Abstract
IL-17A is a proinflammatory cytokine with critical effects on many cell types. It is part of a larger family of cytokines with important roles in protection against pathogens. The IL-17A receptors are IL-17RA and IL-17RC. They are expressed on most cell types. IL-17A, in synergy with other cytokines such as TNF-α, induces the production of cytokines, chemokines, and mediators of tissue destruction in several cell types. The identification of IL-17A and, later on, of Th17 cells has modified the established Th1 and Th2 paradigm, led to the definition of a new CD3+ CD4+ effector T cell subset, and introduced a new paradigm to explain the origin of several autoimmune events. However, this paradigm shift tended also to identify the effects of IL-17A with those of Th17 cells and vice versa. This view might be insufficient to explain the role of IL-17A in infections and autoimmune models. IL-17A is in fact produced by several other cell types involved in host defense, autoimmunity, and inflammation, and they might also be involved in IL-17A induced pathology.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 IL-17A and Other IL17 Family Members
In 1993, Rouvier et al. [1] described the cloning of a rodent cDNA sequence, termed CTLA8, from an activated T cell hybridoma using a subtractive hybridization approach and reported the sequence of the corresponding protein. This sequence had homology to an open reading frame encoded within a T cell-tropic c-herpesvirus, Herpesvirus Saimiri, and they suggested that it might be a novel cytokine [1]. Later on, the protein described was found to be a rat protein [2]. The human homologue was reported in 1995 and was mainly produced by activated CD4+ T cells, induced the secretion of IL-6 and IL-8 in human foreskin fibroblasts, and enhanced the expression of ICAM-1 [2]. This molecule was named IL-17. The mouse homolog was reported a year later [3, 4] and, in one case, was cloned from mouse NKT cells [3].
Human IL-17A is a glycoprotein containing 155 amino acid residues and shares 63 % amino acid identity with murine IL-17A (147 amino acids). Both human and mouse IL-17A are secreted as disulfide-linked homodimers. Now, the IL-17 family includes seven members (IL-17 or IL-17A, IL-17B, IL-17C, IL-17D, IL-17E or IL-25, and IL-17F plus the viral homologue ORF13 or vIL-17). In the mouse system, IL-17F has 45 % amino acid homology to IL-17A, followed by IL-17C (24 %), IL-17B (21 %), IL-17D (16 %), and IL-17E (16 %) [5]. IL-17A and IL-17F are secreted by overlapping populations of T cells and form both homodimers and heterodimers [6, 7]. IL-17B, IL-17C, and IL-17E differ substantially from IL-17A and IL-17F in the N terminus, having longer extensions. Furthermore, IL-17B is secreted as a noncovalent dimer [8]. Common features of the IL-17 family are a highly conserved carboxyl terminus and five spatially conserved cysteine residues. Four of these cysteines form a cystine knot fold. Transforming growth factor (TGF)-β, bone morphogenic protein, and nerve growth factor superfamilies show structural similarities but have an additional disulfide bond [8].
The genes for IL-17A and IL-17F are located on human chromosome 6p12.2, while the genes of the other IL-17 family members are located on different chromosomes: IL-17B on Chr 5q32, IL-17C on Chr 16q24.3, IL-17D on Chr 13q12.111, and IL-25 on Chr 14q11.2. Polymorphisms in IL-17A (rs2275913) gene have been weakly associated with rheumatoid arthritis [9], while polymorphism in IL-17F (rs763780) gene has been associated with a protective role in asthma [10]. Polymorphism at the 197A (IL-17A) and 7488T (IL-17F) alleles may influence the susceptibility to and pathophysiological features of ulcerative colitis [11]. In the Chinese population, rs2275913 and rs763780 are associated with immune diseases and infectious diseases, including asthma, Vogt–Koyanagi–Harada syndrome, bacterial bronchiolitis, and neuromyelitis optica [12–14].
2 The Receptors
When the mouse and the human IL-17 receptors (IL-17RA) were cloned, IL-17RA was found to be ubiquitously expressed and unique. It consisted of a single transmembrane receptor with an unusually long cytoplasmic tail [15, 16]. Now, five different receptors have been described, IL-17RA, IL-17RB, IL-17RC, IL-17RD, and IL-17RE [17]. The IL-17R family contains conserved structural characteristics such as extracellular fibronectin III-like domains and cytoplasmic similar expression to fibroblast growth factor, IL-17R, and Toll-IL-1R family (SEFIR) domains. IL-17A and IL-17F signal through a receptor complex composed of IL-17RA and IL-17RC [18]. In humans IL-17RA binds with higher affinity to IL-17A than to IL-17F, whereas IL-17RC associates with both IL-17A and IL-17F [18, 19]. Cristal structure of the IL-17F-IL-17RA interaction reveals the presence of a loop of IL-17RA which interacts with the deep groove at the interface of IL-17F homodimer [18]. IL-17RD has also been associated with the IL-17R complex and may contribute to IL-17A signaling [20]. IL-17RB is the receptor chain for IL-25 and IL-17B [21, 22]. The receptor for IL-17C appears to be a heterodimeric complex formed by IL-17RA (low-affinity interaction) and IL-17RE (high-affinity interaction) [23–25].
In conclusion, IL-17RA is shared among the members of the IL-17 family, while the specificity of the interaction mainly resides in the second subunit of the heterodimeric receptor complex. IL-17RC is common to IL-17A and IL-17F homo- and heterodimers; IL-17RB is specific for IL-25 and IL-17B, IL-17RE for IL-17C, and IL-17RD may be part of the IL-17A receptor complex [23–25]. Since IL-17RA is ubiquitously expressed, also the tissue specificity of the responses resides in the second receptor chain which is differently expressed in various tissues.
3 IL-17A as a Proinflammatory Cytokine
Several reports suggest that IL-17A has a critical role in the protection of the organism against bacteria and fungi due to the ability of IL-17A to recruit neutrophils to the infected organs/tissues [26, 27]. IL-17A and IL-17F can induce the production of proinflammatory cytokines (IL-1, IL-6, TNF-α, G-CSF, and GM-CSF), chemokines (CXCL1, CXCL5, IL-8, CCL2, and CCL7), antimicrobial peptides (defensins, lipocalin2, and S100 proteins), and matrix metalloproteinases (MMP1, MMP3, and MMP13) from different cell types. IL-17A also controls the expression of intercellular cell adhesion molecule 1 (ICAM-1) in keratinocytes as well as iNOS and cyclooxygenase-2 in chondrocytes. IL-17F is a weaker inducer of proinflammatory cytokine but seems to be produced by a wider range of cell types, including innate immune cells and epithelial cells. Not unexpectedly, IL-17A and IL-17F act in concert with other cytokines such as TNF-α, with which they strongly synergize in in vitro cellular assays and in vivo. It is expected that this characteristic is shared by the other IL-17 family members. Moreover, IL-17A contributes to germinal center formation and class switch recombination by its action on B cells [28, 29].
In summary, IL-17A, acting in synergy with other cytokines, contributes to modulate and control the inflammatory response. Some events, e.g., neutrophil recruitment, are indirectly mediated through the activity of IL-17A on cells which secrete chemotactic factors for neutrophils. Therefore, the mediators, secreted by activated cells, modulate the final outcome of the response to IL-17A.
4 IL-17A-Producing Cells
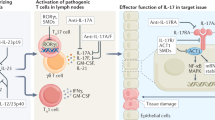
In 2000, Infante-Duarte et al. [30] recognized that IL-17A cannot be categorized as either a Th1 or a Th2 cytokine, but a major advance in IL-17 research came with the recognition of a novel T cell lineage producing IL-17A and/or IL-17F [31, 32]. This finding modified the established Th1-Th2 paradigm, led to the definition of the CD3+ CD4+ Th17 cell subset, and introduced a new paradigm to explain the origin of several autoimmune events.
In the mouse, IL-17A and/or IL-17F are however produced by several other cell types involved in host defense, autoimmunity, and inflammation, including subpopulations of CD8+ T cells (Tc17), γδ T cells [33], invariant natural killer (iNKT) cells [34], lymphoid tissue inducers [35], neutrophils [36], and macrophages [37]. In addition, IL-17A is produced by intestinal Paneth cells in an IL-23 independent fashion being one of the causes of a systemic inflammatory response syndrome [38], and IL-17F mRNA is expressed in colonic epithelial cells [39]. In particular, Jensen et al. [33] found that γδ T cells are uniquely suited for the initial IL-17A response, which often is elicited without a clear antigen exposure. They also found evidence for two distinct functional subsets of γδ T cells (Tγδ-17s and Tγδ-IFN-γs).
Yoshiga et al. [40] demonstrated that also iNKT cells synthesize and release IL-17A, accelerate IL-17 production by Th17 cells, and play a role in the development of collagen-induced arthritis (CIA). Moreover, they found that iNKT cells can be induced to secrete IL-17A through two pathways, one involving the direct activation of the TCR by glycolipid antigen and the other the IL-23-IL-23R signaling pathway [40]. It appears therefore that IL-17A is produced by different subsets of T cells, which are involved in both innate and adaptive immunity, and it is well suited to participate to both early and late phases of the immune response.
In addition, in IL-10- or IL-10R-deficient mice, even macrophages stimulated with lipopolysaccharide appear to produce high levels of IL-17A and IL-22 [41]. Addition of exogenous IL-10 abolishes IL-17A production both in Th17 cells and in macrophages [41], again suggesting that IL-17A production is highly regulated.
In humans, IL-17A has been detected in several T cell subsets, such as naive, central memory, and effector memory CD4+ IL-17+ T cells [42], NKT-like cells [43], macrophages [44], astrocytes [45], oligodendroglia [45], mast cells [46], neutrophils [47], and also a human myeloma cell line [48].
The large number of cell types able to secrete IL-17A and IL-17F, the migratory potential of the cells secreting these cytokines, the ubiquitous distribution of the receptor, and the synergism with other proinflammatory cytokines allow IL-17A and IL-17F to exert effects in multiple pathological conditions [26, 49]. In addition, Th17 cells act in close contact with their target cell, and IL-17A might have a short range of action and act in synergy with other cytokines secreted by Th-17 cells [50]. The role of other family members might be more restricted due to a more limited expression and tissue distribution of their receptors.
A pathological role for IL-17A in several autoimmune models of disease, such as experimental autoimmune encephalomyelitis (EAE) and arthritis, has been documented [5], and increased production of IL-17A has been reported in various human autoimmune and allergic diseases, such as rheumatoid arthritis [51], multiple sclerosis [45], psoriasis [52], and asthma [53]. All these findings suggest a pathogenic role in several human diseases which has been confirmed by the clinical efficacy of IL-17A neutralization in different disease [54].
References
Rouvier E, Luciani M-F, Mattei MG, Denizot F, Golstein P (1993) CTLA-8, cloned from an activated T cell, bearing AU-rich messenger RNA instability sequences, and homologous to a herpesvirus saimiri gene. J Immunol 150:5445–5456
Yao Z, Painter SL, Fanslow WC, Ulrich D, Macduff BM, Spriggs MK, Armitage RJ (1995) Cutting edge: human IL-17: a novel cytokine derived from T cells. J Immunol 155:5483–5486
Kennedy J, Rossi DL, Zurawski SM, Vega F Jr, Kastelein RA, Wagner JL, Hannum CH, Zlotnik A (1996) Mouse IL-17: a cytokine preferentially expressed by alpha beta TCR + CD4-CD8-T cells. J Interferon Cytokine Res 16:611–617
Yao Z, Timour M, Painter S, Fanslow W, Spriggs M (1996) Complete nucleotide sequence of the mouse CTLA8 gene. Gene 168:223–225
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852
Liang SC, Long AJ, Bennett F, Whitters MJ, Karim R, Collins M, Goldman SJ, Dunussi-Joannopoulos K, Williams CMM, Wright JF et al (2007) An IL-17F/A heterodimer protein is produced by mouse Th17 cells and induces airway neutrophil recruitment. J Immunol 179:7791–7799
Wright JF, Guo Y, Quazi A, Luxenberg DP, Bennett F, Ross JF, Qiu Y, Whitters MJ, Tomkinson KN, Dunussi-Joannopoulos K et al (2007) Identification of an interleukin 17F/17A heterodimer in activated human CD4+ T cells. J Biol Chem 282:13447–13455
Hymowitz SG, Filvaroff EH, Yin JP, Lee J, Cai L, Risser P, Maruoka M, Mao W, Foster J, Kelley RF et al (2001) IL-17s adopt a cystine knot fold: structure and activity of a novel cytokine, IL-17F, and implications for receptor binding. EMBO J 20:5332–5341
Nordang GB, Viken MK, Hollis-Moffatt JE, Merriman TR, Førre ØT, Helgetveit K, Kvien TK, Lie BA (2009) Association analysis of the interleukin 17A gene in Caucasian rheumatoid arthritis patients from Norway and New Zealand. Rheumatology (Oxford) 48:367–370
Kawaguchi M, Takahashi D, Hizawa N, Suzuki S, Matsukura S, Kokubu F, Maeda Y, Fukui Y, Konno S, Huang SK et al (2006) IL-17F sequence variant (His161Arg) is associated with protection against asthma and antagonizes wild-type IL-17F activity. J Allergy Clin Immunol 117:795–801
Arisawa T, Tahara T, Shibata T, Nagasaka M, Nakamura M, Kamiya Y, Fujita H, Nakamura M, Yoshioka D, Arima Y et al (2008) The influence of polymorphisms of interleukin-17A and interleukin-17F genes on the susceptibility to ulcerative colitis. J Clin Immunol 28:44–49
Chen J, Deng Y, Zhao J, Luo Z, Peng W, Yang J, Ren L, Wang L, Fu Z, Yang X et al (2010) The polymorphism of IL-17G-152A was associated with childhood asthma and bacterial colonization of the hypopharynx in bronchiolitis. J Clin Immunol 30:539–545
Shu Q, Yang P, Hou S, Li F, Chen Y, Du L, Jiang Z (2010) Interleukin-17 gene polymorphism is associated with Vogt–Koyanagi–Harada syndrome but not with Behçet’s disease in a Chinese Han population. Hum Immunol 71:988–991
Wang H, Zhong X, Wang K, Qiu W, Li J, Dai Y, Hu X (2012) Interleukin 17 gene polymorphism is associated with anti-aquaporin 4 antibody-positive neuromyelitis optica in the Southern Han Chinese—a case control study. J Neurol Sci 314:26–28
Yao Z, Fanslow WC, Seldin MF, Rousseau A-M, Painter SL, Comeau MR, Cohen JI, Spriggs MK (1995) Herpesvirus saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. Immunity 3:811–821
Yao Z, Spriggs MK, Derry JMJ, Strockbine L, Park LS, VandenBos T, Zappone JD, Painter SL, Armitage RJ (1997) Molecular characterization of the human interleukin-17 receptor. Cytokine 9:794–800
Shen F, Gaffen SL (2008) Structure–function relationships in the IL-17 receptor: implications for signal transduction and therapy. Cytokine 41:92–104
Ely LK, Fischer S, Garcia KC (2009) Structural basis of receptor sharing by interleukin 17 cytokines. Nat Immunol 10:1245–1251
Kuestner RE, Taft DW, Haran A, Brandt CS, Brender T, Lum K, Harder B, Okada S, Ostrander CD, Kreindler JL et al (2007) Identification of the IL-17 receptor related molecule IL-17RC as the receptor for IL-17F. J Immunol 179:5462–5473
Rong Z, Wang A, Li Z, Ren Y, Cheng L, Li Y, Wang Y, Ren F, Zhang X, Hu J et al (2009) IL-17RD (Sef or IL-17RLM) interacts with IL-17 receptor and mediates IL-17 signaling. Cell Res 19:208–215
Shi Y, Ullrich SJ, Zhang J, Connolly K, Grzegorzewski KJ, Barber MC, Wang W, Wathen K, Hodge V, Fisher CL et al (2000) A novel cytokine receptor-ligand pair. Identification, molecular characterization, and in vivo immunomodulatory activity. J Biol Chem 275:19167–19176
Rickel EA, Siegel LA, Yoon BR, Rottman JB, Kugler DG, Swart DA, Anders PM, Tocker JE, Comeau MR, Budelsky AL (2008) Identification of functional roles for both IL-17RB and IL-17RA in mediating IL-25-induced activities. J Immunol 181:4299–4310
Chang SH, Reynolds JM, Pappu BP, Chen G, Martinez GJ, Dong C (2011) Interleukin-17C promotes Th17 cell responses and autoimmune disease via interleukin-17 receptor E. Immunity 35:611–621
Ramirez-Carrozzi V, Sambandam A, Luis E, Lin Z, Jeet S, Lesch J, Hackney J, Kim J, Zhou M, Lai J et al (2011) IL-17C regulates the innate immune function of epithelial cells in an autocrine manner. Nat Immunol 12:1159–1166
Song X, Zhu S, Shi P, Liu Y, Shi Y, Levin SD, Qian Y (2011) IL-17RE is the functional receptor for IL-17C and mediates mucosal immunity to infection with intestinal pathogens. Nat Immunol 12:1151–1158
Ye P, Rodriguez FH, Kanaly S, Stocking KL, Schurr J, Schwarzenberger P, Oliver P, Huang W, Zhang P, Zhang J et al (2001) Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J Exp Med 194:519–527
Ye P, Garvey PB, Zhang P, Nelson S, Bagby G, Summer WR, Schwarzenberger P, Shellito JE, Kolls JK (2001) Interleukin-17 and lung host defense against Klebsiella pneumoniae infection. Am J Respir Cell Mol Biol 25:335–340
Mitsdoerffer M, Lee Y, Jaeger A, Kim HJ, Korn T, Kolls JK, Cantor H, Bettelli E, Kuchroo VK (2010) Proinflammatory T helper type 17 cells are effective B-cell helpers. Proc Natl Acad Sci USA 107:14292–14297
Wu HJ, Ivanov II, Darce J, Hattori K, Shima T, Umesaki Y, Littman DR, Benoist C, Mathis D (2010) Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32:815–827
Infante-Duarte C, Horton HF, Byrne MC, Kamradt T (2000) Microbial lipopeptides induce the production of IL-17 in Th cells. J Immunol 165:6107–6115
Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT (2005) Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol 6:1123–1132
Park H, Li Z, Yang XO, Chang SH, Nurieva R, Wang YH, Wang Y, Hood L, Zhu Z, Tian Q et al (2005) A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol 6:1133–1141
Jensen KD, Su X, Shin S, Li L, Youssef S, Yamasaki S, Steinman L, Saito T, Locksley RM, Davis MM et al (2008) Thymic selection determines gammadelta T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon gamma. Immunity 29:90–100
Michel ML, Keller AC, Paget C, Fujio M, Trottein F, Savage PB, Wong CH, Schneider E, Dy M, Leite-de-Moraes MC (2007) Identification of an IL-17-producing NK1.1(neg) iNKT cell population involved in airway neutrophilia. J Exp Med 204:995–1001
Cua DJ, Tato CM (2010) Innate IL-17-producing cells: the sentinels of the immune system. Nat Rev Immunol 10:479–489
Ferretti S, Bonneau O, Dubois GR, Jones CE, Trifilieff A (2003) IL-17, produced by lymphocytes and neutrophils, is necessary for lipopolysaccharide-induced airway neutrophilia: IL-15 as a possible trigger. J Immunol 170:2106–2112
Song C, Luo L, Lei Z, Li B, Liang Z, Liu G, Li D, Zhang G, Huang B, Feng ZH (2008) IL-17-producing alveolar macrophages mediate allergic lung inflammation related to asthma. J Immunol 181:6117–6124
Takahashi N, Vanlaere I, de Rycke R, Cauwels A, Joosten LA, Lubberts E, van den Berg WB, Libert C (2008) IL-17 produced by Paneth cells drives TNF-induced shock. J Exp Med 205:1755–1761
Ishigame H, Kakuta S, Nagai T, Kadoki M, Nambu A, Komiyama Y, Fujikado N, Tanahashi Y, Akitsu A, Kotaki H et al (2009) Differential roles of interleukin-17A and -17F in host defense against mucoepithelial bacterial infection and allergic responses. Immunity 30:108–119
Yoshiga Y, Goto D, Segawa S, Ohnishi Y, Matsumoto I, Ito S, Tsutsumi A, Taniguchi M, Sumida T (2008) Invariant NKT cells produce IL-17 through IL-23-dependent and –independent pathways with potential modulation of Th17 response in collagen-induced arthritis. Int J Mol Med 22:369–374
Gu Y, Yang J, Ouyang X, Liu W, Li H, Yang J, Bromberg J, Chen SH, Mayer L, Unkeless JC, Xiong H (2008) Interleukin 10 suppresses Th17 cytokines secreted by macrophages and T cells. Eur J Immunol 38:1807–1813
Jandus C, Bioley G, Rivals JP, Dudler J, Speiser D, Romero P (2008) Increased numbers of circulating polyfunctional Th17 memory cells in patients with seronegative spondylarthritides. Arthritis Rheum 58:2307–2317
Cosmi L, De Palma R, Santarlasci V, Maggi L, Capone M, Frosali F, Rodolico G, Querci V, Abbate G, Angeli R et al (2008) Human interleukin 17-producing cells originate from a CD161+ CD4+ T cell precursor. J Exp Med 205:1903–1916
Fujino S, Andoh A, Bamba S, Ogawa A, Hata K, Araki Y, Bamba T, Fujiyama Y (2003) Increased expression of interleukin 17 in inflammatory bowel disease. Gut 52:65–70
Tzartos JS, Friese MA, Craner M, Palace J, Newcombe J, Esiri MM, Fugger L (2007) Interleukin-17 production in CNS-infiltrating T-cells and glial cells is associated with active disease in multiple sclerosis. Am J Pathol 172:146–155
Hueber AJ, Asquith DL, Miller AM, Reilly J, Kerr S, Leipe J, Melendez AJ, McInnes IB (2010) Mast cells express IL-17A in rheumatoid arthritis synovium. J Immunol 184:3336–3340
Lin AM, Rubin CJ, Khandpur R, Wang JY, Riblett M, Yalavarthi S, Villanueva EC, Shah P, Kaplan MJ, Bruce AT (2011) Mast cells and neutrophils release IL-17 through extracellular trap formation in psoriasis. J Immunol 187:490–500
Zhou L, Peng S, Duan J, Zhou J, Wang L, Wang J (1998) A human B cell line AF10 expressing HIL-17. Biochem Mol Biol Int 45:1113–1119
Iwakura Y, Ishigame H, Saijo S, Nakae S (2011) Functional specialization of interleukin-17 family members. Immunity 34:149–162
Pöllinger B, Junt T, Metzler B, Walker UA, Tyndall A, Allard C, Bay S, Keller R, Raulf F, Di Padova F et al (2011) Th17 cells, not IL-17+ γδ T cells, drive arthritic bone destruction in mice and humans. J Immunol 186:2602–2612
Ziolkowska M, Koc A, Luszczykiewicz G, Ksiezopolska-Pietrzak K, Klimczak E, Chwalinska-Sadowska H, Maslinski W (2000) High levels of IL-17 in rheumatoid arthritis patients: IL-15 triggers in vitro IL-17 production via cyclosporin A-sensitive mechanism. J Immunol 164:2832–2838
Pène J, Chevalier S, Preisser L, Vénéreau E, Guilleux MH, Ghannam S, Molès JP, Danger Y, Ravon E, Lesaux S et al (2008) Chronically inflamed human tissues are infiltrated by highly differentiated Th17 lymphocytes. J Immunol 180:7423–7430
Molet S, Hamid Q, Davoine F, Nutku E, Taha R, Page N, Olivenstein R, Elias J, Chakir J (2001) IL-17 is increased in asthmatic airways and induces human bronchial fibroblasts to produce cytokines. J Allergy Clin Immunol 108:430–438
Hueber W, Patel DD, Dryja T, Wright AM, Koroleva I, Bruin G, Antoni C, Draelos Z, Gold MH, Durez P et al (2010) Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci Transl Med 2:52ra72
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Basel
About this chapter
Cite this chapter
Di Padova, F., Ryffel, B., Quesniaux, V. (2013). IL-17A Family, Receptors, Proinflammatory Effects, and Production. In: Quesniaux, V., Ryffel, B., Padova, F. (eds) IL-17, IL-22 and Their Producing Cells: Role in Inflammation and Autoimmunity. Progress in Inflammation Research. Springer, Basel. https://doi.org/10.1007/978-3-0348-0522-3_1
Download citation
DOI: https://doi.org/10.1007/978-3-0348-0522-3_1
Published:
Publisher Name: Springer, Basel
Print ISBN: 978-3-0348-0521-6
Online ISBN: 978-3-0348-0522-3
eBook Packages: MedicineMedicine (R0)