Abstract
The study of the ‘smell of death’ is an important part of the thanatochemistry, the chemistry of death. Since 2004 (Vass et al. 2004), an increasing number of studies have been conducted to understand the body decomposition process by measuring the Volatile Organic Compounds (VOCs) released by decaying bodies. However, the chemical profile of the decomposition odor is still far for from being resolved. Indeed, the complexity of the VOC mixture makes it difficult to be carried out by classical GC-MS. A better understanding of the decomposition process could thus possibly be achieved using a multidimensional technique such as Comprehensive Two Dimensional Gas Chromatography coupled to time of flight mass spectrometry (GC×GC-TOFMS). The high peak capacity of this multidimensional technique combined with the visualization power of multivariate statistical methods allows a deeper understanding of complex VOC matrices.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Forensic science is a dynamic area. Police officers always need advanced analytical methods to help them in their investigations. For several years, researchers have been working on volatile evidence analysis. Many cases of investigation are linked with volatile organic compound (VOC) mixtures including drug, contraband, and terrorism. Nowadays, police dogs are mainly used for searching for volatile evidence in investigations (Lorenzo et al. 2003; Oesterhelweg et al. 2008). However, the dog detection mechanism is not yet completely understood. Indeed, it is not yet known which compounds police dogs react to during the detection of drugs, explosives and bodies. Lorenzo et al. studied the olphactometric fingerprint for some illicit products. These results assisted with dog training improvement (Lorenzo et al. 2003).
Another area of investigation for forensic volatiles is decomposition odor analysis. People start to study VOC originating from decomposition in 2004. A decomposition VOC database was created during this study containing approximately 400 compounds (Vass et al. 2004). Several additional studies tried to elucidate the process of decomposition following the release of VOCs (Statheropoulos et al. 2005, 2007, 2011; Vass et al. 2004, 2008; Paczkowski and Schütz 2011; Pandey and Kim 2011; Hoffman et al. 2009; DeGreeff and Furton 2011; Dekeirsschieter et al. 2009). Allmost all of these studies were based on gas chromatography (GC) methods coupled with different types of sampling methods.
Unfortunately, VOC mixtures from decaying bodies belong to the most complex volatile matrixes from Life Sciences. Indeed, the decomposition process is dynamic and the VOC concentration profile is changing over time. However, a better understanding of this process could help dog trainers to improve their training techniques. Improvement of cadaver dog training would not only help in crime solving but also improve the efficiency in finding trapped people after a natural disaster (Statheropoulos et al. 2006). In the past, gas chromatography – mass spectrometry (GC-MS) methods were used for this kind of forensic VOC investigation. Nevertheless, the resolution limits has not yet allowed a full understanding of the decomposition VOC profile.
In 2011, GC×GC was applied to the forensic field for the first time. Mitrevski et al. used it for the analysis of the volatile profile of ecstasy to determine the drug’s origin. de Vos et al. also applied GC×GC for environmental forensic analysis (de Vos et al. 2011). They used GC×GC for Persistent Organic Pollutants (POPs) analysis in developing countries that do not have access to GC systems coupled to high-resolution mass spectrometry (HRMS). Their study shows that even though GC×GC-TOFMS is a non-target method, it allows screening of the compounds that are present in the environment and it can provide a good estimation of the concentration levels (de Vos et al. 2011). GC×GC was also successfully applied for complex matrix analysis in different metabolomics studies (Seeley and Seeley 2013). Hence, it is considered as a good approach to solve the many co-elution problems often encountered in classical GC methods.
In 2012, Brasseur et al. used multidimensional gas chromatography (MDGC) to analyze grave soil samples in order to overcome the 1D GC limitations (Brasseur et al. 2012). In subsequent years, several decomposition studies were conducted using GC×GC-TOFMS. Dekeirsshieter et al. monitored the VOC profile from pig carcasses during the different stages of decomposition. This study demonstrated that the list of compounds in the decomposition database is not exhaustive. Therefore, a method with high separation power is required (Dekeirsschieter et al. 2012). Stadler et al. conducted different studies on decomposition VOCs. The first study analyzed the VOC profile of synthetic training aids used for cadaver dog training solution analysis. That study determined that these training aids are very simple mixtures when compared to the true VOC profile from a decaying body. However, dogs trained with these solutions are able to locate human remains (Stadler et al. 2012). The second study was focused on the comparison of the decomposition process between different environments in Canada and Belgium. They used TD-GC×GC-TOFMS. This method combined the sampling advantage of TD (Thermal Desorption) and the strong separation capability of GC×GC. This research demonstrated for the first time the similarities between two different decomposition trials conducted in different countries (Stadler et al. 2013).
An additional chromatographic dimension is helpful to improve the resolution of individual VOCs. Moreover, data treatment with multivariate statistical methods, such as Principal Component Analysis (PCA), can improve the data visualization and simplify the interpretation. These kinds of statistical methods were first applied to decomposition studies in 2006 by Statheropoulos et al. (2006). The aim of this current paper is to demonstrate the advantage of combining the chromatographic resolution power of GC×GC-TOFMS and the visualization enhancement of multivariate statistics for data handling. This combination of methods is applied to grave soil VOC analysis to demonstrate the different aspects of these tools and their value to forensic investigators.
2 Comprehensive Two Dimensional Gas Chromatography
Comprehensive Two Dimensional Gas Chromatography is a multidimensional approach that offers the possibility to isolate and identify compounds present in complex mixtures (Seeley and Seeley 2013). GC×GC was introduced almost 25 years ago by Liu and Phillips (Liu and Phillips 1991). This method is based on two chromatographic separations that separate the compounds on a chromatographic plane. GC×GC allows the screening of large numbers of compounds in one GC run. The main advantages of this method are the increase in peak capacity and the possibility to obtain a structured chromatogram (Dallüge et al. 2003).
The majority of hardware equipment in a GC×GC system is the same as in classical GC except the addition of a secondary column and the presence of a special device, i.e. a modulator (Seeley and Seeley 2013). The modulator is the interface between the two dimensions of separation (Ryan and Marriott 2003). The design of efficient modulators was crucial for GC×GC development. The key of a GC×GC application is the ability to rapidly pulse segments of effluent from the first to the second dimension (Phillips and Beens 1999). The modulator samples peaks eluting from the first dimension and injects them into the second dimension. The co-eluting peaks from the first dimension will be sent together into the second column where they will separate according to the selectivity of the second dimension. This additional separation contributes to cleaner mass spectra but the ultimate aim is, of course, to correctly resolve all the peaks on the chromatographic space to use the mass spectra for identification purposes. The slicing process also improves the deconvolution of the co-eluting peaks. The peak height is increased by the compression process in the modulator. This process increases the peak intensity and the limit of detection compared with classical GC (Patterson et al. 2011; Shellie et al. 2001). There are two major types of modulator: thermic and valve systems. Both types must serve three functions: (1) Continuously trap small adjacent fractions of the eluent from the primary dimension; (2) Refocus the trapped sections; and (3) Inject the refocused trapped slices into the secondary dimension (Dallüge et al. 2003).
The selection of the best columns combination for a specific analysis is not obvious to achieve a proper comprehensive separation. There are three important parameters to take into account to ensure a comprehensive separation that were developed from the Giddings’s rules (Giddings 1987). First, all peaks must pass through the two dimensions. Second, the two dimensions of separation must be orthogonal (meaning that the process of separation is entirely different on each column). Finally, the second dimension must be in fast GC condition.
Nowadays, there are numerous GC columns available on the market. Column manufacturers are introducing new kinds of stationary phases, either to increase the existing polarity range or using completely different separation principles, e.g. with liquid crystalline phases. Recently, Supelco® introduced the ionic liquid columns (de Boer et al. 1992). These columns allow GC users to obtain very high polarity selectivity (Armstrong et al. 2009). The constant development of new columns is really helpful to determine the best separation conditions for a specific analysis. This wide selection of phases offers a bench of possibilities to tune the separation. A suitable column combination will ensure a full separation (Dallüge et al. 2003; Ryan et al. 2005; Ryan and Marriott 2003). In multidimensional methods, each added dimension gives additional information that can be used for identification (Dallüge et al. 2003). The column set must be carefully selected taking into account the sample composition (Dallüge et al. 2003; Ryan et al. 2005). For example, in polychlorinated biphenyls (PCBs) congeners analysis, a structured chromatograms can give important additional information that is helpful for the identification (Korytar et al. 2002; Focant et al. 2004). More recently, Seeley et al. combined two semi-polar columns to obtain a specific separation for the FAME compounds in diesel samples (Seeley et al. 2012). These studies show the importance of the structure in multidimensional chromatography.
The last Giddings’s rule is very important as the separation obtained in the first dimension must be maintained in the secondary column. An important consideration is the length ratio between the two columns. The first column length is similar to that used in classical GC. Thus, the separation is similar to a 1D GC temperature program analysis. The second dimension must be a series of fast GC isothermal separations. The second column has to be smaller to fit with this restriction. The combination of these two separations gives a GC×GC two dimensional chromatogram (e.g. Fig. 20.1) (Seeley and Seeley 2013). Additionally, the modulation period must be chosen carefully to obtain the best space occupation on the two dimensional separation space (Ryan et al. 2005).
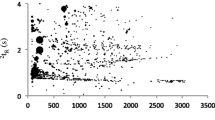
3D chromatogram of the VOCs trapped in the headspace of the grave soil. The two retention axes are in seconds and the 1D traces for each dimension are shown. The peak occupation is important due to the sample complexity. Without a multidimensional approach, the complete resolution of this mixture is not possible
Peaks elute from the second dimension with a Full Width at Half Maximum (FWHM) in the order of 100–300 ms. This represents less than 10 % of the peak width in conventional GC analysis. Hence, GC×GC detectors need to have a fast response (Seeley and Seeley 2013). There are several detectors that can reach the acquisition rate required for GC×GC. There are electron capture detectors (ECD, μECD), flam ionization detectors (FID) and other elemental detectors that do not allow full identification (Dallüge et al. 2003). But GC×GC systems can also be connected with a mass spectrometer. The mass spectra can be compared with databases (e.g. NIST and Wiley) to obtain peak identification. Due to the acquisition speed required, quadrupole MS is not suitable. A Time of Flight Mass Spectrometer (TOFMS) is the most viable technology available to provide this rapid acquisition capability (Ryan and Marriott 2003; Dallüge et al. 2003). The absence of concentration skewing ensures spectral continuity and allows for effective mass spectral deconvolution of the co-eluting peaks characterized by different fragmentation patterns (Cochran 2002; Focant et al. 2004). Recently, High Resolution Time of Flight Mass Spectrometers (HRTOFMS) have also become available coupled to GC×GC systems. These new instruments will improve the mass spectra dimension without losing the required acquisition rate. HRMS provides exact masses for the ions that can be linked to their chemical formula improving the identification capabilities (Ochiai et al. 2011; Ieda et al. 2011).
For sample injection, GC×GC systems can be linked to all the classical injection devices that are used for classical GC. All the methods usually used in 1D GC can be implemented on 2D GC. The most used one is probably the liquid injection but to study VOC mixtures Thermal Desorption (TD) methods are more efficient (Barro et al. 2009; Ramírez et al. 2010; Brokl et al. 2013).
One of the major drawback of the GC×GC technique is the number of parameters that can affect the separation. The additional parameters make the optimization steps more complicated. GC×GC users need to choose the best column combination and set the best modulation period… (Dallüge et al. 2003). Condition optimization is important to ensure optimal separation by GC×GC (Semard et al. 2011; Mostafa et al. 2012).
The data treatment software is another critical part in GC×GC. The commercial availability of GC×GC instruments was strongly linked to the development of dedicated software. Due to the raw data complexity, GC×GC instruments must be linked to powerful software for the data processing and to obtain the final chromatogram (Fig. 20.1). The chromatogram obtained from the detector must be transformed to obtain a 2D chromatogram and as a result, the data process follows different steps. A raw (1D) chromatogram is cut in several slices due to the modulation process. Based on the modulation period (PM), the software will divide the 1D traces into different pieces. Each piece will be rotated by 90° to obtain the 2D space chromatogram. After that, each modulation slice will be combined to reconstruct the real GC peaks. If a mass spectrometer is used for the detection, a mass spectra comparison is applied to determine if two consecutive slices are part of the same peak and have to be combined. The slice-to-slice combination also improves the deconvolution capabilities. Slices from the same peak must be separated by one PM and the mass spectra of each slice must be the same. The deconvolution improvement is critical for very complex samples because even with the increased peak capacity, co-elutions can remain. Finally, a 2D chromatogram is obtained with two retention times, one on the x-axis and one on the y-axis. The z-axis is used to show the peak intensity based on the detector response (see Fig. 20.1). If a mass detector is used, a mass library identification can be performed to identify the compound linked to each peak (Dimandja 2004; Seeley and Seeley 2013; Ramos 2009; Mondello et al. 2008).
When the 2D chromatogram is obtained, different data comparison tools are available. To compare a set of data robust comparison tools are recommended (Almstetter et al. 2011; Castillo et al. 2011). The LECO® ChromaTOF software contains a Statistical Compare (SC) option. This feature allows chromatogram alignment and Fisher Ratio (FR) calculations. SC aligns the chromatograms based on the two retention times and on a mass spectra comparison. When the chromatogram is aligned, the FR can be calculated. A Fisher Ratio is the ratio of “between class” variance to “within class” variance. This factor identifies the compounds that are significantly different from one class to another (Almstetter et al. 2011). This tool assists with finding biomarkers that are responsible for group segregation when different compounds have to be compared (e.g. different origin of drugs).
Chromatogram comparison is like DNA analysis. It helps to identify the links that exist between different samples. Each peak is comparable to a gene and the complete peak pattern represents the genetic fingerprint of the sample. If different samples have some groups of peaks in common, it means that they are probably linked.
3 GC×GC and Multivariate Statistics: A Helpful Combination!
As explained before, GC×GC offers a real solution for complete separation of complex samples. However, this improvement of separation is linked to an increase in the data complexity. Dallüge notes that “The amount of data generated per run is overwhelming and data handling is, consequently, rapidly becoming the real analytical problem” (Dallüge et al. 2003). To solve this problem, GC×GC users need tools that can reduce the amount of data. A way to achieve this is the use of multivariate statistics.
The combination of GC×GC resolution power and robust statistical tools can be helpful when a large number of different samples need to be compared. GC×GC separation provides all the information about the composition of one sample. It is its chemical fingerprint. The comparison of this chemical signature with databases or other samples can identify the origin of one substance. The combination of comprehensive GC and multivariate statistic has been employed to analyze the VOC profile from beers and olive oils (Cajka et al. 2010a, b). These products are much appreciated by customers all over the world and their geographic origin can have a significant impact on the prices. Economic fraud by incorrect labeling is sometimes observed and should be avoided. Thus, analytical methods that can verify the traceability of these products are important to the producers. In these two studies, the traceability was studied using GC×GC and multivariate analysis (Cajka et al. 2010a, b). This combination of multidimensional chromatography and statistics was also applied in 2011 to a forensic application. Mitrevski et al. used GC×GC-TOFMS to analyze the VOC profiles of ecstasy from different countries. Based on the volatile fingerprint, Principal Component Analysis (PCA) was applied to observe clustering of the different kind of drugs according to their country of origin. The improvement of peak capacity and signal enhancement due to the use of GC×GC method allowed the detection of low level markers of the drug’s origin (Mitrevski et al. 2011).
These studies are the result of the combination of the chromatographic resolution from GC×GC analysis and the data visualization power of multivariate statistic.
4 Application for Grave Soils Analysis
In this study, TD-GC×GC-TOFMS was used in combination with multivariate statistics to monitor the VOC profile from grave soil samples collected above a grave at different depths. The method was based on a study by Brasseur et al., which used GC × GC for grave soil investigations. The study identified different kinds of biomarkers with the highest number coming from the soil below the pig. Notably, branched alkane compounds were found through all grave soil depths above the pig carcass up to the surface. Based on a scripting method, an algorithm was developed to search the different peaks and identify the branched alkanes. The scripts were based on the specific fragmentation pattern of alkanes in electron ionization source (Brasseur et al. 2012).
The research was conducted by trapping VOCs on tubes and desorption using a liquid solvent. However, Brokl et al. demonstrated that the volatile profile is influenced if the sampling is conducted using liquid extraction versus thermal desorption. Moreover, the number of peaks detected in the headspace were two times higher using thermal desorption (Brokl et al. 2013). Based on this result, it was decided to investigate the volatile organic compound profile of grave soil samples using TD-GC × GC-TOFMS. This experiment follow the pig were the same as in a previous study realized by Brasseur et al. (Brasseur et al. 2012). The pigs for the two studies were buried at the same time but the excavation took place after a different delay (17 months). Three hundred grams of the soil above the pig carcass was sampled every 10 centimeters from the surface to the carcass. The VOCs were trapped using the same kind of pumping device as Brasseur reported (Brasseur et al. 2012) but the sorbent tubes were chosen to be more polyvalent. A combination of Tenax® and Carbopack B® wad used to trap a higher number of VOCs. The efficiency of these sorbent tubes has been previously reported in other decomposition studies (Stadler et al. 2013).
The TD-GC×GC-TOFMS system used was a Markes® Unity 2 TD (Llantrisant, U.K) linked to a LECO® Pegasus 4D (St. Joseph, MI). The separation was conducted on a reverse column set: a polar ionic liquid (SLB-IL-111; 30 m × 0.25 mm × 0.25 μm) column in the first dimension and a non-polar polysiloxane (Rtx-1; 1 m × 0.1 mm × 0.08 μm) column in the second dimension. This particular configuration was chosen because most of the decomposition biomarkers found in previous studies are polar or semi-polar compounds. These compounds are better separated on a reverse column combination (polar – non-polar) (Dimandja et al. 2003).
The chemical composition of the headspace was analyzed at each depth. The results are displayed on Fig. 20.2. It shows that the VOCs from the top of the grave are mostly hydrocarbon compounds. This observation is consistent with the result obtained by Brasseur et al. in the headspace of a grave. At a depth of 10 cm, the most abundant chemical class is the carboxylic acids. Carboxylic acids likely result from the degradation of amino acids and lipids. At a depth of 30 and 34 cm, the most abundant class is that of the sulfur compounds. Sulfides are typically the most abundant compounds in the headspace of decomposition. Dimethyl disulfide and dimethyl trisulfide are predominantly responsible for this abundance (Stadler et al. 2013).
The Statistical Compare feature of the LECO Software (see section above) was also used to determine which compounds are different between grave soil samples and control soil samples. Fig. 20.3 shows on the top PCA that the grave soil samples (red) are correctly separated from the control soil samples (blue). The principal component axis responsible for this segregation is PCA (1). On the lower plot, the PCA (1) component was plotted vs. the different chemical families (the variables of the multivariate analysis). The green points represent the classes that have a medium influence on the PCA (1). The three points have different colors and demonstrated the greatest impact. These points correspond to the chemical classes with the highest impact on the separation. This observation is consistent with the conclusion from Fig. 20.2. The chemicals, which are significantly different between the grave soil and the control, are present in greater quantity.
The top figure displays the Principal Component Analysis of the VOC profiles from a grave (red) and a control (blue). The first axes clearly separates the two samples. The plot at the bottom shows which chemical families are responsible for this separation: the carboxylic acids (blue), the hydrocarbons (red) and the sulfur compounds (yellow)
The major advantage of the Statistical Compare approach is the speed of the data processing. It allows direct alignment of all the samples. Moreover, the comparison of all the peaks through the different samples allows multivariate statistics to visualize how the different classes are clustering and which variables (i.e. compounds) are responsible for this separation.
5 Conclusion
A comprehensive decomposition VOC profile is still unknown due to the complexity and the dynamic nature of the decomposition process. Moreover, the decomposition markers are often linked to other complex matrices (e.g. soils). Since 2004, an increasing number of studies have investigated the VOCs profile of decaying bodies. However, a robust analytical method is required to ensure a comprehensive analysis of decomposition samples.
This study aimed to demonstrate the real analytical improvements offered by GC × GC-TOFMS. The high resolution of separation can isolate the markers from the matrix and the mass spectral data can provide an identification of the compounds. The implementation of multivariate analysis (e.g. Principal Component Analysis) in the data processing is helpful to obtain a better visualization of the results. This visualization tool helps to reduce the data dimensionality of GC×GC-TOFMS analysis.
This combination of multidimensional chromatography and multivariate statistic offers solutions to overcome the analytical limitations of decomposition studies. Moreover, it offers solutions for VOC profiling in different areas of forensic sciences.
References
Almstetter MF et al (2011) Comparison of two algorithmic data processing strategies for metabolic fingerprinting by comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry. J Chromatogr A 1218(39):7031–7038
Armstrong DW, Payagala T, Sidisky LM (2009) The advent and potential impact of ionic liquid stationary phases in GC and GC and GCXGC. LC GC Eur 22(9):459–461
Barro R et al (2009) Analysis of industrial contaminants in indoor air: Part 1. Volatile organic compounds, carbonyl compounds, polycyclic aromatic hydrocarbons and polychlorinated biphenyls. J Chromatogr A 1216(3):540–566
Brasseur C et al (2012) Comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry for the forensic study of cadaveric volatile organic compounds released in soil by buried decaying pig carcasses. J Chromatogr A 1255:163–170
Brokl M et al (2013) Analysis of mainstream Tobacco smoke particulate phase using comprehensive two-dimensional gas chromatography time-of-flight mass spectrometry. J Sep Sci 36(6):1037–1044
Cajka T, Riddellova K, Tomaniova M et al (2010a) Recognition of beer brand based on multivariate analysis of volatile fingerprint. J Chromatogr A 1217(25):4195–4203
Cajka T, Riddellova K, Klimankova E et al (2010b) Traceability of olive oil based on volatiles pattern and multivariate analysis. Food Chem 121:282–289
Castillo S, Mattila I, Miettinen J (2011) Data analysis tool for comprehensive Two-dimensional Gas chromatography/time-of-flight mass spectrometry – analytical chemistry (ACS publications). Anal Chem 83(8):3058–3067
Cochran JW (2002) Fast gas chromatography-time-of-flight mass spectrometry of polychlorinated biphenyls and other environmental contaminants. J Chromatogr Sci 40(5):254–268
Dallüge J, Beens J, Th UAB (2003) Comprehensive two-dimensional gas chromatography: A powerful and versatile analytical tool. J Chromatogr A 100:69–108
de Boer J, Dao QT, van Dortmond R (1992) Retention times of fifty one chlorobiphenyl congeners on seven narrow bore capillary columns coated with different stationary phases. J High Resolut Chromatogr 5(4):249–255
de Vos J et al (2011) Comprehensive two-dimensional gas chromatography time of flight mass spectrometry (GC × GC-TOFMS) for environmental forensic investigations in developing countries. Chemosphere 82:1230–1239
DeGreeff LE, Furton KG (2011) Collection and identification of human remains volatiles by non-contact, dynamic airflow sampling and SPME-GC/MS using various sorbent materials. Anal Bioanal Chem 401:1295–1307
Dekeirsschieter J et al (2009) Cadaveric volatile organic compounds released by decaying pig carcasses (Sus domesticus L.) in different biotopes. Forensic Sci Int 189(1–3):46–53
Dekeirsschieter J et al (2012) Enhanced characterization of the smell of death by comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry (GCxGC-TOFMS) PLoS ONE 7(6) p.e39005
Dimandja J-M (2004) Comprehensive 2-D GC provides high-performance separations in terms of selectivity, sensitivity, speed, and structure. Anal Chem 76(9):167A–174A
Dimandja J-M, Clouden G, Colón I (2003) Standardized test mixture for the characterization of comprehensive two-dimensional gas chromatography columns: the Phillips mix. J Chromatogr A 1019:261–272
Focant J-F, Sjödin A, Patterson DG (2004) Improved separation of the 209 polychlorinated biphenyl congeners using comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry. J Chromatogr A 1040(2):227–238
Giddings JC (1987) Concepts and comparisons in multidimensional separation. J High Resolut Chromatogr 10(5):319–323
Hoffman E, Curran A, Dulgerian N (2009) Characterization of the volatile organic compounds present in the headspace of decomposing human remains. Forensic Sci Int 186(1–3):6–13
Ieda T et al (2011) Environmental analysis of chlorinated and brominated polycyclic aromatic hydrocarbons by comprehensive two-dimensional gas chromatography coupled to high-resolution time-of-flight mass spectrometry. J Chromatogr A 1218(21):3224–3232
Korytar P et al (2002) High-resolution separation of polychlorinated biphenyls by comprehensive two-dimensional gas chromatography. J Chromatogr A 958:203–218
Liu Z, Phillips JB (1991) Comprehensive two-dimensional gas chromatography using an on-column thermal modulator interface. J Chromatogr Sci 29(6):227–231
Lorenzo N et al (2003) Laboratory and field experiments used to identify Canis lupus var. Familiaris active odor signature chemicals from drugs, explosives, and humans. Anal Bioanal Chem 376(8):1212–1224
Mitrevski B et al (2011) Chemical signature of ecstasy volatiles by comprehensive two-dimensional gas chromatography. Forensic Sci Int 209(1–3):11–20
Mondello L et al (2008) Comprehensive two-dimensional gas chromatography-mass spectrometry: a review. Mass Spectrom Rev 27(2):101–124
Mostafa A, Edwards M, Górecki T (2012) Optimization aspects of comprehensive two-dimensional gas chromatography. J Chromatogr A 1255:38–55
Ochiai N et al (2011) Stir bar sorptive extraction and comprehensive two-dimensional gas chromatography coupled to high-resolution time-of-flight mass spectrometry for ultra-trace analysis of organochlorine pesticides in river water. J Chromatogr A 1218(39):6851–6860
Oesterhelweg L et al (2008) Cadaver dogs–a study on detection of contaminated carpet squares. Forensic Sci Int 174(1):35–39
Paczkowski S, Schütz S (2011) Post-mortem volatiles of vertebrate tissue. Appl Microbiol Biotechnol 91(4):917–935
Pandey SK, Kim K-H (2011) Human body-odor components and their determination. Trends Anal Chem 30(5):784–796
Patterson DG et al (2011) Cryogenic zone compression for the measurement of dioxins in human serum by isotope dilution at the attogram level using modulated gas chromatography couple to high resolution magnetic sector mass spectrometry. J Chromatogr A 1218:3274–3281
Phillips JB, Beens J (1999) Comprehensive two-dimensional gas chromatography: a hyphenated method with strong coupling between the two dimensions. J Chromatogr A 856:331–347
Ramírez N et al (2010) Comparative study of solvent extraction and thermal desorption methods for determining a wide range of volatile organic compounds in ambient air. Talanta 82(2):719–727
Ramos L (2009) Comprehensive two dimensional gas chromatography. Elsevier, Amsterdam
Ryan D, Marriott P (2003) Comprehensive two-dimensional gas chromatography. Anal Bioanal Chem 376(3):295–297
Ryan D, Morrison P, Marriott P (2005) Orthogonality considerations in comprehensive two-dimensional gas chromatography. J Chromatogr A 1071(1–2):47–53
Seeley JV, Seeley SK (2013) Multidimensional gas chromatography: fundamental advances and new applications. Anal Chem 85(2):557–578
Seeley JV et al (2012) Stationary phase selection and comprehensive two-dimensional gas chromatographic analysis of trace biodiesel in petroleum-based fuel. J Chromatogr A 1226:103–109
Semard G et al (2011) Comparative study of differential flow and cryogenic modulators comprehensive two-dimensional gas chromatography systems for the detailed analysis of light cycle oil. J Chromatogr A 1218(21):3146–3152
Shellie R, Marriott P, Morrison P (2001) Concepts and preliminary observations on the triple-dimensional analysis of complex volatile samples by using GC × GC − TOFMS. Anal Chem 73(6):1336–1344
Stadler S et al (2012) Analysis of synthetic canine training aids by comprehensive two-dimensional gas chromatography-time of flight mass spectrometry. J Chromatogr A 1255:202–206
Stadler S et al (2013) Characterization of volatile organic compounds from human analogue decomposition using thermal desorption coupled to comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry. Anal Chem 85(2):998–1005
Statheropoulos M et al (2005) Preliminary investigation of using volatile organic compounds from human expired air, blood and urine for locating entrapped people in earthquakes. J Chromatogr B Anal Technol Biomed Life Sci 822(1–2):112–117
Statheropoulos M et al (2006) Discriminant analysis of volatile organic compounds data related to a new location method of entrapped people in collapsed buildings of an earthquake. Anal Chim Acta 566(2):207–216
Statheropoulos M et al (2007) Environmental aspects of VOCs evolved in the early stages of human decomposition. Sci Total Environ 385(1–3):221–227
Statheropoulos M et al (2011) Combined chemical and optical methods for monitoring the early decay stages of surrogate human models. Forensic Sci Int 210(1–3):154–163
Vass AA et al (2004) Decompositional odor analysis database. J Forensic Sci 49(4):1–10
Vass AA et al (2008) Odor analysis of decomposing buried human remains. J Forensic Sci 53:384–391
Acknowledgement
We wish to thank Restek® Corp., SGE® and Supelco Sigma-Aldrish® for providing GC phases and various GC consumables. We would also like to thank JSB® and LECO® for the technical support on the system. We also want to thank the University of Bradford, the University of Ontario Institute of Technology, University of Technology Sydney and Gembloux agro biotech (University of Liège) for their contribution on the sampling.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Stefanuto, PH., Focant, JF. (2016). GC×GC-TOFMS, the Swiss Knife for VOC Mixtures Analysis in Soil Forensic Investigations. In: Kars, H., van den Eijkel, L. (eds) Soil in Criminal and Environmental Forensics. Soil Forensics. Springer, Cham. https://doi.org/10.1007/978-3-319-33115-7_20
Download citation
DOI: https://doi.org/10.1007/978-3-319-33115-7_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33113-3
Online ISBN: 978-3-319-33115-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)




 Alcohol,
Alcohol,  Aldehyde,
Aldehyde,  Aromatic,
Aromatic,  Carboxylic acid,
Carboxylic acid,  Ester,
Ester,  Halogen,
Halogen,  Hydrocarbon,
Hydrocarbon,  Ketone,
Ketone,  Nitrogen,
Nitrogen,  Sulfur,
Sulfur,  Other
Other