Abstract
Diabetes mellitus, along with hypertension and dyslipidaemia, are important risk factors for atherosclerotic cardiovascular disease (ASCVD), which is a major cause of mortality and morbidity worldwide. Several pathophysiologic mechanisms are common to both diabetes and hypertension, including oxidative stress due to reactive oxygen species (ROS), activation of the renin-angiotensin-aldosterone system (RAAS), inflammation, blunted insulin-mediated vasodilatation, sympathetic nervous system (SNS) activation, dysfunctional innate and adaptive immune responses, and impaired renal handling of sodium. The organs affected are similar in both essential hypertension and diabetes mellitus, with the vascular tree being the common target. The relative risk of hypertension in populations with versus without diabetes varies from 4.7 to 7.7. In addition, each 20-mmHg increase in systolic blood pressure increases the risk of new-onset type 2 diabetes mellitus by 77%.
The “rule of halves” in diabetes and hypertension states that more than 50% of people with either of these conditions remain undiagnosed, 50% of those in whom the disease is detected are untreated treatment, and 50% of those receiving treatment do not achieve disease control. Thus, the overall proportion of patients with diabetes mellitus or hypertension who are adequately controlled is small. The ideal management of diabetes requires identification and optimisation of the “ABCDEs” of diabetes: A1C (glycosylated haemoglobin; HbA1c), blood pressure, cholesterol (i.e., dyslipidaemia), diet and exercise. Targeting diabetes at all levels of prevention, including primordial prevention, primary prevention, secondary prevention and tertiary prevention, along with control of comorbidities, especially hypertension, and good strategies to promote vascular health, will be important in reducing the microvascular and macrovascular complications of diabetes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
12.1 Introduction
Diabetes mellitus, hypertension and dyslipidaemia are all risk factors for atherosclerotic cardiovascular disease (ASCVD), which is a major cause of mortality and morbidity worldwide. The global prevalence of diabetes is increasing in epidemic proportions, as outlined in an International Diabetes Federation (IDF) report. The IDF estimated that 366 million people worldwide had diabetes mellitus in 2011 and, if current trends continue, 552 million people (or one in ten adults) will have diabetes by 2030 [1]. In contrast, over the period 1975–2015, the prevalence of hypertension decreased in high-income, and some middle-income countries and remained unchanged elsewhere [2]. Hypertension and type 2 diabetes are common comorbidities based on data from studies conducted in Western countries and in Japan [3, 4]. It was estimated that 20% of patients with hypertension had type 2 diabetes and the 50% of patients with type 2 diabetes had hypertension; having either of these conditions increased the risk of developing the other by 1.5–2.0 times [5].
Individuals with both hypertension and diabetes have a four-fold higher risk of developing cardiovascular disease (CVD) compared with age-matched normotensive nondiabetic controls [6]. Diabetes was associated with a two- to four-fold increase in the risk of myocardial infarction (MI), congestive heart failure, peripheral arterial disease, stroke and death in the Framingham Heart Study [7]. A recent analysis of the Framingham data revealed that the population with hypertension at the time of a diabetes mellitus diagnosis had significantly higher rates of mortality from all causes (32 vs 20 events per 1000 person-years) and cardiovascular events (52 vs 31 events per 1000 person-years) compared to normotensive subjects with diabetes mellitus, suggesting that major part of this excess risk is due to coexistent HTN [8].
12.2 Pathophysiology: Converging Pathways
The complex mechanisms involved in the pathogenesis of essential hypertension act on a genetic background. Development of hypertension in genetically vulnerable people is related to increased salt intake, obesity, excess alcohol consumption, mental stress, decreased physical activity and poor sleep [9]. While the majority of patients have essential hypertension that does not have an identifiable cause, it is important to recognise secondary causes of hypertension because they may be curable. Causes of secondary hypertension include primary aldosteronism, pheochromocytoma or renal artery stenosis, among others.
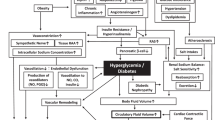
Several pathophysiologic mechanisms coexist in diabetes mellitus and hypertension including oxidative stress due to reactive oxygen species (ROS), pathogenic activation of the renin-angiotensin-aldosterone system (RAAS), inflammation, blunted insulin-mediated vasodilatation, sympathetic nervous system (SNS) activation, dysfunctional innate and adaptive immune responses, and impaired renal handling of sodium [10]. In addition, major pathogenic factors underlying the coexistence of both diabetes mellitus and hypertension are obesity and increased visceral adiposity. Tissue RAAS activation is caused by chronic low-grade inflammation and oxidative stress in adipose tissue, which leads to increased production of angiotensinogen (AGT) and angiotensin II (Ang II) [8]. Ultimately, overexpression of AGT in adipose tissue results in elevated blood pressure (BP) [11]. Thus, AGT and Ang II have local and systemic effects on BP regulation. One of the many deleterious effects of Ang II is mediated via activation of the Ang II type 1 receptor (AT1R) [12]. The production of ROS, reduced insulin metabolic signalling, and proliferative and inflammatory vascular responses culminating in endothelial dysfunction, insulin resistance and hypertension are caused by the activation of AT1R in non-adrenal tissues [12]. Figure 12.1 shows the RAAS pathway and its association with chronic low-grade inflammation in diabetes.
12.3 Coexistence of Hypertension and Diabetes Mellitus
There is a lot of evidence for an increased prevalence of hypertension in people with versus without diabetes mellitus [13]. In a paper published on American Indian and Alaska Native communities to study the prevalence of clinical hypertension and look at its coexistence with diabetes mellitus, 37% of individuals with diabetes were diagnosed with hypertension [14]. In addition, the relative risk of hypertension in those with versus without diabetes varied from 4.7 to 7.7 [11]. Individuals from ethnic minorities in the United Kingdom who were aged 40–59 years and had hypertension were more likely to have diabetes than those without hypertension, and people with diabetes were at much higher risk of having hypertension [15]. Data from a meta-analysis suggest that each 20-mmHg increase in systolic BP increases the risk of new-onset type 2 diabetes mellitus by 77% [16].
The affected organs are similar in both essential hypertension and diabetes mellitus, and the vascular tree is the common target. As a result, coexisting diabetes mellitus and hypertension significantly increase the risk of developing renal failure, retinopathy, coronary heart disease [17], left ventricular hypertrophy [18], congestive heart failure [19] and stroke [20] compared with either condition alone. Rates of microvascular complications, retinopathy and nephropathy are highest in patients with hypertension and diabetes [21, 22]. Lowering BP is therefore of critical importance in patients with diabetes [23, 24]. However, how low the BP target should be is the subject of debate. The “rule of halves” in diabetes and hypertension states that more than 50% of people with either of these conditions remain undiagnosed, 50% of those in whom the disease is detected are untreated treatment, and 50% of those receiving treatment do not achieve disease control. Thus, the overall proportion of patients with diabetes mellitus or hypertension who are adequately controlled is small [25, 26].
12.4 Diagnosis of Hypertension in Diabetes Mellitus
BP should be measured routinely at each clinical visit, and should be measured in both arms to detect any differences. For patients with elevated office BP (>140/90 mmHg), this should be confirmed using multiple readings, including measurements on a separate day, to diagnose hypertension. All patients with hypertension and diabetes are advised to monitor their BP at home. Orthostatic measurement of BP should be performed at the first visit and periodically during follow-up [27, 28].
BP needs to be measured by a trained person [29] with the individual in the seated position, with feet on the floor and arm supported at heart level. Cuff size should be appropriate for the upper arm circumference [30]. To reduce within-patient variability, BP should be measured after 5 min of rest, and 2–3 readings should ideally be taken 1–2 min apart, and BP measurements should be averaged [31].
Autonomic neuropathy can be assessed by checking for postural changes in BP and heart rate, which would require adjustment of BP targets [32]. Home BP monitoring and 24-h ambulatory BP monitoring can provide evidence of white coat hypertension, masked hypertension, or other discrepancies between office and “true” BP [33].
12.5 Blood Pressure Target in DM
BP targets for all patients with diabetes and hypertension must be individualised, taking into account patient preference, cardiovascular risk, and the potential beneficial and adverse effects of antihypertensive medications. A BP target of <130/80 mmHg is appropriate for individuals with diabetes and hypertension at higher cardiovascular risk (existing or 10-year ASCVD risk ≥15%) [34]. BP should be maintained at <140/90 mmHg in individuals with diabetes and hypertension who are at lower risk for CVD (10-year ASCVD risk <15%) [34].
12.6 Glycaemic Goals
Optimal management of diabetes requires identification and optimisation of the “ABCDEs” of diabetes: A1C (glycosylated haemoglobin; HbA1c), BP, cholesterol (i.e., dyslipidaemia), diet and exercise. Glucose goals should be established on an individual basis for each patient, taking both clinical characteristics and the patient’s psycho-socioeconomic circumstances into consideration [35, 36].
The American Diabetes Association (ADA) also recommends individualising glycaemic targets based on patient attitude and expected treatment efforts, risks potentially associated with hypoglycaemia and other adverse events, disease duration, life expectancy, important comorbidities, established vascular complications, resources and support system [37].
Glycaemic recommendations for many non-pregnant adults with diabetes are summarised as follows [37]:
-
1.
HbA1c: <7.0%.
-
2.
Preprandial blood glucose: 80–130 mg/dL.
-
3.
Postprandial blood glucose: <180 mg/dL.
-
4.
Time in range as measured by continuous glucose monitoring should be >70%.
12.7 Prevention
Treating diabetes and hypertension in developing countries such as India presents a real challenge because prevention is a major goal before starting to treat. Effective management is essential to lessen the mortality and morbidity related to both diabetes and hypertension, and there are now better treatment tools than ever before [38,39,40]. There are multiple levels at which hypertension and diabetes can be prevented and treated.
Primordial prevention: This refers to avoidance and prevention of risk factors. Given the overlap between major risk factors for hypertension and diabetes mellitus, an integrated approach to the prevention and control of both can be undertaken. Primordial prevention focuses on health policies that create an environment that promotes healthy behaviours and necessary education programmes. This requires political commitment, involvement by health professionals, and efforts of community leaders and the mass media [41].
The extremely low proportion of patients with hypertension or diabetes mellitus who achieve adequate disease control [25, 26] represents a massive challenge and underlines the need to urgently raise community awareness of these conditions. It is important to detect diabetes and hypertension early, before any organ damage occurs, and provide patients with the best possible and yet affordable treatment. A research paper titled “Improving the prevention and management of chronic disease in low-income and middle-income countries: a priority for primary health care” talks about management of chronic diseases and how this is totally different from acute care, and dependent on several factors, including opportunistic case finding for risk factor assessment, early disease detection and high-risk status identification [42]. All these should be followed by a combination of social, psychological and pharmacological interventions, often in a stepped-care manner, and finally long-term follow-up with frequent monitoring and promotion of adherence to treatment [42].
Primary prevention: The concept of primary prevention refers to a stage where risk factors have already emerged and efforts are needed to prevent the condition in individuals with pre-diabetes or prehypertension. The co-ordination and collaboration of public primary care and private health systems will be required to make many of these recommendations a reality. Primary prevention is very important as a complementary and integrated strategy for several reasons [43]:
-
1.
The immense public health burden associated with diabetes and hypertension justifies action at the population level.
-
2.
Many of the presently available treatments, while valuable, come with the risk of undesirable side effects (e.g. hypoglycaemia, electrolyte imbalance), may have limited efficacy, and may not be accessible over the long term, especially for individuals who have problems accessing medical care or adhering to self-care regimens [44].
-
3.
Lifestyle modifications designed to facilitate the prevention of hypertension and diabetes are likely to have other beneficial effects (e.g. decreased lipid levels, and prevention of heart disease and certain cancers).
-
4.
Many important determinants, such as a balanced diet, weight management and physical activity, are not amenable to implementation or influence by medical care practitioners alone, and are likely to be better addressed by public health efforts and educators in the society.
-
5.
The integration of primary care and public health interventions is needed to address racial/ethnic and socioeconomic disparities because these are multifactorial in origin [45].
-
6.
In addition to lifestyle modification, pharmacotherapy as part of primary prevention strategies in high-risk individuals has been shown to be effective in randomised controlled trials (RCTs) [46].
RCTs of structured lifestyle modification have found that reducing calorie intake plus physical activity leading to modest weight loss reduces the incidence of type 2 diabetes in high-risk adults by 50–70% [47]. The Diabetes Prevention Program (DPP) research trial led by the US National Institutes of Health (NIH) was performed in a US population, but is generalisable to other countries [48]. Participants were randomly assigned to one of three groups:
-
1.
Lifestyle intervention to encourage reduction in calorie intake and increased physical activity.
-
2.
Metformin, the most prescribed oral antidiabetic medication, which is known to improve insulin sensitivity.
-
3.
A placebo control group.
The DPP lifestyle intervention group had an initial body weight reduction of ∼6% after 12 months, decreasing to ∼4% after 3 years. This group had showed an increase in self-reported physical activity (equivalent to brisk walking) from 100 to 190 min per week. This intervention had an impressive effect on the incidence of diabetes, which was decreased by 58% over 4 years compared with the placebo control group. These benefits were seen in men and women across race and ethnic groups and were even greater at older ages [48].
Major trials have also documented the efficacy of lifestyle modification to reduce BP and prevent hypertension in high-risk adults [49]. A prospective study on the contribution of physical activity and body mass index to the risk of hypertension conducted in drug-naïve adults from Finland showed that subjects with higher levels of physical activity had a lower prevalence of hypertension [50]. In combination with increased community detection programmes and compelling epidemiologic data, these trials of primary and secondary prevention led to a series of strong national recommendations for the prevention, detection and treatment of high BP [28].
In the Trials of Hypertension Prevention-phase I (TOHP-I), which enrolled 2182 participants with prehypertension, of the three lifestyle changes proposed (weight reduction, sodium reduction and stress management), weight reduction was the most powerful strategy, producing a net weight loss of 3.9 kg and a BP change of −2.3/−2.9 mmHg [51].
Secondary prevention: Secondary prevention in people with diabetes and hypertension is essential because of the significant financial burden associated with the complications of these conditions. Unless proper treatment is provided to patients with hypertension and/or diabetes, these individuals face a significant burden of complications in the future. Three landmark studies on glycaemic control in diabetes mellitus—the Diabetes Complications and Control Trial (DCCT) [52], the United Kingdom Prospective Diabetes Study (UKPDS) [53] and the Kumamoto Study [54]—have documented the beneficial effects of glycaemic control in preventing microvascular complications. In the UKPDS study there was a 16% reduction in the occurrence of MI but this did not reach statistical significance, indicating that blood sugar control alone is not sufficient to prevent MI. Thus, a multifaceted approach, including controlling glucose, BP, and serum lipid levels, is needed to prevent CVD in patients with diabetes, as shown by the STENO-2 Study [55].
Unfortunately, replicating the international consensus on the treatment and care of patients with diabetes mellitus and hypertension is a huge task in developing nations such as India, especially in rural areas. One major challenge is the availability of trained physicians to screen, identify and treat the conditions in these regions. Screening can be successfully performed by non-physicians, as shown in the Chunampet Rural Diabetes Prevention Project (CRDPP) [56]. The CRDPP is a successful model of diabetes healthcare and prevention to underserved rural areas in developing countries such as India, which provides an example for others on how to deliver quality screening and treatment tools. Large-scale screening for diabetes mellitus and hypertension is possible, but the challenge then becomes long-term follow-up and delivery of pharmacological therapy in a cost-effective manner because the diseases are lifelong.
12.8 Conclusion
The prevalence of diabetes and hypertension is rising in epidemic proportions, and these conditions represent some of the biggest challenges facing healthcare systems in developing countries like India. Diabetes increases the risk of CVD, and this risk is multiplied by coexisting hypertension. All major molecular mechanisms that contribute to the microvascular and macrovascular complications of diabetes, including oxidative stress, inflammation and fibrosis, also cause vascular remodelling and dysfunction in hypertension. Targeting diabetes at all levels of prevention including primordial prevention, primary prevention, secondary prevention and tertiary prevention along with controlling comorbidities, especially hypertension, and good strategies to promote vascular health will be important for reducing the microvascular and macrovascular complications of diabetes. There is an urgent need to improve monitoring and management of risk factors through primary care-linked programmes. Public health policies and large-scale public education have a huge role in reducing the risk of both diabetes and hypertension, and thus of CVD, in the community.
References
Unwin N, Whiting D, Guariguata L, Ghyoot G, Gan D, editors. Diabetes atlas. 5th ed. Brussels: International Diabetes Federation; 2011.
Zhou B, Bentham J, Di Cesare M, Bixby H, Danaei G, Cowan MJ, et al. Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19· 1 million participants. Lancet. 2017;389(10064):37–55.
Kaplan NM. The deadly quartet. Upper-body obesity, glucose intolerance, hypertriglyceridemia, and hypertension. Arch Intern Med. 1989;149(7):1514–20.
Hayashi T, Tsumura K, Suematsu C, Endo G, Fujii S, Okada K. High normal blood pressure, hypertension, and the risk of type 2 diabetes in Japanese men. The Osaka Health Survey. Diabetes Care. 1999;22(10):1683–7.
Tatsumi Y, Ohkubo T. Hypertension with diabetes mellitus: significance from an epidemiological perspective for Japanese. Hypertens Res. 2017;40(9):795–806.
Lastra G, Syed S, Kurukulasuriya LR, Manrique C, Sowers JR. Type 2 diabetes mellitus and hypertension: an update. Endocrinol Metab Clin N Am. 2014;43(1):103–22.
Fox CS. Cardiovascular disease risk factors, type 2 diabetes mellitus, and the Framingham Heart Study. Trends Cardiovasc Med. 2010;20(3):90–5.
Chen G, McAlister FA, Walker RL, Hemmelgarn BR, Campbell NR. Cardiovascular outcomes in Framingham participants with diabetes: the importance of blood pressure. Hypertension. 2011;57(5):891–7.
Liang M. Epigenetic mechanisms and hypertension. Hypertension. 2018;72(6):1244–54.
Sowers JR, Whaley-Connell A, Hayden MR. The role of overweight and obesity in the cardiorenal syndrome. Cardiorenal Med. 2011;1(1):5–12.
Massiéra F, Bloch-Faure M, Ceiler D, Murakami K, Fukamizu A, Gasc JM, et al. Adipose angiotensinogen is involved in adipose tissue growth and blood pressure regulation. FASEB J. 2001;15(14):2727–9.
Mehta PK, Griendling KK. Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am J Physiol Cell Physiol. 2007;292(1):C82–97.
Berraho M, El Achhab Y, Benslimane A, El Rhazi K, Chikri M, Nejjari C. Hypertension and type 2 diabetes: a cross-sectional study in Morocco (EPIDIAM Study). Pan Afr Med J. 2012;11:52.
Broussard BA, Valway SE, Kaufman S, Beaver S, Gohdes D. Clinical hypertension and its interaction with diabetes among American Indians and Alaska Natives. Estimated rates from ambulatory care data. Diabetes Care. 1993;16(1):292–6.
Cappuccio FP, Barbato A, Kerry SM. Hypertension, diabetes and cardiovascular risk in ethnic minorities in the UK. Br J Diabetes Vasc Dis. 2003;3(4):286–93.
Emdin CA, Anderson SG, Woodward M, Rahimi K. Usual blood pressure and risk of new-onset diabetes: evidence from 4.1 million adults and a meta-analysis of prospective studies. J Am Coll Cardiol. 2015;66(14):1552–62.
Assmann G, Schulte H. The Prospective Cardiovascular Munster (PROCAM) study: prevalence of hyperlipidemia in persons with hypertension and/or diabetes mellitus and the relationship to coronary heart disease. Am Heart J. 1988;116:1713–24.
Somaratne JB, Whalley GA, Poppe KK, ter Bals MM, Wadams G, Pearl A, et al. Screening for left ventricular hypertrophy in patients with type 2 diabetes mellitus in the community. Cardiovasc Diabetol. 2011;10:29.
Govind S, Saha S, Brodin LA, Ramesh SS, Arvind SR, Quintana M. Impaired myocardial functional reserve in hypertension and diabetes mellitus without coronary artery disease: searching for the possible link with congestive heart failure in the myocardial Doppler in diabetes (MYDID) study II. Am J Hypertens. 2006;19(8):851–7; discussion 858.
Grossman E, Messerli FH, Goldbourt U. High blood pressure and diabetes mellitus: are all antihypertensive drugs created equal? Arch Intern Med. 2000;160(16):2447–52.
Lea JP, Nicholas SB. Diabetes mellitus and hypertension: key risk factors for kidney disease. J Natl Med Assoc. 2002;94(8 Suppl):7S–15S.
Knowler WC, Bennett PH, Ballintine EJ. Increased incidence of retinopathy in diabetics with elevated blood pressure. A six-year follow-up study in Pima Indians. N Engl J Med. 1980;302(12):645–50.
Parving HH. Hypertension Optimal Treatment (HOT) trial. Lancet. 1998;352(9127):574–5.
Hansson L, Zanchetti A, Carruthers SG, Dahlöf B, Elmfeldt D, Julius S, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet. 1998;351(9118):1755–62.
Deepa R, Shanthirani CS, Pradeepa R, Mohan V. Is the ‘rule of halves’ in hypertension still valid? Evidence from the Chennai Urban Population Study. J Assoc Physicians India. 2003;51:153–7.
Ranjit Unnikrishnan I, Anjana RM, Mohan V. Importance of controlling diabetes early—the concept of metabolic memory, legacy effect and the case for early insulinisation. J Assoc Physicians India. 2011;59(Suppl):8–12.
Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur Heart J. 2018;39:3021–104.
Whelton PK, Carey RM, Aronow WS, et al. 2017ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/vASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults. Hypertension. 2018;71:e13–e115.
Pickering TG, Hall JE, Appel LJ, Falkner BE, Graves J, Hill MN, et al.; Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension. 2005;45(1):142–61.
Powers BJ, Olsen MK, Smith VA, Woolson RF, Bosworth HB, Oddone EZ. Measuring blood pressure for decision making and quality reporting: where and how many measures? Ann Intern Med. 2011;154(12):781–8.
Shah SN, Core Committee Members: Billimoria AB, Mukherjee S, Kamath S, Munjal YP, Maiya M, Wander GS, Mehta N. Indian guidelines on management of hypertension (I.G.H) – IV 2019. Suppl J Assoc Phys India (JAPI). 2019;67(9):1–48.
Shibao C, Gamboa A, Diedrich A, Biaggioni I. Management of hypertension in the setting of autonomic failure: a pathophysiological approach. Hypertension. 2005;45(4):469–76. https://doi.org/10.1161/01.HYP.0000158835.94916.0c. Epub 2005 Feb 28. PMID: 15738343.
de Boer IH, Bangalore S, Benetos A, Davis AM, Michos ED, Muntner P, et al. Diabetes and hypertension: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(9):1273–84.
American Diabetes Association. 10. cardiovascular disease and risk management: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(Suppl 1):S111–34.
Garber AJ, Abrahamson MJ, Barzilay JI, Blonde L, Bloomgarden ZT, Bush MA, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2017 executive summary. Endocr Pract. 2017;23(2):207–38.
American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(Suppl 1):S66–76.
Handelsman Y, Bloomgarden ZT, Grunberger G, Umpierrez G, Zimmerman RS, Bailey TS, et al. American association of clinical endocrinologists and American College of Endocrinology—clinical practice guidelines for developing a diabetes mellitus comprehensive care plan—2015. Endocr Pract. 2015;21:1–87.
Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, et al.; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353(25):2643–53.
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837–53.
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med. 2003;348(5):383–93.
Falkner B, Lurbe E. Primordial prevention of high blood pressure in childhood: an opportunity not to be missed. Hypertension. 2020;75(5):1142–50.
Beaglehole R, Epping-Jordan J, Patel V, Chopra M, Ebrahim S, Kidd M, et al. Improving the prevention and management of chronic disease in low-income and middle-income countries: a priority for primary health care. Lancet. 2008;372(9642):940–9.
Krousel-Wood MA, Muntner P, He J, Whelton PK. Primary prevention of essential hypertension. Med Clin North Am. 2004;88(1):223–38.
Whelton PK, He J, Appel LJ, Cutler JA, Havas S, Kotchen TA, et al.; National High Blood Pressure Education Program Coordinating Committee. Primary prevention of hypertension: clinical and public health advisory from the National High Blood Pressure Education Program. JAMA. 2002;288(15):1882–8.
National Health Care Disparities Report. 2010. https://fodh.phhp.ufl.edu/files/2011/05/AHRQ-disparities-2010.pdf.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403.
Klein S, Sheard NF, Pi-Sunyer X, Daly A, Wylie-Rosett J, Kulkarni K, et al.; American Diabetes Association; North American Association for the Study of Obesity; American Society for Clinical Nutrition. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: rationale and strategies. A statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Am J Clin Nutr. 2004;80(2):257–63.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403.
Whelton PK, Appel LJ, Espeland MA, Applegate WB, Ettinger WH Jr, Kostis JB, et al. Sodium reduction and weight loss in the treatment of hypertension in older persons: a randomized controlled trial of nonpharmacologic interventions in the elderly (TONE). TONE Collaborative Research Group. JAMA. 1998;279(11):839–46.
Hu G, Barengo NC, Tuomilehto J, Lakka TA, Nissinen A, Jousilahti P. Relationship of physical activity and body mass index to the risk of hypertension: a prospective study in Finland. Hypertension. 2004;43(1):25–30.
The Trials of Hypertension Prevention Collaborative Research Group. The effects of nonpharmacologic interventions on blood pressure of persons with high normal levels. Results of the trials of hypertension prevention, phase I. JAMA. 1992;267:1213–20.
Nathan DM, Genuth S, Lachin J, Cleary P, Crofford O, Davis M, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977–86.
Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352(9131):837–53.
Shichiri M, Kishikawa H, Ohkubo Y, Wake N. Long-term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care. 2000;23(Suppl 2):B21–9.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358(6):580–91.
Mohan V, Deepa M, Pradeepa R, Prathiba V, Datta M, Sethuraman R, et al. Prevention of diabetes in rural India with a telemedicine intervention. J Diabetes Sci Technol. 2012;6(6):1355–64.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Aswin, M., Mohan, V. (2022). Diabetes and Hypertension: What Is the Connection?. In: Ram, C.V.S., Teo, B.W.J., Wander, G.S. (eds) Hypertension and Cardiovascular Disease in Asia. Updates in Hypertension and Cardiovascular Protection. Springer, Cham. https://doi.org/10.1007/978-3-030-95734-6_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-95734-6_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-95733-9
Online ISBN: 978-3-030-95734-6
eBook Packages: MedicineMedicine (R0)