Abstract
Aplastic anemia (AA) is characterized by diminished or absent hematopoietic precursors in the bone marrow (BM), most often due to injury to the hematopoietic stem cell. AA can be congenital but is more frequently acquired. Acquired AA preferentially affects young adults (20–25 years) and individuals over the age of 55–60 years [1]. The incidence of acquired AA is estimated at two per million annually, and occurs at higher rates in countries with increased rates of viral hepatitis [2]. Table 4.1 lists the causative agents that have been reported associated with acquired AA. However, despite numerous, diverse possible causes, from chemicals and drugs to viral, collagen vascular diseases and pregnancy, in about 70% of patients, no clear cause can be determined and the AA is considered to be idiopathic [3]. It has been suggested that immune-mediated destruction/suppression may be the underlying cause in many of the patients with idiopathic AA. It has been postulated that damage induced by chemicals, drugs, viruses, or antigens leads to lymphocyte activation resulting in destruction of BM hematopoietic cells [4]. Studies have shown that autoreactive T-lymphocytes from the BM of patients with AA can inhibit hematopoiesis when co-cultured with normal marrows [5, 6]. This inhibition may be mediated by the release of marrow-suppressing cytokines, such as interferon gamma (IFN-γ), tumor necrosis factor (TNF), and interleukin-2 [1], ultimately leading to apoptotic death of BM hematopoietic stem cells. IFN-γ may also lead to increased expression of the Fas and Fas receptor [7]. Telomeric attrition resulting in critically shortened telomeres, prompting cellular senescence or crisis, has also been considered one of the underlying causes of AA. Inherited heterozygous mutations in the genes that repair or protect telomere may limit marrow stem cell self-renewal and predispose some patients to marrow failure [8, 9].
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Overview
Acquired bone marrow failure can attribute to a number of mechanisms, such as the loss of pluripotent hematopoietic stem cells; bone marrow replacement by metastatic carcinoma, lymphoma, leukemia or fibrosis; maturation arrest as a result of nutritional/metabolic deficiency or clonal hematopoietic stem neoplasm such as myelodysplastic syndrome (MDS). BM failure due to an infiltrative process and MDS are discussed under Chapter 2, and BM failure associated with a congenital genetic predisposition under Chapter 3. This chapter will focus on Aplastic anemia and Paroxysmal nocturnal hemoglobinuria (PNH).
Aplastic Anemia
Aplastic anemia (AA) is characterized by diminished or absent hematopoietic precursors in the bone marrow (BM), most often due to injury to the hematopoietic stem cells. AA can be congenital but is more frequently acquired. Acquired AA preferentially affects young adults (20–25 years) and individuals over the age of 55–60 years [1]. The incidence of acquired AA is estimated at two per million annually, and occurs at higher rates in countries with increased rates of viral hepatitis [2]. Table 4.1 lists the causative agents that have been reported associated with acquired AA. However, despite numerous, diverse possible causes, from chemicals and drugs to viral, collagen vascular diseases and pregnancy, in about 70% of patients, the cause cannot be clearly determined and the AA is considered to be idiopathic [3]. It has been suggested that immune-mediated destruction/suppression may be the underlying cause in many of the patients with idiopathic AA. It has been postulated that damage induced by chemicals, drugs, viruses, or antigens leads to lymphocyte activation resulting in destruction of BM hematopoietic cells [4]. Studies have shown that autoreactive T-lymphocytes from the BM of patients with AA can inhibit hematopoiesis when co-cultured with normal marrows [5, 6]. This inhibition may be mediated by the release of marrow-suppressing cytokines, such as interferon gamma (IFN-γ), tumor necrosis factor (TNF), and interleukin-2 [1], ultimately leading to apoptosis of BM hematopoietic stem cells. IFN-γ may also lead to increased expressions of the Fas and Fas receptor [7]. Telomeric attrition resulting in critically shortened telomeres, prompting cellular senescence or crisis, has also been considered as one of the underlying causes of AA. Inherited heterozygous mutations in the genes that repair or protect telomere may limit marrow stem cell self-renewal and predispose some patients to marrow failure [8, 9].
In recent years, it has been increasingly recognized that acquired AA, at least in a large fraction of patients, is linked to clonal hematopoiesis [10] (see “Molecular Genetics” section below).
Clinical Features
The clinical presentation of acquired AA is variable. Disease onset is often insidious, and the initial symptoms are usually related to anemia or bleeding, and infections. Anemia is usually normocytic but occasionally may be macrocytic, and is associated with absolute reticulocytopenia. Infections are typically bacterial, including sepsis, pneumonia, and urinary tract infection. Invasive fungal infection is a common cause of death, especially in subjects with prolonged and severe neutropenia [11]. Patients usually do not have splenomegaly or lymphoadenopathy.
Serologic testing for hepatitis and other viral pathogens, such as Epstein-Barr virus (EBV), cytomegalovirus (CMV), and human immunodeficiency virus (HIV), and an evaluation for autoimmune diseases may be helpful. However, the association between onset of AA and exposure to the offending agent varies greatly, and in many patients, a cause is never identified.
The clinical outcome and management decision of acquired AA depend in part upon the severity of AA, which is based on the degrees of the cytopenia(s). Significant cytopenias are defined as HGB < 10 g/L, absolute neutrophil count (ANC) < 1.5 × 109/L, and platelets <50 × 109/L [12]. To define AA, there must be at least two of the three cytopenias reaching the above levels [12, 13]. Moderate (non-severe) AA is defined as no severe pancytopenia but shows at least two cytopenia(s) below the above mentioned ranges but with an ANC ≥0.5 × 109/L. Severe and very severe AA are defined as the presence of severe pancytopenia, of which an ANC count of 0.2–0.5 × 109/L is considered as “severe” and ANC <0.2 as “very severe.” The definitions of AA severity are shown in Table 4.2.
Morphology
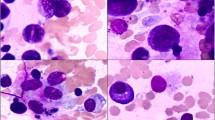
Bone marrow (BM) biopsy is required to assess BM cellularity. Non-severe (moderate) AA usually has a cellularity lower than age-appropriate cellularity, often <50% in children and <30% in adults, while severe and very severe AA have a cellularity <25%. Typical BM biopsy (Fig. 4.1) shows a profound hypocellularity with decreased trilineage hematopoiesis. Erythroid islands are generally small if present. Megakaryocytes may be difficult to visualize. In addition, there is a relative increase in small lymphocytes, plasma cells, stromal cells, and mast cells. In some patients with severe and very severe AA, the BM cellularity may be higher than 30%, but comprises predominantly of inflammatory cells rather than hematopoietic cells. The BM aspirate is often hypocellular, containing acellular spicules.
Bone marrow findings of very severe aplastic anemia (AA) (Left) versus hypoplastic MDS (Right). In AA, the bone marrow trephine biopsy is profoundly hypocellular with trilineage hypoplasia (Upper Left). The small areas of preserved cellularity comprise mainly small lymphocytes, plasma cells, and stromal cells (Middle Left). CD34 immunohistochemistry stain shows decreased or absent CD34+ hematopoietic cells (Lower Left). Hypoplastic MDS also shows a hypocellularity, but with focal clusters of hematopoietic cells (Upper Right). The aspirate smear is hypocellular, but erythroid clusters with dyserythropoiesis are present (Middle Right). CD34 immunohistochemistry stain shows easily identifiable CD34+ blasts, focally clustering (Lower Right). These morphologic features differ from those of AA
In AA, the residual hematopoietic cells are often morphologically normal, but some mild dyserythropoiesis with megaloblastoid maturation may be observed. Megakaryocytes in AA are within the normal limits morphologically, but the low number of megakaryocytes may make the evaluation of megakaryocytic dysplasia difficult; applying immunohistochemistry stains to help visualize megakaryocytes (such as CD61 and/or CD42b) may aid in revealing if the megakaryocytes appear normal or dysplastic. Morphological assessment for dysgranulopoiesis can be performed on a peripheral smear in addition to the BM aspirate.
Immunophenotype
Flow cytometric studies show that the bone marrow of AA patients has a decreased CD34+ cell count. Within the CD34+ cell compartment, there is retained differentiation to immature precursor B cells (hematogones) (Fig. 4.2). The CD34+ cells have a normal immunophenotype (Fig. 4.2). The myelomonocytic maturation analyzed by flow cytometry is often normal in AA.
Flow cytometry comparison of aplastic anemia (AA, left column) and hypoplastic myelodysplastic syndrome (MDS, right column). On CD45/SSC (a), compared to AA, MDS shows decreased side scatter of granulocytes (short arrows) and lack of hematogones (long arrows). (b) CD34+ cells are very low in number (0.08%) in AA versus a more distinct CD34+ population (0.49%) in hypoplastic MDS. (c)–(e) Within the CD34+ population (all red dots), AA shows retained stage I hematogones (arrows, about 40–50% of total CD34+ cells are CD19+ CD10+ cells) and normal expressions of CD13, CD123, and CD117 on myeloid precursors (not pointed by arrows). In contrast, hypoplastic MDS (Right) shows an absence of stage I hematogones and increased expression of CD13, CD123, and CD117 on myeloblasts. The mean florescence intensity (MFI) of each marker on immature myeloid cells is shown on the respective plot
Blood cells deficient in glycosyl phosphatidylinositol-anchored membrane proteins (GPI proteins)-paroxysmal nocturnal hemoglobinuria (PNH) cells have been reported in 30–60% AA patients, and the presence of a PNH clone can be used to support a diagnosis of AA [1, 14]. The mechanism of PNH appearance in AA likely involves an escape of PNH cells from the immune attack associated with acquired AA [15]. Interestingly, PNH virtually never arises from inherited forms of AA [16]. Thus, in children and young adults, the presence of a small PNH clone in the setting of a hypocellular BM virtually excludes a diagnosis of an inherited BM failure disorder.
The PNH clones are frequently below 1%, and in the majority, <10% of the respective cell lineage cells analyzed. Since most of these clones are small, they do not lead to clinical manifestations of hemolysis or thrombosis. The presence of a minor population of PNH-type cells in AA correlates with a positive immunosuppressive therapy response and a favorable prognosis [17]. The detection of a small PNH clone relies on a high-sensitivity flow cytometric analytic method (please see the details under PNH disease). FLAER (fluorochrome-conjugated version of a non-lysing mutated form of proaerolysin) [18] assay is the recommended test in the detection of small PNH clones (Fig. 4.3). Analysis should be performed on granulocytes and monocytes, which are generally more sensitive than red blood cells in detecting a small PNH clone. To define PNH+ cells, GPI-deficient cells should be found in ≥2 different cell lineages at a frequency of >0.01% of all cells [19, 20].
Flow cytometry assay for paroxysmal nocturnal hemoglobinuria (PNH). A lineage marker is required to define (gate) the cell population for analysis. The left column shows CD15 for neutrophils, CD64 for monocytes, and CD235a (glycophorin) for red blood cell (RBC) gating. Leukocytes are better than RBCs for the detection of small PNH clones. In this case, 61.93% granulocytes, 58.83% of monocytes and 5.695% of RBCs completely lack GPI-linked proteins (PNH type III cells). In addition, 0.063% of RBCs show partial loss of GPI proteins (PNH type II cells). Type II PNH cells are not seen in the granulocytes and monocytes
It is noteworthy that classic PNH can be associated with marrow failure (the so-called aplastic anemia/PNH syndrome), and all PNH patients show evidence of underlying hematopoietic deficiency [21]. It has been suggested that some HLA-dependent immune mechanism plays an important role in the occurrence or selection of a PNH clone and GPI itself may be a target for cytotoxic T lymphocytes [22].
In AA patients treated with immunosuppressive therapy, the PNH clones may reduce in size (Fig. 4.4), persist, or expand.
Aplastic anemia with a paroxysmal nocturnal hemoglobinuria (PNH) clone, pre- and post treatment. A patient with severe aplastic anemia at the time of diagnosis (Left side figures) showed a profound marrow hypocellularity and a large PNH clone (arrows) in granulocytes (33.27%), monocytes (Type III = 33.11%, Type II = 7.23%), and RBCs (Type III = 1.928%, Type II = 1.107%). After immunosuppressive therapy (Right side figures), the bone marrow recovered trilineage hematopoiesis and a normal cellularity. The PNH clone decreased to 4.5% in granulocytes and 7.3% in monocytes, but the PNH clone relatively increased in RBCs, likely due to no longer requiring blood transfusions
Cytogenetics and Molecular Genetics
In recent years, with the advance of next-generation sequencing (NGS) technology, clonal hematopoiesis has been found in nearly half of AA patients [23]. Healthy individuals are now known to accumulate structural genomic rearrangements and somatic mutations as a part of normal aging mutations [24]; in AA, these preexisting age-related somatic mutations are expanded through autoimmune selection and hematopoietic stress. It has been shown that aging-related mutations in DNMT3A, TET2, and spliceosome factor genes are more frequent and occur at a higher mutant allele fraction in older AA patients than younger AA patients [23, 25]. Additionally, mutations in ASXL1 and BCOR/BCORL1 [23, 26] are significantly overrepresented in AA compared to their relative prevalence in similarly aged individuals, outnumbering DNMT3A and TET2. Notably, in all age groups of AA patients, mutations in PIGA and loss of HLA class I alleles (such as copy number neutral loss of heterozygosity on chromosome 6p: 6p CN-LOH), two prototypical markers of immune escape in AA, are the most prevalent [27, 28]. It is likely that genetic mutations in hematopoietic stem cells confer a selective advantage in the AA BM environment.
The implications of age- and MDS-related somatic mutations in AA are complex and require a cautious interpretation. The detection of a PNH clone, or loss of HLA alleles through 6p CN-LOH or somatic inactivation, supports a diagnosis of AA. Patients with PIGA, BCOR, or BCORL1 mutations are found to have an improved response to immunosuppressive therapy and therefore prognostically favorable [23]. DNMT3A, ASXL1, TP53, RUNX1, and PRM1D have been reported to be associated with a shorter overall survival [23], particularly in patients <60 years of age, whereas the significance of other age-related somatic mutations is less clear.
Bone marrow cytogenetic abnormalities can occur infrequently at the time of AA diagnosis, with an incidence of approximately 4–5% [29,30,31,32]. The most common abnormalities are del(6), del(5q), del(13), del(20q), -7, and +6, which may persist or disappear with hematopoietic recovery after treatment. Except for +6 which often shows no response to immunosuppressive therapy [31, 32], AA with other clonal chromosomal abnormalities and AA with a normal karyotype show a similar response to immunosuppressive therapies and have a comparable risk for MDS or AML progression. Therefore, the presence of clonal cytogenetic abnormalities should not be used as presumptive evidence of MDS in the absence of other features of MDS. Cytogenetic abnormality detected in AA at MDS evolution include -7, 11q23 abnormalities, and chromosomal 9 abnormalities [33, 34]. Unlike a cytogenetic abnormality present at diagnosis, the emergence of a new clonal karyotypic abnormality in a patient with AA generally heralds MDS progression.
Clonal evolution occurs in about 10–20% of AA patients; in other cases, the disease may be stable for many years without progression. The incidence of clonal evolution increases substantially among patients who are successfully treated by immunosuppressive therapy and hematopoietic growth factors [6, 33, 35,36,37]. AA can progress to PNH, MDS, or AML.
Approximately half of patients with AA have a detectable PNH clone (see “Immunophenotyic Studies” section above) that can expand during immunosuppressive therapy, and these patients may have increased vulnerability to complement-mediated hemolysis. Progression to clinically significant PNH has been described in 15–25% of the treated AA patients [16, 38, 39]. The evolution rate of AA to MDS is estimated at 5–15%.
Differential Diagnosis
The main differential diagnosis of AA is hypoplastic/hypocellular MDS; in some cases, it is virtually impossible to separate these entities clinically and morphologically. The presence of mild dyserythropoiesis is common in AA, and should not be used to exclude an AA diagnosis. A number of subtle bone marrow findings may assist in the differential diagnosis of AA versus hypoplastic MDS (Table 4.3). Accurate evaluation of the blast percentage and presence of myeloid and megakaryocytic dysplasia in a severely hypoplastic marrow can be particularly challenging [38]. A CD34 stain by immunohistochemistry often reveals decreased CD34+ cells in AA [40] (Fig. 4.1) while they are normal or increased in hypocellular MDS (Fig. 4.1). P53 immunohistochemistry should not show strong-expressing cells in AA (<1%), but can be positive in a subset of MDS. Age-related mutations are very common in AA and therefore cannot be used to reliably differentiate AA from MDS.
Determining MDS evolution in AA patients who are treated with immunosuppressive therapy can be even more challenging. Notably, AA patients who are treated with immunosuppressive therapy may recover the BM cellularity, but frequently remain cytopenic; and when a complete clinical response is obtained, recurrence is common. MDS evolution in AA is characterized by a diffuse or patchy increase in bone marrow cellularity (27% of cases), while a continued hypocellularity is found in one-third of the patients with MDS evolution. Treated AA often displays mild-to-moderate dysplasia, especially in the erythroid lineage. Certain drugs that may be used in AA patients, such as antimetabolites, and superimposed viral infections can also produce BM dyspoietic changes that can complicate the evaluation for an evolving MDS.
A diagnosis of MDS evolution in AA can only be made by combining clinical, laboratory, BM morphology, cytogenetic, and immunophenotypic data. The following findings in this setting are considered as highly suggestive of evolution to MDS: (1) increased BM or peripheral blood blasts; (2) marked dysplasia in the granulocytic or megakaryocytic lineage; or (3) a newly emerging MDS-related clonal cytogenetic abnormality. As for mutations, in treated patients, clonal hematopoiesis can be detected by NGS in about 85% of patients with adult-onset AA and in over 60% of patients with pediatric-onset AA. It is debatable if the presence of unfavorable somatic mutations, such as DNMT3A, ASXL1, TP53, RUNX1, and PRM1D, in patients with persistent cytopenia but lack of other criteria of MDS is sufficient to diagnose a MDS evolution in AA patients.
Flow cytometry immunophenotyping can be particularly useful in this setting. While identification of a PNH clone is common in AA patients and supports an AA diagnosis, a PNH clone, often detected at a lower level (<1%), has been reported in about 10% of MDS cases without excess blasts. MDS cases with a PNH clone often show dysplasia limited to erythroid lineage cells; with little or no dysplasia in myeloid and megakaryocytic lineages [41]. Flow cytometry immunophenotying of CD34+ cells is a very useful method to determine a neoplastic versus nonneoplastic process in this setting (Fig. 4.2). On flow cytometry study of bone marrow from AA patients, CD34+ blasts are often very low in number, do not form a discrete population, and show retained hematogones (normal immature B-cell precursors), and lack immunophenotypic aberrancies. In contrast, hypoplastic MDS or MDS evolving from AA often show a discrete myeloblast population, which demonstrates various immunophenotypic aberrancies [42]. The most common findings include loss of hematogones; altered expression level of CD13 (increased), CD38 (decreased), CD117 (increased), and/or CD123 (increased); or aberrant expression of lymphoid antigens. The identification of immunophenotypic aberrancies in blasts is highly suggestive of a diagnosis of MDS rather than AA.
Idiopathic AA must also be distinguished from transient myelosuppression caused by drug, toxin, or infection. In transient cytopenia(s) with a hypocellular marrow, interrogation of the clinical history is critical in identifying the causative agent; in some cases, the cytopenias resolve spontaneously without identifying an underlying cause, which may be an unrecognized viral infection. In order to exclude the possibility of a transient hypoplastic process due to an exogenous factor, it is recommended that an AA diagnosis should only be considered if cytopenias are persistent and the clinical and laboratory work-up is extensive and thorough but unrevealing.
Other conditions that can cause a profound hypocellular BM include anorexia nervosa or prolonged starvation, mycobacterial infection, and lymphomas. The differential diagnosis of AA with hereditary bone marrow failure syndromes and with pediatric MDS (refractory cytopenia of childhood) is discussed separately in Chap. 3.
Paroxysmal Nocturnal Hemoglobinuria (PNH)
Paroxysmal nocturnal hemoglobinuria (PNH) has long been classified as acquired hemolytic anemia, but it is now recognized as a stem cell disorder due to an acquired mutation in the PIGA gene that is located on the X chromosome. PIGA is involved in the synthesis of the glycosylphosphatidylinositol (GPI) anchor proteins that link dozens of cell-surface proteins to the plasma membrane on hematopoietic cells. GPI-link protein CD55 (decay-accelerating factor) prevents the formation of C3 convertases in the complement cascade, and CD59 inhibits assembly of the membrane attack complex of complement. Deficiency in these proteins on red cell surface is responsible for complement sensitivity of red cells in PNH. Classical PNH is characterized by complement-mediated intravascular hemolysis, as well as extravascular hemolysis. The latter is due to complement-mediated destruction of red blood cells by reticuloendothelial macrophages in the liver and spleen. PNH can arise de novo or in the setting of an underlying bone marrow disorder such as aplastic anemia (AA). The reported incidence of clinically significant PNH is 1–10 cases per million population [43].
In recent years, the clinical heterogeneity of PNH has been increasingly recognized. While classic PNH manifests complement-mediated hemolysis, in other patients, bone marrow failure dominates the clinical picture with modest or even no evidence of hemolysis. This clinical heterogeneity likely reflects the close relationship between PNH and immune-mediated bone marrow failure, and the fact that PNH is an acquired, clonal disease of the hematopoietic stem cells. Bone marrow failure complicates the management of PNH, because the intrinsically defective hematopoiesis contributes at varying degrees of anemia and other cytopenias; in addition, the extent to which the mutant stem cell clone expands in an individual patient determines the magnitude of the hemolytic component of the disease. The International PNH Interest Group (I-PIG) has proposed a working diagnostic classification that includes the following three categories: (1) classic PNH; (2) PNH in the context of another bone marrow disorder; and (3) subclinical PNH. The definitions, clinical and laboratory features, bone marrow findings, and PNH clone size of these three groups are summarized in Table 4.4.
Clinical Features
PNH is mostly a disease of adults, with the median age of onset in the 30s, and occurs equally in men and women [44, 45]. From the data from the international PNH registry [44], frequently reported symptoms include fatigue (80%), dyspnea (64%), hemoglobinuria (62%), abdominal pain (44%), and chest pain (33%).
Patients with classic PNH present with unexplained hemolytic anemia with jaundice, and red or pink urine. Laboratory workup shows anemia, increased lactate dehydrogenase (LDH) and bilirubin, decreased haptoglobin with a negative direct antiglobulin (Coombs) test, and an increased reticulocyte count, consistent with hemolysis. Thrombosis, mainly venous, occurs in about 5% of patients at the time of diagnosis, but the incidence of thrombotic events is as high as 40% over the entire course of disease. Thrombosis can involve the hepatic vein, inferior vena cava, and portal or splenic veins, and is the leading cause of death in patients with PNH. Patients with classic PNH have preserved overall bone marrow function and mainly present with anemia. However, many cases of PNH have some degree of bone marrow failure that either exacerbates the anemia or leads to pancytopenia. One study consisting of 220 PNH patients reported a 15% incidence of pancytopenia at the 8 years of follow-up [45]. Some of these patients may belong to the group “PNH in the context of another bone marrow disorder,” who show evidence of not only hemolysis but also another primary BM abnormality such as AA, MDS, or primary myelofibrosis (PMF) (Table 4.4).
“Subclinical PNH” is defined by the presence of a small population of PNH cells, without clinical or laboratory evidence of hemolysis, often in association with AA or MDS. It may be also observed in some patients with cytopenia(s) but not filling the criteria for AA or MDS. As discussed above, a PNH clone is identified in up to 60% of AA patients and in about 10% of MDS without excess blasts. Over time, the PNH clone size in AA may increase, and a subset of patients may develop classic PNH [46].
Morphology
In classic PNH where there is significant hemolysis, PB smears often show anisopoikilocytosis, increased polychromatic cells, and some nucleated RBC, features of intravascular and extravascular hemolysis. In patients who develop BM failure, PB often shows pancytopenia.
Bone marrow examination is not required for a diagnosis of PNH, but should be performed in patients with significant cytopenia(s) other than anemia. The BM in classic PNH (Fig. 4.5) is usually normocellular or slightly hypercellular with erythroid hyperplasia due to the intravascular and extravascular hemolysis. Mild dyserythropoiesis is common as a manifestation of “stress erythropoiesis.” Stainable iron is often absent. Of patients with PNH in the context of another bone marrow disorder, the most commonly associated disease is AA, followed by MDS, and with rare cases of primary myelofibrosis [47] reported. Similarly, patients with subclinical PNH mostly have underlying AA and less frequently MDS. The diagnosis of these entities in patients with PNH clones should follow the standard diagnostic approach for AA and MDS.
Classic paroxysmal nocturnal hemoglobinuria (PNH). The bone marrow biopsy shows a marked hypercellularity (Top Figure) with normal megakaryocytes and erythroid hyperplasia (Middle Figure). The bone marrow aspirate smear shows erythroid predominance with dyspoietic changes (“stress erythropoiesis) secondary to hemolysis (Bottom Figure)
Immunophenotype
Flow cytometry to detect populations of GPI-deficient cells is the method of choice for diagnosis and monitoring PNH. A consensus guideline on screening criteria, reagents, methodology, and data analysis has been published by the International Clinical Cytometry Society (ICCS) [20]. The recommendation guidelines can be summarized as follows: (1) since PNH flow cytometry testing is to detect cells that are negative for GPI proteins, a lineage marker that is not GPI-anchored protein is required to define the population being analyzed. For example, CD15 is recommended to use for granulocytes, CD64 for monocytes, and CD235a (glycophorin) for red blood cells; (2) assessment of PNH populations in leukocytes is the best method to determine the size of a PNH clone. Neutrophils and monocytes should be assessed separately; (3) FLAER, a fluorochrome-conjugated inactive variant of aerolysin, which binds specifically to GPI anchors, is a better reagent than other individual antibodies for the detection of the PNH phenotype in neutrophils and monocytes. In addition to FLAER, assessing another GPI-linked protein is recommended to increase the reliability. The recommended markers are shown in Table 4.5. (4) Testing of red blood cells alone is not adequate for the evaluation of PNH patients, because hemolysis and transfusion may greatly underestimate the size of the PNH clone. However, if a large PNH clone is identified in leukocytes, RBC analysis is required to assess the severity of hemolysis by measuring cells lacking the expression of GPI-anchored proteins (PNH type III and type II cells, see below). (5) In order to detect a small PNH clone, the assay should reach 0.01% sensitivity. The flow cytometry test is illustrated in Fig. 4.3.
CD55 and CD59 are GPI proteins on RBCs. CD59 is often brighter than CD55, and is more closely associated with intravascular hemolysis. Three types of PNH cells have been described according to the abundance of GPI-anchored proteins on the cell surface. PNH type I cells have a normal level of GPI protein expressions; PNH type II cells show a low level (partial) of GPI protein expression and PNH type III cells show absence of GPI-linked proteins. PNH type II cells may be shown as a distinct population or a “transitional” population between PNH type I and PNH type III cells (Figs. 4.3 and 4.4). PNH type II cells may be due to a mutation in the PIGA gene [48] causing limited rather than completely abolished production of GPI-linked proteins. In general, the degree of hemolysis correlates with the size of the erythrocyte PNH clone; however, patients with a large percentage of type II RBCs often have less hemolysis than patients with mostly or exclusively type III RBCs. Type II and type III PNH cells can be determined in leukocytes as well (Fig. 4.4). Figure 4.6 shows an algorithm of flow cytometry immunophenotyping studies of PNH phenotype cells.
Cytogenetics and Molecular Genetics
Acquired mutations in PIGA gene, which is located on the X chromosome, occur in early hematopoietic stem cells. These acquired PIGA mutations show a large spectrum of involved loci, spreading throughout the entire coding region of the PIGA gene, without any specific mutational “hot spot” [49, 50]. The majority are frame-shift mutations that create a premature stop codon, resulting in a truncated protein product that is unstable and rapidly degraded. Interestingly, rare circulating blood cells with PIGA mutations and a PNH immunophenotype have been detected in healthy blood donors [51]. These mutations are polyclonal and do not involve lymphocytes, indicating that the mutations occur in the hematopoietic cells. In contrast to patients with classic PNH and/or small PNH clones associated with AA/MDS, the PIGA mutations in healthy individuals appear to arise in hematopoietic precursor cells that lack the capacity for self-renewal, rather than in hematopoietic stem cells or earlier progenitor cells [52]. In PNH secondary to AA and MDS, the hematopoietic stem cell carrying a PIGA mutation can undergo clonal expansion due to a number of reasons, including immune escape, survival advantage, and clonal evolution.
Differential Diagnosis
Coombs-negative hemolytic anemia can mimic PNH, and may be caused by (1) hereditary red blood cell membrane or enzymatic defects, such as hereditary spherocytosis, sickle cell anemia, glucose-6-phosphate dehydrogenase deficiency, and pyruvate kinase deficiency; (2) drug- or toxin-induced hemolysis; (3) paroxysmal cold hemoglobinuria due to the presence of cold-reacting IgG antibodies; (4) microangiopathic hemolytic anemia; and (5) rarely autoimmune hemolysis, due to excessive hemolysis of antibody-coated cells that prevent their detection by the Coombs test. Unlike PNH, these conditions do not have PNH-type cells and are associated with characteristic red blood cell morphologies on the peripheral blood smear and other distinctive clinical features.
Thrombosis caused by an inherited or acquired hypercoagulable state can mimic the thrombosis associated with PNH. A variety of conditions can cause abdominal and cerebral venous thrombosis. Inherited conditions include factor V Leiden, prothrombin gene mutation, and protein S and C deficiency. Acquired conditions include myeloproliferative neoplasms, solid tumors associated with hypercoagulability, inflammatory diseases, compression of blood vessels, and antiphospholipid syndrome. Thrombosis in PNH is usually associated with evidence of intravascular hemolysis and often has a large population of PNH-type cells detected by flow cytometry.
Finally, as mentioned above, PNH and other bone marrow failure syndromes or myeloid neoplasms can coexist, and a diagnosis of one does not eliminate the possibility of the other. Therefore, if there is evidence of hemolysis, it is important to follow the standard criteria for PNH screening in order not to miss the diagnosis of PNH whenever it may occur together with other disease processes.
References
Maciejewski JP, et al. Distinct clinical outcomes for cytogenetic abnormalities evolving from aplastic anemia. Blood. 2002;99(9):3129–35.
Nathan DG, Oski FA. Hematology of infancy and childhood. Philadelphia: WB Saunders; 2009.
Fuhrer M, et al. Immunosuppressive therapy for aplastic anemia in children: a more severe disease predicts better survival. Blood. 2005;106(6):2102–4.
Young NS, Maciejewski J. The pathophysiology of acquired aplastic anemia. N Engl J Med. 1997;336(19):1365–72.
Shimamura A, Guinan EA. Acquired aplastic anemia. In: Nathan D, Orkin SH, editors. Hematology of infancy and childhood. Philadelphia: WB Saunders; 2003. p. 256.
Young NS. Acquired aplastic anemia. Ann Intern Med. 2002;136(7):534–46.
Maciejewski JP, et al. Increased expression of Fas antigen on bone marrow CD34+ cells of patients with aplastic anaemia. Br J Haematol. 1995;91(1):245–52.
Ly H, et al. Functional characterization of telomerase RNA variants found in patients with hematologic disorders. Blood. 2005;105(6):2332–9.
Yamaguchi H, et al. Mutations in TERT, the gene for telomerase reverse transcriptase, in aplastic anemia. N Engl J Med. 2005;352(14):1413–24.
Stanley N, Olson TS, Babushok DV. Recent advances in understanding clonal haematopoiesis in aplastic anaemia. Br J Haematol. 2017;177:509.
Torres HA, et al. Infections in patients with aplastic anemia: experience at a tertiary care cancer center. Cancer. 2003;98(1):86–93.
Killick SB, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol. 2016;172(2):187–207.
Marsh JC, et al. Guidelines for the diagnosis and management of aplastic anaemia. Br J Haematol. 2009;147(1):43–70.
Nakao S, Sugimori C, Yamazaki H. Clinical significance of a small population of paroxysmal nocturnal hemoglobinuria-type cells in the management of bone marrow failure. Int J Hematol. 2006;84(2):118–22.
Young NS, Calado RT, Scheinberg P. Current concepts in the pathophysiology and treatment of aplastic anemia. Blood. 2006;108(8):2509–19.
de Planque MM, et al. Long-term follow-up of severe aplastic anaemia patients treated with antithymocyte globulin. Severe Aplastic Anaemia Working Party of the European Cooperative Group for Bone Marrow Transplantation (EBMT). Br J Haematol. 1989;73(1):121–6.
Sugimori C, et al. Minor population of CD55-CD59- blood cells predicts response to immunosuppressive therapy and prognosis in patients with aplastic anemia. Blood. 2006;107(4):1308–14.
Sutherland DR, et al. Diagnosing PNH with FLAER and multiparameter flow cytometry. Cytometry B Clin Cytom. 2007;72(3):167–77.
Morado M, et al. Diagnostic screening of paroxysmal nocturnal hemoglobinuria: prospective multicentric evaluation of the current medical indications. Cytometry B Clin Cytom. 2016;92:361.
Borowitz MJ, et al. Guidelines for the diagnosis and monitoring of paroxysmal nocturnal hemoglobinuria and related disorders by flow cytometry. Cytometry B Clin Cytom. 2010;78(4):211–30.
Shichishima T, Noji H. A new aspect of the molecular pathogenesis of paroxysmal nocturnal hemoglobinuria. Hematology (Amsterdam, Netherlands). 2002;7(4):211–27.
Young NS. Paroxysmal nocturnal hemoglobinuria: current issues in pathophysiology and treatment. Curr Hematol Rep. 2005;4(2):103–9.
Yoshizato T, et al. Somatic mutations and clonal hematopoiesis in aplastic anemia. N Engl J Med. 2015;373(1):35–47.
McKerrell T, et al. Leukemia-associated somatic mutations drive distinct patterns of age-related clonal hemopoiesis. Cell Rep. 2015;10(8):1239–45.
Young NS, Ogawa S. Somatic mutations and clonal hematopoiesis in aplastic anemia. N Engl J Med. 2015;373(17):1675–6.
Kulasekararaj AG, et al. Somatic mutations identify a subgroup of aplastic anemia patients who progress to myelodysplastic syndrome. Blood. 2014;124(17):2698–704.
Inaguma Y, et al. Induction of HLA-B*40:02-restricted T cells possessing cytotoxic and suppressive functions against haematopoietic progenitor cells from a patient with severe aplastic anaemia. Br J Haematol. 2016;172(1):131–4.
Katagiri T, et al. Frequent loss of HLA alleles associated with copy number-neutral 6pLOH in acquired aplastic anemia. Blood. 2011;118(25):6601–9.
Ohga S, et al. Treatment responses of childhood aplastic anaemia with chromosomal aberrations at diagnosis. Br J Haematol. 2002;118(1):313–9.
Mikhailova N, et al. Cytogenetic abnormalities in patients with severe aplastic anemia. Haematologica. 1996;81(5):418–22.
Appelbaum FR, et al. Clonal cytogenetic abnormalities in patients with otherwise typical aplastic anemia. Exp Hematol. 1987;15(11):1134–9.
Moormeier JA, et al. Trisomy 6: a recurring cytogenetic abnormality associated with marrow hypoplasia. Blood. 1991;77(6):1397–8.
Fuhrer M, et al. Relapse and clonal disease in children with aplastic anemia (AA) after immunosuppressive therapy (IST): the SAA 94 experience. German/Austrian Pediatric Aplastic Anemia Working Group. Klin Padiatr. 1998;210(4):173–9.
Kojima S, et al. Risk factors for evolution of acquired aplastic anemia into myelodysplastic syndrome and acute myeloid leukemia after immunosuppressive therapy in children. Blood. 2002;100(3):786–90.
Yamazaki E, et al. The evidence of clonal evolution with monosomy 7 in aplastic anemia following granulocyte colony-stimulating factor using the polymerase chain reaction. Blood Cells Mol Dis. 1997;23(2):213–8.
Doney K, et al. Primary treatment of acquired aplastic anemia: outcomes with bone marrow transplantation and immunosuppressive therapy. Seattle Bone Marrow Transplant Team. Ann Intern Med. 1997;126(2):107–15.
Socie G, et al. Malignant tumors occurring after treatment of aplastic anemia. European Bone Marrow Transplantation-Severe Aplastic Anaemia Working Party. N Engl J Med. 1993;329(16):1152–7.
Frickhofen N, et al. Antithymocyte globulin with or without cyclosporin A: 11-year follow-up of a randomized trial comparing treatments of aplastic anemia. Blood. 2003;101(4):1236–42.
Rosenfeld S, et al. Antithymocyte globulin and cyclosporine for severe aplastic anemia: association between hematologic response and long-term outcome. JAMA. 2003;289(9):1130–5.
Orazi A, et al. Hypoplastic myelodysplastic syndromes can be distinguished from acquired aplastic anemia by CD34 and PCNA immunostaining of bone marrow biopsy specimens. Am J Clin Pathol. 1997;107(3):268–74.
Wang SA, et al. Detection of paroxysmal nocturnal hemoglobinuria clones in patients with myelodysplastic syndromes and related bone marrow diseases, with emphasis on diagnostic pitfalls and caveats. Haematologica. 2009;94(1):29–37.
Stetler-Stevenson M, et al. Diagnostic utility of flow cytometric immunophenotyping in myelodysplastic syndrome. Blood. 2001;98(4):979–87.
Gulbis B, et al. Epidemiology of rare anaemias in Europe. Adv Exp Med Biol. 2010;686:375–96.
Schrezenmeier H, et al. Baseline characteristics and disease burden in patients in the International Paroxysmal Nocturnal Hemoglobinuria Registry. Haematologica. 2014;99(5):922–9.
Socie G, et al. Paroxysmal nocturnal haemoglobinuria: long-term follow-up and prognostic factors. French Society of Haematology. Lancet. 1996;348(9027):573–7.
Moyo VM, et al. Natural history of paroxysmal nocturnal haemoglobinuria using modern diagnostic assays. Br J Haematol. 2004;126(1):133–8.
Parker C, et al. Diagnosis and management of paroxysmal nocturnal hemoglobinuria. Blood. 2005;106(12):3699–709.
Rollinson S, et al. Both paroxysmal nocturnal hemoglobinuria (PNH) type II cells and PNH type III cells can arise from different point mutations involving the same codon of the PIG-A gene. Blood. 1997;89(8):3069–71.
Ostendorf T, et al. Heterogeneous PIG-A mutations in different cell lineages in paroxysmal nocturnal hemoglobinuria. Blood. 1995;85(6):1640–6.
Nafa K, et al. The spectrum of somatic mutations in the PIG-A gene in paroxysmal nocturnal hemoglobinuria includes large deletions and small duplications. Blood Cells Mol Dis. 1998;24(3):370–84.
Araten DJ, et al. Clonal populations of hematopoietic cells with paroxysmal nocturnal hemoglobinuria genotype and phenotype are present in normal individuals. Proc Natl Acad Sci U S A. 1999;96(9):5209–14.
Hu R, et al. PIG-A mutations in normal hematopoiesis. Blood. 2005;105(10):3848–54.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Wang, S.A. (2018). Cytopenias: Acquired Bone Marrow Failure. In: Wang, S., Hasserjian, R. (eds) Diagnosis of Blood and Bone Marrow Disorders. Springer, Cham. https://doi.org/10.1007/978-3-319-20279-2_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-20279-2_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-20278-5
Online ISBN: 978-3-319-20279-2
eBook Packages: MedicineMedicine (R0)