Abstract
Advanced echocardiographic techniques, such as 3D echocardiography, tissue Doppler imaging (TDI), and speckle-tracking strain imaging, have recently been introduced for the echocardiographic evaluation of patients with dilated cardiomyopathy (DCM) and have demonstrated significant incremental value over basic echocardiography. For instance, 3D echocardiography has shown improved accuracy for left ventricular (LV) volumes and ejection fraction assessment, which might play a substantial role in better selecting candidates for device therapy. Moreover, TDI of the mitral annulus is one of the principal methods for assessing diastolic function, provides noninvasive information on left atrial pressure, is related to exercise capacity in DCM patients, and has demonstrated prognostic value. LV dyssynchrony with speckle-tracking strain analysis was an independent determinant of response to cardiac resynchronization therapy and long-term survival in this patient population.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cardiac Magnetic Resonance
- Cardiac Resynchronization Therapy
- Pulmonary Capillary Wedge Pressure
- Tissue Doppler Imaging
- Functional Mitral Regurgitation
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Advanced echocardiographic techniques, such as 3D echocardiography, tissue Doppler imaging (TDI), and speckle-tracking strain imaging, have recently been introduced for the echocardiographic evaluation of patients with dilated cardiomyopathy (DCM) and have demonstrated significant incremental value over basic echocardiography.
2 Left Ventricular Dimensions and Systolic Function
Accurate left ventricular (LV) volumes and ejection fraction (EF) quantitation is crucial in the echocardiographic evaluation of patients with DCM. For instance, improved accuracy of LV volumes and EF assessment might play a substantial role in helping physicians select candidates for implantable cardioverter defibrillator or cardiac resynchronization therapy (CRT). However, it is well known that M-mode and 2D evaluations of LV volumes and EF have limitations [1]. Conversely, 3D echocardiography has been recently validated with cardiac magnetic resonance (CMR) for quantification of ventricular volumes in various cardiomyopathies, including DCM, and demonstrated a higher accuracy and lower inter- and intraobserver variability compared with 2D echocardiography [2, 3]. Some advantages of 3D echocardiography include independence from geometric assumptions, semiautomatic delineation of the endocardial border, and absence of errors deriving from “foreshortening” of the LV apex (Fig. 6.1, Clip 6.1) [4]. Three-dimensional echocardiography also provides the possibility for quantitative assessment of LV regional wall motion by measuring the volume change of each segment in the cardiac cycle. This technique demonstrated high feasibility in DCM patients [5] and provided good correlation with CMR [6]. Finally, 3D echocardiography has also been applied in DCM patients and validated against CMR for LV mass assessment, showing excellent correlation with CMR, significant superiority over 2D echocardiography, and low inter- and intraobserver variability [7, 8].
Left ventricular (LV) volumes and ejection fraction (EF) quantification with 3D echocardiography in a patient with dilated cardiomyopathy (DCM) with severe LV dilatation and dysfunction. The endocardial border is manually traced in the apical four-chamber (upper left panel ), two-chamber (upper right panel ), and short-axis (mid left panel ) views, and a 3D LV model is automatically generated (mid right panel ); subsequently, 3D LV volumes and EF are measured, and a time–volume change curve of all segments during the cardiac cycle is provided (lower panel ). EDV end-diastolic volume, ESV end-systolic volume, SV stroke volume
Technological advances in the field of cardiac ultrasound have lead to further new noninvasive techniques, such as TDI and speckle-tracking strain imaging, for assessing cardiac mechanics and segmental and global LV function. The peak systolic myocardial velocity S’, a simple TDI index of systolic longitudinal function, is a marker of impaired subendocardial fiber contraction and correlates with myocardial fibrosis [9]. In addition, strain evaluation (by TDI, 2D and 3D speckle-tracking echocardiography) allows discrimination between active and passive movement of all myocardial segments and permits separate assessments of distinct components of myocardial deformation (i.e., longitudinal versus circumferential shortening and radial thickening) [10]. Also, all myocardial deformation parameters, including longitudinal, circumferential, and radial strain and torsion, are reduced in DCM patients (Fig. 6.2, Clips 6.2a, 6.2b, and 6.2c) [11].
Left ventricular (LV) longitudinal strain evaluation by 2D speckle-tracking echocardiography in apical four- (a), two- (b), and long-axis (c) views in a dilated cardiomyopathy (DCM) patient. In each view, the LV region of interest in the 2D speckle-tracking analysis is shown (upper half of each panel), together with longitudinal strain curves for each segment (lower half of each panel). Final results of the strain analysis for each LV segment are displayed in a bulls-eye plot (d). Global longitudinal strain is automatically calculated by the software and is importantly reduced (−12 %)
3 Left Ventricular Diastolic Function
TDI of the mitral annulus is a relatively novel method for assessing diastolic function. It is performed in apical four-chamber view, placing the pulsed-wave tissue Doppler on the septal or lateral annulus of the mitral valve (MV), showing a Doppler pattern with E’ and A’ waves. The ratio of transmitral E velocity to mitral annular E’ velocity (E/E’) is a marker of left atrial (LA) pressure, is related to exercise capacity in DCM, and provides prognostic value [12]. On the other hand, E/E’ ratio has a wide grey zone, and its accuracy is questionable, particularly in patients with advanced DCM and severe heart failure (HF). In fact, a study with invasive hemodynamic correlations showed that E/E’ ratio had a weak correlation with LV filling pressures in DCM patients, particularly in those with severe LV dilation and after CRT [13].
Other new indices for diastolic function evaluation obtained by speckle-tracking analysis are promising. Circumferential strain and strain rate during late diastolic LV filling, E/circumferential strain rate at early diastolic LV filling, and E/circumferential strain at the time of peak E wave showed greater area under the curve than the E/E’ ratio for predicting pulmonary capillary wedge pressure (PCWP) >12 mmHg [14]. Also, LA strain assessment with speckle-tracking technique demonstrated a better correlation than other Doppler indices, such as E/E’ ratio, with LV filling pressure as measured by right catheterization, in patients with advanced systolic HF [15]. Specifically, peak atrial longitudinal strain is a parameter for functional assessment of the atrial reservoir phase (which is essential for LV filling), and results progressively reduced with the increase of LV filling pressure. Therefore, peak atrial longitudinal strain demonstrated a strong inverse correlation with PCWP and excellent diagnostic accuracy in predicting elevated filling pressure [15].
4 Left Ventricular Dyssynchrony
There is evidence that LV mechanical dyssynchrony is an independent determinant of response to CRT and long-term survival [16]. Advanced indices of intraventricular mechanical dyssynchrony are based on TDI, speckle-tracking imaging, and 3D echocardiography [17].
The rocking motion of the apex (apical rocking) can be quantified by TDI by measuring the transverse motion of the LV apex perpendicular to the LV long axis. This index is clinically feasible, reproducible, and has predictive value for response and long-term survival following CRT [18]. Furthermore, the time from QRS onset to peak systolic velocity can be measured by TDI imaging at the level of different basal and mid-LV segments: the time difference between peak contraction of opposite segments is an index for LV dyssynchrony. Contraction delay between the basal septum and the basal lateral wall in the apical four-chamber view is usually considered, but this index can be measured from multiple apical views in up to 12 segments. Moreover, TDI-derived longitudinal strain and strain-rate imaging allows LV dyssynchrony evaluation by measuring the time delay between peak systolic strain rate [19]. The Predictors of Response to CT (PROSPECT) trial demonstrated a low sensitivity and specificity of echocardiographic TDI markers of LV dyssynchrony for predicting response to CRT [20]. The TDI markers indeed show low feasibility and reproducibility. In particular, in advanced DCM patients with extended areas of akinesis, identifying systolic contraction timing and measurements of TDI parameters are particularly problematic and can be misleading, as it is impossible to discriminate passive from active myocardial movements.
New echocardiographic techniques not included in the PROSPECT trial involve speckle-tracking strain evaluation and 3D echocardiography, even if the temporal resolution of these techniques is relatively low and inferior compared with TDI [21, 22].
LV dyssynchrony with speckle-tracking strain analysis (Fig. 6.3) is calculated as the time difference in peak strain values (radial and circumferential in the short-axis plane, longitudinal and transverse in the long-axis plane) between opposing segments (most frequently between the anteroseptal and posterolateral wall) [21]. Longitudinal and radial strain parameters appear to be more reproducible and accurate for quantifying LV mechanical dyssynchrony [21, 23]. In the prospective multicenter Speckle Tracking and Resynchronization (STAR) study [24], radial and transverse LV strains were both significantly associated with EF response and long-term outcome (death, heart transplant, LV assist device) after CRT. Lack of baseline radial or transverse LV dyssynchrony (defined as ≥130 ms opposing wall delay) appeared to be a marker of adverse prognosis following CRT [24].
Evaluation of left ventricular (LV) dyssynchrony with 2D speckle-tracking strain analysis calculated as the time difference in peak circumferential strain between opposing segments (mid-inferoseptal and mid-anterolateral wall) in the short-axis plane. In this case of advanced dilated cardiomyopathy (DCM), there was significant LV dyssynchrony (130 ms). MA mid anterior, MAL mid anterolateral, MAS mid anteroseptal, MI mid inferior, MIL mid inferolateral, MIS mid inferoseptal
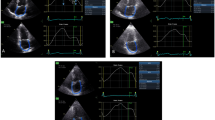
Furthermore, 3D echocardiography has emerged as a novel technique for dyssynchrony quantification based on analysis of volume variations of each segment in the same cardiac cycle: if the contraction is dyssynchronous, there is dispersion in the time each segment takes to reach its minimum volume. The 3D systolic dyssynchrony index is defined as the standard deviation of the time to minimum systolic regional volume using a 16-segment model (Fig. 6.4). A cutoff value of systolic dyssynchrony index ≥5.6 % has been proposed to predict response to CRT [25]. More recently, 3D speckle-tracking imaging has emerged for calculating LV dyssynchrony by assessing myocardial deformation within the LV 3D full volume [26]. With this technique, the maximal opposing wall delay and the standard deviation of time to peak (radial) strain of 16 LV segments are derived as LV dyssynchrony indices. HF patients show higher 3D dyssynchrony indices compared with normal individuals; moreover, in patients who underwent CRT, 3D imaging demonstrated effective LV resynchronization with considerably improved systolic function [26].
Larger studies are needed to determine the best LV mechanical dyssynchrony echocardiographic parameter for predicting CRT response and long-term survival. Moreover, besides LV mechanical dyssynchrony, it seems important to use advanced imaging to evaluate other aspects to maximize response to CRT, including detecting necrotic tissue, presence of viability, and availability of coronary veins for optimal LV lead position [27]. Echocardiography might play a role in identifying the optimal site for LV pacing, first by detecting the myocardial area of latest mechanical activation (in particular, by 2D and 3D speckle-tracking imaging) [28]; and second, by excluding the presence of scar tissue in the area considered for LV pacing (for instance, by speckle-tracking imaging or dobutamine stress echocardiography). Scarred LV tissue assessed using speckle-tracking global longitudinal strain was a strong determinant of response to CRT in patients with ischemic DCM and was significantly related with total scar burden quantified by CMR [29].
5 Functional Mitral Regurgitation
It is well known that the 2D proximal isovelocity surface area (PISA) method for evaluating mitral regurgitation (MR) has several limitations, as it is based on geometric assumptions. There are advantages of 3D over 2D echocardiography in evaluating MR grade due to the direct planimetry of the effective regurgitant orifice area, which is typically elliptical in functional MR (Fig. 6.5, Clip 6.3). Previous studies demonstrated the additional value of 3D echocardiography in patients with functional MR, whereas 2D imaging significantly underestimated the size of the regurgitant orifice [30]. Furthermore, dedicated software permits 3D quantification of MV annulus dimensions, MV leaflet surface, tenting volume, aortomitral angle, and papillary muscle geometry [31].
Evaluation of mitral regurgitation (MR) grade with 3D transthoracic echocardiography from a 3D full volume with color Doppler acquisition of the regurgitant jet (mid right panel): the software permits measurement of the vena contracta in two perpendicular planes (as shown in the upper panels); furthermore, it is possible to trace the direct planimetry of the effective regurgitant orifice area, which is typically elliptical in functional MR (mid left and lower panels). In this case of severe functional MR, the effective regurgitant orifice area was 0.5 cmq
6 Other Features
Echocardiography with integrated backscatter imaging (i.e., a technique that allows assessment of myocardial ultrasound reflectivity) permits identification of myocardial fibrosis, which appears as high-echogenic myocardial areas. Integrated backscattering is an index of myocardial echogenicity that demonstrated a significant correlation with the quantity of fibrosis at biopsy [32]. Other authors [33] demonstrated a reduction of integrated backscattering in patients with DCM and worse prognosis, possibly due to reduced contractility and/or myocardial fibrosis.
References
Otterstad JE, Froeland G, St John Sutton M et al (1997) Accuracy and reproducibility of biplane two-dimensional echocardiographic measurements of left ventricular dimensions and function. Eur Heart J 18:507–513
Gutierrez-Chico JL, Zamorano JL, Perez de Isla L et al (2005) Comparison of left ventricular volumes and ejection fractions measured by three-dimensional echocardiography versus by two-dimensional echocardiography and cardiac magnetic resonance in patients with various cardiomyopathies. Am J Cardiol 95:809–813
Shiota T, McCarthy PM, White RD et al (1999) Initial clinical experience of real-time three-dimensional echocardiography in patients with ischemic and idiopathic dilated cardiomyopathy. Am J Cardiol 84:1068–1073
Lang RM, Badano LP, Tsang W et al (2012) EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. Eur Heart J Cardiovasc Imaging 13:1–46
Mu Y, Chen L, Tang Q et al (2010) Real time three-dimensional echocardiographic assessment of left ventricular regional systolic function and dyssynchrony in patients with dilated cardiomyopathy. Echocardiography 27:415–420
Corsi C, Lang RM, Veronesi F et al (2005) Volumetric quantification of global and regional left ventricular function from real-time three-dimensional echocardiographic images. Circulation 112:1161–1170
Gopal AS, Schnellbaecher MJ, Shen Z et al (1997) Freehand three-dimensional echocardiography for determination of left ventricular volume and mass in patients with abnormal ventricles: comparison with magnetic resonance imaging. J Am Soc Echocardiogr 10:853–861
Mor-Avi V, Sugeng L, Weinert L et al (2004) Fast measurement of left ventricular mass with real-time three-dimensional echocardiography: comparison with magnetic resonance imaging. Circulation 110:1814–1818
Shan K, Bick RJ, Poindexter BJ et al (2000) Relation of tissue Doppler derived myocardial velocities to myocardial structure and beta-adrenergic receptor density in humans. J Am Coll Cardiol 36:891–896
Mor-Avi V, Lang RM, Badano LP et al (2011) Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. Eur J Echocardiogr 12:167–205
Meluzin J, Spinarova L, Hude P et al (2009) Left ventricular mechanics in idiopathic dilated cardiomyopathy: systolic-diastolic coupling and torsion. J Am Soc Echocardiogr 22:486–493
Oki T, Tabata T, Yamada H et al (1997) Clinical application of pulsed Doppler tissue imaging for assessing abnormal left ventricular relaxation. Am J Cardiol 79:921–928
Mullens W, Borowski AG, Curtin RJ et al (2009) Tissue Doppler imaging in the estimation of intracardiac filling pressure in decompensated patients with advanced systolic heart failure. Circulation 119:62–70
Meluzin J, Spinarova L, Hude P et al (2011) Estimation of left ventricular filling pressures by speckle tracking echocardiography in patients with idiopathic dilated cardiomyopathy. Eur J Echocardiogr 12:11–18
Cameli M, Lisi M, Mondillo S et al (2010) Left atrial longitudinal strain by speckle tracking echocardiography correlates well with left ventricular filling pressures in patients with heart failure. Cardiovasc Ultrasound 8:14
Delgado V, Bax JJ (2011) Assessment of systolic dyssynchrony for cardiac resynchronization therapy is clinically useful. Circulation 123:640–655
Gorcsan J 3rd, Abraham T, Agler DA et al (2008) Echocardiography for cardiac resynchronization therapy: recommendations for performance and reporting–a report from the American Society of Echocardiography Dyssynchrony Writing Group endorsed by the Heart Rhythm Society. J Am Soc Echocardiogr 21:191–213
Szulik M, Tillekaerts M, Vangeel V et al (2010) Assessment of apical rocking: a new, integrative approach for selection of candidates for cardiac resynchronization therapy. Eur J Echocardiogr 11:863–869
Bax JJ, Bleeker GB, Marwick TH et al (2004) Left ventricular dyssynchrony predicts response and prognosis after cardiac resynchronization therapy. J Am Coll Cardiol 44:1834–1840
Chung ES, Leon AR, Tavazzi L et al (2008) Results of the Predictors of Response to CRT (PROSPECT) trial. Circulation 117:2608–2616
Delgado V, Ypenburg C, van Bommel RJ et al (2008) Assessment of left ventricular dyssynchrony by speckle tracking strain imaging comparison between longitudinal, circumferential, and radial strain in cardiac resynchronization therapy. J Am Coll Cardiol 51:1944–1952
Mele D, Agricola E, Galderisi M et al (2009) Real-time three-dimensional echocardiography: current applications, advantages and limits for the evaluation of the left ventricle. G Ital Cardiol (Rome) 10:516–532
Faletra FF, Conca C, Klersy C et al (2009) Comparison of eight echocardiographic methods for determining the prevalence of mechanical dyssynchrony and site of latest mechanical contraction in patients scheduled for cardiac resynchronization therapy. Am J Cardiol 103:1746–1752
Tanaka H, Nesser HJ, Buck T et al (2010) Dyssynchrony by speckle-tracking echocardiography and response to cardiac resynchronization therapy: results of the Speckle Tracking and Resynchronization (STAR) study. Eur Heart J 31:1690–1700
Marsan NA, Bleeker GB, Ypenburg C et al (2008) Real-time three-dimensional echocardiography permits quantification of left ventricular mechanical dyssynchrony and predicts acute response to cardiac resynchronization therapy. J Cardiovasc Electrophysiol 19:392–399
Tanaka H, Hara H, Saba S et al (2010) Usefulness of three-dimensional speckle tracking strain to quantify dyssynchrony and the site of latest mechanical activation. Am J Cardiol 105:235–242
Ypenburg C, van Bommel RJ, Delgado V et al (2008) Optimal left ventricular lead position predicts reverse remodeling and survival after cardiac resynchronization therapy. J Am Coll Cardiol 52:1402–1409
Van de Veire NR, Yu CM, Ajmone-Marsan N et al (2008) Triplane tissue Doppler imaging: a novel three-dimensional imaging modality that predicts reverse left ventricular remodelling after cardiac resynchronisation therapy. Heart 94:e9
D’Andrea A, Caso P, Scarafile R et al (2009) Effects of global longitudinal strain and total scar burden on response to cardiac resynchronization therapy in patients with ischaemic dilated cardiomyopathy. Eur J Heart Fail 11:58–67
Iwakura K, Ito H, Kawano S et al (2006) Comparison of orifice area by transthoracic three-dimensional Doppler echocardiography versus proximal isovelocity surface area (PISA) method for assessment of mitral regurgitation. Am J Cardiol 97:1630–1637
Sugeng L, Spencer KT, Mor-Avi V et al (2003) Dynamic three-dimensional color flow Doppler: an improved technique for the assessment of mitral regurgitation. Echocardiography 20:265–273
Fujimoto S, Mizuno R, Nakagawa Y et al (1999) Ultrasonic tissue characterization in patients with dilated cardiomyopathy: comparison with findings from right ventricular endomyocardial biopsy. Int J Card Imaging 15:391–396
Dagdeviren B, Akdemir O, Eren M et al (2002) Prognostic implication of myocardial texture analysis in idiopathic dilated cardiomyopathy. Eur J Heart Fail 4:41–48
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Abate, E., Pinamonti, B. (2014). Advanced Echocardiographic Technologies in Dilated Cardiomyopathy. In: Pinamonti, B., Sinagra, G. (eds) Clinical Echocardiography and Other Imaging Techniques in Cardiomyopathies. Springer, Cham. https://doi.org/10.1007/978-3-319-06019-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-06019-4_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-06018-7
Online ISBN: 978-3-319-06019-4
eBook Packages: MedicineMedicine (R0)