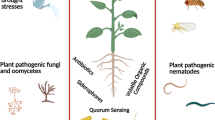
Abstract
Plant growth-promoting rhizobacteria (PGPR) are being used as an alternative approach to combat plant diseases. About 80–90% of plant diseases are caused by bacterial and fungal pathogens, which remain an inevitable cause for the loss of several crops. Phytopathogenic bacteria and fungi are the major constraints to sustainable agriculture by adversely affecting crop growth and productivity. Owing to the increased pollution and harmful impacts of chemicals to control these pathogens, scientists are now centering on safer biological organisms and their byproducts. Secondary metabolites and volatile organic compounds (VOCs) emitted by various beneficial bacterial strains have a lot of potential for enhancing plant growth and preventing plant diseases. The VOCs produced by the most researched bacterial strains, such as Pseudomonas genera, are well recognized for protecting economically imperative plants and inducing resistance against bacterial and fungal phytopathogens. This chapter concentrates on throwing up a better grasp of biological activities of secondary metabolites such as hydrogen cyanide, siderophores, antibiotics, and VOCs produced by Pseudomonas spp. Hundreds of various bacterial VOCs, including alcohols, terpenoids, esters, and sulfur compounds, have been discovered. The VOCs emitted by Pseudomonas sp., for instance, acetophenone, 1,3-butadiene, 2-undecanone, benzaldehyde, 1,2-benzisothiazol-3(2H)-one, dimethyl trisulfide, dimethyl disulfide, benzothiazole, nonanal, N,N-dimethyldodecylamin, 3,5,5-trimethyl-1-hexanol, isovaleric acid, cyclohexanol, 2-ethyl 1-hexanol, n-decanal, decyl alcohol, etc., are reported for their antagonistic potential, inducing resistance in host plants against several bacterial and fungal pathogens. Crop growth enhancement and protection via VOCs is a promising and an ecofriendly method, substituting the harmful impacts of chemicals and ensuring the long-term sustainability in agriculture.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
In the twenty-first century, according to United Nations the global population is increasing continuously which is projected to reach 9.7 billion in 2050,, which may lead to increase in global agricultural production, to fulfill the requirements of rapidly growing population (UNDESAP 2017; Rohr et al. 2019). Our agricultural sector largely depends upon the use of synthetic chemicals in order to revamp the crop production, i.e., synthetic fertilizers, which are used to increase the crop biomass, while synthetic pesticides are used to control pest and diseases in crops to reduce crop loss by 17–30%, particularly for the major staple crops (Naz and Bano 2014; Savary et al. 2019). However, these synthetic pesticides are unendurable due to their harmful residual effects and heavy manufacturing costs (Naz et al. 2014, 2018, 2021a). According to an estimation, around $250 million are required to take single active ingredients in market, having very low success rate about only 1 out of 140,000 synthetic compounds are successful, which is a very unsustainable way to develop synthetic pesticides (Lamberth et al. 2013).
Besides these, the continued use of pesticides makes them less effective because of the production of pesticide-resistant genes in plants (Butt et al. 2019; Ullah et al. 2020; Naz et al. 2021b). Furthermore, the continuous increase in global population has increased the demands for crops and agricultural growth, which has further caused increase in the applications of synthetic compounds. As projected increase in the demand for crops, agricultural growth might result in increased pesticide use of 10-fold and increased fertilizer application of 2.7-fold (Rohr et al. 2019; Jabborova et al. 2020).
Agronomic practices should be taken in consideration to lessen this dependency on synthetic compounds as well as to evolve the viable control measures, and different collaborative efforts should be made, i.e., improving agricultural practices by agronomic practices (Naz and Bano 2015; Ahluwalia et al. 2021). However, the introduction of soil beneficial microorganisms is another effective method to reduce the use of synthetic compounds in agricultural practices, as they have potential to antagonizing soil pathogenic microbes and are capable of increasing plant biomass (Yasmin et al. 2019; Luh Suriani et al. 2020). An extensive range of secondary metabolites is produced by these soil microorganisms which strengthen them to fight with other pathogenic soil microbes, as they compete with each other for same resources in soil (Naz et al. 2017; Garbeva and Weisskopf 2020; Hamid et al. 2021).
The production of antibiotics, volatile organic compounds (VOCs), and secondary metabolites during microbial lifecycles are some other microbial inhibition tools to cope with pathogenic microbes within soil (Naz et al. 2020; Ye et al. 2020; Khan et al. 2021). The scientific world requires more attention on the production of VOCs due to multiple benefits of their utilization. VOCs are a mixture of volatile metabolites that may be emitted by all living microorganisms and have been shown to be very potent to control the growth of phytopathogenic bacteria and fungus through cross-talk interactions and antibacterial activities. Their antimicrobial effects, along with the reduced hazard for both the environment and human beings and their possible application without the need of a supplemental spray or drench, make the use of VOCs a promising and sustainable approach to replace fungicides of synthetic origin in the control of plant pathogens (Parafati et al. 2017; Tilocca and Migheli 2020; Zhang et al. 2020).
Although numerous modes of action are involved in phytopathogen obliteration, this chapter will dig into novel visions and ideas in biological control of phytopathogens via PGPR by dint of antibiotics and VOCs. Some Pseudomonas spp. have been associated with plant growth, suppression of fungal pathogens affecting plants, and detrimental rhizobacteria presenting considerable upsurge in root colonization. These aspects suggest that Pseudomonas spp. can serve as excellent biocontrol agents (Gomez-Lama et al. 2018; Reshma et al. 2018).
In this chapter, we focus to explore the role of secondary metabolites, antibiotics, and VOCs produced by the Pseudomonas species to sustain plant health by directly suppressing pathogens, inducing plant resistance against phytopathogens, and promoting plant growth, emphasizing their potential as alternatives to synthetic fertilizers and pesticides.
2 Microorganisms Emitting Volatile Organic Compounds
The volatile metabolites emitted from both plant and microbial sources are receiving a steady increase in interest. The word “volatilome” has been relatively recently used to describe this diverse and heterogeneous collection of metabolites (Farbo et al. 2018; Tilocca and Migheli 2020). The volatile metabolites of plant and microbial origin are mainly differentiated into organic and inorganic volatile molecules. Among inorganic volatile molecules, most relevant are CO2, H2S, CO, HCN, SO3, H2, NH3, NO2−, and SO2. The inorganic volatile molecules play an important role in different biological functions, i.e., acting as defense compounds by donating/accepting electrons (Rad et al. 2016; Zhang et al. 2020). These metabolites also have a role in various ecological and biological features along with their antibiotic resistance potential (Avalos et al. 2019; Kenawy et al. 2019).
In agriculture, the application of VOCs with microbial source in the biological control of plants pathogens has been given an unintentional decrease over the last few years. However, the progress recently made and the general trend to an integrative approach have highlighted the potential advantages of microbiological VOCs in this area. The VOCs are known to be very effective at very low levels (Reshma et al. 2018; Tilocca and Migheli 2020).
In addition to pathogen inhibition and negative impact on fungal spore germination and function of morphogenesis enzymes, VOCs from microbial species have been found to play a role in a variety of biological processes (Deveau et al. 2018; Zhang et al. 2020). VOCs have the capability to kill nematodes which are known to be parasitic for plants (de Freitas Silva et al. 2020; Khoja et al. 2021) to increase plant growth (Hernández-León et al. 2015; Fincheira and Quiroz 2018) and to activate the mechanisms associated with resistance within plants, thus averting the plant from being infected by pathogens (Sharifi and Ryu 2016; Tahir et al. 2017; Zhang et al. 2020).
2.1 Production of VOCs by Consortium of Different Microbes
A single organism can produce a diverse mixture of VOCs when applied, which leads to different outcomes proved by many experiments (Tilocca et al. 2019), whereas in a single ecological niche, there reside many microbial entities through which unexpected achievements can be obtained by consortium application as compared to the application of single microbial strain (Khan et al. 2019). Microbial strains interact with each other irrespective of their genera, phyla, and kingdom (Shaikh et al. 2016; Schulz-Bohm et al. 2017). These interactions lead to the essential biological and ecological outcomes in ensembled role of all the microbiota as a single unique entity. The effectiveness of the interactions occurring between microbiota members Pseudomonas helmanticensis Sc-B94 and Bacillus cereus Rs-MS53 has been reported to control the pathogenic fungus R. solani (Mülner et al. 2019), which was proved to be a strong strain compatibility and cooperative interaction (Asari et al. 2016; Che and Men 2019; Kramer et al. 2020). The production of volatile and nonvolatile compounds by different strains of Pseudomonas and Bacillus spp. can directly inhibit the growth of pathogen or can help in the acclimatization of the microbial community already residing in the same ecological niche which can also inhibit growth and infection caused by pathogen (Schulz-Bohm et al. 2017; Tilocca et al. 2020; Dimkić et al. 2022).
3 Bacterial Volatiles: Tool to Biocontrol of Phytopathogens
Bacterial VOCs play a role in the complex network of interactions that are established between bacteria, bacterial species, and bacteria with other microorganisms as well as with plants. Similarly, these interactions play a variable ecological role including beneficial interaction as well as antagonistic interaction. However, beneficial cooperation encompasses symbiosis, mutualism, and host resistance interaction, while in antagonistic relationship, one of the interacting species exerts microbicidal activity on other species (Kanchiswamy et al. 2015; Tilocca et al. 2020). The useful bacterial-plant interaction has recently been recognized, which has extend new approaches for the use of bacterial volatilome in promoting plant growth. Furthermore, due to high flexibility of bacterial origin VOCs as well as their efficacy in controlling other pathogens, investigation is made on the utilization of VOCs produced by natural bacteria in defense against plant pathogenic microbes (Reshma et al. 2018; Mulero-Aparicio et al. 2019).
A wide variety of VOCs have been produced by rhizobacteria (Serratia odorifera, S. plymuthica, Stenotrophomonas maltophilia, P. fluorescens, Stenotrophomonas rhizophila, and Pseudomonas trivialis) which are active against an extensive variety of pathogenic microorganisms including bacteria and fungi (Kanchiswamy et al. 2015; Gotor-Vila et al. 2017; Mulero-Aparicio et al. 2019). Bacteria-fungi interaction usually produces some common volatile molecules including 1-octen3-ol, 2-nonanone, 2-undecanone, γ-patchoulene, 3-methylbutanoate, 3-methylbutanal, 2-methylbutan-1-ol, ethanethioic acid, dimethyl trisulfide 2,3,6-trimethylphenol, and 4-methyl-2-heptanone. Among these antifungal activities of some VOCs have already been tested (Tilocca et al. 2020).
4 Pseudomonas Volatilome
Various studies revealed that VOCs can inhibit a wide range of plant pathogens, also emphasizing VOCs as possible viable alternatives to pesticides and chemical fertilizers. One of the first examples of VOCs, produced by Pseudomonas species from canola and soyabean, exhibits plant growth stimulatory and inhibitory effect in case of plant pathogenic microbes (Agisha et al. 2019). About 23 VOCs are identified, which are produced by Pseudomonas species; among these six VOCs inhibited the mycelium growth of S. sclerotiorum, a pathogen of more than 400 plant species (Effmert et al. 2012; Thomas et al. 2020). A growth of widespread soil-borne R. solani pathogen was inhibited by VOCs from Pseudomonas spp. (Elkahoui et al. 2015) and by a variety of other rhizobacterial isolates (Velivelli et al. 2015). However, inhibitory activity against various bacterial pathogens exhibited by many VOCs is reported; for instance, nonanal, benzaldehyde, acetophenone, and benzothiazole are reported to inhibit the proliferation of Clavibacter michiganensis, a causative agent of bacterial ring rot disease of potato (Rajer et al. 2017). Similarly, Xanthomonas oryzae causing bacterial leaf blight of rice has been reported to be inhibited by 3,5,5-trimethyl-1-hexanol and decyl alcohol (Xie et al. 2018).
Moreover, the bacterial VOCs are also known to inhibit fungal mycelial growth; e.g., isovaleraldehyde, 3-methyl-1-butanol, isovaleric acid, 2-heptanone, and 2-ethylhexanol decrease the mycelium growth of Phytophthora capsica (Syed-Ab-Rahman et al. 2019; Freitas et al. 2022). Anti-oomycete activity is displayed by the VOCs of Nodulisporium against different Pythium species, while VOCs which are causing inhibition of pathogens were not assayed individually (Sánchez-Fernández et al. 2016). However, these studies spotlight inhibitory activity of VOCs against a wide range of bacterial and pathogens, which could be good alternatives to pesticides.
4.1 Role of Pseudomonas Volatilome in Biocontrol of Phytopathogens
Pseudomonas is widely recognized for having a diverse storage of plant growth-enhancing and antifungal metabolites, and many of these molecules are volatile compounds (Hernández-León et al. 2015; Yan et al. 2017; Dahiya et al. 2020). For instance, recently, it is reported by Hunziker et al. (2015) that P. infestans (a well-known oomycete phytopathogen of potato) can be inhibited by high potential volatiles emitted by Pseudomonas. The VOCs produced by P. fluorescens and P. trivialis are also reported to drastically inhibit the mycelial growth of R. solani (Kai et al. 2007). In recent studies, it was revealed that P. donghuensis P482 in the rhizosphere of tomato plants emits volatiles that play a significant role in inhibiting the growth of different plant pathogens for instance Pythium ultimum, R. solani, Verticillium dahlia, and F. culmorum (Ossowicki et al. 2017).
Evidences related to bacteriostatic were also found in the volatilomes of several strains of Pseudomonas spp. particularly in P. chlororaphis, which was tested against Agrobacterium tumefaciens and fungal, nematode, and insect pathogens for its antagonistic potential (Popova et al. 2014). The VOCs emitted from P. putida BP25 including 2-ethyl-5-methyl pyrazine, 2,5-dimethyl pyrazine, 2-ethyl-3,6-dimethyl pyrazine, 2-methyl pyrazine, and dimethyl trisulfide exhibited significant in vitro antimicrobial potential against several pathogens, for instance, C. gloeosporioides, P. capsici, G. moniliformis, P. myriotylum, R. solani, R. pseudosolanacearum, A. rolfsii, R. similis, and M. oryzae (Agisha et al. 2019).
From rhizosphere of soybean, common bean, and canola plants, the Pseudomonas strains were isolated and further reported for antagonistic potential against S. sclerotiorum owing to their VOCs including dimethyl trisulfide, n-decanal, benzothiazole, nonanal, cyclohexanol, and 2-ethyl 1-hexanol (Fernando et al. 2005; Giorgio et al. 2015). The antagonistic ability of VOCs produced by P. fluorescens B-4117 and P. fluorescens Q8r1-96 has been reported against plant pathogenic bacterial strains including A. vitis and A. tumefaciens. Here, it is suggested that Pseudomonas species are known to produce VOCs which can be used as a potential tool to control many diseases particularly the crown gall tumors which can be effectively prevented in tomato plants (Dandurishvili et al. 2011). The VOCs produced by P. fluorescens WR-1 are also reported to significantly affect and decrease the virulence characteristics of R. solanacearum in tomato (Raza et al. 2016). The active VOCs produced by Pseudomonas spp. and their biocontrol potential against target phytopathogens have been described in Table 6.1.
4.2 Role of Secondary Metabolites Produced by Pseudomonas spp. in Plant Disease Control
Fluorescent pseudomonads are predominant antagonistic bacteria that live in soil. Nowadays, the significance of these bacteria has been acknowledged all over the world, owing to the fact that they are capable of synthesizing a variety of antifungal compounds such as siderophores; fluorescent pigments along with volatile elements, namely, hydrocyanic acid (HCN); lytic enzymes; as well as antibiotics (Ciancio et al. 2016; Jadhav et al. 2017; Yasmin et al. 2020). Some of the noteworthy lytic enzymes produced by Pseudomonas spp. are chitinase, protease, and β-1,3-glucanase. These enzymes instigate lysis and hyperparasitism of antagonistic bacteria toward lethal fungal pathogens (Jadhav et al. 2017; Zia et al. 2021).
Various fluorescent pseudomonads are impervious to cyanide due to the existence of a thiosulfate (RhdA): cyanide sulfur transferase that modifies the cyanide to thiocyanate which is less toxic. In many Pseudomonas spp., approximately 300 μM cyanide is produced by the oxidative decarboxylation of glycine (Blumer and Haas 2000). Gupta et al. (2002) investigated the Pseudomonas to biologically control the charcoal rot instigated by Macrophomina phaseolina in peanut.
Sindhu et al. (1997) reported the role of secondary metabolites in the inhibition of phytopathogens and also the inhibiting role of siderophore-producing rhizobacteria and several fluorescent Pseudomonas spp. against many bacterial and fungal phytopathogens. Siderophore- producing pseudomonads have been reported in chickpea to markedly reduce the root rot disease (Akhtar and Siddiqui 2009).
Pseudomonas fluorescens are known to produce siderophore and control Pythium ultimum, and Pseudomonas stutzeri produces chitinase which lyse the cell wall of Fusarium solani. Antifungal metabolites produced by these Rhizobacteria were identified as antibiotics (iturin, surfactins, fengycin, DAPG, phenazine, etc.), cell wall degrading enzymes (proteses, chitinases, cellulases), plant growth-promoting enzymes and hormones (indole-3-acetic acid, ACC-deaminase, phosphatase, nitrogen fixation), N-acyl homoderine lactones, and siderophore (Dahiya et al. 2020).
Another siderophore as pseudobactin produced by P. putida was able to suppress the growth of Fusarium oxysporum in iron-deficient soil; this suppression/inhibition was abandoned when iron was provided in that soil (de Boer et al. 2003). Several studies have explained the inhibition of fungal pathogens by fluorescent pseudomonads from the excretion of siderophores (iron-chelating), making it inaccessible to other several microorganisms (Shaikh et al. 2014).
The biocontrol potential of siderophore as an antifungal metabolite produced by Pseudomonas spp. is shown in Table 6.2.
4.3 Antibiotics Produced by Pseudomonas spp.
According to Haas and Défago (2005), six antibiotic classes are best to perform their biocontrol potential particularly to control root fungal diseases: pyoluteorin, phenazines, pyrrolnitrin, phloroglucinols, hydrogen cyanide (which is volatile), and cyclic lipopeptides. Most recently, lipopeptide biosurfactants produced by Pseudomonas spp. have been implied in biocontrol due to their potential positive impact on the competitive contacts with organisms involving fungi, bacteria, oomycetes, nematodes, protozoa, and plants (Raaijmakers et al. 2010; Shafi et al. 2017; Fira et al. 2018).
Many bacterial species are reported for the isolation of several antibiotics that are known to inhibit cell wall composition of the pathogen, interrupt the cell membrane structures, and impede the synthesis of ribosomal subunits (Maksimov et al. 2011). Fluorescent pseudomonads primarily achieve biocontrol of pathogens by synthesizing specific antibiotics like pyoluteorin, 2,4-diacetylphloroglucinol, pyrrolnitrin, 2-hydroxy phenazines, and phenazine-1-carboxamide and phenazine-1-carboxyclic acid (Mustafa et al. 2019). Antibiotics are not just solely involved in antipathogenic activity; they are also major contributors in instigating ISR in plants as they vigorously suppress disease by offering competitive leverage to biocontrol agents. Host resistance toward plant pathogens is enhanced significantly when ISR and antibiotics act synergistically (Hashem et al. 2019; Ullah et al. 2020).
More than 6000 compounds have been characterized and identified for strong antifungal potential, including phenazine (PHZ) as a key molecule and over 100 more derivatives of PHZ (Mavrodi et al. 2006). Moreover, the products containing PHZ (even more than 180) are known for their strong antifungal, antibiotic, anticancer, insecticidal, anti-protozoan, and antitumor potential (Briard et al. 2015; Guttenberger et al. 2017). Several studies attributed the antimicrobial potential of PHZ produced by Pseudomonas strain PCL1391 to the production of ROS (reactive oxygen species) (Laursen and Nielsen 2004) and found very effective against Botrytis cinerea, Gaeumannomyces graminis, and F. oxysporum (Schoonbeek et al. 2002; Chin-A-Woeng et al. 2003). Several PHZ and its derivatives are efficient in controlling numerous fungal diseases (Chincholkar et al. 2013). The P. chlororaphis PCL1391 strain has been reported to produce phenazine-1-carboxamide, which can nourish plants with soluble iron at neutral pH (Hernandez et al. 2004; Haas and Défago 2005).
The fluorescent pseudomonads producing DAPG are reported for their strong biocontrol potential (Weller et al. 2007; Troppens et al. 2013); several other research studies have confirmed DAPG as a key antimicrobial metabolite engaged in the biocontrol of fungal phytopathogens (Sonnleitner and Haas 2011; Khare et al. 2018). The DAPG is an efficient and extensively researched antibiotic which is released by pseudomonads to control oomycete and Pythium spp. (de Souza et al. 2003).
Pyoluteorin (PLT) is a phenolic polyketide, which has initially been isolated and identified from P. aeruginosa and then from fluorescent pseudomonads (Nowak-Thompson et al. 1997). PLT has herbicidal, bactericidal, and fungicidal properties (Takeda 1959). PLT has also been stated to function as an intercellular signal and auto-inducer among distinctive rhizospheric populations of bacterial strains (Brodhagen et al. 2004). It has recently been observed that phloroglucinol in P. protegens has a significant impact on PLT gene expression and production (Clifford et al. 2016).
Different metabolites are produced at different concentration of phloroglucinol with distinct phytopathogenic target (Khare et al. 2018). Limited range of gram-negative bacteria are involved in the production of pyrrolnitrin from Pseudomonas species (Mujumdar et al. 2014; Weller et al. 2016). Fluorescent pseudomonads produce pyrrolnitrin which has antagonistic nature against fungi, yeast, and Gram-positive bacteria (Jani et al. 2015). P. fluorescens BL915 strain secretes pyrrolnitrin which has a property to protect Rhizoctonia solani during damping off of cotton (Hill et al. 1994).
Currently, cyclic lipopeptides (CLPs) have been identified as biosurfactant and antimicrobials which is found effective against broad spectrum of phytopathogen involving enveloped viruses, Gram-positive bacteria, and mycoplasmas (Raaijmakers et al. 2006; Tran et al. 2007; Raju et al. 2016). Research has reported that CLPs released by pseudomonads are involved in colonization of seeds and roots. In addition, it also contributes to the formation of biofilm and virulence (Li et al. 2013; Raaijmakers et al. 2010). Fluorescent pseudomonads release different types of CLPs; many of them have not characterized completely. The well-documented and studied groups of CLPs are amphisin, viscosin syringomycin, and tolaasin (Nybroe and Sørensen 2004).
CLPs secreted by Pseudomonas are categorized into eight different groups on the basis of variation in length and composition of the oligopeptide and fatty acid tails (Olorunleke et al. 2017). The ability to agitate biological membranes are associated with the antimicrobial properties (Raaijmakers et al. 2006; Dumée et al. 2015). P. protegens produce orfamide which is a type of potential CLPs having insecticidal property (Nandi et al. 2015). Fluorescent pseudomonads releasing several metabolites having broad-spectrum phytopathogenic activities are preferred in the field of agriculture. Currently, Izzah-Shahid et al. (2017) reported that application of PCA, CLP, and lahorenoic acid A substantially enhanced growth of wheat by producing P. chlororaphis and P. aurantiaca during development. Sharifazizi et al. (2017) also found that fluorescent pseudomonad strain Ps170 has the capability to control blight-causing pathogen in pear by releasing DAPG, PLT, PRN, and PCA. Metabolites of fluorescent pseudomonads are currently being used as biological controls to secure the plant from causative agents such as causing protozoa and nematodes (Meyer et al. 2009; Jousset et al. 2010; Clifford et al. 2016). Antibiotics produced by Pseudomonas spp. and their biocontrol potential against phytopathogens have been described in Table 6.3 and Fig. 6.1.
5 Conclusion
Pseudomonas spp. are plant growth-stimulating bacteria that are often observed with diverse phyto-beneficial characteristics. The biological activities of hydrogen cyanide, siderophore, antibiotics, and VOCs produced by these species highlight their potential to act as alternatives to unsustainable agricultural chemical inputs and to feed a continuously growing population. In this chapter, we have investigated the biocontrol potential of secondary metabolites and VOCs produced by Pseudomonas species (Tables 6.1, 6.2, and 6.3), which have more and diverse abilities to fight phytopathogens. Therefore, future research should focus on the growth-stimulating effects of antibiotics and VOCs on various crop and vegetable species. This chapter represented here focuses on the antibiotics and particularly VOCs emitted by Pseudomonas spp. in axenic culture conditions, whereas growing evidence suggests that interaction between different microorganisms could boost the production of VOCs which have been shown to have inhibition against pathogens. This will allow to identify the biologically relevant VOCs that are effectively involved in the inhibition of microbial pathogens. While a number of studies have also investigated the impact of VOCs in one biological function, there are likely to be similarities in the functions of these VOCs. For instance, nonadecane and heptadecane exhibited their role in pathogen suppression, plant growth promotion, and induced resistance, which suggests that the biological activities are not the isolated entity. Studies have shown the pathogenic suppression in the presence of the VOCs, but it is also important to know the involvement of these inhibitory VOCs on plant growth. Further investigation on the efficacy of VOC under field conditions can be a promising approach. There is a dire need for further exploration for the testing of a wider range of VOCs for field applications.
In conclusion, studies reviewed here demonstrate antibiotics, siderophore, hydrogen cyanide, and VOCs can be manipulated to serve as sustainable alternatives to agricultural chemical inputs, which can potentially reduce our overreliance on the current unsustainable methods at a time when population growth, and food demand, is likely to substantially increase.
References
Agisha VN, Kumar A, Eapen SJ, Sheoran N, Suseelabhai R (2019) Broad-spectrum antimicrobial activity of volatile organic compounds from endophytic Pseudomonas putida BP25 against diverse plant pathogens. Biocontrol Sci Technol 29(11):1069–1089
Ahluwalia O, Singh PC, Bhatia R (2021) A review on drought stress in plants: Implications, mitigation and the role of plant growth promoting rhizobacteria. Res Environ Sust. 5:100032
Akhtar MS, Siddiqui ZA (2009) Use of plant growth-promoting rhizobacteria for the biocontrol of root-rot disease complex of chickpea. Australas Plant Pathol 38(1):44–50
Anjaiah V, Cornelis P, Koedam N (2003) Effect of genotype and root colonization in biological control of fusarium wilts in pigeonpea and chickpea by Pseudomonas aeruginosa PNA1. Can J Microbiol 49(2):85–91
Asari S, Matzén S, Petersen MA, Bejai S, Meijer J (2016) Multiple effects of Bacillus amyloliquefaciens volatile compounds: plant growth promotion and growth inhibition of phytopathogens. FEMS Microbiol Ecol 92(6)
Ayyadurai N, Ravindra Naik P, Sreehari Rao M, Sunish Kumar R, Samrat SK, Manohar M, Sakthivel N (2006) Isolation and characterization of a novel banana rhizosphere bacterium as fungal antagonist and microbial adjuvant in micropropagation of banana. J Appl Microbiol 100(5):926–937
Blumer C, Haas D (2000) Mechanism regulation and ecological role of bacterial cyanide biosynthesis. Arch Microbiol 173(3):170–177
Briard B, Bomme P, Lechner BE, Mislin GL, Lair V, Prévost MC, Beauvais A (2015) Pseudomonas aeruginosa manipulates redox and iron homeostasis of its microbiota partner Aspergillus fumigatus via phenazines. Sci Reports 5:8220
Burkhead KD, Schisler DA, Slininger PJ (1994) Pyrrolnitrin production by biological control agent Pseudomonas cepacia B37w in culture and in colonized wounds of potatoes. Appl Environ Microbiol 60(6):2031–2039
Che S, Men Y (2019) Synthetic microbial consortia for biosynthesis and biodegradation: promises and challenges. J Ind Microbiol 46(9-10):1343–1358
Chin-A-Woeng TF, Bloemberg GV, Lugtenberg BJ (2003) Phenazines and their role in biocontrol by Pseudomonas bacteria. New phytol 157(3):503–523
Chincholkar S, Patil S, Sarode P, Rane M (2013) Fermentative production of bacterial phenazines. In: Microbial phenazines. Springer, Berlin, Heidelberg, pp 89–100
Ciancio A, Pieterse CM, Mercado-Blanco J (2016) Harnessing useful rhizosphere microorganisms for pathogen and pest biocontrol. Front Microbiol 7:1620
Clifford JC, Buchanan A, Vining O, Kidarsa TA, Chang JH, McPhail KL, Loper JE (2016) Phloroglucinol functions as an intracellular and intercellular chemical messenger influencing gene expression in Pseudomonas protegens. Environ Microbiol 18(10):3296–3308
Dahiya P, Kaushik R, Sindhu A (2020) An Introduction to Plant Growth Promoting Rhizobacteria, Antifungal Metabolites Biosynthesis using PRPR with reference to Pseudomonas species and It’s other characteristics like Antagonistic and Biocontrolling properties. IRJAS 2:95–100
Dandurishvili N, Toklikishvili N, Ovadis M, Eliashvili P, Giorgobiani N, Keshelava R et al (2011) Broad-range antagonistic rhizobacteria Pseudomonas fluorescens and Serratia plymuthica suppress Agrobacterium crown gall tumors on tomato plants. J Appl Microbiol 110:341–352. https://doi.org/10.1111/j.1365-2672.2010.04891.x
de Boer M, Bom P, Kindt F, Keurentjes JJ, van der Sluis I, Van Loon LC, Bakker PA (2003) Control of Fusarium wilt of radish by combining Pseudomonas putida strains that have different disease-suppressive mechanisms. Phytopathology 93(5):626–632
de Freitas SM, Campos VP, Barros AF, da Silva JC, Pedroso MP, de Jesus SF, Gomes VA, Justino JC (2020) Medicinal plant volatiles applied against the root-knot nematode Meloidogyne incognita. Crop Protection. 130:105057
de Souza JT, Weller D, Raaijmakers JM (2003) Frequency, diversity, and activity of 2, 4-diacetylphloroglucinol-producing fluorescent Pseudomonas spp. in Dutch take-all decline soils. J Phytopathol 93(1):54–63
Deveau A, Bonito G, Uehling J, Paoletti M, Becker M, Bindschedler S, Hacquard S, Hervé V, Labbé J, Lastovetsky OA, Mieszkin S (2018) Bacterial–fungal interactions: ecology, mechanisms and challenges. FEMS Microbiol Rev 42(3):335–352
Dimkić I, Janakiev T, Petrović M, Degrassi G, Fira D (2022) Plant-associated Bacillus and Pseudomonas antimicrobial activities in plant disease suppression via biological control mechanisms-A review. Physiol Mol Plant Pathol 117:101754
Dodd SL, Stewart A (1992) Biological control of Pythium induced damping-off of beetroot (Beta vulgaris) in the glasshouse. N Z J Crop Horti Sci 20(4):421–426
Dumée LF, He L, King PC, Le Moing M, Güller I, Duke M, Hodgson PD, Gray S, Poole AJ, Kong L (2015) Towards integrated anti-microbial capabilities: Novel bio-fouling resistant membranes by high velocity embedment of silver particles. J Mem Sci 475:552–561
Effmert U, Kalderás J, Warnke R, Piechulla B (2012) Volatile mediated interactions between bacteria and fungi in the soil. J Chem Ecol 38(6):665–703
Elkahoui S, Djébali N, Yaich N, Azaiez S, Hammami M, Essid R, Limam F (2015) Antifungal activity of volatile compounds-producing Pseudomonas P2 strain against Rhizoctonia solani. World J Microbiol Biotechnol 31(1):175–185
Farbo MG, Urgeghe PP, Fiori S, Marcello A, Oggiano S, Balmas V, Migheli Q (2018) Effect of yeast volatile organic compounds on ochratoxin A-producing Aspergillus carbonarius and A. ochraceus. Int J Food Microbiol 284:1–10
Fenton AM, Stephens PM, Crowley J, O'callaghan M, O'gara F (1992) Exploitation of gene (s) involved in 2, 4-diacetylphloroglucinol biosynthesis to confer a new biocontrol capability to a Pseudomonas strain. App Environ Microbiol 58(12):3873–3878
Fernando WD, Ramarathnam R, Krishnamoorthy AS, Savchuk SC (2005) Identification and use of potential bacterial organic antifungal volatiles in biocontrol. Soil Biol Biochem 1;37(5):955–64
Fincheira P, Quiroz A (2018) Microbial volatiles as plant growth inducers. Microbiol Res 208:63–75
Freitas CS, Maciel LF, Corrêa dos Santos RA, Costa OM, Maia FC, Rabelo RS, Franco HC, Alves E, Consonni SR, Freitas RO, Persinoti GF (2022) Bacterial volatile organic compounds induce adverse ultrastructural changes and DNA damage to the sugarcane pathogenic fungus Thielaviopsis ethacetica. Environ Microbiol
Garagulia OD, Kiprianova OA, Boiko OI (1974) Antibiotic effect of bacteria from the genus Pseudomonas on phytopathogenic fungi. Mikrobiol Zh
Garbeva P, Weisskopf L (2020) Airborne medicine: bacterial volatiles and their influence on plant health. New Phytologist 226(1):32–43
Giorgio A, De Stradis A, Lo Cantore P, Iacobellis NS (2015) Biocide effects of volatile organic compounds produced by potential biocontrol rhizobacteria on Sclerotinia sclerotiorum. Front Microbiol 6:1056
Gomez-Lama Cabanas C, Legarda G, Ruano-Rosa D, Pizarro-Tobías P, Valverde-Corredor A, Niqui JL, Triviño JC, Roca A, Mercado-Blanco J (2018) Indigenous Pseudomonas spp. strains from the olive (Olea europaea L.) rhizosphere as effective biocontrol agents against Verticillium dahliae: from the host roots to the bacterial genomes. Front Microbiol 9:277
Gotor-Vila A, Teixidó N, Di Francesco A, Usall J, Ugolini L, Torres R, Mari M (2017) Antifungal effect of volatile organic compounds produced by Bacillus amyloliquefaciens CPA-8 against fruit pathogen decays of cherry. Food Microbiol 64:219–225
Gupta C, Dubey R, Maheshwari D (2002) Plant growth enhancement and suppression of Macrophomina phaseolina causing charcoal rot of peanut by fluorescent Pseudomonas. Biol Fertil Soils 35(6):399–405
Guttenberger N, Blankenfeldt W, Breinbauer R (2017) Recent developments in the isolation, biological function, biosynthesis, and synthesis of phenazine natural products. Bioorg Med Chem 25(22):6149–6166
Gutterson NEAL, Ziegle JS, Warren GJ, Layton TJ (1988) Genetic determinants for catabolite induction of antibiotic biosynthesis in Pseudomonas fluorescens HV37a. J Bacteriol 170(1):380–385
Haas D, Défago G (2005) Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat Rev Microbiol:307–319
Hagedorn C (1990) Evaluation of a Pseudomonas fluorescens Strain for Repression of Seedling Disease in Cotton. Va J Sci 41:413
Hamid B, Zaman M, Farooq S, Fatima S, Sayyed RZ, Baba ZA, Sheikh TA, Reddy MS, El Enshasy H, Gafur A, Suriani NL (2021) Bacterial plant biostimulants: a sustainable way towards improving growth, productivity, and health of crops. Sustainability 13(5):2856
Hammer PE, Hill DS, Lam ST, Van Pée KH, Ligon JM (1997) Four genes from Pseudomonas fluorescens that encode the biosynthesis of pyrrolnitrin. Appl Environ Microbiol 63(6):2147–2154
Han SH, Lee SJ, Moon JH, Park KH, Yang KY, Cho BH, Kim KY, Kim YW, Lee MC, Anderson AJ, Kim YC (2006) GacS-dependent production of 2R, 3R-butanediol by Pseudomonas chlororaphis O6 is a major determinant for eliciting systemic resistance against Erwinia carotovora but not against Pseudomonas syringae pv. tabaci in tobacco. Mol. Plant-Microbe Interact 19(8):924–930
Hashem A, Tabassum B, AbdAllah EF (2019) Bacillus subtilis: A plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J Biol Sci 26(6):1291–1297
Hernandez ME, Kappler A, Newman DK (2004) Phenazines and other redox-active antibiotics promote microbial mineral reduction. Appl Environ Microbiol 70(2):921–928
Hernández-León R, Rojas-Solís D, Contreras-Pérez M (2015) Characterization of the antifungal and plant growth-promoting effects of diffusible and volatile organic compounds produced by Pseudomonas fluorescens strains. Biol Control 81:83–92
Hill DS, Stein JI, Torkewitz NR, Morse AM, Howell CR, Pachlatko JP, Ligon JM (1994) Cloning of genes involved in the synthesis of pyrrolnitrin from Pseudomonas fluorescens and role of pyrrolnitrin synthesis in biological control of plant disease. Appl Environ Microbiol 60(1):78–85
Homma Y (1994) Mechanisms in biological control focused on antibiotic pyrrolnitrin. Improving Plant Productivity with Rhizosphere Bacteria, 100–103
Howell CR, Stipanovic RD (1979) Control of Rhizoctonia solani on cotton seedlings with Pseudomonas fluorescens and with an antibiotic produced by the bacterium. J Phytopathol 69(5):480–482
Hunziker L, Bönisch D, Groenhagen U, Bailly A, Schulz S, Weisskopf L (2015) Pseudomonas strains naturally associated with potato plants produce volatiles with high potential for inhibition of Phytophthora infestans. Appl Environ Microbiol 81:821–830
Jaaffar AKM, Parejko JA, Paulitz TC, Welle DM, Thomashow LS (2017) Sensitivity of Rhizoctonia isolates to phenazine-1-carboxylic acid and biological control by phenazine-producing Pseudomonas spp. J Phytopathol 107(6):692–703
Jabborova D, Wirth S, Kannepalli A, Narimanov A, Desouky S, Davranov K, Sayyed RZ, El Enshasy H, Malek RA, Syed A, Bahkali AH (2020) Co-inoculation of rhizobacteria and biochar application improves growth and nutrients in soybean and enriches soil nutrients and enzymes. Agronomy 10(8):114
Jadhav HP, Shaikh SS, Sayyed RZ (2017) Role of hydrolytic enzymes of rhizoflora in biocontrol of fungal phytopathogens: an overview. Rhizotrophs: Plant growth promotion to bioremediation. Springer, pp 183–203
Jani J, Parvez N, Mehta D (2015) Metabolites of Pseudomonads: a new avenue of plant health management. In: New Horizons in insect science: towards sustainable pest management. Springer, New Delhi, pp 61–69
Jousset A, Rochat L, Scheu S, Bonkowski M, Keel C (2010) Predator-prey chemical warfare determines the expression of biocontrol genes by rhizosphere-associated Pseudomonas fluorescens. Appl Environ Microbiol 76(15):5263–5268
Kai M, Effmert U, Berg G, Piechulla B (2007) Volatiles of bacterial antagonists inhibit mycelial growth of the plant pathogen Rhizoctonia solani. Arch Microbiol:351–360
Kanchiswamy CN, Malnoy M, Maffei ME (2015) Chemical diversity of microbial volatiles and their potential for plant growth and productivity. Front Plant Sci 6:151
Keel C, Schnider U, Maurhofer M, Voisard C, Laville J, Burger U, Défago G (1992) Suppression of root diseases by Pseudomonas fluorescens CHA0: importance of the bacterial secondary metabolite 2, 4-diacetylphloroglucinol. Mol Plant Microbe Interact 5(1):4–13
Kenawy A, Dailin DJ, Abo-Zaid GA, Abd Malek R, Ambehabati KK, Zakaria KH, Sayyed RZ, El Enshasy HA (2019) Biosynthesis of antibiotics by PGPR and their roles in biocontrol of plant diseases. In: Plant growth promoting Rhizobacteria for sustainable stress management, vol 1. Springer, Singapore, p 35
Kerr A, Tate ME (1984) Agrocins and the biological control of crown gall. Microbiol Sci 1(1):1–4
Khan A, Sayyed RZ, Seifi S (2019) Rhizobacteria: legendary soil guards in Abiotic stress management. In: Plant Growth promoting Rhizobacteria for sustainable stress management. Singapore, Springer, pp 327–343
Khan N, Ali S, Shahid MA, Mustafa A, Sayyed RZ, Curá JA (2021) Insights into the Interactions among roots, rhizosphere, and rhizobacteria for improving plant growth and tolerance to Abiotic stresses: a review. Cells 10(6):1551
Khare, Mishra J, Arora NK (2018) Multifaceted interactions between endophytes and plant: developments and prospects. Front Microbiol 9:2732
Khoja S, Eltayef KM, Baxter I, Myrta A, Bull JC, Butt T (2021) Volatiles of the entomopathogenic fungus, Metarhizium brunneum, attract and kill plant parasitic nematodes. Biological Control 152:104472
Kloepper JW, Leong J, Teintze M, Schroth MN (1980) Enhanced plant growth by siderophores produced by plant growth-promoting rhizobacteria. Nature 286(5776):885–886
Kramer J, Özkaya Ö, Kümmerli R (2020) Bacterial siderophores in community and host interactions. Nat Rev Microbiol 18(3):152–163
Kraus J, Loper JE (1995) Characterization of a genomic region required for production of the antibiotic pyoluteorin by the biological control agent Pseudomonas fluorescens Pf-5. Appl Environ Microbiol 61(3):849–854
Lagzian A, Saberi Riseh R, Khodaygan P, Sedaghati E, Dashti H (2013) Introduced Pseudomonas fluorescens VUPf5 as an important biocontrol agent for controlling Gaeumannomyces graminis var. tritici the causal agent of take-all disease in wheat. Arch Phytopathol pflanzenschutz 46(17):2104–2116
Lamberth C, Jeanmart S, Luksch T, Plant A (2013) Current challenges and trends in the discovery of agrochemicals. Science 341(6147):742–746
Lanteigne C, Gadkar VJ, Wallon T, Novinscak A, Filion M (2012) Production of DAPG and HCN by Pseudomonas spp. LBUM300 contributes to the biological control of bacterial canker of tomato. J Phytopathol 102(10):967–973
Laursen JB, Nielsen J (2004) Phenazine natural products: biosynthesis, synthetic analogues, and biological activity. Chem Rev 104(3):1663–1686
Le CN, Kruijt M, Raaijmakers JM (2012) Involvement of phenazines and lipopeptides in interactions between Pseudomonas species and Sclerotium rolfsii, causal agent of stem rot disease on groundnut. J Appl Microbiol 112(2):390–403
Lee JY, Moon SS, Hwang BK (2003) Isolation and antifungal and antioomycete activities of aerugine produced by Pseudomonas fluorescens strain MM-B16. Appl Environ Microbiol 69(4):2023–2031
León, M, Yaryura PM, Montecchia MS, Hernández AI, Correa OS, Pucheu NL, Garcia AF (2009) Antifungal activity of selected indigenous Pseudomonas and Bacillus from the soybean rhizosphere. Int J Microbiol 2009
Luh Suriani N, Ngurah Suprapta D, Nazir N, Made Susun Parwanayoni N, Agung Ketut Darmadi A, Andya Dewi D, Sudatri NW, Fudholi A, Sayyed RZ, Syed A, Elgorban AM (2020) A mixture of piper leaves extracts and Rhizobacteria for sustainable plant growth promotion and bio-control of blast pathogen of organic bali rice. Sustainability 12(20):8490
Maksimov IV, Abizgil’Dina RR, Pusenkova LI (2011) Plant growth promoting rhizobacteria as alternative to chemical crop protectors from pathogens. Appl Biochem Microbiol 47(4):333–345
Mavrodi DV, Blankenfeldt W, Thomashow LS (2006) Phenazine compounds in fluorescent Pseudomonas spp. biosynthesis and regulation. Annu Rev Phytopathol 44:417–445
McLoughlin TJ, Quinn JP, Bettermann A, Bookland R (1992) Pseudomonas cepacia suppression of sunflower wilt fungus and role of antifungal compounds in controlling the disease. Appl Environ Microbiol 58(5):1760–1763
Meyer SL, Halbrendt JM, Carta LK, Skantar AM, Liu T, Abdelnabby HM, Vinyard BT (2009) Toxicity of 2, 4-diacetylphloroglucinol (DAPG) to plant-parasitic and bacterial-feeding nematodes. J Nematol 41(4):274
Mujumdar SS, Bashetti SP, Chopade BA (2014) Plasmid pUPI126-encoded pyrrolnitrin production by Acinetobacter haemolyticus A19 isolated from the rhizosphere of wheat. World J Microbiol Biotechnol 30(2):495–505
Mulero-Aparicio A, Cernava T, Turrà D, Schaefer A, Di Pietro A, López-Escudero FJ, Trapero A, Berg G (2019) The role of volatile organic compounds and rhizosphere competence in mode of action of the non-pathogenic Fusarium oxysporum FO12 toward Verticillium wilt. Front Microbiol 10:1808
Mülner P, Bergna A, Wagner P, Sarajlić D, Gstöttenmayr B, Dietel K, Berg G (2019) Microbiota associated with sclerotia of soilborne fungal pathogens–A novel source of biocontrol agents producing bioactive volatiles. Phytobiomes J 3(2):125–136
Mustafa S, Kabir S, Shabbir U, Batool R (2019) Plant growth promoting rhizobacteria in sustainable agriculture: from theoretical to pragmatic approach. Symbiosis 78(2):115–123
Nandi M, Selin C, Brassinga AKC, Belmonte MF, Fernando WD, Loewen PC, De Kievit TR (2015) Pyrrolnitrin and hydrogen cyanide production by Pseudomonas chlororaphis strain PA23 exhibits nematicidal and repellent activity against Caenorhabditis elegans. PloS One 10(4):e0123184
Naz R, Bano A (2014) Effects of allelochemical extracts from medicinal plants on physiological and biochemical mechanisms of maize (Zea mays L.) seedlings. Int J Agr Agri Res 5(2):31–39
Naz R, Bano A (2015) Molecular and physiological responses of sunflower (Helianthus annuus L.) to PGPR and SA under salt stress. Pak J Bot 47(1):35–42
Naz R, Bano A, Wilson NL, Guest D, Roberts TH (2014) Pathogenesis-related protein expression in the apoplast of wheat leaves protected against leaf rust following application of plant extracts. Phytopathology 104(9):933–944
Naz R, Ayub H, Nawaz S, Islam ZU, Yasmin T, Bano A, Wakeel A, Zia S, Roberts TH (2017) Antimicrobial activity, toxicity and anti-inflammatory potential of methanolic extracts of four ethnomedicinal plant species from Punjab, Pakistan. BMC Comp Alt Med 17(1):1–13
Naz R, Nosheen A, Yasmin H, Bano A, Keyani R (2018) Botanical-chemical formulations enhanced yield and protection against Bipolaris sorokiniana in wheat by inducing the expression of pathogenesis-related proteins. Plos One 13(4):e0196194
Naz R, Roberts TH, Bano A, Nosheen A, Yasmin H, Hassan MN, Keyani R, Ullah S, Khan W, Anwar Z (2020) GC-MS analysis, antimicrobial, antioxidant, antilipoxygenase and cytotoxic activities of Jacaranda mimosifolia methanol leaf extracts and fractions. PloS One 15(7):e0236319
Naz R, Batool S, Shahid M, Keyani R, Yasmin H, Nosheen A, Hassan MN, Mumtaz S, Siddiqui MH (2021a) Exogenous silicon and hydrogen sulfide alleviates the simultaneously occurring drought stress and leaf rust infection in wheat. Plant Physiol Biochem 2021 Jun 21
Naz R, Bano A, Nosheen A, Yasmin H, Keyani R, Shah ST, Anwar Z, Roberts TH (2021b) Induction of defense-related enzymes and enhanced disease resistance in maize against Fusarium verticillioides by seed treatment with Jacaranda mimosifolia formulations. Sci Rep 11(1):1–5
Nielsen MN, Sørensen JAN, Fels J, Pedersen HC (1998) Secondary metabolite-and endochitinase-dependent antagonism toward plant-pathogenic microfungi of Pseudomonas fluorescens isolates from sugar beet rhizosphere. Appl Environ Microbiol 64(10):3563–3569
Nielsen TH, Thrane C, Christophersen C, Anthoni U, Sørensen J (2000) Structure production characteristics and fungal antagonism of tensin–a new antifungal cyclic lipopeptide from Pseudomonas fluoresces strain 96.578. J Appl Microbiol 89(6):992–1001
Nielsen TH, Sørensen D, Tobiasen C, Andersen JB, Christophersen C, Givskov M, Sørensen J (2002) Antibiotic and biosurfactant properties of cyclic lipopeptides produced by fluorescent Pseudomonas spp. from the sugar beet rhizosphere. Appl Environ Microbiol 68(7):3416–3423
Nowak-Thompson B, Gould SJ, Loper JE (1997) Identification and sequence analysis of the genes encoding a polyketide synthase required for pyoluteorin biosynthesis in Pseudomonas fluorescens Pf-5. Gene 204(1-2):17–24
Nybroe O, Sørensen J (2004) Production of cyclic lipopeptides by fluorescent pseudomonads. In: Pseudomonas. Springer, Boston, pp 147–172
Olorunleke FE, Kieu NP, De Waele E, Timmerman M, Ongena M, Höfte M (2017) Coregulation of the cyclic lipopeptides orfamide and sessilin in the biocontrol strain Pseudomonas spp CMR 12a. Microbiologyopen 6(5):e00499
Ossowicki A, Jafra S, Garbeva P (2017) The antimicrobial volatile power of the rhizospheric isolate Pseudomonas donghuensis P482. PloS one 12(3):e0174362
Parafati L, Vitale A, Restuccia C, Cirvilleri G (2017) Performance evaluation of volatile organic compounds by antagonistic yeasts immobilized on hydrogel spheres against gray, green and blue postharvest decays. Food Microbiol 63:191–198
Park JY, Oh SA, Anderson AJ, Neiswender J, Kim JC, Kim YC (2011) Production of the antifungal compounds phenazine and pyrrolnitrin from Pseudomonas chlororaphis O6 is differentially regulated by glucose. Lett Appl Microbiol 52(5):532–537
Patil S, Nikam M, Anokhina T, Kochetkov V, Chaudhari A (2017) Multi-stress tolerant plant growth promoting Pseudomonas spp. MCC 3145 producing cytostatic and fungicidal pigment. Biocatal Agric Biotechnol 10:53–63
Popova AA, Koksharova OA, Lipasova VA, Zaitseva JV, Katkova-Zhukotskaya OA, Eremina SI (2014) Inhibitory and toxic effects of volatiles emitted by strains of Pseudomonas and Serratia on growth and survival of selected microorganisms, Caenorhabditis elegans, and Drosophila melanogaster. Biomed Res Int 4. https://doi.org/10.1155/2014/125704
Prasad RM, Sagar BV, Devi GU, Triveni S, Rao SK, Chari DK (2017) Isolation and screening of bacterial and fungal isolates for plant growth promoting properties from tomato (Lycopersicon esculentum Mill.). Int J Curr Microbiol App Sci 6(8):753–761
Priyanka, TA, Kotasthanem AS, Kosharia A, Kushwah R, Zaidi NW, Singh US (2017) Crop specific plant growth promoting effects of ACCd enzyme and siderophore producing and cynogenic fluorescent Pseudomonas. 3 Biotech 7(1)
Raaijmakers JM, De Bruijn I, de Kock MJ (2006) Cyclic lipopeptide production by plant-associated Pseudomonas spp. diversity, activity, biosynthesis, and regulation. Mol Plant Microbe Interact 19(7):699–710
Raaijmakers JM, De Bruijn I, Nybroe O, Ongena M (2010) Natural functions of lipopeptides from Bacillus and Pseudomonas: more than surfactants and antibiotics. FEMS Microbiol Rev 34(6):1037–1062
Rad AS, Esfahanian M, Ganjian E, Tayebi HA, Novir SB (2016) The polythiophene molecular segment as a sensor model for H 2 O, HCN, NH 3, SO 3, and H 2 S: a density functional theory study. J Mol Model 22(6):127
Rajer FU, Wu H, Xie Y, Xie S, Raza W, Tahir HAS, Gao X (2017) Volatile organic compounds produced by a soil-isolate, Bacillus subtilis FA26 induce adverse ultra-structural changes to the cells of Clavibacter michiganensis ssp. sepedonicus, the causal agent of bacterial ring rot of potato. Microbiology 163(4):523–530
Raju R, Kandhasamy S, Nalliappan GK, Natarajan KV, Gandhi K, Chandrasekaran B (2016) Cyclic depsipeptide producing fluorescent pseudomonads exerts antifungal activity against fungal pathogens of maize (Zea mays). Afr J Microbiol Res 10(42):1767–1774
Raza W, Ling N, Yang L, Huang Q, Shen Q (2016) Response of tomato wilt pathogen Ralstonia solanacearum to the volatile organic compounds produced by a biocontrol strain Bacillus amyloliquefaciens SQR-9. Sci Rep 6(1):1–3
Reetha AK, Pavani SL, Mohan S (2014) Hydrogen cyanide production ability by bacterial antagonist and their antibiotics inhibition potential on Macrophomina phaseolina (Tassi.) Goid. Int J Curr Microbiol Appl Sci 3(5):172–178
Reshma P, Naik MK, Aiyaz M, Niranjana SR, Chennappa G, Shaikh SS, Sayyed RZ (2018) Induced systemic resistance by 2, 4-diacetylphloroglucinol positive fluorescent Pseudomonas strains against rice sheath blight
Rodriguez F, Pfender WF (1997) Antibiosis and antagonism of Sclerotinia homoeocarpa and Drechslera poae by Pseudomonas fluorescens Pf-5 in vitro and in planta. J Phytopathol 87(6):614–621
Rohr JR, Barrett CB, Civitello DJ, Craft ME, Delius B, DeLeo GA, Hudson PJ, Jouanard N, Nguyen KH, Ostfeld RS, Remais JV (2019) Emerging human infectious diseases and the links to global food production. Nat Sust 2(6):445–456
Rojas-Solís D, Zetter-Salmón E, Contreras-Pérez M, del Carmen R-GM, Macías-Rodríguez L, Santoyo G (2018) Pseudomonas stutzeri E25 and Stenotrophomonas maltophilia CR71 endophytes produce antifungal volatile organic compounds and exhibit additive plant growth-promoting effects. Biocatal Agric Biotechnol 13:46–52
Rosales Sout AM, Thomashow L, Cook RJ, Mew TW (1995) Isolation and identification of antifungal metabolites produced by rice-associated antagonistic Pseudomonas spp. J Phytopathol 85(9):1028–1032
Sánchez-Fernández RE, Diaz D, Duarte G, Lappe-Oliveras P, Sánchez S, Macías-Rubalcava ML (2016) Antifungal volatile organic compounds from the endophyte Nodulisporium sp. strain GS4d2II1a: a qualitative change in the intraspecific and interspecific interactions with Pythium aphanidermatum. Microb Ecol 71(2):347–364
Savary S, Willocquet L, Pethybridge SJ, Esker P, McRoberts N, Nelson A (2019) The global burden of pathogens and pests on major food crops. Nat Ecol Evol 3(3):430–439
Sayyed RZ, Chincholkar SB, Reddy MS, Gangurde NS, Patel PR (2013) Siderophore producing PGPR for crop nutrition and phytopathogen suppression. In: Bacteria in agrobiology: disease management. Springer, Berlin, Heidelberg, pp 449–471
Schoonbeek HJ, Raaijmakers JM, De Waard MA (2002) Fungal ABC transporters and microbial interactions in natural environments. Mol Plant Microbe Interact 15(11):1165–1172
Schulz-Bohm K, Martín-Sánchez L, Garbeva P (2017) Microbial volatiles: small molecules with an important role in intra-and inter-kingdom interactions. Front Microbiol 8:2484
Shafi J, Tian H, Ji M (2017) Bacillus species as versatile weapons for plant pathogens: a review. Biotechol Biotechnol Equip 31(3):446–459
Shahid MR, Baig DN, Saleem RS, Mali KA, Mehnaz S (2017) Secondary Metabolites production and plant growth promotion by Pseudomonas chlororaphis and P. aurantiaca strains isolated from cactus, cotton, and para grass. J Microbiol Biotechnol 27(3):480–491
Shaikh SS, Sayyed RZ (2015) Role of plant growth-promoting rhizobacteria and their formulation in biocontrol of plant diseases. In: Plant microbes symbiosis: applied facets. Springer, New Delhi, pp 337–351
Shaikh SS, Patel PR, Patel SS, Nikam SD, Rane TU, Sayyed RZ (2014) Production of biocontrol traits by banana field fluorescent Pseudomonads and comparison with chemical fungicide
Shaikh SS, Sayyed RZ, Reddy MS (2016) Plant growth-promoting rhizobacteria: an eco-friendly approach for sustainable agroecosystem. In: Plant, soil and microbes. Springer, Cham, pp 181–201
Shanahan P, O'Sullivan DJ, Simpson P, Glennon JD, O’Gara F (1992) Isolation of 2, 4-diacetylphloroglucinol from a fluorescent pseudomonad and investigation of physiological parameters influencing its production. Appl Environ Microbiol 58(1):353–358
Sharifazizi M, Harighi B, Sadeghi A (2017) Evaluation of biological control of Erwinia amylovora, causal agent of fire blight disease of pear by antagonistic bacteria. Biol Control 104:28–34
Sharifi R, Ryu CM (2016) Are bacterial volatile compounds poisonous odors to a fungal pathogen Botrytis cinerea, alarm signals to Arabidopsis seedlings for eliciting induced resistance, or both? Front Microbiol 7:e196
Sherathia D, Dey R, Thomas M, Dalsania T, Savsani K, Pal KK (2016) Biochemical and molecular characterization of DAPG-producing plant growth-promoting rhizobacteria (PGPR) of groundnut (Arachis hypogaea L). Legume Res Int J 39(4):614–622
Sindhu SS, Suneja S, Dadarwal KR (1997) Plant growth promoting rhizobacteria and their role in crop productivity. Biotechnological approaches in soil microorganisms for sustainable crop production, Sci Pub Jodhpur, pp 149–193
Sonnleitner E, Haas D (2011) Small RNAs as regulators of primary and secondary metabolism in Pseudomonas species. Appl Microbiol Biotechnol 91(1):63–79
Strano CP, Bella P, Licciardello G, Caruso A, Catara V (2017) Role of secondary metabolites in the biocontrol activity of Pseudomonas corrugata and Pseudomonas mediterranea. Eur J Plant Pathol 149(1):103–115
Sulochana MB, Jayachandra SY, Kumar SKA, Dayanand A (2014) Antifungal attributes of siderophore produced by the Pseudomonas aeruginosa JAS-25. J Basic Microbiol 54(5):418–424
Syed-Ab-Rahman SF, Carvalhais LC, Chua ET, Chung FY, Moyle PM, Eltanahy EG, Schenk PM (2019) Soil bacterial diffusible and volatile organic compounds inhibit Phytophthora capsici and promote plant growth. Sci Total Environ 692:267–280
Tahir HAS, Gu Q, Wu H, Niu Y, Huo R, Gao X (2017) Bacillus volatiles adversely affect the physiology and ultra-structure of Ralstonia solanacearum and induce systemic resistance in tobacco against bacterial wilt. Sci Rep 7:e40481
Takeda R (1959) Pseudomonas pigments. III. Derivatives of pyoluteorin. J Agric Chem 23(2):126–130
Thiribhuvanamala G, Rajeswari E, Duraiswamy S (1999). Biological control of stem rot of tomato caused by Sclerotium rolfsii Sacc (No. RESEARCH)
Thomas G, Withall D, Birkett M (2020) Harnessing microbial volatiles to replace pesticides and fertilizers. Microb Biotechnol 13(5):1366–1376
Thomashow LS, Weller DM (1988) Role of a phenazine antibiotic from Pseudomonas fluorescens in biological control of Gaeumannomyces graminis var. tritici. J Bacteriol 170(8):3499–3508
Thomashow LS, Weller DM, Bonsall RF, Pierson LS (1990) Production of the antibiotic phenazine-1-carboxylic acid by fluorescent Pseudomonas species in the rhizosphere of wheat. Appl Environ Microbiol 56(4):908–912
Tilocca B, Balmas V, Hassan ZU, Jaoua S, Migheli Q (2019) A proteomic investigation of Aspergillus carbonarius exposed to yeast volatilome or to its major component 2-phenylethanol reveals major shifts in fungal metabolism. Int J Food Microbiol 306:108265
Tilocca B, Cao A, Migheli Q (2020) Scent of a killer: Microbial volatilome and its role in the biological control of plant pathogens. Front Microbiol 11:41
Tran H, Ficke A, Asiimwe T, Höfte M, Raaijmakers JM (2007) Role of the cyclic lipopeptide massetolide A in biological control of Phytophthora infestans and in colonization of tomato plants by Pseudomonas fluorescens. New Phytol 175(4):731–742
Troppens DM, Moynihan JA, Barret M, O'Gara F, Morrisse JP (2013) Genetics and evolution of 2, 4-Diacetylphloroglucinol Synthesis in Pseudomonas fluorescens. Mol Microbial Ecol Rhizosphere 1:593–605
Ullah H, Yasmin H, Mumtaz S, Jabeen Z, Naz R, Nosheen A, Hassan MN (2020) Multitrait Pseudomonas spp. isolated from monocropped wheat (Triticum aestivum) suppress Fusarium root and crown rot. Phytopathology 110(3):582–592
UNDESAP (United Nations, Department of Economic and Social Affairs, Population Division) (2017) World population prospects: the 2017 revision, key findings and advance tables. Working paper no. ESA/P/WP/248
Velivelli SL, Kromann P, Lojan P, Rojas M, Franco J, Suarez JP, Prestwich BD (2015) Identification of mVOCs from Andean rhizobacteria and field evaluation of bacterial and mycorrhizal inoculants on growth of potato in its center of origin. Microl Ecol 69(3):652–667
Velusamy P, Gnanamanickam SS (2003) Identification of 2, 4-diacetylphloroglucinol production by plant-associated bacteria and its role in suppression of rice bacterial blight in India. Curr Sci 85(9):1270–1273
Voisard C, Keel C, Haas D, Defago G (1989) Cyanide production by Pseudomonas fluorescens helps suppress black root rot of tobacco under gnotobiotic conditions. EMBO J 8:351–358
Weller DM, Landa BB, Mavrodi OV, Schroeder KL, De La Fuente L, Blouin Bankhead S, Thomashow LS (2007) Role of 2, 4-diacetylphloroglucinol-producing fluorescent Pseudomonas spp. in the defense of plant roots. Plant Biol 9(1):4–20
Weller DM, Thomashow LS, Mavrodi, DV, Yang M, Zhang, J (2016) U.S. Patent No. 9,528,115. U.S. Patent and Trademark Office, Washington, DC
Wright SAI, Lindberg A, Gerhardson B (1999) The genetic basis for the production of a fungitoxic compound by the biocontrol agent MA 342. In: Proceeding of 9th International Symposium of Mol Plant Microbe Interact. pp 25–30
Xie S, Zang H, Wu H, Uddin Rajer F, Gao X (2018) Antibacterial effects of volatiles produced by Bacillus strain D13 against Xanthomonas oryzae pv. oryzae. Mol Plant Pathol 19(1):49–58
Yan DD, Wang QX, Li Y (2017) Analysis of the inhibitory effects of chloropicrin fumigation on nitrification in various soil types. Chemosphere 175:459–464
Yasmin H, Nosheen A, Naz R, Keyani R, Anjum S (2019) Regulatory role of rhizobacteria to induce drought and salt stress tolerance in plants. In: Field crops: sustainable management by PGPR. Springer, Cham, pp 279–335
Yasmin H, Naz R, Nosheen A, Hassan MN, Ilyas N, Sajjad M, Anjum S, Gao X, Geng Z (2020) Identification of new biocontrol agent against charcoal rot disease caused by Macrophomina phaseolina in soybean (Glycine max L.). Sustainability 12(17):6856
Zhang Y, Li T, Liu Y, Li X, Zhang C, Feng Z, Peng X, Li Z, Qin S, Xing K (2019) Volatile organic compounds produced by Pseudomonas chlororaphis subsp. aureofaciens SPS-41 as biological fumigants to control Ceratocystis fimbriata in postharvest sweet potatoes. Journal of agricultural and food chemistry 67(13):3702–3710
Zhang D, Yu S, Yang Y, Zhang J, Zhao D, Pan Y, Fan S, Yang Z, Zhu J (2020) Antifungal effects of volatiles produced by Bacillus subtilis against Alternaria solani in potato. Front Microbiol 11:1196
Zhou T, Chen D, Li C, Sun Q, Li L, Liu F, Shen B (2012) Isolation and characterization of Pseudomonas brassicacearum J12 as an antagonist against Ralstonia solanacearum and identification of its antimicrobial components. Microbiol Res 167(7):388–394
Zia MA, Riaz R, Batool A, Yasmin H, Nosheen A, Naz R, Hassan MN (2021) Glucanolytic rhizobacteria associated with wheat-maize cropping system suppress the Fusarium wilt of tomato (Lycopersicum esculentum L). Sci Hort 287:110275
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Naz, R., Khushhal, S., Asif, T., Mubeen, S., Saranraj, P., Sayyed, R.Z. (2022). Inhibition of Bacterial and Fungal Phytopathogens Through Volatile Organic Compounds Produced by Pseudomonas sp.. In: Sayyed, R.Z., Uarrota, V.G. (eds) Secondary Metabolites and Volatiles of PGPR in Plant-Growth Promotion. Springer, Cham. https://doi.org/10.1007/978-3-031-07559-9_6
Download citation
DOI: https://doi.org/10.1007/978-3-031-07559-9_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-07558-2
Online ISBN: 978-3-031-07559-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)