Abstract
Hypertension has several effects on the retinal circulation. Assessment of retinal alterations has gained increasing interest, since it offers the unique opportunity to visualize human microvasculature noninvasively, directly, and repeatedly. This chapter is focused on the most established methods, funduscopy and scanning laser Doppler flowmetry (SLDF). Large-scale studies using funduscopy have shown that prevalence of retinal alterations is common in general and in particular in hypertensive patients. SLDF has expanded the notion of retinal changes due to functional and structural changes. Preliminary data suggest that these functional and structural changes, assessed with SLDF, can be improved by antihypertensive treatment. However, there are also unresolved questions, such as the prognostic value of changes in retinal parameters, since with both methods only indirect evidence exists that treatment-induced changes may be related to improved cardiovascular outcome.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Hypertension causes alterations in vascular structure and function. Structural changes in small arterioles can be diverged to two different patterns: first eutrophic remodeling characterized by a rearrangement of the smooth muscle cells around a narrowed lumen but without growth response (meaning that media cross-sectional area remains unchanged) and second hypertrophic remodeling, a growth response with increment of media cross-sectional area observed in patients with long-standing and/or severe hypertension [1]. Regardless of the pattern, both are characterized by an increased wall-to-lumen ratio (WLR).
The analysis of retinal vessels offers the exceptional opportunity to assess directly and noninvasively human microvasculature in vivo. In the last years several methods have been introduced for the assessment of retinal changes. Since this book is proposed as a practical approach guiding the reader in the assessment, the focus is on the most established methods, namely, funduscopy and scanning laser Doppler flowmetry (SLDF).
1 Assessment of the Retinal Arterioles
For a long time, direct ophthalmoscopic examination using the traditional four-grade classification system with increasing severity (Table 11.1, Fig. 11.1) introduced by Keith, Wagener, and Barker [1], modified by Scheie [3], was regarded as part of standard evaluation of patients suffering from hypertension [4]. Nowadays, its clinical usefulness in current clinical practice has been questioned due to its unreliable reproducibility [5], and hence routine funduscopic examination is no longer recommended [6]. Reliable assessment was only shown for advanced alterations like hemorrhages and exudates referring to at least grade 3 [7].
Funduscopic changes (e.g., cotton wool) (Republished from Ott and Schmieder [2])
1.1 Funduscopy
In the last decade several approaches have been developed, assessing more sensitive and quantitative alterations of retinal microvascular changes. Although protocols may differ in some minor points, the principles are similar. According to standardized protocols, one (e.g., 45°) nonstereoscopic color retinal photograph centered between the optic disk and the macula and approximately two disk diameters nasal to the optic disk has to be done in a darkened room. Hence, due to dark adaption, mydriatic agents are no longer necessary. However, in some studies (e.g., Rotterdam Study), pharmacological mydriasis was routinely done. For quantitative assessment of retinal vessels, the photographs have to be converted to digital pictures and analyzed by specific imaging software, e.g., the “Interactive Vessels Analysis” (IVAN) (University of Wisconsin, Madison, WI, USA). This software analysis provides semiautomated measurement of retinal arterioles and venules. Using formulas (e.g., Parr and Spears [8] or Knudtson et al. [9]), a single “central retinal artery equivalent (CRAE)” and a “central retinal vein equivalent (CRVE)” are calculated. Subsequently, arteriole-to-venule ratio (AVR) can be computed; for details see Hubbard et al. [10]. However, by this method, it is not possible to evaluate the retinal vascular wall thickness or vessel diameter directly.
1.2 Scanning Laser Doppler Flowmetry
SLDF, introduced by our study group about 10 years ago, allows the dynamic assessment of both functional (i.e., vascular tone) and structural parameters (i.e., wall and lumen diameter). In brief, SLDF is performed in the juxtapapillary area of the right eye, 2–3 mm temporal superior of the optic nerve at 670 nm (Heidelberg Retina Flowmeter, Heidelberg Engineering, Germany). A retinal sample of 2.56 × 0.64 × 0.30 nm is scanned within 2 s (at least one full systolic and one diastolic phase) and measured every 10 μm of this specific length of the retinal arteriole (80–140 μm). The confocal technique of the device ensures that only capillary flow of the superficial layer of 300 μm is measured. No pupil dilation is necessary (i.e., no constriction of patient daily routine) [11].
For assessment of functional parameters, mean retinal capillary flow (RCF) is assessed in the area of interest, and for further dynamic analysis, non pharmacological and pharmacological tools can be applied. Flicker light increases RCF at least in part via a nitric oxide (NO)-dependent mechanism and represents a non pharmacological tool to investigate vasodilatory capacity of retinal arterioles. It is noteworthy to mention that flicker light exposure has no effects on systemic blood pressure (BP), thereby minimizing potential systemic hemodynamic influences on RCF. Moreover, basal NO activity can be assessed by administration of the NO synthase inhibitor NG-monomethyl-l-arginine (l-NMMA).
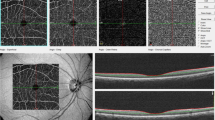
For assessment of structural parameters, the outer arteriole diameter (AD) is measured by reflection images, and the lumen diameter (LD) is measured by perfusion images. From the raw parameters, wall thickness (WT, [AD−LD]/2), WLR ([AD−LD]/LD) (Fig. 11.2), and wall cross-sectional area (WCSA, п/4 × [AD2–LD2]) can be calculated.
Scanning laser Doppler flowmetry (SLDF) (Republished from Ott and Schmieder [2]). (a) Differentiation between retinal arteriole and venule (SLDF live image before measurement). (b) Scanned area – reflection image. (c) Scanned area – perfusion image. (d) Scanned area – corrected and analyzed flow image. (e) Pulse curve run as mean retinal capillary flow (RCF) and time plot. (f) Localization of systolic and diastolic RCF on the image (d). (g) Localization of systolic and diastolic RCF on the image (c). (h) Calculation of wall-to-lumen ratio
Importantly, also individual pulsatile pattern of functional (RCF) and structural (e.g., WT) parameters of retinal arterioles in systole and diastole can be reliably assessed (Fig. 11.2).
All analyses are performed offline with automatic full-field perfusion imaging analysis (AFFPIA) (SLDF Version 4.0 by Welzenbach with improved resolution) [11].
2 Prevalence and Incidence (General Population, Hypertension)
2.1 Funduscopy
Several population-based studies have provided data on prevalence of retinal signs using standardized funduscopic photographs in the general population, partly with subsequent categorization according to (among others) hypertension status. In general, retinal signs are common in people aged 40 years or older, even in those without arterial hypertension. However, these findings can only be respected with caution, since different definitions of arterial hypertension have been used. Moreover, the reported prevalence of retinal signs depends also largely on the assessed parameter, e.g., retinopathy per se, AV nicking, or focal/generalized arteriolar narrowing (for details see Table 11.2).
In the Cardiovascular Health Study (CHS) (aged ≥65 years), 16.6 % (men 19.7 %; women 14.3 %) of normotensive participants and 25.4 % (men 30.0 %; women 23.0 %) of hypertensive patients (defined as BP ≥140/90 mmHg or history of hypertension with use of antihypertensive drugs) were reported to have generalized arteriolar narrowing (defined as the lowest twentieth percentile of AVR). In contrast, retinopathy was by far less frequently documented in this study, i.e., in 5.6 % (men 4.7 %; women 6.3 %) of normotensive participants and in 10.4 % (men 7.5 %; women 11.9 %) of hypertensive patients [16]. Confirmatory results were found in the Beijing study (aged ≥ 40 years), which also used the accepted criteria of hypertension (BP ≥140/90 mmHg or history of hypertension with use of antihypertensive drugs) [19]. Moreover, in the former study another important point was found, namely, differences in the prevalence of retinal signs according gender [16].
Other influencing factors are age and ethnicity. In the Blue Mountains Eye Study (BMES) [14] and the Atherosclerosis Risk in Communities (ARIC) Study [20], the prevalence of retinopathy increased with advancing age, whereas in the CHS only some retinal signs revealed an age-dependent relationship [16]. Regarding ethnicity, an enhanced prevalence of retinopathy was suggested in Afro-Caribbeans compared to Europeans, but in this study the use of standardized protocols was not clearly outlined [12]. In the ARIC study higher prevalence of retinopathy has been documented in blacks compared to whites [17]; however this difference was largely explained by the severity of hypertension.
Much less data are available addressing the frequency of new retinal signs. In the Beaver Dam Eye Study (BDES), the 5-year incidence of focal arteriolar narrowing was 7.7 % and of retinopathy 4.6 %, respectively, in normotensive (BP <160/95 mmHg) participants. Both incidences were about doubled in hypertensive patients [13]. In contrast, in the BMES the 5-year incidence for retinopathy was numerically higher in normotensive (BP <140/90 mmHg) subjects (8.2 %) but not clearly increased in hypertensive subjects (10.4 %) [15]. On the other hand, there is good evidence from several epidemiological studies that retinal alterations (i.e., generalized arteriolar narrowing) precedes the development of hypertension (Table 11.3), as a preclinical marker of hypertension.
2.2 Scanning Laser Doppler Flowmetry
Large epidemiological studies addressing prevalence and incidence of functional and structural microvascular alterations assessed with SLDF are lacking. Regarding RCF, similar values were found in young hypertensive patients compared to normotensive controls, which was confirmed by findings of an unaltered RCF between middle-aged patients with and without hypertension [49, 50]. Moreover, mean RCF was found to be similar in patients with hypertension stage 1–2 compared to patients with advanced stage of hypertensive disease, e.g., patients with treatment-resistant hypertension (TRH). However, by analyzing the individual pulsatile pattern of RCF in latter both groups, we were able to demonstrate a different pattern. RCF in systole was higher, whereas RCF in diastole was lower, and hence an exaggerated pulsed RCF (difference in RCF between systole and diastole) in patients with TRH was observed compared to patients with hypertension stage 1–2 [51].
Regarding structural parameters, small (monocentric) studies point toward similar findings as seen with funduscopy, namely, an increment of retinal alterations (e.g., WLR) with increased BP [52]. Moreover, a pooled analysis comprising ≥500 patients suggests an increased WLR with aging (Schmieder RE, Ott C, unpublished data). Prevalence and incidence rates of retinal alterations assessed by SLDF are difficult to describe since no thresholds values for the parameters are yet established.
3 Change with Treatment (Criteria for Significant Change, Incidence During Treatment)
3.1 Funduscopy
It was repeatedly shown that initiating effective antihypertensive therapy resulted in disappearance of severe (grade III and IV) hypertensive retinopathy [53, 54]. In a case report of a 34-year-old woman with a short history of hypertension, headache, and blurred vision, all indicative of malignant hypertension (her BP was 240/150 mmHg), funduscopic examination found swelling of the optic disk, widespread hemorrhages, and soft and hard exudates, consistent with grade IV or malignant hypertensive retinopathy, respectively. Antihypertensive treatment was initiated, and 10 months follow-up revealed a good BP control (110/70 mmHg). In accordance, funduscopy demonstrated an improvement of hypertensive retinopathy [55]. In a small cohort (n = 28), comprising previously untreated men with hypertension stage 1–2, scored (0–4) funduscopic changes were evaluated before and after 26 weeks of treatment with enalapril or hydrochlorothiazide, respectively. Both treatments resulted in a significant BP reduction but numerically higher after enalapril (−14.3 vs. −7.1 mmHg) compared to hydrochlorothiazide without reaching significant difference. Treatment with enalapril reduced numerically but nonsignificantly the frequency of arteriolar narrowing and arteriovenous crossing, whereas no changes were seen after treatment with hydrochlorothiazide [56].
Whether these observed changes are associated with improved cardiovascular (CV) and cerebrovascular prognosis remains to be determined. Indirect evidence comes from epidemiological studies. For example, in the BMES prevalence of hemorrhages and/or microaneurysm was comparable between normotensive and controlled (BP <160/95 mmHg) hypertensive men but not in women [14]. A subsequent sub-analysis of BMES revealed that prevalence of focal arteriolar narrowing was similar between normotensive (4.6 %) and controlled (BP <160/95 mmHg) hypertensive subjects (6.5 %), whereas its prevalence was more than doubled in treated uncontrolled (14.5 %) and untreated (15.3 %) hypertensive patients. In contrast, generalized arteriolar narrowing (narrowest quintile of AVR) was similarly prevalent in treated and controlled (22.0 %), treated and uncontrolled (22.5 %), and untreated hypertensive (27.2 %) patients but significantly greater compared to normotensive subjects (17.0 %) [57].
Thus, in contrast to data on improvement or even disappearance of qualitative hypertensive retinal abnormalities (e.g., papilledema, exudates), data are much less clear for quantitative retinal signs (e.g., arteriolar narrowing). On the other hand, generalized retinal arteriolar narrowing and AV nicking appear to be (irreversible) markers of mild to moderate hypertension, related not only to current and past BP levels but to cerebrovascular diseases as well [58].
3.2 Scanning Laser Doppler Flowmetry
Again data are limited with SLDF compared to funduscopy and are based on small studies only. Data with SLDF revealed that endothelial function (basal NO activity) was impaired in young hypertensive patients and improved after treatment with the angiotensin receptor blocker (ARB) candesartan [49], whereas no improvement was demonstrated in elderly hypertensive men after treatment with ARB valsartan [59]. Whether this discrepancy is related to the different ARB, different duration of therapy, or a potential irreversibility of vascular changes in the elderly patients is subject of ongoing investigations. Moreover, we were able to demonstrate that vasodilatory capacity (magnitude of vasodilation to flicker light) was lower in untreated hypertensive patients compared to normotensive controls, and systolic BP was inversely related to the percent increase of RCF due to flicker light exposure, independently of other CV risk factors [60]. Another study of our group suggests that BP and hence pulse pressure (PP) changes have an impact on pulsed RCF. In hypertensive patients with TRH, we observed a decrease of systolic and pulsed RCF 6 and 12 months after renal denervation (RDN), in parallel to decreases of BP and heart rate (HR). The reduction of pulsed RCF after RDN transfers into less shear stress on the vascular wall and, thereby, suggests an improvement of retinal (and potentially cerebral) microcirculation [61].
In a cross-sectional study, we observed that in treated hypertensive patients with BP control <140/90 mmHg, WLR was at the same level as observed in normotensive subjects but significantly lower than in treated hypertensive subjects with BP >140/90 mmHg [62]. Previously, two small prospective studies assessed the effect of antihypertensive treatment on retinal structural parameters using SLDF. In one study, hypertensive patients with non-insulin-dependent diabetes mellitus were treated with either aliskiren (n = 9) or ramipril (n = 7) for one year. To achieve equivalent BP control, open-label hydrochlorothiazide could be added, and hence only one patient in each group had BP ≥140/90 mmHg. Both treatment regimes resulted in a significant regression of retinal WLR after 1-year treatment without a difference between the groups [63]. In a second small unblinded study, hypertensive patients were treated with lercanidipine for 4 weeks and thereafter randomized to additional antihypertensive therapy with either enalapril (n = 10) or hydrochlorothiazide (n = 10) for 24 weeks. There was an improvement of WLR already after 4 weeks of treatment with lercanidipine alone, and only enalapril on top further reduced WLR (but not hydrochlorothiazide) [64]. Of note, both studies had small sample sizes, and surprisingly high values of WLR (>0.5) at baseline were reported. The reduction of WLR of about 50 % in both studies is very high in comparison to changes observed after treatment in analyses relying on the assessment in vascular remodeling of subcutaneous small arteries [65]. In a double-blind randomized study comprising in total of 40 patients with mild to moderate hypertension, treatment with manidipine or amlodipine for 4 weeks resulted not in any significant changes in WLR compared to baseline values (Ott et al., unpublished data). Overall data are sparse, and to clarify the effects of various antihypertensive agents on reversal of WLR, multicenter double-blind randomized studies with large number of patients are required.
4 Prognostic Value of Change
4.1 Funduscopy
No data are available whether treatment-induced regression of retinal alterations is related with reduction of other target-organ damages (e.g., left ventricular hypertrophy) or incident CV outcomes. So far, only epidemiological studies uniformly found that qualitative retinal signs of hypertensive retinopathy are related with incidence of CV disease. In accordance, quantitative retinal vascular caliber was associated with BP, target-organ damage, and CV disease (Table 11.2). Cross-sectional studies also indicated that the prevalence in retinal signs differs between normotensive, treated and controlled, treated and uncontrolled, and never-treated hypertensive patients. Thus, in addition to our pathophysiological understanding of vascular remodeling and its consequences, there seems to be a strong rationale that treatment-induced changes may also result in an improved CV outcome.
4.2 Scanning Laser Doppler Flowmetry
No prospective study analyzing the effects of treatment changes of retinal alterations assessed by SLDF, and hence its prognostic significance, is available. Again, at least indirect evidence exists that is based on findings of media-to-lumen ratio of subcutaneous small arterioles (measured with a myograph ex vivo) and cross-sectional studies.
In an Italian study (n = 126) with an averaged follow-up of 5.4 years including both patients with primary and secondary hypertension (e.g., pheochromocytoma) as well as normotensive subjects, an increment of subcutaneous media-to-lumen ratio was predictive of a diminished event-free survival [66]. Subsequent analysis, with an increased study population (n = 303) and a mean follow-up of 6.9 years, revealed that increased media-to-lumen ratio was of prognostic significance with regard to adverse CV and cerebrovascular outcome [67]. Accordingly, a Danish study comprising 159 patients with primary hypertension and moderate CV risk reported that media-to-lumen ratio was an independent predictor for the incidence of CV outcome even after adjustment of the Heart Score level over the follow-up of 4.6 years [68]. Recently, it was shown in a long-term follow-up survey (comprising 124 hypertensive patients) that after 9–12 months of antihypertensive treatment, SBP was reduced from 164 ± 15 to 134 ± 14 mmHg, which was accompanied by an regression of media-to-lumen ratio of subcutaneous small arteries (0.084 ± 0.03 vs. 0.075 ± 0.02, p < 0.01). Importantly, in the subsequent follow-up period of 15 years, the extent of the reduction in the media-to-lumen ratio of subcutaneous small arteries was demonstrated to be an independent predictor of CV events [69].
The relevance of these data on subcutaneous small arteries for retinal arteriolar changes comes from the nowadays recognized concept that changes seen in the small subcutaneous small arterioles are reflecting alterations seen also in other vascular beds. Indeed, Rizzoni et al. have previously demonstrated that WLR assessed by SLDF (retinal arterioles in vivo) and media-to-lumen ratio measured with myograph (subcutaneous small arteries taken from a biopsy) showed close correlation in hypertensive subjects (r = 0.80, p < 0.001), suggesting that SLDF may provide similar information about microcirculation alterations compared to subcutaneous small arteries [68].
References
Keith NM, Wagener HP, Barker NW. Some different types of essential hypertension: their course and prognosis. Am J Med Sci. 1939;197:332–43.
Ott C, Schmieder RE. Retinal circulation in arterial disease. In: Berbari A, Mancia G, editors. Arterial disorders. Springer; 2015.
Scheie HG. Evaluation of ophthalmoscopic changes of hypertension and arteriolar sclerosis. AMA Archiv Ophthalmol. 1953;49:117–38.
Guidelines Subcommittee. 1999 World Health Organization-International Society of Hypertension guidelines for the management of hypertension. J Hypertens. 1999;17:151–83.
Dimmitt SB, West JN, Eames SM, Gibson JM, Gosling P, Littler WA. Usefulness of ophthalmoscopy in mild to moderate hypertension. Lancet. 1989;1:1103–6.
Mancia G, Fagard R, Narkiewicz K, et al. 2013 ESH/ESC Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2013;31:1281–357.
van den Born BJ, Hulsman CA, Hoekstra JB, Schlingemann RO, van Montfrans GA. Value of routine funduscopy in patients with hypertension: systematic review. BMJ. 2005;331:73.
Parr JC, Spears GF. General caliber of the retinal arteries expressed as the equivalent width of the central retinal artery. Am J Ophthalmol. 1974;77:472–7.
Knudtson MD, Lee KE, Hubbard LD, Wong TY, Klein R, Klein BE. Revised formulas for summarizing retinal vessel diameters. Curr Eye Res. 2003;27:143–9.
Hubbard LD, Brothers RJ, King WN, et al. Methods for evaluation of retinal microvascular abnormalities associated with hypertension/sclerosis in the Atherosclerosis Risk in Communities Study. Ophthalmology. 1999;106:2269–80.
Harazny JM, Raff U, Welzenbach J, et al. New software analyses increase the reliability of measurements of retinal arterioles morphology by scanning laser Doppler flowmetry in humans. J Hypertens. 2011;29:777–82.
Sharp PS, Chaturvedi N, Wormald R, McKeigue PM, Marmot MG, Young SM. Hypertensive retinopathy in Afro-Caribbeans and Europeans. Prevalence and risk factor relationships. Hypertension. 1995;25:1322–5.
Klein R, Klein BE, Moss SE. The relation of systemic hypertension to changes in the retinal vasculature: the Beaver Dam Eye Study. Trans Am Ophthalmol Soc. 1997;95:329–48; discussion 348–50.
Yu T, Mitchell P, Berry G, Li W, Wang JJ. Retinopathy in older persons without diabetes and its relationship to hypertension. Arch Ophthalmol. 1998;116:83–9.
Cugati S, Cikamatana L, Wang JJ, Kifley A, Liew G, Mitchell P. Five-year incidence and progression of vascular retinopathy in persons without diabetes: the Blue Mountains Eye Study. Eye. 2006;20:1239–45.
Wong TY, Klein R, Sharrett AR, et al. The prevalence and risk factors of retinal microvascular abnormalities in older persons: The Cardiovascular Health Study. Ophthalmology. 2003;110:658–66.
Wong TY, Klein R, Duncan BB, et al. Racial differences in the prevalence of hypertensive retinopathy. Hypertension. 2003;41:1086–91.
van Leiden HA, Dekker JM, Moll AC, et al. Risk factors for incident retinopathy in a diabetic and nondiabetic population: the Hoorn study. Arch Ophthalmol. 2003;121:245–51.
Wang S, Xu L, Jonas JB, et al. Major eye diseases and risk factors associated with systemic hypertension in an adult Chinese population: the Beijing Eye Study. Ophthalmology. 2009;116:2373–80.
Klein R, Sharrett AR, Klein BE, et al. Are retinal arteriolar abnormalities related to atherosclerosis?: The Atherosclerosis Risk in Communities Study. Arterioscler Thromb Vasc Biol. 2000;20:1644–50.
Sharrett AR, Hubbard LD, Cooper LS, et al. Retinal arteriolar diameters and elevated blood pressure: the Atherosclerosis Risk in Communities Study. Am J Epidemiol. 1999;150:263–70.
Wong TY, Klein R, Couper DJ, et al. Retinal microvascular abnormalities and incident stroke: the Atherosclerosis Risk in Communities Study. Lancet. 2001;358:1134–40.
Wong TY, Klein R, Sharrett AR, et al. Retinal arteriolar narrowing and risk of coronary heart disease in men and women. The Atherosclerosis Risk in Communities Study. JAMA. 2002;287:1153–9.
Wong TY, Klein R, Sharrett AR, et al. Retinal arteriolar diameter and risk for hypertension. Ann Intern Med. 2004;140:248–55.
Tikellis G, Arnett DK, Skelton TN, et al. Retinal arteriolar narrowing and left ventricular hypertrophy in African Americans. The Atherosclerosis Risk in Communities (ARIC) study. Am J Hypertens. 2008;21:352–9.
Yatsuya H, Folsom AR, Wong TY, et al. Retinal microvascular abnormalities and risk of lacunar stroke: Atherosclerosis Risk in Communities Study. Stroke J Cerebral Circ. 2010;41:1349–55.
Wong TY, Klein R, Nieto FJ, et al. Retinal microvascular abnormalities and 10-year cardiovascular mortality: a population-based case-control study. Ophthalmology. 2003;110:933–40.
Wong TY, Klein R, Klein BE, Meuer SM, Hubbard LD. Retinal vessel diameters and their associations with age and blood pressure. Invest Ophthalmol Vis Sci. 2003;44:4644–50.
Wong TY, Shankar A, Klein R, Klein BE, Hubbard LD. Prospective cohort study of retinal vessel diameters and risk of hypertension. BMJ. 2004;329:79.
Wang JJ, Liew G, Klein R, et al. Retinal vessel diameter and cardiovascular mortality: pooled data analysis from two older populations. Eur Heart J. 2007;28:1984–92.
Leung H, Wang JJ, Rochtchina E, et al. Relationships between age, blood pressure, and retinal vessel diameters in an older population. Invest Ophthalmol Vis Sci. 2003;44:2900–4.
Leung H, Wang JJ, Rochtchina E, Wong TY, Klein R, Mitchell P. Impact of current and past blood pressure on retinal arteriolar diameter in an older population. J Hypertens. 2004;22:1543–9.
Smith W, Wang JJ, Wong TY, et al. Retinal arteriolar narrowing is associated with 5-year incident severe hypertension: the Blue Mountains Eye Study. Hypertension. 2004;44:442–7.
Wang JJ, Liew G, Wong TY, et al. Retinal vascular calibre and the risk of coronary heart disease-related death. Heart. 2006;92:1583–7.
Wong TY, Hubbard LD, Klein R, et al. Retinal microvascular abnormalities and blood pressure in older people: the Cardiovascular Health Study. Br J Ophthalmol. 2002;86:1007–13.
Wong TY, Kamineni A, Klein R, et al. Quantitative retinal venular caliber and risk of cardiovascular disease in older persons: the cardiovascular health study. Arch Intern Med. 2006;166:2388–94.
Wong TY, Islam FM, Klein R, et al. Retinal vascular caliber, cardiovascular risk factors, and inflammation: the multi-ethnic study of atherosclerosis (MESA). Invest Ophthalmol Vis Sci. 2006;47:2341–50.
Kawasaki R, Cheung N, Wang JJ, et al. Retinal vessel diameters and risk of hypertension: the Multiethnic Study of Atherosclerosis. J Hypertens. 2009;27:2386–93.
Yau JW, Xie J, Kawasaki R, et al. Retinal arteriolar narrowing and subsequent development of CKD Stage 3: the Multi-Ethnic Study of Atherosclerosis (MESA). Am J Kidney Dis Off J Nat Kidney Found. 2011;58:39–46.
Ikram MK, de Jong FJ, Vingerling JR, et al. Are retinal arteriolar or venular diameters associated with markers for cardiovascular disorders? The Rotterdam Study. Invest Ophthalmol Vis Sci. 2004;45:2129–34.
Ikram MK, Witteman JC, Vingerling JR, Breteler MM, Hofman A, de Jong PT. Retinal vessel diameters and risk of hypertension: the Rotterdam Study. Hypertension. 2006;47:189–94.
Ikram MK, de Jong FJ, Bos MJ, et al. Retinal vessel diameters and risk of stroke: the Rotterdam Study. Neurology. 2006;66:1339–43.
Wieberdink RG, Ikram MK, Koudstaal PJ, Hofman A, Vingerling JR, Breteler MM. Retinal vascular calibers and the risk of intracerebral hemorrhage and cerebral infarction: the Rotterdam Study. Stroke J Cerebral Circ. 2010;41:2757–61.
Sun C, Liew G, Wang JJ, et al. Retinal vascular caliber, blood pressure, and cardiovascular risk factors in an Asian population: the Singapore Malay Eye Study. Invest Ophthalmol Vis Sci. 2008;49:1784–90.
Sabanayagam C, Shankar A, Koh D, et al. Retinal microvascular caliber and chronic kidney disease in an Asian population. Am J Epidemiol. 2009;169:625–32.
Jeganathan VS, Sabanayagam C, Tai ES, et al. Effect of blood pressure on the retinal vasculature in a multi-ethnic Asian population. Hypertension Res Off J Jpn Soc Hypertens. 2009;32:975–82.
Sabanayagam C, Tai ES, Shankar A, Lee J, Sun C, Wong TY. Retinal arteriolar narrowing increases the likelihood of chronic kidney disease in hypertension. J Hypertens. 2009;27:2209–17.
Mitchell P, Cheung N, de Haseth K, et al. Blood pressure and retinal arteriolar narrowing in children. Hypertension. 2007;49:1156–62.
Delles C, Michelson G, Harazny J, Oehmer S, Hilgers KF, Schmieder RE. Impaired endothelial function of the retinal vasculature in hypertensive patients. Stroke J Cerebral Circ. 2004;35:1289–93.
Ritt M, Harazny JM, Ott C, et al. Analysis of retinal arteriolar structure in never-treated patients with essential hypertension. J Hypertens. 2008;26:1427–34.
Harazny JM, Ott C, Raff U, et al. First experience in analysing pulsatile retinal capillary flow and arteriolar structural parameters measured noninvasively in hypertensive patients. J Hypertens. 2014;32:2246–52; discussion 2252.
Ott C, Raff U, Harazny JM, Michelson G, Schmieder RE. Central pulse pressure is an independent determinant of vascular remodeling in the retinal circulation. Hypertension. 2013;61:1340–5.
Kirkendall WM, Armstrong ML. Vascular changes in the eye of the treated and untreated patient with essential hypertension. Am J Cardiol. 1962;9:663–8.
Bock KD. Regression of retinal vascular changes by antihypertensive therapy. Hypertension. 1984;6:III158–62.
Strachan MW, McKnight JA. Images in clinical medicine. Improvement in hypertensive retinopathy after treatment of hypertension. New Engl J Med. 2005;352:e17.
Dahlof B, Stenkula S, Hansson L. Hypertensive retinal vascular changes: relationship to left ventricular hypertrophy and arteriolar changes before and after treatment. Blood Press. 1992;1:35–44.
Wang JJ, Mitchell P, Leung H, Rochtchina E, Wong TY, Klein R. Hypertensive retinal vessel wall signs in a general older population: the Blue Mountains Eye Study. Hypertension. 2003;42:534–41.
Wong TY, Klein R, Klein BE, Tielsch JM, Hubbard L, Nieto FJ. Retinal microvascular abnormalities and their relationship with hypertension, cardiovascular disease, and mortality. Surv Ophthalmol. 2001;46:59–80.
Oehmer S, Harazny J, Delles C, et al. Valsartan and retinal endothelial function in elderly hypertensive patients. Blood Press. 2006;15:185–91.
Ritt M, Harazny JM, Ott C, et al. Impaired increase of retinal capillary blood flow to flicker light exposure in arterial hypertension. Hypertension. 2012;60:871–6.
Ott C, Harazny JM, Schmid A, et al. Retinal microperfusion after renal denervation in treatment resistant hypertensive patients. Clin Res Cardiol. submitted.
Harazny JM, Ritt M, Baleanu D, et al. Increased wall:lumen ratio of retinal arterioles in male patients with a history of a cerebrovascular event. Hypertension. 2007;50:623–9.
De Ciuceis C, Savoia C, Arrabito E, et al. Effects of a long-term treatment with aliskiren or ramipril on structural alterations of subcutaneous small-resistance arteries of diabetic hypertensive patients. Hypertension. 2014;64:717–24.
De Ciuceis C, Salvetti M, Rossini C, et al. Effect of antihypertensive treatment on microvascular structure, central blood pressure and oxidative stress in patients with mild essential hypertension. J Hypertens. 2014;32:565–74.
Agabiti-Rosei E, Heagerty AM, Rizzoni D. Effects of antihypertensive treatment on small artery remodelling. J Hypertens. 2009;27:1107–14.
Rizzoni D, Porteri E, Boari GE, et al. Prognostic significance of small-artery structure in hypertension. Circulation. 2003;108:2230–5.
De Ciuceis C, Porteri E, Rizzoni D, et al. Structural alterations of subcutaneous small-resistance arteries may predict major cardiovascular events in patients with hypertension. Am J Hypertens. 2007;20:846–52.
Rizzoni D, Porteri E, Duse S, et al. Relationship between media-to-lumen ratio of subcutaneous small arteries and wall-to-lumen ratio of retinal arterioles evaluated noninvasively by scanning laser Doppler flowmetry. J Hypertens. 2012;30:1169–75.
Buus NH, Mathiassen ON, Fenger-Gron M, et al. Small artery structure during antihypertensive therapy is an independent predictor of cardiovascular events in essential hypertension. J Hypertens. 2013;31:791–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Ott, C., Schmieder, R.E. (2015). Damage of Retinal Arterioles in Hypertension. In: Agabiti Rosei, E., Mancia, G. (eds) Assessment of Preclinical Organ Damage in Hypertension. Springer, Cham. https://doi.org/10.1007/978-3-319-15603-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-15603-3_11
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-15602-6
Online ISBN: 978-3-319-15603-3
eBook Packages: MedicineMedicine (R0)