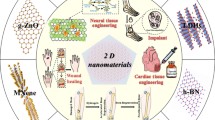
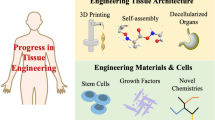
Abstract
Regenerative medicine aims to repair tissues or organs for restoring normal functions, which represents one of the greatest challenges in modern day science and medicine. Diverse techniques and materials are required to truly understand the process of tissue repairing and build a proper scaffold for cells attachment, proliferation and differentiation. Functionalized nanomaterials with nanotechnologies are the ideal to solve most of the problems of regenerative medicine. Multifunctionalized nanoparticles and nanostructured biomaterials can be powerful tools for cell tracking and matrix-like scaffold rebuilding.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Regenerative medicine is an interdisciplinary field of research and clinical applications focused on the repair, replacement or regeneracy of cells, tissues or organs to restore impaired function resulting from any cause (Daar and Greenwood 2007), which combines diverse techniques to stimulate or support the body’s own self-healing capacity. Though numerous implants and engineered tissues have been developed which are based on the current knowledge of the superstructure and the microstructure of tissue in last several decades, the truly regenerative therapies require significant understanding and controlling the underlying nanostructures in cells, the extracellular matrix (ECM) and also the cell behaviors during the tissue regeneracy (Harrison 2008; Zhang and Webster 2009).
In order to truly understand of the regenerative process during the tissue reconstruction, the knowledge of cell migration, proliferation and/or differentiation after the regenerative therapies is critical. Cell imaging and cell tracking as one of these methods is used to investigate the detail process during tissue regeneracy. The knowledge from cell tracking helps to advance new technologies for improving regenerative medicine in vice versa (Vaccaro et al. 2008; Solanki et al. 2008). An ideal cell tracking technology should be multifunctional (able to be both imaged and quantified), high sensitive, nontoxic, have long life time after labeling, and label cells with high efficiency (Vaccaro et al. 2008).
Furthermore, in a regenerative strategy, a scaffold normally required for promoting new tissue formation by providing adequate space (porosity) and appropriate surface to foster and direct cellular attachment, migration, proliferation, desired differentiation of specific cell phenotypes throughout the scaffold where new tissue formation is needed (Wei and Ma 2008; Chaikof et al. 2002).
Nanomaterials is a prosperous field in materials science based on the nanotechnology which was first defined by Taniguchi (1974). Nanotechnology, as a tool for fabricating nanomaterials, is the study of the control of matter on an atomic and molecular scale, which has the potential to create many new materials and devices with wide-ranging applications, such as in medicine, electronics, and energy production. The scale of nanomaterials made by nanotechnology is usually smaller than 100 nm meter in at least one dimension (Buzea et al. 2007), though sometimes also smaller than 1 μm, especially in the biological area.
Nanotechnology or the use of nanomaterials may have the answers since only these materials can be a powerful tool to track cell and mimic surface properties (including topography, composite, etc.) of natural tissues or delivery growth factors for tissue regeneracy. Nanomaterials are the materials with complex nanostructures, normally are fabricated by bottom-up or top-down methods. At the nanometer scale, where many biological processes operate, for example, the functional structures on the cell membrane, enzyme reactions, protein dynamics and DNA all possess some aspect of nanodimensionality (Harrison 2008). With significant advancements in synthetic and modification methodologies, nanomaterials can be modified to desired sizes, shapes, compositions and properties, which can be used as an ideal cell tracking label the cells without toxicity (Solanki et al. 2008). Furthermore, the ECM that the cells interact with also abounds with nanosized features which not only adjust the behaviors of the cells contacted with, but also influence the other cells and even tissues. These nanosized features, such as the size of fibers, the pores of matrix, and the chemical composition control the mechanical properties, the cell adhesion, proliferation and even differentiation on the matrix (Harrison 2008).
6.2 Principles
Since tissue regeneracy is a complex and precise process, in which cell behaviors, nanosized structures, chemical components of ECM and cytokines play critical roles. Understanding these critical aspects in the tissue repairing process helps to develop new techniques for fabricating proper materials in the regenerative medicine.
6.2.1 Nanoparticles for Cell Tracking
The importance of tracking cells in regenerative medicine is increasing because of the developing of basic cell therapy science, which is critical for cell delivery optimization and for accurate biodistribution studies (Vaccaro et al. 2008; Solanki et al. 2008).
In general, there are little cells retained in the target site after cell injection, which is found by cell tracking method. Wentworth et al. (2007) labeled skeletal myoblasts, and bone marrow stromal cells with Europium nanoparticles in advance, then the labeled cells were injected in vivo into the rat heart. The results showed that only approximately 15 % of the delivered cells were retained shortly after cell injection and the cells kept losing during the following 5 days. Other groups have reported similar cell retention numbers at the therapeutic site after injection, ranging from 5 to 15 % (Wentworth et al. 2007; Freyman et al. 2006). By immunohistochemical detection, the authors found that the macrophage infiltrate contribute to losses of both cell types (Wentworth et al. 2007).
Nanoparticles, especially iron oxide nanoparticles and quantum dots (QDs), are one of exciting materials for cell labeling, cell tracking and in vivo imaging, because of ease to synthesize in large quantities from various materials using relatively simple methods. The diameter of the nanoparticles can be tuned from several to a few hundred nanometers with controlled size distribution. Among them, QDs are considered as the ideal tool to label cells for tracking the cells, because of broad adsorption spectra, narrow emission spectra, high fluorescent intensity and long fluorescence lifetime (Solanki et al. 2008).
6.2.2 Scaffold for Tissue Regeneracy
As mentioned above, cell lost is a big problem for the cells injection method for tissue repair. It’s a really necessary requirement to make cells adhere to the surface of a scaffold which can prevent cell losing, support three-dimensional tissue formation. Furthermore, depending upon the setting, progenitor cells may need to mature into a tissue-specific phenotype, and fully differentiated cells will need to operate with appropriate functional responses (Chaikof et al. 2002).
Actually, the ECM, which is the natural environment for cells growing, is full of nanosized structure. Bone, as an example, is a nanocomposite that consists of a protein network (i.e., collagen, laminin, fibronectin, and vitronectin) and hard inorganic components (hydroxyapatite (HA), Ca10(PO4)6(OH)2) (Zhang and Webster 2009; Webster 2001). Specifically, 70 % of the bone matrix is composed of nanocrystalline HA which is typically 20–80 nm long and 2–5 nm thick (Simon 1994). Other protein components in the bone ECM same with the other tissues are also nanometer in dimension. This self-assembled nanostructured ECM in all tissues closely surrounds and affects these cell behaviors, for example, cell adhesion, proliferation and differentiation. Apparently, the design of novel nanomaterials which possess not only excellent mechanical properties but that are also biomimetic in terms of their nanostructure, has become quite popular in order to improve the functions of cells in regenerative medicine (Zhang and Webster 2009).
In 2004, Miller et al. (2004) reported endothelial and vascular smooth muscle cells adhesion and proliferation were enhanced comparing a nanostructured PLGA surfaces with smooth one. Later, a series of PLGA surfaces were fabricated by composing of submicron scale spheres on them. The results revealed that surfaces with 200 nm lateral diameter spherical features exhibited highest fibronectin and collagen type IV adsorption comparing the 100 nm or 500 nm lateral diameter spherical surface features. Furthermore, the higher fibronectin and collagen IV adsorption, the more endothelial and vascular smooth muscle cell adhesion was found as well (Miller et al. 2007). Since chemistry was similar between all PLGA surfaces investigated, this study provided strong evidence of the influence of nanometer features on optimizing fibronectin interactions and subsequently vascular cell adhesion. Similar results were also found that nanostructured titanium implant surfaces promote bone cell responses leading to accelerated calcium deposition improving integration with surrounding bone compared to conventional titanium surfaces (Ergun et al. 2008; Webster et al. 1999; Yao et al. 2008).
Collagen, the major ECM component of most of these tissues, has been proved as a substrate or scaffold for cell attachment, proliferation, and differentiation (Elsdale and Bard 1972; Strom and Michalopoulos 1982). Wang et al formed a gene activated collagen substrate via assembly of complexes of plasmid encoding SDF-1alpha into a collagen substrate to create a microenvironment favoring stem cell homing. In vivo experiments showed that local release of SDF-1alpha from the transfected cells on the gene activated collagen substrate could effectively home CD117(+) stem cell to the surrounding tissues(Wang et al. 2010). Moreover, the nanosized collagen fibrillar structure (50–500 nm in diameter) has been demonstrated to enhance cell/matrix interactions (Grinnell and Bennett 1982; Kuntz and Saltzman 1997). For serving as a scaffold for regenerative cells, the nanosized fibers like collagen may be help to improve the cell/scaffold interactions and to be a better environment for cell growing. Several techniques, for example, self-assembly, phase separation, and electrospinning are developed to fabricate porous scaffolds composed with nanosized fibers (Wei and Ma 2008).
For example, electrospun poly(L-lactide-co-ε-caprolactone) (PLCL) fibrous scaffolds of varying fiber diameters (ranging from 300 nm to 7 mm) were used as scaffolds for culturing human umbilical vein endothelial cells (HUVECs). A higher cell adhesion and proliferation potential was found cultured with nanosized PLCL scaffolds (Kwon et al. 2005). Ma’s group developed a thermally induced phase separation method to fabricate nanofibrous scaffold (Zhang and Ma 1999). With this method, nanofibrous poly(L-lactide) (PLLA) scaffolds with diameters ranging from 50 to 500 nm has been prepared using tetrahydrofuran (THF) as solvent (Zhang and Ma 1999, 2000). Nanofibrous scaffolds of PLLA prepared from this technique have demonstrated to adsorb/absorb cell adhesive proteins (fibronectin and vitronectin) 2–4 times higher and an almost twofold increased osteoblast attachment in comparison to solid walled PLLA scaffolds (Yang et al. 2004a).
So, an ideal 3D-scaffold for tissue regeneracy should have similarity to native ECM in terms of both chemical composition and physical nanostructure. Nanostructured biomaterials having physical features in the nanometer range, such as nanocrystals, nanofibers, nanosurfaces and nanocomposites, have gained much interest recently in regenerative medicine (Thomas et al. 2006a; Layrolle and Daculsi 2006).
Furthermore, except ECMs, intrinsic regulators (e.g., growth factors and signaling molecules) are another prime factors that have critical roles in regulating cell behaviors during the tissue reparation (Solanki et al. 2008; Kiritsy and Lynch 1993). For example, during the cutaneous wound repair process, the growth factors (platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), epidermal growth factor (EGF), and fibroblast growth factor (FGF) et. al) play important roles in all of three repair phases (hemostasis and inflammation, granulation tissue formation, and matrix formation and remodeling) (Kiritsy and Lynch 1993). However, direct injection of growth factors into a regeneracy site is generally not effective because of their rapid diffusion and short lifetime. To enable growth factors to efficiently exert their biological effects during the tissue regeneracy process, a drug delivery system should be used (Tabata 2003).
6.3 Technological and Biological Opportunities for Therapeutic Devices
6.3.1 Functionalized Nanoparticles for Cell Tracking
Over the past decade, cell tracking is becoming more and more important for optimizing cell delivery or accurate biodistribution studies in regenerative medicine as mentioned above.
Nanoparticles, because of the size-dependent properties and dimensional similarities to biomacromolecules, are suitable as contrast agents (Chan et al. 2002; Bruchez-Jr et al. 1998; Chan and Nie 1998; Dubertret et al. 2002; Jaiswal et al. 2002; Ishii et al. 2003; Dahan et al. 2003; Lidke et al. 2004) or probes for biomedical imaging (Josephson et al. 1999; Bulte et al. 2001).
Magnetic nanoparticles in magnetic resonance imaging (MRI) , QDs and other bioengineered nanoparticles are commonly used for cell labeling and tracking which provide several unique features and capabilities. Firstly, the size-dependent optical and electronic properties can be tuned continuously by changing the particles size (Alivisatos 1996). Secondly, nanoparticles have big specific surface which can be useful for surface modification in order to target a specific organ or tissue in human body (Rhyner et al. 2006). These nanoparticles are functionalized by diverse techniques in order to meet the requirements for cell imaging and cell tracking.
6.3.1.1 Magnetic Nanoparticles
Magnetic nanoparticles, especially superparamagnetic iron oxide particles (SPIO), have a variety of applications on molecular and cellular imaging for enhancing magnetic resonance contrast. The popularity of SPIO particle is mainly because of these following several properties: (1) they provide the most change in signal (albeit hypointensity) per unit of metal which can obtain sharp images with different iron concentration; (2) they are composed of biodegradable iron, which is biocompatible and can thus be reused/recycled by cells using normal biochemical pathways for iron metabolism; (3) they can be magnetically manipulated and change their magnetic properties according to size (Bulte and Kraitchman 2004).
Normally, SPIO consist of two components, an iron oxide core and a hydrophilic coating. Typically, the core is magnetite (Fe3O4) and/or maghemite (γ Fe2O3) which plays important role in the MRI. In some case, SPIO nanoparticles can be functionalized simply by doping some other metal ions during the preparation process. Groman et al. (2007) fabricated one new mixed ferrite colloidal magnetic iron oxides by adding informational atoms (Lanthanide) during formation of the iron oxides core. The new functionalized nanoparticles, not only can be visualized by iron-based MRI, but also can be quantized by neutron activation (Eu, Sm, La, Tb add) and even visualized histologically using time resolved fluorescence (Eu, Tb added).
The SPIO nanoparticles must be functionalized by hydrophilic coating in order to stabilize iron oxide crystals in aqueous colloidal solutions or in vivo, reduce unspecific protein adsorption or cell interactions in vitro or in vivo. Most commonly, surface molecules are biocompatible hydrophilic polymers, for example polysaccharides-dextran (Weissleder and Papisov 1992). A rich dextran density can also enhance circulation time because of the flexible dextran layer forming a “molecular brush” (Papisov et al. 1993) because of the low protein adsorption in plasma. As a fact, long circulation time is critically necessary for better targeting and tracking cells. For this purpose, other biological macromolecules have been investigated for functionalizing iron nanoparticles, e.g. poly(sialic acid), heparin etc., but because of their high cost, efforts have been directed to the design of synthetic hydrophilic macromolecules.
Among these synthetic macromolecules, block-copolymers such as poloxamers and poloxamines has been widely used for enhancing circulation time in vivo because of the effect from poly(ethylene glycol) (PEG) molecules extended in the solution (Moghimi and Hunter 2000). To achieve coupling PEG on iron oxide particles, associated dextran on particles was oxidized and poly-L-lysine (PLL) were attached to the surface by electrostatic force. Finally, methoxy(polyethylene glycol)-O-succinyl succinate was immobilized on PLL covered particles (Weissleder et al. 1995). Then, Butterworth et al. (2001) developed a new method for the grafting of PEG onto magnetite particles by the use of trimethoxysilane-PEG which is more convenient for controlling the grafting process. The PEG immobilized iron oxide particles produced showed greatly enhanced colloidal stability with respect to uncoated particles.
Targeting specific tissue, organ, or cells precisely are necessary for cellular imaging and tracking. Passive targeting is easier to achieve in application which can make nanoparticles accumulate in a specific tissue or organ (liver, spleen, cancer et al.) by simply controlling the particles size (Thorek et al. 2006). But passive targeting is not a universal method for targeting because of its limitation. In order to achieve active targeting of SPIO against specific tissue, organ or cells, it is necessary to first conjugate targeting agents onto the SPIO surface directly or onto its hydrophilic coating. In this case, reactive moieties (i.e., amines, sulfhydryls, carboxyls, etc.) are needed in order to immobilize targeting moieties (i.e., antibodies, folic acid, galactose etc.). Take dextran coated SPIO particles as an example, these hydroxyl groups on dextran molecules were oxidized by sodium metaperiodate (Weissleder et al. 1995), then further modification can be applied. Josephson et al. (1999) crosslinked dextran with epichlohydrin, then amination was used to induce amino groups on SPIO particles. Finally, particles were functionalized by tat peptide and fluorochrome for imaging cells (Josephson et al. 1999; Groman et al. 2007; Koch et al. 2003).
In order to improve specific interactions with certain kind of cell, targeting moieties, for example peptides, antibodies, small molecule (folic acid, galactose etc.) are normally immobilized on SPIO particles. HIV tat peptide, which contains a membrane translocating signal, was immobilized on surface for efficiently transporting the iron oxides into cells (Josephson et al. 1999; Koch et al. 2003; Lewin et al. 2000). Monoclonal antibodies (mABs) are the proteins which can only interact with a specific substance, and achieve precisely targeting for one kind of cell. Bulte et al. coupled mouse anti-transferrin receptor mAB OX-26 with magnetic nanoparticles and then magnetically labeled oligodendrocyte progenitors (Bulte et al. 1999) and neural precursor cells (Bulte et al. 2003) by receptor-mediated endocytosis for monitoring cell migration. Schellenberger et al. (2002, 2004) found that Annexin V conjugated nanoparticles could detect apoptotic cells at nanoparticle concentrations as low as 0.1 μg Fe/ml in vitro. Targeting moieties are immobilized with PEG as space, for reducing unspecific interactions and increasing specific interactions. Iron oxide nanoparticle surface was modified by folic acid (FA) with PEG as spacer successfully (Kohler et al. 2004; Sun et al. 2006). Then the cell uptake properties were obviously increased after FA immobilized on particles. The specific interactions was found with FA receptor overexpressed cell line—HeLa, but not with non FA receptor overexpressed cells—MG-63 (Sun et al. 2006).
Moreover, Fe2+ released from iron oxide nanoparticles may have potential toxic effects on the cells. In order to prevent Fe2+ toxic effects of SPIONs, the gold-coated shell was combined on the surface, which is well know as a stable metal. More importantly, gold has well-defined surface chemistry with thiol or amine moieties. This offers an attractive and convenient route for further functionalization of the SPIONs with biomolecules through thiol- or amine-coupling chemistry (Niemeyer and Ceyhan 2001).
Magnetic nanoparticles labeled cell not only make cell visible in the regenerative medicine, but also may be helpful for guiding cells into usable tissues for transplantation with the help of magnetic fields. Sasaki et al. (2008) fabricated novel magnetic nanoparticles coated with chitosan. When bound to fibroblasts and exposed to an external magnetic field, these magnetic nanoparticles improved cell seeding into the center of a 3D scaffold.
6.3.1.2 Quantum Dots
QDs are crystalline semiconductors typically less than 10 nm in diameter that have been studied for over 20 years. Recently, more and more applications are developed in biomedicine including regenerative medicine field (Bruchez-Jr et al. 1998; Chan and Nie 1998). In the past decades, several production methods are available, from photolithography to wet chemical synthesis. The QDs produced in colloidal solutions are the most useful for biomedical applications since high-quality nanocrystals can be prepared in large quantities at low costs (Rhyner et al. 2006).
QDs have unique optical and electronic properties comparing with organic dyes and fluorescent proteins because of higher molar extinction coefficients, emission wavelengths size tunable and long term photostability (Yu et al. 2003; Cui et al. 2007; Maysinger et al. 2007). These properties have made QDs a topic of intensive research in tracking cell migration, differentiation and metastasis (Rhyner et al. 2006).
The highest quality QDs are composed of II–VI, IV–VI or III–V semiconductors (Lemon and Crooks 2000; Rogach et al. 1999). The most common QD structure is a CdSe core functionalized with a thin shell of ZnS in order to reduce potential toxicity of core (Rhyner et al. 2006). No acute and obvious CdSe QD toxicity has been detected in studies of cell proliferation and viability in live cells (Jaiswal et al. 2002; Winter et al. 2001; Parak et al. 2002; Derfus et al. 2004) and animal models (Larson et al. 2003; Akerman et al. 2002). However, cytotoxicity was observed when Cd2+ was released by oxidization the CdSe in air or UV. This happened when the QD surface coating was not stable enough. But after larger molecules, such as proteins (e.g., streptavidin and bovine serum albumin) are used to functionalize on the surface, slower oxidation is found of the core (Alivisatos et al. 2005). Bioconjugation of QDs with biomolecules, such as arginine-glycine-aspartic acid, did not show any toxic effect on hMSCs as compared with unlabeled human umbilical vein endothelial cells (hMSCs) (Shah et al. 2007).
In general, surface modifications or functionalization must be taken place after the QDs are synthesized in order to transfer to an aqueous phase for medical applications. To accomplish this, the hydrophobic surface ligands can either be exchanged with bifunctional ligands or the entire QD can be coated with an amphiphilic polymer layer. In recent work, Gao and colleagues (2004) encapsulated luminescent QDs with a biocompatible copolymer and linked this amphiphilic polymer to tumor-targeting ligands. Using either subcutaneous injection of QD-tagged cells or systemic injection of multifunctional QD probes, sensitive and multicolor fluorescence imaging of cells can be achieved under in vivo conditions which may be quite useful for regenerative medicine applications (Rhyner et al. 2006).
Although optical imaging with QDs is highly sensitive, a limitation in depth is a major disadvantage. Other imaging techniques, such as MRI, are more suited for tomography and 3D imaging. By functionalizing on QDs, dual imaging can be achieved. For example, gadolinium, which is visible by MRI, was used to link on the surface of QDs by polymer conjugated lipids (Mulder et al. 2006). These functionalized QDs can be easily detected both by fluorescence imaging and MRI in vitro (Mulder et al. 2006). Furthermore, these Gd-based dual-modality nanoparticle probes are promising for in vivo apoptosis after immobilized Annexin A5 on the surface through a PEG spacer (van-Tilborg et al. 2006). The dual imaging nanoparticle probes can be achieved by linking QDs with Fe2O3 or FePt as well (Gu et al. 2004).
Normally, fluorescent QDs require excitation from external illumination sources to fluoresce, which limits their application for imaging living opaque subjects because of the resultant strong autofluorescence background and a paucity of excitation light at non-superficial locations. So et al. (2006) reported self-illuminating quantum dot conjugates designed by mimicking a natural bioluminescence resonance energy transfer (BRET) system, with a mutant of R. reniformis luciferase as the energy donor and quantum dots as the acceptor, and have demonstrated that BRET emission can be imaged in cells and small animals. These self-illuminating QD conjugates can emit long-wavelength (from red to near-infrared) bioluminescent light in living cells and in living animals, even in deep tissues, and can be applied for multiplex in vivo imaging (So et al. 2006).
6.3.1.3 Other Nanoparticles
Except wildly used magnetic nanoparticles and QDs, diverse nanoparticles made by either organic or inorganic are applied as cell tracking or imaging probes.
For the specific application, bioactive inorganic particles, for example hydroxyapatite (HA) particles, were functionalized by fluorescence molecules. Zaheer et al. (2001) synthesized a near-infrared (NIR) fluorescent bisphosphonate derivative that exhibits rapid and specific binding to hydroxyapatite (HA). They demonstrate NIR light–based detection of osteoblastic activity in the living animal, and discuss how this technology can be used to study skeletal development. Fluorescence imaging of osteoblastic activity in living animals has also met with success using an active probe: a tetrasulfonated heptamethine indocyanine conjugated to the hydroxyapatite-binding ligand pamidronate (Rao et al. 2007).
A large group of organic nanoparticles such as liposomes, dendrimers and polymersomes have not only been developed for drug delivery, but can also be applied to in vivo optical imaging. Therien and colleagues (Ghoroghchian et al. 2005) reported the synthesis of NIR-emissive polymersomes (polymer vesicles with a diameter of 50 nm to 50 mm) through the cooperative self-assembly of amphiphilic diblock copolymers and conjugated multi(porphyrin)-based NIR fluorophores. Dendrimers were previously used as carriers for magnetic resonance imaging (MRI) contrast reagents and, recently, McIntyre et al. (2004) designed a polyamidoamine dendrimer-based fluorogenic substrate to image tumorassociated matrix metalloproteinase-7 in vivo. A boronated dendrimer labeled with a vascular endothelial growth factor (VEGF) and an NIR dye Cy5 has been shown to selectively bind upregulated VEGF receptors in mouse breast carcinoma (Backer et al. 2005).
When multiple fluorescent dyes are attached to the same molecule, such as an antibody, the fluorescent intensity can decrease instead of increase owing to dye–dye quenching. However, when a viral capsid is used as the scaffold for labeling, more than 40 Cy5 dyes can be loaded onto a single virus particle via specific chemical coupling and no fluorescence quenching is observed due to the large intermolecular distances (Soto et al. 2006). This approach has resulted in the synthesis of highly fluorescent viral nanoparticles with a defined structure and a size of 30 nm in diameter. The local dye concentration was reported to be as high as 1.8 mM without significant quenching (Wu et al. 2005). Cowpea mosaic virus nanoparticles labeled with Alexa dyes have been used successfully to visualize the vasculature and blood flow and for imaging human fibrosarcoma-mediated tumor angiogenesis in living mouse and chick embryos (Lewis et al. 2006).
6.3.2 Functionalized Nanomaterials for Tissue Regeneracy
Besides multifunctionalized nanoparticles for better understanding tissue regenerative process through cell tracking and cell imaging, functionalized nanomaterials could become important for tissue regeneracy. They could be utilized directly for tissue regeneration or as cornerstone for artificial tissues. Li et al tried to guide magnetic nanoparticle polymer/DNA complexes after systemic administration to the heart with a magnetic field. They showed that an epicardial magnet could effectively attract the complexes in the left side of the thorax, resulting in strong reporter and therapeutic gene expression in the left lung and the heart (Li et al. 2008). Zhang et al conjugated magnetic nanoparticles with adenoviral vectors-encoded hVEGF gene. In a rat acute myocardial infarction (AMI) model, they injected magnetic nanoparticle/adenoviral vector (hVEGF) complexes intravenously. With magnetic targeting, the complexes significantly improved left ventricular function and exhibited higher capillary and arteriole density and lower collagen deposition in the infarcted area (Zhang et al. 2012).
The principles of the design of an ideal 3D scaffold for tissue engineering remain unclear. The scaffolds should mimic the structure, composition and biological functions of native extra cellular matrix (ECM) as much as possible. Moreover, most of the scaffold fabrication strategies have not given importance to mimic the nanoscale physical features of the natural ECM. It is well known that cells and proteins interact at the nanoscale (Thomas et al. 2006a; Cao 2008). For instance, researchers have engineered a variety of scaffolds made from nanotubes, nanofibers, and nano composites that can be used to grow lifelike networks of cells from the liver, bladder, kidney, bones and cardiovascular system. These artificial tissues could be developed into new therapies for patients with diseased or damaged organs.
It is still early, but many laboratories are experimenting with a wide variety of nanomaterial scaffolds that can be infused with cells to form artificial tissues, such as bone and liver. It appears possible to repair damaged nerves by injecting them with nanomaterials that form bridge-like lattices. Other nanostructures show promise as foundations for growing three-dimensional networks of blood vessels.
Considerable efforts have been made to develop ideal scaffolds for tissue engineering so far. Various techniques such as solvent casting/particulate leaching (Mikos et al. 1994), gas foaming (Mooney et al. 1996; Nam et al. 2000), and phase separation/emulsification (Nam and Park 1999a, b) have been employed to fabricate conventional porous polymeric foams. Peptide self-assembling, phase separation, and electrospinning are normally techniques used for fabricating nanostructured scaffold materials.
6.3.2.1 Self-Assembly
Self-assembly is a process in which molecules and supramolecular aggregates organize and arrange themselves into an ordered structure through weak and non-covalent bonds (Murugan and Ramakrishna 2007; Whitesides and Grzybowski 2002). It is a common process in nature, for example, collagen has a triple helix secondary structure, which consists of three polypeptide chains in an extended left handed helix (Ramachandran 1988). Self-assembly could be used to produce natural or synthetic polymers into nanoscale structures including nanofibers (Zhang 2003; Hartgerink et al. 2001; Chiti et al. 2003), especially scaffolds based on peptides and proteins. The biological ECMs made by this technique are able to interact with cells at the molecular level to control the processes of tissue regeneracy effectively.
Several studies report promising results of this strategy. For example, a peptide amphiphile (chemical compound possessing both hydrophilic and hydrophobic properties) nanofiber network could be mineralized with hydroxyapatite to recreate the nanoscale structure of bone (Hartgerink et al. 2001). Certain peptide amphiphiles can be designed in order to get functionalized nanomaterials for specific applications. For example, these amphiphile nanofibers have been designed to mimic the collagen structure-building protein-like structural motifs that incorporate sequences of biological interest (Fields et al. 1998; Yu et al. 1998, 1999; Berndt et al. 1995). These nanofibers have been also applied to promote rapid and selective differentiation of neural progenitor cells into neurons (Silva et al. 2004). Self-assembly was also used successfully to encapsulate chondrocytes within a self-assembled peptide hydrogel scaffold for cartilage repair (Kisiday et al. 2002; Engel et al. 2008). Self-assembly of PAs can be promoted by various factors such as pH change, presence of Ca2+ ions, and drying on surface. Hong et al. (2003) developed another kind of peptide containing 16 alternating hydrophobic and hydrophilic amino acids and studies the effect of amino acid sequence and pH on self-assembly into nanofibers (Thomas et al. 2006a).
6.3.2.2 Phase Separation
Phase separation techniques have been used to prepare porous polymer membranes for purification and separation purposes. In last two decades, it is becoming a frequently used and convenient method to prepare porous tissue regenerative scaffolds. A variety of biodegradable polymers have been fabricated into three-dimensional porous scaffolds using phase separation techniques (Zhang and Ma 1999; Ma et al. 2003; Gong et al. 2006). In order to meet the requirement of nanoscaled scaffold for tissue regenerative process, a novel phase separation technique has been developed to generate nanofibrous structures by manipulating the phase separation process (Zhang and Ma 2000; Chen and Ma 2006). The poly(L-lactic acid) (PLLA) fibrous scaffold contains nanofibers ranging from 50 to 500 nm in diameter (Chen and Ma 2006), which is similar to natural collagen fibers in size (Elsdale and Bard 1972; Hay 1991). Nanofibrous scaffolds of PLLA prepared from this technique have demonstrated to adsorb/absorb cell adhesive proteins (fibronectin and vitronectin) 2–4 times higher and an almost twofold increased osteoblast attachment in comparison to solid walled PLLA scaffolds (Zhang and Ma 2000). Due to the substantial surface area difference, degradation is much more rapid in such nanofibrous scaffolds, in which the overall mass loss is 51 % while mass loss in solid-walled nonfibrous foams is only 6 % after 15 months (Chen and Ma 2006).
One limitation of the early nanofibrous materials generated using the phase-separation technique is the lack of interconnected macropores, which are critical for cell seeding and recruiting, mass transfer, vascularization, and tissue organization. To overcome this problem, phase separation techniques are used in combination with other scaffold fabrication techniques such as porogen leaching. The combined technique provides broader control over porous architectures from macro-, micro- to nanoscales (Chen and Ma 2004; Wei and Ma 2006; Zhou et al. 2005; Gong et al. 2008; Ma et al. 2005a). Gong et al. (Zhou et al. 2005; Gong et al. 2008) fabricated well connected PLLA scaffolds via porogen leaching with phase separation technique in which gelatin particles was used as porogens. The biological performance of the scaffold was evaluated by in vitro chondrocyte culture and in vivo implantation. In comparison with the control scaffold fabricated with NaC1 particles as porogen under the same conditions, the experimental scaffold had better biological performance because the gelatin molecules were stably entrapped onto the pore surfaces (Gong et al. 2008). Surface modification was also taken place in order to improve the biocompatibility of these PLLA scaffolds. Ma et al. (2005a) immobilized collagen and introduced basic fibroblast growth factor (bFGF) on PLLA scaffold. Chondrocyte culturing on the collagen immobilized PLLA surfaces showed significantly improved cell spreading and growth. Incorporation of fibroblast growth factors in the collagen layer further enhanced the cell growth (Ma et al. 2005a).
6.3.2.3 Electrospinning
Electrospinning, as another method to produce nanoscale fibers, is a simple and cost-effective fabrication process that uses an electric field to control the deposition of polymer fibers onto a target substrate (Engel et al. 2008). The generated fibers can mimic the structural profile of the proteins found in the native ECM. The use of electrospinning process in biomaterials field was first reported by Martin and Cockshott (1977) as early as 1977. Since then, electrospinning process has been continuously investigated for the fabrication of nanofibrous matrices for divers applications (Reneker and Chun 1996; Li et al. 2002; J-s and Reneker 1999; Fong et al. 1999; Yoshimoto et al. 2003; Ma et al. 2005b; Chiu et al. 2005). Various synthetic polymer, PLA (Zong et al. 2002; Zeng et al. 2003), PLGA (Li et al. 2002), PCL (Silva et al. 2004; Yoshimoto et al. 2003), poly(dioxanone) (PDS) (Boland et al. 2005) and synthetic polypeptide (Huang et al. 2000), natural proteins such as collagen (Matthews et al. 2002; Huang et al. 2001; Matthews et al. 2003), silk protein (Li et al. 2005), elastin fibrinogen (Wnek et al. 2003) etc, are used for fabricating biodegradable scaffold. Electrospun nanofibers have been shown to support cell attachment and proliferation of a variety of cells as they have large surface area and well-interconnectivity of inter-fiber spaces, in addition to the nano sized diameters mimicking the physical nanoscaled dimensions of native ECM (Thomas et al. 2006a).
Biomolecules such as growth factors, drugs, and genes can be directly mixed into the polymer solution and electrospun to prepare functionalized polymer nanofibers. These functionalized bioactive nanofibers have potential applications in both tissue regeneracy and drug delivery systems. Co-spinning of growth factors for cells in future may enable to fabricate scaffolds with controlled release of cellular nutrients. Luu et al. (2003) and Liang et al. (Ye and Huang 2005) have encapsulated plasmid DNA in PLA-PEG and co-electrospun with PLGA in DMF and electrospun the mixture into nanofibers. Release of plasmid DNA from the scaffolds was studied for 20 days and found that the release of DNA sustained over 20 day period with a maximum release occurring at 2 h. Verreck et al. (2003) prepared polyurethane nanofibers containing model drugs itraconazole and ketanserin to study the pattern of drug release. Co-spinning of growth factors for cells in future may enable to fabricate scaffolds with controlled release of cellular nutrients (Thomas et al. 2006a).
Bioactive nanoscale fillers, e.g. hydroxyapatite (HA), tricalcium phosphate (TCP) et al., are incorporated into polymer solution to electrospun nanocomposite nanofibers for better interactions with cells. Thomas et al. (2006b) examined the physical property changes after nanoHA incorporated into PCL nanofibers. They demonstrated that it is possible to tailor subtle mechanical properties in a nanofibrous matrix by incorporating nanofillers of desired amount. Higher percentage loadings of nanoHA resulted in poor dispersion of the nanoHA powder as particle size of nanoHA used was ~100 nm. If the particle size of HA is small enough (~20–40 nm), PCL/nanoHA composite with more than 20-wt% produces fibers with well dispersed nanoHA (Thomas et al. 2006b). It has been reported that chondrocyte adhesion and proliferation on polymer/nanoHA composite materials are better than the pure polymer (Hong et al. 2005). MSCs seeded onto nanocomposite scaffolds exhibited well cell spreading and growth on PCL/nanoHA nanocomposites, revealing favorable cell-matrix interactions (Thomas et al. 2006a).
As mentioned earlier, electrospinning of collagen into nanofibers have opened the door to make nanofibrous matrices mimicking nano structures of bone for bone tissue engineering. However an ideal scaffold for bone tissue engineering should mimic not only the nanofibrous physical structure but also the chemical composition. Electrospun nanofibrous nanobiocomposite scaffolds based on Type I collagen and nanoHA have been prepared as biologically inspired scaffolds mimicking the chemical and morphological features of natural ECM (Thomas et al. 2007).
Very recently, Badami et al. (2006) have electrospun PLA as well as PEG-PLA di-block copolymers of PEG-PLA into fibers with diameters ranging from 140 nm-2.1 mm and cultured MC3T3-E1 mouse calvaria-derived osteoprogenitor cells on the scaffolds up to 14 days. The results of study focussed on the effect of fiber diameter on spreading, proliferation and differentiation of osteoblastic cells on fibrous scaffolds with and without osteogenic factors The authors concluded that in the absence of osteogenic factors such as β-glycerophosphate and Lascorbat-2-phosphate, cell growth (cell density) was lower on polymer fibers than smooth polymer surfaces, while in the presence of osteogenic factors cell density on fibers was equal or greater than that on smooth surfaces (Thomas et al. 2006a).
Venugopal et al. (2005) have coated collagen over electrospun PCL by soaking the PCL matrix in collagen solution (10 mg/mL) and cultured human coronary artery smooth muscle cells. It was observed that SMCs migrated towards inside the nanofibrous matrices and formed smooth muscle tissue in 72 h. According to the authors, PCL scaffold supporting the cell growth needs collagen support for migration of cells inside the nanofibrous matrices. In another study He et al. (2005) showed that collagen coated PLLA-CL nanofibers exhibited enhanced cell attachment, spreading and viability of human coronary artery endothelial cells. It was found that coating of collagen on PCL scaffold definitely favored cell proliferation.
The co-use of these adhesion proteins and biodegradable synthetic polymers enables the construction of cell-adhesive scaffolds for vitally functioning engineered tissues (Kwon and Matsuda 2005; Kwon et al. 2001; Chen et al. 2000; Almany and Seliktar 2005; Zhang et al. 2005a). Co-electrospinning is a feasible approach to provide a compromise solution for overcoming the shortcomings of synthetic and natural polymers that is producing new porous nanofibrous biomaterials with good biocompatibility and improved mechanical, physical and chemical properties and biological performance.
Stitzel et al. (2006) have recently fabricated a vascular graft scaffold from electrospun polymer blends of Type I collagen (45 wt%), elastin (15 wt%) and PLGA (45 wt%). They found that by controlling the compositional ratio of collagen, elastin, and PLGA have resulted in improved electrospun fiber characteristics and physical strength of the vascular graft.
Core-shell types of multi component nanofibers by coaxial electrospinning are of another interesting mixed polymer system in tissue engineering for bioactive scaffolds. Functionalization of fibers without affect the core is desirable in tissue engineering and in controlled drug delivery for preserving an unstable biological agent from an aggressive environment and delivering a biomolecular drug in a sustained way. Co-axial electrospinning is a method for incorporation of water-soluble macromolecules as the core of nanofibers during electrospinning. The production of coreshell nanofibers from co axial electrospinning was first demonstrated by Sun et al. (2003). Zhang et al. (2004) fabricated bi-component nanofibers of PCL and gelatin in the form of a core-shell structure by coaxial electrospinning. A quantitative analysis of the effect of gelatin concentration on the diameters of core and shell of nanofibers was carried out that when the concentration of gelatin was below 12.5 w/v % the diameter of core and shell were, respectively, less than 200 nm and 400 nm. Zhang et al. (2005b) have fabricated collagen-PCL nanofibrous scaffold (collagen-r-PCL) by coaxial electrospinning and compared the surface biocompatibility with electrospun neat PCL, electrospun neat collagen scaffold and collagen coated PCL scaffolds by culturing human dermal fibroblasts. As compared to neat PCL scaffold and collagen coated PCL, scaffold human dermal fibroblasts cell density on collagen-r-PCL linearly increased over period. However, cell proliferations data of collagen-r-PCL are not significantly differ from those of electrospun neat collagen. In another study Jiang et al. (2005) fabricated biodegradable core-shell nanofibers with PCL as shell and protein containing PEG as core for controlled release of incorporated proteins such as lysozyme and BSA.
Recently, a group based in Singapore developed an alternative approach to wound healing, which they termed autologous layered dermal reconstitution (ALDR) (Chong et al. 2007). This technique relies upon novel TE scaffolds which consist of electrospun fibers made of PCL and gelatin, between 300 and 600 nm in diameter, with a total thickness of only 28 μm. The scaffolds were seeded with human dermal fibroblasts, which remained viable in the scaffold for all time points tested (up to 2 weeks) and doubled in population approximately every 3 days. Although no in vivo results are currently available, ALDR using electrospun scaffolds should offer a distinct advantage over traditional techniques. Namely, ALDR will allow for a rapid, layer-by-layer buildup of tissue in deep wounds, with dermal fibroblasts distributed throughout. This can occur because the electrospinning process takes place on top of a commercially available polyurethane wound dressing. As little as 48–72 h after implantation, the wound dressing can be removed, and another scaffold/wound dressing construct placed in the wound site. This is repeated until the wound area is fully repaired. Since each scaffold will be individually seeded with dermal fibroblasts prior to implantation, this layer by layer technique eliminates the long in vitro culture times otherwise needed for cellular infiltration and growth within larger, single-layer scaffolds. The end result is a continuous layer of tissue, wherein the use of a porous, nanostructured scaffolds allows for rapid cellular proliferation and integration between layers (Khang et al. 2010).
6.3.2.4 Nanocomposite Scaffold
Nanocomposite scaffold are made of regenerative scaffold with certain nanostructure system, for example, bioactive molecules and particles. Nanocomposites can be reinforced polymers or ceramics with low quantities of nanometric-sized particles (silicate, carbon nanotubes (CNT)) which give them improved properties. The properties of nano-composite materials depend not only on the properties of their individual parents but also on their morphology and interfacial characteristics. This rapidly expanding field is generating many exciting new materials with novel properties. Nanocomposites have attracted a great deal of attention in biomedical applications also. Many natural tissues such as bone possess a composite micro/nano structure. These complex composite structures play roles for the physical and biological properties of the tissues. To mimic the natural tissue structure, biomedical polymers, bioceramics and other organic/inorganic materials are to be combined for superior properties. Composite materials often show an excellent balance between strength and toughness and usually improved characteristics compared to their separate components. Recently, Kothapalli et al. have shown that by incorporation of 50 wt% nanoHA into PLA scaffold, the yield strength increased 150 % and compression modulus almost doubled in comparison to pure PLA. Addition of nanoHA can improve osteoconductivity to the polymer scaffolds (Kothapalli et al. 2005).
One of the most interested nanocomposite biomaterials for bone regeneracy is comprised of biodegradable polymers with nanoHA or other calcium phosphate bioceramic composition. Experiments prove that micro/nanometer features on biomaterial surfaces can be used to guide cell behavior along a desired biological response (Webster et al. 1999; Liu and Webster 2007). Xu et al. found that structural signals from microstructured substrates comprising arrays of square-shaped or round-shaped microwells could influence the migration, proliferation, and osteogenesis of the stem cells on the substrate. The results open a window to control stem cell functions by the combination of structured microwells with the manipulation of cellular signaling (Xu et al. 2014). In bone-regeneracy applications, promising results have been obtained with the nanophase materials ceramics and metals, with which increased osteoblast adhesion, proliferation and calcium deposition have been observed compared with conventional materials (i.e. with micrometer-scaled grains) (Webster and Ejiofor 2004).
Biologically inspired nanobiocomposites of collagen and nanoHA for bone substitute have a long history in biomedical field (Clarke et al. 1993; Rovira et al. 1993; TenHuisen et al. 1995; Itoh et al. 2001). There is possibility of enhancing the functionalities of collagen by incorporating other bone materials such as HA, bone morphogenic proteins (BMP) etc. A combination of collagen and nanoHA materials is bioactive, osteoconductive and osteoinductive and seems to be a natural choice for bone grafting. I.e., it mimics the bone components. The unique characteristics of this biocomposite is the spatial orientation between HA and collagen macromolecules, which seems to be the source of the mechanical strength of the composite. Conventionally, collagen/HA nanocomposites can be made by blending or mixing the collagen and HA or by biomimetic methods (Du et al. 1999; Tampieri et al. 2003; Liao et al. 2004; Itoh et al. 2004; Yang et al. 2004b). However most of the collagen/HA composites are conventionally processed by anchoring microHA particles into the matrix of collagen, which makes it quite difficult to obtain a uniform to a homogeneous composite graft. Further, large size crystalline microHA, which is in contrast to natural bone apatite, may take a longer time to remodel into bone tissue up on implantation. In addition some of the composites exhibit very poor mechanical properties, probably due to the lack of strong interfacial bonding between constituents. There is a chance for improving osteointegration by reducing the grain size HA particles by activating the nucleation of ultra fine apatite growth into the matrix. This may lead to enhance mechanical properties and osteointegration with improved biological and biochemical affinity to the host bone (Thomas et al. 2006a).
Nanoparticle within regenerative medicine has been addressed mainly towards the development of entrapment and delivery systems for genetic material, biomolecules, such as growth and differentiation factors, and bone morphogenetic proteins and also as reinforcing- or bioactivity-enhancement phase for polymeric matrices in 3D scaffolds for tissue regeneracy (Engel et al. 2008).
Controlled delivery of biomolecules is crucial in the support and enhancement of tissue growth in tissue regeneracy applications. Nanotechnology approaches in delivery systems can enhance the success of specific therapeutic agents, such as growth factors and DNA among others, which are of paramount importance for tissue regeneracy (Reddy et al. 2006). Carriers in the nanoscale enable the intracellular delivery of molecules and the possibility of reaching targets that are inaccessible normally, such as the blood–brain barrier, tight junctions and capillaries, whereas the control over biomolecule dosage and delivery period are increased. The ultimate challenge is to develop artificial nanocarriers that can target cells with efficiency and specificity similar to that of viruses (Mastrobattista et al. 2006).
Examples of nanoparticles for delivery systems include currently microspheres, microcapsules, liposomes, micelles and also dendrimers. The different types of nanoparticles have been developed as solid, hollow or porous. The most common development methods are molecular self-assembly, nanomanipulation, bioaggregation and photochemical patterning (Allemann et al. 1993; Cade et al. 2004).
Biodegradable polymers are the most commonly used materials in drug delivery. Polylactic acid (PLA), polyglycolic acid (PGA), polyethylene glycol (PEG) and its copolymers have been used widely in combination with hydrogels to attain nanocarriers that exhibit different release properties. Particularly important for the development of nanoparticles for delivery purposes are ‘smart’ or ‘stimuli-responsive’ polymers that can undergo conformational changes, such as swelling or shrinkage, on variations in temperature, pH and magnetic field (Engel et al. 2008). Neffe et al. formed a 3D architectured hydrogel consisting of gelatin and lysine connected by urea junction units. The 3D architectured hydrogel could provide growing pores during the degradation, hence supporting cell adhesion and providing tailorable microscopic and macroscopic elastic properties of environment for cells. It could effectively induce bone regeneration without requiring addition of cells or growth factors (Neffe et al. 2015).
In sophisticated tissue-engineering strategies, the biodegradable scaffold is preferred to serve as both a 3D substrate and a growth factor delivery vehicle to promote cellular activity and enhance tissue neogenesis (Jain et al. 2008). A novel approach has been described for fabrication of tissue-engineering scaffolds capable of controlled growth factor delivery whereby growth factor containing microspheres are incorporated into 3D scaffolds with good mechanical properties, well-interconnected macroporous and nanofibrous structures (Wei et al. 2006).
Incorporation of microspheres into scaffolds significantly reduced the initial burst release. Sustained release from several days to months was achieved through different microspheres in scaffolds. Released platelet derived growth factor (PDGF) was demonstrated to possess biological activity as evidenced by stimulation of human gingival fibroblast DNA synthesis in vitro. The successful generation of 3D nanofibrous scaffold incorporating controlled-release factors indicates significant potential for more complex tissue regeneracy (Jain et al. 2008). Growth factors are able to be incorporated on regenerative scaffold by other techniques, e.g. layer-by-layer self assembly. Collagen scaffolds functionalized with acid fibroblast growth factor (aFGF) or basic fibroblast growth factor (bFGF) via assembly with heparin/PEI or chondroitin sulfate (Mao et al. 2005; Ma et al. 2007). The results prove that both aFGF and bFGF can be successfully deposited onto the scaffold. The FGFs in the multilayers obviously enhances fibroblast proliferation and viability (Mao et al. 2005; Ma et al. 2007).
We show here that the bioactive aFGF has been successfully deposited onto the TCPS sheet surface in the presence of heparin via a layer-by-layer manner. The aFGF built in the multilayers obviously enhances fibroblast proliferation and viability.
For the skin tissue engineering, Chung et al. (2006) explored the use of poly(ε-caprolactone) (PCL) grafted with nanostructured chitosan (CS) as a regenerative scaffold for the growth of human dermal fibroblasts. Resultant nano-CS/PCL surfaces exhibited significantly higher surface roughness values as compared to smooth CS/PCL surfaces: 106.0 nm compared to 3.6 nm, respectively. Furthermore, these nano-CS/PCL constructs exhibited significantly (p < 0.001) higher rates of fibroblast proliferation and viability as compared to smooth CS/PCL surfaces or nano-rough PCL surfaces. As such, the technique of solvent spin-etching for polymers may represent an inexpensive means to prepare nanoscale TE scaffolds as improved artificial skin grafts (Khang et al. 2010).
6.4 Applications for Therapeutic Devices
After several decades development, dextran and other polymer-coated SPIONs are currently used in a number of biomedical applications; for example, Endorem® (Geurbet, France) is a commercially available contrast agent based on SPIONs surface coated with dextran (Corot et al. 2006). It is a suitable contrast agent for labeling human MSCs (hMSCs) and human ESCs (hESCs) as it does not need a transfection agent (which may damage the stem cells) to facilitate its cellular uptake. Feridex ® and Sinerem® are other commercially available dextran-coated SPIONs that are combined with commercially available transfection agents, such as Fungene™, Superfect™ or Lipofectamine (Bulte and Kraitchman 2004; Corot et al. 2006). The use of transfection agents at higher concentrations may increase toxicity and, at lower concentrations, may not lead to sufficient cellular uptake (Bulte and Kraitchman 2004). Thus, the amount of transfection agent needed to enhance internalization is optimized carefully before combining it with SPIONs. The amount also depends on the stem cell type to be labeled.
With the further development and investigation, more and more products will be commercially available.
6.5 Barriers to Practice and Prospects
Although research on nanoparticles for non-invasive detecting is developing continually, there are still a lot of barriers which should be overcome.
Every technique and or application has it limitations, and the use of iron oxides or quantum dots for molecular and cellular imaging is no exception. (1) Resolution of MRI and fluorescence imaging is not good enough. For better understanding of cell behaviors, high resolution is required to investigate one single cell’s migration, proliferation, and differentiation. (2) Better targeting and lower dose for imaging. Highly specific targeting is necessary for labeling interested cells only in order to reduce dose and get higher resolution. (3) for cellular imaging, as labeling is not permanent and self-replicable like reporter genes, with dilution of label upon cell division, iron oxide detection may rapidly become impossible, both in vitro (Bulte et al. 2001; Schaffer et al. 1993) and in vivo. (4) Finally, careful iron oxide titration and cellular differentiation studies need to be performed, as labeling may lead to inhibition of differentiation into certain cell types, without affecting cell viability or proliferation (Kostura et al. 2004).
For the scaffold for tissue regeneracy, they have not been used extensively but major contributions are expected in two areas. The first is growth of complex tissue, where microfluidic structures ensure a steady blood supply, thereby circumventing the well-known problem of providing larger tissue structures with a continuous flow of oxygen as well as nutrition and removal of waste products. The second, and probably more important function of microfluidics, combined with micro/nanotechnology, lies in the development of in vitro physiological systems for studying fundamental biological phenomena (Jain et al. 2008).
6.6 Conclusions and Future Challenges
Nanomaterials are considered as a new class of materials possessing superior properties over its microscale counterparts. Nanostructured biomaterials having physical nanofeatures such as nanocrystals, nanofibers nanosurfaces, nanocomposites, etc. have gained much interest in regenerative medicine.
The coregistration of in vivo fluorescence imaging with anatomical imaging modalities such as MRI helps traverse the shortcomings of fluorescence imaging, such as limited tissue penetration of photons and low three-dimensional spatial resolution, and provides complementary information. The development of multifunctional probes is attracting increasing attention and several studies have already appeared - from iron-oxide- and dendrimer-based dual MRI–fluorescence imaging contrast agents.
Effective and innovative imaging approaches are in great demand as new proteins and genes, particularly within the field of oncology, are being discovered at an ever-increasing pace. This provides a constantly multiplying library of molecules and pathways to be studied for prevention, diagnosis, and treatment of diseases. The full potential of new discoveries is however limited by the void between the advances in bioscience and the means to accurately, effectively and—critically—non-invasively image the molecular interactions in biological systems. Many challenges clearly remain in the pursuit of ideal SPIO probes for molecular imaging; increased target affinity, less complex conjugation schemes, reduction of cost, a means for MRS to avoid sequestration in lysosomes and more effective activatable probes. With persistent advances, this system continues to demonstrate its potential as a means to probe deeper into our biological universe.
Nanostructured scaffolds are interested in regenerative medicine, mainly because of their resemblance of nanomorphology and physical nanofeatures to natural extra cellular matrices. The nanoscaled features such as surface roughness and topography of nanocrystalline biocermics and nanofibrous scaffolds promote the cell behavior such as adhesion, proliferation and migration and differentiated functions. Polymeric nanofiber based nonwoven matrix is among the most promising nanostructured biomaterials for native ECM analogs. Electrospinning is a versatile technique to fabricate nanofibrous matrices of polymers for tissue engineering scaffold applications. One of the particular advantages of electrospinning in regenerative medicine is the ability to co-spin various components such as cell adhesive proteins and other cell-growth factors along with biodegradable synthetic or biopolymers. An ideal 3Dscaffold for tissue engineering should have similarity to native ECM in terms of both chemistry and physical nanostructure. Electrostatic co-spinning of nanocomposite fibers of polymers with nanoHA to fabricate hybrid scaffolds of improved mechanical properties and cellular behaviors has been established in our group. The unique characteristics of collagen/nanoHA composite system in native bone is the special orientation between HA and collagen molecules. Therefore, future efforts in nanofibrous collagen/nanoHA composite are required mimicking exactly the complex nano structured architecture of collagen matrix with the c-axis orientation of nanoHA particles (Thomas et al. 2006a).
Regenerative medicine aspects that focus on TE have evolved into two main strategies. The first strategy consists of an elegant approach in which stem cells harvested from the patient are expanded and seeded on 3D scaffolds within a bioreactor. The resulting hybrid construct is then implanted into the patient (together with growth factors) as a tissue matrix. However, the need to harvest and expand stem cells poses great efficacy and efficiency problems that define the success of the entire process. The second strategy relies on the development of intelligent materials that would be able to send signals to the stem cells already present in the diseased or damaged tissue niches that would then trigger the regeneracy process. Nanotechnology is a powerful tool for creating these ‘smart’ materials. This approach is challenging and is still far from being achieved. Among other advantages, it would raise the possibility to have such cell-free materials ready ‘off the shelf’ and to be able to use them as and when required (Engel et al. 2008).
References
Akerman ME, Chan WC, Laakkonen P et al (2002) Nanocrystal targeting in vivo. Proc Natl Acad Sci 99:12617–12621
Alivisatos AP (1996) Semiconductor clusters, nanocrystals, and quantum dots. Science 271:933–937
Alivisatos AP, Gu W, Larabell C (2005) Quantum dots as cellular probes. Annu Rev Biomed Eng 7:55–76
Allemann E, Gurny R, Doelker E (1993) Drug-loaded nanoparticles: preparation methods and drug targeting issues. Eur J Pharm Biopharm 39:173–191
Almany L, Seliktar D (2005) Biosynthetic hydrogel scaffolds made from fibrinogen and polyethylene glycol for 3D cell cultures. Biomaterials 26:2467–2477
Backer MV, Gaynutdinov TI, Patel V et al (2005) Vascular endothelial growth factor selectively targets boronated dendrimers to tumor vasculature. Mol Cancer Ther 4:1423–1429
Badami AS, Kreke MR, Thompson MS et al (2006) Effect of fiber diameter on spreading, proliferation, and differentiation of osteoblastic cells on electrospun poly(lactic acid) substrates. Biomaterials 27:596–606
Berndt P, Fields GB, Tirrell M (1995) Synthetic lipidation of peptides and amino acids: monolayer structure and properties. J Am Chem Soc 117:9515–9522
Boland ED, Coleman BD, Barnes CP et al (2005) Electrospinning polydioxanone for biomedical applications. Acta Biomater 1:115–123
Bruchez-Jr M, Moronne M, Gin P et al (1998) Semiconductor nanocrystals as fluorescent biological labels. Science 281:2013–2016
Bulte JWM, Kraitchman DL (2004) Iron oxide MR contrast agents for molecular and cellular imaging. NMR Biomed 17:484–499
Bulte JWM, Zhang S-C, Gelderen PV et al (1999) Neurotransplantation of magnetically labeled oligodendrocyte progenitors: Magnetic resonance tracking of cell migration and myelination. Proc Natl Acad Sci 96:15256–15261
Bulte JW, Douglas T, Witwer B (2001) Magnetodendrimers allow endosomal magnetic labeling and in vivo tracking of stem cells. Nat Biotechnol 19:1141–1147
Bulte JWM, Ben-Hur T, Miller BR et al (2003) MR microscopy of magnetically labeled neurospheres transplanted into the Lewis EAE rat brain. Magn Reson Med 50:201–205
Butterworth MD, Illum L, Davis SS (2001) Preparation of ultrafine silica- and PEG-coated magnetite particles. Colloids Surf A Physicochem Eng Asp 179:93–102
Buzea C, Blandino IIP, Robbie K (2007) Nanomaterials and nanoparticles: sources and toxicity. Biointerphases 2:17–71
Cade D, Ramus E, Rinaudo M et al (2004) Tailoring of bioresorbable polymers for elaboration of sugar-functionalized nanoparticles. Biomacromolecules 5:922–927
Cao YC (2008) Nanomaterials for biomedical applications. Nanomedicine 3:467–469
Chaikof EL, Matthew H, Kohn J et al (2002) Biomaterials and scaffolds in reparative medicine. Ann N Y Acad Sci 961:96–105
Chan WCW, Nie S (1998) Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281:2016–2018
Chan WCW, Maxwell DJ, Gao X et al (2002) Luminescent quantum dots for multiplexed biological detection and imaging. Curr Opin Biotechnol 13:40–46
Chen VJ, Ma PX (2004) Nano-fibrous poly(L-lactic acid) scaffolds with interconnected spherical macropores. Biomaterials 25:2065–2073
Chen VJ, Ma PX (2006) The effect of surface area on the degradation rate of nano-fibrous poly(l-lactic acid) foams. Biomaterials 27:3708–3715
Chen G, Ushida T, Tateishi T (2000) Hybrid biomaterials for tissue engineering: a preparative method for PLA or PLGA-collagen hybrid sponges. Adv Mater 12:455–457
Chiti F, Stefani M, Taddei N et al (2003) Rationalization of the effects of mutations on peptide and protein aggregation rates. Nature 424:805–808
Chiu JB, Luu YK, Fang D et al (2005) Electrospun nanofibrous scaffolds for biomedical applications. J Biomed Nanotechnol 1:115–132
Chong EJ, Phan TT, Lim IJ et al (2007) Evaluation of electrospun PCL/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater 3:321–330
Chung T-W, Wang Y-Z, Huang Y-Y et al (2006) Poly (ε-caprolactone) grafted with nano-structured chitosan enhances growth of human dermal fibroblasts. Artif Organs 30:35–41
Clarke KI, Graves SE, Wong ATC et al (1993) Investigation into the formation and mechanical properties of a bioactive material based on collagen and calcium phosphate. J Mater Sci Mater Med 4:107–110
Corot C, Robert P, Idée J-M et al (2006) Recent advances in iron oxide nanocrystal technology for medical imaging. Adv Drug Deliv Rev 58:1471–1504
Cui B, Wu C, Chen L et al (2007) One at a time, live tracking of NGF axonal transport using quantum dots. Proc Natl Acad Sci 104:13666–13671
Daar AS, Greenwood HL (2007) A proposed definition of regenerative medicine. J Tissue Eng Regener Med 1:179–184
Dahan M, Lévi S, Luccardini C et al (2003) Diffusion dynamics of glycine receptors revealed by single-quantum dot tracking. Science 302:442–445
Derfus AM, Chan WCW, Bhatia SN (2004) Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 4:11–18
Du C, Cui FZ, Zhu XD et al (1999) Three-dimensional nano-HAp/collagen matrix loading with osteogenic cells in organ culture. J Biomed Mater Res Part A 44:407–415
Dubertret B, Skourides P, Norris DJ et al (2002) In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science 298:1759–1762
Elsdale T, Bard J (1972) Collagen substrata for studies on cell behavior. J Cell Biol 54:626–637
Engel E, Michiardi A, Navarro M et al (2008) Nanotechnology in regenerative medicine: the materials side. Trends Biotechnol 26:39–47
Ergun C, Liu HN, Webster TJ et al (2008) Increased osteoblast adhesion on nanoparticulate calcium phosphates with higher ca/p ratios. J Biomed Mater Res Part A 85A:236–241
Fields GB, Lauer JL, Dori Y et al (1998) Proteinlike molecular architecture: biomaterial applications for inducing cellular receptor binding and signal transduction. Pept Sci 47:143–151
Fong H, Chun I, Reneker DH (1999) Beaded nanofibers formed during electrospinning. Polymer 40:4585–4592
Freyman T, Polin G, Osman H et al (2006) A quantitative, randomized study evaluating three methods of mesenchymal stem cell delivery following myocardial infarction. Eur Heart J 27:1114–1122
Gao X, Cui Y, Levenson RM et al (2004) In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol 22:969–976
Ghoroghchian PP, Frail PR, Susumu K et al (2005) Near-infrared-emissive polymersomes: Self-assembled soft matter for in vivo optical imaging. Proc Natl Acad Sci 102:2922–2927
Gong Y, Ma Z, Gao C et al (2006) Specially elaborated thermally induced phase separation to fabricate poly(L-lactic acid) scaffolds with ultra large pores and good interconnectivity. J Appl Polym Sci 101:3336–3342
Gong Y, Ma Z, Zhou Q et al (2008) Poly(lactic acid) scaffold fabricated by gelatin particle leaching has good biocompatibility for chondrogenesis. J Biomater Sci Polym Ed 19:207–221
Grinnell F, Bennett MH (1982) Ultrastructural studies of cell--collagen interactions. Methods Enzymol 82A:535–544
Groman EV, Bouchard JC, Reinhardt CP et al (2007) Ultrasmall mixed ferrite colloids as multidimensional magnetic resonance imaging, cell labeling, and cell sorting agents. Bioconjug Chem 18:1763–1771
Gu H, Zheng R, Zhang X et al (2004) Facile one-pot synthesis of bifunctional heterodimers of nanoparticles: A conjugate of quantum dot and magnetic nanoparticles. J Am Chem Soc 126:5664–5665
Harrison BS (2008) Applications of nanotechnology. In: Atala A (ed) Principles of regenerative medicine. Academic, New York
Hartgerink JD, Beniash E, Stupp SI (2001) Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 294:1684–1688
Hay ED (1991) Cell biology of extra cellular matrix. Plenum Press, New York
He W, Ma Z, Yong T et al (2005) Fabrication of collagen-coated biodegradable polymer nanofiber mesh and its potential for endothelial cells growth. Biomaterials 26:7606–7615
Hong Y, Legge RL, Zhang S et al (2003) Effect of amino acid sequence and pH on nanofiber formation of self-assembling peptides EAK16-II and EAK16-IV. Biomacromolecules 4:1433–1442
Hong Z, Zhang P, He C et al (2005) Nano-composite of poly(l-lactide) and surface grafted hydroxyapatite: mechanical properties and biocompatibility. Biomaterials 26:6296–6304
Huang L, McMillan RA, Apkarian RP et al (2000) Generation of synthetic elastin-mimetic small diameter fibers and fiber networks. Macromolecules 33:2989–2997
Huang L, Nagapudi K, Apkarian RP et al (2001) Engineered collagen: PEO nanofibers and fabrics. J Biomater Sci Polym Ed 12:979–993
Ishii D, Kinbara K, Ishida Y et al (2003) Chaperonin-mediated stabilization and ATP-triggered release of semiconductor nanoparticles. Nature 423:628–632
Itoh S, Kikuchi M, Takakuda K et al (2001) The biocompatibility and osteoconductive activity of a novel hydroxyapatite/collagen composite biomaterial, and its function as a carrier of rhBMP-2. J Biomed Mater Res Part A 54:445–453
Itoh S, Kikuchi M, Koyama Y et al (2004) Development of a hydroxyapatite/collagen nanocomposite as a medical device. Cell Transplant 13:451–461
Jain KK et al (2008) Regenerative medicine and tissue engineering. In: The handbook of nanomedicine. Humana Press Inc, Totowa
Jaiswal JK, Mattoussi H, Mauro JM et al (2002) Long-term multiple color imaging of live cells using quantum dot bioconjugates. Nat Biotechnol 21:47–51
Jiang H, Hu Y, Li Y et al (2005) A facile technique to prepare biodegradable coaxial electrospun nanofibers for controlled release of bioactive agents. J Control Release 108:237–243
Josephson L, Tung C-H, Moore A et al (1999) High-efficiency intracellular magnetic labeling with novel superparamagnetic-tat peptide conjugates. Bioconjug Chem 10:186–191
Khang D, Carpente J, Chun YW et al (2010) Nanotechnology for regenerative medicine. Biomed Microdevices 12:575–587
Kim J-s, Reneker DH (1999) Mechanical properties of composites using ultrafine electrospun fibers. Polym Compos 20:124–131
Kiritsy CP, Lynch SE (1993) Role of growth factors in cutaneous wound healing: a review. Critical Rev Oral Biol Med 4:729–760
Kisiday J, Jin M, Kurz B et al (2002) Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair. Proc Natl Acad Sci 99:9996–10001
Koch AM, Reynolds F, Kircher MF et al (2003) Uptake and metabolism of a dual fluorochrome Tat-nanoparticle in HeLa cells. Bioconjug Chem 14:1115–1121
Kohler N, Fryxell GE, Zhang M (2004) A bifunctional poly(ethylene glycol) silane immobilized on metallic oxide-based nanoparticles for conjugation with cell targeting agents. J Am Chem Soc 126:7206–7211
Kostura L, Kraitchman DL, Mackay AM et al (2004) Feridex labeling of mesenchymal stem cells inhibits chondrogenesis but not adipogenesis or osteogenesis. NMR Biomed 17:513–517
Kothapalli CR, Shaw MT, Wei M (2005) Biodegradable HA-PLA 3-D porous scaffolds: Effect of nano-sized filler content on scaffold properties. Acta Biomater 1:653–662
Kuntz R, Saltzman W (1997) Neutrophil motility in extracellular matrix gels: mesh size and adhesion affect speed of migration. Biophys J 72:1472–1480
Kwon IK, Matsuda T (2005) Co-electrospun nanofiber fabrics of poly(l-lactide-co-ε-caprolactone) with type I collagen or heparin. Biomacromolecules 6:2096–2105
Kwon IK, Park KD, Choi SW et al (2001) Fibroblast culture on surface-modified poly (glycolide-co-ε-caprolactone) scaffold for soft tissue regeneration. J Biomater Sci Polym Ed 12:1147–1160
Kwon IK, Kidoaki S, Matsuda T (2005) Electrospun nano- to microfiber fabrics made of biodegradable copolyesters: structural characteristics, mechanical properties and cell adhesion potential. Biomaterials 26:3929–3939
Larson DR, Zipfel WR, Williams RM et al (2003) Water-soluble quantum dots for multiphoton fluorescence imaging in vivo. Science 300:1434–1436
Layrolle P, Daculsi G (2006) Nanostructured biomaterials. Nanomedicine 1:493–494
Lemon BI, Crooks RM (2000) Preparation and characterization of dendrimer-encapsulated CdS semiconductor quantum dots. J Am Chem Soc 122:12886–12887
Lewin M, Carlesso N, Tung C-H et al (2000) Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nat Biotechnol 18:410–414
Lewis JD, Destito G, Zijlstra A et al (2006) Viral nanoparticles as tools for intravital vascular imaging. Nat Med 12:354–360
Li W-J, Laurencin CT, Caterson EJ et al (2002) Electrospun nanofibrous structure: a novel scaffold for tissue engineering. J Biomed Mater Res Part A 60:613–621
Li M, Mondrinos MJ, Gandhi MR et al (2005) Electrospun protein fibers as matrices for tissue engineering. Biomaterials 26:5999–6008
Li W, Ma N, Ong LL et al (2008) Enhanced thoracic gene delivery by magnetic nanobead-mediated vector. J Gene Med 10:897–909
Liao SS, Cui FZ, Zhang W et al (2004) Hierarchically biomimetic bone scaffold materials: nano-HA/collagen/PLA composite. J Biomed Mater Res B Appl Biomater 68B:158–165
Lidke DS, Nagy P, Heintzmann R et al (2004) Quantum dot ligands provide new insights into erbB/HER receptor − mediated signal transduction. Nat Biotechnol 22:198–203
Liu H, Webster TJ (2007) Nanomedicine for implants: a review of studies and necessary experimental tools. Biomaterials 28:354–369
Luu YK, Kim K, Hsiao BS et al (2003) Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA-PEG block copolymers. J Control Release 89:341–353
Ma L, Gao C, Mao Z et al (2003) Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering. Biomaterials 24:4833–4841
Ma Z, Gao C, Gong Y et al (2005a) Cartilage tissue engineering PLLA scaffold with surface immobilized collagen and basic fibroblast growth factor. Biomaterials 26:1253–1259
Ma Z, Kotaki M, Inai R et al (2005b) Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng 11:101–109
Ma L, Zhou J, Gao C et al (2007) Incorporation of basic fibroblast growth factor by a layer-by-layer assembly technique to produce bioactive substrates. J Biomed Mater Res B Appl Biomater 83B:285–292
Mao Z, Ma L, Zhou J et al (2005) Bioactive thin film of acidic fibroblast growth factor fabricated by layer-by-layer assembly. Bioconjug Chem 16:1316–1322
Martin GE, Cockshott ID (1977) Fibrillar product of electrostatically spun organic material. US Patent 4043331 A
Mastrobattista E, van-der Aa MAEM, Hennink WE et al (2006) Artificial viruses: a nanotechnological approach to gene delivery. Nat Rev Drug Discov 5:115–121
Matthews JA, Wnek GE, Simpson DG et al (2002) Electrospinning of collagen nanofibers. Biomacromolecules 3:232–238
Matthews JA, Boland ED, Wnek GE et al (2003) Electrospinning of collagen type II: a feasibility study. J Bioact Compat Polym 18:125–134
Maysinger D, Behrendt M, Lalancette-Hébert M et al (2007) Real-time imaging of astrocyte response to quantum dots: in vivo screening model system for biocompatibility of nanoparticles. Nano Lett 7:2513–2520
McIntyre JO, Fingleton B, Wells KS et al (2004) Development of a novel fluorogenic proteolytic beacon for in vivo detection and imaging of tumour-associated matrix metalloproteinase-7 activity. Biochem J 377:617–628
Mikos AG, Thorsen AJ, Czerwonka LA et al (1994) Preparation and characterization of poly(L-lactic acid) foams. Polymer 35:1068–1077
Miller DC, Thapa A, Haberstroh KM et al (2004) Endothelial and vascular smooth muscle cell function on poly(lactic-co-glycolic acid) with nano-structured surface features. Biomaterials 25:53–61
Miller DC, Haberstroh KM, Webster TJ (2007) PLGA nanometer surface features manipulate fibronectin interactions for improved vascular cell adhesion. J Biomed Mater Res Part A 81A:678–684
Moghimi SM, Hunter AC (2000) Poloxamers and poloxamines in nanoparticle engineering and experimental medicine. Trends Biotechnol 18:412–420
Mooney DJ, Baldwin DF, Suh NP et al (1996) Novel approach to fabricate porous sponges of poly(D, L-lactic-co-glycolic acid) without the use of organic solvents. Biomaterials 17:1417–1422
Mulder WJM, Koole R, Brandwijk RJ et al (2006) Quantum dots with a paramagnetic coating as a bimodal molecular imaging probe. Nano Lett 6:1–6
Murugan R, Ramakrishna S (2007) Design strategies of tissue engineering scaffolds with controlled fiber orientation. Tissue Eng 13:1845–1866
Nam YS, Park TG (1999a) Porous biodegradable polymeric scaffolds prepared by thermally induced phase separation. J Biomed Mater Res Part A 47:8–17
Nam YS, Park TG (1999b) Biodegradable polymeric microcellular foams by modified thermally induced phase separation method. Biomaterials 20:1783–1790
Nam YS, Yoon JJ, Park TG (2000) A novel fabrication method of macroporous biodegradable polymer scaffolds using gas foaming salt as a porogen additive. J Biomed Mater Res B Appl Biomater 53:1–7
Neffe AT, Pierce BF, Tronci G et al (2015) One step creation of multifunctional 3D architectured hydrogels inducing bone regeneration. Adv Mater 27:1738–1744
Niemeyer CM, Ceyhan B (2001) DNA-directed functionalization of colloidal gold with proteins. Angew Chem Int Ed 40:3685–3688
Papisov MI, Bogdanov A, Schaffer B et al (1993) Colloidal magnetic resonance contrast agents: effect of particle surface on biodistribution. J Magn Magn Mater 122:383–386
Parak WJ, Boudreau R, Le-Gros M et al (2002) Cell motility and metastatic potential studies based on quantum dot imaging of phagokinetic tracks. Adv Mater 14:882–885
Ramachandran GN (1988) Stereochemistry of collagen. Int J Pept Protein Res 31:1–16
Rao J, Dragulescu-Andrasi A, Yao H (2007) Fluorescence imaging in vivo: recent advances. Curr Opin Biotechnol 18:17–25
Reddy ST, Rehor A, Schmoekel HG et al (2006) In vivo targeting of dendritic cells in lymph nodes with poly(propylene sulfide) nanoparticles. J Control Release 112:26–34
Reneker DH, Chun I (1996) Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 7:216–223
Rhyner MN, Smith AM, Gao X et al (2006) Quantum dots and multifunctional nanoparticles: new contrast agents for tumor imaging. Nanomedicine 1:209–217
Rogach A, Kershaw SV, Burt M et al (1999) Colloidally prepared HgTe nanocrystals with strong room-temperature infrared luminescence. Adv Mater 11:552–555
Rovira A, Bareille R, Lopez I et al (1993) Preliminary report on a new composite material made of calcium phosphate, elastin peptides and collagens. J Mater Sci Mater Med 4:372–380
Sasaki T, Iwasaki N, Kohno K et al (2008) Magnetic nanoparticles for improving cell invasion in tissue engineering. J Biomed Mater Res Part A 86A:969–978
Schaffer BK, Linker C, Papisov M et al (1993) MION-ASF: biokinetics of an MR receptor agent. Magn Reson Imaging 11:411–417
Schellenberger EA, Bogdanov AJ, Högemann D et al (2002) Annexin V-CLIO: a nanoparticle for detecting apoptosis by MRI. Mol Imaging 1:102–107
Schellenberger EA, Sosnovik D, Weissleder R et al (2004) Magneto/optical annexin V, a multimodal protein. Bioconjug Chem 15:1062–1067
Shah BS, Clark PA, Moioli EK et al (2007) Labeling of mesenchymal stem cells by bioconjugated quantum dots. Nano Lett 7:3071–3079
Silva GA, Czeisler C, Niece KL et al (2004) Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science 303:1352–1355
Simon SR (1994) Orthopaedic basic science. American Academy of Orthopaedic Surgeons, Rosemont
So M-K, Xu C, Loening AM et al (2006) Self-illuminating quantum dot conjugates for in vivo imaging. Nat Biotechnol 24:339–343
Solanki A, Kim JD, Lee K-B (2008) Nanotechnology for regenerative medicine: nanomaterials for stem cell imaging. Nanomedicine 3:567–578
Soto CM, Blum AS, Vora GJ et al (2006) Fluorescent signal amplification of carbocyanine dyes using engineered viral nanoparticles. J Am Chem Soc 128:5184–5189
Stitzel J, Liu J, Lee SJ et al (2006) Controlled fabrication of a biological vascular substitute. Biomaterials 27:1088–1094
Strom SC, Michalopoulos G (1982) Collagen as a substrate for cell growth and differentiation. Methods Enzymol 82A:544–555
Sun Z, Zussman E, Yarin AL et al (2003) Compound core-shell polymer nanofibers by co-electrospinning. Adv Mater 15:1929–1932
Sun C, Sze R, Zhang M (2006) Folic acid-PEG conjugated superparamagnetic nanoparticles for targeted cellular uptake and detection by MRI. J Biomed Mater Res Part A 78A:550–557
Tabata Y (2003) Tissue regeneration based on growth factor release. Tissue Eng 9:S5–S15
Tampieri A, Celotti G, Landi E et al (2003) Biologically inspired synthesis of bone-like composite: self-assembled collagen fibers/hydroxyapatite nanocrystals. J Biomed Mater Res Part A 67A:618–625
Taniguchi N (1974) On the basic concept of ‘nano-technology’. In: Proceedings of the international conference of production engineering, Tokyo, pp 18–23
TenHuisen KS, Martin RI, Klimkiewicz M et al (1995) Formation and properties of a synthetic bone composite: hydroxyapatite-collagen. J Biomed Mater Res 29:803–810
Thomas V, Dean DR, Vohra YK (2006a) Nanostructured biomaterials for regenerative medicine. Curr Nanosci 2:155–177
Thomas V, Jagani S, Johnson K et al (2006b) Electrospun bioactive nanocomposite scaffolds of polycaprolactone and nanohydroxyapatite for bone tissue engineering. J Nanosci Nanotechnol 6:487–493
Thomas V, Dean DR, Jose MV et al (2007) Nanostructured biocomposite scaffolds based on collagen co-electrospun with nanohydroxyapatite. Biomacromolecules 8:631–637
Thorek DLJ, Chen AK, Czupryna J et al (2006) Superparamagnetic iron oxide nanoparticle probes for molecular imaging. Ann Biomed Eng 34:23–38
Vaccaro DE, Yang M, Weinberg JS et al (2008) Cell tracking using nanoparticles. J Cardiovasc Transl Res 1:217–220
van-Tilborg GAF, Mulder WJM, Chin PTK et al (2006) Annexin A5-conjugated quantum dots with a paramagnetic lipidic coating for the multimodal detection of apoptotic cells. Bioconjug Chem 17:865–868
Venugopal J, Ma LL, Yong T et al (2005) In vitro study of smooth muscle cells on polycaprolactone and collagen nanofibrous matrices. Cell Biol Int 29:861–867
Verreck G, Chun I, Rosenblatt J et al (2003) Incorporation of drugs in an amorphous state into electrospun nanofibers composed of a water-insoluble, nonbiodegradable polymer. J Control Release 92:349–360
Wang W, Li W, Ong LL et al (2010) Localized SDF-1alpha gene release mediated by collagen substrate induces CD117 stem cells homing. J Cell Mol Med 14:392–402
Webster TJ (2001) Nanophase ceramics: the future orthopedic and dental implant material. In: Ying JY-R (ed) Nanostructured materials. Academic, New York
Webster TJ, Ejiofor JU (2004) Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials 25:4731–4739
Webster TJ, Siegel RW, Bizios R (1999) Osteoblast adhesion on nanophase ceramics. Biomaterials 20:1221–1227
Wei G, Ma PX (2006) Macroporous and nanofibrous polymer scaffolds and polymer/bone-like apatite composite scaffolds generated by sugar spheres. J Biomed Mater Res Part A 78A:306–315
Wei G, Ma PX (2008) Nanostructured biomaterials for regeneration. Adv Funct Mater 18:3568–3582
Wei G, Jin Q, Giannobile WV et al (2006) Nano-fibrous scaffold for controlled delivery of recombinant human PDGF-BB. J Control Release 112:103–110
Weissleder R, Papisov M (1992) Pharmaceutical iron oxides for MR imaging. Rev Magn Reson Med 4:1–20
Weissleder R, Bogdanov A, Neuwelt EA et al (1995) Long-circulating iron oxides for MR imaging. Adv Drug Deliv Rev 16:321–334
Wentworth B, Stewart J, Westrich J, et al (2007) Studies on the retention of cells delivered to the rat heart. In: European society of cardiology congress, Vienna
Whitesides GM, Grzybowski B (2002) Self-assembly at all scales. Science 295:2418–2421
Winter JO, Liu TY, Korgel BA et al (2001) Recognition molecule directed interfacing between semiconductor quantum dots and nerve cells. Adv Mater 13:1673–1677
Wnek GE, Carr ME, Simpson DG et al (2003) Electrospinning of nanofiber fibrinogen structures. Nano Lett 3:213–216
Wu C, Barnhill H, Liang X et al (2005) A new probe using hybrid virus-dye nanoparticles for near-infrared fluorescence tomography. Opt Commun 255:366–374
Xu X, Wang W, Kratz K, et al (2014) Controlling major cellular processes of human mesenchymal stem cells using microwell structures. Adv Healthc Mater 3:1991–2003
Yang F, Murugan R, Ramakrishna S et al (2004a) Fabrication of nano-structured porous PLLA scaffold intended for nerve tissue engineering. Biomaterials 25:1891–1900
Yang XB, Bhatnagar RS, Li S et al (2004b) Biomimetic collagen scaffolds for human bone cell growth and differentiation. Tissue Eng 10:1148–1159
Yao C, Slamovich EB, Webster TJ (2008) Enhanced osteoblast functions on anodized titanium with nanotube-like structures. J Biomed Mater Res Part A 85A:157–166
Ye L, Huang X (2005) MAP2: multiple alignment of syntenic genomic sequences. Nucleic Acids Res 33:162–170
Yoshimoto H, Shin YM, Terai H et al (2003) A biodegradable nanofiber scaffold by electrospinning and its potential for bone tissue engineering. Biomaterials 24:2077–2082
Yu Y-C, Tirrell M, Fields GB (1998) Minimal lipidation stabilizes protein-like molecular architecture. J Am Chem Soc 120:9979–9987
Yu Y-C, Roontga V, Daragan VA et al (1999) Structure and dynamics of peptide-amphiphiles incorporating triple-helical proteinlike molecular architecture. Biochemistry 38:1659–1668
Yu WW, Qu L, Guo W et al (2003) Experimental determination of the extinction coefficient of CdTe, CdSe, and CdS nanocrystals. Chem Mater 15:2854–2860
Zaheer A, Lenkinski RE, Mahmood A et al (2001) In vivo near-infrared fluorescence imaging of osteoblastic activity. Nat Biotechnol 19:1148–1154
Zeng J, Chen X, Xu X et al (2003) Ultrafine fibers electrospun from biodegradable polymers. J Appl Polym Sci 89:1085–1092
Zhang S (2003) Fabrication of novel biomaterials through molecular self-assembly. Nat Biotechnol 21:1171–1178
Zhang R, Ma PX (1999) Poly(α-hydroxyl acids)/hydroxyapatite porous composites for bone-tissue engineering. I. Preparation and morphology. J Biomedical Mater Res Part A 44:446–455
Zhang R, Ma PX (2000) Synthetic nano-fibrillar extracellular matrices with predesigned macroporous architectures. J Biomed Mater Res Part A 52:430–438
Zhang L, Webster TJ (2009) Nanotechnology and nanomaterials: promises for improved tissue regeneration. Nano Today 4:66–80
Zhang Y, Huang Z-M, Xu X et al (2004) Preparation of core − shell structured PCL-r-gelatin bi-component nanofibers by coaxial electrospinning. Chem Mater 16:3406–3409
Zhang Y, Ouyang H, Lim CT et al (2005a) Electrospinning of gelatin fibers and gelatin/PCL composite fibrous scaffolds. J Biomed Mater Res B Appl Biomater 72B:156–165
Zhang YZ, Venugopal J, Huang Z-M et al (2005b) Characterization of the surface biocompatibility of the electrospun PCL-collagen nanofibers using fibroblasts. Biomacromolecules 6:2583–2589
Zhang Y, Li W, Ou L et al (2012) Targeted delivery of human VEGF gene via complexes of magnetic nanoparticle-adenoviral vectors enhanced cardiac regeneration. PLoS One 7:e39490
Zhou Q, Gong Y, Gao C (2005) Microstructure and mechanical properties of poly(L-lactide) scaffolds fabricated by gelatin particle leaching method. J Appl Polym Sci 98:1373–1379
Zong X, Kim K, Fang D et al (2002) Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 43:4403–4412
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Zhou, J., Gao, C., Li, W. (2016). Functionalized Nanomaterials. In: Steinhoff, G. (eds) Regenerative Medicine - from Protocol to Patient. Springer, Cham. https://doi.org/10.1007/978-3-319-28274-9_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-28274-9_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-28272-5
Online ISBN: 978-3-319-28274-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)