Abstract
The development and validation of an analytical method based on a solid-phase extraction (SPE) prior to liquid chromatography-tandem mass spectrometry (LC-MS/MS) to determine methyl benzylidene camphor (MBC) and its major metabolite, i.e., carboxy benzylidene camphor (CBC), in urine from users of sunscreen cosmetic products containing MBC has been carried out.
Some content of this chapter has been published in Anal Methods (2013) 664:178–184.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Urine Sample
- Select Reaction Monitoring
- Cosmetic Product
- Anhydrous Magnesium Sulfate
- Aqueous Standard Solution
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
1.1 Aim of the Study
The development and validation of an analytical method based on a solid-phase extraction (SPE) prior to liquid chromatography-tandem mass spectrometry (LC-MS/MS) to determine methyl benzylidene camphor (MBC) and its major metabolite, i.e., carboxy benzylidene camphor (CBC), in urine from users of sunscreen cosmetic products containing MBC has been carried out.
Since CBC is not a commercially available standard, it was synthesized from MBC. The purified product was used as analytical standard in the development and validation of the analytical methodology.
Furthermore, the aim of the study also includes the detection of the other major phase I metabolite of MBC, i.e., CBC-OH, and the formation and excretion assessment of the phase II metabolites, mainly glucuronide conjugates, by appropriate enzymatic hydrolysis treatment of urine samples.
1.2 Background and Current Status of the Issue
Different methodologies that allow the determination of MBC in human plasma, urine and breast milk by liquid chromatography with UV/VIS detection (LC-UV/VIS) (Janjua et al. 2008), LC-MS/MS (Volkel et al. 2006; Schauer et al. 2006) and gas chromatography-mass spectrometry (GC-MS) (Hany and Nagel 1995; Schlumpf et al. 2008) have been found. However, some of these methods involve the use of long and laborious extraction procedures such as liquid-liquid extraction (LLE) (Schlumpf et al. 2008) and gel permeation chromatography (Hany and Nagel 1995), while others do not consider the contribution of metabolites to consider the overall body disposition process of MBC (Janjua et al. 2008). Furthermore, the methodologies described in the literature considering the contribution of metabolites of MBC do not allow, however, the determination of analytes using a single analytical method, and then different ionization sources are employed depending on the compound. Moreover, the analytical validation of these methods is not fully complete.
2 Experimental
2.1 Reagents and Samples
Methyl benzylidene camphor (MBC), also named 3-(4′-methylbenzylidene) camphor, 99.7 % from Guinama SL (Valencia) and carboxy benzylidene camphor (CBC), also named 3-(4′-carboxibenziliden) camphor, >95 %, which was synthesized as described in Sect. 9.2.3, were used as analytical standards. Benzophenone-d10 (BZ-d), 99 % D atom from Sigma-Aldrich (Schnelldorf) was used as deuterated internal standard. Some properties of interest of these compounds are shown in Table 9.1.
The solvents used were absolute ethanol (EtOH), LC grade methanol (MeOH), LC grade acetonitrile (ACN) and LC ultrapure grade acetone, all from Scharlab (Barcelona). Deionized water was obtained from a NANOpure II water purification system (R ≥ 18 mΩ cm) from Barnstead (Boston).
Formic acid (FA) from Fluka Chemie (Steinheim), analysis grade 37 % hydrochloric acid (d = 1.19 g mL−1) and analysis grade sodium hydroxide, both from Scharlab (Barcelona), and ß-glucuronidase from Helix pomatia (type HP-2) with 116 300 U mL−1 activity, and sulfatase activity ≤7,500 U mL−1, from Sigma-Aldrich (Steinheim), were also employed.
Urine samples used to develop and validate the method were obtained from different volunteers who had not applied any cosmetic product containing MBC and stored at 4 °C until analysis.
All reagents and solvents used in the synthesis of CBC (see Sect. 9.2.3) were of analytical or purest grade and obtained from Fluka Chemika (Buchs) or Strem Chemicals (Newburyport). Deuterochloroform (CDCl3) 99.9 % D atom, and NMR grade trimethylsilane (TMS) both from Sigma-Aldrich (Steinheim) were also used.
2.2 Instruments and Material
A L-7100 liquid chromatograph® connected to a L-7420® UV/VIS detector both from Hitachi (Tokyo) was used. The chromatographic separation was carried out on a LiChrospher® RP-18 analytical column (5 μm particle size, 125 mm long, 4 mm internal diameter) from Merck (Darmstadt). The UV/VIS detection was performed at the wavelength of 300 nm. Data acquisition was controlled by the D-7000 HSM software from Hitachi (Tokyo).
The LC-MS/MS system consisted of an Acquity UltraPerformance LC® liquid chromatograph coupled to a TQD® triple quadrupole mass spectrometer from Waters (Barcelona). A CLAN Technology high purity generator (Sevilla) provided nitrogen that was used as both ionization source and nebulizer gas. The MS/MS spectra were acquired by collision-induced dissociation (CID) of the selected precursor ions using Premier® argon from Air Products (Barcelona) as collision gas. The acquisition of data, and qualitative and quantitative analyses were performed using the MassLynx software. An Acquity UPLC BEH C18® analytical column (1.7 μm particle size, 50 mm long, 2.1 mm internal diameter) with an Acquity BEH C18 Vanguard® precolumn (1.7 μm particle size, 5 mm length, 2.1 mm internal diameter) also from Waters were used.
An Avance 300® spectrometer from Bruker (Madrid) to obtain the nuclear magnetic resonance (NMR) spectra at 299.95 MHz (CDCl3) for 1H NMR and at 75.43 MHz (CDCl3) for 13C NMR was also employed.
C18 solid-phase extraction cartridges (100 mg, 1 cm long, 5 mm internal diameter) and a SPE vacuum system from Varian (Barcelona) were used.
A MicropH 2001 pH meter from Crison (Alella), a ZX3® vibrating agitator from VELP Scientifica (Usmate), Silica Gel 60 F-254® thin layer plates for thin-layer chromatography (TLC) from Merck (Barcelona), a R-210/R-215® rotavapor from Buchi (Flawil) and a Precisterm® thermostated water bath, an incubation chamber and a Digitheat® oven, all from JP Selecta (Barcelona) were also used.
2.3 Synthesis and Characterization of Carboxy Benzylidene Camphor
The synthesis CBC was conducted through an initial reaction of MBC bromination followed by the Kornblum reaction to obtain the aldehyde analogue and a final oxidation with the Jones reagent (see Fig. 9.1).
Scheme of the CBC synthesis from MBC. Experimental conditions are described in Sect. 9.2.3
All reactions were performed using glassware dried in oven at 120 °C for at least 12 h and monitored by TLC analysis. Samples were dissolved in CDCl3 and TMS was used as internal standard (δ 0 ppm) in both 1H and 13C NMR experiments.
2.3.1 Step 1: Bromination
1.45 g (5.7 mmol) of MBC and 1.06 g (5.95 mmol) of N-bromosuccinimide were added to a 10 mL of carbon tetrachloride in a round bottom flask equipped with a refrigerator and a septum under nitrogen atmosphere.
The mixture was stirred at 80 °C under reflux and exposed to the action of a lamp (150 W) that provided white light. The reaction was monitored by TLC until MBC reacted completely (48 h). Then, the suspension was filtered and the filtrate was concentrated in vacuo. The solid residue [see Fig. 9.1, compound (1)] was washed with deionized water and recrystallized from isopropanol. 0.823 g of a pale yellow solid in the form of needles was obtained.
2.3.2 Step 2: Kornblum Reaction
0.411 g (4.9 mmol) of sodium bicarbonate was added to the stirred solution consisting of 0.823 g (1) (2.5 mmol) in 7 mL (200 mmol) of dimethylsulfoxide and the reaction mixture was kept stirring at room temperature for 48 h.
Next, the reaction mixture was diluted with deionized water (50 mL), and a liquid-liquid extraction (LLE) was proceeded in triplicate with 30 mL of ethyl acetate. The organic phases were washed with deionized water and dried with anhydrous magnesium sulfate. After concentrating in vacuo, 0.56 g of a brown oil [see Fig. 9.1, compound (2)] was obtained.
2.3.3 Step 3: Oxidation
The Jones reagent was prepared by adding 0.67 g (6.7 mmol) of chromium trioxide to 0.6 mL (11.3 mmol) of concentrated sulfuric acid, and the mixture was cautiously diluted with deionized water (5 mL).
1 mL of Jones reagent was added to a stirred solution consisting of 0.56 g of (2) (2 mmol) and 20 mL (272.9 mmol) of acetone. The reaction mixture was stirred at 15 °C and monitored by TLC until the reaction was completed (4 h). Next, 10 mL of an aqueous sodium hydroxide (1 M) was added and the reaction mixture was placed in an ice bath for 30 min. The organic phase obtained by LLE with diethyl ether (2 × 20 mL) was discarded and hydrochloric acid (2 M) was added to the basic aqueous phase until pH 1 conditions. Next, the reaction mixture was extracted with diethyl ether (3 × 20 mL) and the combined organic phases were dried with anhydrous magnesium sulfate. After evaporating in vacuo of the solvent, 0.258 g of CBC (see Fig. 9.1) was obtained. Spectral data for the characterization of CBC are shown in Table 9.2.
2.4 Analytical Method for the Determination of Methyl Benzylidene Camphor and its Major Metabolite in Urine
2.4.1 Preparation of Solutions
Separate solutions at a concentration level of 400 μg mL−1 of both the analytical standards (MBC, CBC) and the internal standard (BZ-d) were prepared in EtOH and kept at 4 °C.
To correct the error caused by the matrix effects (see Sect. 9.3.2), the standard addition calibration was used. Thus, a BZ-d solution (4 μg mL−1) in EtOH and a MBC and CBC solution (4 μg mL−1) in the urine sample to be analyzed were prepared from the respective stock solutions.
The calibrate solutions were prepared fortifying five aliquots (4,750 μL) of urine containing 0, 30, 60, 90 and 120 μL of the MBC and CBC standard solution (4 μg mL−1), to which 150, 120, 90, 60 and 30 μL of urine was added, respectively, to set the same final volume. Likewise, 50 μL of the BZ-d solution (4 μg mL−1) was also added to each calibrate solution.
2.4.2 Enzymatic Hydrolysis
To determine the total analyte content, i.e., unconjugated and glucuronide conjugated forms, 50 μL of the commercial solution of ß-glucuronidase was added to each calibrate solution. Alternatively, urine samples were also treated with deionized water to determine only the unconjugated content of analyte. Urine solutions were stirred and subjected to enzymatic hydrolysis by incubation at 37 °C for 12 h with ß-glucuronidase.
Subsequently, 50 μL of FA was added to the urine samples to stop the reaction. Hence, The urine solutions were adjusted to pH 3 for the subsequent SPE procedure of the analytes.
2.4.3 Solid-Phase Extraction
The C18 solid-phase extraction cartridge was conditioned with 2 mL of EtOH followed by 2 mL of deionized water. Next, the cartridge was loaded with 4 mL of each incubated urine solutions at a flow rate of 0.5 mL min−1, washed with 8 × 1 mL of deionized water and dried under vacuum for 10 min. The analytes were eluted with 3 × 0.35 mL of acetone. Then, the eluted fractions were evaporated to dryness in an oven at 50 °C (30 min) and reconstituted with 100 μL of deionized water:MeOH:ACN (2:1:1), (v/v/v) containing 0.1 % FA. Finally, the reconstituted samples were injected into the LC-MS/MS system.
2.4.4 LC-MS/MS Analysis
The mobile phase consisted of deionized water and MeOH:ACN (1:1, v/v), both containing 0.1 % FA. The elution gradient shown in Table 9.3 was used at a flow rate of 0.4 mL min−1 and at room temperature. The analytical column was equilibrated with a post-analysis time of 3 min. The injection volume was 5 μL.
The analytes were determined by selected reaction monitoring (SRM) in both positive and negative electrospray ionization (ESI) modes. The conditions of the triple quadrupole mass analyzer were as follows: temperature of gas ionization source, 120 °C; gas flow ionization source, 700 L h−1; nebulizer gas temperature, 300 °C; nebulizer gas flow, 25 L h−1; capillary voltage, 3,900 V. Dwell time was set to 37 ms. Two SRM transitions to quantify and to confirm were selected for each compound of interest. They are specified in Table 9.4, along with other relevant information.
Calibrates were obtained by representing the ratio of areas between each analyte and internal standard (BZ-d) versus the added concentration. Figure 9.2 shows the SRM chromatograms for the compounds of interest obtained by applying the described SPE-LC-MS/MS procedure to a urine sample containing no analytes, fortified (at 200 ng mL−1) and unfortified.
Total ion chromatograms (TIC) in positive (a) and negative (b) ionization modes and selected reaction monitoring (SRM) chromatograms of MBC (c, ESI+), BZ- d (d, ESI+) and CBC (e, ESI-) obtained by analyzing a urine sample containing no compounds (red line) and the same urine sample fortified at a concentration of 200 ng mL−1 (blue line). Experimental conditions are described in Sect. 9.2.4
3 Results and Discussion
3.1 SPE Procedure: Study of the Variables
Preliminary assays were carried out to verify the SPE conditions using the LC-UV/VIS system.
C18 cartridges were used to show the suitable capacity to retain the compounds of interest. Thus, after duly conditioning of the cartridge with 2 mL of EtOH followed by 2 mL of deionized water, aqueous standard solution of MBC and CBC (1 μg mL−1) under different conditions depending on the performed study was loaded and quantitative elution of the analytes with 3 × 0.35 mL of acetone was proceeded. The effectiveness of the elution conditions was checked (R > 95 %).
The eluted fraction was evaporated under a stream of air, reconstituted with 100 μL of deionized water:MeOH:ACN, 2:1:1 (v/v/v) containing 0.1 % FA and injected into the LC-UV/VIS system. The mobile phase consisted of deionized water (0.1 % FA) and (MeOH:ACN, 1:1, 0.1 % FA). The employed gradient of elution at a flow rate of 1 mL min−1 and at room temperature was as follows: 0–5 min, 60 % (MeOH:ACN, 1:1, 0.1 % FA); 5–10 min, linear gradient from 60 to 70 % (MeOH:ACN, 1:1, 0.1 % FA); 10–10.5 min, linear gradient from 70 to 100 % (MeOH:ACN, 1:1, 0.1 % of FA), maintained for 5 min. The analytical column was equilibrated with a post-analysis time of 5 min. The injection volume was 20 μL. Under these conditions, the resulting retention times were 2.6 min for CBC and 8.2 min for MBC.
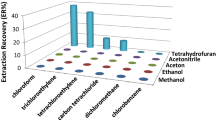
First, the retaining process of the SPE procedure was studied. Hence, 50 μL of FA, deionized water or sodium hydroxide was added to three aqueous standard solutions, resulting pH 3, 5 and 12, respectively. Then, 1 mL of each of these solutions was loaded into the C18 cartridges. The results obtained from triplicate LC-UV/VIS analysis showed that the signal of CBC at pH 3 was greater than at pH 5 (see Fig. 9.3).
At pH 12, no signal was observed for CBC, thus showing that no retention was produced for CBC in the C18 cartridges under these conditions. The findings are consistent with its pKa value (see Table 9.1), so that CBC remains quantitatively in its non-ionized form at pH below 3, enhancing hydrophobic interactions with C18 cartridges. The signal of MBC was kept constant at all three pH conditions, being slightly lower at pH 12. Therefore, urine solutions were adjusted at pH 3 prior to the SPE step.
To study the load capacity, different aliquots of aqueous standard solutions (1 μg mL−1) were loaded into two C18 cartridges assembled in series. Thereafter, SPE cartridges were disassembled and the procedure described above was carried out in both cartridges. As the retention process in the first cartridge when loading 4 mL of solution was almost quantitative (R > 99 %), it was concluded that there was no analyte loss under these conditions. Recovery of the SPE procedure was estimated comparing the signal obtained from the eluted and reconstituted fraction and external standards. The obtained values were around 60 and 95 % for MBC and CBC, respectively. Due to the low obtained value of MBC, an alternative way to dry the eluted fraction from the SPE procedure was considered. Thus, the oven assisted evaporation at 50 °C for 30 min was studied and compared with the results obtained under the air stream evaporation. The recovery of MBC by using the oven assisted evaporation was improved up to 90 %, while CBC results were maintained constant (95 %). Therefore, SPE eluted fractions were dried before reconstitution with the mobile phase in the oven at 50 °C (30 min) to avoid the analyte losses showed when air stream was used to evaporate to dryness.
3.2 LC-MS/MS Analysis
The MS/MS spectra of the compounds of interest were acquired by CID experiments by direct infusion for standard solutions of each target compound (200 μg mL−1) prepared in MeOH, operating with electrospray ionization in positive (ESI+) or negative (ESI−) modes.
The most intense MS/MS transitions for MBC and BZ-d were obtained with positive electrospray ionization, through the corresponding protonated molecular ions ([M+H]+). However, the corresponding deprotonated molecular ion ([M-H]−) obtained by negative electrospray ionization was more intense for CBC. Under these conditions, tandem mass spectrometry parameters were optimized (see Table 9.4).
3.3 Study of Sample Stability
The stability of MBC and CBC in fortified urine samples stored at 4 °C was studied. As can be seen in Fig. 9.4, the reproducibility of the LC-MS/MS analysis from the first to the sixth day indicates the stability of the analytes during at least this period of time, under the study conditions. Therefore, the urine samples were kept at 4 °C until analysis.
Peak area ratios between MBC (a) and CBC (b) and the internal standard obtained by analyzing a urine sample fortified at 50 ng mL−1 and stored at 4 °C at different times. Experimental conditions are described in Sect. 9.2.4
3.4 Validation of the Analytical Method: Study of the Interferences
The matrix effects, which may occur in both extraction and detection processes, were evaluated. Hence, the differences between the responses obtained from a series of standard solutions (30–150 ng mL−1) prepared in a urine sample containing no analytes from a single volunteer (single urine calibrate) were statistically compared to those obtained from a series prepared in a pool of urine samples containing no analytes from five volunteers (pool urine calibrate). Moreover, all the solutions were fortified (50 μL) with BZ-d (4 μg mL−1) and pH was adjusted by adding 50 μL of AF (pH 3). Then, the described SPE procedure followed by the LC-MS/MS analysis was carried out.
As can seen in Table 9.5, statistically different slopes were obtained when comparing both calibrates for MBC. In the case of CBC, they were statistically comparable.
Therefore, the developed analytical methodology considered standard addition calibration to correct the proportional errors caused by potential matrix interferences in order to correctly determine the content of the target compounds in the urine samples.
3.4.1 Accuracy
Accuracy was evaluated using the developed method by analyzing urine samples fortified with known amounts of MBC and CBC, from volunteers who had not applied any cosmetic product containing MBC. The obtained values are shown in Table 9.6.
Standard deviations were obtained as the standard deviation of the extrapolated value in the standard addition line (Miller and Miller 2005). The accuracy of the analytical method was statistically confirmed by Student’s t-test, thus showing the absence of significant differences between concentration values found by the developed method and concentration values of the fortified urine samples (see Annex III.3).
3.4.2 Other Analytical Parameters
Calibration curves (N = 5) were found to be linear for CBC and MBC over a range of concentrations from 30 to 150 ng mL−1 (working range) with a regression coefficient greater than 0.995 in all cases.
The limits of detection (LOD) and quantification (LOQ) of the analytes were estimated based on the International Conference on Harmonization on validation of analytical procedures (ICH guidelines 2005) (see Table 9.7).
The intra-day variability (repeatability) was assessed by analyzing in the same batch five fortified (50 ng mL−1) urine samples containing initially no analytes. The inter-day variability (reproducibility) was determined by analyzing in five different batches a single fortified (50 ng mL−1) urine sample containing initially no analytes. The intra- and inter-day variabilities expressed as relative standard deviation (RSD) are shown in Table 9.8.
3.5 Application of the Analytical Method
To apply the proposed analytical method to urine samples from users of cosmetic products containing MBC in their composition, a volunteer was instructed to apply 13 g of the cosmetic product in the morning and evening for three consecutive days, making a total of 6 applications. The cosmetic product was prepared in the laboratory according to an adapted protocol (Jordán and Jordán 1991) containing 4 % of MBC (see Annex II.5). The applied doses are in the normal range of application thickness for sunscreen products (0.5–1 mg cm−2) which, moreover, is well below from the recommended dose to get the labelled value of Sun Protection Factor (SPF) (2 mg cm−2) (Chisvert and Salvador 2007). A shower per day just before the second daily application was allowed to the volunteer. The reason to conduct this study was to simulate a real situation of sunscreen cosmetic product application, which usually involves the application of repeated doses over sunbathing time.
During the first 4 days from the application of the cosmetic product, the total excreted urine was collected over periods of 24 h in sterile commercially available containers. The urine volume was measured and stored at 4 °C until analysis. Taking into account the total collected urine is possible to determine the content of MBC excreted by urine. Thus, urine sample 1 (U1) corresponded to the urinary excretions collected during the first 24 h, urine sample 2+3 (U2+3) was prepared by mixing the excreted urine collected during the second and third day and finally, urine sample 4 (U4) corresponded to the urine excretion collected during the fourth day since the first application. An additional urine sample was collected just before the first application (U0) to verify the initial absence of MBC and/or CBC.
Urine samples (U1, U2+3 and U4) were divided into two fractions. To each urine fraction, deionized water or ß-glucuronidase solution were added, thus allowing the determination of the unconjugated analyte content or the total (i.e., unconjugated and glucuronide conjugate) analyte content, respectively. Thereby, each urine sample was analyzed in duplicate (with and without enzymatic treatment) by applying the SPE-LC-MS/MS analytical methodology described above.
As can be seen in Fig. 9.5, MBC was determined only in U1, thus concluding that MBC follows an extensive biotransformation process in human body. By contrast, increasing amounts of both unconjugated and glucuronide conjugated forms of CBC were detected along with the study. The highest concentration of CBC was found in U4, that corresponds to the urine sample collected the day after the last application of the cosmetic product, showing a clear process of bioaccumulation.
Found amounts of MBC (a) and CBC (b) in urine samples collected at times specified in Sect. 9.3.3 from a volunteer after repeated topical application of a sunscreen cosmetic product containing MBC (4 %). Error bars show the standard deviation of the results. The results are expressed in terms of absolute mass considering the dilution factor corresponding to the total volume collected in each case. Unconjugated form content, (square); glucuronide conjugated form content, (filled square)
Figure 9.6 shows a SRM chromatogram obtained after the application of the SPE-LC-MS/MS methodology to the U2+3 urine sample, with and without enzymatic treatment.
SRM chromatograms for CBC obtained by analyzing U0 (black line) and U2+3 (red line) urine samples from a volunteer who had repeatedly applied a sunscreen containing MBC. U2+3 sample was also treated by enzymatic hydrolysis (blue line). Experimental conditions are described in Sect. 9.2.4 and the conditions of the study can be found in Sect. 9.3.3
The effectiveness of the deconjugation reaction is evident, thus showing that the glucuronide conjugated species of CBC contribute greatly to the total content of CBC excreted by urine when topical incorporation of MBC is increased.
New conditions were included into the LC-MS/MS analysis to consider the detection of another phase I metabolite of MBC described in literature, i.e., CBC-OH. SRM parameters were adapted from Schauer et al. (2006), applying the same conditions of voltage and collision energy as for CBC (see Table 9.9).
The contribution of the different isomers of CBC-OH as a consequence of the in vivo biotransformation process of MBC can be seen in Fig. 9.7, with predominance of one isomer as described in the literature (Schauer et al. 2006).
SRM chromatograms for CBC-OH obtained by analyzing urine samples U0 (black line) and U2+3 (red line) from a volunteer who had repeatedly applied a sunscreen cosmetic product containing MBC. U2+3 sample was also treated by enzymatic hydrolysis process (blue line). Experimental conditions are described in Sect. 9.2.4 and the conditions of the study can be found in Sect. 9.3.3
Furthermore, it can be concluded that the contribution of the conjugated species by glucuronidation to the total content of this metabolite was also important, when comparing both enzymatic treated and untreated samples.
4 Conclusions
An analytical method based on the combination of SPE and a subsequent process by LC-MS/MS analysis to determine MBC and its metabolite, CBC, in human urine has been developed and validated. Since CBC was not commercially available, it was synthesized from MBC.
The standard addition calibration was employed to correct the proportional errors caused by the observed matrix effects. Under this approach, statistically accurate results were obtained by analyzing urine samples previously fortified with the analytes.
The methodology has been successfully applied to the analysis of urine samples collected from a volunteer that had applied repeatedly a sunscreen cosmetic product containing MBC. A low content of MBC in the urine sample collected 24 h after the first application was determined. However, the largest contribution to the MBC excretion corresponds to the CBC species conjugated by glucuronidation, which increased with the number of sunscreen applications. Furthermore, CBC-OH, another major metabolite of MBC, was identified and detected, and it was found that CBC-OH also prevailed mainly as glucuronide conjugated in urine samples.
References
Chisvert, A., & Salvador, A. (2007). UV filters in sunscreens and other cosmetics. Regulatory aspects and analytical methods. In A. Salvador & A. Chisvert (Eds.), Analysis of cosmetic products (pp. 83–120). Amsterdam: Elsevier.
Hany, J., Nagel, R. (1995). Detection of sunscreen agents in human breast milk. Dtsch Lebensm Rundsch, 91, 341–345.
ICH validation of analytical procedures methodology: Text and methodology Q2(R1), ICH harmonised tripartite guidelines, Adopted November (2005).
Janjua, N.R., Kongshoj, B., Andersson, A.M., Wulf, H.C. (2008). Sunscreens in human plasma and urine after repeated whole-body topical application. Journal of the European Academy of Dermatology and Venearology, 22, 456–461.
Jordán, M.C., Jordán, A.M. (1991). Formulario de Cosmética. NAU Llibres, Valencia.
Miller, J. C., & Miller, J. N. (2005). Statistics and chemometrics for analytical chemistry (5th ed.). Upper Saddle River: Prentice Hall.
Schauer, U.M.D., Völkel, W., Heusener, A., Colnot, T., Broschard, T.H., von Landenberg, F., Dekant, W. (2006). Kinetics of 3-(4-methylbenzylidene)camphor in rats and humans after dermal application. Toxicology and Applied Pharmacology, 216, 339–346.
Schlumpf, M., Kypke, K., Voekt, C.C., Birchler, M., Durrer, S., Faass, O., Ehnes, C., Fuetsch, M., Gaille, C., Henseler, M., Hofkamp, L., Maerkel, K., Reolon, S., Zenker, A., Timms, B., Tresguerres, J.A.F., Lichtensteiger, W. (2008). Endocrine active UV filters: Developmental toxicity and exposure through breast milk. Chimia, 62, 345–351.
Völkel, W., Colnot, T., Schauer, U.M.D., Broschard, T.H., Dekant, W. (2006). Toxicokinetics and biotransformation of 3-(4-methylbenzylidene)camphor in rats after oral administration. Toxicology and Applied Pharmacology, 216, 331–338.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
León González, Z. (2014). Determination of Methyl Benzylidene Camphor and its Main Metabolite in Urine by Solid-Phase Extraction and Liquid Chromatography Tandem Mass Spectrometry. In: Percutaneous Absorption of UV Filters Contained in Sunscreen Cosmetic Products. Springer Theses. Springer, Cham. https://doi.org/10.1007/978-3-319-01189-9_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-01189-9_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-01188-2
Online ISBN: 978-3-319-01189-9
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)