Abstract
Clostridioides (formerly Clostridium) difficile is an increasingly common cause of diarrheal illness ranging in presentation from simple diarrhea to toxic pseudomembranous colitis in the most severe cases. The past several years have seen the association of strains such as ribotype 027 with more severe infections. This chapter will cover the epidemiology of this disease, as well as the most current testing strategies, medical and surgical treatment, and the evolving role for fecal microbiota transplantation.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Clostridioides difficile
- Risk factors
- Colectomy
- Antibiotic treatment
- Fecal microbiota transplant
- Fidaxomicin
-
More virulent strains of C. difficile such as ribotype 027 are associated with severe clinical infection.
-
CDI in IBD patients should be treated with vancomycin.
-
CDI is often associated with an exaggerated leukocytosis.
-
Fecal transplant is effective therapy for recurrent CDI.
Introduction
Clostridioides (formerly Clostridium) difficile is an anaerobic, Gram-positive rod bacterium, which often is part of normal colonic flora, but can become a pathogenic organism under the appropriate circumstances when the microbiome is altered. While this was once an uncommon clinical entity, Clostridium difficile infection (CDI) is now responsible for at least 20% of the cases of antibiotic-associated diarrhea, and its incidence continues to steadily rise [1]. As the incidence of this disease has increased, the emergence of more virulent strains, such as ribotype 027, has led to increasingly severe clinical presentations [2,3,4]. As we have seen evolutions in the Clostridium difficile bacterium, management strategies have drifted away from the older recommendation of metronidazole as a first-line agent to newer strategies using vancomycin or fidaxomicin [5,6,7,8,9]. This chapter will discuss the epidemiology of CDI and the most pertinent clinical risk factors. We will discuss current microbiological nomenclature and the evolving testing strategies. Finally, we will discuss the current best recommendations for medical and surgical treatment of this disease, as well as the evolving role for fecal microbiota transplantation (FMT) [10] as an alternative to classical medical and surgical options for CDI.
Epidemiology
Evidence is mounting that both the incidence and severity of CDI are increasing [11,12,13]. In fact, recent evidence suggests that in many hospital settings, CDI has now become the most common cause of hospital-acquired infection, surpassing methicillin-resistant Staphylococcus aureus (MRSA) infection [14, 15]. Data from the Centers for Disease Control and Prevention (CDC) suggested a fourfold increase in CDI from 1993 to 2009, while the incidence in those over age 65 increased by over 200% [16].
In addition to an overall increasing incidence, there appears to be an increasing severity as well. At least one ribotype (ribotype 027) has been associated with >15 times the production of toxins A and B compared to other strains, as well as producing a third toxin called binary toxin. This strain has been associated with a higher incidence of toxic colitis and increased mortality [17, 18]. A study by Falcone examined clinical response to antibiotic treatment in 027+ vs. 027− subtypes. Overall, metronidazole monotherapy (HR 2.38, 95% CI 1.55–3.60, p < 0.001) and immunosuppressive treatment (HR 3.11, 95% CI 1.91–5.09, p < 0.001) were associated with recurrent CDI in ribotype 027-positive patients [4]. The authors advocated for vancomycin as primary treatment over metronidazole in cases where ribotype 027 is identified.
The increasing incidence of this condition is both clinically and financially important. A recent meta-analysis by Nanwa examined studies from 1988 to 2014 to determine the economic impact of CDI. The main outcome was the total direct cost attributable from hospital stays for CDI. Attributable direct costs of CDI ranged from $8911 to $30,049 [19]. A second study by McGlone agreed with these estimates for cost of a hospitalization and extrapolated to an annual US economic burden of $496 million (hospital perspective) and $547 million from a third-party payer perspective [20]. When both estimates from an inpatient and outpatient perspective are considered, the cost to the US healthcare system has been estimated to be a staggering $3 billion [21]. Given the increasing incidence and cost of CDI, it is important to fully understand the most current risk factors, diagnostic testing, and treatment strategies.
Clinical Risk Factors
Advanced Age
Though increasing age is commonly cited as a risk factor for CDI [22,23,24,25], patients of any age may be affected. The majority of the literature on age in CDI suggests that both young and older patients with CDI have similar clinical presentations and comorbidities, indicating that medical comorbidities rather than age may be the most important risk factors predisposing to CDI [22, 24]. Lee compared patients younger than 65 to those older than 65 and found more severe colitis in older patients and more frequent failure of first-line treatment, suggesting the need for more aggressive initial treatment in older patients hospitalized with CDI [23]. Louie randomized patients to be treated with vancomycin or fidaxomicin for severe CDI. Compared to patients age 18–40, clinical cure rate was lower, and clinical recurrence was higher for each successive decade [25].
Antibiotic Treatment
The most consistent and potentially modifiable risk factor for the development of CDI is antibiotic use. Though the most commonly reported antibiotics implicated are clindamycin, fluoroquinolones, carbapenems, and cephalosporins [26,27,28,29,30,31,32,33,34], it appears that almost any antibiotic, used over any period of time, can be associated with the development of CDI. A recent systematic review compared the impact of different classes of antibiotics on the development of CDI in the setting of randomized trials. The results compared all classes of antibiotics and quantified individual risks of CDI. Clindamycin and carbapenems appeared to be the most strongly associated antibiotics with CDI [28]. Another contemporary review by Slimings of 13 heterogeneous studies indicated that second-, third-, and fourth-generation cephalosporins (OR 3.2), clindamycin (OR 2.8), and fluoroquinolones (OR 1.6) were the most commonly cited agents [35]. Though these studies are quite heterogeneous and it is difficult to assess the impact of antibiotics in comparison of the other myriad of risk factors, it does serve to remind us that antibiotics should be used judiciously in both the prophylactic and therapeutic settings and that inciting antibiotics should be discontinued as soon as possible once CDI is diagnosed [26, 28,29,30,31,32,33,34]. Though it is postulated that antibiotics cause changes in colonic bacterial flora leading to susceptibility to CDI, most of the available data is descriptive in nature. Certain species of bacteria such as Bacteroides, Bifidobacteriae, and Lachnospiraceae seem to be the most prevalent among species which confer resistance to C. difficile colonization [36].
There is some degree of controversy as to whether probiotics should be used for primary prevention of CDI when a course of antibiotic therapy is prescribed. There are meta-analyses suggesting that probiotics could decrease the incidence of CDI when given to patients on antibiotics with no prior history of CDI [37,38,39]. Arguments against this strategy suggest that the incidence of CDI in hospitalized patients > age 65 on antibiotics tends to be <3% even without probiotics [40]. Additionally, the meta-analyses contained some studies with higher than expected incidence of CDI; excluding these studies makes the magnitude of effect of probiotics much less impressive. Additionally, due to the heterogeneity in types of probiotic and the numerous clinical confounding factors, it is difficult to say at this time whether these agents should be used as a means of primary prevention of CDI.
Contact with a Healthcare Facility
CDI is common both in acute care and longer-term health facility stays. This is likely due to a concentration of patients with the typical risk factors for CDI, as well as the transmission from patient to patient via the fecal-oral route. In addition to the usual clinical risk factors, hospital-related factors such as increasing bed occupancy have been associated with an increased risk of developing CDI [41]. It is increasingly clear that hospitals need to be aware of the potential to transmit CDI among patients and to have proper infection control practices in place [42]. These practices include antibiotic stewardship, contact precautions in the appropriate setting, hand washing, disinfection practices, and CDI treatment protocols. Though literature has shown decreased incidence in CDI when proper disinfection practices [43], antibiotic stewardship [30, 44], and hand washing with soap rather than alcohol-based disinfectants [42, 45] decrease the incidence of CDI, not all hospitals follow these practices. A recent study by Aquina used a New York statewide database to study 150,878 patients in New York having either a segmental colectomy or proctectomy. C. difficile incidence ranged from 0% to 11.3% among surgeons and 0% to 6.8% among hospitals. Importantly, patient factors only explained 24% of the variation, while approximately 70% of the variation was from unexplained hospital factors [46]. This highlights the need to be vigilant when caring for hospitalized patients, as the risks for CDI transmission are significant.
Immunocompromised States
Regardless of the cause, compromise of the immune system appears to be associated with the development of CDI. It remains unclear whether immunosuppression alone is sufficient for the development of CDI, or whether immunocompromised patients frequently become hospitalized and have several of the other clinical risk factors for CDI. HIV patients seem to be at risk to develop CDI, with several of the common risk factors for infection also being cited such as low serum albumin, clindamycin use, prolonged hospital stay, and proton pump inhibitor use [47,48,49,50]. The strongest risk factor for CDI in HIV patients appears to be a CD4 count ≤50/mm3, with an adjusted odds ratio of 5.2–27.6 [48, 51]. There are many reports in the literature of CDI in other settings of immunocompromise such as general oncology patients [52], solid organ transplant recipients [53, 54], or stem cell transplant recipients [55,56,57]. Although these patients do have compromised immune systems, most of them seem to exhibit the classic risk factors for CDI as well such as prolonged hospitalization and antibiotic use [52, 54, 55, 57].
Inflammatory Bowel Disease
CDI has been increasing in inflammatory bowel disease (IBD) patients and may be associated with increased morbidity, mortality, and need for surgery [58, 59]. Patients with both Crohn’s disease and ulcerative colitis both appear to be at risk, and clinical presentation with CDI most often happens during an acute disease exacerbation. There is some evidence that specific genetic polymorphisms may be associated with the development of CDI. One such study noted that the TNFRSF14 locus was associated with a sixfold increase in development of CDI [60]. Some of the clinical risk factors for the development of CDI in the setting of IBD are low serum albumin, hemoglobin level below 9 g/dl, active colitis from IBD, biologic use, and antibiotic use [61, 62].
Though most studies consistently show a relationship between active colitis, steroid use, and antibiotic use with CDI, some studies failed to show an association between biologic medication use and CDI [63]. Testing for CDI is recommended in patients with IBD and severe colitis. Though the evidence supports the early administration of vancomycin to treat the CDI [64], these patients need close clinical monitoring in the hospital, as at least one study has estimated a sixfold increase in the need for colectomy in this setting [65].
Perioperative Prophylactic Antibiotics and Mechanical Bowel Preparation
In preparation for elective colectomy , patients for several decades have typically received a combination of mechanical and oral antibiotic bowel preparation [66]. This was thought to minimize infectious complications by decreasing both the load of stool and bacteria in the colon. For many years, bowel preparation was used less and less, as the oral antibiotic preparation was abandoned. Recently, many studies have highlighted improved outcomes with mechanical bowel preparation and oral antibiotics including decreased surgical site infection (SSI) and anastomotic leak [67,68,69,70,71]. Some data also suggests that even in the absence of a mechanical bowel preparation, oral antibiotics may still decrease the incidence of SSI following colectomy [72]. It is possible that beneficial changes in the microbiome may be responsible for these improved surgical outcomes [73, 74]. However, there are some concerns that changes in the colonic microbiome may make a patient more susceptible to pathogens such as C. difficile. Literature on this topic is quite sparse. While on one hand, a single study by Morris noted that 9.5% of CDI patients had only preoperative oral antibiotic utilization as the only clinical risk factor [75], this likely does not outweigh the benefits of these agents in reducing SSI and potentially anastomotic leak. Further, the comparative data described above actually showed that patients receiving mechanical bowel preparation with oral antibiotics had a lower incidence of CDI postoperatively.
Likely the most important factor in reducing CDI is the appropriate adherence to guidelines and cessation of perioperative antibiotics as soon as feasible. A study by Balch showed that patients who had perioperative antibiotics continued for greater than 24 hours had a 6.7-fold increase in the incidence of CDI compared to those who received 24 hours of therapy alone [27].
Proton Pump Inhibitors
There appears to be a clinical association between proton pump inhibitors and CDI, though it is difficult to directly attribute causation owing to the numerous other clinical risk factors that are often present. The exact mechanism for this association remains unclear, but at least one recent in vitro study by Stewart indicated that omeprazole stimulated the production of C. difficile toxins in both acidic and basic environments [76]. Since many conflicting studies have been published, there were 4 recent meta-analyses performed, combining data on over 300,000 patients [77,78,79,80]. These analyses were limited due to significant heterogeneity of data, and two of the studies noted publication bias. Combined studies suggest an odds ratio of 1.6–1.7 for the development of CDI for hospitalized patients on PPIs [77,78,79,80].
One observational study by Chitnis examined 984 cases of community-acquired CDI. In this population, 36% had not received antibiotics. The most common risk factor was use of PPI medication, which was present in 31% of patients [81]. Though there appears to be a clinical correlation between use of PPIs and development of CDI, the quality of the evidence is poor. Randomized clinical studies with proper consideration of other clinical risk factors are lacking. Though this does not support the global discontinuation of PPIs in hospitalized patients, physicians should be encouraged to use these medications judiciously, when there is a clinical indication to do so. Many patients are routinely placed on PPI medication as a means of “prophylaxis” or for other questionable clinical indications [82]. The risk of CDI could be a reason to call these practices into question.
Nomenclature and Genetics of C. difficile Infection
The proper genus designation for this pathogen is Clostridioides and not Clostridium. This change in terminology, introduced in 2016, was prompted by phylogenetic studies indicating that the genus Clostridium should be restricted to Clostridium butyricum and similarly evolutionarily related species that shared genetic and functional characteristics common to Clostridium cluster [83,84,85]. Despite being an anaerobic, spore-forming, Gram-positive rod, Clostridioides has more in common with genus Peptoclostridium. Its official reassignment to this genus never occurred, however, due to concerns that the pathogen’s former name recognition would make the introduction of such a different moniker a source of confusion and generate a significant financial burden to research and medical fields due to re-labelling costs. Therefore, a new genus was proposed (Clostridioides), one similar enough to the former name to allow continued broad recognition and one clever enough to allow the continued use of the term “C. difficile.”
CDI is clinically characterized by a colitis that is, in large part, mediated by bacterial envenomation. The genetic basis for C. difficile toxin production includes a 19.6 kb region known as the pathogenicity locus (PaLoc) that contains the genes for clostridial toxins A (tcdA) and B (tcdB) [86, 87]. These toxin genes, and the regulatory genes that increase and decrease their expression, are actually of bacteriophage (viral) origin [88, 89]. With successive replicative errors, loss of portions of these previously viral genes, under selective pressure, resulted in their retention as bacterial genes that increase the fitness of C. difficile [90]. Some, though not all, strains of C. difficile can produce an additional toxin known as binary toxin (CDT) encoded by genes outside of the PaLoc. Toxins A (308 kDa) and B (269 kDa) are classified as large clostridial toxins due to their larger molecular weight [91]. Both of these toxins initiate monoglucosylation of a variety of intracellular Rho GTPases that result in depolymerization of cytoskeletal elements, with resultant cytopathy of colonocytes [92]. Binary toxin has an ADP-ribosyltransferase function that requires internalization by colonocytes; to promote this internalization, binary toxin is able to induce colonocytes to alter the apical aspect of their cell membrane to produce microtubular protrusions, increasing the membrane surface area up to fivefold and promoting the adherence of both C. difficile and its toxins, promoting further mucosal damage [93, 94]. Although updated studies are needed, the most current data suggests that binary toxin is present in up to 6% of all C. difficile isolates [95]. In virtually all symptomatic infections among humans, toxins A and B will be present.
Ribotype and Clinical Severity
Although more frequently incorporated into research than clinical care, efforts at characterizing the dominant C. difficile strain on the basis of bacterial genotype have most commonly involved a process called ribotyping, a process using restriction enzymes to characterize the heterogeneity of the bacterial ribosomal intergenic spacer region [96, 97]. The most frequently identified ribotype associated with severe forms of C. difficile infection is ribotype 027 [98]. Though there are exceptions to the following, several associations between this ribotype and the clinical characteristics of CDI have emerged [99]. Ribotype 027 is the most frequently encountered strain among patients admitted from long-term healthcare facilities, with one study identifying an odds ratio of 4.87 for 027 being present compared to patients admitted with CDI from a private residence [100]. This may be a reflection of this ribotype having a selective advantage in terms of colonization and the promotion of a carriage state. Ribotype 027 is able to outcompete endemic bacteria for resources while producing larger volumes of bacterial toxins and producing higher rates of symptomatic infections; this strain also frequently has the ability to produce binary toxin in addition to toxins A and B [101]. Investigations focused on comparative bacterial genomics reveals that ribotype 027 has more than 200 genes not found in other strains of C. difficile, with many of these genes having a plausible role in promoting virulence [102, 103]. Other ribotypes, such as 078, represent potentially virulent strains that also have a zoonotic link between human and animal CDI. C. difficile as a pathogen has soil, animal, and human reservoirs, creating an important interaction between humans and their environment in terms of the emergence of new virulent strains of this bacteria [104, 105].
Diagnosis of C. difficile Infection
CDI is a disease capable of producing both toxin-mediated and toxin-independent forms of colitis (160), and thus the hallmark of symptomatic infection is diarrhea. The term “C. difficile infection” should be kept distinct from carrier states, defined as patients without symptoms of infection who nonetheless also have a positive stool test for C. difficile. The exact incidence of carrier states in the general population is not known, though small studies of patient populations at risk for CDI suggest the incidence is not small. In 1 study of geriatric patients without diarrhea, 43 (16.4%) out of 262 consecutive patients tested positive for toxin B based on PCR stool testing. Of those 43 patients, 7 (16.3%) eventually developed symptomatic CDI, confirming that carrier states are both more common among patients with frequent healthcare facility contacts and predispose patients to symptomatic infection [106]. Studies have suggested that patients who are able to form antibodies to C. difficile toxin A are more likely to remain asymptomatic carriers compared to patients who develop symptoms of CDI [107].
Diarrhea, which is typically grossly non-bloody in this disease, is the primary symptom of CDI. Depending on the severity of CDI, other findings will include abdominal distention, abdominal pain that is frequently colicky due to colitis, tachycardia, and hypotension. In fulminant cases, localized or generalized peritonitis may develop, which serves as an indication for surgery. CT findings include colonic wall thickening and pericolic fat stranding, as would be observed with any form of colitis. Colonic wall thickening is characteristic of severe forms of CDI and can provide a heightened index of suspicion for CDI prior to the results of stool testing. Transudative ascites is frequently associated with more severe forms of colitis. Pneumoperitoneum and portal venous gas are rarer radiographic findings and are generally encountered in patients with fulminant forms of the infection with septic shock and the need for vasopressors; the patients often develop non-viable colon due to severe mesenteric vasoconstriction.
Diagnostic Tests for CDI
In general, the diagnosis of CDI is founded on a clinical suspicion with support from specific laboratory testing. The original confirmatory test for CDI was bacterial culture, which proved to be both difficult and was plagued by a lengthy time interval to final results. This technique was replaced by the cell culture cytotoxicity neutralization assay (CCCNA). This approach uses a filtrate of patient stool applied to a monolayer of one of various cell lines. After a 24–48-hour incubation period, cells are evaluated for evidence of cytopathy attributable to C. difficile toxins. If cytopathic changes are observed, a neutralization assay is then performed to assure that these changes are due to C. difficile toxins. Historically, this technique was considered to be the gold standard; but with sensitivities as low as 65%, this test has been replaced by more sensitive tests that provide more expeditious results and do not require technical expertise that may limit generalizability [108].
Another, now outdated, method involves toxigenic culture. Though there are multiple methods to accomplish this test, all of them focus on isolating C. difficile from stool samples and confirming the presence of its toxin. There were numerous steps and difficulties with this approach. First, there is no one superior approach to isolating C. difficile from candidate stool samples. Secondly, once C. difficile colonies are isolated, those colonies have to be evaluated for their ability to produce toxin. This technique is considered by some to be a gold standard in terms of serving as a reference for new testing methods, though in terms of current practice, it has been supplanted by simpler, quicker, and less exacting approaches.
A current method of testing involves immunoassays that detect glutamate dehydrogenase (GDH), a conserved metabolic enzyme ubiquitous among C. difficile. As it is present among toxigenic and non-toxigenic forms of C. difficile, GDH alone lacks specificity for clinical purposes. Therefore, GDH is a useful screening test that, when positive, must be followed by a confirmatory test, which generally involves a toxin enzyme immunoassay (EIA). This approach allows for the use of a GDH as a high sensitivity test (80–100%) with an excellent negative predictive value, with limited cost and limited requirement for expertise and with rapid turnaround. When combined with toxin EIA, the cost per test remains less than PCR-based diagnostics while providing rapid results (<24 hours) and excellent sensitivity.
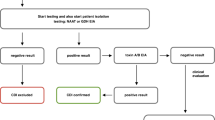
Nucleic acid amplification tests (NAATs) are a collection of PCR-based diagnostics that are designed to amplify highly conserved C. difficile genes, such as those related to toxins A (tcdA) and B (tcdB). The sensitivity of these tests is greater than EIA and possibly GDH-EIA combinations, though their cost is two to three times greater than GDH-EIA, and NAATs may require longer result times depending on the particular test considered. To leverage sensitivity, specificity, and cost-related issues, many hospitals have adopted an algorithmic approach, where diarrheal stools are first tested using a GDH assay. A negative result marks the end of the testing algorithm, while a positive GDH assay is followed by a confirmatory test, usually a toxin EIA. A positive GDH assay followed by a positive toxin EIA in patients with symptoms of CDI is treated as a case of CDI. A stool that is GDH positive and toxin EIA negative is tested using a NAAT. Routine retesting with NAATs to confirm resolution of CDI is not recommended, given the potential for bacterial DNA to linger for as long as 30 days resulting in patients who no longer have CDI but who test positive for this infection.
Clinical Measures of Severity
Several clinical severity scoring systems are available, though none has emerged clearly superior to the others. Perhaps the two most commonly utilized are the 2017 Infectious Disease Society of America (IDSA) guidelines [109] and those published by the American College of Gastroenterology (ACG) [21]. In the IDSA schema, initial episodes are classified as (1) non-severe, if a patient’s white blood cell count is <15,000 cells/mL and if their serum creatinine is <1.5 mg/dL; (2) severe, if a patient’s white blood cell is >15,000 cells/mL and if their serum creatinine is >1.5 mg/dL; or (3) fulminant, if the patient demonstrates evidence of cardiovascular shock, ileus, or megacolon. The ACG schema defines severe CDI as patients with a white blood cell count of ≥15,000 cells/mL or a serum albumin of <3 g/dL. Fulminant disease is then defined as any patient requiring ICU admission, with a temperature of ≥38.5 °C, ileus, significant abdominal distention, altered mental status, a white blood cell count of >35,000 cells/mL or <2000 cells/mL, a serum lactate of >2.2 mmol/L, or any evidence of organ dysfunction. Using scoring systems such as these offers the potential advantages of standardizing patient assessments across providers and institutions.
Antibiotic Therapy for CDI
For more than 20 years, the two principal antibiotics used to treat CDI were metronidazole and vancomycin. Data has accumulated to suggest that vancomycin is the superior treatment option compared to metronidazole. For example, in a 2007 randomized, prospective, double-blind, placebo-controlled trial by Zar, 150 patients with either mild or severe CDI were randomized to either oral metronidazole (250 mg 4 times daily) or vancomycin (125 mg 4 times daily). While no significant difference in clinical cure rates was noted among patients with mild disease, for study subjects with severe disease, vancomycin was associated with a significantly higher cure rate (97% versus 76%; p = 0.02) [110].
Additionally, retrospective data specifically focused on the treatment of mild disease suggests that vancomycin is superior to metronidazole in these cases as well, with one study reporting that compared to vancomycin, metronidazole was an independent risk factor for treatment failure [110]. Given these data, the most recent IDSA recommendations state “Use of oral metronidazole, however, should be restricted to an initial episode of nonsevere CDI in cases where other therapies are contraindicated or not available, and treatment should be limited to one course due to case reports of neurotoxicity with prolonged or repeated use.” Between these two drugs, vancomycin has emerged as the recommended drug for mild and severe forms of CDI [109].
Fidaxomicin is a macrocyclic drug designed to exhibit a narrow antibiotic spectrum mediated through inhibition of the sigma subunit of RNA polymerase. The first Phase III study comparing fidaxomicin to vancomycin enrolled 629 subjects, demonstrating that fidaxomicin was non-inferior to vancomycin with respect to clinical cure rates [111]. Additionally, lower recurrence rates were noted with fidaxomicin, though only with strains that were not NAP-1 (a nomenclature of strains associated with more virulent infections). A more recent study (the EXTEND study) evaluated 364 patients randomly assigned to either vancomycin or extended pulsed fidaxomicin. Fidaxomicin was associated with an 11% improvement in clinical cure at 30 days after the end of treatment compared to vancomycin [112]. Further, fidaxomicin was just as safe as vancomycin, with no difference in treatment-related adverse events. A recent Cochrane Database Systematic Review evaluated 22 studies representing a total of 3215 patients. This study concluded that “moderate quality evidence suggests that vancomycin is superior to metronidazole and fidaxomicin is superior to vancomycin. The differences in effectiveness between these antibiotics were not too large and the advantage of metronidazole is its far lower cost compared to the other two antibiotics” [113].
For a first episode of non-severe CDI, the best available evidence supports the use of either vancomycin 125 mg orally every 6 hours or fidaxomicin 200 mg every 12 hours, either for 10 days. For fulminant cases, the most effective treatment remains the use of oral vancomycin 500 mg every 6 hours. In many instances, patients with fulminant CDI will develop an ileus, potentially decreasing the safety and efficacy of orally administered drugs. Therefore, for patients with fulminant CDI with concerns regarding the appropriateness of orally administered therapies, vancomycin can be administered as a retention enema (500 mg diluted in 100 cc of normal saline every 6 hours) [114], though the quality and strength of clinical data supporting vancomycin enemas is weak. In the setting of fulminant CDI, the Infectious Diseases Society of America also recommends administering 500 mg of parenteral metronidazole every 8 hours in conjunction with oral or rectal vancomycin, as the risks and side effects of a limited course of metronidazole are small in comparison to the risk of progressively worsening CDI.
Surgery for CDI
The incidence of patients who undergo surgery (Figs. 52.1, 52.2, and 52.3) for CDI has been estimated to be as high as 10% [115,116,117]. Inherent in these estimates are decisions assessing whether a patient has received maximal medical therapy for CDI with surgery remaining as the only option, as well as selecting which patients might meet this qualification but are not considered to be candidates for surgical intervention. Multiple series indicate that only a small percentage of patients with fulminant CDI will undergo surgery; this suggests that the majority of CDI patients will not develop medically refractory disease severe enough to warrant surgery [115,116,117]. With the more recent introduction of diversion and colonic lavage [115], if it becomes more commonplace, it may increase the incidence of surgical intervention for CDI as it may be considered a “more survivable” surgery than total colectomy, which serves as the traditional procedure of choice.
There are two systematic reviews that compare the survival benefit of total abdominal colectomy to continued medical therapy in the setting of fulminant CDI, although the particular vantage points by which they evaluate this question differ in important respects. In a review by Bhangu, 31 studies were included in an effort to compare survival rates among patients with fulminant CDI who either underwent colectomy or who continued with medical therapy. In the surgical cohort, 89% of patients underwent a total colectomy, but patients who underwent a partial colectomy were also included. Findings from this review included a statistically significant association between preoperative clinical findings of septic shock and the incidence of postoperative mortality. Of interest, patients who underwent a partial colectomy experienced a 16% reoperation rate with resection of an additional length of colon. This review provided support for a survival benefit for surgical intervention in cases of fulminant CDI, with total colectomy arguably the preferred form of surgery compared to partial colectomy [118].
A second systematic review described outcomes among 510 patients for the purpose of evaluating whether total colectomy was associated with a survival benefit in the setting of medically refractory, fulminant CDI. The pooled odds ratio for mortality was significantly lower in patients undergoing total colectomy (OR = 0.70) [119]. Importantly, this study excluded patients undergoing partial colectomy.
In 2011, Neal described an alternative to total colectomy, involving the construction of a diverting loop ileostomy allowing for intraoperative colonic lavage with 8 liters of polyethylene glycol, followed by postoperative intraluminal vancomycin provided per stoma. In this study, 42 patients with fulminant CDI were treated with intestinal diversion, and their outcomes were compared to a historical control group of patients treated with colectomy and an end ileostomy. The colectomy and diversion populations demonstrated similar preoperative APACHE scores as well as preoperative clinical indices, suggesting similar degrees of CDI severity. The group undergoing diversion was observed to have a significantly lower postoperative mortality (19% versus 50%) and shorter length of surgery, and 83% of the patients undergoing diversion had their surgeries completed laparoscopically [115]. This study provided the first modern description of diversion for life-threatening colitis of an infectious etiology, providing ostensibly superior survival utilizing a much smaller and more tolerable surgery that would also offer a higher likelihood of restoring gut continuity.
Since the study by Neal and colleagues, a number of primarily retrospective studies on diversion for fulminant CDI have been published using either institutional- or population-level data. One such study was undertaken in 2017 by the EAST Multi-Center Trials Committee that collected data from ten participating centers [120]. Certain details, such as the definition of CDI and the form of stool testing used at the participating centers, were not described. Comparing patients who underwent total colectomy to those who underwent diversion, there were no statistically significant differences in median vital sign measurements, white blood cell counts, lactate levels, INRs, or APACHE scores.
There were no differences between the surgical cohorts with respect to postoperative outcomes such as the rate of any complication, pneumonia, acute renal failure, sepsis, and acute respiratory distress syndrome. Just as importantly, there was no difference in rates of overall as well as unplanned reoperations, while ventilator days, ICU, and hospital lengths of stay were also similar between these groups. Unadjusted mortality was comparable between the cohorts (23.8% in the diversion cohort and 33.8% in the colectomy cohort; p = 0.44). The authors of this study also performed calculations for what they termed adjusted mortality, which involved an inverse probability of treatment weights propensity score analysis. With this adjustment, mortality was significantly lower among the diversion group (17.2% versus 39.7%; p = 0.002) [120].
A 2019 study using data from the Nationwide Inpatient Sample suggests that more surgeons are adopting diversion as the surgery of choice for fulminant CDI. In a retrospective review of this dataset from 2011 to 2015, 2408 patients were identified as undergoing surgery for CDI. Of these, 613 patients (approximately 20% of the study population) underwent diversion with a loop ileostomy; during the study period, the use of this procedure increased from 11% in 2011 to 25% in 2015 [121]. Although important details regarding the severity of CDI and the management of study subjects were not available using this data source, the authors also reported that in-hospital mortality did not significantly differ between these cohorts. One important limitation with the data source used for this study was the inability to identify the selection criteria used for choosing the form of surgical intervention, which has implications for the measured outcomes such as mortality rates.
In summary, patients with severe, complicated/fulminant CDI should be managed in a multidisciplinary fashion. Surgical consultation is recommended when CDI of this severity is first recognized, in an effort to allow for surgical intervention at the earliest appropriate time. CDI is frequently associated with organ failure, including hematologic failure characterized by an exaggerated leukocytosis that is often >30,000 cells/mm. It is critical that patients are euvolemic when assessing severity of sepsis, with a greater emphasis on trending organ function than on absolute cut-off values for laboratory abnormalities. De-escalating or discontinuing antibiotics that are not C. difficile targeted is also important; though the data is limited [122], CDI outcomes are worse when the inciting antibiotics are continued while CDI is being treated. While de-escalation of antibiotics is not always appropriate, certain infections (mild bladder infections) that are not life-threatening can have their treatment deferred, while life-threatening CDI is first addressed.
When patients are deemed to be too ill to allow for ongoing medical therapy, or demonstrate continued deterioration, surgery should be recommended for patients who are candidates for surgery. The largest body of evidence supports the use of total abdominal colectomy with an end ileostomy; partial colectomies have an extremely limited utility given a significantly higher incidence of reoperation due to postoperative fulminant CDI. Loop ileostomy has enough data at this juncture to be an acceptable alternative to total colectomy, though as described in a letter to the editor [123] in response to the recent EAST study [120] on loop ileostomy, there are several unanswered questions regarding this newer surgery. In the EAST study, diversion provided no advantage compared to total colectomy in terms of postoperative sepsis, renal failure, acute lung injury, overall mortality, and mortality related to unplanned reoperation. This may indicate that the particular strain of C. difficile, which is information frequently missing from the surgical literature on CDI, may influence postoperative outcomes, especially when a diverting stoma is created and an infected colon is left in situ.
Fecal Microbiota Transplant (FMT)
With access to commercially screened and prepared stool from vendors, FMT in either a liquid form for endoscopic or nasogastric application, or in capsule form for oral consumption, is now more readily available for inpatient use than in earlier times when providers had to collect, screen, and prepare stool from donors. FMT is extremely effective for treating recurrent CDI, with cure rates greater than 80% routinely described [124]. FMT for fulminant CDI has the least exploration of all FMT applications, though the few studies on this topic are promising [125, 126]. One of the challenges with FMT for fulminant CDI is that of disease recrudescence and a measurable mortality from that disease recurrence. The incorporation of vancomycin in addition to FMT for fulminant cases appears to be important for ensuring reliable cure rates [127].
FMT is no longer an experimental therapeutic for outpatients, or inpatients, with CDI. Its cure rates for recurrent forms of CDI are superior to conventional antibiotics, leading some to question whether FMT should be the first intervention to address the recurrence of this infection. Of note, FMT does not currently represent the first-line therapy for a primary case of FMT. The availability of commercial vendors who can provide screened and prepared transplant material has simplified the process of FMT, making it more accessible for clinicians. The use of inpatient FMT for inpatient cases of CDI has increased in recent years, though physicians continue to treat the majority of cases of this infection with conventional antibiotics. At this time, there is insufficient data to recommend the routine use of FMT for the treatment of fulminant CDI; though there are limited numbers of small series that suggest safety and efficacy with FMT, more data in this patient population is required before FMT can be recommended for patients with life-threatening CDI.
References
Luo R, Barlam TF. Ten-year review of Clostridium difficile infection in acute care hospitals in the USA. J Hosp Infect. 2005-2014;98:40–3.
Pepin J, Valiquette L, Gagnon S, Routhier S, Brazeau I. Outcomes of Clostridium difficile-associated disease treated with metronidazole or vancomycin before and after the emergence of NAP1/027. Am J Gastroenterol. 2007;102:2781–8.
Krutova M, Matejkova J, Tkadlec J, Nyc O. Antibiotic profiling of Clostridium difficile ribotype 176--A multidrug resistant relative to C. difficile ribotype 027. Anaerobe. 2015;36:88–90.
Falcone M, Tiseo G, Iraci F, Raponi G, Goldoni P, Delle Rose D, Santino I, Carfagna P, Murri R, Fantoni M, Fontana C, Sanguinetti M, Farcomeni A, Antonelli G, Aceti A, Mastroianni C, Andreoni M, Cauda R, Petrosillo N, Venditti M. Risk factors for recurrence in patients with Clostridium difficile infection due to 027 and non-027 ribotypes. Clin Microbiol Infect. 2019;25:474–80.
Wilcox MH, Rooney CM. Comparison of the 2010 and 2017 Infectious Diseases Society of America guidelines on the diagnosis and treatment of Clostridium difficile infection. Curr Opin Gastroenterol. 2019;35:20–4.
Steele SR, McCormick J, Melton GB, Paquette I, Rivadeneira DE, Stewart D, Buie WD, Rafferty J. Practice parameters for the management of Clostridium difficile infection. Dis Colon Rectum. 2015;58:10–24.
Martin M, Zingg W, Knoll E, Wilson C, Dettenkofer M, Group PS. National European guidelines for the prevention of Clostridium difficile infection: a systematic qualitative review. J Hosp Infect. 2014;87:212–9.
Madden GR, Poulter MD, Sifri CD. Diagnostic stewardship and the 2017 update of the IDSA-SHEA Clinical Practice Guidelines for Clostridium difficile Infection. Diagnosis (Berl). 2018;5:119–25.
Bauer MP, Kuijper EJ, van Dissel JT, European Society of Clinical M, Infectious D. European Society of Clinical Microbiology and Infectious Diseases (ESCMID): treatment guidance document for Clostridium difficile infection (CDI). Clin Microbiol Infect. 2009;15:1067–79.
Camacho-Ortiz A, Gutierrez-Delgado EM, Garcia-Mazcorro JF, Mendoza-Olazaran S, Martinez-Melendez A, Palau-Davila L, Baines SD, Maldonado-Garza H, Garza-Gonzalez E. Randomized clinical trial to evaluate the effect of fecal microbiota transplant for initial Clostridium difficile infection in intestinal microbiome. PLoS One. 2017;12:e0189768.
Kuijper EJ, Coignard B, Tull P, difficile ESGfC, States EUM, European Centre for Disease P, Control. Emergence of Clostridium difficile-associated disease in North America and Europe. Clin Microbiol Infect. 2006;12(Suppl 6):2–18.
Freeman J, Bauer MP, Baines SD, Corver J, Fawley WN, Goorhuis B, Kuijper EJ, Wilcox MH. The changing epidemiology of Clostridium difficile infections. Clin Microbiol Rev. 2010;23:529–49.
Gravel D, Miller M, Simor A, Taylor G, Gardam M, McGeer A, Hutchinson J, Moore D, Kelly S, Boyd D, Mulvey M, Canadian Nosocomial Infection Surveillance P. Health care-associated Clostridium difficile infection in adults admitted to acute care hospitals in Canada: a Canadian Nosocomial Infection Surveillance Program Study. Clin Infect Dis. 2009;48:568–76.
Magill SS, Edwards JR, Beldavs ZG, Dumyati G, Janelle SJ, Kainer MA, Lynfield R, Nadle J, Neuhauser MM, Ray SM, Richards K, Rodriguez R, Thompson DL, Fridkin SK, Emerging Infections Program Healthcare-Associated I, Antimicrobial Use Prevalence Survey T. Prevalence of antimicrobial use in US acute care hospitals, May-September 2011. JAMA. 2014;312:1438–46.
Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile Infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32:387–90.
Gupta A, Khanna S. Community-acquired Clostridium difficile infection: an increasing public health threat. Infect Drug Resist. 2014;7:63–72.
Warny M, Pepin J, Fang A, Killgore G, Thompson A, Brazier J, Frost E, McDonald LC. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366:1079–84.
Loo VG, Poirier L, Miller MA, Oughton M, Libman MD, Michaud S, Bourgault AM, Nguyen T, Frenette C, Kelly M, Vibien A, Brassard P, Fenn S, Dewar K, Hudson TJ, Horn R, Rene P, Monczak Y, Dascal A. A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. N Engl J Med. 2005;353:2442–9.
Nanwa N, Kendzerska T, Krahn M, Kwong JC, Daneman N, Witteman W, Mittmann N, Cadarette SM, Rosella L, Sander B. The economic impact of Clostridium difficile infection: a systematic review. Am J Gastroenterol. 2015;110:511–9.
McGlone SM, Bailey RR, Zimmer SM, Popovich MJ, Tian Y, Ufberg P, Muder RR, Lee BY. The economic burden of Clostridium difficile. Clin Microbiol Infect. 2012;18:282–9.
Surawicz CM, Brandt LJ, Binion DG, Ananthakrishnan AN, Curry SR, Gilligan PH, McFarland LV, Mellow M, Zuckerbraun BS. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108:478–98; quiz 99.
Kim HH, Kim YS, Han DS, Kim YH, Kim WH, Kim JS, Kim H, Kim HS, Park YS, Song HJ, Shin SJ, Yang SK, Ye BD, Eun CS, Lee KM, Lee SH, Jang BI, Jung SA, Cheon JH, Choi CH, Huh K, Diseases IBDSGotKAftSoI. Clinical differences in Clostridium difficile infection based on age: a multicenter study. Scand J Infect Dis. 2008;46:46–51.
Lee HC, Kim KO, Jeong YH, Lee SH, Jang BI, Kim TN. Clinical outcomes in hospitalized patients with Clostridium difficile infection by age group. Korean J Gastroenterol. 2016;67:81–6.
Dharmarajan T, Sipalay M, Shyamsundar R, Norkus E, Pitchumoni C. Co-morbidity, not age predicts adverse outcome in clostridium difficile colitis. World J Gastroenterol. 2000;6:198–201.
Louie TJ, Miller MA, Crook DW, Lentnek A, Bernard L, High KP, Shue YK, Gorbach SL. Effect of age on treatment outcomes in Clostridium difficile infection. J Am Geriatr Soc. 2013;61:222–30.
Bernatz JT, Safdar N, Hetzel S, Anderson PA. Antibiotic overuse is a major risk factor for Clostridium difficile infection in surgical patients. Infect Control Hosp Epidemiol. 2017;38:1254–7.
Balch A, Wendelboe AM, Vesely SK, Bratzler DW. Antibiotic prophylaxis for surgical site infections as a risk factor for infection with Clostridium difficile. PLoS One. 2017;12:e0179117.
Vardakas KZ, Trigkidis KK, Boukouvala E, Falagas ME. Clostridium difficile infection following systemic antibiotic administration in randomised controlled trials: a systematic review and meta-analysis. Int J Antimicrob Agents. 2016;48:1–10.
Deshpande A, Pant C, Jain A, Fraser TG, Rolston DD. Do fluoroquinolones predispose patients to Clostridium difficile associated disease? A review of the evidence. Curr Med Res Opin. 2008;24:329–33.
Cruz-Rodriguez NC, Hernández-Garcia R, Salinas-Caballero AG, Perez-Rodriguez E, Garza-Gonzalez E, Camacho-Ortiz A. The effect of pharmacy restriction of clindamycin on Clostridium difficile infection rates in an orthopedics ward. Am J Infect Control. 2014;42:e71–3.
Lee S, Prasad P, Lin M, Garritson S, Nichols A, Liu C. Ertapenem prophylaxis associated with an increased risk of Clostridium difficile infection among surgical patients. Infect Control Hosp Epidemiol. 2015;36:1351–4.
McCusker ME, Harris AD, Perencevich E, Roghmann MC. Fluoroquinolone use and Clostridium difficile-associated diarrhea. Emerg Infect Dis. 2003;9:730–3.
McFarl, LV, Clarridge JE, Beneda HW, Raugi GJ. Fluoroquinolone use and risk factors for Clostridium difficile-associated disease within a Veterans Administration health care system. Clin Infect Dis. 2007;45:1141–51.
Bruns AH, Oosterheert JJ, Kuijper EJ, Lammers JW, Thijsen S, Troelstra A, Hoepelman AI. Impact of different empirical antibiotic treatment regimens for community-acquired pneumonia on the emergence of Clostridium difficile. J Antimicrob Chemother. 2010 Nov;65(11):2464–71.
Slimings C, Armstrong P, Beckingham WD, Bull AL, Hall L, Kennedy KJ, Marquess J, McCann R, Menzies A, Mitchell BG, Richards MJ, Smollen PC, Tracey L, Wilkinson IJ, Wilson FL, Worth LJ, Riley TV. Increasing incidence of Clostridium difficile infection, Australia, 2011-2012. Med J Aust. 2014;200:272–6.
Reeves AE, Koenigsknecht MJ, Bergin IL, Young VB. Suppression of Clostridium difficile in the gastrointestinal tracts of germfree mice inoculated with a murine isolate from the family Lachnospiraceae. Infect Immun. 2012;80:3786–94.
Goldenberg JZ, Ma SS, Saxton JD, Martzen MR, vik PO, Thorlund K, Guyatt GH, Johnston BC. Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst Rev:CD006095.
Johnson S, Maziade PJ, McFarl, LV, Trick W, Donskey C, Currie B, Low DE, Goldstein EJ. Is primary prevention of Clostridium difficile infection possible with specific probiotics? Int J Infect Dis;16:e786–92.
Pattani R, Palda VA, Hwang SW, Shah PS. Probiotics for the prevention of antibiotic-associated diarrhea and Clostridium difficile infection among hospitalized patients: systematic review and meta-analysis. Open Med. 2013;7:e56–67.
Dubberke ER, Reske KA, Noble-Wang J, Thompson A, Killgore G, Mayfield J, Camins B, Woeltje K, McDonald JR, McDonald LC, Fraser VJ. Prevalence of Clostridium difficile environmental contamination and strain variability in multiple health care facilities. Am J Infect Control. 2007;35:315–8.
Ahyow LC, Lambert PC, Jenkins DR, Neal KR, Tobin M. Bed occupancy rates and hospital-acquired Clostridium difficile infection: a cohort study. Infect Control Hosp Epidemiol. 2013;34:1062–9.
Stuart RL, Marshall C, Harrington G, Sasko L, McLaws ML, Ferguson J. ASID/ACIPC position statement – Infection control for patients with Clostridium difficile infection in healthcare facilities. Infect Dis Health. 2019;24:32–43.
Ray AJ, Deshp, e A, Fertelli D, Sitzlar BM, Thota P, Sankar CT, Jencson AL, Cadnum JL, Salata RA, Watkins RR, Sethi AK, Carling PC, Wilson BM, Donskey CJ. A Multicenter Randomized Trial to Determine the Effect of an Environmental Disinfection Intervention on the Incidence of Healthcare-Associated Clostridium difficile Infection. Infect Control Hosp Epidemiol. 2017;38:777–83.
Baur D, Gladstone BP, Burkert F, Carrara E, Foschi F, Dobele S, Tacconelli E. Effect of antibiotic stewardship on the incidence of infection and colonisation with antibiotic-resistant bacteria and Clostridium difficile infection: a systematic review and meta-analysis. Lancet Infect Dis. 2017;17:990–1001.
Brown KA, Daneman N, Jones M, Nechodom K, Stevens V, Adler FR, Goetz MB, Mayer J, Samore M. The Drivers of Acute and Long-term Care Clostridium difficile Infection Rates: A Retrospective Multilevel Cohort Study of 251 Facilities. Clin Infect Dis. 2014;65:1282–8.
Aquina CT, Probst CP, Becerra AZ, Hensley BJ, Iannuzzi JC, Noyes K, Monson JR, Fleming FJ. High Variability in Nosocomial Clostridium difficile Infection Rates Across Hospitals After Colorectal Resection. Dis Colon Rectum. 2017;59:323–31.
Di Bella S, Friedrich AW, Garcia-Almodovar E, Gallone MS, Taglietti F, Topino S, Galati V, Johnson E, D’Arezzo S, Petrosillo N. Clostridium difficile infection among hospitalized HIV-infected individuals: epidemiology and risk factors: results from a case-control study (2002–2013). BMC Infect Dis. 2015;15:194.
Barbut F, Meynard JL, Guiguet M, Avesani V, Bochet MV, Meyohas MC, Delmee M, Tilleul P, Frottier J, Petit JC. Clostridium difficile-associated diarrhea in HIV-infected patients: epidemiology and risk factors. J Acquir Immune Defic Syndr Hum Retrovirol. 1997;16:176–81.
Imlay H, Kaul D, Rao K. Risk factors for Clostridium difficile infection in HIV-infected patients. SAGE Open Med. 2016;4:2050312116684295.
Hutin Y, Molina JM, Casin I, Daix V, Sednaoui P, Welker Y, Lagrange P, Decazes JM, Modai J. Risk factors for Clostridium difficile-associated diarrhoea in HIV-infected patients. AIDS. 1993;7:1441–7.
Haines CF, Moore RD, Bartlett JG, Sears CL, Cosgrove SE, Carroll K, Gebo KA. Clostridium difficile in a HIV-infected cohort: incidence, risk factors, and clinical outcomes. AIDS. 2013;27:2799–807.
Abughanimeh O, Qasrawi A, Kaddourah O, Al Momani L, Abu GM. Clostridium difficile infection in oncology patients: epidemiology, pathophysiology, risk factors, diagnosis, and treatment. Hosp Pract. 1995;46:266–77.
Boutros M, Al-Shaibi M, Chan G, Cantarovich M, Rahme E, Paraskevas S, Deschenes M, Ghali P, Wong P, Fern, ez M, Giannetti N, Cecere R, Hassanain M, Chaudhury P, Metrakos P, Tchervenkov J, Barkun JS. Clostridium difficile colitis: increasing incidence, risk factors, and outcomes in solid organ transplant recipients. Transplantation. 2013;93:1051–7.
Honda H, Dubberke ER. Clostridium difficile infection in solid organ transplant recipients. Curr Opin Infect Dis. 2014;27:336–41.
Aldrete SD, Kraft CS, Magee MJ, Chan A, Hutcherson D, Langston AA, Greenwell BI, Burd EM, Friedman-Moraco R. Risk factors and epidemiology of Clostridium difficile infection in hematopoietic stem cell transplant recipients during the peritransplant period. Transpl Infect Dis. 2017;19(1).
Alonso CD, Dufresne SF, Hanna DB, Labbe AC, Treadway SB, Neofytos D, Belanger S, Huff CA, Laverdiere M, Marr KA. Clostridium difficile infection after adult autologous stem cell transplantation: a multicenter study of epidemiology and risk factors. Biol Blood Marrow Transplant. 2013;19:1502–8.
Alonso CD, Kamboj M. Clostridium difficile Infection (CDI) in Solid Organ and Hematopoietic Stem Cell Transplant Recipients. Curr Infect Dis Rep. 2014;16:414.
Ananthakrishnan AN, Binion DG. Impact of Clostridium difficile on inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2010;4:589–600.
Bossuyt P, Verhaegen J, Van Assche G, Rutgeerts P, Vermeire S. Increasing incidence of Clostridium difficile-associated diarrhea in inflammatory bowel disease. J Crohns Colitis. 2009;3:4–7.
Ananthakrishnan AN, Oxford EC, Nguyen DD, Sauk J, Yajnik V, Xavier RJ. Genetic risk factors for Clostridium difficile infection in ulcerative colitis, Aliment Pharmacol Ther. 2013;38:522–30.
Ananthakrishnan AN, Guzman-Perez R, Gainer V, Cai T, Churchill S, Kohane I, Plenge RM, Murphy S. Predictors of severe outcomes associated with Clostridium difficile infection in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2012;35:789–95.
Balram B, Battat R, Al-Khoury A, D’Aoust J, Afif W, Bitton A, Lakatos PL, Bessissow T. Risk factors associated with Clostridium difficile infection in inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis. 2019;13:27–38.
Schneeweiss S, Korzenik J, Solomon DH, Canning C, Lee J, Bressler B. Infliximab and other immunomodulating drugs in patients with inflammatory bowel disease and the risk of serious bacterial infections. Aliment Pharmacol Ther. 2009;30:253–64.
Horton HA, Dezfoli S, Berel D, Hirsch J, Ippoliti A, McGovern D, Kaur M, Shih D, Dubinsky M, Targan SR, Fleshner P, Vasiliauskas EA, Grein J, Murthy R, Melmed GY. Antibiotics for treatment of Clostridium difficile infection in hospitalized patients with inflammatory bowel disease. Antimicrob Agents Chemother. 2014;58:5054–9.
Ananthakrishnan AN, McGinley EL, Binion DG. Excess hospitalisation burden associated with Clostridium difficile in patients with inflammatory bowel disease. Gut. 2008;57:205–10.
Nichols RL, Condon RE, DiSanto AR. Preoperative bowel preparation. Erythromycin base serum and fecal levels following oral administration. Arch Surg. 1977;112:1493–6.
Al-Mazrou AM, Hyde LZ, Suradkar K, Kiran RP. Effect of inclusion of oral antibiotics with mechanical bowel preparation on the risk of Clostridium Difficile infection after colectomy. J Gastrointest Surg. 2018;22:1968–75.
Kiran RP, Murray AC, Chiuzan C, Estrada D, Forde K. Combined preoperative mechanical bowel preparation with oral antibiotics significantly reduces surgical site infection, anastomotic leak, and ileus after colorectal surgery. Ann Surg. 2015;262:416–25; discussion 23–5
Murray AC, Kiran RP. Bowel preparation: are antibiotics necessary for colorectal surgery? Adv Surg. 2016;50:49–66.
Murray AC, Kiran RP. Benefit of mechanical bowel preparation prior to elective colorectal surgery: current insights. Langenbeck’s Arch Surg. 2016;401:573–80.
Midura EF, Jung AD, Hanseman DJ, Dhar V, Shah SA, Rafferty JF, Davis BR, Paquette IM. Combination oral and mechanical bowel preparations decreases complications in both right and left colectomy. Surgery. 2018;163:528–34.
Atkinson SJ, Swenson BR, Hanseman DJ, Midura EF, Davis BR, Rafferty JF, Abbott DE, Shah SA, Paquette IM. In the absence of a mechanical bowel prep, does the addition of pre-operative oral antibiotics to parental antibiotics decrease the incidence of surgical site infection after elective segmental colectomy? Surg Infect. 2015;16:728–32.
Gaines S, Shao C, Hyman N, Alverdy JC. Gut microbiome influences on anastomotic leak and recurrence rates following colorectal cancer surgery. Br J Surg. 2018;105:e131–e41.
Alverdy JC, Hyoju SK, Weigerinck M, Gilbert JA. The gut microbiome and the mechanism of surgical infection. Br J Surg. 2017;104:e14–23.
Morris AM, Jobe BA, Stoney M, Sheppard BC, Deveney CW, Deveney KE. Clostridium difficile colitis: an increasingly aggressive iatrogenic disease? Arch Surg. 2002;137:1096–100.
Stewart DB, Hegarty JP. Correlation between virulence gene expression and proton pump inhibitors and ambient pH in Clostridium difficile: results of an in vitro study. J Med Microbiol. 2013;62:1517–23.
Janarthanan S, Ditah I, Adler DG, Ehrinpreis MN. Clostridium difficile-associated diarrhea and proton pump inhibitor therapy: a meta-analysis. Am J Gastroenterol. 2012;107:1001–10.
Tleyjeh IM, Bin Abdulhak AA, Riaz M, Alasmari FA, Garbati MA, AlGhamdi M, Khan AR, Al Tannir M, Erwin PJ, Ibrahim T, Allehibi A, Baddour LM, Sutton AJ. Association between proton pump inhibitor therapy and clostridium difficile infection: a contemporary systematic review and meta-analysis. PLoS One. 2012;7:e50836.
Kwok CS, Arthur AK, Anibueze CI, Singh S, Cavallazzi R, Loke YK. Risk of Clostridium difficile infection with acid suppressing drugs and antibiotics: meta-analysis. Am J Gastroenterol. 2012;107:1011–9.
Garey KW, Sethi S, Yadav Y, DuPont HL. Meta-analysis to assess risk factors for recurrent Clostridium difficile infection. J Hosp Infect. 2008;70:298–304.
Chitnis AS, Holzbauer SM, Belflower RM, Winston LG, Bamberg WM, Lyons C, Farley MM, Dumyati GK, Wilson LE, Beldavs ZG, Dunn JR, Gould LH, MacCannell DR, Gerding DN, McDonald LC, Lessa FC. Epidemiology of community-associated Clostridium difficile infection, 2009 through 2011. JAMA Intern Med. 2013;173:1359–67.
Heidelbaugh JJ, Goldberg KL, Inadomi JM. Overutilization of proton pump inhibitors: a review of cost-effectiveness and risk [corrected]. Am J Gastroenterol. 2009;104(Suppl 2):S27–32.
The Lancet Infectious D. C difficile-a rose by any other name. Lancet Infect Dis. 2019;19:449.
Lawson PA, Rainey FA. Proposal to restrict the genus Clostridium Prazmowski to Clostridium butyricum and related species. Int J Syst Evol Microbiol. 2016;66:1009–16.
Dohrmann AB, Walz M, Lowen A, Tebbe CC. Clostridium cluster I and their pathogenic members in a full-scale operating biogas plant. Appl Microbiol Biotechnol. 2015;99:3585–98.
Monot M, Eckert C, Lemire A, Hamiot A, Dubois T, Tessier C, Dumoulard B, Hamel B, Petit A, Lalande V, Ma L, Bouchier C, Barbut F, Dupuy B. Clostridium difficile: new insights into the evolution of the pathogenicity locus. Sci Rep. 2015;5:15023.
Aktories K, Schwan C, Jank T. Clostridium difficile toxin biology. Annu Rev Microbiol. 2017;71:281–307.
El Meouche I, Peltier J, Monot M, Soutourina O, Pestel-Caron M, Dupuy B, Pons JL. Characterization of the SigD regulon of C. difficile and its positive control of toxin production through the regulation of tcdR. PLoS One. 2013;8:e83748.
Popoff MR, Bouvet P. Genetic characteristics of toxigenic clostridia and toxin gene evolution. Toxicon. 2013;75:63–89.
Fortier LC. The contribution of bacteriophages to the biology and virulence of pathogenic clostridia. Adv Appl Microbiol. 2017;101:169–200.
Chandrasekaran R, Lacy DB. The role of toxins in Clostridium difficile infection. FEMS Microbiol Rev. 2017;41:723–50.
Jank T, Giesemann T, Aktories K. Rho-glucosylating Clostridium difficile toxins A and B: new insights into structure and function. Glycobiology. 2007;17:15R–22R.
Holbourn KP, Shone CC, Acharya KR. A family of killer toxins. Exploring the mechanism of ADP-ribosylating toxins. FEBS J. 2006;273:4579–93.
Schwan C, Stecher B, Tzivelekidis T, van Ham M, Rohde M, Hardt WD, Wehland J, Aktories K. Clostridium difficile toxin CDT induces formation of microtubule-based protrusions and increases adherence of bacteria. PLoS Pathog. 2009;5:e1000626.
Carter GP, Lyras D, Allen DL, Mackin KE, Howarth PM, O’Connor JR, Rood JI. Binary toxin production in Clostridium difficile is regulated by CdtR, a LytTR family response regulator. J Bacteriol. 2007;189:7290–301.
Gurtler V, Grando D. New opportunities for improved ribotyping of C. difficile clinical isolates by exploring their genomes. J Microbiol Methods. 2013;93:257–72.
Janezic S. Direct PCR-Ribotyping of Clostridium difficile. Methods Mol Biol. 2016;1476:15–21.
Peretz A, Tkhawkho L, Pastukh N, Brodsky D, Halevi CN, Nitzan O. Correlation between fecal calprotectin levels, disease severity and the hypervirulent ribotype 027 strain in patients with Clostridium difficile infection. BMC Infect Dis. 2016;16:309.
Sirard S, Valiquette L, Fortier LC. Lack of association between clinical outcome of Clostridium difficile infections, strain type, and virulence-associated phenotypes. J Clin Microbiol. 2011;49:4040–6.
Archbald-Pannone LR, Boone JH, Carman RJ, Lyerly DM, Guerrant RL. Clostridium difficile ribotype 027 is most prevalent among inpatients admitted from long-term care facilities. J Hosp Infect. 2014;88:218–21.
Yakob L, Riley TV, Paterson DL, Marquess J, Magalhaes RJ, Furuya-Kanamori L, Clements AC. Mechanisms of hypervirulent Clostridium difficile ribotype 027 displacement of endemic strains: an epidemiological model. Sci Rep. 2015;5:12666.
Gerding DN, Johnson S, Rupnik M, Aktories K. Clostridium difficile binary toxin CDT: mechanism, epidemiology, and potential clinical importance. Gut Microbes. 2014;5:15–27.
Stabler RA, He M, Dawson L, Martin M, Valiente E, Corton C, Lawley TD, Sebaihia M, Quail MA, Rose G, Gerding DN, Gibert M, Popoff MR, Parkhill J, Dougan G, Wren BW. Comparative genome and phenotypic analysis of Clostridium difficile 027 strains provides insight into the evolution of a hypervirulent bacterium. Genome Biol. 2009;10:R102.
Wu YC, Lee JJ, Tsai BY, Liu YF, Chen CM, Tien N, Tsai PJ, Chen TH. Potentially hypervirulent Clostridium difficile PCR ribotype 078 lineage isolates in pigs and possible implications for humans in Taiwan. Int J Med Microbiol. 2016;306:115–22.
Stewart DB, Sr., Wright JR, Fowler M, McLimans CJ, Tokarev V, Amaniera I, Baker O, Wong HT, Brabec J, Drucker R, Lamendella R. Integrated meta-omics reveals a fungusassociated bacteriome and distinct functional pathways in clostridioides difficile infection. mSphere. 2019 Aug 28;4(4):e00454–19.
Nissle K, Kopf D, Rosler A. Asymptomatic and yet C. difficile-toxin positive? Prevalence and risk factors of carriers of toxigenic Clostridium difficile among geriatric in-patients. BMC Geriatr. 2016;16:185.
Crobach MJT, Vernon JJ, Loo VG, Kong LY, Pechine S, Wilcox MH, Kuijper EJ. Understanding Clostridium difficile Colonization. Clin Microbiol Rev. 2018 Mar 14;31(2):e00021–17.
Burnham CA, Carroll KC. Diagnosis of Clostridium difficile infection: an ongoing conundrum for clinicians and for clinical laboratories. Clin Microbiol Rev. 2013;26:604–30.
McDonald LC, Gerding DN, Johnson S, Bakken JS, Carroll KC, Coffin SE, Dubberke ER, Garey KW, Gould CV, Kelly C, Loo V, Shaklee Sammons J, Sandora TJ, Wilcox MH. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66:987–94.
Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302–7.
Louie TJ, Miller MA, Mullane KM, Weiss K, Lentnek A, Golan Y, Gorbach S, Sears P, Shue YK, Group OPTCS. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364:422–31.
Guery B, Menichetti F, Anttila VJ, Adomakoh N, Aguado JM, Bisnauthsing K, Georgopali A, Goldenberg SD, Karas A, Kazeem G, Longshaw C, Palacios-Fabrega JA, Cornely OA, Vehreschild M, Group ECS. Extended-pulsed fidaxomicin versus vancomycin for Clostridium difficile infection in patients 60 years and older (EXTEND): a randomised, controlled, open-label, phase 3b/4 trial. Lancet Infect Dis. 2018;18:296–307.
Nelson RL, Suda KJ, Evans CT. Antibiotic treatment for Clostridium difficile-associated diarrhoea in adults. Cochrane Database Syst Rev. 2017;3:CD004610.
Malamood M, Nellis E, Ehrlich AC, Friedenberg FK. Vancomycin enemas as adjunctive therapy for Clostridium difficile infection. J Clin Med Res. 2015;7:422–7.
Neal MD, Alverdy JC, Hall DE, Simmons RL, Zuckerbraun BS. Diverting loop ileostomy and colonic lavage: an alternative to total abdominal colectomy for the treatment of severe, complicated Clostridium difficile associated disease. Ann Surg. 2011;254:423–7; discussion 7–9
Dallal RM, Harbrecht BG, Boujoukas AJ, Sirio CA, Farkas LM, Lee KK, Simmons RL. Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complications. Ann Surg. 2002;235:363–72.
Sailhamer EA, Carson K, Chang Y, Zacharias N, Spaniolas K, Tabbara M, Alam HB, DeMoya MA, Velmahos GC. Fulminant Clostridium difficile colitis: patterns of care and predictors of mortality. Arch Surg. 2009;144:433–9; discussion 9–40
Bhangu A, Nepogodiev D, Gupta A, Torrance A, Singh P, Midl W, Research C. Systematic review and meta-analysis of outcomes following emergency surgery for Clostridium difficile colitis. Br J Surg. 2012;99:1501–13.
Stewart DB, Hollenbeak CS, Wilson MZ. Is colectomy for fulminant Clostridium difficile colitis life saving? A systematic review. Colorectal Dis. 2013;15:798–804.
Ferrada P, Callcut R, Zielinski MD, Bruns B, Yeh DD, Zakrison TL, Meizoso JP, Sarani B, Catalano RD, Kim P, Plant V, Pasley A, Dultz LA, Choudhry AJ, Haut ER, Committee EM-IT. Loop ileostomy versus total colectomy as surgical treatment for Clostridium difficile-associated disease: an Eastern Association for the Surgery of Trauma multicenter trial. J Trauma Acute Care Surg. 83:36–40.
Juo YY, Sanaiha Y, Jabaji Z, Benharash P. Trends in diverting loop ileostomy vs total abdominal colectomy as surgical management for Clostridium Difficile colitis. JAMA Surg. 2019.
Appaneal HJ, Caffrey AR, Beganovic M, Avramovic S, LaPlante KL. Predictors of Clostridioides difficile recurrence across a national cohort of veterans in outpatient, acute, and longtermcare settings. Am J Health Syst Pharm. 2019 Apr 17;76(9):581–90.
Stewart DB. Loop ileostomy for Clostridium difficile infection: know thy enemy. J Trauma Acute Care Surg. 83:1214–5.
van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG, Speelman P, Dijkgraaf MG, Keller JJ. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407–15.
Zainah H, Hassan M, Shiekh-Sroujieh L, Hassan S, Alangaden G, Ramesh M. Intestinal microbiota transplantation, a simple and effective treatment for severe and refractory Clostridium difficile infection. Dig Dis Sci. 2015;60:181–5.
Weingarden AR, Hamilton MJ, Sadowsky MJ, Khoruts A. Resolution of severe Clostridium difficile infection following sequential fecal microbiota transplantation. J Clin Gastroenterol. 2013;47:735–7.
Fischer M, Sipe BW, Rogers NA, Cook GK, Robb BW, Vuppalanchi R, Rex DK. Faecal microbiota transplantation plus selected use of vancomycin for severe-complicated Clostridium difficile infection: description of a protocol with high success rate. Aliment Pharmacol Ther. 2015;42:470–6.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Paquette, I.M., Stewart, D.B. (2022). Clostridium difficile Infection. In: Steele, S.R., Hull, T.L., Hyman, N., Maykel, J.A., Read, T.E., Whitlow, C.B. (eds) The ASCRS Textbook of Colon and Rectal Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-66049-9_52
Download citation
DOI: https://doi.org/10.1007/978-3-030-66049-9_52
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-66048-2
Online ISBN: 978-3-030-66049-9
eBook Packages: MedicineMedicine (R0)