takashi et al. 2009; Heitzeberg et al. 2004). Functional characterization of mutants of these homologs suggests that the DNA sensing mechanisms via the ATM and ATR pathway are conserved between animals and plants.
Transducing the Signal
For proper response, information about the damage detected by the sensors is amplified and transduced to the effector proteins by the mediation of transducers. These transducers are mostly serine/threonine kinases which initiate a sequence of phosphorylation steps. ATM/ATR and CHK1/CHK2 are the main transducers in animals. Activation of ATM involves its autophosphorylation and subsequent monomerization of the ATM dimer (Bakkenist and Kastan 2003), whereas ATR is brought into action by its recruitment to RPA-ssDNA complex by ATRIP (Ball and Cortez 2005; Warmerdam et al. 2010). The downstream substrates for phosphorylation by ATM and ATR include CHK1, CHK2, p53, NBS1, and BRCA1 (Matsuoka et al. 2000; Gatei et al. 2003; Zhao and Piwnica-Worms 2001). Of these, CHK1 and CHK2 are the main transducers which have common phosphorylation substrates like p53, BRCA1, E2F1, and CDC25A (Kim et al. 2007).
AtATM and AtATR, the Arabidopsis homologs of mammalian ATM and ATR have been identified. Analysis of the atm and atr mutants revealed their sensitivity to DNA damage inducing agents signifying the conservation of the role of these proteins as signal transducers in both animals and plants (Garcia et al. 2003; Culligan et al. 2004). Orthologs of ATRIP have also been reported in A. thaliana (Sweeney et al. 2009; Sakamoto et al. 2009). Although the orthologs of CHK1 and CHK2 have not been identified in plants the presence of other kinases having functions similar to these two proteins has been alluded to, in view of the presence of mediator proteins like BRCA1 and E2F in plants (Lafarge 2003; Inze and de Veylder 2006).
Between the Signal and Response: The Mediators
Different mediator proteins that regulate DDR have been identified and their roles have been described. The two most common mediators involved in the ATM pathway are BRCA1, mediator of DNA-damage checkpoint protein 1 (MDC1), and p53-binding protein (53BP1; Stewart et al. 2003; Stucki and Jackson 2004). DNA topoisomerase-2-binding protein 1(TOPBP1) and CLASPIN have been demonstrated to regulate the ATR pathway (Garcia et al. 2005; Kumagai et al. 2004). Many of these proteins interact with other phosphorylated proteins through a conserved BRCA1 associated C-terminal (BRCT) domain (Manke et al. 2003). These mediator proteins function as scaffolds for many DNA damage repair proteins. AtBRCA1, a homolog of BRCA1 with two BRCT domains, which is strongly induced by IR, has been reported in A. thaliana (Culligan et al. 2006; Lafarge 2003). Also, an Arabidopsis MEIOSIS DEFECTIVE 1 (ME1) protein with five BRCT domains having 40 % similarity to the human TOPBP1 has been reported although its role as a mediator in DRR remains to be confirmed (Mathilde et al. 2003). Other mediators are yet to be identified in plants.
Responding to the Damage: The Effectors
Cellular response to DNA damage depends on the severity of the damage. A cell tries to repair the damage by halting its progress in the cell cycle and stopping DNA replication followed by the activation of DNA repair pathways. If the damage is extensive and beyond repair, the cell prefers to initiate apoptosis. The ultimate result of DNA damage is decided by the action of effector proteins. In animals, the tumor suppressor protein p53 is the most important effector (Helton and Chen 2007). The amount and activity of this transcription factor is regulated very precisely by post-translational modifications, of which phosphorylation is the most crucial (Taira and Yoshida 2012). Under normal conditions, p53 levels are maintained at low levels by MDM2-mediated ubiquitination and proteasomal degradation (Xirodimas et al. 2004). DNA damage leads to phosphorylation of p53 by ATM, ATR, CHK1, and CHK2; phosphorylation inhibits the interaction of p53 with MDM2, thereby, resulting in its stabilization and activation (Shieh et al. 1997; Appella and Anderson 2001). Despite the conservation of many DRR proteins between animals and plants, a plant p53 homolog has long eluded plant scientists. Presence of factors with functions similar to that of p53 has been envisaged; SOG1 has been described as one such factor.
An essential prerequisite for initiating DNA repair is the halt or arrest of cell cycle to prevent the replication of damaged DNA or allow the division of cells carrying such DNA. Cyclin-dependent kinase (CDK) inhibitors, p21 and WEE1 kinase, are the major effectors that halt the cell cycle in response to DNA damage in animals (Abbas and Dutta 2009; Sorensen and Syljuasen 2012). While p21 arrests the cells at G1 in a p53-mediated manner (Reinhardt and Schumacher 2012), WEE1 prevents mitotic entry (McGowan and Russell 1995). In addition, several CDC25 phosphatases are also cell-cycle effectors. These phosphatases remove the inhibitory phosphate groups on CDKs to control the progression of cell cycle. Upon DNA damage, CDC25 is itself inactivated by phosphorylation mediated by CHK1 and CHK2 leading to the cell-cycle arrest (Karlsson-Rosenthal and Millar 2006). Although, homologs of p21, like its immediate regulator p53, have not been identified in plants several other CDK inhibitors (CDKIs) have been reported from A. thaliana. These CDKIs belong to two families, namely Kip-related protein (KRP) family and SIAMESE/SIAMESE-RELATED (SIM/SMR) family. DSBs strongly induce the expression of SMR4 and SMR5, indicating their involvement in cell-cycle arrest (Yoshiyama et al. 2009). AtWEE1 is the WEE1 homolog in A. thaliana that is activated by DNA damage or DNA replication arrest in an AtATM- or AtATR-dependent manner, respectively (de Schutter et al. 2007). It has been demonstrated to control cell-cycle arrest in the DDR pathway. Although a CDC25-like protein has been identified in A. thaliana, its role in cell-cycle regulation is not confirmed (Spadafora et al. 2011).
By applying brakes on the cell cycle, cell buys time to make amends to the damaged DNA . A number of conserved DNA repair pathways viz., nucleotide excision repair (NER), base excision repair (BER), homologous recombination (HR), non-homologous end-joining (NHEJ), and direct repair (DR) have been identified in yeast, animals, and plants. If the damage is beyond repair, animal cells activate apoptosis, the exquisitely regulated pathway to kill cells with compromised genomes. The diversion of cells from DNA repair pathways to apoptosis is mediated by the transcriptional activation of genes such as BAX, PUMA, and NOXA by p53 (Vousden and Lu 2002). Plants, too, have programmed cell death (PCD), but it is distinct from the apoptotic cell death in animals because plants lack the core apoptotic machinery. PCD involving AtATM and AtATR has been observed in shoot and root apical meristem in plants in response to DNA damage (Fulcher and Sablowski 2009). Metacaspases (MCs) that are structurally similar to animal caspases, the initiator proteins for apoptosis have also been reported in plants (Uren et al. 2000; Lam and Zhang 2012). Overexpression of some of these MCs increased the cell death upon treatment with ROS-inducing agents while their loss resulted in delay or decrease of cell death (Lam and Zhang 2012). It has been suggested that some of these pathways might be regulated by SOG1 in plants.
DNA Repair Pathways in Plants
Maintenance of genomic integrity is crucial to life. This comes at a huge premium in terms of energy expenditure and elaborate repair framework that organisms need to maintain. Although damages that do not interfere with the replication or transcription can sometimes be innocuous, those that hinder the activity of DNA and RNA polymerases are very harmful for the organism and need to be compulsorily repaired. Most of the repair mechanisms have been conserved through evolution and homologs of different elements have been identified across kingdoms. The whole genome sequence of A. thaliana and rice has revealed the presence of many repair proteins that are similar to those in humans. Characterization of mutants and overexpressing lines has provided functional validation for these proteins. A comprehensive list of DNA repair genes in the rice genome was provided by Kimura and Sakaguchi (2006). Like animals, plants are also equipped with a variety of repair pathways catering to the different types of DNA damages. These damages can be rectified through a number of pathways. Herein, we have described the main pathways, namely photoreactivation/photorepair (PR), base excision repair (BER), and nucleotide excision repair (NER).
Photoreactivation/Photorepair (PR) Pathway
As inherent in the name, photoreactivation is a light-dependent pathway majorly responsible for the repair of UV-B-induced DNA damage, especially CPDs. This pathway utilizes light-mediated reversion of the damage by photolyases (Yasui and Eker 1998). Photolyases generally contain two chromophoric co-factors, one of which is always the two electron reduced form of FAD (FADH−; Sancar 2003). FADH− acts as a transient electron donor to reverse the cross-link between the bases. The second chromophore acts as an antenna pigment to excite the electron donor. The reaction mechanism proceeds via a free radical mechanism. Photolyases bind specifically to DNA lesions absorbing blue/UV-A (320–400 nm) light and reducing pyrimidine dimers to monomers (Kimura et al. 2004; Fig. 7.2). A number of factors including quality, timing, and quantity of photoreactivating light as well as the severity of the damage affect this repair (Sutherland et al. 1996; Takeuchi et al. 1996; Stapleton et al. 1997). Although, PR is present in prokaryotes and eukaryotes including some species of plants and animals, it is not universal, with many species, including humans, lacking it (Todo 1999). The genes for photolyases have been cloned from higher plants such as A. thaliana, Cucumis sativus, Oryza sativa, and Spinacia oleracea. Six genes with photolyase activity have been identified in the Arabidopsis genome. The common UV-induced lesions, CPD and 6-4 PP, are specifically recognized by photolyases PHR1/UVR2 and UVR3, respectively. Arabidopsis mutants for these photolyases were defective in PR (Jiang et al. 1997). Similarly, rice cultivars with deficient PR of CPDs were reported to carry a mutation in the CPD photolyase (Hidemal et al. 2000; Teranishi et al. 2004). In contrast, transgenic rice overexpressing CPD photolyase were demonstrated to be tolerant to growth inhibition caused by UV-B-induced damage and accumulated lower CPD lesions in leaves during growth under UV-B irradiation (Hidemal et al. 2007). Further, diurnal changes in CPD photolyase expression have been observed in cucumber, suggesting regulation of the gene to prevent growth suppression by UV (Takahashi et al. 2002). In Arabidopsis, the overexpression of CPD photolyase resulted in a modest increase in biomass under UV-irradiated conditions (Kaiser et al. 2009). In addition, different ecotypes of A. thaliana have been demonstrated to possess variable UV-B response (Kalbina and Strid 2006).
Schematic representation of photoreactivation mechanism . The enzyme photolyase binds to the site of structural distortion in the DNA caused because of pyrimidine (thymine in this case) dimer (Step 1) and catalyzes the light (blue) activated reversion of the dimer to the monomeric form (Step 2). The enzyme is released from the DNA after the repair (Step 3)
Base Excision Repair
Only a minority of damaged bases in DNA are repaired by direct damage reversal; most of these are removed by excision repair (Lindahl and Wood 1999). Damaged bases that do not distort the helical structure of DNA are repaired by BER. Such damaged bases could be the result of spontaneous deamination or base loss or could be induced by IR, oxidative or methylating agents. In this multi-step repair pathway, specific DNA glycosylases excise the damaged base thereby creating an abasic (AP) site that is subsequently filled in by the correct DNA sequence (Lindahl and Wood 1999; Mol et al. 1995; Tuteja and Tuteja 2001). The repair mechanism has been classified into two sub-pathways: short-patch BER and long-patch BER. In short-patch BER, a glycosylase with specificity for a particular base-adduct slides along the minor grove of DNA helix and localizes to the site of the lesion where it removes the damaged base creating an AP site (Fromme et al. 2004). The abasic site is then recognized by an AP-endonuclease that nicks the DNA backbone on 5′-end of the AP site. Thereafter, the 5′-terminal deoxyribose-phosphate residue is excised by DNA polymerase β using its AP lyase activity (Matsumoto and Kim 1995). The missing base is then put in place by DNA polymerase β, and the nick is sealed by DNA ligase I or III with the help of XRCC1 (Fig. 7.3). In the case of long-patch BER, instead of the replacement of a single damaged base, a longer patch (2–10 nucleotides) is resynthesized by nick translation in conjunction with strand displacement in the 5′–3′ direction, generating a flap-like structure. The flap is removed by a flap endonuclease, FEN-1, with the aid of PCNA (Wu et al 1996; Klungland and Lindahl 1997; Fig. 7.3). The nick translation is catalyzed by DNA polymerase β or δ and the backbone is sealed by DNA ligase 1 (Fortini et al. 1998).
The study of BER in plants has revealed the presence of several homologs of different component proteins in A. thaliana and O. sativa (Kimura and Sakaguchi 2006). For example, glycosylases with specificity for particular modified bases have been reported (Santerre and Britt 1994; Garcia-Ortiz et al. 2001; Dany and Tissier 2001; Murphy and Gao 2001). Similarly, Arabidopsis XRCC1-like protein and rice FEN1 and DNA polymerase δ have been isolated (Martínez-Macías et al. 2013; Kimura et al. 2003; Uchiyama et al. 2002). However, plant homologs of DNA polymerase β and DNA ligase III have not yet been reported.
Nucleotide Excision Repair
Lesions that cause large distortions in the helical structure of DNA are generally restored by nucleotide excision repair. The steps involved in this mechanism include recognition of the damaged site, opening of the double helix by the action of helicases and endonucleases, excision of a DNA segment carrying the lesion, and replacement of the segment using the template provided by the intact complementary strand followed by ligation of the nick (Costa et al. 2003; Fig. 7.4). The proteins involved in NER are sequentially assembled at the site of the lesion (Volker et al 2001). NER is classified into two distinct sub-pathways that differ in the DNA damage recognition: transcription coupled repair (TCR) and global genome repair (GGR) . RNA polymerase II detects the lesion in TCR with the help of CSB and CSA. CSB has been demonstrated to alter DNA conformation and remodel chromatin in an ATP dependent manner (Citterio et al. 2000). In GGR) , XPC-hHR23B is the first factor to be involved in the lesion detection (Volker et al. 2001). In general, XPC-hHR23B complex is stabilized by hCEN2. Some other factors like XPE may aid in this recognition (Kusumoto et al. 2001). Subsequent to damage recognition other NER factors are recruited, which include TFIIH, XPD, XPA, XPB, RPA, XPF, and XPG (Evans et al. 1997). TFIIH is the basal transcription initiation factor that comprises nine different proteins (Winkler et al. 1998; Coin et al. 1999). XPD and XPB are ATP-dependent helicases that unwind the DNA helix in 5′–3′ and 3′–5′ direction, respectively (Schaeffer et al. 1994; Roy et al. 1994). The replication protein A (RPA) binds to DNA to provide complete opening of the helix around the lesion (Christians and Hanawalt 1993). XPA homodimer binds to RPA to form XPA2-RPA complex that provides proper three-dimensional orientation of the NER components required by excision of damage carrying DNA segment (Missura et al. 2001; Yang et al. 2002). With the DNA lesion recognized and helix around it unwound, XPG carries out structure-specific 3′-endonucleolytic activity (2–8 nucleotide away from the lesion) in conjunction with the 5′-endonucleolytic activity (15–24 nucleotide away from the lesion) of an XPF-ERCC1 complex. Thus, the lesion carrying segment of DNA is excised and is dissociated. Thereafter, the resultant gap is filled by DNA polymerase ε and δ using the 3′-end left by the XPF-ERCC1 incision as the primer (Hunting et al. 1991; Coverley et al. 1992). These polymerases require the presence of PCNA and RFC (Wood and Shivij 1997). Finally, the 5′-end of the newly synthesized DNA segment is sealed with the parent strand by DNA ligase I (Tomkinson and Levin 1997).
Schematic representation of nucleotide excision repair. Please refer to the text for explanation of the steps in the pathway. (Adapted from Fuss and Cooper 2006)
Most of the genes of NER pathway have been reported from A. thaliana (The Arabidopsis Genome Initiative 2000). Many genes with sequence similar to those of yeast and mammals have been cloned (Schultz and Quatrano 1997; Sturm and Lienhard 1998; Taylor et al. 1998; Costa et al. 2001; Liu et al. 2003; Dubest et al. 2004; Kimura et al. 2004). In addition, NER-related genes like PCNA, DNA polymerase δ, RPA, and CSB have been cloned from rice (Kimura and Sakaguchi 2006). The functional analysis of these genes and characterization of many mutants defective in some of them confirms the importance of NER in DNA damage repair in plants and the conservation of this pathway across the living kingdoms.
Conclusions
DNA damage response and repair pathways play a crucial role in maintenance of the integrity of genome and have been conserved to a great extent in various organisms. Many of these pathways have been thoroughly examined in mammals and yeast . Studies on plants have lagged behind in this regard. However, with the increasing genomic data available as a consequence of high-throughput sequencing efforts, homologs of different proteins involved in these pathways have been identified and functionally evaluated. Although most of the components are present, some crucial factors like p53, XRCC, DNA polymerase γ, DNA ligase III, etc. have been conspicuously absent. Furthermore, some genes like CSB, RPA, PCNA, and FEN1 are present in multiple copies in plants. Some repair defects, for example, RAD50 knockout, which are lethal in animals, have been reported to have no physiological abnormalities in plants. Existence of novel plant-specific DDR components in plants has been alluded to in plants. The ongoing genetic, proteomic, and knock-out based strategies are expected to unveil novel DNA damage sensing and repair components in plants.
References
Abbas T, Dutta A (2009) p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer 9:400–414
Akutsu N, Iijima K, Hinata T, Tauchi H (2007) Characterization of the plant homolog of Nijmegen breakage syndrome 1: involvement in DNA repair and recombination. Biochem Biophys Res Commun 353:394–398
Alscher RG, Donahue JL, Cramer CL (1997) Reactive oxygen species and antioxidants: relationships in green cells. Physiol Plant 100:224–233
Amiard S, Charbonnel C, Allain E, Depeiges A, White CI, Gallego ME (2010) Distinct roles of the ATR kinase and the MRE11-RAD50-NBS1 complex in the maintenance of chromosomal stability in Arabidopsis. Plant Cell 22:3020–3033
Appella E, Anderson CW (2001) Post-translational modifications and activation of p53 by genotoxic stresses. Eur J Biochem 268:2764–2772
Bakkenist CJ, Kastan MB (2003) DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421:499–506
Ball HL, Cortez D (2005) AtRIP oligomerization is required for ATR-dependent checkpoint signaling. J Biol Chem 280:31390–31396
Belli M, Sapora O, Tabocchini MA (2002) Molecular targets in cellular response to ionizing radiation and implications in space radiation protection. J Radiat Res 43:S13-9
Bermudez VP, Lindsey-Boltz LA, Cesare AJ, Maniwa Y, Griffith JD, Hurwitz J, Sancar A (2003) Loading of the human 9-1-1 checkpoint complex onto DNA by the checkpoint clamp loader hRAD17-replication factor C complex in vitro. Proc Natl Acad Sci U S A 100:1633–1638
Biedermann S, Mooney S, Hellmann H (2011) Recognition and repair pathways of damaged DNA in higher plants. In: Chen C (ed) Selected topics in DNA repair. InTech, Rijeka, pp 201–236
Britt A (1999) Molecular genetics of DNA repair in higher plants. Trends Plant Biol 4:20–25
Christians FC, Hanawalt PC (1993) Lack of transcription-coupled repair in mammalian ribosomal RNA genes. Biochemistry 32:10512–10518
Ciccia A, Elledge SJ (2010) The DNA damage response: making it safe to play with knives. Mol Cell 40:179–204
Cimprich KA, Cortez D (2008) Atr: an essential regulator of genome integrity. Nat Rev Mol Cell Biol 9:616–627
Citterio E, van den Boom V, Schnitzler G, Kanaar R, Bonte E, Kingston RE, Hoeijmakers JHJ (2000) RNA polymerase II elongation complexes containing the Cockayne’s syndrome group B protein interacts with a molecular complex containing the transcription factor IIH component Xeroderma pigmentosum B and p62. Mol Cell Biol 20:7643–7653
Coin F, Bergmann E, Bravard AT, Egly JM (1999) Mutations in XPB and XPD helicases found in Xeroderma pigmentosum patients impair the transcription function of TFII-H. EMBO J 18:1357–1366
Costa RM, Morgante PG, Berra CM, Nakabashi M, Bruneau D, Bouchez D, Sweder KS, Van Sluys MA, Menck CF (2001) The participation of AtXPB1, the XPB/RAD25 homologue gene from Arabidopsis thaliana, in DNA repair and plant development. Plant J 28:385–395
Costa RMA, Chigancas V, Galhardo RS, Carvalho H, Menck CFM (2003) The eukaryotic nucleotide excision repair pathway. Biochimie 85:1083–1099
Coverley D, Kenny MK, Lane DP, Wood RD (1992) A role for the human single-stranded DNA binding protein HSSB/RPA in an early stage of nucleotide excision repair. Nucleic Acids Res 20:3873–3880
Culligan K, Tissier A, Britt A (2004) ATR regulates a G2-phase cell-cycle checkpoint in Arabidopsis thaliana. Plant Cell 16:1091–1104
Culligan KM, Robertson CE, Foreman J, Doerner P, Britt AB (2006) ATR and ATM play both distinct and additive roles in response to ionizing radiation. Plant J 48:947–961
Dany AL, Tissier A (2001) A functional OGG1 homologue from Arabidopsis thaliana. Mol Genet Genom 265:293–301
Davis CD, Uthus EO, Finley JW (2000) Dietary selenium and arsenic affect DNA methylation in vitro in Caco-2 cells and in vivo in rat liver and colon. J Nutr 130:2903–2909
de Schutter K, Joubes J, Cools T, Verkest A, Corellou F, Babiychuk E, van Der Schueren E, Beeckman T, Kushnir S, Inze D et al (2007) Arabidopsis WEE1 kinase controls cell cycle arrest in response to activation of the DNA integrity checkpoint. Plant Cell 19:211–225
Demple B, Harrison L (1994) Repair of oxidative damage to DNA—enzymology and biology. Annu Rev Biochem 63:915–948
Dickey JS, Redon CE, Nakamura AJ, Baird BJ, Sedelnikova OA, Bonner WM (2009) H2AX: functional roles and potential applications. Chromosoma 118:683–692
Dubest S, Gallego ME, White CI (2004) Roles of the AtErcc1 protein in recombination. Plant J 39:334–342
Evans E, Fellows J, Coffer A, Wood RD (1997) Open complex formation around the lesion during nucleotide excision repair provides a structure for cleavage by human XPG protein. EMBO J 16:625–638
Fortini P, Pascucci B, Parlanti E, Sobol RW, Wilson SH, Dogliotti E (1998) Different DNA polymerases are involved in the short- and long-patch base excision repair in mammalian cells. Biochemistry 37:3575–3580
Friedberg EC, Walker GC, Siede W, Wood RD, Schultz RA, Ellenberger T (2006) DNA repair and mutagenesis. ASM Press, Washington, DC
Friesner JD, Liu B, Culligan K, Britt AB (2005) Ionizing radiation-dependent gamma-H2AX focus formation requires ataxia telangiectasia mutated and ataxia telangiectasia mutated and rad3-related. Mol Biol Cell 16:2566–2576
Fromme JC, Banerjee A, Verdine GL (2004) DNA glycosylase recognition and catalysis. Curr Opin Struct Biol 14:43–49
Fulcher N, Sablowski R (2009) Hypersensitivity to DNA damage in plant stem cell niches. Proc Natl Acad Sci U S A 106:20984–20988
Fuss JO, Cooper PK (2006) DNA repair: dynamic defenders against cancer and aging. PLoS Biol 4:e203
Gallego ME, White CI (2001) Rad50 function is essential for telomere maintenance in Arabidopsis. Proc Natl Acad Sci U S A 98:1711–1716
Garcia V, Bruchet H, Camescasse D, Granier F, Bouchez D, Tissier A (2003) AtATM is essential for meiosis and the somatic response to DNA damage in plants. Plant Cell 15:119–132
Garcia V, Furuya K, Carr AM (2005) Identification and functional analysis of TOBP1 and its homologs. DNA Repair 4:1227–1239
Garcia-Ortiz MV, Ariza RR, Roldan-Arjona T (2001) An OGG1 orthologue encoding a functional 8-oxoguanine DNA glycosylase/lyase in Arabidopsis thaliana. Plant Mol Biol 47:795–804
Gatei M, Sloper K, Sorensen C, Syljuasen R, Falck J, Hobson K, Savage K, Lukas J, Zhou BB, Bartek J et al (2003) Ataxia-telangiectasia-mutated (atm) and nbs1-dependent phosphorylation of CHK1 on Ser-317 in response to ionizing radiation. J Biol Chem 278:14806–14811
Gentil A, Margot A, Sarasin A (1984) Apurinic sites cause mutations in simian virus 40. Mutat Res 129:141–147
Hartung HPF (1999) Isolation of the complete cDNA of the Mre11 homolog of Arabidopsis (accession no. Aj243822) indicates conservation of DNA recombination mechanisms between plants and other eucaryotes. Plant Physiol 121:312
Heitzeberg F, Chen IP, Hartung F, Orel N, Angelis KJ, Puchta H (2004) The RAD17 homologue of Arabidopsis is involved in the regulation of DNA damage repair and homologous recombination. Plant J Cell Mol Biol 38:954–968
Helton ES, Chen X (2007) p53 modulation of the DNA damage response. J Cell Biochem 100:883–896
Henner WD, Grunberg SM, Haseltine WA (1983) Enzyme action at 3′ termini of ionizing radiation-induced DNA strand breaks. J Biol Chem 258:15198–15205
Hidemal J, Kumagai T, Sutherland BM (2000) UV radiation-sensitive Norin 1 rice contains defective cyclobutane pyrimidine dimer photolyase. Plant Cell 12:1569–1578
Hidemal J, Taguchil T, Onol T, Teranishil M, Yamamoto K, Kumagai T (2007) Increase in CPD photolyase activity functions effectively to prevent growth inhibition caused by UVB radiation. Plant J 50:70–79
Hunting DJ, Gowans BJ, Dresler SL (1991) DNA polymerase delta mediate excision repair in growing cells damaged with ultraviolet radiation. Biochem Cell Biol 69:303–308
Huang X-D, Dixon DG, Greenberg BM (1993) Impacts of UV radiation and photomodification on the toxicity of pahs to the higher plant Lemna gibba (duckweed). Environ Toxicol Chem 12:1067–1077
Imlay JA, Linn S (1988) DNA damage and oxygen radical toxicity. Science 240:1302–1309
Inze D, de Veylder L (2006) Cell cycle regulation in plant development. Annu Rev Genet 40:77–105
Iovine B, Nino M, Irace C, Bevilacqua MA, Monfrecola G (2009) Ultraviolet B and A irradiation induces fibromodulin expression in human fibroblasts in vitro. Biochimie 91:364–372
Ito K, Inoue S, Yamamoto K, Kawanishi S (1993) 8-Hydroxydeoxyguanosine formation at the 5′ site of 5′-GG-3′ sequences in double-stranded DNA by UV radiation with riboflavin. J Biol Chem 268:13221–13227
Jiang CZ, Yee J, Mitchell D, Britt A (1997) Photorepair mutants of Arabidopsis. Proc Natl Acad Sci U S A 94:7441–7445
Kaiser G, Kleiner O, Beisswenger C, Batschauer A (2009) Increased DNA repair in Arabidopsis plants overexpressing CPD photolyase. Planta 230:505–515
Kalbina I, Strid Å (2006) Supplementary ultraviolet-B irradiation reveals differences in stress responses between Arabidopsis thaliana ecotypes. Plant Cell Environ 29:754–763
Karlsson-Rosenthal C, Millar JB (2006) Cdc25: mechanisms of checkpoint inhibition and recovery. Trends Cell Biol 16:285–292
Kim MA, Kim HJ, Brown AL, Lee MY, Bae YS, Park JI, Kwak JY, Chung JH, Yun J (2007) Identification of novel substrates for human checkpoint kinase CHK1 and CHK2 through genome-wide screening using a consensus CHK phosphorylation motif. Exp Mol Med 39:205–212
Kimura S, Sakaguchi K (2006) DNA repair in plants. Chem Rev 106:753–766
Kimura S, Furukawa T, Kasai N, Mori Y, Kitamoto HK, Sugawara F, Hashimoto J, Sakaguchi K (2003) Functional characterization of two flap endonuclease-1 homologues in rice. Gene 314:63–71
Kimura S, Tahira Y, Ishibashi T, Mori Y, Mori T, Hashimoto J, Sakaguchi K (2004) DNA repair in higher plants; photoreactivation is the major DNA repair pathway in non-proliferating cells while excision repair (nucleotide excision repair and base excision repair) is active in proliferating cells. Nucleic Acids Res 32:2760–2767
Klungland A, Lindahl T (1997) Second pathway for completion of human DNA base excision-repair; reconstitution with purified proteins and requirement for DNase IV (FEN1). EMBO J 16:3341
Kumagai A, Kim SM, Dunphy WG (2004) Claspin and the activated form of ATR-ATRIP collaborate in the activation of CHK1. J Biol Chem 279:49599–49608
Kusumoto R, Masutani C, Sugasava K, Iwai S, Araki M, Uchida A, Mizukoshi T, Hanaoka F (2001) Diversity of the damage recognition step in the global genomic nucleotide excision repair in vitro. Mutat Res 485:219–227
Lafarge S (2003) Characterization of Arabidopsis thaliana ortholog of the human breast cancer susceptibility gene 1: AtBRCA1, strongly induced by gamma rays. Nucleic Acids Res 31:1148–1155
Lam E, Zhang Y (2012) Regulating the reapers: activating metacaspases for programmed cell death. Trends Plant Sci 17:487–494
Lindahl T, Wood RD (1999) Quality control by DNA repair. Science 286:1897–1905
Liu Z, Hong SW, Escobar M, Vierling E, Mitchell DL, Mount DW, Hall JD (2003) Arabidopsis UVH6, a homolog of human XPD and yeast RAD3 DNA repair genes, functions in DNA repair and is essential for plant growth. Plant Physiol 132:1405–1414
Maki H, Sekiguchi M (1992) MutT protein specifically hydrolyses a potent mutagenic substrate for DNA synthesis. Nature 355:273–275
Manke IA, Lowery D, Nguyen A, Yaffe MB (2003) BRCT repeats as phosphopeptide-binding modules involved in protein targeting. Science 302:636–639
Mannuss A, Trapp O, Puchta H (2012) Gene regulation in response to DNA damage. Biochim Biophys Acta 1819:154–165
Martínez-Macías MI, Córdoba-Cañero D, Ariza RR, Roldán-Arjona T (2013) The DNA repair protein XRCC1 functions in the plant DNA demethylation pathway by stimulating cytosine methylation (5-meC) excision, gap tailoring, and DNA ligation. J Biol Chem 288:5496–5505
Mathilde G, Ghislaine G, Daniel V, Georges P (2003) The Arabidopsis ME1 gene encodes a protein with five BRCT domains that is involved in meiosis-specific DNA repair events independent of SPO11-induced DSBs. Plant J 35:465–475
Matsumoto Y, Kim K (1995) Excision of deoxyribose phosphate residues by DNA polymerase beta during DNA repair. Science 269:699–702
Matsuoka S, Rotman G, Ogawa A, Shiloh Y, Tamai K, Elledge SJ (2000) Ataxia telangiectasia-mutated phosphorylates CHK2 in vivo and in vitro. Proc Natl Acad Sci U S A 97:10389–10394
McGowan CH, Russell P (1995) Cell cycle regulation of human WEE1. EMBO J 14:2166–2175
McGregor WG (1999) DNA repair, DNA replication, and UV mutagenesis. J Invest Dermatol Symp Proc 4:1–5
Missura M, Buterin T, Hindges R, Hubscher U, Kasparkova J, Brabec V, Naegeli H (2001) Double-check probing of DNA bending and unwinding by XPA-RPA: an architectural function in DNA repair. EMBO J 20:3554–3564
Mitchell DL, Nairn RS (1989) The biology of the (6-4) photoproduct. Photochem Photobiol 49:805–819
Mol CD, Arvai AS, Slupphaug G, Kavli B, Alseth I (1995) Crystal structure and mutational analysis of human uracil-DNA glycosylase: structural basis for specificity and catalysis. Cell 80:1–20
Mouret S, Baudouin C, Charveron M, Favier A, Cadet J, Douki T (2006) Cyclobutane pyrimidine dimers are predominant DNA lesions in whole human skin exposed to UVA radiation. Proc Natl Acad Sci U S A 103:13765–13770
Murphy TM, Gao MJ (2001) Multiple forms of formamidopyrimidine-DNA glycosylase produced by alternative splicing in Arabidopsis thaliana. J Photochem Photobiol B 61:87–93
Pang Q, Hays JB (1991) UV‐B‐inducible and temperature‐sensitive photoreactivation of cyclobutane pyrimidine dimers in Arabidopsis thaliana. Plant Physiol 95:536–543
Reinhardt HC, Schumacher B (2012) The p53 network: cellular and systemic DNA damage responses in aging and cancer. Trends Genet 28:128–136
Roy R, Adamczewski JP, Seroz T, Vermeulen W, Tassan JP, Schaeffer L, Nigg EA, Hoeijmakers JH, Egly JM (1994) The M015 cell cycle kinase is associated with the TFIIH transcription-DNA repair factor. Cell 79:1093–1101
Rupnik A, Lowndes NF, Grenon M (2010) Mrn and the race to the break. Chromosoma 119:115–135
Sakamoto AN, Lan VT, Puripunyavanich V, Hase Y, Yokota Y, Shikazono N, Nakagawa M, Narumi I, Tanaka AA (2009) UVB-hypersensitive mutant in Arabidopsis thaliana is defective in the DNA damage response. Plant J Cell Mol Biol 60:509–517
Sancar A (2003) Structure and function of DNA photolyase and cryptochrome bluelight photoreceptors. Chem Rev 103:2203–2237
Santerre A, Britt AB (1994) Cloning of a 3-methyladenine-DNA glycosylase from Arabidopsis thaliana. Proc Natl Acad Sci U S A 91:2240–2244
Schaeffer L, Moncollin V, Roy R, Staub A, Mezzina M, Sarasin A, Weeda G, Hoeijmakers JH, Egly JM (1994) The ERCC2/DNA repair protein is associated with class II BTF2/TFIIH transcription factor. EMBO J 13:2388–2392
Schultz TF, Quatrano RS (1997) Characterization and expression of a rice RAD23 gene. Plant Mol Biol 34:557–562
Shieh SY, Ikeda M, Taya Y, Prives C (1997) DNA damage-induced phosphorylation of p53 alleviates inhibition by mdm2. Cell 91:325–334
Singh SK, Roy S, Choudhury SR, Sengupta DN (2010) DNA repair and recombination in higher plants: insights from comparative genomics of Arabidopsis and rice. BMC Genomics 11:443
Sirbu BM, Cortez D (2013) DNA damage response: three levels of DNA repair regulation. Cold Spring Harb Perspect Biol 5(8):a012724. doi:10.1101/cshperspect.a012724
Sorensen CS, Syljuasen RG (2012) Safeguarding genome integrity: the checkpoint kinases ATR, CHK1 and WEE1 restrain CDK activity during normal DNA replication. Nucleic Acids Res 40:477–486
Spadafora ND, Doonan JH, Herbert RJ, Bitonti MB, Wallace E, Rogers HJ, Francis D (2011) Arabidopsis T-DNA insertional lines for cdc25 are hypersensitive to hydroxyurea but not to zeocin or salt stress. Ann Bot 107:1183–1192
Stapleton AE, Thornber CS, Walbot V (1997) UV-B component of sunlight causes measurable damage in field-grown maize (Zea mays L.): developmental and cellular heterogeneity of damage and repair. Plant Cell Environ 20:279–290
Stewart GS, Wang B, Bignell CR, Taylor AM, Elledge SJ (2003) Mdc1 is a mediator of the mammalian DNA damage checkpoint. Nature 421:961–966
Stucki M, Jackson SP (2004) Mdc1/nfbd1: a key regulator of the DNA damage response in higher eukaryotes. DNA Rep 3:953–957
Sturm A, Lienhard S (1998) Two isoforms of plant Rad23 complement a UV-sensitive rad23 mutant in yeast. Plant J 13:815–821
Sutherland BM, Takayanagi S, Sullivan JH, Sutherland JC (1996) Plant responses to changing environmental stress: cyclobutyl pyrimidine dimer repair in soybean leaves. Photochem Photobiol 64:464–468
Sweeney PR, Britt AB, Culligan KM (2009) The Arabidopsis ATRIP ortholog is required for a programmed response to replication inhibitors. Plant J Cell Mol Biol 60:518–526
Taira N, Yoshida K (2012) Post-translational modifications of p53 tumor suppressor: determinants of its functional targets. Histol Histopathol 27:437–443
Takahashi S, Nakajima N, Saji H, Kondo N (2002) Diurnal change of cucumber CPD photolyase gene (CsPHR) expression and its physiological role in growth under UV-B irradiation. Plant Cell Physiol 43:342–349
Takashi Y, Kobayashi Y, Tanaka K, Tamura K (2009) Arabidopsis replication protein a 70a is required for DNA damage response and telomere length homeostasis. Plant Cell Physiol 50:1965–1976
Takeuchi Y, Murakami M, Nakajima N, Kondo N, Nikaido O, Takeuchi Y (1996) The photorepair of photoisomerization of DNA lesions in etiolated cucumber cotyledons after irradiation by UV-B depends on wavelength. Plant Cell Physiol 39:745–750
Taylor RM, Hamer MJ, Rosamond J, Bray CM (1998) Molecular cloning and functional analysis of the Arabidopsis thaliana DNA ligase I homologue. Plant J 14:75–81
Teranishi M, Iwamatsu Y, Hidema J, Kumagai T (2004) Ultraviolet-B sensitivities in Japanese lowland rice cultivars: cyclobutane pyrimidine dimer photolyase activity and gene mutation. Plant Cell Physiol 45:1848–1856
The Arabidopsis Genome Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408:796–815
Todo T (1999) Functional diversity of the DNA photolyase/blue light receptor family. Mutat Res 434:89–97
Tomkinson AE, Levin DS (1997) Mammalian DNA ligases. Bioeassays 19:893–901
Tuteja N, Tuteja R (2001) Unraveling DNA repair in human: molecular mechanisms and consequences of repair defect. Crit Rev Biochem Mol Biol 36:261–290
Tuteja N, Singh MB, Misra MK, Bhalla PL, Tuteja R (2001) Molecular mechanisms of DNA damage and repair: progress in plants. Crit Rev Biochem Mol Biol 36:337–397
Uchiyama Y, Hatanaka M, Kimura S, Ishibashi T, Ueda T, Sakakibara Y, Matsumoto T, Furukawa T, Hashimoto J, Sakaguchi K (2002) Characterization of DNA polymerase delta from a higher plant, rice (Oryza sativa L.). Gene 295:19–26
Uren AG, O’Rourke K, Aravind LA, Pisabarro MT, Seshagiri S, Koonin EV, Dixit VM (2000) Identification of paracaspases and metacaspases: two ancient families of caspase-like proteins, one of which plays a key role in malt lymphoma. Mol Cell 6:961–967
Vespa L, Couvillion M, Spangler E, Shippen DE (2005) ATM and ATR make distinct contributions to chromosome end protection and the maintenance of telomeric DNA in Arabidopsis. Genes Dev 19:2111–2115
Volker M, Mone MJ, Karamakar P, van Hoffen A, Schul W, Vermuelen W, Hooeijmakers JHJ, van Driel R, van Zeeland A, Mullenders LHF (2001) Sequential assembly of nucleotide excision repair factors in vivo. Moll Cell 8:213–224
Vousden KH, Lu X (2002) Live or let die: the cell’s response to p53. Nat Rev Cancer 2:594–604
Ward JF (1975) Molecular mechanisms of radiation-induced damage to nucleic acids. Adv Radiat Biol 5:181–239
Warmerdam DO, Kanaar R, Smits VA (2010) Differential dynamics of ATR-mediated checkpoint regulators. J Nucleic Acids 319142:1–16
Wasi S, Tabrez S, Ahmad M (2013) Toxicological effects of major environmental pollutants: an overview. Environ Monit Assess 185:2585–2593
Winkler GS, Vermeulen W, Coin F, Egly JM, Hoeijmakers JH, Weeda G (1998) Affinity purification of human DNA repair/transcription factor TFIIH using epitope-tagged Xeroderma pigmentosum B protein. J Biol Chem 273:1092–1098
Wiseman H, Halliwell B (1996) Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem J 313:17–29
Wood RD, Shivij MKK (1997) Which DNA polymerase are used for DNA repair in eucaryotes? Carcinogenesis 18:605–610
Wu X, Li J, Li X, Hsieh CL, Burgers PM, Lieber MR (1996) Processing of branched DNA intermediates by a complex of human FEN-1 and PCNA. Nucleic Acids Res 24:2036–2043
Xirodimas DP, Saville MK, Bourdon JC, Hay RT, Lane DP (2004) Mdm2-mediated nedd8 conjugation of p53 inhibits its transcriptional activity. Cell 118:83–97
Yang ZG, Liu Y, Mao LY, Zhang JT, Zhou Y (2002) Dimerization of human XPA and formation of XPA-RPA protein complex. Biochemistry 41:13012–13020
Yasui A, Eker APM (1998) DNA photolyases. In: Nickoloff JA, Hoekstra MF (eds) DNA damage and repair, vol II. Humana Press, Totowa, pp 9–32
Yoshiyama K, Conklin PA, Huefner ND, Britt AB (2009) Suppressor of gamma response 1 (sog1) encodes a putative transcription factor governing multiple responses to DNA damage. Proc Natl Acad Sci U S A 106:12843–12848
Yoshiyama KO, Sakaguchi K, Kimura S (2013) DNA damage response in plants: conserved and variable response compared to animals. Biology 2:1338–1356
Zhao H, Piwnica-Worms H (2001) Atr-mediated checkpoint pathways regulate phosphorylation and activation of human CHK1. Mol Cell Biol 21:4129–4139
Zou L, Elledge SJ (2003) Sensing DNA damage through AtRIP recognition of RPA-ssDNA complexes. Science 300:1542–1548
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Fatima, U., Khan, M.F., e Fatima, J., Shahab, U., Ahmad, S., Yusuf, M.A. (2017). DNA Damage, Response, and Repair in Plants Under Genotoxic Stress. In: Sarwat, M., Ahmad, A., Abdin, M., Ibrahim, M. (eds) Stress Signaling in Plants: Genomics and Proteomics Perspective, Volume 2. Springer, Cham. https://doi.org/10.1007/978-3-319-42183-4_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-42183-4_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-42182-7
Online ISBN: 978-3-319-42183-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)