Abstract
Inherited cerebellar malformations cause lifelong disability and are not well studied in the newborns because there is a lack of appropriate clinical examination tools. Recently, inherited cerebellar malformations have been investigated using emerging advanced neuroimaging technologies such as magnetic resonance imaging (MRI), which has revealed many developmental disorders of the cerebellum. These malformations cause impairments that affect motor and nonmotor functions. Cerebellar hypoplasia (CH), cerebellar dysplasia (CD), Dandy–Walker malformation (DWM), Joubert syndrome and related disorders (JSRDs), pontocerebellar hypoplasia (PCH), rhombencephalosynapsis (RES), lissencephaly with cerebellar hypoplasia (LCH), and Lhermitte–Duclos disease (LDD) are examples of cerebellar malformations which this chapter will focus on using characteristic symptoms and signs. The current approaches for evaluation of the affected patients, differential diagnosis, and management of the malformations will be discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cerebellar hypoplasia
- Cerebellar dysplasia
- Dandy–Walker malformation
- Joubert syndrome
- Pontocerebellar hypoplasia
- Rhombencephalosynapsis
- Lhermitte–Duclos disease
Introduction
The cerebellar development and structure have been discussed in chapter “The Embryology and Anatomy of the Cerebellum”. The cerebellar development begins during an early embryonic stage with a complicated developmental process that continues well into the first year after birth in human. Recent advances in neonatal intensive care and breakthroughs in neuroimaging techniques such as positron emission tomography (PET), structural MRI (sMRI), and functional MRI (fMRI) have improved our ability to understand the structural and functional anomalies that implicate cerebellar involvement in numerous motor and nonmotor functions, ranging from motor/sensory integration and working memory to various higher order cognitive processes [1,2,3,4]. Despite the advanced technologies, understanding cerebellar malformations in children requires additional research regarding their prognosis as well as their lifelong consequences. Because of a lack of an appropriate treatment, up to 80% of parents choose to terminate pregnancy after a prenatal diagnosis of a cerebellar malformation [1, 5]. The prolonged developmental process in the cerebellum makes it more vulnerable to perturbation caused by genetic and environmental factors, or a combination of both that occur during development. Cerebellar abnormalities range from subtle impairments including cognitive impairments to significant structural defects with life-threatening or lifelong disabilities [6].
Cerebellar dysfunction that disturbs the regulation of muscle tone, motor control, and coordination of movement is called ataxia – a broad term that refers to a disturbance in the smooth performance of the motor activities. The nonmotor dysfunction that results from cerebellar manifestations includes cognitive affective syndrome that includes impairment in executive function, spatial cognition, personality changes, and language deficits [7,8,9]. Cerebellar structural and functional abnormalities have been reported in psychiatric disorders such as schizophrenia, bipolar disorder, depression, anxiety disorders, attention deficit hyperactivity disorder (ADHD), and autism [10,11,12,13,14,15].
The specific constellation of symptoms is sometimes useful for localizing the cerebellar lesion, but often there is considerable overlap. Because of a complex developmental process during the formation of cerebellum, clinical classification of cerebellar neurodevelopmental disorder is difficult; however, there are classification that are based on embryological and genetic considerations [16, 17]. Before the introduction of MRI, Dandy–Walker variants was a term used to characterize several types of cerebellar malformations. Now, cerebellar malformations can be classified into primary (malformation) and secondary (disruptive) lesions [17].
Secondary disruptive cerebellar defects are secondary to a developmental disorder in structures around the cerebellum such as Chiari malformation and vein of Galen malformation. Chiari malformations (Fig. 1) are posterior cranial fossa defects that range from herniation of the cerebellar tonsils through the foramen magnum to complete agenesis of the cerebellum, which are classified into four types (I–IV), with type IV being the most severe malformations [18]. Vein of Galen malformation is another secondary cerebellar malformation that results from the presence of one or more arteriovenous fistulas, which constitute up to 30% of intracranial vascular malformations presenting among pediatric patients [19, 20]. In patients with vein of Galen malformation, the superior cerebellar arteries also discharge into the vein of Galen [21]. It is reasonable to assume that the dilated vein causes direct compression of cerebrospinal fluid (CSF) flow, increased intracranial pressure, and caudal displacement of the cerebellar tonsils [22], leading to cerebellar signs and symptoms.
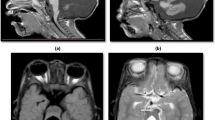
Chiari malformation type I. (a, b) Sagittal and coronal T2-weighted brain MR images. There is slight inferior herniation of cerebellar tonsils through the foramen magnum that is less than 5 mm and shows benign tonsillar ectopia that could be a mild variant of Chiari malformation. (c, d) Axial and sagittal T2-weighted MR images of the brainstem and cervical spinal cord. Note the presence of a large syrinx in association with tonsillar ectopia in Chiari malformation type I
Primary cerebellar malformations are classified into two broad categories: (1) those with hypoplasia and (2) those with dysplasia. Both hypoplasia and dysplasia categories have their own subgroups, which are categorized in Diagram 1 [17]. This chapter aims to discuss primary cerebellar malformations and the current treatment approaches in affected patients. The included primary cerebellar malformations are the cerebellar hypoplasia (CH), cerebellar dysplasia (CD), Dandy–Walker malformation (DWM), pontocerebellar hypoplasia (PCH), Joubert syndrome and related disorders (JSRDs), rhombencephalosynapsis (RES), lissencephaly with cerebellar hypoplasia (LCH), and dysplastic cerebellar gangliocytoma or Lhermitte–Duclos disease (LDD).
Cerebellar Hypoplasia
Cerebellar hypoplasia (CH) is a heterogeneous group of disorders that was first reported by Crouzon in 1929. From the embryological aspect, the cerebellar primordium emerges at approximately 28 days after fertilization in human (embryonic days 7–8 in the mouse) as a neuroepithelial swelling of the rostral lip of the fourth ventricle, which is part of the alar plate of the metencephalon (rhombomere-1) [6, 23,24,25]. Therefore, any developmental dysregulation that targets the rhombomere-1 causes failure to specify the anterior hindbrain and results in cerebellar aplasia/hypoplasia because of defects in dorsal patterning mechanisms [26,27,28].
Distinguishing CH from cerebellar atrophy is very important because the treatment approaches are different. CH refers to a cerebellum with reduced volume, but with normal shape, which is stable over time and normal interfoliate fissures and sulci. On the other hand, cerebellar atrophy represents a progressive loss of cerebellar parenchyma along with secondary enlargement of both interfolial spaces and the fourth ventricle (29).
The causes of the CH are broad and include chromosomal aberrations (such as trisomy 9, 13, and 18), metabolic disorders [29], teratogens (drugs and infections: see chapter “Hormonal Regulation of Cerebellar Development and Its Disorders”), or isolated genetic CH (such as very low density lipoprotein receptor (VLDLR) – a reelin receptor [30, 31], dyskerin pseudouridine synthase 1 (DKC1) [32], oligophrenin 1 (OPHN1) [33], pancreas-specific transcription factor 1a (PTF1A) [34], and carbohydrate-deficient glycoprotein syndrome Types I and II (CDG 1 and 2) [35, 36]). Mutations in CHD7 (chromodomain helicase DNA binding protein 7) and retinoic acid exposure during early pregnancy are associated with variable cerebellar CH [31]. Similar to most developmental anomalies, CH may be associated with other brain malformations and there may be multi-organ involvement. Based on recent studies, CH is classified into three categories: (1) focal hypoplasia which has three subgroups – (i) isolated vermis hypoplasia, which is mostly characterized by partial absence of the inferior portion of the vermis, (ii) predominantly vermis hypoplasia including DWM [37], and (iii) unilateral hemispheric hypoplasia; (2) global (diffuse or generalized) hypoplasia (involvement of both vermis and hemisphere), which is more common in congenital cytomegalovirus (CMV) infection; and (3) CH with brainstem involvement including PCH [17, 38,39,40].
Clinically, in cerebellar hypoplasia, ataxia and poor motor learning are the most common and nonprogressive presentations compared with atrophic cerebellar disorders [41]. In infancy, hypotonia and global developmental delay are present earlier, and other signs include ocular motor disorders, dysarthria, intention tremor, and microcephaly. Behavioral abnormalities, intellectual disability, and speech and language disorders can vary from mild to severe impairment [42].
Management
It is important to consider that ataxia or other neurological signs in cerebellar hypoplasic patients usually do not worsen over time compared with atrophic cerebellar disorder. There is no standard course of treatment; therefore, the principal treatment is supportive including physical therapy, occupational therapy, speech therapy, psychiatric/behavioral medications, and special education (see chapters “Clinical Features, Assessment, and Management of Patients with Developmental and Other Cerebellar Disorders” and “Rehabilitation in Cerebellar Ataxia”).
Cerebellar Dysplasia
Cerebellar dysplasia (CD) is defined by abnormal pattern in foliation, abnormal white matter arborization, heterotopic nodules of gray matter, and abnormal gray-white matter junction. CD may be associated with cysts resulting from disorganized cortical structures and pia matter disruption in which subarachnoid space is abnormally engulfed by dysplastic cerebellar folia [43]. CD can be subdivided into: (1) global CD, which has been reported in some posterior fossa malformations such as Chudley-McCullough syndrome, α-dystroglycanopathies, GPR56-related polymicrogyria, and Poretti-Boltshauser syndrome; and (2) focal CD including dysplasia of the superior cerebellar vermis in Joubert syndrome and increased volume of multiple cerebellar folia in Lhermitte-Duclos disease [43]. In addition, it has been reported a rare number of isolated unilateral cerebellar hemispheric dysplasia [44, 45].
It has been reported that CD with abnormal folia orientation may be a core finding in PROS (PIK3CA-related overgrowth spectrum) patients that show somatic mutations in PIK3CA pathway genes, especially in those presenting MCAP (megalencephaly-capillary malformation) condition. Brain MRI of PROS patients, who are presented in clinics with vascular anomalies (mainly capillary malformations), segmental overgrowth dysregulation, and distal limb anomalies such as syndactyly and polydactyly, should be focused on cerebellum to detect any cerebellar dysplasia, which could be followed by proper genetic testing [46].
Management
Like CH, treatment of CD is symptomatic and supportive.
Dandy–Walker Malformation
The fundamental structure that is affected in Dandy–Walker malformation (DWM) is the cerebellum [47,48,49]. DWM is a genetic disorder, with the most common and severe type being the Dandy–Walker syndrome malformation [47]. Deletion of Zinc finger 1 and 4 (ZIC1, ZIC4) genes on chromosome 3q24 [37, 50] and the Forkhead Box 1 (FOXC1) gene on chromosome 6p25 are candidates involving in DWM [37, 51]. It is suggested that ZIC1 and ZIC4 are required for the full responsiveness of granule cell precursors (GCPs) to sonic hedgehog (SHH) [28]. It seems that FOXC1 directly regulates the size of posterior fossa and FOXC1-dependent SDF1α-CXCR4 (stromal cell derived factor 1α–CXC motif chemokine receptor 4) signaling from the surrounding mesenchyme to the developing cerebellar anlage regulates a plethora of cerebellar developmental programs [37]. Some other congenital abnormalities, especially eye malformations consistent with Axenfeld-Rieger syndrome, are seen in FOXC1-related DWM, and sometimes overlap with 3C (cranio-cerebello-cardiac) syndrome in severely affected patients [31]. Recently discovered mutations in CCDC22 (coiled-coil domain containing 22) gene in X-linked cases of 3C syndrome indicate that the CCDC22 mutations may be another cause of DWM [31]. Deletion of FOXC1 can lead to vermian tail (a common extended and dysplastic posterior vermis with an indistinct choroid plexus) in infants of DWM [52]. Autosomal dominant mutations in LAMC1 (laminin subunit gamma 1) and NID1 (nidogen 1) can also be considered as another causes of DWM [31].
DWM is characterized by agenesis or hypoplasia of the cerebellar vermis, upwardly rotated vermis, cystic dilatation of the fourth ventricle into the posterior cranial fossa, and an enlarged posterior cranial fossa [1, 37, 38, 53]. Enlargement of the posterior cranial fossa causes an abnormally high tentorium above the internal occipital protuberance and transverse occipital sulcus (location of transverse sinus), as well as a variable degree of hydrocephalus [1, 54]. During cerebellar development, the right and left cerebellar primordia are fused at the midline. Any misregulation in this developmental process leads to a lack of cerebellar fusion at the midline. The lack of midline fusion causes the extension of membranous area/roof plate anteriorly, resulting in a large fourth ventricle. Cerebrospinal fluid pulsations cause roof plate expansion posteriorly within the posterior fossa, forming a large posterior cyst that represents the fourth ventricle [55].
Clinically, DWM can be defined via the characteristic triad consisting of the following: (1) complete or partial agenesis of the vermis, (2) cystic dilatation of the fourth ventricle, and (3) an enlarged posterior cranial fossa with upward displacement of the transverse sinuses [56, 57]. If hydrocephalus is present, it suggests a common developmental disorder in which multiple brain regions are affected [58].
The signs and symptoms associated with DWM are broad. DWM patients often have global developmental delay (GDD), language delay, intellectual disability (ID), hypotonia, motor delay, ataxia, lack of coordination, jerky movements of the eyes, and progressive enlargement of the skull. Some patients may have normal cognition, whereas others have mild to severe mental retardation, even when hydrocephalus is effectively treated. The enlarged head circumference, which may bulge at the back of the skull, can increase pressure on the brainstem and nerves and can cause difficulties in controlling face and neck, and abnormal breathing patterns. Sagittal and axial MR images (Fig. 2) can distinguish DWM from other cerebellar malformations. In DWM, it is important to consider mega cisterna magna, retro-cerebellar cysts, and Blake’s pouch cyst [55, 59]. It should be noted that in addition to the absence of the middle part of the cerebellum, midline structures in the forebrain such as the corpus callosum may be absent, a condition known as agenesis of corpus callosum (ACC). Systemic malformations associated with DWM may include cardiac anomalies, urogenital anomalies, and other abnormalities may occur collectively in about half of the patients [60,61,62,63].
Dandy–Walker malformation. (a) Sagittal T2-weighted brain MR image showing hypoplasia of the inferior vermis. A connection between the cisterna magna and the fourth ventricle is seen. (b) Axial T1-weighted brain MR image showing a cerebellum with isolated inferior vermian hypoplasia and normal cerebellar hemispheres, which is referred to as part of the Dandy–Walker variant
Management
If there is hydrocephalous, treatment could include shunting and CSF drainage from the lateral ventricles and/or posterior fossa cyst, which is currently considered the ordinary surgical treatment of choice [49, 64]. Another way of shunting is endoscopic method, including (1) endoscopic third ventriculostomy (ETV), (2) aqueduct stent with shunt insertion, and (3) trans-tentorial proximal catheter insertion with endoscopic shunting [64]. The treatment consists of physiotherapy, occupational therapy, speech therapy, and specialized education. Although diagnosis of DWS during intrauterine development is difficult, if an ultrasound suggests DWS, then amniocentesis should be performed to aid in the diagnosis [65]. It is important that the families of affected children be referred for genetic counseling.
Joubert Syndrome and Related Disorders
Joubert syndrome (JS) was first identified by Marie Joubert in Montreal, Canada [66]. JS is a group of autosomal recessive conditions that are characterized by developmental anomalies, which are caused by defects in the structure or function of the primary cilium [67, 68]. Molar tooth sign (MTS) observed on axial images of plain MRI (Fig. 3) is one of the gold standards of JS, which is formed by cerebellar vermis hypoplasia and dysplasia (most likely with a cleft in the superior vermis) accompanied by long, thick, elevated, and horizontally oriented superior cerebellar peduncles, with a deep interpeduncular fossa at the level of a thin midbrain–hindbrain junction (isthmus) [17, 37, 69]. In addition, diffusion tensor imaging (DTI), an MRI technique for white matter tractography, can further demonstrate laterally displaced and dysmorphic cerebellar nuclei, hypoplastic medial lemnisci, absent transverse fibers in vermis, and deficient superior cerebellar peduncle decussation [31]. When other organs, such as the retina, kidney, and liver are involved, it is called Joubert syndrome and related disorders (JSRDs), and these patients also have the MTS [67].
Joubert syndrome and related disorders. (a) Coronal T2 FLAIR brain image. The cerebellar vermis is aplastic and superior cerebellar peduncles are elongated. (b) Axial T2 FLAIR brain image. This image shows a deep interpeduncular fossa, elongated superior cerebellar peduncles with cerebellar vermis hypoplasia, which are characteristic of the molar tooth sign in Joubert syndrome
JSRDs is the most common inherited congenital cerebellar malformation in which ciliopathy is a fundamental mechanism. The primary cilia are important in neuronal development and function as cellular antenna that are found in nearly all cell types. Two main conditions related to defective function of primary cilia are retinal dystrophy and nephronophthisis that are found in many patients with JS [70]. The function of cilia in cells includes protein trafficking, photoreception, embryonic axis patterning, and cell cycle regulation. Therefore, dysfunction of this microtubule-based extension of cellular membranes can affect a single tissue or manifest as having multi-organ involvement, which is called ciliopathy [71]. Within the developing cerebellum, primary cilia have been shown to be essential for reception of the cell signaling ligand sonic hedgehog, which in turn is essential for proliferation of cerebellar neurons such as granule cells [72, 73].
The causative gene of many ciliopathies in individuals with JSRDs has defined a new class of neurological diseases [71]. To date, over 16 causative genes have been associated with JSRDs and all encode proteins in the primary cilium or its apparatus [67]. For example, mutations in genes such as AHII, INPP5E, CC2D2A, and ARL13B cause JS with MTS and retinal blindness [74]. However, mutations in TMEM216 and RPGRIPIL genes lead to MTS and renal involvement. In more severe cases, mutations in the CEP290 gene causes MTS together with retinal and renal involvement and complete situs inversus [70], while mutations in TMEM67 are the most common cause of MTS with liver involvement [75].
Clinically, JSRDs patients have developmental delay, motor disability, hypotonia, ataxia, dysregulated breathing rhythms such as apnea and tachypnea (that results from dysfunction of the respiratory centers in the brainstem or cerebellum [69, 76]), abnormal eye and tongue movements, and subsequent mental retardation [70]. As ciliopathy interrupts a broad range of developmental process, a defect could be seen in other organs such as kidney, retina, and liver, and there were also facial abnormalities (cleft lip or palate, tongue abnormalities) and polydactyly (extra fingers and toes) [77,78,79]. In mild JSRDs, ataxic movement lessens with age and the ability to walk is delayed to age 4–5 years.
Management
The treatment is symptomatic and supportive such as physical therapy, occupational therapy, and speech therapy. Infants with abnormal breathing patterns should be monitored closely for apnea, and this may be required during the first year of life because some neonates have died as a result of apnea. In this case, caffeine may be helpful to promote respiratory drive. These patients should be periodically examined for any non-neurological signs and symptoms. Because of the heterogeneity of these conditions, genetic testing will show specific gene mutations, which can help predict the range of organ involvement such as retina, kidney, and liver [6, 80].
Pontocerebellar Hypoplasia
Pontocerebellar hypoplasia (PCH) is a group of autosomal recessive neurodevelopmental and neurodegenerative disorders with hypoplasia of the cerebellum and ventral pons, followed by atrophy. It is also characterized by variable cerebral involvement such as microcephaly, seizures, and a severe delay in cognitive and motor development, which in many cases is fatal early in life [17, 38, 81, 82].
Ten different subtypes have been reported based on clinical and genetic features (i.e., PCH1–10) [83], and they are summarized in Table 1. Mutations in the following genes cause PCH because of molecular malfunctions that are important for normal development of the neurons and non-neuronal cells. Mutations in the vaccinia-related kinase 1 (VRK1) gene on chromosome 14q32.2 cause PCH1A (or spinal muscular atrophy with pontocerebellar hypoplasia; SMA-PCH), in which there is spinal cord anterior horn cell degeneration [84, 85]. Mutations in the EXOSC3 (exosome component 3) gene on chromosome 9p13.2 lead to PCH1B [86]. Mutations in three genes, TSEN54, TSEN34, and TSEN2, encoding three of four subunits of the tRNA splicing endonuclease (TSEN) complex have been found to underlie PCH2, PCH4, and PCH5 [81]. PCH2 is characterized by CH in which the hemispheres are more severely affected than the vermis, and in contrast to PCH1, there is no anterior horn cell degeneration in the spinal cord. These patients have other signs and symptoms such as progressive cerebral atrophy, microcephaly, dyskinesia, seizures [81, 82], early hyperreflexia, developmental delay, and feeding problems [31]. In brief, it is known that mutations in TSEN54 on chromosome 17q25.1 cause PCH2A; mutations in TSEN2 on chromosome 3p25.2 cause PCH2B; mutations in TSEN34 on chromosome 19q13.42 cause PCH2C; mutations in SEPSECS (O-phosphoseryl-tRNA:selenocysteinyl-tRNA synthase) on chromosome 4p15.2 cause PCH2D (as known as progressive cerebello-cerebral atrophy; PCCA); mutations in the gene VPS53, a subunit of the Golgi-associated retrograde protein (GARP) complexes, on chromosome 17p13.3 cause PCH2E; and mutations in TSEN15 on chromosome 1q25 cause PCH2F [87,88,89]. PCH3 that seems to be associated with optic atrophy is caused by mutations in the gene encoding PCLO (piccolo presynaptic cytomatrix protein) on chromosome 7q21 [90, 91]. PCH4 is caused by a mutation in the TSEN54 gene on chromosome 17q25.1 [87]. PCH4 is associated with polyhydramnios, contractures, severe hyperreflexia, and early death because of central respiratory failure [31]. A mutation in the TSEN54 gene on chromosome 17q25 causes PCH5, and mutations in the RARS2 (mitochondrial arginyl-tRNA synthetase 2) encoding gene on chromosome 6q15 cause PCH6, which is associated with elevated CSF lactate level [92]. The gene involved in PCH7 is unknown [93, 94]. PCH8 is caused by recessive loss-of-function mutations in the CHMP1A (charged multivesicular body protein 1A) encoding gene on chromosome 16q24 [95]. Mutations in the AMPD2 (adenosine monophosphate deaminase 2) gene on chromosome 1p13 cause PCH9, which is associated with severely delayed psychomotor involvement, progressive microcephaly, spasticity, and seizures [31, 96], and mutations in CLP1 (cleavage factor polyribonucleotide kinase subunit 1) gene on chromosome 11p12 cause PCH10, which is associated with progressive neurodegenerative features or static encephalopathy [31, 97]. Finally, loss-of-function mutations in SLC25A46 (solute carrier family 25 member 46) cause lethal congenital PCH [98].
Disorders presenting with PCH are constantly growing. Some examples are calcium/calmodulin-dependent serine protein kinase (CASK)-related PCH (associated with progressive microcephaly, hypoplasia of pons and cerebellum, intellectual disability, and epilepsy in female), congenital disorders of glycosylation (CDG; associated with GDD, language delay, eye anomalies, coagulation defects, neuropathy, impaired liver function, abnormal fat distribution, and cerebellar and pons atrophy imitating PCH), cerebellofaciodental syndrome (BRF1-related PCH; associated with microcephaly, short stature, intellectual disability, cerebellar and brainstem hypoplasia, and dystrophic features including taurodontism), and osteogenesis imperfecta (WNT1-related PCH; associated with developmental defects of the midbrain, pons, and cerebellum including variable degree of cerebellar and brainstem hypoplasia) [17, 38].
Clinically, PCH patients have hypotonia and difficulty with coordination of sucking and swallowing, and problems with handling their oral and respiratory secretions [99]. There are no criteria to distinguish precisely between the different subtypes based on clinical signs and symptoms, and therefore genetic testing is important. The cerebellum and pontine hypoplasia can be revealed by MRI in which the cerebellar hemispheres may be more severely affected than the midline vermis. Flattened cerebellar hemispheres (the “wings”) and a slightly preserved vermis (the “body”) together create dragonfly appearance on coronal MRI of PCH patients [17] (Fig. 4).
Pontocerebellar hypoplasia. (a) Sagittal T2-weighted brain MR images. The pons is very small but has a relative sparring bulging in its superior part. Vermis hypoplasia predominates at the inferior site. (b) Coronal T2-weighted brain MR images. Cerebellar hemispheric hypoplasia with vermis relatively spared leading to classic dragonfly image
Management
There is no known cure for PCH, and treatment is symptomatic and palliative and requires the teamwork of health-care professionals. Patients with PCH need a gastrostomy tube and airway control, and they may not survive beyond 1 year of age. It is important to refer families of affected children for genetic counseling.
Rhombencephalosynapsis
Rhombencephalosynapsis (RES) is a neurodevelopmental malformation that is characterized by midline fusion of the two cerebellar hemispheres, which is caused by failure of the midline structure development in the rhombencephalon. It is suggested that disruption of dorsoventral patterning of the rhombencephalon may cause RES [100, 101]. RES is a rare condition with unknown etiology, and the most specific and key MRI finding is agenesis or hypogenesis of the vermis, in which the cerebellar vermis is completely or partially absent with a fused cerebellar hemisphere, a fused superior cerebellar peduncle, and midline dentate nucleus, creating a horseshoe-shaped arch across the midline [17, 37, 100]. Coronal T2-weighted MR images show horizontal folia pattern and mid-sagittal T1-weighted MR images show the dentate nucleus [17].
RES may be associated with other cerebellar abnormalities, such as Purkinje cell heterotopias [102]. Although RES is seen most frequently in isolated form, it can also be seen together with other developmental malformations in the nervous system or other organs. RES is a highly consistent finding in Gomez–Lopez–Hernandez syndrome (GLHS), which is also known as cerebellotrigeminal-dermal dysplasia (a neurocutaneous disorder) presenting with parietal/temporal alopecia (focal dermal dysplasia or lack of hair), trigeminal anesthesia (loss of sensation in the face), mid-face hypoplasia with towering skull shape, corneal opacities, mental retardation, and short stature. RES is also associated with midline brain structural defects including absent olfactory bulbs, dysgenesis of the corpus callosum, absent septum pellucidum, and in rare patients, atypical forms of holoprosencephaly [100]. RES has also been reported in VACTERL (vertebral anomalies, anal atresia, cardiovascular anomalies, trachea–esophageal fistula, renal anomalies, limb defects) association and hydrocephalus [42, 102,103,104,105].
Ishak et al. (2012) proposed four groups based on the severity of cerebellar vermis defect: (1) mild, in which the nodulus, anterior, and posterior vermis are partially absent; (2) moderate, where there is a lack of posterior vermis with some anterior vermis but the nodulus is present; (3) severe, which is a lack of posterior and anterior vermis with the nodulus partially absent; and (4) complete, where there is a lack of the entire vermis [100]. They also divided RES-affected patients into four clinical categories using the following criteria: (1) RES in patients with GLHS; (2) RES plus at least one of the VACTERL association features without scalp alopecia; (3) RES plus a focal or diffuse forebrain midline fusion defect without alopecia; and (4) RES in patients with malformations that do not fit into the categories (1)–(3) (with abnormal head shape, midface hypoplasia, low-set and/or posteriorly rotated ears, telecanthus and/or hypertelorism). Based on other literatures, RES includes some specific clinical phenotypes such as biparietal alopecia, craniosynostosis, trigeminal anesthesia, and atresia of the fourth ventricle [106].
Clinically, signs and symptoms in patients with the isolated form of RES are variable such as developmental delay, in which motor learning and skills develop between 3 and 6 years of age, hypotonia, ataxia, abnormal eye movements, and head stereotypies [17, 107].
Management
Treatment for RES infants is generally supportive and includes physical therapy and occupational therapy. If hydrocephalus is present in patients with RES and it is symptomatic, this can be an indication for surgical intervention with a ventriculostomy or ventricular shunt. It is important to refer families of affected children for genetic counseling.
Lissencephaly with Cerebellar Hypoplasia
Lissencephaly with CH is a neurodevelopmental malformation in which cellular migration is severely impaired. The cerebellum in patients with lissencephaly is underdeveloped with prominent vermis hypoplasia or aplasia [108,109,110,111]. Mutations in the gene encoding reelin (RELN), which is mapped on chromosome 7q22, cause lissencephaly with severe abnormalities of the cerebellum, hippocampus, and brainstem. Reelin is a large extracellular matrix-associated protein [112] that is involved in migration of neurons through binding to its receptors (VLDLR), the apolipoprotein E receptor 2 (ApoER2) [113,114,115], and also α3β1 integrin and protocadherins [116]. In a mouse model of lissencephaly, mutations in RELN and DAB1 prominently cause neuronal migration defects in the brain with accompanying cerebellar hypoplasia, and there is also abnormal circuitry development [117, 118]. Mutations in RELN also show abnormal developmental disorders outside the brain such as neuromuscular connectivity and congenital lymphedema [110]. It is also reported that mutations in α-dystroglycan may result in lissencephaly and central nervous system developmental malformations [119].
Clinically, the important approach to diagnose is MRI of the cerebellum, which shows severe vermis and CH and cerebellar peduncle malformation.
Management
Treatment of patients who have lissencephaly with CH are supportive care and symptom management. In case of difficulties with feeding, a gastrostomy tube may be considered. If seizures are present, anti-seizure medications are administered, and in the case of hydrocephalus, shunting is performed. It is important to refer families of affected children for genetic counseling.
Dysplastic Cerebellar Gangliocytoma or Lhermitte–Duclos Disease
The first case of the Lhermitte–Duclos disease (LDD) was reported by Lhermitte and Duclos in 1920 as a cerebellar ganglion cell tumor or dysplastic cerebellar gangliocytoma [120, 121]. LDD is a rare developmental disorder of the cerebellum and features both malformation and benign neoplasm. Most patients with LDD appear to have mutations in the phosphatase and tensin homologue (PTEN) gene [121,122,123]. Most frequently, LDD occurs in young adults in the third and fourth decades of life [124, 125]. Because LDD presents in previously healthy children with features of a unilateral cerebellar mass, the main considerations are the posterior fossa tumor and secondary hydrocephalus. LDD is not diagnosed as medulloblastoma in most patients because of differences in the age group, medical history, and unique imaging features. Neuroimaging with MRI is sufficient and important in the diagnostic process. Long-standing unilateral space-occupying skull lesions in the posterior fossa leads to thinning of the skull in the occipital region [126, 127]. Histopathological findings show dysplastic gangliocytoma of the cerebellum in front of a hamartoma lesion with widening of the molecular layer occupied by abnormal ganglion cells, absence of the Purkinje cell layer, and hypertrophy of the granular layer [128].
Clinically, patients with LDD present with headache, nausea, cerebellar signs, hydrocephalus, and increased intracranial pressure. Patients may have symptoms for many years, such as cranial nerve palsies and cerebellar symptoms, because of the slowly progressive nature of this disease [126]. LDD patients may show mental retardation. LLD is commonly associated with other disorders of cortical formation, megalencephaly, gray matter heterotopia, polymicrogyria, polydactyly, macroglossia, localized gigantism, and other congenital malformations such as familial hamartoma–neoplasia syndrome and Cowden’s disease (CD), an inherited cancer/hamartoma syndrome involving the breast, thyroid gland, and other organs [17, 129]. Widened cerebellar folia with striated appearance can be seen on T2-weighted MRI [17]. Elevated lactate, slightly reduced N-acetyl aspartate (NAA), reduced myoinositol, reduced choline, and reduced choline/creatine ratio are common on MR spectroscopy (MRS) [17].
Management
Decompressive surgery for symptomatic patients is the accepted choice of treatment. The risk of performing surgery is the lack of clear tumor margins. Symptomatic and supportive treatments such as physical therapy and occupational therapy should be offered.
Summary
In this chapter, cerebellar malformations and current treatment approaches were summarized. Based on available knowledge and our clinical experience, there is no curative treatment and most of the patients are managed using conservative approaches (see chapters “Clinical Features, Assessment, and Management of Patients with Developmental and Other Cerebellar Disorders” and “Rehabilitation in Cerebellar Ataxia”). Treatment is in response to symptoms and requires a team of specialists (neonatologists, pediatricians, neurologists, and therapists), health-care professionals, and genetic counselors.
References
Bolduc ME, Limperopoulos C. Neurodevelopmental outcomes in children with cerebellar malformations: a systematic review. Dev Med Child Neurol. 2009;51(4):256–67.
Allen G, Buxton RB, Wong EC, Courchesne E. Attentional activation of the cerebellum independent of motor involvement. Science. 1997;275(5308):1940–3.
Middleton FA, Strick PL. Anatomical evidence for cerebellar and basal ganglia involvement in higher cognitive function. Science. 1994;266(5184):458–61.
Leiner HC, Leiner AL, Dow RS. The human cerebro-cerebellar system: its computing, cognitive, and language skills. Behav Brain Res. 1991;44(2):113–28.
Hutchinson S, Lee LH, Gaab N, Schlaug G. Cerebellar volume of musicians. Cereb Cortex. 2003;13(9):943–9.
Haldipur P, Millen KJ, Aldinger KA. Human cerebellar development and transcriptomics: implications for neurodevelopmental disorders. Annu Rev Neurosci. 2022;
Bhatia MS, Saha R, Gautam P. Cerebellar Cognitive Affective Syndrome: a case report. The primary care companion for CNS disorders. 2016;18(2).
Chang C, Siao SW. Cerebellar cognitive affective syndrome: attention deficit-hyperactivity disorder episode of adolescent with cerebellar atrophy in a psychiatric ward. Kaohsiung J Med Sci. 2016;32(1):52–4.
Marien P, Wackenier P, De Surgeloose D, De Deyn PP, Verhoeven J. Developmental coordination disorder: disruption of the cerebello-cerebral network evidenced by SPECT. Cerebellum. 2010;9(3):405–10.
Marko MK, Crocetti D, Hulst T, Donchin O, Shadmehr R, Mostofsky SH. Behavioural and neural basis of anomalous motor learning in children with autism. Brain. 2015;138(Pt 3):784–97.
Salman MS, Tsai P. The role of the pediatric cerebellum in motor functions, cognition, and behavior: a clinical perspective. Neuroimaging Clin N Am. 2016;26(3):317–29.
Mothersill O, Knee-Zaska C, Donohoe G. Emotion and theory of mind in schizophrenia-investigating the role of the cerebellum. Cerebellum. 2016;15(3):357–68.
Minichino A, Bersani FS, Trabucchi G, Albano G, Primavera M, Delle Chiaie R, et al. The role of cerebellum in unipolar and bipolar depression: a review of the main neurobiological findings. Rivista di psichiatria. 2014;49(3):124–31.
Schutter DJ. A cerebellar framework for predictive coding and homeostatic regulation in depressive disorder. Cerebellum. 2016;15(1):30–3.
Phillips JR, Hewedi DH, Eissa AM, Moustafa AA. The cerebellum and psychiatric disorders. Front Public Health. 2015;3:66.
Barkovich AJ, Millen KJ, Dobyns WB. A developmental and genetic classification for midbrain-hindbrain malformations. Brain. 2009;132(Pt 12):3199–230.
Ibrahim RS, Hachem RH. Pediatric cerebellar malformations: magnetic resonance diagnostic merits and correlation with neurodevelopmental outcome. Egypt J Radiol Nucl Med. 2020;51(1):1–10.
Abd-El-Barr MM, Strong CI, Groff MW. Chiari malformations: diagnosis, treatments and failures. J Neurosurg Sci. 2014;58(4):215–21.
Wilkins RH. Natural history of intracranial vascular malformations: a review. Neurosurgery. 1985;16(3):421–30.
Raybaud CA, Strother CM, Hald JK. Aneurysms of the vein of Galen: embryonic considerations and anatomical features relating to the pathogenesis of the malformation. Neuroradiology. 1989;31(2):109–28.
Rao VR, Mathuriya SN. Pediatric aneurysms and vein of Galen malformations. J Pediatr Neurosci. 2011;6(Suppl 1):S109–17.
Jones BV, Ball WS, Tomsick TA, Millard J, Crone KR. Vein of Galen aneurysmal malformation: diagnosis and treatment of 13 children with extended clinical follow-up. AJNR Am J Neuroradiol. 2002;23(10):1717–24.
Marzban H, Del Bigio MR, Alizadeh J, Ghavami S, Zachariah RM, Rastegar M. Cellular commitment in the developing cerebellum. Front Cell Neurosci. 2014;8:450.
Millet S, Bloch-Gallego E, Simeone A, Alvarado-Mallart RM. The caudal limit of Otx2 gene expression as a marker of the midbrain/hindbrain boundary: a study using in situ hybridisation and chick/quail homotopic grafts. Development. 1996;122(12):3785–97.
Millen KJ, Millonig JH, Wingate RJ, Alder J, Hatten ME. Neurogenetics of the cerebellar system. J Child Neurol. 1999;14(9):574–81; discussion 81–2
Eddison M, Toole L, Bell E, Wingate RJ. Segmental identity and cerebellar granule cell induction in rhombomere 1. BMC Biol. 2004;2:14.
Chizhikov VV, Lindgren AG, Currle DS, Rose MF, Monuki ES, Millen KJ. The roof plate regulates cerebellar cell-type specification and proliferation. Development. 2006;133(15):2793–804.
Basson MA, Wingate RJ. Congenital hypoplasia of the cerebellum: developmental causes and behavioral consequences. Front Neuroanat. 2013;7:29.
Vermeer S, Kremer HP, Leijten QH, Scheffer H, Matthijs G, Wevers RA, et al. Cerebellar ataxia and congenital disorder of glycosylation Ia (CDG-Ia) with normal routine CDG screening. J Neurol. 2007;254(10):1356–8.
Turkmen S, Hoffmann K, Demirhan O, Aruoba D, Humphrey N, Mundlos S. Cerebellar hypoplasia, with quadrupedal locomotion, caused by mutations in the very low-density lipoprotein receptor gene. Eur J Human Genet. 2008;16(9):1070–4.
Aldinger KA, Doherty D, editors. The genetics of cerebellar malformations. In: Seminars in fetal and neonatal medicine. Elsevier; 2016.
Pearson T, Curtis F, Al-Eyadhy A, Al-Tamemi S, Mazer B, Dror Y, et al. An intronic mutation in DKC1 in an infant with Hoyeraal-Hreidarsson syndrome. Am J Med Genet A. 2008;146A(16):2159–61.
des Portes V, Boddaert N, Sacco S, Briault S, Maincent K, Bahi N, et al. Specific clinical and brain MRI features in mentally retarded patients with mutations in the Oligophrenin-1 gene. Am J Med Genet A. 2004;124A(4):364–71.
Sellick GS, Barker KT, Stolte-Dijkstra I, Fleischmann C, Coleman RJ, Garrett C, et al. Mutations in PTF1A cause pancreatic and cerebellar agenesis. Nat Genet. 2004;36(12):1301–5.
Jaeken J, Matthijs G. Congenital disorders of glycosylation: a rapidly expanding disease family. Annu Rev Genomics Hum Genet. 2007;8:261–78.
Tentler D, Gustavsson P, Leisti J, Schueler M, Chelly J, Timonen E, et al. Deletion including the oligophrenin-1 gene associated with enlarged cerebral ventricles, cerebellar hypoplasia, seizures and ataxia. Eur J Human Genet. 1999;7(5):541–8.
Haldipur P, Millen KJ. What cerebellar malformations tell us about cerebellar development. Neurosci Lett. 2019;688:14–25.
Accogli A, Addour-Boudrahem N, Srour M. Diagnostic approach to cerebellar hypoplasia. Cerebellum. 2021;20(4):631–58.
Patel S, Barkovich AJ. Analysis and classification of cerebellar malformations. AJNR Am J Neuroradiol. 2002;23(7):1074–87.
Massoud M, Cagneaux M, Garel C, Varene N, Moutard ML, Billette T, et al. Prenatal unilateral cerebellar hypoplasia in a series of 26 cases: significance and implications for prenatal diagnosis. Ultrasound Obstetr Gynecol. 2014;44(4):447–54.
Wichman A, Frank LM, Kelly TE. Autosomal recessive congenital cerebellar hypoplasia. Clin Genet. 1985;27(4):373–82.
Poretti A, Boltshauser E, Doherty D. Cerebellar hypoplasia: differential diagnosis and diagnostic approach. Am J Med Genet C: Semin Med Genet. 2014;166C(2):211–26.
Poretti A, Boltshauser E. Terminology in morphological anomalies of the cerebellum does matter. Cerebellum Ataxias. 2015;2(1):1–6.
Chatur C, Balani A, Vadapalli R, Murthy MG. Isolated unilateral cerebellar hemispheric dysplasia: a rare entity. Can J Neurol Sci. 2019;46(6):760–1.
Pascual B, Hodics T, Funk Q, Cykowski MD, Nakawah MO, Masdeu JC. Translocator protein 18 kDa PET imaging highlights asymptomatic isolated cerebellar dysplasia. Neurology. 2022;
Di Stasi M, Izzo G, Cattaneo E, Baraldini V, Doneda C, Righini A, et al. Cerebellar dysplasia related to PIK3CA mutation: a three-case series. Neurogenetics. 2021;22(1):27–32.
Osenbach RK, Menezes AH. Diagnosis and management of the Dandy-Walker malformation: 30 years of experience. Pediatr Neurosurg. 1992;18(4):179–89.
Cueva-Nunez JE, Lozano-Bustillo A, Irias-Alvarez MS, Vasquez-Montes RF, Varela-Gonzalez DM. [Dandy-Walker variant: case report]. Rev Chil Pediatr. 2016;87(5):406–10.
Klein JL, Lemmon ME, Northington FJ, Boltshauser E, Huisman TA, Poretti A. Clinical and neuroimaging features as diagnostic guides in neonatal neurology diseases with cerebellar involvement. Cerebellum Ataxias. 2016;3:1.
Grinberg I, Northrup H, Ardinger H, Prasad C, Dobyns WB, Millen KJ. Heterozygous deletion of the linked genes ZIC1 and ZIC4 is involved in Dandy-Walker malformation. Nat Genet. 2004;36(10):1053–5.
Aldinger KA, Lehmann OJ, Hudgins L, Chizhikov VV, Bassuk AG, Ades LC, et al. FOXC1 is required for normal cerebellar development and is a major contributor to chromosome 6p25.3 Dandy-Walker malformation. Nat Genet. 2009;41(9):1037–42.
Chapman T, Kapur R. Cerebellar vermian dysplasia: the tale of the tail. J Pediatr Neurol Neurosci. 2018;2(1):17–22.
Alexiou G, Prodromou N. Dandy-walker malformation. Pediatr Neurosurg Clin. 2022:83–7.
Abdel Razek AA, Castillo M. Magnetic resonance imaging of malformations of midbrain-hindbrain. J Comput Assist Tomogr. 2016;40(1):14–25.
Cotes C, Bonfante E, Lazor J, Jadhav S, Caldas M, Swischuk L, et al. Congenital basis of posterior fossa anomalies. Neuroradiol J. 2015;28(3):238–53.
D’Agostino AN, Kernohan JW, Brown JR. The Dandy-Walker Syndrome. J Neuropathol Exp Neurol. 1963;22:450–70.
Hart MN, Malamud N, Ellis WG. The Dandy-Walker syndrome. A clinicopathological study based on 28 cases. Neurology. 1972;22(8):771–80.
Spennato P, Mirone G, Nastro A, Buonocore MC, Ruggiero C, Trischitta V, et al. Hydrocephalus in Dandy-Walker malformation. Child’s Nervous Syst. 2011;27(10):1665–81.
Nelson MD Jr, Maher K, Gilles FH. A different approach to cysts of the posterior fossa. Pediatr Radiol. 2004;34(9):720–32.
Tonni G, Lituania M, Chitayat D, Bonasoni MP, Keating S, Thompson M, et al. Complete trisomy 9 with unusual phenotypic associations: Dandy-Walker malformation, cleft lip and cleft palate, cardiovascular abnormalities. Taiwanese J Obstetr Gynecol. 2014;53(4):592–7.
Zaki MS, Masri A, Gregor A, Gleeson JG, Rosti RO. Dandy-Walker malformation, genitourinary abnormalities, and intellectual disability in two families. Am J Med Genet A. 2015;167A(11):2503–7.
Klein O, Pierre-Kahn A, Boddaert N, Parisot D, Brunelle F. Dandy-Walker malformation: prenatal diagnosis and prognosis. Child’s Nervous Syst. 2003;19(7-8):484–9.
Sasaki-Adams D, Elbabaa SK, Jewells V, Carter L, Campbell JW, Ritter AM. The Dandy-Walker variant: a case series of 24 pediatric patients and evaluation of associated anomalies, incidence of hydrocephalus, and developmental outcomes. J Neurosurg Pediatr. 2008;2(3):194–9.
Haddadi K, Zare A, Asadian L. Dandy-Walker Syndrome: a review of new diagnosis and management in children. J Pediatr Rev. 2018;6(2):47–52.
Guibaud L, Larroque A, Ville D, Sanlaville D, Till M, Gaucherand P, et al. Prenatal diagnosis of ‘isolated’ Dandy-Walker malformation: imaging findings and prenatal counselling. Prenat Diagn. 2012;32(2):185–93.
Joubert M, Eisenring JJ, Robb JP, Andermann F. Familial agenesis of the cerebellar vermis. A syndrome of episodic hyperpnea, abnormal eye movements, ataxia, and retardation. Neurology. 1969;19(9):813–25.
Valente EM, Dallapiccola B, Bertini E. Joubert syndrome and related disorders. Handb Clin Neurol. 2013;113:1879–88.
Usta M, Urganci N, Ozcelik G, Cetincelik U, Kafadar I, Ozguven BY. Joubert syndrome and related disorders: a rare cause of intrahepatic portal hypertension in childhood. Eur Rev Med Pharmacol Sci. 2015;19(12):2297–300.
Taytard J, Valence S, Sileo C, Rodriguez D, Bokov P, Aubertin G, et al. Severe central apnea secondary to cerebellar dysplasia in a child: look past Joubert syndrome. J Clin Sleep Med. 2020;16(12):2113–6.
Millen KJ, Gleeson JG. Cerebellar development and disease. Curr Opin Neurobiol. 2008;18(1):12–9.
Sattar S, Gleeson JG. The ciliopathies in neuronal development: a clinical approach to investigation of Joubert syndrome and Joubert syndrome-related disorders. Dev Med Child Neurol. 2011;53(9):793–8.
Chizhikov VV, Davenport J, Zhang Q, Shih EK, Cabello OA, Fuchs JL, et al. Cilia proteins control cerebellar morphogenesis by promoting expansion of the granule progenitor pool. J Neurosci. 2007;27(36):9780–9.
Spassky N, Han YG, Aguilar A, Strehl L, Besse L, Laclef C, et al. Primary cilia are required for cerebellar development and Shh-dependent expansion of progenitor pool. Dev Biol. 2008;317(1):246–59.
Bachmann-Gagescu R, Dona M, Hetterschijt L, Tonnaer E, Peters T, de Vrieze E, et al. The ciliopathy protein CC2D2A associates with NINL and functions in RAB8-MICAL3-regulated vesicle trafficking. PLoS Genet. 2015;11(10):e1005575.
Brancati F, Iannicelli M, Travaglini L, Mazzotta A, Bertini E, Boltshauser E, et al. MKS3/TMEM67 mutations are a major cause of COACH Syndrome, a Joubert Syndrome related disorder with liver involvement. Hum Mutat. 2009;30(2):E432–42.
Kamdar BB, Nandkumar P, Krishnan V, Gamaldo CE, Collop NA. Self-reported sleep and breathing disturbances in Joubert syndrome. Pediatr Neurol. 2011;45(6):395–9.
Brancati F, Dallapiccola B, Valente EM. Joubert Syndrome and related disorders. Orphanet J Rare Dis. 2010;5:20.
Nag C, Ghosh M, Das K, Ghosh T. Joubert syndrome: the molar tooth sign of the mid-brain. Ann Med Health Sci Res. 2013;3(2):291–4.
Lopez Ruiz P, Garcia ME, Dicapua Sacoto D, Marcos-Dolado A. Uncrossed epileptic seizures in Joubert syndrome. BMJ Case Rep 2015;2015.
Parisi MA, Dobyns WB. Human malformations of the midbrain and hindbrain: review and proposed classification scheme. Mol Genet Metab. 2003;80(1-2):36–53.
Bierhals T, Korenke GC, Uyanik G, Kutsche K. Pontocerebellar hypoplasia type 2 and TSEN2: review of the literature and two novel mutations. Eur J Med Genet. 2013;56(6):325–30.
Sanchez-Albisua I, Frolich S, Barth PG, Steinlin M, Krageloh-Mann I. Natural course of pontocerebellar hypoplasia type 2A. Orphanet J Rare Dis. 2014;9:70.
Eggens VR, Barth PG, Niermeijer JM, Berg JN, Darin N, Dixit A, et al. EXOSC3 mutations in pontocerebellar hypoplasia type 1: novel mutations and genotype-phenotype correlations. Orphanet J Rare Dis. 2014;9:23.
Rudnik-Schoneborn S, Sztriha L, Aithala GR, Houge G, Laegreid LM, Seeger J, et al. Extended phenotype of pontocerebellar hypoplasia with infantile spinal muscular atrophy. Am J Med Genet A. 2003;117A(1):10–7.
Renbaum P, Kellerman E, Jaron R, Geiger D, Segel R, Lee M, et al. Spinal muscular atrophy with pontocerebellar hypoplasia is caused by a mutation in the VRK1 gene. Am J Hum Genet. 2009;85(2):281–9.
Wan J, Yourshaw M, Mamsa H, Rudnik-Schoneborn S, Menezes MP, Hong JE, et al. Mutations in the RNA exosome component gene EXOSC3 cause pontocerebellar hypoplasia and spinal motor neuron degeneration. Nat Genet. 2012;44(6):704–8.
Budde BS, Namavar Y, Barth PG, Poll-The BT, Nurnberg G, Becker C, et al. tRNA splicing endonuclease mutations cause pontocerebellar hypoplasia. Nat Genet. 2008;40(9):1113–8.
Samanta D, Willis E. Intractable epileptic spasms in a patient with Pontocerebellar hypoplasia: Severe phenotype of type 2 or another subtype? Ann Indian Acad Neurol. 2016;19(3):385–7.
Feinstein M, Flusser H, Lerman-Sagie T, Ben-Zeev B, Lev D, Agamy O, et al. VPS53 mutations cause progressive cerebello-cerebral atrophy type 2 (PCCA2). J Med Genet. 2014;51(5):303–8.
Rajab A, Mochida GH, Hill A, Ganesh V, Bodell A, Riaz A, et al. A novel form of pontocerebellar hypoplasia maps to chromosome 7q11-21. Neurology. 2003;60(10):1664–7.
Ahmed MY, Chioza BA, Rajab A, Schmitz-Abe K, Al-Khayat A, Al-Turki S, et al. Loss of PCLO function underlies pontocerebellar hypoplasia type III. Neurology. 2015;84(17):1745–50.
Edvardson S, Shaag A, Kolesnikova O, Gomori JM, Tarassov I, Einbinder T, et al. Deleterious mutation in the mitochondrial arginyl-transfer RNA synthetase gene is associated with pontocerebellar hypoplasia. Am J Hum Genet. 2007;81(4):857–62.
Anderson C, Davies JH, Lamont L, Foulds N. Early pontocerebellar hypoplasia with vanishing testes: A new syndrome? Am J Med Genet A. 2011;155A(4):667–72.
Namavar Y, Barth PG, Poll-The BT, Baas F. Classification, diagnosis and potential mechanisms in pontocerebellar hypoplasia. Orphanet J Rare Dis. 2011;6:50.
Mochida GH, Ganesh VS, de Michelena MI, Dias H, Atabay KD, Kathrein KL, et al. CHMP1A encodes an essential regulator of BMI1-INK4A in cerebellar development. Nat Genet. 2012;44(11):1260–4.
Akizu N, Cantagrel V, Schroth J, Cai N, Vaux K, McCloskey D, et al. AMPD2 regulates GTP synthesis and is mutated in a potentially treatable neurodegenerative brainstem disorder. Cell. 2013;154(3):505–17.
Karaca E, Weitzer S, Pehlivan D, Shiraishi H, Gogakos T, Hanada T, et al. Human CLP1 mutations alter tRNA biogenesis, affecting both peripheral and central nervous system function. Cell. 2014;157(3):636–50.
Wan J, Steffen J, Yourshaw M, Mamsa H, Andersen E, Rudnik-Schoneborn S, et al. Loss of function of SLC25A46 causes lethal congenital pontocerebellar hypoplasia. Brain. 2016;
Christiansen S, Roos LK, Miranda MJ. [Pontocerebellar hypoplasia is a rare cause of floppy infant syndrome]. Ugeskr Laeger. 2015;177(40):V05150380.
Ishak GE, Dempsey JC, Shaw DW, Tully H, Adam MP, Sanchez-Lara PA, et al. Rhombencephalosynapsis: a hindbrain malformation associated with incomplete separation of midbrain and forebrain, hydrocephalus and a broad spectrum of severity. Brain. 2012;135(Pt 5):1370–86.
Rahimi-Balaei M, Marzban H, Hawkes R. Early cerebellar development in relation to the trigeminal system. Cerebellum. 2022;
Pasquier L, Marcorelles P, Loget P, Pelluard F, Carles D, Perez MJ, et al. Rhombencephalosynapsis and related anomalies: a neuropathological study of 40 fetal cases. Acta Neuropathol. 2009;117(2):185–200.
Sukhudyan B, Jaladyan V, Melikyan G, Schlump JU, Boltshauser E, Poretti A. Gomez-Lopez-Hernandez syndrome: reappraisal of the diagnostic criteria. Eur J Pediatr. 2010;169(12):1523–8.
Gomez MR. Cerebellotrigeminal and focal dermal dysplasia: a newly recognized neurocutaneous syndrome. Brain Dev. 1979;1(4):253–6.
Lopez-Hernandez A. Craniosynostosis, ataxia, trigeminal anaesthesia and parietal alopecia with pons-vermis fusion anomaly (atresia of the fourth ventricle). Report of two cases. Neuropediatrics. 1982;13(2):99–102.
Sarnat HB. Cerebellar networks and neuropathology of cerebellar developmental disorders. Handb Clin Neurol. 2018;154:109–28.
Kruer MC, Blasco PA, Anderson JC, Bardo DM, Pinter JD. Truncal ataxia, hypotonia, and motor delay with isolated rhombencephalosynapsis. Pediatr Neurol. 2009;41(3):229–31.
Ross ME, Swanson K, Dobyns WB. Lissencephaly with cerebellar hypoplasia (LCH): a heterogeneous group of cortical malformations. Neuropediatrics. 2001;32(5):256–63.
al Shahwan SA, Bruyn GW, al Deeb SM. Non-progressive familial congenital cerebellar hypoplasia. J Neurol Sci. 1995;128(1):71–7.
Hong SE, Shugart YY, Huang DT, Shahwan SA, Grant PE, Hourihane JO, et al. Autosomal recessive lissencephaly with cerebellar hypoplasia is associated with human RELN mutations. Nat Genet. 2000;26(1):93–6.
Kroon AA, Smit BJ, Barth PG, Hennekam RC. Lissencephaly with extreme cerebral and cerebellar hypoplasia. A magnetic resonance imaging study. Neuropediatrics. 1996;27(5):273–6.
D’Arcangelo G, Miao GG, Chen SC, Soares HD, Morgan JI, Curran T. A protein related to extracellular matrix proteins deleted in the mouse mutant reeler. Nature. 1995;374(6524):719–23.
D’Arcangelo G, Homayouni R, Keshvara L, Rice DS, Sheldon M, Curran T. Reelin is a ligand for lipoprotein receptors. Neuron. 1999;24(2):471–9.
Hiesberger T, Trommsdorff M, Howell BW, Goffinet A, Mumby MC, Cooper JA, et al. Direct binding of Reelin to VLDL receptor and ApoE receptor 2 induces tyrosine phosphorylation of disabled-1 and modulates tau phosphorylation. Neuron. 1999;24(2):481–9.
Trommsdorff M, Gotthardt M, Hiesberger T, Shelton J, Stockinger W, Nimpf J, et al. Reeler/Disabled-like disruption of neuronal migration in knockout mice lacking the VLDL receptor and ApoE receptor 2. Cell. 1999;97(6):689–701.
Senzaki K, Ogawa M, Yagi T. Proteins of the CNR family are multiple receptors for Reelin. Cell. 1999;99(6):635–47.
Caviness VS Jr, Rakic P. Mechanisms of cortical development: a view from mutations in mice. Annu Rev Neurosci. 1978;1:297–326.
Lambert de Rouvroit C, Goffinet AM. The reeler mouse as a model of brain development. Adv Anat Embryol Cell Biol. 1998;150:1–106.
Yis U. Lissencephaly with brainstem and cerebellar hypoplasia and congenital cataracts. J Child Neurol. 2015;30(5):625–6.
Klisch J, Juengling F, Spreer J, Koch D, Thiel T, Buchert M, et al. Lhermitte-Duclos disease: assessment with MR imaging, positron emission tomography, single-photon emission CT, and MR spectroscopy. Am J Neuroradiol. 2001;22(5):824–30.
Shinagare AB, Patil NK, Sorte SZ. Case 144: dysplastic cerebellar gangliocytoma (Lhermitte-Duclos disease). Radiology. 2009;251(1):298–303.
Zhou XP, Marsh DJ, Morrison CD, Chaudhury AR, Maxwell M, Reifenberger G, et al. Germline inactivation of PTEN and dysregulation of the phosphoinositol-3-kinase/Akt pathway cause human Lhermitte-Duclos disease in adults. Am J Hum Genet. 2003;73(5):1191–8.
Maehama T, Dixon JE. The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem. 1998;273(22):13375–8.
Roessmann U, Wongmongkolrit T. Dysplastic gangliocytoma of cerebellum in a newborn. Case report. J Neurosurg. 1984;60(4):845–7.
Vieco PT, del Carpio-O’Donovan R, Melanson D, Montes J, O’Gorman AM, Meagher-Villemure K. Dysplastic gangliocytoma (Lhermitte-Duclos disease): CT and MR imaging. Pediatr Radiol. 1992;22(5):366–9.
Milbouw G, Born JD, Martin D, Collignon J, Hans P, Reznik M, et al. Clinical and radiological aspects of dysplastic gangliocytoma (Lhermitte-Duclos disease): a report of two cases with review of the literature. Neurosurgery. 1988;22(1 Pt 1):124–8.
Ashley DG, Zee CS, Chandrasoma PT, Segall HD. Lhermitte-Duclos disease: CT and MR findings. J Comput Assist Tomogr. 1990;14(6):984–7.
Nowak DA, Trost HA. Lhermitte-Duclos disease (dysplastic cerebellar gangliocytoma): a malformation, hamartoma or neoplasm? Acta Neurol Scand. 2002;105(3):137–45.
Padberg GW, Schot JD, Vielvoye GJ, Bots GT, de Beer FC. Lhermitte-Duclos disease and Cowden disease: a single phakomatosis. Ann Neurol. 1991;29(5):517–23.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Marzban, A., Ghiyamihoor, F., Vafaee-shahi, M., Azarkhish, K. (2023). Clinical Aspects of the Inherited Cerebellar Malformations. In: Marzban, H. (eds) Development of the Cerebellum from Molecular Aspects to Diseases. Contemporary Clinical Neuroscience. Springer, Cham. https://doi.org/10.1007/978-3-031-23104-9_22
Download citation
DOI: https://doi.org/10.1007/978-3-031-23104-9_22
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-23103-2
Online ISBN: 978-3-031-23104-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)