Abstract
Cell-based therapy has become an attractive solution to the high incidence of heart failure post-infarction. Many current approaches to cell delivery post-infarction result in poor cell engraftment resulting in limited functional benefits. Thus, the use of engineered tissues to deliver cells to the injured myocardium or replace myocardium post infarction has been a topic of increasing interest. Tissue engineering provides a platform for the delivery of a large number of cells to the injured myocardium with high retention, allowing for in vitro development of cellular organization, intracellular communication and ECM deposition. This chapter will discuss the currently used methods to create engineered cardiac tissues, including scaffolds, cells, and cellular conditioning. This chapter will also review the efficacy of these patches in limiting left ventricular remodeling post-infarction in vivo.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Tissue engineering
- Myocardial infarction
- Embryonic stem cells
- Induced pluripotent stem cells
- Endothelial cells
- Bioreactor conditioning
- Cyclic stretch
- Electrical stimulation
- Perfusion
- Biomaterials
1 Introduction
Heart failure has many causes, but the most prevalent initiator is a myocardial infarction , in which myocardial tissue is deprived of oxygen for an extended period of time, usually through a blockage of one of the coronary arteries. In the ischemia induced by an infarction, one-quarter of the 4 billion cells in the left ventricle can be lost [1]. With this level of cell death, heart failure can develop as a result of the limited capacity of the injured myocardial tissue to recover or regenerate, leading to fibrosis and scar formation of the damaged myocardium, left ventricular dilation and thinning due to a resulting pressure and volume overload , and the inhibition of proper action potential propagation. Current treatments, whether they are pharmaceutical or medical device-based, act merely as palliative measures and the only effective long term treatment to date for heart failure is to replace the damaged myocardium via total heart transplantation [2].
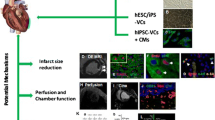
With the limited number of donors and the inherent risks of surgery and immunogenicity, cellular therapy has become an attractive solution to the high incidence of heart failure post-infarction. However, direct injection of cells into the myocardium has shown limited efficacy due to poor grafting efficiency [3]. Low rates of retention may be a product of cell loss due to inability to create focal adhesions with neighboring cells, the inflammatory response to myocardial injury, or the hypoxic environment of the infarct zone. Thus, the use of engineered cardiac tissues has been a topic of increasing interest. Tissue engineering not only provides a platform for the delivery of a large number of cells to the injured myocardium, it provides a means to replace damaged myocardium. Tissue engineering allows for in vitro development of cellular organization, intracellular communication and ECM deposition while also isolating the cells from the inflammatory infarct environment when implanted in vivo (see Fig. 1).
In this chapter, we will review the currently used methods used to create engineered cardiac tissues, or “cardiac patches”, and their efficacy when delivered in vivo. We will begin by surveying the cells, scaffold and in vitro conditioning used to create these patches, and then move on to how they are characterized, and finally their efficacy to limit left ventricular remodeling post-infarction.
2 Cells for Cardiac Tissue Engineering
2.1 Neonatal Rat Cardiomyocytes
Until functional cardiomyocytes were able to be differentiated from human pluripotent stem cell sources, primary isolates of neonatal rat cardiomyocytes were the standard cell source for the development of cardiac patches [4–6]. These cells have been well characterized in vitro and can be obtained in large numbers, making them a useful tool for developing cardiac patches and interrogating the effects of in vitro conditioning on these tissues .
2.2 Pluripotent Stem Cell Derived Cardiomyocytes
Stem cells, whether taken from an embryo or reprogrammed from an adult cell, have enormous therapeutic potential due to their ability to be expanded in an undifferentiated state, and differentiated into cells of any lineage found in the body. In their undifferentiated state, pluripotent stem cells cannot be transplanted into patients as they can form teratomas. However, as the ability to not only differentiate pluripotent stem cells into functional cardiomyocytes but also in numbers large enough to be used therapeutically has become available, these cells have become the new standard cell source for creating cardiac patches.
2.2.1 Embryonic Stem Cell Derived Cardiomyocytes
Spontaneous differentiation of embryonic stem cells (ESCs) to cardiogenic cells was first observed in mouse ESCs in 1981 when these cells were cultured in 3D aggregates, called embryoid bodies [7]. Human ESCs were first obtained from a blastocyst in 1998 [8], but it wasn’t shown until 2001 that they could successfully be differentiated into functional cardiomyocytes that exhibited contractile force generation and recordable action potentials [9]. Human ESC-CMs are now commonly used as a cell source for cardiac tissue engineering [10, 11].
2.2.2 Induced Pluripotent Stem Cell Derived Cardiomyocytes
In 2006 it was discovered that the introduction of four transcription factors (Oct3/4, Sox2, c-Myc, and Klf4) into adult murine fibroblasts reprogrammed these cells into an embryonic state, called induced pluripotent stem cells (iPSCs) [12]. This was a landmark discovery, as the reprogramming of adult cells into an embryonic state not only alleviates the ethical concerns of using embryonic cardiomyocytes but also allows for potentially autologous cell transplantation, eliminating the immunogenicity obstacle of allogeneic transplantation. Human iPSCs were reported in 2007 using the same factors or a new combination of Oct4, NANOG, LIN28, and SOX2 [13, 14], and were differentiated into functional cardiomyocytes in 2009 [15]. They have been used in parallel with hESC-CMs for tissue engineering applications, and will likely become the preferred cell source for cardiac patches for the foreseeable future [10].
2.2.3 Differentiation Methods
Initially, pluripotent stem cell derived cardiomyocytes were differentiated via the embryoid body method, where undifferentiated pluripotent stem cells are cultured in 3D aggregates on top of irradiated mouse embryonic fibroblast feeder cells and allowed to spontaneously differentiate into cells from all three embryonic germ layers, among them being cardiomyocytes. However, as can be expected, this method results in low percentages and yields of cardiomyocytes and requires purification steps in order to obtain high percentages of cardiomyocytes. In efforts to efficiently obtain high yields of cardiomyocytes from stem cell differentiation, Zhang et al developed a new method in which cells are cultured between two layers of matrigel [16], resulting in a significant increase in differentiation efficiency to cardiomyocytes. Recently, small molecule methods of reprogramming and differentiation under defined conditions have been developed to eliminate the need for viral vectors and to increase reprogramming efficiency [17].
2.3 Cardiac Progenitor Cells
The heart has a limited capacity to regenerate via the presence of sparsely distributed resident cardiac progenitor cells (CPCs) and cardiac side population (SP) cells within the myocardium, approximately one CPC for every 30,000–40,000 cells in the myocardium [18]. These cells have been isolated based on their expression of a number of cell surface markers and gene expression, including c-kit [19], Sca-1 [20], and islet-1 [21] as well as from their migration of progenitor cells out of excised cardiac tissue when cultured in vitro [22]. Cells selected through these methods display the capacity to self-renew and differentiate into cardiomyocyte as well as endothelial phenotypes and can be used to create cardiac patches [11, 23].
2.4 Non-Cardiomyocyte Cells and Co-Culture of Cells for Cardiac Patches
Cardiomyocytes are not the only cell type used to create engineered tissues for cardiac repair. Non-cardiomyocyte cells are required for the creation of cardiac patches that utilize compacting biopolymer hydrogels, as cardiomyocytes do not contract collagen and fibrin hydrogels. Despite improvements in differentiation techniques, pluripotent stem cell-derived CM populations are only 60–90 % pure CMs, with the remaining 10–40 % of cells primarily being fibroblast-like cells, though those cells remain poorly characterized. Neonatal rat cell isolates also contain between 50–60 % of a heterogeneous population of non-CM cells, consisting primarily of fibroblasts, but also containing smooth muscle cells and endothelial cells from the vasculature and other interstitial cells from the myocardium [24]. The addition or inclusion of non-CM cells into cardiac patches has proven to be beneficial. Co-culture of hESC and hiPSc-derived cardiomyocytes with endothelial cells with or without mesenchymal stromal cells has resulted in an increase in contractile force generation by the resulting cardiac patches [10], and the force generated per cardiomyocyte has shown to be higher with less pure populations of hESC-CMs [25]. Other non-CM cells used in cardiac tissue engineering include blood outgrowth endothelial cells (BOECs) [26], human umbilical endothelial cells (HUVECs) [27], pericytes (PCs) [28], and human dermal fibroblasts (HDFs) [29]. These cells are used either in combination with cardiomyocytes or by themselves in engineered tissues, all with the goal of rescuing the injured myocardium post-infarction.
3 Scaffolds for Cardiac Tissue Engineering
Cardiac patches are typically formed by the in vitro seeding and culturing cells in 3-dimensional scaffolds. The choice of scaffold influences cell survival, phenotype, and function through its physical and chemical properties, including stiffness , microstructure, and surface chemistry. Two general classes of scaffolds are utilized to create cardiac patches: prefabricated scaffolds, in which cells are seeded onto a pre-existing scaffold structure; and biopolymer scaffolds, in which cells are present during scaffold polymerization and become entrapped in a fibrous network .
3.1 Prefabricated Scaffolds
Synthetic biodegradable, elastomeric polymers including polycaprolactone (PCL) [30], poly(glycerol-sebacate) (PGS) [31], and poly(lactide-co-glycolide) (PLGA) [32] copolymers and natural materials such as collagen I [33], alginate [34], and gelatin [35] are materials that have been used to create prefabricated foam or nanofiber scaffolds. Foam scaffolds allow for control of pore size and structure through the use of different material processing techniques. Highly porous scaffolds have proven successful in creating cardiac patches with high cell viability, but they are often isotropic and do not result in cellular alignment. Nanofiber scaffolds can be created through electrospinning [36], in which fiber diameter and orientation can be controlled, mimicking organization of collagen fibers in the native myocardium. These scaffolds can be fabricated in advance and can incorporate surface modifications to enhance cell adhesion and activity, such as the conjugation of RGD peptides [23].
3.2 Biopolymer Scaffolds
Biopolymers are another class of scaffolds used in cardiac tissue engineering. These are native protein fibril networks in hydrogel form made from either collagen I or fibrin [5, 37, 38]. Rather than being seeded onto pre-formed scaffolds, cells are suspended in a gel-forming solution and fibrillogenesis occurs around the cells, entrapping them in a hydrated, fibrillar network. Initially, collagen and fibrin gels are extremely soft andisotropic. Over time, entrapped non-CM cells compact the gel, resulting in a denser fibril network. Eventually, the entrapped cells not only compact the gel, but begin to degrade the fibrils (in the case of fibrin gels) and replace them with cell-produced matrix. By constraining the compaction of the gel, fibrillar and cellular alignment can be induced in these gels. Induction of alignment allows the native architecture of the myocardium to be mimicked and has been shown to result in an increase in both contractile force generation by entrapped cardiomyocytes and the presence of gap junctions between cardiomyocytes [5]. Collagen and fibrin biopolymer scaffolds can be altered by the addition of Matrigel [10, 25, 39]. Other biopolymer scaffold materials include alginate, which can be 3D printed into various geometries [23].
3.3 Tissue-Based Scaffolds
Decellularized heart tissue has been used as a scaffold to culture cardiac cells. Heart tissues are decellularized by perfusion with SDS followed by Triton-X to remove cells while leaving most of the heart ECM content and structure intact [40]. Cells can then be reperfused into the heart [40], or entrapped in an ECM-hydrogel matrix to create cardiac patches [41].
3.4 Scaffold-Free Tissues
Scaffold-free approaches have been used to create cardiac patches in the form of cell sheets [42] and cell aggregate patches [43]. Cell sheets are created by seeding cells onto temperature-responsive membranes, allowing them to deposit cell-produced ECM and form cellular connections, and releasing them from the culture surfaces through temperature reduction. Cell sheets can then be stacked to create thin 3D tissues or layered with cell sheets of endothelial cells to facilitate rapid vascularization upon implantation [42]. Cell aggregate patches are prepared by culturing cells on a low-attachment plate placed on an orbital shaker, allowing for large numbers of cells to aggregate in solution and form tissues.
4 In vitro Cellular Conditioning
In addition to seeding cells onto or into scaffolds, external stimulation of the cells is often required to induce them to convert the scaffold into a functional cardiac tissue. This may be achieved through mechanical, electrical, or pharmacological stimulation.
4.1 Mechanical Stimulation
Cardiomyocytes in the native myocardium experience cyclic mechanical stretch as part of the cardiac cycle. Taking inspiration from this, in vitro mechanical stimulation has been widely used to stimulate alignment, hypertrophy, and maturation of cardiomyocytes in engineered tissues. Stimulation through cyclic stretching has been achieved for ring-shaped cardiac patches using two methods: looping the patch around one fixed post and another coupled to a motorized stretching device [44], or through pneumatically-controlled distension of a latex mandrel on which the patch is mounted [37]. In both instances, cyclic stretching resulted in over a two-fold increase in contractile force generation.
4.2 Electrical Stimulation
Ventricular cardiomyocytes in the heart beat under the continuous regulation of their electrical activity through the cardiac conduction system originating at the sinoatrial node. In vitro, cardiomyocytes in cardiac patches beat spontaneously, but often at an irregular and variable rate, and not in synchrony with cardiomyocytes in other regions of the patch. Electrical stimulation of cardiac patches throughout culture has resulted in increases in the maximum frequency at which these patches can be stimulated and elicit a contractile response synchronous with pacing, a reduction in the voltage threshold at which cardiomyocytes contract, increased contractile force and also stimulated cardiomyocyte hypertrophy [45].
4.3 In vitro Perfusion
Due to the high metabolic demand of cardiomyocytes, diffusion limitations hamper the ability to make the thicker cardiac patches that are necessary to achieve clinical relevance. To increase oxygen and nutrient availability to cells, different methods of in vitro perfusion have been utilized in cardiac patches. Pulsatile perfusion of cell culture medium through porous pre-formed scaffolds has proven effective in increasing cell viability and myofibril assembly through media flow [6] and activates the ERK 1/2 signaling pathway [46]. In biopolymer hydrogels, microchannels have been included in the gel [47], and perfusable microvessels have been created within the gel to allow for medium flow through the gel during culture [48–50]. Though promising, this latter approach has yet to be combined with cardiomyocyte culture in vitro.
4.4 Medium Supplementation
Supplementation of culture medium with pharmacological agents can also be used to stimulate development of cardiac patches. The addition of platelet-derived growth factor BB (PDGF-BB) to cultures of collagen/matrigel scaffolds containing neonatal rat cardiomyocytes protected cells from apoptotic death, thus increasing the final contractile performance of the patch [51]. Insulin and ascorbic acid have been used to promote matrix deposition by entrapped non-CM cells [37].
4.5 Combination Treatments
In addition to exposing cardiac patches to one form of in vitro conditioning, combinations of the above mentioned techniques have been used in combination to further condition tissues. Electrical stimulation has been used in concert with perfusion and resulted in an increase in cell elongation, enhanced expression of gap junction protein connexin-43, increase in cell number, and an increase in contractile performance [52, 53]. However, the combination of stretch and β-adrenergic stimulation resulted in no additional benefit [53].
5 Characterization of Cardiac Patch Function
One additional benefit of using engineered tissues to treat heart failure post-infarction is that these engineered tissues and the cells entrapped in them can be functionally characterized in vitro, allowing for quality control and a more thorough understanding of the benefits of cell transplantation. Functional performance of patches is primarily assessed through contractile force measurements and optical mapping of electrical conductivity.
5.1 Contractile Force Measurements
Cardiac patches should, once developed, function as a piece of cardiac muscle tissue and generate contractile stresses close to physiological values. Quantification of this contractile activity is widely used to assess the quality of a cardiac patch. Contractile activity can be assessed through the magnitude of patch contraction both spontaneously and in response to pacing, the maximum frequency by which a patch can be paced and still elicit a contractile response in phase with the electrical stimulus, the force-frequency relationship of contractile amplitude with increasing pacing frequency, and the contractile response of the patch to exposure to pharmacological agents. Such agents include β-adgrenergic agonists, gap junction blocker 1-Heptanol [54], or cholinergic agonist carbacol [39].
5.2 Optical Mapping
Electrical activity of cardiac patches can be assessed through optical mapping. To do this, tissues are submerged in medium containing a voltage sensitive dye, such as Di-4ANEPPS or ANNINE-6. Tissues are then point-stimulated and the action potential propagation is recorded by a high speed camera. This data can be analyzed to assess both conduction velocity (the rate at which action potentials propagate across the tissue), and action potential duration (the time it takes for a cell to re polarize to either 50 or 90 % of its resting potential). This data can be used to obtain the conduction velocities of action potentials across the patch, as well as the action potential duration. These values can then be compared to physiological values to assess the functional quality of the cardiac patch [55]. Additionally, calcium transients can be similarly imaged with a calcium sensitive dye [56].
6 In vivo Assessments of Cardiac Patches
Although cardiac patches can be fully characterized in vitro and can be conditioned in vitro to obtain functional values close to that of the native myocardium, cardiac patches can only truly be effective if their implantation results in the restoration of cardiac function post-infarction. Some studies have made progress towards achieving this goal, utilizing both small and large animal models, and acute or delayed implantation. The timing of patch implantation is a crucial aspect of in vivo assessments as a way to help determine the mechanisms by which any benefits provided by the patch occur. Acute transplantation is transplantation of the patch immediately after infarction, delivering it during the initial inflammatory phase before fibrosis begins to occur. Chronic, or delayed transplantation, requires a second surgery and applies the patch after the inflammatory phase has passed and either before or after mature scar formation, depending on the time of implantation.
6.1 Rodent Models
Rodent models of myocardial infarction treatments are commonly used as the first initial model due to the smaller size of patch required for treatment. Acute placement of a cardiac patch using fibrin as the scaffold and syngeneic neonatal rat cardiac cells, with in vitro stretch conditioning of the patch prior to implantation, resulted in minimal scar formation and restoration of cardiac function 4 weeks post-infarct, with 36 % cardiomyocyte retention [37]. A similar patch made with a collagen/Matrigel scaffold rather than fibrin and implanted 2 weeks post-infarction limited further scar formation and preserved cardiac function from the time of implantation [4]. Transplantation of a layered neonatal rat cell sheet patch into a nude rat infarct model 14 days post-infarction resulted in significant improvements in cardiac function, though cell retention was minimal after 4 weeks [57]. Few studies to date have been published assessing the functional consequences of a human stem cell derived cardiac patch in an infarcted rat model. Acute transplantation into non-infarcted rats of scaffold-free tissue patches utilizing hESC-CMs and HUVECs resulted in rapid vascularization of the patches in vivo. Patches containing only an enriched CM population resulted in poor CM retention [58].
6.2 Large Animal Models
The use of large animal models of myocardial infarction is a necessary step towards bringing cardiac patches to clinical relevance Few studies to date have been able to generate the large numbers of human stem cell derived cardiomyocytes required to create cardiac patches large enough to be implemented in large animal models. Human ESC-CMs in cell sheet form were transplanted into a porcine ischemia-reperfusion model 4 weeks after ligation in a feasibility and safety study, and it was found that the cell sheet treatment resulted in a reduction in LV dilation and an improvement in cardiac function both 4 and 8 weeks post-transplantation [59].
7 Current Deficiencies and Obstacles
Despite the many advances in cardiac tissue engineering , there remain deficiencies in the field and many obstacles still to be overcome. Contractile force generation and cellularity of cardiac patches average far below physiological values of 44 mN/mm2 and 20–40 M cells/gram for human ventricular myocardium and 56.4 N/mm2 rat ventricular myocardium [60]. Diffusional limitations may contribute to this, and may be overcome by the development of a microvascular network- withing the patch during its fabrication. As mentioned earlier, progress has been made, but microvascular network have yet to be created and perfused in the presence of a cardiomyocyte-containing patch of size relevant to even a rat infarct. Other issues that will be addressed in the coming years are maturation mismatch of donor cells to recipient hearts. Currently used stem cell-derived cardiomyocytes are a mixture of atrial, ventricular, and pacemaker phenotypes, and all exhibit a fetal or neonatal phenotype. Immature phenotypes also have different electrophysiological, metabolic, and contractile properties than adult CMs. It has yet to be determined if transplantation of immature cells will elicit arrhythmic events, or if cells need to be differentiated to maturity prior to transplantation. However, more mature cells may not survive transplantation with the same efficiency as less mature cells.
References
Caulfield JB, Leinbach R, Gold H (1976) The relationship of myocardial infarct size and prognosis. Circulation 53(3 Suppl):I141–144
Miniati DN, Robbins RC (2011) Heart transplantation: a thirty-year perspective. Annu Rev Med 53(1):189–205
Reinecke H, Murry CE (2002) Taking the death toll after cardiomyocyte grafting: a reminder of the importance of quantitative biology. J Mol Cell Cardiol 34(3):251–253
Zimmermann WH, Melnychenko I, Wasmeier G, Didie M, Naito H, Nixdorff U, Hess A, Budinsky L, Brune K, Michaelis B, Dhein S, Schwoerer A, Ehmke H, Eschenhagen T (2006) Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nat Med 12(4):452–458
Black LD 3rd, Meyers JD, Weinbaum JS, Shvelidze YA, Tranquillo RT (2009) Cell-induced alignment augments twitch force in fibrin gel-based engineered myocardium via gap junction modification. Tissue Eng Part A 15(10):3099–3108
Brown MA, Iyer, Rohin K., Radisic, M. (2008) Pulsatile perfusion bioreactor for cardiac tissue engineering. Biotechnol Prog 2008:907–920
Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292(5819):154–156
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282(5391):1145–1147
Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, Livne E, Binah O, Itskovitz-Eldor J, Gepstein L (2001) Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest 108(3):407–414. doi:10.1172/jci12131
Tulloch NL, Muskheli V, Razumova MV, Korte FS, Regnier M, Hauch KD, Pabon L, Reinecke H, Murry CE (2011) Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ Res 109(1):47–59
Liau B, Christoforou N, Leong KW, Bursac N (2011) Pluripotent stem cell-derived cardiac tissue patch with advanced structure and function. Biomaterials 32(35):9180–9187
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676
Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, Slukvin, II, Thomson JA (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318(5858):1917–1920
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872
Zhang J, Wilson GF, Soerens AG, Koonce CH, Yu J, Palecek SP, Thomson JA, Kamp TJ (2009) Functional cardiomyocytes derived from human induced pluripotent stem cells. Circ Res 104(4):e30–e41
Zhang J, Klos M, Wilson GF, Herman AM, Lian X, Raval KK, Barron MR, Hou L, Soerens AG, Yu J, Palecek SP, Lyons GE, Thomson JA, Herron TJ, Jalife J, Kamp TJ (2012) Extracellular matrix promotes highly efficient cardiac differentiation of human pluripotent stem cells: the matrix sandwich method. Circ Res 111(9):1125–1136
Lian X, Zhang J, Azarin SM, Zhu K, Hazeltine LB, Bao X, Hsiao C, Kamp TJ, Palecek SP (2013) Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/beta-catenin signaling under fully defined conditions. Nat Protoc 8(1):162–175
Leri A, Kajstura J, Anversa P (2005) Cardiac stem cells and mechanisms of myocardial regeneration. Physiol Rev 85:1373–1416
Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, Kasahara H, Rota M, Musso E, Urbanek K, Leri A, Kajstura J, Nadal-Ginard B, Anversa P (2003) Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114(6):763–776
Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y, Pocius J, Michael LH, Behringer RR, Garry DJ, Entman ML, Schneider MD (2003) Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci U S A 100(21):12313–12318
Laugwitz K-L, Moretti A, Lam J, Gruber P, Chen Y, Woodard S, Lin L-Z, Cai C-L, Lu MM, Reth M, Platoshyn O, Yuan JX-J, Evans S, Chien KR (2005) Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte. Nature 433(7026):647–653
Messina E, De Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MVG, Coletta M, Vivarelli E, Frati L, Cossu G, Giacomello A (2004) Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res 95(9):911–921
Gaetani R, Doevendans PA, Metz CH, Alblas J, Messina E, Giacomello A, Sluijter JP (2012) Cardiac tissue engineering using tissue printing technology and human cardiac progenitor cells. Biomaterials 33(6):1782–1790
Banerjee I, Fuseler JW, Price RL, Borg TK, Baudino TA (2007) Determination of cell types and numbers during cardiac development in the neonatal and adult rat and mouse. Am J Physiol Heart Circ Physiol 293:H1883–H1891
Zhang D, Shadrin IY, Lam J, Xian HQ, Snodgrass HR, Bursac N (2013) Tissue-engineered cardiac patch for advanced functional maturation of human ESC-derived cardiomyocytes. Biomaterials 34(23):5813–5820
Morin KT, Tranquillo RT (2011) Guided sprouting from endothelial spheroids in fibrin gels aligned by magnetic fields and cell-induced gel compaction. Biomaterials 32(26):6111–6118
Chouinard JA, Gagnon S, Couture MG, Levesque A, Vermette P (2009) Design and validation of a pulsatile perfusion bioreactor for 3D high cell density cultures. Biotechnol Bioeng 104(6):1215–1223
Stratman AN, Malotte KM, Mahan RD, Davis MJ, Davis GE (2009) Pericyte recruitment during vasculogenic tube assembly stimulates endothelial basement membrane matrix formation. Blood 114:5091–5101
Kellar RS, Landeen LK, Shepherd BR, Naughton GK, Ratcliffe A, Williams SK (2001) Scaffold-based three-dimensional human fibroblast culture provides a structural matrix that supports angiogenesis in infarcted heart tissue. Circulation 104(17):2063–2068
Yeong W, Sudarmadji N, Yu H, Chua C, Leong K, Venkatraman S, Boey Y, Tan L (2010) Porous polycaprolactone scaffold for cardiac tissue engineering fabricated by selective laser sintering. Acta Biomater 6(6):2028–2034
Engelmayr GC Jr, Cheng M, Bettinger CJ, Borenstein JT, Langer R, Freed LE (2008) Accordion-like honeycombs for tissue engineering of cardiac anisotropy. Nat Mater 7(12):1003–1010. 6
Caspi O, Lesman A, Basevitch Y, Gepstein A, Arbel G, Habib IH, Gepstein L, Levenberg S (2007) Tissue engineering of vascularized cardiac muscle from human embryonic stem cells. Circ Res 100(2):263–272
Radisic M, Euloth M, Yang L, Langer R, Freed LE, Vunjak-Novakovic G (2003) High-density seeding of myocyte cells for cardiac tissue engineering. Biotechnol Bioeng 82(4):403–414
Dar A, Shachar M, Leor J, Cohen S (2002) Optimization of cardiac cell seeding and distribution in 3D porous alginate scaffolds. Biotechnol Bioeng 80(3):305–312
Li R-K, Jia Z-Q, Weisel RD, Mickle DAG, Choi A, Yau TM (1999) Survival and function of bioengineered cardiac grafts. Circulation 100(suppl 2):II63–Ii69
Ifkovits JL, Devlin JJ, Eng G, Martens TP, Vunjak-Novakovic G, Burdick JA (2009) Biodegradable fibrous scaffolds with tunable properties formed from photo-cross-linkable poly(glycerol sebacate). ACS Appl Mater Interfaces 1(9):1878–1886
Wendel JS, Ye L, Zhang P, Tranquillo RT, Zhang JJ (2014) Functional consequences of a tissue-engineered myocardial patch for cardiac repair in a rat infarct model. Tissue Eng Part A 20(7–8):1325–1335
Boudou T, Legant WR, Mu A, Borochin MA, Thavandiran N, Radisic M, Zandstra PW, Epstein JA, Margulies KB, Chen CS (2012) A microfabricated platform to measure and manipulate the mechanics of engineered cardiac microtissues. Tissue Eng Part A 18(9–10):910–919
Zimmermann WH (2001) Tissue engineering of a differentiated cardiac muscle construct. Circ Res 90 (2):223–230
Ott HC, Matthiesen TS, Goh SK, Black LD, Kren SM, Netoff TI, Taylor DA (2008) Perfusion-decellularized matrix: using nature’s platform to engineer a bioartificial heart. Nat Med 14(2):213–221
Duan Y, Liu Z, O’Neill J, Wan LQ, Freytes DO, Vunjak-Novakovic G (2011) Hybrid gel composed of native heart matrix and collagen induces cardiac differentiation of human embryonic stem cells without supplemental growth factors. J Cardiovasc Transl Res 4(5):605–615
Sekine H, Shimizu T, Hobo K, Sekiya S, Yang J, Yamato M, Kurosawa H, Kobayashi E, Okano T (2008) Endothelial cell coculture within tissue-engineered cardiomyocyte sheets enhances neovascularization and improves cardiac function of ischemic hearts. Circulation 118(14 Suppl):S 145–S152
Stevens KR, Kreutziger KL, Dupras SK, Korte FS, Regnier M, Muskheli V, Nourse MB, Bendixen K, Reinecke H, Murry CE (2009) Physiological function and transplantation of scaffold-free and vascularized human cardiac muscle tissue. Proc Natl Acad Sci U S A 106(39):16568–16573
Fink C, Ergün S, Kralisch D, Remmers U, Weil J, Eschenhagen T (2000) Chronic stretch of engineered heart tissue induces hypertrophy and functional improvement. FASEB J 14 (5):669–679
Chiu LLY, Iyer RK, King J-P, Radisic M (2011) Biphasic electrical field stimulation aids in tissue engineering of multicell-type cardiac organoids. Tissue Eng Part A 17(11–12):1465–1477
Dvir T, Levy O, Shachar M, Granot Y, Cohen S (2007) Activation of the ERK1/2 cascade via pulsatile interstitial fluid flow promotes cardiac tissue assembly. Tissue Eng 13(9):2185–2193
Kolesky DB, Truby RL, Gladman AS, Busbee TA, Homan KA, Lewis JA (2014) 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv Mater 26(19):3124–3130
Morin KT, Dries-Devlin JL, Tranquillo RT (2014) Engineered microvessels with strong alignment and high lumen density via cell-induced fibrin gel compaction and interstitial flow. Tissue Eng Part A 20(3–4):553–565
Moya ML, Hsu YH, Lee AP, Hughes CC, George SC (2013) In vitro perfused human capillary networks. Tissue Eng Part C Methods 19(9):730–737
Raghavan S, Nelson CM, Baranski JD, Lim E, Chen CS (2010) Geometrically controlled endothelial tubulogenesis in micropatterned gels. Tissue Eng Part A 16(7):2255–2263
Vantler M, Karikkineth BC, Naito H, Tiburcy M, Didie M, Nose M, Rosenkranz S, Zimmermann WH (2010) PDGF-BB protects cardiomyocytes from apoptosis and improves contractile function of engineered heart tissue. J Mol Cell Cardiol 48(6):1316–1323
Morgan KY, Black LD (2014) Mimicking isovolumic contraction with combined electromechanical stimulation improves the development of engineered cardiac constructs. Tissue Eng Part A. 20(11–12):1654–1667. doi:10.1089/ten.TEA.2013.0355
Barash Y, Dvir T, Tandeitnik P, Ruvinov E, Guterman H, Cohen S (2011) Electric field stimulation integrated into perfusion bioreactor for cardiac tissue engineering. Tissue Eng Part C Methods 16(6):1417–1426
Shapira-Schweitzer K, Habib M, Gepstein L, Seliktar D (2009) A photopolymerizable hydrogel for 3-D culture of human embryonic stem cell-derived cardiomyocytes and rat neonatal cardiac cells. J Mol Cell Cardiol 46(2):213–224
Radisic M PD, Fast VG PhD, Sharifov OF PhD, Iyer RK BASc, Park H PhD, Vunjak-Novakovic G PhD (2009) Optical mapping of impulse propagation in engineered cardiac tissue. Tissue Eng Part A 15(4):851–860
Herron TJ, Lee P, Jalife J (2012) Optical imaging of voltage and calcium in cardiac cells & tissues. Circ Res 110(4):609–623
Sekine H, Shimizu T, Dobashi I, Matsuura K, Hagiwara N, Takahashi M, Kobayashi E, Yamato M, Okano T (2011) Cardiac cell sheet transplantation improves damaged heart function via superior cell survival in comparison with dissociated cell injection. Tissue Eng Part A 17(23–24):2973–2980
Mihic A, Li J, Miyagi Y, Gagliardi M, Li SH, Zu J, Weisel RD, Keller G, Li RK (2014) The effect of cyclic stretch on maturation and 3D tissue formation of human embryonic stem cell-derived cardiomyocytes. Biomaterials 35(9):2798–2808
Kawamura M, Miyagawa S, Miki K, Saito A, Fukushima S, Higuchi T, Kawamura T, Kuratani T, Daimon T, Shimizu T, Okano T, Sawa Y (2012) Feasibility, safety, and therapeutic efficacy of human induced pluripotent stem cell-derived cardiomyocyte sheets in a porcine ischemic cardiomyopathy model. Circulation 126:S29–S37
Hasenfuss G, Mulieri LA, Blanchard EM, Holubarsch C, Leavitt BJ, Ittleman F, Alpert NR (1991) Energetics of isometric force development in control and volume-overload human myocardium. Comparison with animal species. Circ Res 68(3):836–846
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Wendel, J., Tranquillo, R. (2015). Cardiac Tissue Engineering for the Treatment of Heart Failure Post-Infarction. In: Dixon, I., Wigle, J. (eds) Cardiac Fibrosis and Heart Failure: Cause or Effect?. Advances in Biochemistry in Health and Disease, vol 13. Springer, Cham. https://doi.org/10.1007/978-3-319-17437-2_20
Download citation
DOI: https://doi.org/10.1007/978-3-319-17437-2_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-17436-5
Online ISBN: 978-3-319-17437-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)