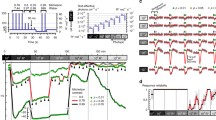
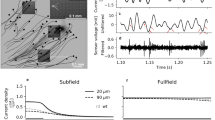
Abstract
The retina continually adapts its kinetics, average response and sensitivity to the conditions of the environment. Retinal neurons adapt essentially to the mean light intensity and its temporal fluctuations over the mean, also called temporal contrast. Contrast adaptation has two distinct temporal expressions with fast and slow components. Here, we present a configurable retina simulation environment that accurately reproduces both contrast components. A contrast increase in the visual input accelerates kinetics of the filter, reduces sensitivity and depolarizes the membrane potential. Slow adaptation does not affect the temporal response but produces a progressive hyperpolarization of membrane potential. The implemented model for contrast adaptation provides a neural basis of each retinal stage, from photoreceptors up to ganglion cells, to explain the observed retina behavior. Both forms of contrast adaptation, fast and slow, are captured by a combined model of shunting feedback of bipolar cells and short-term plasticity (STP) at the bipolar-to-ganglion synapse. Biological accuracy of the model is evaluated by comparison of the measured neural response with the simulated response fitted to published physiological data. One problem with the simulated model is finding its optimal parameter settings, since the model response is described by a complex system of different retina stages with linear, nonlinear and feedback connections. We propose to use a multiobjective genetic optimization to automatically search the parameter space and easily find a feasible configuration solution.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
References
Rieke, F., Rudd, M.E.: The challenges natural images pose for visual adaptation. Neuron 64(5), 605–616 (2009)
Demb, J.B.: Functional circuitry of visual adaptation in the retina. The Journal of Physiology 586(18), 4377–4384 (2008)
Kohn, A.: Visual adaptation: physiology, mechanisms, and functional benefits. Journal of Neurophysiology 97(5), 3155–3164 (2007)
Ozuysal, Y., Baccus, S.A.: Linking the computational structure of variance adaptation to biophysical mechanisms. Neuron 73(5), 1002–1015 (2012)
Beaudoin, D.L., Borghuis, B.G., Demb, J.B.: Cellular basis for contrast gain control over the receptive field center of mammalian retinal ganglion cells. The Journal of Neuroscience 27(10), 2636–2645 (2007)
Baccus, S.A., Meister, M.: Fast and slow contrast adaptation in retinal circuitry. Neuron 36(5), 909–919 (2002)
Kim, K.J., Rieke, F.: Temporal contrast adaptation in the input and output signals of salamander retinal ganglion cells. The Journal of Neuroscience 21(1), 287–299 (2001)
Chander, D., Chichilnisky, E.: Adaptation to temporal contrast in primate and salamander retina. The Journal of Neuroscience 21(24), 9904–9916 (2001)
Manookin, M.B., Demb, J.B.: Presynaptic mechanism for slow contrast adaptation in mammalian retinal ganglion cells. Neuron 50(3), 453–464 (2006)
Wohrer, A., Kornprobst, P.: Virtual retina: a biological retina model and simulator, with contrast gain control. Journal of Computational Neuroscience 26(2), 219–249 (2009)
Mante, V., Bonin, V., Carandini, M.: Functional mechanisms shaping lateral geniculate responses to artificial and natural stimuli. Neuron 58(4), 625–638 (2008)
van Hateren, J.V., Rüttiger, L., Sun, H., Lee, B.: Processing of natural temporal stimuli by macaque retinal ganglion cells. The Journal of Neuroscience 22(22), 9945–9960 (2002)
Victor, J.D.: The dynamics of the cat retinal x cell centre. The Journal of Physiology 386(1), 219–246 (1987)
Martínez-Cañada, P., Morillas, C., Nieves, J.L., Pino, B., Pelayo, F.: First stage of a human visual system simulator: the retina. In: Trémeau, A., Schettini, R., Tominaga, S. (eds.) CCIW 2015. LNCS, vol. 9016, pp. 118–127. Springer, Heidelberg (2015)
Rieke, F.: Temporal contrast adaptation in salamander bipolar cells. The Journal of Neuroscience 21(23), 9445–9454 (2001)
Euler, T., Haverkamp, S., Schubert, T., Baden, T.: Retinal bipolar cells: elementary building blocks of vision. Nature Reviews Neuroscience 15(8), 507–519 (2014)
Jarsky, T., Cembrowski, M., Logan, S.M., Kath, W.L., Riecke, H., Demb, J.B., Singer, J.H.: A synaptic mechanism for retinal adaptation to luminance and contrast. The Journal of Neuroscience 31(30), 11003–11015 (2011)
Dunn, F.A., Rieke, F.: Single-photon absorptions evoke synaptic depression in the retina to extend the operational range of rod vision. Neuron 57(6), 894–904 (2008)
Singer, J.H., Diamond, J.S.: Vesicle depletion and synaptic depression at a mammalian ribbon synapse. Journal of Neurophysiology 95(5), 3191–3198 (2006)
Kim, K.J., Rieke, F.: Slow na+ inactivation and variance adaptation in salamander retinal ganglion cells. The Journal of Neuroscience 23(4), 1506–1516 (2003)
Zaghloul, K.A., Boahen, K., Demb, J.B.: Contrast adaptation in subthreshold and spiking responses of mammalian y-type retinal ganglion cells. The Journal of Neuroscience 25(4), 860–868 (2005)
Zaghloul, K.A., Boahen, K., Demb, J.B.: Different circuits for on and off retinal ganglion cells cause different contrast sensitivities. The Journal of Neuroscience 23(7), 2645–2654 (2003)
Dayan, P., Abbott, L.: Theoretical neuroscience: computational and mathematical modeling of neural systems. Journal of Cognitive Neuroscience 15(1), 154–155 (2003)
Wohrer, A.: Model and large-scale simulator of a biological retina, with contrast gain control. PhD thesis, Nice (2008)
Rodieck, R.W.: Quantitative analysis of cat retinal ganglion cell response to visual stimuli. Vision Research 5(12), 583–601 (1965)
Enroth-Cugell, C., Robson, J.G.: The contrast sensitivity of retinal ganglion cells of the cat. The Journal of Physiology 187(3), 517–552 (1966)
Smith, V.C., Pokorny, J., Lee, B.B., Dacey, D.M.: Primate horizontal cell dynamics: an analysis of sensitivity regulation in the outer retina. Journal of Neurophysiology 85(2), 545–558 (2001)
Torre, V., Poggio, T.: A synaptic mechanism possibly underlying directional selectivity to motion. Proceedings of the Royal Society of London. Series B. Biological Sciences 202(1148), 409–416 (1978)
Amthor, F.R., Grzywacz, N.M.: Nonlinearity of the inhibition underlying retinal directional selectivity. Visual Neuroscience 6(03), 197–206 (1991)
Carandini, M., Heeger, D.J., Movshon, J.A.: Linearity and normalization in simple cells of the macaque primary visual cortex. The Journal of Neuroscience 17(21), 8621–8644 (1997)
Kastner, D.B., Baccus, S.A.: Coordinated dynamic encoding in the retina using opposing forms of plasticity. Nature Neuroscience 14(10), 1317–1322 (2011)
Fortin, F.-A., De Rainville, F.-M., Gardner, M.-A., Parizeau, M., Gagné, C.: DEAP: Evolutionary algorithms made easy. Journal of Machine Learning Research 13, 2171–2175 (2012)
Gewaltig, M.-O., Diesmann, M.: Nest (neural simulation tool). Scholarpedia 2(4), 1430 (2007)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this paper
Cite this paper
Martínez-Cañada, P., Morillas, C., Romero, S., Pelayo, F. (2015). Modeling Retina Adaptation with Multiobjective Parameter Fitting. In: Rojas, I., Joya, G., Catala, A. (eds) Advances in Computational Intelligence. IWANN 2015. Lecture Notes in Computer Science(), vol 9095. Springer, Cham. https://doi.org/10.1007/978-3-319-19222-2_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-19222-2_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-19221-5
Online ISBN: 978-3-319-19222-2
eBook Packages: Computer ScienceComputer Science (R0)