Abstract
Clostridium difficile is a Gram-positive, anaerobic, spore-producing anaerobe [1] responsible for approximately 50–70% of gastrointestinal infections in hospitalized patients [2, 3]. An episode of C. difficile infection (CDI) is defined as a clinical picture compatible with CDI (i. e., diarrhea, ileus and toxic megacolon) with microbiological evidence of C. difficile (ideally free C. difficile toxins) in stool, without reasonable evidence of another cause of diarrhea, or identification of pseudomembranous colitis during endoscopy, after colectomy or on autopsy [4, 5]. Life-threatening cases are associated with severe colitis and shock, and can require intensive care unit (ICU) admission and colectomy [4, 6].
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Intensive Care Unit
- Intensive Care Unit Admission
- Intensive Care Unit Patient
- Infection Control Measure
- Fecal Microbiota Transplantation
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Clostridium difficile is a Gram-positive, anaerobic, spore-producing anaerobe [1] responsible for approximately 50–70% of gastrointestinal infections in hospitalized patients [2, 3]. An episode of C. difficile infection (CDI) is defined as a clinical picture compatible with CDI (i. e., diarrhea, ileus and toxic megacolon) with microbiological evidence of C. difficile (ideally free C. difficile toxins) in stool, without reasonable evidence of another cause of diarrhea, or identification of pseudomembranous colitis during endoscopy, after colectomy or on autopsy [4, 5]. Life-threatening cases are associated with severe colitis and shock, and can require intensive care unit (ICU) admission and colectomy [4, 6].
CDI is increasingly recognized as a leading public health threat. European surveillance data indicate that CDI rates among hospitalized patients have increased in many countries [2] and that approximately one in ten cases of CDI cause – or contribute to – ICU admission or death, or lead to colectomy [6]. The infection significantly prolongs hospitalization, and total length of hospital stay in studies was on average 15 days [7]. In the US, the incidence of CDI among hospitalized adults almost doubled from 2001 to 2010, to 8.2 discharges per 1,000 total adult discharges [8]. Indeed, C. difficile is the most common pathogen isolated from patients with healthcare-associated infections in the US [3], causing an estimated 250,000 infections and 14,000 deaths a year [9]. CDI is regarded by the Centers for Disease Control and Prevention (CDC) as one of its top three ‘urgent’ (antibiotic resistant) threats [9].
An understanding of current knowledge and guidelines on CDI is essential for intensivists both to manage patients admitted to ICU facilities with, or as a result of, CDI and because critically ill patients are at risk of developing the infection while in the ICU.
Epidemiology and Outcomes
Data on the epidemiology of CDI in ICU patients are limited and heterogeneous. Mixing data from patients with CDI that results in ICU admission, as opposed to CDI that begins after a patient has entered the ICU, contributes to this heterogeneity. Retrospective cohort studies in various types of ICU have typically found that approximately 0.5–5% of patients acquire CDI during an ICU stay [10–17]. In France, for example, 512/5260 (9.7%) patients admitted to three ICUs had diarrhea and were tested for CDI. Of these, 69 patients (13.5% of tested patients and 1.3% of all admitted patients) had CDI; 68.1% of CDI cases were ICU-acquired [12].
Crude ICU or in-hospital mortality rates among ICU patients with CDI are typically around 21–31% [10, 11, 12, 16, 18]. Generally, 30-day or in-hospital mortality typically reaches 33–40% among patients who undergo emergency surgery for fulminant CDI [19, 20]. Studies in hospitalized patients (not ICU-specific) clearly show that CDI increases the risk of 30-day mortality by approximately 2 or 2.5-fold [5, 21]. For example, a recent very large study of 6,522 inpatient diarrhea episodes showed an approximate doubling of 30-day mortality in CDI cases (defined according to the presence of toxin in fecal samples) versus controls (odds ratio 1.61; 95% confidence intervals 1.12–2.31; p = 0.0101) [5]. The attributable contribution of CDI to mortality risk specifically in critically ill patients is less clear and studies have not shown a significant effect after adjusting for confounding factors [12, 13, 18]. However, CDI has been shown to independently extend hospital or ICU stay [12, 13, 18]. A key feature of CDI is recurrence of symptoms, which is reported in ~20–25% of patients treated with metronidazole or vancomycin [22, 23]. The CDI recurrence rate was 47% lower in patients given fidaxomicin versus vancomycin in studies [22]. However, recurrence in critically ill patients has not yet been studied in great detail.
Pathogenesis and Risk Factors
CDI results from transmission of C. difficile spores by the fecal-oral route. Infection control measures employed in relation to CDI focus on preventing infection transmission from symptomatic patients with CDI (see below). However, recent research has revealed that, outside of an outbreak setting, the majority of CDI cases cannot be linked to earlier cases using whole genome sequencing of isolates [24]. This finding highlights the potential importance of other sources of C. difficile, possibly asymptomatic patients or environmental reservoirs, in the transmission of infection.
Ingested spores pass through the stomach and into the upper intestine, where they germinate into vegetative cells. The vegetative cells proliferate in the colon, a process facilitated when the normal gut microbiota are altered by antibiotics. C. difficile produces two enterotoxins, known as toxins A and B, the principal virulence factors in CDI. Studies in recent years have demonstrated that these toxins trigger not only various inflammatory processes and cell death locally [25], but also a comprehensive systemic inflammatory response [26]. Notably, excess mortality correlates with changes in inflammatory biomarkers that are specific to particular C. difficile genotypes, implicating the host inflammatory pathways as a major influence on poor outcome [27]. A third C. difficile toxin, known as binary toxin, has also been identified, although its clinical significance is still unclear [28]. Virulence also appears to be determined by other non-toxin factors that modulate germination, sporulation and colonization, and by the effect of the microbiota on colonic metabolite levels [25, 29].
Studies into CDI risk factors have generally been heterogeneous in terms of their methods and quality [30] and there are few data specific to ICU patients.
Primary Infection
The two main risk factors for CDI are exposure to antibiotics and exposure to C. difficile in the hospital setting [1]. Most antibiotic classes have been implicated, but fluoroquinolones and third-generation cephalosporins are associated with higher risk, including in ICU patients [16]. CDI is particularly common in elderly patients [6]. Other important risk factors include multiple co-morbidities, frailty, immunosuppression and gastrointestinal surgery [1]. Gastric acid suppression for stress ulcer prevention, especially with proton pump inhibitors (PPI), also increases the risk of CDI in ICU patients [16, 17]. In one recent study in critically ill medical patients, PPIs increased the risk of CDI by an odds ratio of 3.1 (1.11–8.74) in a multivariate analysis, compared with ratios of < 2 for all antibiotic classes [16]. However, it should be cautioned that the microbiological definition of CDI in this retrospective study was not optimized. Speculation that elemental, non-residue enteral feeds could predispose patients to CDI [31] is supported by recent evidence of an independent effect of nasogastric tube use [32]. Evidence suggests that patients receiving prolonged mechanical ventilation are at a high risk of CDI: 5.3% of such adults were discharged with a concomitant diagnosis of CDI in one large study [33].
Severe/Complicated Infection and Mortality
Severe/complicated CDI and mortality (not limited to ICU patients), typically associated with leukocytosis, is seen more commonly in the elderly, those with multiple co-morbidities, and/or patients with renal failure or hypoalbuminemia [1, 4, 30]. Infection by hypervirulent ribotypes (e. g., 027 and 078) is also associated with increased mortality risk [27, 30]. Although ribotype epidemiology has been relatively well characterized in some regions (especially Europe and North America) relatively few data are available from other areas. Regional variations are often marked, suggesting that clonal expansion/transmission of particular strain(s) drives local epidemiology. Clinical risk factor scores are in development [34], and these will become more germane as the treatment options for CDI increase.
Diagnosis of CDI within the ICU is itself a strong predictor of a complicated disease course [34]. However, few data exist on risk factors for severe/complicated CDI or mortality specifically in ICU patients, and there is no validated score to aid treatment stratification. Risk factors associated with mortality among ICU patients with CDI, determined via multivariate analyses, have variously included advanced age, septic shock, ward-to-ICU transfer, increasing Acute Physiology and Chronic Health Evaluation (APACHE) score, end-stage liver disease, and length of hospital stay prior to CDI [18, 35, 36]. In addition, male sex, rising C-reactive protein (CRP) levels and previous exposure to fluoroquinolones have been independently associated with severe CDI in the ICU [14]. Additional risk factors for poor outcomes identified by univariate analyses include immunosuppression, high Logistic Organ Dysfunction Score, high McCabe score [12], hypoalbuminemia, history of corticosteroid prescription, prolonged ICU stay, high Sequential Organ Failure Assessment (SOFA) score at the time of CDI diagnosis, and high Simplified Acute Physiology Score (SAPS II) [37].
Recurrence
Diagnosis
CDI remains under-diagnosed, in part owing to low clinical suspicion among healthcare staff and low laboratory testing rates [2, 39]. Rapid and accurate diagnosis is important, however, to avoid delays in appropriate therapy and to reduce empirical therapy [40]. Laboratory testing should be performed on loose stool samples in patients with typical signs and symptoms (usually unexplained diarrhea) of CDI to confirm the diagnosis [1, 4]. All patients who are immunosuppressed (as a result of malignancy, chemotherapy, corticosteroid therapy, organ transplantation or cirrhosis) should be tested if they develop diarrhea [1]. Routine screening for C. difficile in hospitalized patients without diarrhea is not recommended [1].
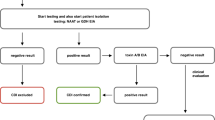
Advances in recent years have resulted in an array of different types of laboratory tests for C. difficile. These can be categorized as: (1) Tests to detect C. difficile toxins, i. e., cell culture cytotoxicity assay and enzyme immunoassays (EIA) or membrane immunoassays for toxins A/B, or glutamate dehydrogenase (GDH); (2) toxigenic culture of C. difficile; and (3) nucleic acid amplification tests (NAAT), such as polymerase chain reaction (PCR) for the genes that code for C. difficile toxins [1, 4]. The best standard test has not been established. European guidelines recommend the use of a two or three-stage algorithm in which a positive sensitive screening test is followed by use of a more specific test [4, 41]. Recent evidence that presence of C. difficile toxin in the stool predicts mortality from CDI [5] implies that testing algorithms should certainly include toxin testing. Moreover, there is good evidence that use of PCR tests alone leads to over-diagnosis of CDI, principally because these highly sensitive tests will detect colonization by a toxigenic strain in some patients with diarrhea who do not have true infection [5].
Treatment
Updated guidelines for specific therapy for CDI, based on disease severity, have recently been published in Europe [4, 42] and North America [1]. The response to CDI treatment should be monitored on a daily basis to detect patients who fail to respond or have worsening symptoms. Treatment response is defined as a reduction in stool frequency or an improvement in stool consistency, together with improvements in markers of disease severity and no new signs of severe disease [4].
Supportive Measures
Supportive measures recommended for patients with CDI include fluid resuscitation and electrolyte replacement. Anti-motility therapy should be avoided in acute CDI, PPI use should be reviewed, and unnecessary antimicrobial therapy discontinued [1, 4]. In the absence of ileus or significant abdominal distention, oral or enteral feeding should be continued [1]. Fecal collection systems can be useful in critically ill patients with diarrhea, including that caused by CDI [43].
Mild-moderate CDI
Patients with mild CDI may not need specific antibiotic therapy against C. difficile [4, 42]. In non-epidemic situations, in which a mild CDI case is clearly induced by antibiotics, it may be acceptable to stop the inducing antibiotic and closely observe the clinical response for 24–48 h [4]. European guidelines recommend 10 days of metronidazole (500 mg three times daily [TID]), vancomycin (125 mg four times daily [QID]) or fidaxomicin (200 mg twice daily [BID]) for initial episodes of non-severe CDI (Table 1) [4]. It is noted that fidaxomicin was not associated with a reduced rate of recurrent CDI due to PCR ribotype 027 as opposed to non-027 ribotypes [22]. US and English guidelines recommend metronidazole for mild-to-moderate CDI [1, 42]. Recent data have questioned the relative efficacy of metronidazole in comparison with vancomycin. In a pooled analysis of data from two phase 3 clinical trials, three factors were strongly associated with clinical success: Vancomycin treatment, treatment-naive status, and mild or moderate CDI severity [23].
European guidelines recommend metronidazole, vancomycin or fidaxomicin for a first CDI recurrence and for patients at risk of recurrence [4], although it is probably prudent to avoid metronidazole given increasing evidence of its lower efficacy [23]. Vancomycin and fidaxomicin are recommended in Europe for multiple recurrences [4]. Fecal microbiota transplantation (FMT), in combination with oral antibiotic treatment, is strongly recommended for multiple recurrent CDI episodes unresponsive to repeated antibiotic treatment [4]. US guidelines recommend pulsed vancomycin for a second recurrence and that FMT should be considered in the case of a third recurrence [1]. In general, guidelines recommend against the use of probiotics or toxin binding agents for treatment of any severity of CDI [4].
Severe or Complicated CDI
Vancomycin and fidaxomicin are recommended in Europe for severe infection [4]. The dose of vancomycin can be increased in life-threatening infection (500 mg QID). These guidelines caution that there is no evidence to support the use of fidaxomicin in life-threatening CDI [4]. In England, national guidelines recommend that fidaxomicin should be considered for patients with severe CDI who are considered at high risk of recurrence (elderly, multiple co-morbidities, or concomitant antibiotic therapy) and for those with recurrent CDI of any severity [42]. If oral therapy for severe CDI is not possible, it is recommended that intravenous (i.v.) metronidazole (500 mg TID) should be combined with vancomycin (500 mg QID for 10 days) administered either by intracolonic retention enema, oro- or nasogastric tube. Intravenous tigecycline (50 mg BID for 14 days) may be an alternative [4], although it does not have a licensed indication for CDI treatment.
US guidelines recommend oral vancomycin for severe CDI, defined as the presence of hypoalbuminemia plus either leukocytosis or abdominal tenderness [1]. Severe and complicated CDI is defined as that necessitating ICU admission, or with various signs of shock and severe disease. In these circumstances, oral vancomycin (125 mg QID) plus i.v. metronidazole (500 mg TID) is recommended in the absence of significant abdominal distension. Vancomycin delivered orally (500 mg QID) and by enema (500 mg QID) plus i.v. metronidazole is recommended when there is ileus or toxic colon and/or significant abdominal distension [1].
Surgery
Surgery (usually total colectomy with ileostomy) is indicated when there is perforation of the colon or systemic inflammation and a deteriorating clinical condition not responding to antibiotic therapy (including toxic megacolon, an acute abdomen and severe ileus) [4]. A future alternative to colectomy may be diverting loop ileostomy and colonic lavage [44], combined with intracolonic antegrade vancomycin and i.v. metronidazole [4].
Infection Control and Prevention
Infection control measures are mandatory following a diagnosis of CDI [4]. Recommended measures include hand hygiene (with soap and water instead of alcohol hand rubs), protective clothing, sporicidal decontamination of the hospital environment and the use of dedicated patient care equipment for infected patients, with appropriate disinfection [1, 45, 46] (Table 2). More widely, hospital-based infection control programs, including antibiotic stewardship programs, can help to decrease the incidence of CDI but are beyond the scope of this review.
Conclusion
Critically ill patients in the ICU typically have multiple risk factors for the acquisition of CDI. Accordingly, all healthcare staff in the ICU should be aware of the risk of CDI. Crucially, all patients with unexplained diarrhea in the ICU should be tested promptly for CDI using optimized laboratory assays. Supportive care and specific therapy should be provided in patients with suspected or diagnosed CDI according to current guidelines. Rigorous, multifaceted infection control measures are vital to prevent the onward spread of infection.
Acknowledgements
The authors are members of the CDI Europe expert group, which is supported by Astellas Pharma EMEA (Chertsey, UK). Astellas Pharma EMEA supported and funded the development of this chapter, with medical writing support provided by Lee Baker (Interel, London, UK).
References
Surawicz CM, Brandt LJ, Binion DG et al (2013) Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol 108:478–498
European Centre for Disease Prevention (2013) Point prevalence survey of healthcare-associated infections and antimicrobial use in European acute care hospitals. Stockholm, ECDC. http://www.ecdc.europa.eu/en/publications/Publications/healthcare-associated-infections-antimicrobial-use-PPS.pdf. Accessed 17 October 2014
Magill SS, Edwards JR, Bamberg W et al (2014) Multistate point-prevalence survey of health care-associated infections. N Engl J Med 370:1198–1208
Debast SB, Bauer MP, Kuijper EJ (2014) European Society of Clinical Microbiology and Infectious Diseases (ESCMID): update of the treatment guidance document for Clostridium difficile infection (CDI). Clin Microbiol Infect 20(Suppl 2):1–26
Planche TD, Davies KA, Coen PG et al (2013) Differences in outcome according to Clostridium difficile testing method: a prospective multicentre diagnostic validation study of C difficile infection. Lancet Infect Dis 13:936–945
Bauer MP, Notermans DW, van Benthem BH et al (2011) Clostridium difficile infection in Europe: a hospital-based survey. Lancet 377:63–73
Wiegand PN, Nathwani D, Wilcox MH, Stephens J, Shelbaya A, Haider S (2012) Clinical and economic burden of hospital onset health care facility acquired Clostridium difficile infection in Europe: a systematic review. J Hops Infect 81:1–14
Reveles KR, Lee GC, Boyd NK, Frei CR (2014) The rise in Clostridium difficile infection incidence among hospitalized adults in the United States: 2001–2010. Am J Infect Control 42:1028–1032
Centres for Disease Control and Prevention (2013) Antibiotic resistance threats in the United States, 2013. CDC. http://www.cdc.gov/drugresistance/threat-report-2013/pdf/ar-threats-2013. Accessed 17 October 2014
Ang CW, Heyes G, Morrison P, Carr B (2008) The acquisition and outcome of ICU-acquired Clostridium difficile infection in a single centre in the UK. J Infect 57:435–440
Musa SA, Moran C, Thomson SJ et al (2011) Clostridium difficile-associated disease acquired in the cardiothoracic intensive care unit. J Cardiothorac Vasc Anesth 25:263–267
Zahar JR, Schwebel C, Adrie C et al (2012) Outcome of ICU patients with Clostridium difficile infection. Crit Care 16:R215
Dodek PM, Norena M, Ayas NT, Romney M, Wong H (2013) Length of stay and mortality due to Clostridium difficile infection acquired in the intensive care unit. J Crit Care 28:335–340
Khanafer N, Touré A, Chambrier C (2013) Predictors of Clostridium difficile infection severity in patients hospitalised in medical intensive care. World J Gastroenterol 19:8034–8041
Tripathy S, Nair P, Rothburn M (2013) Clostridium difficile associated disease in a neurointensive care unit. Front Neurol 4:82
Buendgens L, Bruensing J, Matthes M et al (2014) Administration of proton pump inhibitors in critically ill medical patients is associated with increased risk of developing Clostridium difficile-associated diarrhea. J Crit Care 29:696.e11–696.e15
MacLaren R, Reynolds PM, Allen RR (2014) Histamine-2 receptor antagonists vs proton pump inhibitors on gastrointestinal tract hemorrhage and infectious complications in the intensive care unit. JAMA Intern Med 174:564–574
Micek ST, Schramm G, Morrow L et al (2013) Clostridium difficile infection: a multicenter study of epidemiology and outcomes in mechanically ventilated patients. Crit Care Med 41:1968–1975
Bhangu A, Nepogodiev D, Gupta A, Torrance A, Singh P, West Midlands Research Collaborative (2012) Systematic review and meta-analysis of outcomes following emergency surgery for Clostridium difficile colitis. Br J Surg 99:1501–1513
Lee DY, Chung EL, Guend H, Whelan RL, Wedderburn RV, Rose KM (2014) Predictors of mortality after emergency colectomy for Clostridium difficile colitis: An analysis of ACS-NSQIP. Ann Surg 259:148–156
Hensgens MP, Goorhuis A, Dekkers OM, van Benthem BH, Kuijper EJ (2013) All-cause and disease-specific mortality in hospitalized patients with Clostridium difficile infection: a multicenter cohort study. Clin Infect Dis 56:1108–1116
Mullane KM, Gorbach S (2011) Fidaxomicin: first-in-class macrocyclic antibiotic. Expert Rev Anti Infect Ther 9:767–777
Johnson S, Louie TJ, Gerding DN et al (2014) Vancomycin, metronidazole, or tolevamer for Clostridium difficile infection: results from two multinational, randomized, controlled trials. Clin Infect Dis 59:345–354
Eyre DW, Cule ML, Wilson DJ et al (2013) Diverse sources of C. difficile infection identified on whole-genome sequencing. N Engl J Med 369:1195–1205
Peniche AG, Savidge TC, Dann SM (2013) Recent insights into Clostridium difficile pathogenesis. Curr Opin Infect Dis 26:447–453
Rao K, Erb-Downward JR, Walk ST et al (2014) The systemic inflammatory response to Clostridium difficile infection. PLoS One 9:e92578
Walker AS, Eyre DW, Wyllie DH et al (2013) Relationship between bacterial strain type, host biomarkers, and mortality in Clostridium difficile infection. Clin Infect Dis 56:1589–1600
Gerding DN, Johnson S, Rupnik M, Aktories K (2014) Clostridium difficile binary toxin CDT: mechanism, epidemiology, and potential clinical importance. Gut Microbes 5:15–27
Vedantam G, Clark A, Chu M, McQuade R, Mallozzi M, Viswanathan VK (2012) Clostridium difficile infection: toxins and non-toxin virulence factors, and their contributions to disease establishment and host response. Gut Microbes 3:121–134
Abou Chakra CN, Pepin J, Sirard S, Valiquette L (2014) Risk factors for recurrence, complications and mortality in Clostridium difficile infection: a systematic review. PLoS One 2014 9:e98400
O’Keefe SJ (2010) Tube feeding, the microbiota, and Clostridium difficile infection. World J Gastroenterol 16:139–142
Huang H, Wu S, Chen R et al (2014) Risk factors of Clostridium difficile infections among patients in a university hospital in Shanghai, China. Anaerobe 30 C:65–69
Zilberberg MD, Nathanson BH, Sadigov S, Higgins TL, Kollef MH, Shorr AF (2009) Epidemiology and outcomes of Clostridium difficile-associated disease among patients on prolonged acute mechanical ventilation. Chest 136:752–758
Hensgens MP, Dekkers OM, Goorhuis A, Lecessie S, Kuijper EJ (2014) Predicting a complicated course of Clostridium difficile infection at the bedside. Clin Microbiol Infect 20:O301–O308
Kenneally C, Rosini JM, Skrupky LP et al (2007) Analysis of 30-day mortality for Clostridium difficile-associated disease in the ICU setting. Chest 132:418–424
Zilberberg MD, Shorr AF, Micek ST, Doherty JA, Kollef MH (2009) Clostridium difficile-associated disease and mortality among the elderly critically ill. Crit Care Med 37:2583–2589
Sabau L, Meybeck A, Gois J et al (2014) Clostridium difficile colitis acquired in the intensive care unit: outcome and prognostic factors. Infection 42:23–30
Zilberberg MD, Reske K, Olsen M, Yan Y, Dubberke ER (2014) Development and validation of a recurrent Clostridium difficile risk-prediction model. J Hosp Med 9:418–423
Davies KA, Davis GL, Longshaw CM et al (2014) Second report from the EUropean, multi-centre, prospective bi-annual point prevalence study of Clostridium difficile Infection in hospitalised patients with Diarrhoea (EUCLID). Presented at 24th European Congress on Clinical Microbiology and Infectious Diseases, Barcelona, 10–13 May 2014. (Abstract P0753)
Barbut F, Surgers L, Eckert C et al (2014) Does a rapid diagnosis of Clostridium difficile infection impact on quality of patient management? Clin Microbiol Infect 20:136–144
Crobach MJ, Dekkers OM, Wilcox MH, Kuijper EJ (2009) European Society of Clinical Microbiology and Infectious Diseases (ESCMID): data review and recommendations for diagnosing Clostridium difficile-infection (CDI). Clin Microbiol Infect 15:1053–1066
Public Health England (2013) Updated guidance on the management and treatment of Clostridium difficile infection. London; PHE. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/321891/Clostridium_difficile_management_and_treatment.pdf. Accessed 17 October 2014
Beitz JM (2006) Fecal incontinence in acutely and critically ill patients: options in management. Ostomy Wound Manage 52:56–58, 60, 62–66
Neal MD, Alverdy JC, Hall DE, Simmons RL, Zuckerbraun BS (2011) Diverting loop ileostomy and colonic lavage: an alternative to total abdominal colectomy for the treatment of severe, complicated Clostridium difficile associated disease. Ann Surg 254:423–427
Vonberg RP, Kuijper EJ, Wilcox MH et al (2008) Infection control measures to limit the spread of Clostridium difficile. Clin Microbiol Infect 14(Suppl 5):2–20
Dubberke ER, Carling P, Carrico R et al (2014) Strategies to prevent Clostridium difficile infections in acute care hospitals: 2014 Update. Infect Control Hosp Epidemiol 35:628–645
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Wilcox, M.H., Vehreschild, M.J.G.T., Nord, C.E. (2015). Clostridium difficile Infection. In: Vincent, JL. (eds) Annual Update in Intensive Care and Emergency Medicine 2015. Annual Update in Intensive Care and Emergency Medicine 2015, vol 2015. Springer, Cham. https://doi.org/10.1007/978-3-319-13761-2_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-13761-2_3
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-13760-5
Online ISBN: 978-3-319-13761-2
eBook Packages: MedicineMedicine (R0)