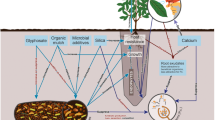
Abstract
Soil-borne diseases and soil fatigue cause relevant economic losses in greenhouse pepper crops from Spain, not only in the South-eastern Mediterranean region but also in the Northern areas with a humid temperate climate. The main soil phytopathological problems in the South-east are the oomycetes Phytophthora parasitica and P. capsici, the nematode Meloidogyne incognita and the soil fatigue by non-pathogenic Fusarium species proliferation. The main problems in the North are P. capsici, P. cryptogea and Verticillium dahliae. The mechanisms involved in disease suppression by organic amendments and the management strategies for the control of protected pepper crops, soil-borne diseases and soil fatigue are reviewed. Biosolarisation provides an effective and stable strategy for soil-borne pathogens control and the mitigation of soil fatigue. When biosolarisation is repeated, its effectiveness against fatigue and pathogens increases either by providing direct action against fungal microbiota and/or increasing plant health through the improvement of soil chemical and physical characteristics. Increase in macro-micronutrients and water infiltration capacity and decrease in apparent density and compaction are among the improvements in soil characteristics that are related to a crop production increase. Biosolarisation combined with organic amendments improves the soil physical characteristics, specifically in relation to the control of Phytophthora root rot which is more important in compact clay soils than in well-ventilated soils with adequate drainage. In recent years numerous alternatives for chemical disinfections have been studied, and of these, those based on organic amendments alone or in combination with solarisation seem to be the most promising (Guerrero et al. 2013) in intensive protected horticultural crops.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Soil Phytopathological Problems and Soil Fatigue in Protected Pepper Crops
Phytophthora root rot is a destructive disease for pepper plants (Capsicum annuum L.) worldwide (Wang et al. 2014). The mortality of pepper plants ranges between 30 and 40 % and in severe cases, even 100 % (Liu et al. 2008). As a consequence of the high plant mortality, relevant economic losses have been reported in pepper crops from Spain, not only in the Mediterranean region (Tello and Lacasa 1997) but also in areas characterised by a humid temperate climate, such as the Basque Country (Northern Spain) (Larregla 2003). The main causal agents of this disease in greenhouse pepper crops are the oomycetes Phytophthora capsici and P. cryptogea in Northern Spain and P. capsici and P. parasitica in South-eastern Spain. In the last region, the nematode Meloidogyne incognita is also a recurring and persistent problem that causes substantial crop damages (Tello and Lacasa 1997; Bello et al. 2004). In South-eastern Spain (Murcia and Alicante provinces) pepper occupies more than 90 % of the area dedicated to greenhouse crops and has been a monoculture for the last 20 years (Lacasa and Guirao 1997). The normal crop cycle lasts 9–10 months (November–December to September–October). Strategies recommended for management of phytophthora root rot involve integrated approaches that focus on cultural practices: reduced soil moisture, reduction of pathogen propagule in soil, utilisation of cultivars with resistance to the disease and the judicious fungicide applications (Ristaino and Johnston 1999). Until the year 2005, methyl bromide (MB) was used to disinfect soils to control both pathogens (Gilreath and Santos 2004) and to lessen the effects of fatigue caused by repeated monocultures (Martínez et al. 2011a). Since 2005, MB has been replaced by a mixture of 1,3-dichloropropene and chloropicrin, but these will also be banned by European legislation in the near future. The increasing demand for ecological foods produced by sustainable agricultural practices must be added to this forthcoming ban, meaning that non-chemical methods will have to be developed for controlling soil-borne pl ant path ogens and plant parasitic nematodes adapted for use in intensive horticulture.
In recent years, numerous alternatives for chemical disinfection have been studied, and of these, those based on organic amendments alone or in combination with solarisation seem to be the most promising (Guerrero et al. 2013) in intensive protected horticultural crops.
In this book chapter, we aim to review both the me chanisms involved in disease suppression and the organic amendment management strategies for the control of protected pepper crops soil-borne diseases and soil fatigue. Several disease suppression mechanisms following the addition of organic matter such as (1) the release of compounds that are toxic to the pathogens, (2) the stimulation of non-pathogenic microorganisms that inhibit or kill the pathogens and (3) the improvement of so il physical, chemica l and biological properties will be explained.
2 Mechanisms Involved in Disease Suppression by Soil Organic Amendments
Several mechanisms have been identified as contributing to disease suppression following the addition of organic matter. These namely include the stimulation of non-pathogenic microorganisms that inhibit or kill the pathogens through competition (Lockwood 1988) or parasitism (Hoitink and Boehm 1999), the release of compounds that are toxic for the pathogens (Bailey and Lazarovits 2003) and the stimulation of the host plant’s disease defence system (Zhang et al. 1996, 1998). Other indirect mechanisms that explain the ability of organic amendments to increase soil suppressiveness are: improvement of nutrition and vigour in the host plants and improvement of physicochemical and biological properties of soil. Some organic amendments are thought to work primarily by altering the structure of the microbial communities in the soil or by changing the physical and chemical properties of the soil.
2.1 Production of Biocidal Compounds
The application of animal manure leads to the generation of ammonia (NH3), which is the mechanism most often implicated in killing soil pathogens (Tenuta and Lazarovits 2002), although other lethal molecules such as nitrous acid and volatile fatty acids (VFA ) have also been reported (Tenuta et al. 2002; Conn et al. 2005). Tenuta and Lazarovits (2002) summarised that NH3 is thought to kill cells by disrupting membranes, eliminating proton gradients across membranes, through the assimilation of NH3 into glutamine and the exhaustion of the chemical energy of cells removing cytosolic NH3. Accumulation of NH3 and VFA derived from manure application have been described as mechanisms capable to kill soil pathogens (Conn et al. 2005), which is largely influenced by several factors that include moisture content, pH, soil organic matter content and quality, soil texture and buffering capacity and nitrification rate soil buffering capacity. These authors concluded that high VFA toxicity was achieved in acid soils (pH about 5.0) while high NH3 toxicity was related with alkaline soils (pH about 7.5).
The application of animal manure followed by soil plastic covering during spring period reduced inoculum survival of the fungal pathogen P. capsici and disease incidence in a greenhouse pepper crop in Northern Spain (Arriaga et al. 2011). Northern Spain is an area characterised by a temperate climate with annual mean temperature of 12 °C, a maximum mean temperature in summer of 25 °C and rainfall of 1200 mm per year.
Ammonia volatilisation , among other volatile comp ounds, and the increase in soil suppressiveness contributed to minimise P. capsici inoculum survival rate and subsequent greenhouse crop disease incidence, respectively. The use of fresh manure favoured NH3 volatilisation as organic nitrogen (Norg) mineralisation was higher than in semicomposted manure, with more stable Norg content. Mean NH3 concentration increased with fresh sheep manure and dry chicken litter (SCM) during biodisinfestation process compared with semicomposted mixture of horse manure and chicken litter (HCM) (Fig. 21.1).
Evolution of NH3 concentration under the plastic sheets from non-amended (control), fresh manure (SCM) and semicomposted manure (HCM) amended plots during soil biodisinfestation starting on March 14, 2008, showing variation of air temperature throughout the biodisinfestation period. The vertical bars indicate least significance difference at 0.05 between treatments. Soil was tarped with 50-μm-thick (two million) transparent low density polyethylene plastic film. The greenhouse field experiment was located in Derio (Biscay) (Northern Spain). Reprinted from Journal of Crop Protection, 30(4), H. Arriaga et al. (2011), Gaseous emissions from soil biodisinfestation by animal manure on a greenhouse pepper crop, 412–419, Copyright (2015), with permission from Elsevier
NH3 concentration increased significantly after manure amendment with respect to control plots (C) and also differed between SCM and HCM manure . Ammonia concentration from C plots averaged 3.9 mg NH3 m−3, while SCM averaged 14.8 mg NH3 m−3 and 9.1 mg NH3 m−3 in HCM. The highest NH3 concentrations were reached at the beginning of the experiment in SCM and HCM treatments and decreased 45.0 % after 35 days of soil biodisinfestation (Fig. 21.1). The reduction of NH3 concentrations could be related to adsorption of NH3 or the increasing anaerobic conditions during manure decomposing process (Kirchmann and Witter 1989). The high water condensation observed on the plastic inner surface, which would trap volatilised NH3 (Kroodsma et al. 1993), and the overall anaerobic conditions under plastic sheets might have reduced NH3 accumulation (Kirchmann and Witter 1989).
P. capsici inoculum survival rate in infected plant re sidues was significantly different among treatments. The application of fresh SCM under plastic sheets reduced Phytophthora inoculum survival in relation to HCM and S treatments (Table 21.1) (P < 0.05). Biodisinfestation by SCM manure reduced by 50 % inoculum survival rate compared with C plots (61.1 %), while Phytophthora inoculum survived in 75.0 % and 94.4 % of plant residues in HCM and S treatments, respectively, which was significantly higher than survival reported in C plots. The higher NH3 concentration in SCM contributed to reduce the inoculum survival rate of P. capsici (30.6 % and 75.0 % in SCM and HCM treatments, respectively). Inoculu m survival rate was not reduced in solarised non-amended plots (94.4 %) as soil temperature at 15 cm depth did not exceed 33 °C under plastic sheets in S, SCM and HCM treatments (a temperature known to be insufficient to inactivate resistant propagules of P. capsici since this pathogen normally shows an optimum temperature range at 24–33 °C) (Erwin and Ribeiro 1996; Etxeberria et al. 2011). Additionally, warm soil temperatures and water condensation detected during the biodisinfestation process might have favoured the conditions for Phytophthora inoculum survival in S and HCM plots when compared with C plots. The lower inoculum survival rate in C plots was related to the higher water evaporation and the subsequent lower soil volumetric water content in these uncovered soils.
Higher P. capsici inoculum inactivation observed in SCM was attributed to the effect of toxic volatile compounds generated from the decomposition of organic amendments. Soil pH of our experiment averaged 6.9 in SCM and HCM treatments, which might suggest that NH3 contributed significantly to the reduction of Phytophthora inoculum survival in SCM. Moreover, Oka et al. (2007) summarised that soil amendments with low C/N ratios have been reported to have fungicidal activity mainly through the release of NH3. In our experiment, the amount of manure amended exceeded those rates applied by Oka et al. (2007), which would support that NH3 was the main factor controlling ino culum survival of Arriaga et al. (2011).
2.2 Increase in Microbial Activity
Phytophthora disease incidence decreased significantly in biodisinfestated SCM and HCM plots compared with C and S treatments (Table 21.1). Plant disease incidence was reduced by 90 % in SCM and HCM plots in relation to inoculum survival rate observed in plant residues 4 months before. Of note, disease was only reduced by 33 % and 54 % in C and S plots, respectively. The application of high amounts of organic amendments contributes to the suppressive capacity of soils through enhanced activity and growth of edaphic microorganisms, which may play an important role in reducing disease incidence by an antagonistic mechanism (Hoitink and Boehm 1999). The significant reduction of disease incidence compared with the high inoculum survival rate could explain this phenomenon in SCM and HCM plots. Several authors have also reported the success of organic matter applications in the control of Phytophthora spp., suggesting that the competition for nutrients and antibiosis are the main mechanism s involved in Phytophthora spp. suppressiveness (Leoni and Ghini 2006).
2.3 Improvement of Plant Nutrition and Vigour
Soil organic matter management affects not only soil biological properties but also soil chemical and physical properties and plant nutrient status. All of them improve plant health and vigour (Stone et al. 2004) and thus may help the plants to overcome pathogen infection. The increase in soil-borne disease suppression by organic amendments may also be attributed to other effec ts such as increase in plant nutritional status and vigour (Hoitink et al. 1997).
Pepper fruit yield increased with manu re amendment in SCM and HCM (Table 21.1), as the application of organic amendments improves soil quality, increasing the amount of plant-available nutrients and, in consequence, crop yield (Liu et al. 2008).
Highest values of crop vigour (plant height) were observed in plots amended with animal manures, and differences increased during crop development (Fig. 21.2) (Núñez-Zofío et al. 2010; Arriaga et al. 2011).
Effect of treatments on pepper plant height, measured at the middle (63 days) and at the end (119 days) of crop development. Error bars represent standard error of the mean (n = 3) from three replicate plots. Different letters indicate significant differences (P < 0.05) (least significance difference = 9.71 and 17.72 cm at 63 and 119 days after transplant, respectively) according to Fisher’s protected LSD test. Reprinted from Acta Horticulturae (ISHS), http://www.actahort.org/books/883/883_44.htm, 883, Núñez-Zofío et al. (2010), Application of organic amendments followed by plastic mulching for the control of Phytophthora root rot of pepper in Northern Spain, 353–360, with permission from International Society for Horticultural Science
2.4 Improvement of Soil Physical and Chemical Properties
Detected differences in plant nutrient status have been generally found between nonamended and amended soils. This could be due to the improved nutrient content, water holding capacity and soil structure imparted to the soil by the amendments (Vallad et al. 2003).
The effects of repeated biodisinfestation with diff er ent organic amendments after three consecutive crop seasons improved soil physical properties through a reduction in soil bulk density and an increase in soil water infiltration (Table 21.2) (Núñez-Zofío et al. 2012). This management strategy provided an effective control of phytophthora root rot in protected pepper crops. Improvements in soil water properties that prevent water flooding are known to facilitate soil-borne pathogen control , mainly in the case of oomycetes (Liu et al. 2008).
In general terms, biodisinfestation with non-composted and semicomposted manures increased the values of all soil chemical properties, except for pH (Table 21.3). Besides, non-composted manure was the only treatment that significantly increased P2O5, Cl−, K+ and Zn2+ contents. Significantly, higher values of Cu2+ content were found only in semicomposted manure-biodisinfected soils. However, no significant differences were observed between Brassica-treated plots and co ntrol soils (Table 21.3) (Núñez-Zofío et al. 2012).
2.5 Improvement of Soil Biological Properties
Biodisinfected soils with non-composted and semicomposted manure showed significantly higher values of all enzyme activities when compared with control non-amended soils, whereas Brassica treatment significantly increased the values of dehydrogenase, β-glucosidase and acid phosphatase activity compared with control non-amended treatments (Table 21.4). Apart from the organic matter input, this increase of enzyme activities could also be attributed, at least partly, to a stimulatory rhizosphere effect caused by a Sinapis alba cover crop during the winter season. This rhizosphere effect could also be responsible for the higher values of microbial population densities (total bacteria, actinomycetes and Pseudomonas spp.) detected in Brassica-amended soil. No significant differences were found between control and plastic-mulched plots (Table 21.4). Significant positive correlations were obtained between organic matter content and the following enzyme activities: FDA, dehydrogenase, urease, alkaline phosphatase and aci d phosphatase.
Biodisinfested soils had significantly higher values of potentially mineralisable nitrogen than control and plastic-mulched soils, but higher values of water s oluble organic carbon and microbial biomass carbon were obtained in manure-biodisinfected soils. Highest values were found in non-composted manure (Table 21.4). The higher values of enzyme activities obtained in non-composted and semicomposted manure-amended soils were concomitant with higher values of microbial biomass carbon, indicating that the higher levels of microbial activity were in this case due to an increase in microbial biomass. It was also observed both in non-composted and semicomposted manure-amended soils, an increase in potentially mineralisable nitrogen and water soluble organic carbon (indicators of biologically active N and C, respectively) (Núñez-Zofío et al. 2012).
Mandal et al. (2007) also found that the incorporation of farmyard manure increased enzyme activities in response to an increase in microbial biomass carbon and an improvement in the soil nutrient status.
The incorporation of fresh manure to the soil must be carried out with caution due to a possible increase in salt content which can alter soil properties and affect crop production an d disease control (Litterick et al. 2004; Moral et al. 2009). Núñez-Zofío et al. (2012) reported that significantly highest values of P2O5, K+ and Cl− were observed in non-composted soils when compared with semicomposted manure-amended soils. Although salinisation can negatively affect soil microbial properties (Rietz and Haynes 2003), Núñez-Zofío et al. (2012) found no differences in soil microbial properties between non-composted and semicomposted soils. In any event, composting provides a more stabilised product, thereb y re ducing the risk of soil salinisation, leaching and phytotoxicity (Moral et al. 2009).
3 Management of Soil-Borne Diseases with Organic Amendments in Protected Pepper Crops
Several reports from farmers show that plant diseases and the need for chemical control measures are reduced over time when practices that improve soil health are used. Research shows that soil management and microbial diversity are key factors in suppressing plant diseases. Organic amendments are quite diverse, including various types of organic materials such as animal manures, food processing wastes, crop residues and sewage sludge. Materials can be composted or uncomposted. The type of organic matter added to the soil could be of use as substrates, and their quality and quantity determine the types of organisms (both pathogens and natural occurring antagonists) that can profit the nutrients (Stone et al. 2004). It is well documented the use of specific organic amendments with suppressive effects against pathogens such as fungi, nematodes and bacteria. These amendments are primarily used for the control of diseases that these pathogens produce (Stone et al. 2004; Bonanomi et al. 2007, 2010; Oka 2010) although also are secondarily used to control soil fatigue caused by microorganisms that take advantage of plant weakness or subclinic pathogens (Manici et al. 2003; Martínez et al. 2011a, b; Mazzola and Manici 2012; Weerakoon et al. 2012; Guerrero 2013). Soil solarisation is an approach for soil disinfestation which uses passive so lar heating of soil with plastic sheeting, usually transparent polyethylene (Stapleton 2000). The resulting soil temperature increase leads to decreased populations of pathogens. Soil solarisation and the application of organic amendments on soil have been described as a valid alternative to the use of chemical fumigants to reduce Phytophthora from pepper crops (Ristaino and Johnston 1999). In the technique known as Anaerobic Soil Disinfestation (also termed Biological Soil Disinfestation /Soil reductive sterilisation /Reductive Soil Disinfestation ), organic amendments are applied in conjunction with soil tarping with an impermeable film for inducing anaerobiosis in order to generate toxic compounds (Blok et al. 2000; Momma 2008; Butler et al. 2012a, b). The advantage of anaerobic soil disinfestation when compared with soil solarisation is that the method does not require high solar radiation so it can be applied in cloudy areas or periods of low sunlight and, thus, a growing season is not lost (Baysal-Gurel et al. 2012).
3.1 Biofumigation
The term biofumigation was originally coined for that part of the suppressive effects of Brassica species on noxious soil-borne organisms (Kirkegaard et al. 1993) that arose quite specifically through liberation of isothiocyanates from hydrolysis of the glucosinolates that characterise the Brassicaceae (Kirkegaard and Matthiessen 2004). Since being coined, the initial term biofumigation has broadened its initial meaning and currently encompasses any beneficial effect arising from green manure or rotation crops and even composts (Matthiessen and Kirkegard 2006).
The use of Brassicaceae in crop rotations or as gree n manure amendment in biofumigation treatments (Stapleton and Bañuelos 2009) has proven to reduce the incidence of some soil-borne pathogens and plant parasites, including nematodes (Smolinska et al. 2003; Larkin and Griffin 2007), through their release of isothiocyanates. Improved pathogen and weed control has been achieved by using amendments obtained from by-products produced during the extraction of oil from Brassica carinata and Sinapis alba seeds (Palmieri 2005; Sachi et al. 2005; Lazzeri and Manici 2000; Cohen et al. 2005; Lazzeri et al. 2010). Brassica carinata (BP) pellets or B. carinata (BP) + fresh sheep manure (M) were evaluated for biodisinfestation treatments which began on two different dates (August and October), and the results were compared with MB-disinfested and untreated controls in greenhouse pepper crops in South-eastern Spain (Guerrero et al. 2013). During the third year, the gall index for BP was lower than that obtained for BP + M, and it was also lower in August than in October (Table 21.5).
The commercial crop of pepper fruit obtaine d in August biodisinfestations was similar or higher than the one obtained with MB, but higher than in Octob er biodisinfestation treatments (Table 21.6). The yield of the October biodisinfestation treatments was higher than that of the untreated. In August of all the studied years, the accumulated exposure times were greater than the thresholds required to kill M. incognita populations at 15 cm soil depth. The incidence of the nematode did not correspond to the reduction achieved during solarisation and seemed to increase during the crop cycle.
3.2 Biosolarisation
The approach of combining soil solarisation together with the application of organic matter has been defined as biosolarisation (Ros et al. 2008) or biodisinfestation (de la Fuente et al. 2009). In this book chapter, the term biodisinfestation will be used in a more general sense than biosolarisation. Biodisinfestation will be applied for the combined use of an organic amendment and soil plastic tarping without implying soil heating (solarisation). Combining soil solarisation with the amendment of fresh organic residues elevates soil temperature by an additional 1–3 °C, in addition to the generation of toxic volatile compounds (Biofumigation) which enhance the vulnerability of soil pathogens (Gamliel and Stapleton 1993; Gamliel et al. 2000; Bello et al. 2004; Stapleton and Bañuelos 2009). Several mechanisms are involved: (1) accumulation of toxic volatile compounds generated during organic matter decomposition; (2) creation of anaerobic conditions in the soil and (3) increase in soil suppressiveness due to high levels of microbial activity (Gamliel et al. 2000). In these processes, the effects of anaerobiosis and temperature (Fig. 21.3) are added to the effect of gases and bioactive compounds released during organic matter decomposition (Lazarovits 2001; Tenuta and Lazarovits 2002; Arriaga et al. 2011).
Hourly temperatures (°C) and oxygen volumetric content (%) continuously recorded at 15 cm soil depth during biodisinfestation treatments with different organic amendments on August 7, 2009. The greenhouse field experiment was located in Derio (Biscay) (Northern Spain). Soil was tarped with 50-μm-thick (two million) transparent low density polyethylene plastic film from August 6 to September 22, 2009 (Larregla S; unpublished data)
Biosolarisation (BS) using fresh sheep manure (M) as ame ndment in August provided similar results to those obtained using MB for Phytophthora spp. control of protected pepper crops in South-eastern Spain (Guerrero et al. 2004a). Production increased when the application was repeated more than 2 years (Guerrero et al. 2004b, 2006; Cándido et al. 2005), but it seemed to hav e little effe ct when applied after the beginning of September (Guerrero et al. 2010).
4 The Problem of Soil Fatigue in Greenhouse Pepper Monocultures and its Control with Organic Amendments
Soil fatigue has been defined as: “the reduced development of certain crops when cultivated two or more times in the same soils” (Scotto-La Massese 1983; Bouhot 1983). Soil fatigue can be caused by one or a combination of several factors of physical, chemical or biological nature. Biotic component of soil fatigue usually includes certain microorganisms that take advantage of plant weakness or subclinic pathogens that tend to accumulate in soil with crop repetition (Manici et al. 2003; Martínez et al. 2011a, b; Mazzola and Manici 2012; Weerakoon et al. 2012; Guerrero 2013; Guerrero et al. 2014). These microorganisms have been isolated from plants showing vegetative depression, when they were repeatedly cultivated in the same soil, but did not produce disease when inoculated, nor reproduced the symptoms of d epressi on, so that they were considered “weakness or subclinical pathogens” by Katan and Vanacher (1990).
In greenhouse pepper monocultures, soil fatigue appears in soils without primary pathogens (Phytophthora capsici or P. parasitica, Meloidogyne incognita) after the second year of crop repetition (Guerrero 2013). The depressive effect on plant development and the loss of production are related to the proliferation of species of Fusarium (Martínez et al. 2009, 2011a). Soil fatigue’s specific depressive effect on the pepper plots is mitigated by soil disinfection (Guerrero 2013) and the reduction of the population densities of Fusarium solani, F. oxysporum and F. equiseti (Martínez et al. 2009). Soil disinfection through the use of chemical disinfectants (methyl bromide or chloropicrin) have less durable effects than when an organic amendment is used (fresh sheep manure + poultry manure), either alone (biofumig ation) or when the soil is covered with plastic (biosolarisation) (Martínez et al. 2011b) (Table 21.7).
When biosolarisation is repeated, its effectiveness against soil fatigue increases (Martínez et al. 2009), either by providing direct action against fungal microbiota and/or increasing plant health through the improvement of soil chemical and physical characteristics (Fernández et al. 2005). Increase in macro- and micronutrients, increase in water infiltration capacity and decrease in apparent density and compaction are among the improvements in soil characteristics that may be mentioned. The use of biosolarisation combined with organic amendments in pepper greenhouses influences the soil physical characteristics, specifically in relation to the control of Phytophthora capsici or P. parasitica. These fungal pathogens are found in greater numbers in compact clay soils than in well-ventilated soils with adequate drainage. Even so, the control of the disease (root rot) can also be attributed to the effects of temperature, the released gases in the amen dments bio-decompo sition (Guerrero et al. 2010; Lacasa et al. 2010) and the suppressiveness connected with bacterial microorganisms (Núñez-Zofío et al. 2011).
5 Conclusions
Biosolarisation provides an effective and stable strategy for soil-borne pathogens control and the mitigation of soil fatigue in protected pepper monocultures.
Biosolarisation reiteration improves soil chemical, physical and biological properties with a subsequent increase in Phytophthora control effectiveness and crop yield.
However, field studies to establish types and rates of organic amendments should be carried out in different horticultural pathosystems in order to optimise pest, soil and crop responses when organic amendment incorporation is combined with soil plastic tarping at moderate soil temperatures. In-depth knowledge of several mechanisms that are contributing to control of soil-borne pathogens is needed. Future research should focus on the complexity of relationships among microbial communities for the establishment of soil management strategies towards a sustainable plant disease control.
References
Arriaga H, Núñez-Zofío M, Larregla S, Merino P (2011) Gaseous emissions from soil biodisinfestation by animal manure on a greenhouse pepper crop. Crop Prot 30:412–419
Bailey KL, Lazarovits G (2003) Suppressing soilborne diseases with residues management and organic amendments. Soil Tillage Res 72:169–180
Baysal-Gurel F, Gardener BM, Miller SA (2012) Soilborne disease management in organic vegetable production. Department of Plant Pathology, The Ohio State University, published online 1 Aug 2012. http://www.extension.org/pages/64951/soilborne-disease-management-in-organic-vegetable-production. Verified 17 Feb 2014
Bello A, López-Pérez JA, García-Álvarez A, Arcos SC, Ros C, Guerrero MM, Guirao P, Lacasa A (2004) Biofumigación con solarización para el control de nematodos en cultivo de pimiento. In: Lacasa A, Guerrero MM, Oncina M, Mora JA (eds) Desinfección de suelos en invernaderos de pimiento. Publicaciones de la Consejería de Agricultura, Agua y Medio Ambiente, Región de Murcia, pp 105–129
Blok WJ, Lamers JG, Termorshuizen AJ, Bollen GJ (2000) Control of soilborne plant pathogens by incorporating fresh organic amendments followed by tarping. Phytopathology 90:253–259
Bonanomi G, Antignani V, Pane C, Scala F (2007) Suppression of soilborne fungal diseases with organic amendments. J Plant Pathol 89:311–324
Bonanomi G, Antignani V, Capodilupo M, Scala F (2010) Identifying the characteristics of organic soil amendments that suppress soilborne plant diseases. Soil Biol Biochem 42:136–144
Bouhot D (1983) Étude de la fatigue des sols dans les aspergerais et les pépinières d’asperge. In: INRA (ed) La fatigue des sols. Diagnostic de la fertilité dans les systèmes culturaux. INRA, Paris, pp 61–64
Butler DM, Rosskopf EN, Kokalis N, Burelle N, Albano JP, Muramoto J, Shennan C (2012a) Exploring warm-season cover crops as carbon sources for anaerobic soil disinfestation (ASD). Plant Soil 335:149–165
Butler DM, Kokalis-Burelle N, Muramoto J, Shennan C, McCollum TG, Rosskopf EN (2012b) Impact of anaerobic soil disinfestation combined with soil solarization on plant–parasitic nematodes and introduced inoculum of soilborne plant pathogens in raised-bed vegetable production. Crop Prot 39:33–40
Candido V, Miccolis V, Basile M, D’Addabbo T, Gatta G (2005) Soil solarization for the control of Meloidogyne javanica on eggplant in Southern Italy. Acta Hortic 698:195–199
Cohen MF, Yamasaki H, Mazzola M (2005) Brassica napus seed meal soil amendment modifies microbial community structure, nitric oxide production and incidence of Rhizoctonia root rot. Soil Biol Biochem 37:1215–1227
Conn KL, Tenuta M, Lazarovits G (2005) Liquid swine manure can kill Verticillium dahliae microsclerotia in soil by volatile fatty acid, nitrous acid, and ammonia toxicity. Phytopathology 95:28–35
de la Fuente E, Soria AC, Díez-Rojo MA, Piedra Buena A, García-Álvarez A, Almendros G, Bello A (2009) Solid-phase micro-extraction (SPME) in the early detection of potentially active volatile compounds from organic wastes used for the management of soil-borne pathogens. J Environ Sci Health A 44:1004–1010
Erwin DC, Ribeiro OK (1996) Phytophthora diseases worldwide. American Phytopathological Society, St. Paul, MN
Etxeberria A, Mendarte S, Larregla S (2011) Thermal inactivation of Phytophthora capsici oospores. Rev Iberoam Micol 28(2):83–90
Fernández P, Guerrero MM, Martínez MA, Ros C, Lacasa A, Bello A (2005) Effects of biofumigation plus solarization on soil fertility. In: Industrial crops and rural development. Proceedings of annual meeting of the association for the advancement of industrial crops, Murcia, 17–21 Sept 2005, pp 229–236
Gamliel A, Stapleton JJ (1993) Characterization of antifungal volatile compounds evolved from solarized soil amended with cabbage residues. Phytopathology 38:899–905
Gamliel A, Austerweil M, Kritzman G (2000) Non-chemical approach to soilborne pest management – organic amendments. Crop Prot 19:847–853
Gilreath JP, Santos BM (2004) Methyl bromide alternatives for weed and soilborne disease management in tomato (Lycopersicon esculentum). Crop Prot 23:1193–1198
Guerrero MM (2013) Biofumigación y desinfección de suelos de invernadero para cultivos de pimiento y la fatiga del suelo. PhD thesis, ETSIA, Universidad Politécnica de Cartagena, Murcia, 215 pp
Guerrero MM, Lacasa A, Ros C, Bello A, Martínez MC, Torres J, Fernández P (2004a) Efecto de la biofumigación con solarización sobre los hongos del suelo y la producción: fechas de desinfección y enmiendas. In: Lacasa A, Guerrero MM, Oncina M, Mora JA (eds) Desinfección de suelos en invernaderos de pimiento. Publicaciones de la Consejería de Agricultura, Agua y Medio Ambiente, Región de Murcia. pp 208–238
Guerrero MM, Ros C, Martínez MA, Barceló N, Martínez MC, Guirao P, Bello A, Contreras J, Lacasa A (2004b) Estabilidad en la eficacia desinfectante de la biofumigación con solarización en cultivos de pimiento. Acta Hortic 42:20–24
Guerrero MM, Ros C, Martínez MA, Martínez MC, Bello A, Lacasa A (2006) Biofumigation vs biofumigation plus solarization to control Meloidogyne incognita in sweet pepper. IOBC-WPRS Bulletin 29:313–318. In: Castañé C, Sánchez JA (eds) Proceedings of the meeting at Murcia, 14–18 May 2006. ISBN 92-9067-187-2. Working group “Integrated Control in Protected Crops, Mediterranean Climate”. International organisation of biological control of noxious animals and plants – west palaearctic regional section. 379 pp. http://www.iobc-wprs.org/pub/bulletins/index.html
Guerrero MM, Ros C, Lacasa C, Martínez V, Lacasa A, Fernández P, Martínez MA, Núñez M, Larregla S, Díez-Rojo MA, Bello A (2010) Effect of biosolarization using pellets of Brassica carinata on soilborne pathogens in protected pepper crops. Acta Hortic 883:337–344
Guerrero MM, Lacasa-Martínez CM, Hernández-Piñera A, Martínez-Alarcon V, Lacasa-Plasencia A (2013) Evaluation of repeated biodisinfestation using Brassica carinata pellets to control Meloidogyne incognita in protected pepper crops. Span J Agric Res 11(2):485–493
Guerrero MM, Guirao P, Martínez MC, Tello J, Lacasa A (2014) Soil fatigue and its specificity towards pepper plants in greenhouses. Span J Agric Res 12:644–652
Hoitink HAJ, Boehm MJ (1999) Biocontrol within the context of soil microbial communities: a substrate-dependent phenomenon. Annu Rev Phytopathol 37:427–446
Hoitink HAJ, Stone AG, Han DY (1997) Suppression of plant diseases by composts. HortSci 32:184–187
Katan J, Vanacher A (1990) Soil and crop health following soil disinfestation. In: Giraud M, Faure J (eds) Fatigue de sol. Méthode de diagnostic en verger et en fraiseraie. Infos, Paris, pp 21–26
Kirchmann H, Witter E (1989) Ammonia volatilization during aerobic and anaerobic manure decomposition. Plant Soil 115:35–41
Kirkegaard JA, Matthiessen JN (2004) Developing and refining the biofumigation concept. Agroindustria 3:233–239
Kirkegaard JA, Gardner PA, Desmarchelier JM, Angus JF (1993) Biofumigation: using Brassica species to control pests and diseases in horticulture and agriculture. In: Wratten N, Mailer R (eds) Proceedings of the 9th Australian Research Assembly on Brassicas. NSW Agric, Wagga Wagga, pp 77–82
Kroodsma W, Huis in’t Veld JWH, Scholtens R (1993) Ammonia emission and its reduction from cubicle houses by flushing. Livest Prod Sci 35:293–302
Lacasa A, Guirao P (1997) Investigaciones actuales sobre alternativas al uso del bromuro de metilo en pimiento en invernaderos del campo de Cartagena. In: López A, Mora JA (eds) Posibilidades de alternativas viables al bromuro de metilo en el pimiento de invernadero. Publicaciones de la Consejería de Agricultura, Agua y Medio Ambiente, Región de Murcia, Jornadas 11, pp 21–36
Lacasa CM, Guerrero MM, Ros C, Martínez V, Lacasa A, Fernández P, Martínez MA, Núñez M, Larregla S, Díez-Rojo MA, Bello A (2010) Efficacy of biosolarization with sugar beet vinasses for soil disinfestation in pepper greenhouses. Acta Hortic 883:345–352
Larkin RP, Griffin TS (2007) Control of soilborne potato diseases using Brassica green manures. Crop Prot 26:1067–1077
Larregla S (2003) Etiología y epidemiología de la “Tristeza” del pimiento en Bizkaia: su control. PhD thesis, Universidad del País Vasco, Bizkaia, 756 pp
Lazarovits G (2001) Management of soilborne plant pathogens with organic soil amendments: a disease control strategy salvaged from the past. Can J Plant Pathol 23:1–7
Lazzeri L, Manici LM (2000) The glucosinolate-myrosinase system: a natural and practical tool for biofumigation. Acta Hortic 532:89–95
Lazzeri L et al (2010) Use of seed flour as soil pesticide. Patent number US7749549B2
Leoni C, Ghini R (2006) Sewage sludge effect on management of Phytophthora nicotianae in citrus. Crop Prot 25:10–22
Litterick AM, Harrier L, Wallace P, Watson CA, Wood M (2004) The role of uncomposted materials, composts, manures, and compost extracts in reducing pest and disease incidence and severity in sustainable temperate agricultural and horticultural crop production – a review. Crit Rev Plant Sci 23:453–479
Liu B, Gumpertz ML, Hu S, Ristaino JB (2008) Effect of prior tillage and soil fertility amendments on dispersal of Phytophthora capsici and infection of pepper. Eur J Plant Pathol 120:273–287
Lockwood JL (1988) Evolution of concepts associated with soilborne plant pathogens. Annu Rev Phytopathol 26:93–121
Mandal A, Patra AK, Singh D, Swarup A, Masto RE (2007) Effect of long-term application of manure and fertilizer on biological and biochemical activities in soil during crop development stages. Bioresour Technol 98:3585–3592
Manici LM, Ciavatta C, Kelderer M, Erschbaumer G (2003) Replant problems in South Tyrol: role of fungal pathogens and microbial population in conventional and organic apple orchards. Plant Soil 256:315–324
Martínez MA, Lacasa A, Tello J (2009) Ecología de la microbiota fúngica de los suelos de los invernaderos de pimiento y su interés agronómico. Ministerio de Medio Ambiente, Madrid, 374 pp
Martínez MA, Martínez MC, Bielza P, Tello J, Lacasa A (2011a) Effect of biofumigation with manure amendments and repeated biosolarization on Fusarium density in pepper crops. J Ind Microbiol Biotechnol 38:3–11
Martínez MA, Martínez MC, Torres J, Tello J, Lacasa A (2011b) Long-term effects of the application of organic amendments on soil fungal communities in pepper crops. Bull IOBC/WPRS 71:81–84
Matthiessen JN, Kirkegard JA (2006) Biofumigation and enahanced biodegradation: opportunity and challenge in soilborne pest and disease management. Crit Rev Plant Sci 25:235–265
Mazzola M, Manici LM (2012) Apple replant disease: role of microbial ecology in cause and control. Annu Rev Phytopathol 50:45–65
Momma N (2008) Biological soil disinfestation (BSD) of soilborne pathogens and its possible mechanisms. Jpn Agric Res Q 42:7–12
Moral R, Paredes C, Bustamante MA, Marhuenda-Egea F, Bernal MP (2009) Utilisation of manure composts by high-value crops: safety and environmental challenges. Bioresour Technol 100:5454–5460
Núñez-Zofío M, Garbisu C, Larregla S (2010) Application of organic amendments followed by plastic mulching for the control of Phytophthora root rot of pepper in northern Spain. Acta Hortic 883:353–360
Núñez-Zofío M, Larregla S, Garbisu C (2011) Application of organic amendments followed by soil plastic mulching reduces the incidence of Phytophthora capsici in pepper crops under temperate climate. Crop Prot 30:1563–1572
Núñez-Zofío M, Larregla S, Garbisu C (2012) Repeated biodisinfection controls the incidence of Phytophthora root and crown rot of pepper while improving soil quality. Span J Agric Res 10(3):794–805
Oka Y (2010) Mechanisms of nematode suppression by organic soil amendments: a review. Appl Soil Ecol 44:101–115
Oka Y, Shapira N, Fine P (2007) Control of root-knot nematodes in organic farming systems by organic amendments and soil solarization. Crop Prot 26:1556–1565
Palmieri S (2005) Biofumigation: a new technology based on the use of sulphur-containing metabolites in Brassica plants. In: Proceedings of annual meeting of the association for the advancement of industrial crops, Murcia, 17–21 Sept 2005, pp 201–213
Rietz DN, Haynes RJ (2003) Effects of irrigation-induced salinity and sodicity on soil microbial activity. Soil Biol Biochem 35:845–854
Ristaino JB, Johnston SA (1999) Ecologically based approaches to management of Phytophthora blight on bell pepper. Plant Dis 83:1080–1089
Ros M, García C, Hernández MT, Lacasa A, Fernández P, Pascual JA (2008) Effects of biosolarization as methyl bromide alternative for Meloidogyne incognita control on quality of soil under pepper. Biol Fertil Soils 45:37–44
Sachi S, Odorizzi S, Lazzeri L, Marciano P (2005) Effect of Brassica carinata seed meal treatment on the Trichoderma harzianum T39-Sclerotinia species interaction. Acta Hortic 698:289–292
Scotto La Massesse C (1983) Mesures susceptibles d’apprécier et de limiter les effets de la fatigue des sols dans le cas de reconstitution des vergers. In: INRA (ed) La fatigue des sols. Diagnostic de la fertilité dans les systèmes culturaux. INRA, Paris, pp 99–111
Smolinska U, Morra MJ, Kanudsen GR, James R (2003) Isothiocyanate produced by Brassicaceae species as inhibitors of Fusarium oxysporum. Plant Dis 87:407–412
Stapleton JJ (2000) Soil solarization in various agricultural production systems. Crop Prot 19:837–841
Stapleton JJ, Bañuelos GS (2009) Biomass crops can be used for biological disinfestation and remediation of soils and water. Calif Agric 63:41–46
Stone AG, Scheuerell SJ, Darby HM (2004) Suppression of soilborne diseases in field agricultural systems: organic matter management, cover cropping, and other cultural practices. In: Magdoff F, Weil RR (eds) Soil organic matter in sustainable agriculture. CRC Press LLC, Boca Raton, FL, pp 166–223
Tello JC, Lacasa A (1997) Problemática fitosanitaria del suelo en el cultivo del pimiento en el campo de Cartagena. In: López A, Mora JA (eds) Posibilidades de Alternativas Viables al Bromuro de Metilo en el Pimiento de Invernadero. Publicaciones de la Consejería de Agricultura, Agua y Medio Ambiente, Región de Murcia, Jornadas 11, pp 11–17
Tenuta M, Lazarovits G (2002) Ammonia and nitrous acid from nitrogenous amendments kill the microsclerotia of Verticillium dahliae. Phytopathology 92:255–264
Tenuta M, Conn KL, Lazarovits G (2002) Volatile fatty acids in liquid swine manure can kill microsclerotia of Verticillium dahliae. Phytopathology 92:548–552
Vallad GE, Cooperband LR, Goodman RM (2003) Plant foliar disease suppression mediated by composted forms of paper mill residuals exhibits molecular features of induced resistance. Physiol Mol Plant Pathol 63:65–77
Wang Q, Ma Y, Wang G, Gu Z, Sun D, An X, Chang Z (2014) Integration of biofumigation with antagonistic microorganism can control Phytophthora blight of pepper plants by regulating soil bacterial community structure. Eur J Soil Biol 61:58–67
Weerakoon DMN, Reardon CL, Paulitz TC, Izzo AD, Mazzola M (2012) Long-term suppression of Pythium abappressorium induced by Brassica juncea seed meal amendment is biologically mediated. Soil Biol Biochem 51:44–52
Zhang W, Dick WA, Hoitink HAJ (1996) Compost-induced systemic acquired resistance in cucumber pythium root rot and anthracnose. Phytopathology 86:1066–1070
Zhang W, Han DY, Dick WA, Davis KR, Hoitink HAJ (1998) Compost and compost water extract-induced systemic acquired resistance in cucumber and Arabidopsis. Phytopathology 88:450–455
Acknowledgements
This research was financially supported by the National Institute for Agricultural and Food Research and Technology (INIA) of the Spanish Ministry of Science and Innovation (projects RTA-2008-00058-C03 and RTA 2011-00005-C03) and by the Department of Environment, Territorial Planning, Agriculture and Fisheries of the Basque Government (projects CIPASAPI and BIOSOL).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Larregla, S., del Mar Guerrero, M., Mendarte, S., Lacasa, A. (2015). Biodisinfestation with Organic Amendments for Soil Fatigue and Soil-Borne Pathogens Control in Protected Pepper Crops. In: Meghvansi, M., Varma, A. (eds) Organic Amendments and Soil Suppressiveness in Plant Disease Management. Soil Biology, vol 46. Springer, Cham. https://doi.org/10.1007/978-3-319-23075-7_21
Download citation
DOI: https://doi.org/10.1007/978-3-319-23075-7_21
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-23074-0
Online ISBN: 978-3-319-23075-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)