Abstract
Introduction of GM crops into agricultural production systems increased public concern and renewed interest in research on the possible environmental consequences of growing GM crops including human health and ecosystem functioning. Globally, Bacillus thuringiensis (Bt) cotton occupies 15 million ha which comprised 43 % of the total cotton area of 35 million ha. Bt cotton was developed by incorporating the cry gene of the soil bacterium Bacillus thuringiensis. This gene expresses the protein endotoxin (Cry) that has insecticidal activity against the common cotton lepidopteran insect pests. While the benefits of Bt cotton are well known, there is a wide spread concern about growing transgenic cotton. This stems from the fact that the Bt toxin produced in leaves, stems and roots of Bt cotton is introduced in soil which might affect general soil health. Several workers have studied the effects of transgene products and transgenic cotton on the soil biological properties. Quite a few studies assessed the risk of growing Bt cotton on flora and fauna in diverse agro-ecosystems. This chapter attempts to review the work done so far related to growing transgenic Bt cotton on the soil microbial diversity and other related soil functions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Genetically Modify
- Genetically Modify Crop
- Microbial Biomass Carbon
- Transgenic Cotton
- Insecticidal Crystal Protein
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
8.1 Introduction
Genetically modified (GM) , genetically engineered, or transgenic crops refer to plants produced by the insertion of specific pieces of nucleic acids into the plant’s DNA using recombinant DNA technology (i.e., Agrobacterium-mediated transformation or direct gene transfer methods) (Griffiths et al. 2005) . This biotechnological approach allows genes to be introduced into a plant genome from any source (i.e., plant, animal, bacterial and fungal) resulting in potential transfer of a wide range of genetic resources between unrelated species, a major difference compared to traditional plant breeding that is limited to exchange of genetic material only between sexually compatible close relatives of a given plant (Mirkov 2003) . Transgenic plants that show herbicide tolerance, resistance to viral, bacterial and fungal diseases, insect resistance, improved product quality and superior agronomic properties were introduced in the mid-1990s. Plant species that have been genetically engineered include: maize, tomato, cotton, soybean, oilseed rape and to a lesser extent potato, squash, beet, rice, flax, papaya and cichorium.
Worldwide 12 major crops such as soybean, maize, cotton, canola, potato, sugar beet, alfalfa, papaya, squash, tomato, poplar and sweet pepper have been genetically modified, commercially cultivated (James 2010) . Soybean is the leading GM crop occupying 75.4 m ha, followed by maize (51.00 m ha), cotton (24.7 m ha) and canola (8.2 m ha). Initially, six countries namely USA, China, Canada, Mexico, Australia and Argentina took up cultivation of GM crops, this number increased to 29 in 2011. Growing awareness of GM crops and acceptance by farmers resulted in an increase in global area under GM crops from 1.7 m ha in 1996 to 160 m ha in 2011 (James 2010). Presently, two principal transgenic technologies dominate the market: (i) herbicide-tolerant (HT) crops and (ii) insect resistant crops (Bt crops). Bt crops increased productivity and reduced insecticide usage, providing additional benefits to human health and the environment (Table 8.1).
There has been a strong debate on the safety of genetically modified plants ever since the introduction of transgenic plant products into the market. This debate is still very much alive and several issues were raised, including the safety of transgenic food and the environmental impact of transgenic plants (Schubert 2002; Dale et al. 2002; Liu et al. 2005) . However, the potential development of resistance to the Bt toxin by insect pests and the indirect damage of Bt toxins to non-target species are major concerns related to their use. Introduction of GM crops took place in 1996, when biotechnology-derived herbicide tolerant (HT) and insect-resistant traits were launched into the market in soybean, cotton, corn, and canola. These ‘input traits’ were designed to benefit the farmer directly and aimed to increase productivity per hectare, reduce agrochemical use, decrease production costs, have greater flexibility and efficiencies in production regimes, and improve grower health (Hossain et al. 2004; Huang et al. 2005) .
8.2 Mechanisms of Transgenic Plants Affect Soil Microorganisms and Functions
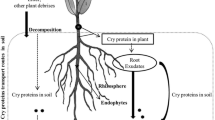
With the introduction of GM crops into agricultural production systems public concern increased. This also renewed interest in research on possible environmental consequences of growing GM crops including human health and ecosystem functioning (Sessitsch et al. 2004; Brookes and Barfoot 2005; Marvier et al. 2007) . Soil microorganisms are responsible for different key functions in ecosystems as they are involved in many decomposing processes as well as in all major biogeochemical cycles, in the recycling of essential elements. Studies of the impact of genetically modified organisms should therefore, also focus on microbial community functions as they are key elements in a healthy ecosystem (Lamarche and Hamelin 2007) . Cultivation of transgenic crops are reported to affect soil functions by direct (transgene proteins) and indirect effects (changes in plant protein, root exudates composition, modification in metabolic pathways). GM crops have the potential to influence soil microbiology through (i) the exudation of transgenic proteins from the root system, (ii) the release of transgenic proteins from broken and dying roots, (iii) the incorporation of above ground plant material into the soil, and (iv) differences in exudation chemistry (Gupta and Watson 2004; Knox et al. 2006; Saxena and Stotzky 2001) .
8.3 Indicators of Soil Biological Quality: Why to Measure?
Soil quality is defined as, “the capacity of a soil to function within its ecosystem boundaries to sustain biological productivity and diversity, maintain environmental quality, and promote plant and animal health” (Brady and Weil 1999) . Soil quality cannot be measured directly, so we evaluates through indicators. Indicators are measurable properties of soil or plants that provide clues about how well the soil can function. Indicators can be physical, chemical, and biological properties, processes, or characteristics of soils. Soil quality indicators are useful to policy makers to monitor the long-term effects of farm management practices on soil quality; assess the economic impact of alternative management practices designed to improve soil quality by including not only environmental values but also taking into account economic and social factors. Some of the key indicators of soil biological quality are presented in Table 8.2.
8.4 What is Bt?
“Bt” is short for Bacillus thuringiensis it is a soil bacterium occurring naturally. Bt was first discovered in 1901 by Shigetane Ishiwatari. In 1911, B. thuringiensis was re-discovered in Germany by Ernst Berliner. He isolated the cause of a disease called Schlaffsucht in flour moth caterpillars. A unique feature is its produce crystal proteins called as “CRY proteins” or “Insecticidal Crystal Protein” (ICP) that selectively kills specific groups of insects for example Lepidopteran caterpillars (moth and butterflies), Diptera (mosquitoes and black flies), Coleoptera (beetles) , and nematodes.
8.5 Mode of Action by ICP
The target organ for Bt toxins is the insect larvae’s mid-gut. The mid-gut of the larvae is a simple, tubular epithelium that dominates the internal architecture of the insects. After ingestion by insect’s larvae, the Bt δ-endotoxin disrupt of the epithelium in the insect mid-gut. The alkalinity of insect mid-gut (pH 12) dissolves the Crystals, releasing the Cry pro-toxin where it is cleaved by insect proteases to generate the trypsin resistant core of the active δ-endotoxin. The active toxin traverses the peritrophic membrane to bind receptor of brush border cells of the insect mid-gut. Integration of the toxin into the epithelial membrane, resulted in osmolysis of the cells, and paralysis occurred and dies within 2 days. Different Bt strains produce different CRY proteins , and there are hundreds of known strains which have identified more than 60 types of Cry-proteins that affect a wide variety of insects.
8.6 Persistence of cry1 Toxins from Bt Cotton
During plant litter decomposition, most transgene protein(s) appear to be rapidly degraded. However, some proteins can bind to surface-active particles and reduce their availability to microbes. Sims and Ream (1997) estimated that a potential maximum of 1.6 mg Cry2A protein kg−1 soil would result from the incorporation of Bt cotton residues into the top soil (Table 8.3). Recently, Sun et al. (2007) reported that the CryIAb protein persisted in soil for at least 56 days after incubating a slit loam soil with Bt cotton tissues.
8.7 Impact of Transgenic Bt Cotton Cultivation
Cotton (Gossypium spp.) belonging to the genus Gossypium in the family Malvaceae is an important fiber crop of global importance. Cotton is grown in tropical and subtropical regions of more than 80 countries. It is an important source of oil and high quality protein and plays a significant role in the national economy. Besides being the backbone of the textile industry, cotton and its byproducts are also part of the livestock feed, seed-oil, fertilizers, papers and other consumer products. Handling, processing and production of various consumer based products of cotton also play an important role in the social and industrial structure. Cotton is long duration crop and it is reported to be attacked by more than 162 species of insect pests including sucking pests, tissue borers and defoliators at various stages of growth, causing losses up to 60 %. Cotton is vulnerable to a number of insect species, especially to the larvae of lepidopteran pests . The cotton bollworm complex is a major and serious threat to the cotton, causing potential yield losses across the world and reported that the annual loss of at least US $ 300 million. High level of insecticide resistance in bollworms necessitates repeated application of insecticides, thereby aggravating the problem of resistance and also leading to heavy expenditure on cultivation and crop failures. Therefore it was important to initiate the development of alternative technologies such as genetic modification to enable plants to resist against the insect attack.
8.8 Transgenic Bt Cotton on Pest Control
The era of transgenic cotton began when Perlak et al. (1990) introduced cry 1A(b) and cry 1A(c) genes into cotton (G. hirsutum) plants and transformed plants showed a high level of resistance to Helicoverpa . During the field and laboratory tests, it was demonstrated that transgenic cotton is highly effective against neonate larvae of H. armigera (cotton bollworm), H. virescens (Tobacco budworm), and Pectinophora gossypiella (pink bollworm). The Bt gene from the original genetically engineered mother plant was Coker-312 . Transferred to advanced cotton cultivars through backcrossing. Later Gene stacking, involving the introduction of more than one gene of similar effects is becoming an attractive alternative for developing durable resistance and for simultaneous and effective control of more than one insect together. For instance, in cotton, Monsanto transgenic event ‘Bollgard-II’ carries two genes viz, cry 1Ac (against American Bollworm and cry 2Ab (Against tobacco bud worm) (Table 8.4).
Bt cotton occupies globally 15 million ha which comprised 43 % of the total cotton area of 35 million ha in nine countries namely USA, Mexico, China, Argentina, South Africa, Colombia, India, and Brazil (ISAAA 2006). GM cotton were developed by incorporating and expressing crystal protein endotoxin (Cry) encoded by the cry gene of the soil bacterium B. thuringiensis having insecticidal activity against the common cotton infecting insects belonging to orders Lepidoptera, Diptera , and Coleoptera (Wallimann 2000) . Although there is large-scale adoption of Bt cotton by the farmers because of immediate financial gain, there is concern that transgenic Bt crops release Bt toxins into the environment which affect associated and succeeding crops due to a reduction in soil chemical and biological activities (O’Callaghan et al. 2005; Sarkar et al. 2008) .
8.9 Transgenic Bt Cotton on Microbial Diversity and Soil Functions
In experiments to evaluate the persistence of Cry1 toxins from Bt cotton leaves incorporated into soil microcosms, Palm et al. (1996) found that degradation of the toxin was microbially mediated as suggested by various reports from the Stotzky group (Koskella and Stotzky 1997; Crecchio and Stotzky 1998) . Bacterial community structure was less affected by the cry1Ab protein than by other environmental factors, such as plant age or field heterogeneity (Baumgarte and Tebbe 2005) . However, undue decrease in microbial community richness with the use of genetically modified cotton is also reported (Dunfield and Germida 2004) . Previous studies have shown that the qualitative and quantitative differences in root exudation of Bt cotton could strongly influence the structure of microbial communities in the rhizosphere (Oger et al. 2000; Yan et al. 2007) . A significant but transient increase in the populations of culturable bacteria and fungi was observed in soil amended with leaves of Bt cotton (Gossypium hirsutum L.) expressing the Cry1Ac protein in comparison to the wild type plant measured by BIOLOG analysis and DNA fingerprinting (Donegan et al. 1995) . Higher microbial counts in transgenic cotton grown soil have also been reported by several workers (Shen et al. 2006) (Table 8.5). Head et al. (2002) demonstrated that the amount of Cry1Ac protein accumulated as a result of continuous use of transgenic Bt cotton, and subsequent incorporation of plant residues into the soil by postharvest tillage for 3 to 6 consecutive years is extremely low and does not result in detectable biological activity. Rui et al. (2005) reported that the fortification of pure Bt toxin into rhizospheric soil did not result in significant changes in the numbers of culturable functional bacteria, except the nitrogen-fixing bacteria when the concentration of Bt toxin was higher than 500 ng/g. The results indicated that Bt toxin was not the direct factor causing decrease of the numbers of bacteria in the rhizosphere, and other factors may be involved. Balachandar et al. (2008) studied the diversity richness of Pink-pigmented facultative methylotrophs (PPFMs) present in the phyllosphere, rhizoplane and internal tissues did not differ between Bt and non-Bt-cotton and reported that there was no evidence to indicate any adverse effects of Bt cotton on the diversity of plant-associated methylobacteria.
Hu et al. (2009) reported that there were no consistent differences in the numbers of different groups of functional bacteria between rhizosphere soil of Bt and non-Bt cotton in the same field. Further, no obvious trends were observed with regard to the numbers of the various groups of functional bacteria with an increasing duration of Bt cotton cultivation. Sarkar et al. (2009) concluded from their study that there were some positive or no negative effects of Bt-cotton on the soil quality indicators (microbial biomass carbon, microbial biomass nitrogen, microbial biomass phosphorus, total organic carbon, microbial quotient, potential nitrogen mineralization, nitrification, nitrate reductase, acid and alkaline phosphatase activities, Root dry weights, and root volume). Therefore cultivation of Bt cotton appears to pose no risk to soil ecosystem functions (Table 8.6). The microbial community structure in soil was not affected by the cropping of Bt cotton and the total microbial population and diversity of experimental fields remain quite similar during the cropping of both Bt cotton and non-Bt cotton (Kapur et al. 2010) . Based on the studies conducted at Central Institute for Cotton Research, Nagpur, it was found that growing Bt cotton does not affect the soil biological properties (soil respiration, urease activity, dehydrogenase activity, and microbial biomass carbon). The results obtained with culturable microbial population and microbial diversity index analysis further proved that the microbial activity in soil was not affected by the cropping of Bt cotton (Velmourougane and Sahu 2013) . Cry proteins from B.thuringiensis was also not reported to affect the soil invertebrates (Table 8.7). These results suggest that cultivation of Bt cotton expressing cry1Ac gene may not poses ecological or environmental risk.
8.10 Conclusion
A major problem in assessing the impacts of transgenic crops on soil microbial attributes is the lack of baseline information on diverse agro-ecosystems to compare with ecosystems in which transgenic crops were introduced and lack of universally approved approach for carrying out impact assessment of the transgenic plants on soil ecosystem. Genetic modifications have been performed with several plant species (maize, cotton, wheat, and rice), with targeted goals such as resistance to insect pests or herbicides, increased growth, and increased nutritional quality. Enormous studies have been conducted to assess the potential beneficial and/or detrimental effects of genetically engineered plants on soil microbes. Special attention was paid to study the impact of Bt plants, which express the cry toxin of B. thuringiensis, on microbial communities. Though few studies demonstrated a negative impact of Bt crops on soil microbes (Castaldini et al. 2005; Wu et al. 2004) , most of the studies showed no adverse effects (Baumgarte and Tebbe 2005, Blackwood and Buyer 2004; Brusetti et al. 2004; Devare et al. 2004, Griffiths et al. 2005; Liu et al. 2005, Muchaonyerwa et al. 2004; Saxena and Stotzky 2001) .
From the already published work, following points emanate:
-
1.
Bt cotton provided effective control against lepidopteran pests and reduced insecticide spray applications.
-
2.
Cry proteins released in root exudates and from Bt cotton residues appear to have no consistent, significant, and long-term effects.
-
3.
Differences in numbers and community structure of microorganisms in soil between Bt and non-Bt crops were not statistically significant and transient.
-
4.
Although Cry proteins bind rapidly on clays and humic substances, there is little evidence for the accumulation of the proteins in soils in the field, even after years of continuous cultivation of Bt cotton.
References
Balachandar D, Raja P, Nirmala K, Rithyl TR, Sundaram SP (2008) Impact of transgenic Bt-cotton on the diversity of pink-pigmented facultative methylotrophs. World J Microbiol Biotechnol 24:2087–2095
Baumgarte S, Tebbe CC (2005) Field studies on the environmental fate of the Cry1Ab Bt-toxin produced by transgenic maize (MON810) and its effect on bacterial communities in the maize rhizosphere. Mol Ecol 14:2539–2551
Blackwood CB, Buyer JS (2004) Soil microbial communities associated with Bt and non-Bt corn in three soils. J Environ Qual 33:832–836
Brady NC, Weil RR (1999) The nature and properties of soils. Prentice Hall Inc., Upper Saddle River
Brookes G, Barfoot P (2005) GM crops: the global economic and environmental impact—the first nine years 1996–2004. Agric Bio Forum 8:187–196
Brusetti L, Francia P, Bertolini C, Pagliuca A, Borin S, Sorlini C, Abruzzese A, Sacchi G, Viti C, Giovannetti L, Giuntini E, Bazzicalupo M, Daffonchio D (2004) Bacterial communities associated with the rhizosphere of transgenic Bt 176 maize (Zea mays) and its non-transgenic counterpart. Plant Soil 266:11–21
Castaldini M, Turrini A, Sbrana C, Benedetti A, Marchionni M, Mocali S, Fabiani A, Landi S, Santomassimo F, Pietrangeli B, Nuti MP, Miclaus N, Giovannetti M (2005) Impact of Bt corn on rhizospheric and soil eubacterial communities and on beneficial mycorrhizal symbiosis in experimental microcosms. Appl Environ Microbiol 71:6719–6729
Crecchio C, Stotzky G (1998) Insecticidal activity and biodegradation of the toxin from Bacillus thuringiensis subsp. kurstaki bound to humic acids from soil. Soil Biol Biochem 30:463–470
Dale PJ, Clarke B, Fontes EMG (2002) Potential for the environmental impact of transgenic crops. Nat Biotechnol 20:567–580
Devare MH, Jones CM, Thies JE (2004) Effect of Cry3Bb transgenic corn and tefluthrin on the soil microbial community: biomass, activity, and diversity. J Environ Qual 33:837–843
Donegan KK, Palm CJ, Fieland VJ, Porteous LA, Ganio LM, Schaller DL, Bucao LQ, Seidler RJ (1995) Changes in levels, species and DNA fingerprints of soil microorganisms associated with cotton expressing the Bacillus thuringiensis var. kurstaki endotoxin. Appl Soil Ecol 2:111–124
Dunfield KE, Germida JJ (2004) Impact of genetically modified crops on soil-and plant-associated microbial communities. J Environ Qual 33:806–815
Griffiths BS, Caul S, Thompson J, Birch ANE, Scrimgeour C, Andersen MN, Cortet J, Messéan A, Sausse C, Lacroix B, Krogh PH (2005) A comparison of soil microbial community structure, protozoa and nematodes in field plots of conventional and genetically modified maize expressing the Bacillus thuringiensis is CryIAb toxin. Plant Soil 275:135–146
Gupta VVSR, Watson S (2004) Ecological impacts of GM cotton on soil biodiversity. Report to Australian Government Department of the Environment and Heritage, Canberra, ACT, Australia
Gupta VVSR, Roberts GN, Neate SM, McClure SG, Crisp P, Watson SK (2002) Impact of Bt-cotton on biological processes in Australian soils. In: Akhurst RJ, Beard CE, Hughes PA (eds) Proceedings of the fourth pacific rim conference on the biotechnology of Bacillus thuringiensis and its environmental impacts. CSIRO, Australia, pp 191–194
Head G, Surber JB, Watson JA, Martin JW, Duan JJ (2002) No detection of Cry1Ac protein in soil after multiple years of transgenic Bt cotton (Bollgard) use. Environ Entomol 31:30–36
Hossain F, Pray CE, Lu Y, Huang J, Fan C, Hu R (2004) Genetically modified cotton and farmers’ health in China. Int J Occup Environ Health 10:296–303
Hu HY, Xiao XL, Zhang WZ, Sun JG, Zhang QW, Liu XZ, Yu Y (2009) Effects of repeated cultivation of transgenic Bt cotton on functional bacterial populations in rhizosphere soil. World J Microbiol Biotechnol 25:357–366
Huang J, Hu R, Rozelle S, Pray C (2005) Insect-resistant GM rice in farmer’s fields: assessing productivity and health effects in china. Science 308:688–690
ISAAA (2006) Global area of biotech crops. International service for the acquisition of agri-biotech applications. http://www.isaaa.org/resources/publications/briefs/35/pptslides/Brief35slides.pdf
James C (2010) Global status of commercialized biotech/GM crops: 2010. ISAAA Brief No. 43, ISAAA, Ithaca, NY
Kapur M, Bhatia R, Pandey G, Pandey J, Paul D, Jain RK (2010) A case study for assessment of microbial community dynamics in genetically modified Bt cotton crop fields. Curr Microbiol 61:118–124
Knox OGG, Constable GA, Pyke B, Gupta VVSR (2006) Environmental impact of conventional and Bt insecticidal cotton expressing one and two cry genes in Australia. Aust J Agric Res 57:501–509
Koskella J, Stotzky G (1997) Microbial utilization of free and clay-bound insecticidal toxins from Bacillus thuringiensis and their retention of insecticidal activity after incubation with microbes. Appl Environ Microbiol 63:3561–3568
Koskella J, Stotzky G (2002) Larvicidal toxins from Bacillus thuringiensis sub spp. kurstaki, morrisoni and israelensis have no microbicidal or microbiostatic activity against selected bacteria, fungi and algae in vitro. Can J Microbiol 48:262–267
Lamarche J, Hamelin RC (2007) No evidence of an impact on the rhizosphere diazotroph community by the expression of Bacillus thuringiensis Cry1Ab toxin by Bt white spruce. Appl Environ Microbiol 73(20):6577–6583
Liu B, Zeng Q, Yan F, Xu H, Xu C (2005) Effects of transgenic plants on soil microorganisms. Plant Soil 271:1–13
Marvier M, McCreedy C, Regetz J, Kareiva P (2007) A meta-analysis of effects of Bt cotton and maize on non-target invertebrates. Science 316:1475–1477
Mina U, Anita C, Anju K (2011) Effect of Bt cotton on enzymes activity and microorganisms in rhizosphere. J Agric Sci 3:96–104
Mirkov TE (2003) The molecular basis of genetic modification and improvement of crops. In: Chrispeels MJ, Sadava DE (eds) Plants, genes, and crop biotechnology 2nd edn. Jones and Bartlett, Boston, pp 124–151
Muchaonyerwa P, Waladde S, Nyamugafata P, Mpepereki S, Ristori GG (2004) Persistence and impact on microorganisms of Bacillus thuringiensis proteins in some Zimbabwean soils. Plant Soil 266:41–46
O’Callaghan M, Travis RG, Burgess EPJ, Malone LA (2005) Effects of plants genetically modified for insect resistance on non-target organisms. Annu Rev Entomol 50:271–292
Oger P, Mansouri H, Dessaux Y (2000) Effect of crop rotation and soil cover on alteration of the soil microflora generated by the culture of transgenic plants producing opines. Mol Ecol 9:881–890
Palm CJ, Schaller DL, Donegan KK, Seidler RJ (1996) Persistence in soil of transgenic plant produced Bacillus thuringiensis var. kurstaki δ-endotoxin. Can J Microbiol 42:1258–1262
Perlak FJ, Deaton RW, Armstrong TA, Fuchs RL, Sims SR, Greenplate JT, Fischhoff DA (1990) Insect resistant cotton plants. Biotechnology (N.Y.) 8:939–943
Ream JE, Sims SR, Leach JN (1994) Aerobic soil degradation of Bacillus thuringiensis var. kurstaki HD-73 protein bioactivity. Monsanto Company Laboratory Project MSL 13267(11), Monsanto, St.Louis, MO
Rui YK, Yi GX, Zhao J, Wang BM, Li JH, Zhai ZX, He Z, Li QX (2005) Changes of Bt toxin in the rhizosphere of transgenic Bt cotton and its influence on soil functional bacteria. World J Microbiol Biotechnol 21:1279–1284
Sarkar B, Patra AK, Purakayastha TJ (2008) Transgenic Bt cotton affects enzyme activity and nutrient availability in a sub-tropical inceptisol. J Agron Crop Sci 194:289–296
Sarkar B, Patra AK, Purakayastha TJ, Megharaj M (2009) Assessment of biological and biochemical indicators in soil under transgenic Bt and non-Bt cotton crop in a sub-tropical environment. Environ Monit Assess 156:595–604
Saxena D, Stotzky G (2001) Bacillus thuringiensis (Bt) toxin released from root exudates and biomass of Bt corn has no apparent effect on earthworms, nematodes, protozoa, bacteria, and fungi in soil. Soil Biol Biochem 33:1225–1230
Schubert D (2002) A different perspective on GM food. Nat Biotechnol 20:969
Sessitsch A, Gyamfi S, Tscherko D, Gerzabek MH, Kandeler E (2004) Activity of microorganisms in the rhizosphere of herbicide treated and untreated transgenic glufosinate-tolerant and wild type oilseed rape grown in containment. Plant Soil 266:105–116
Shen RF, Cai H, Gong WH (2006) Transgenic Bt cotton has no apparent effect on enzymatic activities or functional diversity of microbial communities in rhizosphere soil. Plant Soil 285:149–159
Sims SR, Ream JE (1997) Soil inactivation of the Bacillus thuringiensis subsp. kurstaki Cry2A insecticidal protein within transgenic cotton tissue: Laboratory, microcosm and field studies. J Agric Food Chem 45:1502–1505
Singh RJ, Ahlawat IPS, Singh S (2013) Effects of transgenic Bt cotton on soil fertility and biology under field conditions in subtropical inceptisol. Environ Monit Assess 185:485–495
Sun CX, Chen LJ, Wu ZJ, Zhou LK, Shimizu H (2007) Soil persistence of Bacillus thuringiensis (Bt) toxin from transgenic Bt cotton tissues and its effect on soil enzyme activities. Biol Fertil Soils 43:617–620
US Environmental Protection Agency (EPA) (2001) SAP report no 2000–07, sets of scientific issues being considered by the environmental protection agency regarding: Bt plant pesticides risk and benefits assessments. www.epa.gov/scipoly/sap/meetings/2000/october/octoberfinal.pdf
Valasubramanian R (2001) Transgenic cotton expressing the Bacillus thuringiensis endotoxin: effects on soil microflora and non-target soil organisms with special reference to earthworms. Report submitted to MEC and RCGM by Mahyco Life Science Research Center, Jalna, India
Velmourougane K, Sahu A (2013) Impact of transgenic cottons expressing cry1Ac on soil biological attributes. Plant Soil Environ 59:108–114
Wallimann T (2000) Bt toxin: assessing GM strategies. Science 287:41
Wu WX, Ye QF, Min H, Duan XJ, Jin WM (2004) Bt-transgenic rice straw affects the culturable microbiota and dehydrogenase and phosphatase activities in a flooded paddy soil. Soil Biol Biochem 36:289–295
Yan WD, Shi WM, Li BH, Zhang M (2007) Over expression of a foreign Bt gene in cotton affects the low-molecular-weight components in root exudates. Pedosphere 17:324–330
Yu L, Berry RE, Croft BA (1997) Effects of Bacillus thuringiensis toxins in transgenic cotton and potato on Folsomia candida (Collembola: Isotomidae) and Oppia nitens (Acari: Orbatidae). J Econ Entomol 90:113–118
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Velmourougane, K., Blaise, D. (2014). Transgenic Cotton and Its Impact on Microbial Diversity. In: Maheshwari, D. (eds) Bacterial Diversity in Sustainable Agriculture. Sustainable Development and Biodiversity, vol 1. Springer, Cham. https://doi.org/10.1007/978-3-319-05936-5_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-05936-5_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-05935-8
Online ISBN: 978-3-319-05936-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)