Abstract
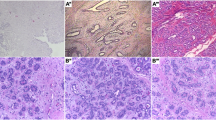
Pancreatic tumors are characterized by marked deposition of extra-cellular matrix, also called desmoplasia, which interacts with tumor cells and facilitates the tumor onset and progression. Thus, it would be relevant to develop a method to quantitatively assess the amount of desmoplasia in images derived from bioptic tissue fragments of the pancreas. To this purpose, we applied the principles of fractal geometry, for the assessment of the fractal dimension of images of Masson’s trichrome stained pancreatic tissue. Thus, we implemented an algorithm for the computation of the Hausdorff dimension, based on the box counting method: the image is split into boxes of identical size, and the number of boxes needed to cover the features of interest in the image is counted. The process is then iterated with boxes of lower size, and finally all box counts obtained at the different steps are considered, to get the estimate of the Hausdorff dimension, D. After validating the algorithm with appropriate tests, we applied it to pancreatic images, where some regions of interest (ROI) were identified, including both healthy and non-healthy (fibrotic) tissue. We found that non-healthy ROI typically show higher D values than healthy ROI (1.927±0.086 vs. 1.750±0.070 (mean±SD), p=0.0013). Thus, our approach may be of help for an accurate quantification of the degree of severity of pancreatic tumors.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Apte M, Pirola RC, Wilson JS (2015) Pancreatic stellate cell: physiologic role, role in fibrosis and cancer. Curr Opin Gastroenterol 31:416–423 DOI 10.1097/MOG.0000000000000196
Dioguardi N, Grizzi F, Franceschini B, Bossi P, Russo C (2006) Liver fibrosis and tissue architectural change measurement using fractal-rectified metrics and Hurst’s exponent. World J Gastroenterol 12:2187–2194
Mandelbrot B (1967) How long is the coast of Britain? Statistical self-similarity and fractional dimension. Science 156:636–638
West BJ (1990) Physiology in fractal dimensions: error tolerance. Ann Biomed Eng 18:135–149
Glenny RW, Robertson HT, Yamashiro S, Bassingthwaighte JB (1991) Applications of fractal analysis to physiology. J Appl Physiol 70:2351–2367
Cross SS (1997) Fractals in pathology. J Pathol 182:1-8
Di Ieva A, Grizzi F, Jelinek H, Pellionisz AJ, Losa GA (2013) Fractals in the neurosciences, Part I: general principles and basic neurosciences. Neuroscientist 20:403-417
Di Ieva A, Esteban FJ, Grizzi F, Klonowski W, Martín-Landrove M (2015) Fractals in the neurosciences, Part II: clinical applications and future perspectives. Neuroscientist 21:30-43
Schaefer HJ (1957) A rapid trichrome stain of Masson type. Am J Clin Pathol 28:646–647
Cohen AH (1976) Masson’s trichrome stain in the evaluation of renal biopsies. An appraisal. Am J Clin Pathol 65:631–643
Eden A (1990) Local estimates for the Hausdorff dimension of an attractor. J Math Anal Appl 150:100–119 DOI 10.1016/0022-247X(90)90198-O
Mattila P (2004) Hausdorff dimension, projections, and the Fourier transform. Publ Mat 48:3–48
Keyes T, Seeley G, Weakliem P, Ohtsuki T (1987) Collision-induced light scattering from growing clusters. Depolarization by fractals. J Chem Soc, Faraday Trans 2 83:1859–1866 DOI 10.1039/F29878301859
Mandelbrot BB, Evertsz CJG (1990) The potential distribution around growing fractal clusters. Nature 348:143–145
Lin DC, Sharif A (2007) Wavelet transform modulus maxima based fractal correlation analysis. Eur Phys J B 60:483–491 DOI 10.1140/epjb/e2008-00004-6
Santolalla C, Chavez-Esquivel G, Reyes-Heredia JADL, Alvarez-Ramirez J (2013) Fractal correlation analysis of x-ray diffraction patterns with broad background. Ind Eng Chem Res 52:8346–8353 DOI 10.1021/ie303069y
Mainieri R (1993) On the equality of Hausdorff and box counting dimensions. Chaos 3:119–126
Dai M, Liu X (2008) The Hausdorff and box-counting dimensions of a class of recurrent sets. Chaos Soliton Fract 36:532–538 DOI 10.1016/j.chaos.2006.06.083
Ebrahimi Khabbazi A, Hinebaugh J, Bazylak A (2015) Analytical tortuosity-porosity correlations for Sierpinski carpet fractal geometries. Chaos Soliton Fract 78:124–133 DOI 10.1016/j.chaos.2015.07.019
Dioguardi N, Grizzi F, Franceschini B, Bossi P, Russo C (2006) Liver fibrosis and tissue architectural change measurement using fractal-rectified metrics and Hurst’s exponent. World J Gastroenterol 12:2187–2194
Dioguardi N, Grizzi F, Fiamengo B, Russo C (2008) Metrically measuring liver biopsy: a chronic hepatitis B and C computer-aided morphologic description. World J Gastroenterol 14:7335–7344
Lin KY, Maricevich M, Bardeesy N, Weissleder R, Mahmood U (2008) In vivo quantitative microvasculature phenotype imaging of healthy and malignant tissues using a fiber-optic confocal laser microprobe. Transl Oncol 1:84–9
Vasilescu C, Giza DE, Petrisor P, Dobrescu R, Popescu I, Herlea V (2012) Morphometrical differences between resectable and non-resectable pancreatic cancer: a fractal analysis. Hepatogastroenterol 59:284–288 DOI 10.5754/hge11277
Constantine VS, Mowry RW (1968) Selective staining of human dermal collagen. II. The use of picrosirius red F3BA with polarization microscopy. J Invest Dermatol 50:419–423
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Scampicchio, A. et al. (2016). Assessment of the Fractal Dimension of Images Derived by Biopsy of Pancreatic Tissue: Implications for Tumor Diagnosis. In: Kyriacou, E., Christofides, S., Pattichis, C. (eds) XIV Mediterranean Conference on Medical and Biological Engineering and Computing 2016. IFMBE Proceedings, vol 57. Springer, Cham. https://doi.org/10.1007/978-3-319-32703-7_77
Download citation
DOI: https://doi.org/10.1007/978-3-319-32703-7_77
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-32701-3
Online ISBN: 978-3-319-32703-7
eBook Packages: EngineeringEngineering (R0)