Abstract
The use of nanoparticles (NPs) is expanding in cosmetics, pharmaceutical, agriculture, biomedical, and several other industries. As its application in different fields is increasing, its presence in the environment is also going to increase. Hence, it is important to examine the overall impacts of NPs on the physiology and metabolism of plants. The release of NPs in water and soil will ultimately reach the plants. So, it is important to unveil the mechanism of entry, translocation, and accumulation of different NPs in plant systems. Also, once they reach plants, they affect the physiology and metabolism of plant cells. In this chapter, we summarized the effects of commonly used NPs on the physiology and metabolism of plants. We also included the studies of NPs affecting the microbiome present along with crop plant in soil. By analyzing this compiled information, it is evident that research on the role of NPs in agriculture is expanding and focusing on holistic approaches including the study of secondary metabolites and the effect of NPs on symbiotic fungi (AMF) and bacteria. These studies show the advancement in research related to the role of NPs in agriculture. In future, more studies explaining the physiological and biochemical effects of NPs on plants will lead to the agricultural application of NPs for better plant growth, yield, and adaptation to stress environment.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
In the last two decades, the use of engineered nanoparticles increases in industrial, agricultural, and biomedical sectors (Maurer-Jones et al. 2013). The engineered nanoparticles are of different shapes and sizes that alter their properties from the naturally occurring materials (Auffan et al. 2009; Joshi et al. 2019; Singh et al. 2020). The extensive use of these nanoparticles makes their presence in the environment inevitable. The water bodies and soil are the most likely to get contaminated with excess nanomaterials that can enter indirectly from industrial discharge or directly from the nanomaterials used for agriculture. So, it is crucial to understand the impact of nanoparticles on different plant species before their release in the environment.
Over the last two decades, the application of nanotechnology in the agricultural field has been proven as an effective tool to ensure food security in a sustainable manner (Singh et al. 2021). Nanofertilizers are more efficient for crop production as they ensure high nutrient use efficiency (Zulfiqar et al. 2019). Site-directed, controlled delivery of these nanofertilizers minimizes the use of fertilizers and increases crop production in a highly efficient manner. The emergence of biopesticides offers the protection of plants without much damage to the natural ecosystem as it offers a reduced amount of active ingredient and high efficacy (Kookana et al. 2014). On the other hand, the use of nanosensors can help to detect the microclimate change in the crop field and can alert the farmer before any biotic or abiotic stress hits the threshold and affect crop production (Pérez-de-Luque et al. 2012; Pérez-de-Luque and Hermosín 2013). The development of nanosensors has improved human control over soil and plant health and contributed to precision farming and sustainable agriculture (Chen et al. 2016).
The increasing use of NPs is directly related to their accumulation in the environment. Besides the NPs used in agriculture, various other NPs that are used in different industries also enter in air, water, and soil by different means. Industrial wastes tend to end up in water bodies that increase the chances of NPs contamination. Soil is the ultimate sink of NPs, which receives the NPs directly or indirectly (Remédios et al. 2012). Plants are the base of the ecosystem and directly in contact with soil, water, and air. Any negative effects of nanomaterials on plants can ultimately affect animals and human beings (Judy et al. 2012; Hawthorne et al. 2014; De la Torre Roche et al. 2015; Tangaa et al. 2016). Hence, it is imperative to intensively study the role of NPs on the plant, before their intensive use in the natural environment. Also, it is important to examine the effect of NPs on microflora present in soil because in the nature, growth and development of plants are affected by them. Any positive or negative effect on them can ultimately affect the crop plants. In this chapter, we will describe the effect of various NPs on plants physiology and their effect on plant microbial associations.
2 Entry and Translocation of Nanoparticles (NPs) in Plant System
Nanoparticles are more reactive than their molecular form due to their smaller size and higher surface to volume ratio (Taylor et al. 2015). The uptake of NPs by plants is dependent on several factors including the nature of nanoparticles and their interaction with environment, along with the physiology of plant species (Pérez-de-Luque 2017).
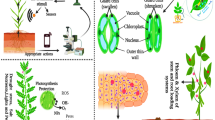
The NPs are generally taken up by plant roots and translocated through vascular tissues to aerial parts of the plant (Fig. 2.1). The uptake and penetration of NPs in plant cells is directly dependent on their size. The small NPs, ranging between 5 and20 nm, can penetrate through the plant cell wall (Dietz and Herth 2011). Few studies reported about the maximum dimensions allowed by plants for the movements and accumulation of NPs inside the plant cells. Usually, 40–50 nm is the size limit of NPs to enter in the plant cells (González-Melendi et al. 2008; Corredor et al. 2009; Sabo-Attwood et al. 2012; Taylor et al. 2015). The morphology of NPs and the means of delivery are also important for their transport in plants. Raliya et al. (2016) had demonstrated that the morphology of gold nanoparticles (nAu) plays a significant role in its translocation in watermelon. The chemical property of NP is an additional factor influencing its interaction with plant cells. The attachment of NPs to the surface of negatively charged plant cell wall is directly dependent on the surface charge of NPs. Zhu et al. (2021) found that the absorption of ZnO onto the leave surface and cell wall of wheat was enhanced by the smaller size and positive charges. In order to understand the uptake, translocation, and accumulation of NPs in plants, standardized laboratory experiments are needed to correlate the physicochemical properties of NPs with their effect on plant tissues.
The NPs of smaller size (3–5 nm) can pass directly through the root epidermis or they penetrate to the plant root along with osmotic pressure and capillary force. The small pores in the cell wall of roots permit the entrance of small-sized NPs while some NPs could form new pores in the root cell wall and enter through it (Du et al. 2011). After crossing the cell wall, NPs enter in extracellular spaces and make their passage to the vascular system. After reaching the vascular bundle, the NPs enter in xylem and they are transferred to shoot and other aerial parts (Ali et al. 2021). When the NPs enter in the cell, they travel from cell to cell through plasmodesmata (Perez-de-Luque 2017). The NPs that cannot enter in the cell are accumulating on the Casparian strip (Wang et al. 2012; Raliya et al. 2016).
NPs can also enter through the stomata or cuticle present on the aerial part of the plant, especially leaves (Larue et al. 2014). The foliar absorption of NPs is dependent on the leaf morphology. The presence of cuticle, wax, trichrome, and leaf exudates are some important factors, which affect the adherence and entrance of NPs from leaves (Larue et al. 2014). The cuticle allows the entrance of NPs smaller than 5 nm while the NPs of 10 nm range can enter through stomata (Ali et al. 2021). The NPs entered through aerial tissues are transported through phloem along with sugar transportation and can accumulate in root, grains, fruits, and young leaves (Wang et al. 2013; Raliya et al. 2016).
The translocation and accumulation of NPs depend upon the plant species and the characteristics of nanomaterial. Accumulation of the same NP was observed in different parts in different plant species (Cifuentes et al. 2010; Zhu et al. 2012). For example, the accumulation of nAu was found in the shoots of Oryza sativa while in Cucurbita pepo and Raphanus raphanistrum nAu accumulation was not found in shoots (Zhu et al. 2012).
3 Physiological Alterations in Plants in Response to Nanoparticles
After entering in plant system, the NPs interact with plants at the cellular and subcellular level, resulting in various morphological and physiological changes. In order to access the effect of NPs, percentage of germination, root elongation, total biomass, and leaf numbers are generally considered physiological parameters (Lee et al. 2010). NPs can affect the plant physiology positively or negatively in a dose-dependent manner and also dependent on plant species.
NPs cover a wide range of materials, but only few of metal and metal oxides nanoparticles are extensively used in various industries. NPs of silver, titanium and its oxide, zinc oxide, iron oxide, aluminum oxide, etc., are the few NPs that are entering in the environment due to their excessive use in different processes. Hence, it is important to understand their physiological impact on the plant. Various methods of NP application, such as foliar application, seed treatment, application in soil, growth media, or in hydroponics, were used to examine the impact of NPs on plant physiology (Table 2.1). Also, various concentrations and sizes of NPs were used to find the threshold level of NPs for different species (Raliya et al. 2016).
Silver nanoparticles (nAg) are extensively used in the preparation of antimicrobial agents, food packaging materials, fabrics, paint, detergents, etc. (Rai et al. 2009; Wijnhoven et al. 2009). Through the industrial waste, nAg can easily enter in the ecosystem and ultimately can affect the plants. It has been observed that the lower concentration of nAg (30 μg/ml) cannot affect the rice, while the higher concentration can affect the cell structure (Mirzajani et al. 2013). Also, it has been observed that the nAg with lower size (˃30 nm) has higher toxic effect on plant and inhibits the growth of root and shoot in different plant species. On contrary, the nAg particles with higher size were able to enhance the growth of root and shoot (Jasim et al. 2017). These results indicate that the penetration and accumulation of nAg in the plant cell can cause a negative impact on plant growth, while its presence in the surrounding has a positive effect on plants.
The higher concentration of NPs can negatively affect plant growth. The increased concentration of NPs can cause stress in plants and in turn, can elevate the production of reactive oxygen species (ROS) as a defense response. There are several studies showing that nAg and nCu/CuO can elevate the level of ROS and various antioxidants in plants (Dimkpa et al. 2013; Jiang et al. 2014; Nair and Chung 2014; Shaw et al. 2014; Cvjetko et al. 2017; Tripathi et al. 2017). These NPs can also affect the expression of genes related to the synthesis of antioxidant. For example, in Arabidopsis thaliana, the expression of various genes related to oxidative stress was changed by applying a high concentration of nCuO (Nair and Chung 2014). The application of NPs can also affect the level of hormones, such as auxin and cytokinin (Yin et al. 2012; Vinkovi’c et al. 2017).
Titanium oxide (TiO2) is known for its photocatalytic property and hence, foliar application of it has shown a positive impact on plants. Foliar application of TiO2 in tomato resulted in an enhanced growth of the plant with an increase in fruit yield and chlorophyll content (Raliya et al. 2016). Similar to nAg and nCu, at high concentration the phytotoxic response of TiO2 was observed in plants (Rafique et al. 2014). Application of TiO2 in colloidal form was observed to inhibit transpiration rate and root growth by physically affecting the root-water interaction (Asli and Neumann 2009).
These studies suggest that the NPs can have impacts on the physiology, morphology, and biochemistry of plants. The effects of NPs on plants can depend upon their size, concentration, and mode of application. It was also observed that the response of the same NPs can differ in genetically diverse plant species (Lee et al. 2008; Van et al. 2016; Wang et al. 2016b). These studies also indicate that the internalization and high accumulation of NPs in plant cells can cause phytotoxic effects in plants, but at lower concentration these NPs can improve plant growth and yield. Application of NPs at lower concentration can help the plant to mitigate various stresses (Jaberzadeh et al. 2013).
4 Molecular and Biochemical Changes in Plants in Response to Cellular Internalization of Nanoparticles
Once the NPs internalize within the plant cells, they start interfering with various metabolic processes. Various reports show that the excess accumulation of NPs in the cell causes oxidative stress and increased production of ROS (Hossain et al. 2015).
NPs present within the cell can enter in the cell organelles. The electron transport chain present on the membrane of mitochondria and chloroplast can be interrupted by the NPs, which ultimately cause oxidative burst (Faisal et al. 2013; Ghosh et al. 2016; Rastogi et al. 2017). This oxidative burst causes the increased production of ROS that ultimately causes the lipid peroxidation, DNA-strand breaks, and cell death (Van Breusegem and Dat 2006; Cvjetko et al. 2017; Saha and Dutta Gupta 2017). The production of ROS also activates the stress regulatory mechanism in plants. The balance between production and scavenging mechanism of ROS determines the level of stress tolerance in plant species (Sharma et al. 2012). The increased level of ROS induces the antioxidant mechanism of cell and as a result, the level of various antioxidant enzymes (peroxidase, catalase, superoxide dismutase (SOD), etc.) and non-enzymatic molecules (ascorbate, glutathione, carotenoids, tocopherols, etc.) increase, to maintain the cellular oxidative state (Sharma et al. 2012).
There are several reports showing an increase in the activities of antioxidant enzymes associated with the application of NPs in plants (Table 2.1). Effects of nickel oxide nanoparticles (nNiO) were examined on tomato. Increased level of ROS, SOD, glutathione, and lipid peroxidation was observed in nNiO-treated tomato plants (Faisal et al. 2013). Application of nAg affected the photosystem II in Spirodela polyrhiza that decreases the fixation of solar energy and promotes the production of ROS in chloroplasts (Jiang et al. 2014). Similarly, nCuO damaged the PSII in rice. The osmotic and oxidative stress was confirmed in nCuO-treated rice plants by the increased level of malondialdehyde, proline, SOD, and ascorbate peroxidase (Costa and Sharma 2016).
Hormonal signaling is liked with the ROS and integrated with the stress signaling pathway. The synthesis of various hormones is differentially regulated by various environmental stresses (O’Brien and Benková 2013). In Arabidopsis, the level of auxin and cytokinin was decreased in shoot, while the level of zeatin, salicylic acid, and abscisic acid (ABA) was increased in presence of nZnO (Vankova et al. 2017). In response to nFe2O3 exposure, an increase in indole acetic acid and ABA was reported in the roots of non-transgenic and transgenic rice (Gui et al. 2015). On contrary, a decrease in phytohormone level was found in seedlings of rice in response to nanocarbon tubes (Hao et al. 2016). These outcomes indicate that the NPs can influence the hormonal synthesis in plants. It has been hypothesized that plants sense the presence of NPs as stress and in response to that they synthesize more stress-related hormones (Vankova et al. 2017). Plant roots are directly in contact with the NPs present in soil/water; hence they are more adversely affected than shoots (Pokhrel and Dubey 2013; Qian et al. 2013; Shaw et al. 2014; Tripathi et al. 2017; Vinkovi’c et al. 2017).
Molecular changes in plants due to NPs are dependent upon their uptake and translocation. NPs of smaller size can penetrate and internalize in the cells by endocytosis. Interaction of NPs with cell organelles like mitochondria, chloroplast, etc., can cause oxidative stress (Fig. 2.1). Various molecular and biochemical changes occur in the plant due to this oxidative stress. The growth and survival of plants depend upon the level of ROS generated in response to NP stress. ROS production at an optimal level can enhance the synthesis of various antioxidant compounds and provide resistance to various stresses. On the other hand, a higher level of ROS can induce apoptosis, cell damage, and ultimately affect plant growth and yield (Foyer and Shigeoka 2011). Hence, intensive studies on the translocation and accumulation of NPs in different plant species at different concentrations are required before their use in the natural environment.
5 Biochemical Alterations in Primary and Secondary Metabolites
In the previous section of this chapter, we mentioned the increase in ROS production as a result of NP-plant interaction. There are several evidences showing ROS-mediated signaling for secondary metabolite production (Simon et al. 2010; Jacobo-Velazquez et al. 2015). Also, ROS can act as a signaling molecule of brassinosteroids (BRs), jasmonic acid (JA), salicylic acid (SA), ethylene, etc., that can modulate the synthesis of secondary metabolites (Wu and Ge 2004; Xia et al. 2009; Maruta et al. 2012; Baxter et al. 2014; Zhang et al. 2016).
Although there is a link between ROS and secondary metabolite production, there are very few reports showing the production of secondary metabolites can be affected by NPs. However, there are few studies related to the alteration in secondary metabolites in plants in response to NPs (Table 2.2). There are many evidences showing the change in SA, JA, BR level, etc., in plants, which are indirectly liked with secondary metabolite production. The level of SA was increased while JA was decreased in Arabidopsis treated with nZnO (Vankova et al. 2017). nAg can reduce the production of ethylene biosynthesis by affecting the production of ACC and ACC oxidase 2 (Syu et al. 2014).
Proteomic studies of some plant species exposed to NPs had shown the change in expression of primary and secondary metabolites. Application of Al2O3, ZnO, and Ag nanoparticles caused alteration in the amount of 104 proteins related to secondary metabolites in soybean roots (Hossain et al. 2016). Application of nZnO increased the proline content, soluble phenols, and phenylalanine ammonia-lyase in tomato (Pejam et al. 2021). In Artemisia annua, the expression of artemisinin was increased by 3.9-fold after the application of nAg in root culture (Zhang et al. 2013). An increase in diosgenin concentration was obtained in fenugreek after nAg application (Jasim et al. 2017). Effects of NPs were examined on lower non-vascular members of plant kingdom. It was found that the application of nTiO2 can increase the secretion of phenolic compound in Arthrospira platensis (cynobacteria) and Haematococcus pluvialis (micoalga) (Comotto et al. 2014).
Although, the studies suggest that NPs are capable of modulating the synthesis of secondary metabolites but the exact mechanism is not clearly understood. Synthesis of secondary metabolites is a defense mechanism of plants against any stress. NPs, after entering in the cell, might interfere with the electron transport system present in mitochondrial and chloroplast membrane, create oxidative stress, and ultimately generate ROS, which can generate secondary metabolite, directly or indirectly.
6 Effect of Nanoparticles in Rhizospheric Environment
Soil is the habitat of diverse microorganisms, which are associated with plants growth and development. Among these soil microorganisms, rhizobia and mycorrhiza are the most important microorganisms that interact with the roots of plants in rhizosphere. Almost all the vascular plant roots establish the symbiotic relationship with arbuscular mycorrhizal fungi (AMF) (Brundrett and Tedersoo 2018). This association helps the plant to access less available minerals in soil, viz. phosphate, improve plant growth, and tolerance to abiotic/biotic stress (Hildebrandt et al. 2007; Smith et al. 2009; Miransari 2010). On the other hand, the symbiotic association of rhizobium with legumes plays a significant role in atmospheric N-fixation. Knowing the importance of rhizobium and mycorrhizal symbiotic association with plants to maintain the health of soil, it is crucial to examine the effect of NPs on these microorganisms.
There is only hand full of studies showing the effect of NPs on symbiotic association of rhizobium and mycorrhiza with plants. Most of the studies show that the presence of NPs has negative impact on rhizobium and mycorrhiza while few show the positive effect of NPs on these microorganisms (Table 2.2). The effect of NPs on mycorrhiza and rhizobium is dependent upon the physicochemical properties of NPs, concentration of NPs, species of fungus and bacteria, and the properties of soil (Tian et al. 2019).
The impact of NPs on mycorrhizal colonization or rhizobial association with plants is dependent on the size and concentration of soil, microbial species, and physicochemical properties of soil. The concentration and size of nanoparticles have a crucial impact on these symbionts. To understand the mechanism by which NPs can affect these microorganisms in the natural environment, it is important to characterize NPs in the possible exposure conditions.
Soil microorganisms are crucial to sustainable agriculture and their association with plants is vital for the growth and development of plants. Hence, it is important to examine the effect of NPs on these microorganisms, individually as well as the effect of NPs on their association with plants. At this point, we know that NPs can affect the symbiotic association of plant-mycorrhiza and legume-mycorrhiza in a positive and negative way. In most of the experiments, unrealistically high concentrations of NPs are used to examine the effect on mycorrhiza (Tian et al. 2019). Also, the experiments with rhizobia were performed in soil-less media (Tian et al. 2019). For a better understanding of the impacts of NPs on soil microorganisms, more studies are required that are closed to natural environmental conditions.
7 Major Concerns
Nowadays, nanoparticles are used in every industry and becoming the part of our lives. The ultimate sink for these NPs is our environment, i.e., air, water, and soil and from there it enters in plants. Hence, it is imperative to observe the impact of NPs on plants. Several studies have reported that the lower concentrations of NPs can positively affect plant growth while its high concentration can cause phytotoxicity. Excessive use of NPs can increase their concentration in the environment. So, it is important to characterize the NPs, their stability in the environment.
The assessment of the impact of NPs on plants is very complicated as it is species-specific (Pérez-de-Luque 2017). Also, the results obtained from experiments done in control conditions, using hydroponics or soil-less media, can differ while using in the field. Crop fields are inhabited by several microorganisms that affect plant growth. Application of NPs in the field can influence the microflora (Table 2.3) so their impact on the plant can differ in the field from the laboratory experiments.
Before the use of NPs in agriculture, experimentation is needed in more realistic conditions. The physicochemical changes of NPs in soil and their long-term effects on mycorrhiza and rhizobia should be investigated. The mechanism of translocation and internalization of NPs in different plant tissues and induction of secondary metabolite pathways by them are still not clearly explained. Studies suggested that NPs can be used to improve the synthesis of bioactive compounds (Table 2.2). This can be a great strategy to enhance the synthesis of commercially important bioactive compounds. But, the potential impact of the NPs as an elicitor of secondary metabolite production needs more studies to assess the overall impact on ecosystem. Furthermore, to bring the NPs in regular agricultural practices, large-scale experiments are required for the better understanding of the NP-plant interaction on the basis of concentration, size, the exposure period of NPs, and its impact on other components of soil.
8 Conclusion
Technological innovation is essential to fill the gap between food production and the exponentially increasing world population. Nanotechnology is one of the promising fields with broader applications. An increase in the use of NPs in industries is ultimately going to end up in our environment. Once they enter in soil, they will affect our crops and ultimately become a part of our food chain. Hence, it is imperative to thoroughly investigate the impacts of commonly used NPs on plants along with their microenvironment. The studies showed that they can affect plant growth and its physiology in positive, negative, or in neutral ways. Also, the effect of the same NPs can vary from species to species.
Similar to plants, the effect of NPs on the microbiome also varies from species to species and can affect positively or negatively in a dose-dependent manner. Before the use of NPs in large-scale crop field, it is crucial to examine the holistic effect of that NP in particular species along with its effect on microbial population present along with that crop. This chapter shows that the study of NPs in agriculture is expanding. Although, more studies are needed to explain the mode of interaction of NPs with different bio-molecules and their impact on gene expressions.
References
M.H. Abd-Alla, N.A. Nafady, D.M. Khalaf, Assessment of silver nanoparticles contamination on faba bean-Rhizobium leguminosarum bv. Viciae-Glomus aggregatum symbiosis: Implications for induction of autophagy process in root nodule. Agric. Ecosyst. Environ. 15(218), 163–177 (2016)
J. Adams, M. Wright, H. Wagner, J. Valiente, D. Britt, A. Anderson, Cu from dissolution of CuO nanoparticles signals changes in root morphology. Plant Physiol. Biochem. 1(110), 108–117 (2017)
M.A. Ahmad, R. Javed, M. Adeel, M. Rizwan, Q. Ao, Y. Yang, Engineered ZnO and CuO nanoparticles ameliorate morphological and biochemical response in tissue culture regenerants of candyleaf (Stevia rebaudiana). Molecules 25(6), 1356 (2020)
A. Ali, S. Mohammad, M.A. Khan, N.I. Raja, M. Arif, A. Kamil, Z.U. Mashwani, Silver nanoparticles elicited in vitro callus cultures for accumulation of biomass and secondary metabolites in Caralluma tuberculata. Artif. Cells Nanomed. Biotechnol 4, 47(1), 715–724 (2019)
S. Ali, A. Mehmood, N. Khan, Uptake, translocation, and consequences of nanomaterials on plant growth and stress adaptation. J. Nanomater. 2021, 6677616 (2021)
S. Asli, P.M. Neumann, Colloidal suspensions of clay or titanium dioxide nanoparticles can inhibit leaf growth and transpiration via physical effects on root water transport. Plant Cell Environ. 32, 577–584 (2009). https://doi.org/10.1111/j.1365-3040.2009.01952.x
M. Auffan, J. Rose, J.Y. Bottero, G.V. Lowry, J.P. Jolivet, M.R. Wiesner, Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat. Nanotechnol. 4(10), 634–641 (2009)
F.P. Baijukya, E. Semu, Effects of Kocide 101® on the bean (Phaseolus vulgaris L.)-Rhizobium symbiosis. Acta Agric. Scandinavica B—Plant Soil Sci. 1 48(3), 175–183 (1998)
A.C. Barrios, C.M. Rico, J. Trujillo-Reyes, I.A. Medina-Velo, J.R. Peralta-Videa, J.L. Gardea-Torresdey, Effects of uncoated and citric acid coated cerium oxide nanoparticles, bulk cerium oxide, cerium acetate, and citric acid on tomato plants. Sci. Total Environ. 1(563), 956–964 (2016)
A. Baxter, R. Mittler, N. Suzuki, ROS as key players in plant stress signalling. J. Exp. Bot. 1, 65(5), 1229–1240 (2014)
M.C. Brundrett, L. Tedersoo, Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 220(4), 1108–1115 (2018)
D.J. Burke, N. Pietrasiak, S.F. Situ, E.C. Abenojar, M. Porche, P. Kraj, Y. Lakliang, A.C. Samia, Iron oxide and titanium dioxide nanoparticle effects on plant performance and root associated microbes. Int. J. Mol. Sci. 16(10), 23630–23650 (2015)
Y.W. Chen, H.V. Lee, J.C. Juan, S.M. Phang, Production of new cellulose nanomaterial from red algae marine biomass Gelidium elegans. Carbohydr. Polym. 151, 1210–1219 (2016)
I.M. Chung, G. Rajakumar, M. Thiruvengadam, Effect of silver nanoparticles on phenolic compounds production and biological activities in hairy root cultures of Cucumis anguria. Acta Biol. Hung. 69(1), 97–109 (2018)
Z. Cifuentes, L. Custardoy, J.M. De la Fuente, C. Marquina, M.R. Ibarra, D. Rubiales, A. Pérez-de-Luque, Absorption and translocation to the aerial part of magnetic carbon-coated nanoparticles through the root of different crop plants. J. Nanobiotechnol. 8(1), 1–8 (2010)
L. Clément, C. Hurel, N. Marmier, Toxicity of TiO2 nanoparticles to cladocerans, algae, rotifers and plants–effects of size and crystalline structure. Chemosphere 1, 90(3), 1083–1090 (2013)
M. Comotto, A.A. Casazza, B. Aliakbarian, V. Caratto, M. Ferretti, P. Perego, Influence of TiO2 nanoparticles on growth and phenolic compounds production in photosynthetic microorganisms. Sci. World J. 28, 2014 (2014)
E. Corredor, P.S. Testillano, M.J. Coronado, P. González-Melendi, R. Fernández-Pacheco, C. Marquina, M.R. Ibarra, J.M. de la Fuente, D. Rubiales, A. Pérez-de-Luque, M.C. Risueño, Nanoparticle penetration and transport in living pumpkin plants: in situ subcellular identification. BMC Plant Biol. 9(1), 1–1 (2009)
P. Cvjetko, A. Milošić, A.M. Domijan, I.V. Vrček, S. Tolić, P.P. Štefanić, I. Letofsky-Papst, M. Tkalec, B. Balen, Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol. Environ. Saf. 1(137), 18–28 (2017)
M.V. Da Costa, P.K. Sharma, Effect of copper oxide nanoparticles on growth, morphology, photosynthesis, and antioxidant response in Oryza sativa. Photosynthetica 1, 54(1), 110–119 (2016)
R. De la Torre Roche, A. Servin, J. Hawthorne, B. Xing, L.A. Newman, X. Ma, G. Chen, J.C. White, Terrestrial trophic transfer of bulk and nanoparticle La2O3 does not depend on particle size. Environ. Sci. Technol. 6, 49(19), 11866–11874 (2015)
K.J. Dietz, S. Herth, Plant nanotoxicology. Trends Plant Sci. 1, 16(11), 582–589 (2011)
C.O. Dimkpa, J.E. McLean, N. Martineau, D.W. Britt, R. Haverkamp, A.J. Anderson, Silver nanoparticles disrupt wheat (Triticum aestivum L.) growth in a sand matrix. Environ. Sci. Technol. 15, 47(2), 1082–1090 (2013)
W. Du, J.L. Gardea-Torresdey, R. Ji, Y. Yin, J. Zhu, J.R. Peralta-Videa, H. Guo, Physiological and biochemical changes imposed by CeO2 nanoparticles on wheat: A life cycle field study. Environ. Sci. Technol. 6 49(19), 11884–11893 (2015)
W. Du, Y. Sun, R. Ji, J. Zhu, J. Wu, H. Guo, TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J. Environ. Monit. 13(4), 822–828 (2011)
M. Faisal, Q. Saquib, A.A. Alatar, A.A. Al-Khedhairy, A.K. Hegazy, J. Musarrat, Phytotoxic hazards of NiO-nanoparticles in tomato: A study on mechanism of cell death. J. Hazard. Mater. 250, 318–332 (2013)
R. Fan, Y.C. Huang, M.A. Grusak, C.P. Huang, D.J. Sherrier, Effects of nano-TiO2 on the agronomically-relevant Rhizobium–legume symbiosis. Sci. Total Environ. 466, 503–512 (2014)
H. Fazal, B.H. Abbasi, N. Ahmad, M. Ali, A.S. Shujait, A. Khan, D.Q. Wei, Sustainable production of biomass and industrially important secondary metabolites in cell cultures of selfheal (Prunella vulgaris L.) elicited by silver and gold nanoparticles. Artif. Cells Nanomed. Biotechnol 47(1), 2553–2561 (2019)
F.Y. Feng, X. Cui, S. He, G. Dong, M. Chen, J. Wang, X. Lin, The role of metal nanoparticles in influencing arbuscular mycorrhizal fungi effects on plant growth. Environ. Sci. Technol. 47(16), 9496–9504 (2013)
C.H. Foyer, S. Shigeoka, Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol. 155(1), 93–100 (2011)
M.R. Ghalamboran, Symbiotic nitrogen fixation enhancement due to magnetite nanoparticles (Doctoral dissertation, Cranfield University) (2011)
F. Ghanati, S. Bakhtiarian, Effect of methyl jasmonate and silver nanoparticles on production of secondary metabolites by Calendula officinalis L (Asteraceae). Trop. J. Pharm. Res. 13(11), 1783–1789 (2014)
M. Ghosh, A. Jana, S. Sinha, M. Jothiramajayam, A. Nag, A. Chakraborty, A. Mukherjee, A. Mukherjee, Effects of ZnO nanoparticles in plants: Cytotoxicity, genotoxicity, deregulation of antioxidant defences, and cell-cycle arrest. Mutat. Res/Genet. Toxicol. Environ. Mutagen 807, 25–32 (2016)
P. González-Melendi, R. Fernández-Pacheco, M.J. Coronado, E. Corredor, P.S. Testillano, M.C. Risueño, C. Marquina, M.R. Ibarra, D. Rubiales, A. Pérez-de-Luque, Nanoparticles as smart treatment-delivery systems in plants: Assessment of different techniques of microscopy for their visualization in plant tissues. Ann. Bot. 101(1), 187–195 (2008)
X. Gui, Y. Deng, Y. Rui, B. Gao, W. Luo, S. Chen, L. Van Nhan, X. Li, S. Liu, Y. Han, L. Liu, Response difference of transgenic and conventional rice (Oryza sativa) to nanoparticles (γFe2O3). Environ. Sci. Pollut. Res. 22(22), 17716–17723 (2015)
Y. Hao, F. Yu, R. Lv, C. Ma, Z. Zhang, Y. Rui, L. Liu, W. Cao, B. Xing, Carbon nanotubes filled with different ferromagnetic alloys affect the growth and development of rice seedlings by changing the C: N ratio and plant hormones concentrations. PLoS One 11(6), e0157264 (2016)
J. Hawthorne, R. De la Torre Roche, B. Xing, L.A. Newman, X. Ma, S. Majumdar, J. Gardea-Torresdey, J.C. White, Particle-size dependent accumulation and trophic transfer of cerium oxide through a terrestrial food chain. Environ. Sci. Technol. 48(22), 13102–13109 (2014)
G.A. Hegazi, W.M. Ibrahim, M.H. Hendawy, H.M. Salem, H.E. Ghareb, Improving α-tocopherol accumulation in Argania spinosa suspension cultures by precursor and nanoparticles feeding. Plant Arch 20, 2431–2437 (2020)
T.A. Hezaveh, F. Rahmani, H. Alipour, L. Pourakbar, Effects of Foliar application of ZnO nanoparticles on secondary metabolite and micro-elements of Camelina (Camelina sativa L.) under salinity stress. J. Stress Physiol. Biochem 16(4), 54–69 (2020)
U. Hidelbrant, M. Regvar, H. Bothe, Asbuscular micorrhiza and heavy metals tolerance. Phytochemisty 68, 138–146 (2007)
Z. Hossain, G. Mustafa, S. Komatsu, Plant responses to nanoparticle stress. Int. J. Mol. Sci. 16(11), 26644–26653 (2015)
Z. Hossain, G. Mustafa, K. Sakata, S. Komatsu, Insights into the proteomic response of soybean towards Al2O3, ZnO, and Ag nanoparticles stress. J. Hazard. Mater. 304, 291–305 (2016)
Y.C. Huang, R. Fan, M.A. Grusak, J.D. Sherrier, C.P. Huang, Effects of nano-ZnO on the agronomically relevant Rhizobium–legume symbiosis. Sci. Total Environ. 497, 78–90 (2014)
M.F. Iannone, M.D. Groppa, M.E. de Sousa, M.B. van Raap, M.P. Benavides, Impact of magnetite iron oxide nanoparticles on wheat (Triticum aestivum L.) development: Evaluation of oxidative damage. Environ. Exp. Bot. 131, 77–88 (2016)
A. Jaberzadeh, P. Moaveni, H.R. Moghadam, H. Zahedi, Influence of bulk and nanoparticles titanium foliar application on some agronomic traits, seed gluten and starch contents of wheat subjected to water deficit stress. Notulae Botanicae, Horti Agrobotanici Cluj-Napoca 41(1), 201–207 (2013)
D.A. Jacobo-Velázquez, M. González-Agüero, L. Cisneros-Zevallos, Cross-talk between signaling pathways: The link between plant secondary metabolite production and wounding stress response. Sci. Rep. 5(1), 1-0 (2015)
M. Jamshidi, F. Ghanati, Taxanes content and cytotoxicity of hazel cells extract after elicitation with silver nanoparticles. Plant Physiol. Biochem. 110, 178–184 (2017)
B. Jasim, R. Thomas, J. Mathew, E.K. Radhakrishnan, Plant growth and diosgenin enhancement effect of silver nanoparticles in Fenugreek (Trigonella foenum-graecum L.). Saudi Pharm. Jl 25(3), 443–447 (2017)
H.S. Jiang, X.N. Qiu, G.B. Li, W. Li, L.Y. Yin, Silver nanoparticles induced accumulation of reactive oxygen species and alteration of antioxidant systems in the aquatic plant Spirodela polyrhiza. Environ. Toxicol. Chem. 33(6), 1398–1405 (2014)
X.X. Jing, Z.Z. Su, H.E. Xing, F.Y. Wang, Z.Y. Shi, X.Q. Liu, Biological effects of ZnO nanoparticles as influenced by arbuscular mycorrhizal inoculation and phosphorus fertilization. Huan jing ke xue= Huanjing kexue 37(8), 3208–3215 (2016)
H. Joshi, P. Choudhary, S.L. Mundra, Future prospects of nanotechnology in agriculture. Int. J. Chem. Stud 7(2), 957–963 (2019)
A. Juarez-Maldonado, H. Ortega-Ortíz, F. Pérez-Labrada, G. Cadenas-Pliego, A. Benavides-Mendoza, Cu Nanoparticles absorbed on chitosan hydrogels positively alter morphological, production, and quality characteristics of tomato. J. Appl. Bot. Food Qual. 24, 89 (2016)
J.D. Judy, J.K. Kirby, C. Creamer, M.J. McLaughlin, C. Fiebiger, C. Wright, T.R. Cavagnaro, P.M. Bertsch, Effects of silver sulfide nanomaterials on mycorrhizal colonization of tomato plants and soil microbial communities in biosolid-amended soil. Environ. Pollut. 206, 256–263 (2015)
J.D. Judy, J.M. Unrine, W. Rao, P.M. Bertsch, Bioaccumulation of gold nanomaterials by Manduca sexta through dietary uptake of surface contaminated plant tissue. Environ. Sci. Technol. 46(22), 12672–12678 (2012)
Ö. Karakaş, Effect of silver nanoparticles on production of indole alkaloids in Isatis constricta. Iranian J. Sci. Technol. Trans. Sci 44, 621–627 (2020)
M. Khajavi, M. Rahaie, A. Ebrahimi, The effect of TiO2 and SiO2 nanoparticles and salinity stress on expression of genes involved in parthenolide biosynthesis in Feverfew (Tanacetum parthenium L.). Caryologia. Int. J. Cytol. Cytosyst. Cytogenet 72(1), 3–14 (2019)
F. Klingenfuss, Testing of TiO2 Nanoparticles on Wheat and Microorganisms in a Soil Microcosm (University of Gothenburg, Gothenburg, 2014)
R.S. Kookana, A.B. Boxall, P.T. Reeves, R. Ashauer, S. Beulke, Q. Chaudhry, G. Cornelis, T.F. Fernandes, J. Gan, M. Kah, I. Lynch, Nanopesticides: Guiding principles for regulatory evaluation of environmental risks. J. Agric. Food Chem. 62(19), 4227–4240 (2014)
C. Krishnaraj, R. Ramachandran, K. Mohan, P.T. Kalaichelvan, Optimization for rapid synthesis of silver nanoparticles and its effect on phytopathogenic fungi. Spectrochim. Acta A Mol. Biomol. Spectrosc. 93, 95–99 (2012)
C. Larue, H. Castillo-Michel, S. Sobanska, L. Cécillon, S. Bureau, V. Barthès, L. Ouerdane, M. Carrière, G. Sarret, Foliar exposure of the crop Lactuca sativa to silver nanoparticles: Evidence for internalization and changes in Ag speciation. J. Hazard. Mater. 264, 98–106 (2014)
N. Le Van, C. Ma, J. Shang, Y. Rui, S. Liu, B. Xing, Effects of CuO nanoparticles on insecticidal activity and phytotoxicity in conventional and transgenic cotton. Chemosphere 144, 661–670 (2016)
C.W. Lee, S. Mahendra, K. Zodrow, D. Li, Y.C. Tsai, J. Braam, P.J. Alvarez, Developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environ. Toxicol. Chem. Int. J 29(3), 669–675 (2010)
W.M. Lee, Y.J. An, H. Yoon, H.S. Kweon, Toxicity and bioavailability of copper nanoparticles to the terrestrial plants mung bean (Phaseolus radiatus) and wheat (Triticum aestivum): Plant agar test for water-insoluble nanoparticles. Environ. Toxicol. Chem. Int. J 27(9), 1915–1921 (2008)
D. Li, Q. An, Y. Wu, J.Q. Li, C. Pan, Foliar application of selenium nanoparticles on celery stimulates several nutrient component levels by regulating the α-linolenic acid pathway. ACS Sustain. Chem. Eng. 8(28), 10502–10510 (2020)
J. Li, J. Hu, C. Ma, Y. Wang, C. Wu, J. Huang, B. Xing, Uptake, translocation and physiological effects of magnetic iron oxide (γ-Fe2O3) nanoparticles in corn (Zea mays L.). Chemosphere 159, 326–334 (2016)
D. Lin, B. Xing, Root uptake and phytotoxicity of ZnO nanoparticles. Environ. Sci. Technol. 42(15), 5580–5585 (2008)
S. Majumdar, J.R. Peralta-Videa, J. Trujillo-Reyes, Y. Sun, A.C. Barrios, G. Niu, J.P. Flores-Margez, J.L. Gardea-Torresdey, Soil organic matter influences cerium translocation and physiological processes in kidney bean plants exposed to cerium oxide nanoparticles. Sci. Total Environ. 569, 201–211 (2016)
T. Maruta, M. Noshi, A. Tanouchi, M. Tamoi, Y. Yabuta, K. Yoshimura, T. Ishikawa, S. Shigeoka, H2O2-triggered retrograde signalling from chloroplasts to nucleus plays specific role in response to stress. J. Biol. Chem. 287(15), 11717–11729 (2012)
M.A. Maurer-Jones, I.L. Gunsolus, C.J. Murphy, C.L. Haynes, Toxicity of engineered nanoparticles in the environment. Anal. Chem. 85(6), 3036–3049 (2013)
M. Miransari, Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biol. 12(4), 563–569 (2010)
F. Mirzajani, H. Askari, S. Hamzelou, M. Farzaneh, A. Ghassempour, Effect of silver nanoparticles on Oryza sativa L. and its rhizosphere bacteria. Ecotoxicol. Environ. Saf. 88, 48–54 (2013)
M.N. Mohaddam, A.H. Sabzevar, Z. Mortazaei, Impact of ZnO and silver nanoparticles on legume-sinorhizobium symbiosis. Adv. Stud. Biol. 9, 83–90 (2017)
J. Moll, A. Gogos, T.D. Bucheli, F. Widmer, M.G. van der Heijden, Effect of nanoparticles on red clover and its symbiotic microorganisms. J. Nanobiotechnol. 14(1), 1–8 (2016)
P.M. Nair, I.M. Chung, Impact of copper oxide nanoparticles exposure on Arabidopsis thaliana growth, root system development, root lignification, and molecular level changes. Environ. Sci. Pollut. Res. 21(22), 12709–12722 (2014)
A. Noori, J.C. White, L.A. Newman, Mycorrhizal fungi influence on silver uptake and membrane protein gene expression following silver nanoparticle exposure. J. Nanopart. Res. 19(2), 66 (2017)
J.A. O'Brien, E. Benková, Cytokinin cross-talking during biotic and abiotic stress responses. Front. Plant Sci. 4, 451 (2013)
C.O. Ogunkunle, E.F. Adegboye, H.K. Okoro, V. Vishwakarma, K. Alagarsamy, P.O. Fatoba, Effect of nanosized anatase TiO2 on germination, stress defense enzymes, and fruit nutritional quality of Abelmoschus esculentus (L.) Moench (okra). Arab. J. Geosci. 13(3), 1–3 (2020)
F. Pejam, Z.O. Ardebili, A. Ladan-Moghadam, E. Danaee, Zinc oxide nanoparticles mediated substantial physiological and molecular changes in tomato. PLoS One 16(3), e0248778 (2021)
A. Pérez-de-Luque, Z. Cifuentes, J.A. Beckstead, J.C. Sillero, C. Ávila, J. Rubio, R.O. Ryan, Effect of amphotericin B nanodisks on plant fungal diseases. Pest Manag. Sci. 68(1), 67–74 (2012)
A. Pérez-de-Luque, M.C. Hermosín, Nanotechnology and its use in agriculture, in Bio-Nanotechnology: A Revolution in Food, Biomedical and Health Sciences, vol. 18, (2013), pp. 383–398
A. Pérez-de-Luque, Interaction of nanomaterials with plants: What do we need for real applications in agriculture? Front. Environ. Sci. 5, 12 (2017)
Z.H. Pinedo-Guerrero, A.D. Hernández-Fuentes, H. Ortega-Ortiz, A. Benavides-Mendoza, G. Cadenas-Pliego, Cu nanoparticles in hydrogels of chitosan-PVA affects the characteristics of post-harvest and bioactive compounds of jalapeño pepper. Molecules 22(6), 926 (2017)
L.R. Pokhrel, B. Dubey, Evaluation of developmental responses of two crop plants exposed to silver and zinc oxide nanoparticles. Sci. Total Environ. 452, 321–332 (2013)
J.H. Priester, Y. Ge, R.E. Mielke, A.M. Horst, S.C. Moritz, K. Espinosa, J. Gelb, S.L. Walker, R.M. Nisbet, Y.J. An, J.P. Schimel, Soybean susceptibility to manufactured nanomaterials with evidence for food quality and soil fertility interruption. Proc. Natl. Acad. Sci. 109(37), E2451–E2456 (2012)
K.P. Priyanka, V.S. Harikumar, K.M. Balakrishna, T. Varghese, Inhibitory effect of TiO2 NPs on symbiotic arbuscular mycorrhizal fungi in plant roots. IET Nanobiotechnol. 11(1), 66–70 (2017)
H. Qian, X. Peng, X. Han, J. Ren, L. Sun, Z. Fu, Comparison of the toxicity of silver nanoparticles and silver ions on the growth of terrestrial plant model Arabidopsis thaliana. J. Environ. Sci. 25(9), 1947–1956 (2013)
R. Rafique, M. Arshad, M.F. Khokhar, I.A. Qazi, A. Hamza, N. Virk, Growth response of wheat to titania nanoparticles application. NUST J. Eng. Sci 7(1), 42–46 (2014)
M. Rai, A. Yadav, A. Gade, Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 27(1), 76–83 (2009)
R. Raliya, C. Franke, S. Chavalmane, R. Nair, N. Reed, P. Biswas, Quantitative understanding of nanoparticle uptake in watermelon plants. Front. Plant Sci. 7, 1288 (2016)
A. Rastogi, M. Zivcak, O. Sytar, H.M. Kalaji, X. He, S. Mbarki, M. Brestic, Impact of metal and metal oxide nanoparticles on plant: A critical review. Front. Chem. 5, 78 (2017)
C. Remédios, F. Rosário, V. Bastos, Environmental nanoparticles interactions with plants: Morphological, physiological, and genotoxic aspects. J. Bot. 2012, 8 (2012)
M.A. Riahi, F. Rezaee, V. Jalali, Effects of alumina nanoparticles on morphological properties and antioxidant system of Triticum aestivum. J. Biotechnol. 254, 1–8 (2012)
C.M. Rico, M.I. Morales, A.C. Barrios, et al. Effect of cerium oxide nanoparticles on the quality of rice (Oryza sativa L.) grains. Journal of Agricultural and Food Chemistry, 61(47):11278–11285 (2013)
T. Sabo-Attwood, J.M. Unrine, J.W. Stone, C.J. Murphy, S. Ghoshroy, D. Blom, P.M. Bertsch, L.A. Newman, Uptake, distribution and toxicity of gold nanoparticles in tobacco (Nicotiana xanthi) seedlings. Nanotoxicology 6(4), 353–360 (2012)
N. Saha, S.D. Gupta, Low-dose toxicity of biogenic silver nanoparticles fabricated by Swertia chirata on root tips and flower buds of Allium cepa. J. Hazard. Mater. 330, 18–28 (2017)
N. Samadi, S. Yahyaabadi, Z. Rezayatmand, Effect of TiO2 and TiO2 nanoparticle on germination, root and shoot Length and photosynthetic pigments of Mentha piperita. Int. J. Plant Soil Sci 3(4), 408–418 (2014)
C.R. Sarabia-Castillo, F. Fernández-Luqueño, TiO2, ZnO, and Fe2O3 nanoparticles effect on Rhizobium leguminosarum- Pisum sativum L. symbiosis. 3rd Biotechnology Summit 2016, Ciudad Obregón, Sonora, Mexico, 144–149 (2016)
R. Sharifi-Rad, S.E. Bahabadi, A. Samzadeh-Kermani, M. Gholami, The effect of non-biological elicitors on physiological and biochemical properties of medicinal plant Momordica charantia L. Iranian J. Sci. Technol. Transact. A: Sci 44(5), 1315–1326 (2020)
P. Sharma, A.B. Jha, R.S. Dubey, M. Pessarakli, Reactive oxygen species, oxidative damage, and antioxidative defence mechanism in plants under stressful conditions. J. Bot. 2012, 217037 (2012)
A.K. Shaw, S. Ghosh, H.M. Kalaji, K. Bosa, M. Brestic, M. Zivcak, Z. Hossain, Nano-CuO stress induced modulation of antioxidative defense and photosynthetic performance of Syrian barley (Hordeum vulgare L.). Environ. Exp. Bot. 102, 37–47 (2014)
C. Simon, M. Langlois-Meurinne, F. Bellvert, M. Garmier, L. Didierlaurent, K. Massoud, S. Chaouch, A. Marie, B. Bodo, S. Kauffmann, G. Noctor, The differential spatial distribution of secondary metabolites in Arabidopsis leaves reacting hypersensitivity to Pseudomonas syringae pv. tomato is dependent on the oxidative burst. J. Exp. Bot. 61(12), 3355–3370 (2010)
O.S. Singh, N.C. Pant, L. Laishram, M. Tewari, R. Dhoundiyal, K. Joshi, C. Pandey, Effect of CuO nanoparticles on polyphenols content and antioxidant activity in Ashwagandha (Withania somnifera L. Dunal). J. Pharmacogn Phytochem. 7(2), 3433–3439 (2018)
A. Singh, V. Rajput, A. Singh, R. Sengar, R. Singh, T. Minkina, Transformation techniques and their role in crop improvements: A global scenario of GM crops, in Policy Issues in Genetically Modified Crops, (2021), pp. 515–542
A. Singh, V. Rajput, et al., Monitoring soil salinity and recent advances in mechanism of salinity tolerance in plants. Biogeosyst. Tech 7(2), 66 (2020)
F.A. Smith, E.J. Grace, S.E. Smith, More than a carbon economy: Nutrient trade and ecological sustainability in facultative arbuscular mycorrhizal symbioses. New Phytol. 182(2), 347–358 (2009)
G. Song, W. Hou, Y. Gao, Y. Wang, L. Lin, Z. Zhang, Q. Niu, R. Ma, L. Mu, H. Wang, Effects of CuO nanoparticles on Lemna minor. Bot. Stud. 57(1), 1–8 (2016)
Y.Y. Syu, J.H. Hung, J.C. Chen, H.W. Chuang, Impacts of size and shape of silver nanoparticles on Arabidopsis plant growth and gene expression. Plant Physiol. Biochemist 83, 57–64 (2014)
S.R. Tangaa, H. Selck, M. Winther-Nielsen, F.R. Khan, Trophic transfer of metal-based nanoparticles in aquatic environments: A review and recommendations for future research focus. Environ. Sci. Nano 3(5), 966–981 (2016)
N.Y. Taran, O.M. Gonchar, K.G. Lopatko, L.M. Batsmanova, M.V. Patyka, M.V. Volkogon, The effect of colloidal solution of molybdenum nanoparticles on the microbial composition in rhizosphere of Cicer arietinum L. Nanoscale Res. Lett. 9(1), 1–8 (2014)
A.F. Taylor, E.L. Rylott, C.W. Anderson, N.C. Bruce, Investigating the toxicity, uptake, nanoparticle formation and genetic response of plants to gold. PLoS One 9(4), e93793 (2015)
H. Tian, M. Kah, K. Kariman, Are nanoparticles a threat to mycorrhizal and rhizobial symbioses? A critical review. Front. Microbiol. 10, 1660 (2019)
D.K. Tripathi, S. Singh, S. Singh, P.K. Srivastava, V.P. Singh, S. Singh, S.M. Prasad, P.K. Singh, N.K. Dubey, A.C. Pandey, D.K. Chauhan, Nitric oxide alleviates silver nanoparticles (AgNps)-induced phytotoxicity in Pisum sativum seedlings. Plant Physiol. Biochem. 110, 167–177 (2017)
F. Van Breusegem, J.F. Dat, Reactive oxygen species in plant cell death. Plant Physiol. 141(2), 384–390 (2006)
R. Vankova, P. Landa, R. Podlipna, I.P. Dobrev, S. Prerostova, L. Langhansova, A. Gaudinova, K. Motkova, V. Knirsch, T. Vanek. ZnO nanoparticle effects on hormonal pools in Arabidopsis thaliana,Science of The Total Environment,Volumes 593–594, 535-542, ISSN 0048-9697, (2017) https://doi.org/10.1016/j.scitotenv.2017.03.160.
C. Vannini, G. Domingo, E. Onelli, B. Prinsi, M. Marsoni, L. Espen, M. Bracale, Morphological and proteomic responses of Eruca sativa exposed to silver nanoparticles or silver nitrate. PLoS One 8(7), e68752 (2013)
T. Vinković, O. Novák, M. Strnad, W. Goessler, D.D. Jurašin, N. Parađiković, I.V. Vrček, Cytokinin response in pepper plants (Capsicum annuum L.) exposed to silver nanoparticles. Environ. Res. 156, 10–18 (2017)
F. Wang, X. Liu, Z. Shi, R. Tong, C.A. Adams, X. Shi, Arbuscular mycorrhizae alleviate negative effects of zinc oxide nanoparticle and zinc accumulation in maize plants–a soil microcosm experiment. Chemosphere 147, 88–97 (2016a)
P. Wang, E. Lombi, F.J. Zhao, P.M. Kopittke, Nanotechnology: A new opportunity in plant sciences. Trends Plant Sci. 21(8), 699–712 (2016b)
Z. Wang, L. Xu, J. Zhao, X. Wang, J.C. White, B. Xing, CuO nanoparticle interaction with Arabidopsis thaliana: Toxicity, parent-progeny transfer, and gene expression. Environ. Sci. Technol. 50(11), 6008–6016 (2016c)
W.N. Wang, J.C. Tarafdar, P. Biswas, Nanoparticle synthesis and delivery by an aerosol route for watermelon plant foliar uptake. J. Nanopart. Res. 15(1), 1–3 (2013)
Z. Wang, X. Xie, J. Zhao, X. Liu, W. Feng, J.C. White, B. Xing, Xylem-and phloem-based transport of CuO nanoparticles in maize (Zea mays L.). Environ. Sci. Technol. 46(8), 4434–4441 (2012)
S.J. Watts-Williams, T.W. Turney, A.F. Patti, T.R. Cavagnaro, Uptake of zinc and phosphorus by plants is affected by zinc fertilizer material and arbuscular mycorrhizas. Plant Soil 376, 165–175 (2014)
S.W. Wijnhoven, W.J. Peijnenburg, C.A. Herberts, W.I. Hagens, A.G. Oomen, E.H. Heugens, B. Roszek, J. Bisschops, I. Gosens, Van, De Meent D., Dekkers, S., Nano-silver–a review of available data and knowledge gaps in human and environmental risk assessment. Nanotoxicology 3(2), 109–138 (2009)
J. Wu, X. Ge, Oxidative burst, jasmonic acid biosynthesis, and taxol production induced by low-energy ultrasound in Taxus chinensis cell suspension cultures. Biotechnol. Bioeng. 85(7), 714–721 (2004)
X.J. Xia, Y.J. Wang, Y.H. Zhou, Y. Tao, W.H. Mao, K. Shi, T. Asami, Z. Chen, J.Q. Yu, Reactive oxygen species are involved in brassinosteroid-induced stress tolerance in cucumber. Plant Physiol. 150(2), 801–814 (2009)
L. Yin, B.P. Colman, B.M. McGill, J.P. Wright, E.S. Bernhardt, Effects of silver nanoparticle exposure on germination and early growth of eleven wetland plants. Sci. Rep. 4, 3782 (2012)
B. Zhang, L.P. Zheng, L.W. Yi, W.J. Wen, Stimulation of artemisinin production in Artemisia annua hairy roots by Ag-SiO2 core-shell nanoparticles. Curr. Nanosci. 9(3), 363–370 (2013)
H. Zhang, A. Li, Z. Zhang, Z. Huang, P. Lu, D. Zhang, X. Liu, Z.F. Zhang, R. Huang, Ethylene response factor TERF1, regulated by ETHYLENE-INSENSITIVE3-like factors, functions in reactive oxygen species (ROS) scavenging in tobacco (Nicotiana tabacum L.). Sci. Rep. 6(1), 1–0 (2016)
L. Zhao, J. Hu, Y. Huang, H. Wang, A. Adeleye, C. Ortiz, A.A. Keller, 1H NMR and GC–MS based metabolomics reveal nano-Cu altered cucumber (Cucumis sativus) fruit nutritional supply. Plant Physiol. Biochem. 110, 138–146 (2017)
J. Zhu, J. Wang, X. Zhan, A. Li, J.C. White, J.L. Gardea-Torresdey, B. Xing, Role of charge and size in the translocation and distribution of zinc oxide particles in wheat cells. ACS Sustain. Chem. Eng. 9(34), 11556–11564 (2021)
Z.J. Zhu, H. Wang, B. Yan, H. Zheng, Y. Jiang, O.R. Miranda, V.M. Rotello, B. Xing, R.W. Vachet, Effect of surface charge on the uptake and distribution of gold nanoparticles in four plant species. Environ. Sci. Technol. 46(22), 12391–12398 (2012)
F. Zulfiqar, M. Navarro, M. Ashraf, N.A. Akram, S. Munné-Bosch, Nanofertilizer use for sustainable agriculture: Advantages and limitations. Plant Sci. 289, 110270 (2019)
Acknowledgments
The research was financially supported by the Russian Foundation for Basic Research, project no. 19-34-60041; project no. 19-05-50097 and the Strategic Academic Leadership Program of the Southern Federal University (“Priority 2030”).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Singh, S., Pradhan, A., Singh, R.K., Singh, A., Minkina, T., Rajput, V.D. (2022). Nanoparticles: Physiology, Chemistry, and Biochemistry. In: Rajput, V.D., Verma, K.K., Sharma, N., Minkina, T. (eds) The Role of Nanoparticles in Plant Nutrition under Soil Pollution. Sustainable Plant Nutrition in a Changing World. Springer, Cham. https://doi.org/10.1007/978-3-030-97389-6_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-97389-6_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-97388-9
Online ISBN: 978-3-030-97389-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)