Abstract
On this planet, crops and insects are two predominating classes as living entities. Among all biodiverse systems, plants hold the foremost commodious part; however, insects are the most plausible class. Both kingdoms have mutualistic associations with one another living divorced life. Green plants are the essential makers of food, and all creatures are heterotrophs which depend straightforwardly or by implication on plant-derived food. Thus, almost three-fourths of all angiosperms require the administrations of creepy crawly pollinators. The entomophilic blossoming plants and their flower pollinators are subsequently addressed as the most obvious and broad material illustration of mutualism among living organic entities. However, a wide assortment of phytophagous insects likewise prospers, enhances, and supports these plants. Thus, plants have advanced a bewildering cluster of morphological and biochemical hindrances for security against insects and different herbivores. Transformative associations among plants and bugs may have added to the expanded biodiversity and achievement of both these gatherings. The investigation of these interrelationships, as illustrated in this chapter, is of incredible importance for future crop development. The advancement of insect-resistant and safe cultivars of plants and progress in integrated pest management need a mind-boggling comprehension of insect plant connections. Best-in-class strategies like freak investigation, metabolomics, RNAi, and proteomics created during the recent 30 years have been useful in giving improved understanding into these interrelationships.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1.1 Introduction
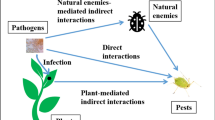
The evolutionary interactions between plants and insect pests are asymmetric: the biochemical and structural assortment of the angiosperms offers a profusion of niches for the evolutionary radiation (cladogenesis) of insects, while insects do not affect plant evolution or may create anagenic adaptations within the plants. So, plants have not developed resistance to insects, but the insects have, or were, evolved ever since their emergence, especially in food selection. Host plant selection is usually a behavioural procedure which is managed basically by chemoreception system. Consequently, the emergence of specific insect/host plant relations mostly is the consequence of evolutionary conversations in the insects’ chemosensory processes. The adaptations to the food preference of the new host plant may be a minor procedure. The ‘plant realm’ and also the ‘class Insecta’ are addressed as the two prevailing gatherings of living organic entities, as far as the bounty of species just as in the measure of biomass (Schoonhoven et al. 2005). Thus, a lion’s share of 300,000 plant species needs bee pollinators for multiplication (Tanda 2019a, b, c, 2020, 2021a, b, c, d, e, f, g). Vivid, scented blossoms and flower nectarines were created by plants for alluring the animal pollinators. The blossom life structures guaranteed that while taking care of, the pollinators additionally got the pollen dust (Kearns et al. 1998). Therefore, to forestall over-abuse, plants have likewise developed a structural and biochemical hindrances for assurance against insects and different herbivores. While a portion of these boundaries are combined by plants paying little heed to the presence of constitutive protection, numerous others are created uniquely because of induced defences in the plant system. Just those insect species, which can beat these impediments at least one plant animal categories by shirking, detoxification, and so forth, can get to that plant species as food. Insects which harm the monetarily significant plants have been named as insect pests. The significant mutualistic and opposing communications among plants and insects are presented hereunder.
1.2 Insect Pollinators and Angiosperm Mutualism
The most clear and broad material illustration of mutualism is between bee-pollinated blossoming plants and their pollinators (Tanda 2019a, b, c, 2020, 2021a, b, c, d, e, f, g). Almost 80% of all blooming plants are bisexual and bear blossoms with stamen and pistils in a same bloom. This advances self-fertilization and subsequently inbreeding occurs. The plants avoid self-fertilization either by separating the sexes in time and space (differences in the timing of maturation) or by self-incompatibility. Both mechanisms promote cross-pollination, which is assisted by various agencies, e.g. wind, water, animals, etc. More than three-fourths of all blooming plants are completely or mostly pollinated by insects (Faegri and Pijl 1971; Tanda 2019a, b, c). The monetary estimation of bug pollinators is colossal. Most of the important plants such as oilseeds, pulses, fruits, vegetables, nuts, spices, and ornamentals (Hill 1997; Atwal 2000) show improved yields with animal pollination (Klein et al. 2007). It has been estimated that animal pollination has an economic value of €153 billion annually, which is nearly one-tenth of the global agricultural production (Galai et al. 2009). Some of the widely accepted estimates of the number of angiosperms pollinated by animals vary from 67 to 96% of all angiosperm species (Axelrod 1960; Nabhan and Buchmann 1997). Ollerton et al. (2011) observed that these estimates are not based on firm data. They compiled data on published and unpublished community-level surveys of plant-pollinator interactions and concluded that proportion of animal-pollinated species was 78% in temperate-zone communities and 94% in tropical communities, with a global mean of 87.5% of all flowering plants. The pollinators benefit from rewards in the form of nectar and pollen. Both are nutrient-rich foods with nectar containing 50% sugars and pollen 15–60% proteins and other essential elements (Proctor et al. 1996; Roulston et al. 2000). Together, they provide nourishment for the bees, which are the most important among insect pollinators (Schoonhoven et al. 2005). The entomophilous flowering plants and the pollinating insects constitute an example par excellence of mutualism. However, the degree of mutualism varies among various plant-pollinator combinations (Schoonhoven et al. 2005). In some cases, there is an obligate mutualism, and a species of plant can only be pollinated by a single species of pollinator, which depends on it for food (Tanda 2019a, b, c, 2020, 2021a, b, c, d, e, f). Figs (Ficus spp.) are reliant upon the fig wasps for pollination (Wiebes 1979). Different species of fig are cross-pollinated by a particular species of wasp, as the pollination in Ficus carica Linnaeus is carried out by the fig wasp, Blastophaga psenes (Linnaeus) (Ramirez 1970). Similarly an obligate relationship is found in yucca moths (Prodoxidae) and yucca plants (Agavaceae). The yucca moths are the alone pollinators of yucca plants and lay eggs in the locule of the ovary so that the hatching caterpillars can start feeding on the seeds (Pellmyr and Krenn 2002). Darwin described that the Christmas orchid, Angraecum sesquipedale Thouars, had a long green nectary and forecasted that there must be a gigantic moth species with long proboscis to suck the nectar from the long flower nectary. Later on, Rothshild and Jordan observed the Morgan’s sphinx moth, Xanthopan morganii Walker, with an enlarged proboscis as the only pollinator of A. sesquipedale, native to Madagascar (Kritsky 2001). This type of mutualistic relationship in plant pollinators is not very common. Burkle and Alarcon (2011) reported that plant-pollinator associations are largely distributed with a high degree of annual turnover of pollinator populations and the significance of an insect pollinator may differ for pollination services for the same plant under dynamic climatic conditions.
Doubtlessly, various insect pollinators have served the development of angiosperms differently, and fossil reports describe that pollination mechanism was found around 250 Myr ago (Labandeira 2013). The old flowering plants might be cross-pollinated by the wind birds or other animals. Due to the benefits of insect pollinators, its significance enhanced in the future (Cox 1991; Crepet et al. 1991). Entomophilic plants possess various flower sizes, shapes, colours, and fragrances which may have been resolved by the needs of flower pollinator species. The pollen grains may have a shape or sticky materials which help to adhere to the body of the insect . The hairy body parts of the insects also assist in the spread of the pollen. Foxglove, (Digitalis purpurea Linnaeus), flowers cross-pollinated by the bumblebees look like a bell; however, the flowers of Calopheria spp. fertilized by the butterfly have tubular corolla, adapted for the enlarged proboscis (Schoonhoven et al. 2005). Additionally, the later consists of maximum amino acids than blossoms nourished on by flies (Baker and Baker 1986). Few brightly coloured flowers develop sterile ‘reward anthers’ to allure pollinators (Nepi et al. 2003). The flowers of orchid Mirror of Venus, Ophrys speculum Link, mimic the virgin female wasps of their pollinator, Dasyscolia ciliata (Fabricius), by discharging female sex pheromone to attract the male wasps. The male wasps attempt to mate with the blooms and operate as pollination agent (Ayasse et al. 2003). At present, hymenopterans are the predominating class working in plant pollination; however, other insect groups are too crucial in the history of pollination. Basically the beetles and flies pollinate the basal flowers (Thien et al. 2000). Honeybees have adapted themselves to a flower nutrition (Atwal 2000) and like pollen grains in spite of impermeable cuticle (Velthius 1992). Honeybees show flower constancy by foraging the blooms of a single plant . It enhances the effectiveness of a pollinator and assists in plant reproductive isolation. The insects’ ability to recall amalgamations of flower odours and colours contributes a crucial role in flower fidelity. Honeybees have been observed to have the capacity to differentiate 700 various floral fragrances (Schoonhoven et al. 2005).
1.3 Antagonistic Plant-Pest Relationships
Insects are the most assorted and massively effective living beings on this globe and attack plants for food. Species in Lepidoptera, Orthoptera, Phasmida, or predominantly Hemiptera and Thysanoptera are herbivorous; however, Coleoptera, Hymenoptera, and Diptera are partially herbivorous but mostly carnivorous (Schoonhoven et al. 2005). Several insects live on all parts of plants; however, solid feeders are defoliators or borers, and others suck the sap (aphids, jassids), lessen plant vigour, and act as vectors such as whitefly. Mostly insects are specific in their food plant choice. Monophagous insects live on a single or a few related plant species, but oligophagous insects feed on a number of plants. Polyphagous species feed on various plants belonging to many families for survival (Panda and Khush 1995). Studies on herbivorous insects have demonstrated that one-tenth of these insects are capable of living on plants of more than three families. Each insect host range is determined by structural, biochemical , and ecological elements. Excluding Orthoptera, all other herbivores are highly consisted of specific species living on specialized species of plant (Schoonhoven et al. 2005). Bruce (2015) described that the herbivores get converted to specialize over time; however, few of polyphages carry on as crop pests. Insects can identify and respond to host signals for nourishing and egg-laying. In spite of this, antagonistic connections between plants and phytophagous insects continue to work, as herbivory has been seen to enhance the plant development and strength in few instances (Owen 1980; Vail 1994; Sadras and Felton 2010). Production minimizes due to insects; however, there are instances of enhanced yield reported in insect-attacked in comparison to insect-unattacked crop plants (Harris 1974). The automatic reply to damage may in few plants more than counterbalance the damage done. It is based on how plants answer to damage by insects or other herbivores. Sesame tissues cultured alone or with okra suppressed egg hatch and penetration of roots by juveniles, delayed adult development, and encouraged development of males in Meloidogyne incognita. Gall formation was inhibited on excised roots of okra by co-culturing with sesame. Sesame callus reduced penetration, discouraged nematode build-up in okra, and caused an increase in numbers of males showing antagonism of sesame to root-knot nematode on okra (Tanda and Atwal 1988; Tanda et al. 1988, 1989).
1.3.1 Plant Defence Mechanisms
Plants are motionless and have to protect themselves against herbivores. Many plants in natural environments exhibit small or no evident attack despite of large populations of plant feeders. Insects feed about 10% of all plant biomass annually (Barbosa and Schulz 1987; Arora and Sandhu 2017). Plants have developed a large range of structural and biochemical attributes to save from herbivores. Contrastingly, insect injury is more in cropping area as many of these attributes have been strayed while breeding more palatable and tasty plants and outyielding the crop genotypes used traditionally. There is a demand to investigate such plant defence mechanisms to manipulate them in agro-industry (see Chap. 10).
1.3.2 Plant Structural Defence Mechanisms
1.3.2.1 Epicuticular Waxes
The epicuticle protects the plant surface by waxes against desiccation, herbivore , and disease attack. Thickness, structure, and wax coating number may be different in plants giving rise to variations in the total plant dry weight. These wax layers work as resistance to the insect pest attack (Jeffree 1986), and the mechanoreceptor and chemoreceptor present on the tarsi of insects and mouth parts get negative tactile and chemical stimuli. In Brassicaceae, leaf epicuticular wax ensues in non-preference for ingesting by the flea beetle, Phyllotreta cruciferae (Goeze) (Bodnaryk 1992). However, wax coating may also have adverse influence by liking few insects. Plants with glossy leaf surfaces have also been reported to be resistant or non-preference to insect pests in many cases (Eigenbrode and Espelie 1995). Indirectly crystals of wax and waxy flowers may also damage the sticking, mobility, and efficiency of predators eventuating in higher herbivore abundance (Eigenbrode et al. 1999).
1.3.2.2 Hairy Structures
In plant mostly, the epidermal surface is protected with hairlike structures, which vary in form, size, position, and their role (Werker 2000). Generally, the hairs on the aerial parts of a plant are called as trichomes; however, the pubescence is mentioned when plant surface is protected by the collective trichomes. These trichomes vary in size from a few microns to several centimetres, and the form differs largely in various plants. They are glandular and non-glandular (Payne 1978). Non-glandular trichomes may work as fence for the attack of insects on the surface of plants or stop the herbivores’ feeding on the plant tissues, thus preventing the plants from any damage (Ram et al. 2004). Glandular trichome structures are developed to produce a number of chemical substances (Fahn 2000), which perform as crucial chemical barricades against insect pests and diseases (Glas et al. 2012). In black bean, Phaseolus vulgaris Linnaeus, curved trichomes were observed to stick the aphid, Aphis craccivora Koch (Johanson 1953), and the leafhopper, Empoasca fabae (Harris), resulting in damage and death (Pillemer and Tingey 1978). In few instances, the density of trichomes has been found to be persuading interestingly, in response to insect nourishing. When plants were attacked by the cabbage white butterfly, Pieris rapae (Linnaeus), and the cabbage looper, Trichoplusia ni (Hubner), on young black mustard, Brassica nigra (Linnaeus) W. D. J. Koch, plants led to enhanced trichome density on new foliage (Traw and Dawson 2002). Few insect pests have also been observed to have evolved morphological and biochemical modifications to counteract the effect of hairy growth. These trichomes may also have role in plant resistance indirectly, by restricting the searching efficiency of predators of herbivores. On glabrous varieties, than on hairy leaf surfaces, the parasitic wasp, Encarsia formosa Gahan, is greatly more effective in searching the whitefly nymphs (van Lenteren et al. 1995).
1.3.2.3 Leaf Surface Rigidity
In a lowland tropical forest, plant toughness was reported as the best forecaster of interspecific differences in herbivory ranking (Coley 1983). By overthrow of cellulose, lignin, suberin, and callose with sclerenchymatous fibres, plant cell walls become stronger and resistant to piercing and sucking by insect mouth parts and ovipositors of adult females and chewing by insects using mandibles. Solid-stemmed cultivars of wheat with toughness were found to be resistant to stem sawfly, Cephus cinctus Norton (Platt and Farstad 1946). Rind hardness was a significant element in sugarcane, in minimizing the internode borer Diatraea saccharalis (Fabricius) attack (Martin et al. 1975). In alfalfa, seed losses due to the seed chalcid Bruchophagus roddi (Gussakovsky) were lower in genotypes possessing more lignified pod walls (Springer et al. 1990).
1.3.2.4 Design and Plant Size
The preference of a plant cultivar to be a host for insect pests may differ with plant design, architecture, and size. The spacing of plant canopy, variations in stem, leaf and bud shapes and their sizes, and angles of branches may impact insect liking and survivance. From monocots through herbs and bushes and trees, the increase in size and design of plants is related with the enhancement of diversity of the connected insect life (Lawton 1983). The indirect impacts of plant structures on herbivory are also arbitrated through their effect on the parasites and predators. As compared to normal leaf varieties, okra-leaved cultivars in cotton are less attacked by bollworms, whitefly, and boll weevil (Ram et al. 2004). Varieties with little cotyledons and unifoliated leaves in soybean were tolerant to the legume seedling fly, Ophiomyia phaseoli (Tryon), and these are the sites where the female insects oviposits (Talekar and Tengkano 1993).
1.3.3 Biochemical Productions as Barriers
Plants have developed a number of chemical structures to avoid attack by insects and other herbivores. As few chemicals are associated with basic metabolism, several other compounds have been reported to deter, repel, kill, or stop insects and other herbivores from feeding on some plants as their food (Chapman 1974; Harborne 1993; Mithofer and Boland 2012). As plant feeding insects have evolved the capacity to manipulate their hosts, the plants have acknowledged by developing defending biochemical secretions to prevent herbivore damage (Johnson 2011). These biochemicals secreted by plants may be grouped into holistic nutrition and allelochemicals.
1.3.3.1 Holistic Nutrition
Crop plant suitability as a host for one or more insect pests is reliant on its capacity to offer holistic nutrition for growth and multiplication of insects. Plants generally provide nutrients at suboptimal ratios, from an insect ’s outlook, which are amalgamated with indigestible structural mixtures of cellulose and lignins, and a diversity of allelochemicals (Schoonhoven et al. 2005). The biochemicals apply a large number of behavioural, physiological, and growth-impeding impacts, some of which may even result in to insect killing. Many insects have the same needs for food, comprising of carbohydrates, amino acids, fatty acids, sterols, and some micronutrients; however, plants are often nutritionally inferior in itself. The key classes of basic plant amino acids, carbohydrates, and lipids intricated in the physiological plant procedures act as important nutrients for herbivores. So, alterations in basic plant metabolism and nutrients highly impact the living and reproduction of plant feeders (Berenbaum 1995). Mainly, nitrogen is crucial as insects are incompetent to utilize plants organic and inorganic nitrogen as it is suboptimal for the insect needs (Schoonhoven et al. 2005). This may work as a main obstacle for the full utilization of plants by a large number of insects. Appealingly, the herbivores consist of about 50% of the total arthropods in less than one-third of insect fauna, showing that once the nitrogen insufficiency is broken, these insects are capable to reach a sufficient nutrition supplies (Strong et al. 1984).
1.3.3.2 Crop Plant Nutrient Deficiency: A Resistance Mechanism Against Insects
Crop host plant , having insufficient one or more important elements of nutrition needed by the herbivores, may demonstrate insect tolerance through antibiotic and antixenotic impacts on the insect development and similarly may also form disparity of accessible essential nutrients (Arora and Dhaliwal 2004).
Cotton Genotypes
Few cotton cultivars with built-in protection depending on essential nutrients have been developed for the leafhopper, Amrasca biguttula (Ishida); whitefly, Bemisia tabaci (Gennadius); stem weevil, Pempherulus affinis (Faust); and thrips complex (Uthamasamy 1996). The whitefly B. tabaci, with genetically resistant genotypes, exhibited more amounts of K, P, and Mg and small of N and Fe in comparison to susceptible cultivars. However, sugars, proteins, Ca, and Cu did not exhibit important correlation with whitefly population multiplication. Another report mentioned that total contents of sugar in some cotton genotypes were positively associated with whitefly attack at the vegetative stages but negatively connected with it after crop flowering period (Rao et al. 1990). For the leafhopper, A. biguttula, more prone cultivars, Acala 4–42, had large contents of reducing sugars (2.55%), proteins (18.49%), and free amino acids (10.15 mg/g) in comparison to highly tolerant BJR 741 holding 1.63% reducing sugar, 13.45% proteins, and 6 mg/g free amino acids (Singh and Agarwal 1988).
Rice Genotypes
Resistant rice cultivars to thrips, Stenchaetothrips biformis (Bagnall), contained notably low reducing sugars and free amino acids as compared to the prone cultivars (Thayumanavan et al. 1990). Contents of asparagine in small amounts in rice cultivar ‘Mudgo’ were contemplated to be the fundamental reason of tolerance to brown plant hopper, Nilaparvata lugens (Stal). Brown plant hopper confined with Mudgo variety showed underdeveloped ovaries with few eggs; however, those kept with susceptible cultivars developed normal ovaries with maximum eggs (Sogawa and Pathak 1970). The gall midge Orseolia oryzae (Wood-Mason)-resistant cultivars PTB 18, PTB 21, and Leuang 152 had greater amounts of free amino acids and low sugars in their shoot apices than non-tolerant cultivars Jaya and IR8. Stems of resistant (TKM6) and moderately resistant (Ratna) cultivars had low amino acids and sugars than susceptible varieties (IR8) against the stem borer, Scirpophaga incertulas (Walker), (Vidyachandra et al. 1981).
Legume Contents
Auclair (1963) demonstrated the significance of amino acid contents in the pea plant on the susceptibility to aphid , Acyrthosiphon pisum (Harris). He reported that the amounts of amino acids in the susceptible cultivars were significantly greater than those in the resistant varieties. High proportions of non-reducing sugars and little percentage of starch in the seeds of chickpea GL 645 might be accountable for the less attack of the pod borer H. armigera in the cultivar under trial in comparison to the infestor (Chhabra et al. 1990). In pigeon pea, genomics against pod borers, small amino acid, protein and sugar amounts, and high phenol contents developed resistance. Sugar amounts were more in seeds (3.64–4.82%) and in the pod coat (3.66–4.92%) of susceptible genotypes (ICPLI, ICPLS7, and UP AS20). Total sugar proportions in the resistant varieties varied between 2.86 (ICPLS3024) and 3.51% (HS9–2) in the seeds and 2.91 (ICPLS3024) and 3.44% (HS9–2) in the pod coat. The amino acid concentrations were small in the pod coating (1.40–1.52 mg/g) and seed (1.39–1.55 mg/g) of resistant pigeon pea genotypes assessed in comparison to the susceptible varieties (1.89–2.57 mg/g in pod coat, 2.04–2.62 mg/g in seed). Extremely significant positive correlation found between amino acid present and infestation of borers helped the potential contribution of amino acids in providing resistance to the pod borers (Sahoo and Patnaik 2003).
1.3.3.3 Phytochemicals
The allelochemicals or phytochemicals secreted by plants are mostly minor metabolites which do not contribute largely in the basic plant pathways of metabolism. As the basic metabolic pathways are the same in almost all angiosperms, these secondary materials differ largely in various crop plants (Schoonhoven et al. 2005). Fraenkel (1959) established that these metabolites serve to repel many herbivores. It has been reported that the plant develop a number of minor substances, and more than 200,000 of these have been recognized (Dixon and Strack 2003). Further, the allelochemicals have been categorized into two classes such as allomones, which help the host plant , and kairomones, which assist the herbivores. In different types of insect-plant associations, the action of allelochemicals can decide the ranking of a plant either as a kairomone host and non-host or as allomone-resistant host plant and allomone-susceptible host (Panda and Khush 1995). Allomone plants are contemplated as a big element accountable for plant defence mechanism against herbivores, and these have been utilized to enhance levels of resistance in many field crops (Green and Hedin 1986). The different secondary plant metabolites used in plant defence mechanisms against insects have been described shortly (Rosenthal and Berenbaum 1991; Arora and Dhaliwal 2004; Schoonhoven et al. 2005; Arora and Sandhu 2017).
Unusual Amino Acids
In many unrelated higher and some lower plants, nonprotein or unusual amino acids are ubiquitous. About 600 amino acids have been described from different legumes. Nonprotein amino acids may provide defence against natural enemies and diseases due to their constructional analogy to the usually important amino acids in nutrition. The biological impacts on herbivores are partially due to the correlated molecules which get involved wrongly into the insect protein synthesis system or through stopping of biosynthetic tracks (Rosenthal 1991; Huang et al. 2011; Yan et al. 2015). In creating insect development disturbance, canavanine, azetidine-2-carboxylic acid, 2,4-diaminobutyric acid, mimosine, 3-hydroxyproline, 5-hydroxynorvaline, β-cyanoalanine, and pipecolic acid are important (Parmar and Walia 2001; Yan et al. 2015). Root exudates and extracts from in vitro grown seedlings of sesame showed an inhibitory effect on egg hatch and juvenile penetration by root-knot nematode, Meloidogyne incognita. Analysis of root exudates of sesame showed seven free amino acids, i.e. aspartic acid, glutamic acid, valine, proline, serine, glycine, and leucine, whereas the exudates of okra had ten free amino acids, viz. glycine, serine, leucine, isoleucine, alanine, arginine, glutamic acid, glutamine, lysine, and cystine. Three sugars, viz. fructose, glucose, and sucrose, were found in sesame root exudates and extracts. Commercial amino acids both singly and in combination inhibited egg-hatching (Tanda et al. 1989).
Organic Compounds
Terpenoids are the biggest and greatest diverse group of organic compounds observed in crop plants. They show gigantic chemical variations and complexness; however, all are established by blend of five-carbon isopentane, and many of them are lipophilic compounds (Ruzicka 1953). Terpenoids attain their highest structural and functional variety in the plant flora. About 30,000 terpenoids are found in plant systems, and a large number of them act as protection against insect pests and diseases or as allures for crop pollinators and fruit scattering organisms. Gershenzon and Croteau (1991) reported that the terpenoids are composed of two or more than five carbon units in their forms: monoterpenoids (2×C5), sesquiterpenoids (3×C5), diterpenoids (4×C5), triterpenoids (6×C5), tetraterpenoids (8×C5), and polyterpenoids [(C5) n where n > 8].
Monoterpenoids have been established to act as toxins and restraints for feeding and laying eggs against many herbivores. Among monoterpenoids, the important example of insect toxin is pyrethrum, working as a botanical insecticide, reported in the leaves and flowers of Chrysanthemum spp. In pyrethrum, the active ingredient is a combination of monoterpene esters commonly called as pyrethroids (Casida 1973).
Cotton and other plants belonging to Malvaceae have pigmented spherical glands found in their foliage, blossoms, and branches of plants. The pigments of these glands, in addition to anthocyanin, possess more amounts of a variety of monoterpenoids and sesquiterpenoids particularly gossypol. Gossypol is a phenolic compound, sesquiterpene dimer with two aldehyde remainders. Gossypol is poisonous to a number of insect pests, resulting in big decline in the survivance, multiplication, and development of numerous major lepidopterous and coleopterous herbivores. The contagion of gossypol to insects is assumed to arise from its irrevocable to proteins in the gastrointestinal area, bringing about a decrease in the digestion of proteins. In the gastrointestinal region, the proteins may be the eaten dietary proteins or the digestive enzymes developed by the herbivore (Meisner et al. 1977). A key secondary metabolite of the common dandelion, Taraxacum officinale G. H. Weber ex Wiggers, the sesquiterpene lactone, beta-d-glucopyranosyl ester (TA-G), saves the plant against its main native root feeders, the common European cockchafer, Melolontha melolontha Linnaeus, by discouraging larval infestation (Huber et al. 2016).
In terpenoids, triterpenoids (C30) are the biggest with six C5 isoprene units. The three main classes of triterpenes are the cucurbitacins, limonoids, and saponins which have important contributions in plant-insect herbivore interplays. In the Cucurbitaceae, cucurbitacins are a class of approximately 20 very bitter and toxic tetracyclic triterpenes, cramped chiefly to the host plants. These substances act as poisons and restraints for feeding against a large number of herbivores (Tallamy et al. 1997). Few specific insects attacking cucurbits are capable to absorb or prevent these poisons and even utilize cucurbitacins as host identifying signals (Abe and Matsuda 2000).
With a fundamental structure of 26 carbon atoms, the limonoids are a big class of very oxygenated compounds and are reported in Rutaceae, Meliaceae, and Cneoraceae. These limonoids are very strong feeding deterrents against many herbivores. More than 100 triterpenoids have been detected from the neem (Azadirachta indica A. Juss.) seeds, and a many of them are working as deterrents and antifeedants against crop insect pests. Azadirachtin is the chief among these triterpenoids, which is effectual at doses as minimum as 50 parts per billion. Over 400 insects have been found to be susceptible to neem compositions at different concentrations. With antifeedant actions, neem is demonstrated to influence the living, growing, multiplication, vigour, and egg-laying capacity of herbivores (Schumutterer 1995; Dhaliwal and Arora 2001).
In many crop plants, saponins are most common and made up of a sugar part (glycoside) associated with a hydrophobic aglycone, which may be a triterpene or a steroid, both of which develop from the C30 precursor, squalene. In soybeans, beans, peas, tea, spinach, sugar beet, and quinoa, triterpenoid saponins have been isolated. In oats, capsicum, peppers, aubergine, tomato seed, allium, and asparagus, steroidal saponins are detected (Francis et al. 2002). Saponins apply a powerful insecticidal reaction against many insect groups resulting in enhanced kill, decreased food intake, weight loss, growth lagging, and moulting faults (Geyter et al. 2007).
Natural Substances
The alkaloids are a diverse group of natural substances that found in all groups of living life; however, they are typical in plant systems. They mostly comprise primary products that have one or more nitrogen atoms, mostly in amalgamation as component of a cyclic system. Many of them are products of usual amino acids, for instance, as lysine, tyrosine, tryptophan, histidine, and ornithine (Facchini 2001). They occur in about 20% of the angiosperms. Mostly, each species carrying alkaloid exhibits its own distinctive, alkaloid shape explained genetically. Many alkaloids have been described to be poisonous or repellent to herbivores. Due to their nature containing nitrogen, several alkaloids impede with the major elements of acetylcholine transference in the nervous system. Nicotine and nornicotine obtained from tobacco were important as botanical insecticides before the invention of organic insecticides made synthetically (Dhaliwal and Arora 2001). Pyrrolizidines, quinolizidines, indole alkaloids, benzylisoquinolines, steroid alkaloids, and methylxanthines are many classes of alkaloids at dietary concentrations over 0.1% and act as insect ’s deterrents and to other herbivores (Schoonhoven et al. 2005).
Glucosinolates
About 100 sulphur or nitrogen carrying unique minor compounds such as glucosinolates comprise a little class of which work as harbingers of oils in mustard. Together with the family Brassicaceae, glucosinolates are found generally in the Brassicales order. Glucosinolates seem to work as successful chemical protections against a number of non-adapted herbivores (Fahey et al. 2001). Heynhold genome, at a minimum 52 genes, is intricated in glucosinolate biosynthesis in the thale cress Arabidopsis thaliana (Linnaeus) (Arabidopsis Genome initiative 2000; Halkier and Gershenzon 2006). When insects infest crop plants, glucosinolates are broken down by myrosinase enzyme into many metabolites acting as deterrents against insects (Hopkins et al. 2009). On the flip side, a small group of Brassica feeders are capable to use glucosinolates in searching and identifying the host. Glucosinolates and their evaporative compounds formed by hydrolysis are also utilized as signals by predators of Brassica feeding herbivores (Louda and Mole 1991).
Juvenoids and Ecdysteroids
For the growth, development, multiplication, and survival of herbivores, the endocrine system is crucial. Though several insect hormones are demonstrated, the juvenile hormone (JH) and the ecdysone or moulting hormone (MH), two strong hormones are established to contribute in these procedures. Juvenoids and ecdysteroids are match of these hormones. It is assumed that plants may have evolved juvenoids and ecdysteroids as fine defence mechanisms against herbivores. Crops possessing more ecdysteroid amounts, i.e. >1000 ppm, are prevented by insect pests. There are few main juvenoids derived from plants such as farnesol, sesamin, juvabione, sterculic acid, bakuchiol, and thujic acid which are familiar to disorder metamorphosis, moulting, and multiplication in herbivores.
Crop Proteinase Inhibitors
In some crop plants, protease inhibitors (PIs) comprise a plentiful and significant group of substances which have a defending mechanism against insect pests (Dunaevsky et al. 2005). New reports utilizing microarrays and proteomic proposals have disclosed that the plant defence mechanisms relying on proteins contribute more significantly against insects than perceived before (Felton 2005; Zhu-Salzman et al. 2008). Arginases, polyphenol oxidases, and peroxidases, the defence proteins, may have properties against microbes, and others, for example, chitinases, cysteine proteases, lectins, and leucine amino peptidases, may also be poisonous (Zhu-Salzman et al. 2008). Nevertheless, the anti-insect action of plant proteins is easily disabled by proteases, and proteolysis-susceptible proteins can be avoided with PIs (Mithofer and Boland 2012). Serine, cysteine, and aspartate proteinases and metallocarboxypeptidases stop the actions of different enzymes in herbivores mostly insect peptidases which are intricated in the development and multiplication of insects. The PIs also minimize the food digesting capability of the herbivores, thus resulting in to the scarcity of amino acids, the major components of food finally decelerating the growth and affecting the starvation. Numerous PIs have been found in plant system (De Leo et al. 2002), which are successful against a number of lepidopteran and hemipteran herbivores (War and Sharma 2014). PIs were positively assessed for their trypsin and H. armigera gut proteinase inhibitory action in various parts of the tomato plants (Damle et al. 2005).
Phytohaemagglutinins
Lectins or phytohaemagglutinins are proteins with a capability to reversibly attach to the carbohydrate moieties of complicated carbohydrates without changing the covalent shape of any of the identified glycosyl myths. Lectins are found globally all over the plant kingdom, where they comprise 6–11% of the total plant proteins. Mostly the legume seeds cotyledons are abundant in lectins. Lectins are connected with the defence mechanisms of crops against insect pests and diseases (Liener 1991). Arisaema helleborifolium Schott lectin showed anti-insect action towards the melon fruit fly second instar larvae, Bactrocera cucurbitae (Coquillett) (Kaur et al. 2006).
Plant Phenolics
In plants phenolics are very common and are fragrant compounds with one or more hydroxyl groups (Harborne 1994). Hydroxybenzoic acids like vanillic acid, hydroxycinnamic acids like caffeic acid, and coumarins are comparatively simple phenolics (Schoonhoven et al. 2005). Coumarins comprise a 5,6-benz-2-pyrone skeleton and may be differently hydroxylated, alkylated, alkoxylated, or acylated. Coumarins can discourage eating and impede with growth of herbivores. Coumarin, bergamottin, is capable of killing eggs of Colorado potato beetle, Leptinotarsa decemlineata (Say), though mammein is insecticidal to the beetles attacking mustard. Coumarins seem to work as kairomones for some insects that are particularly eating on coumarin carrying plants (Berenbaum 1991b). Flavonoids are established mostly in all higher plants in the phenolics, and many plants exhibit their own distinguishing flavonoid contour. To make a water-soluble glycoside, flavonoids have a basic C6-C3-C6 structure, which is related to a sugar moiety. Flavonoids derived from plants are catechin, a botanical insecticide rotenone, and phaseolin, all of them operate as impediments against insect pests (Schoonhoven et al. 2005).
Tannins are polyphenolic combinations generally available in higher plants. The phenolic hydroxyl groups of tannins tie to nearly all soluble proteins, developing insoluble copolymers. Proteins joined to tannins are indigestible and thus reduce the nutritious worth of plant parts (Schoonhoven et al. 2005).
Latex in Laticifers
Latex is found in special tissues known as laticifers, having chemically unspecified milky suspensions or emulsions in aqueous fluids (Agrawal and Konno 2009), and as a defence mechanism. Tiny insects get ensnared in latex physically or their mouthparts may stick together, and chemical constituents in latex including proteins and toxins adversely affect the insect growth (Dussourd 1995). Injuring of laticifers by insects leads to leakage at injury site (Mithofer and Boland 2012). In the milkweed, Hoodia gordonii (Masson) Sweet ex Decne, feeding by larvae and oviposition by T. ni adults, was discouraged when latex was mixed in artificial diet or applied on the leaves of the host plants (Chow et al. 2005).
1.3.3.4 Allelochemicals as Host Plant Defences
Allelochemicals in Maize
Maize is damaged by a variety of insect pests, and anti-herbivore defences in maize comprise small molecules known as benzoxazinoids (Frey et al. 2009), chlorogenic acid (Cortes-Cruz et al. 2003), and maysin (Rector et al. 2003) besides defence-linked proteins (Chuang et al. 2014). Xie et al. (1992) examined for hydroxamic acid many lines of maize resistant to western corn rootworm, Diabrotica virgifera Le Conte. Root extracts in all tests were having four main hydroxamic acids such as 2,4-dihydroxy-7methoxy-l,4-benzoxazin-3-(4H)-one (DIMBOA), 2,4-dihydroxy7,8dimethoxy-l,4-benzoxazin-3(4H)-one (DIM2BOA), 2-hydroxy,7-methoxy,1,4-benzoxazin-3(4H)-one (HMBOA), and 6-methoxy-benzoxazolinone (MBOA). These hydroxamic acids retarded the growth and development and loss in weight and also reduced the head capsule width of rootworm larvae. Wiseman et al. (1992) described a greatly significant negative link between weight variations in corn earworm, Helicoverpa zea (Boddie), and in the fall armyworm, Spodoptera frugiperda J.E. Smith, larvae and maysin contents in the silks of many corn lines.
Allelochemicals in Cotton
In cotton, the allelochemicals such as gossypol, gossypurin, heliocides, hemigossypolone, tannins, anthocyanins, flavonoids, and phenolics have been found to affect negatively on infesting herbivores. Gossypol was described to show resistance to cotton bollworm Heliothis zea (Bottger et al. 1964). Generally, many cotton varieties grown commercially have a gossypol concentration of about 0.5% in squares. With more gossypol cotton varieties (No. 16482, 6501, and Termez-14) had harmful effects such as prolonging incubation period, higher kill in young larvae, and reducing the weight of larvae in comparison to the cultivars having less gossypol contents (Vilkova et al. 1988). They also reported that antibiotic effect of high level of gossypol contents decreased the fecundity (more than 50%) of H. armigera. Gossypol is also described to influence the boll nutritional value adversely by developing complex compounds joining with amino acids, proteins, and enzymes. Mohan et al. (1994) reported that genotypes with maximum gossypol glands on the surface of the ovary in Asiatic cotton Gossypium arboreum Linnaeus decreased the attack of bollworm complexes in H. armigera, Earias vittella (Fabricius), and Pectinophora gossypiella (Saunders). When healthy cotton plants were analysed, for the cotton stem weevil, P. affinis, the amount of tannins was minimum in susceptible MCU5 and more in the resistant successions. The contents grew in the gall region when the plants were attacked, and the concentration was high in resistant accessions in comparison to the susceptible genotype MCU5. No difference was reported in the total phenolic amounts in the resistant and susceptible accessions of healthy stems. The amount of total phenolics, however, enhanced in the gall regions significantly when attacked, even the concentration rose more in resistant accessions. It can be established that more development in tannin and phenolic amounts might offer a defensive tool against the stem weevil infestations (Uthamasamy 1996).
Allelochemicals in Vegetable Crops
In Solanum species against the Colorado potato beetle (CPB), L. decemlineata, and the potato leafhopper, E. fabae, glycoalkaloids in potatoes perform as natural resistance mechanisms. Many wild Solanum species have exhibited a positive relationship between total leaf glycoalkaloid concentration and resistance to Leptinotarsa species. Completely stopping eating, leptin is a very successful antifeedant, whereas tomatine and demissine are halfway in action, followed by the effectiveness of solanine and chaconine (Tingey 1984). Resistance of tetraploid potato (Solanum tuberosum L.) selection ND 2858-1 in the field and its backcross progeny against the Colorado potato beetle is developed by antibiosis mechanism. In detached leaf tests on resistant cultivars, neonates of CPB evolved slowly, and weight gain in larvae after 4 days was discouraged by 75% in proportionate to larval growth and weight gain on susceptible accessions. Foliar glycoalkaloid assays showed low levels of leptins I and II in resistant genotypes (Lorenzen et al. 2001). Against the tomato fruit borer H. zea, the wild species of tomato, Lycopersicon hirsutum and L. hirsutum f. glabratum, exhibited the phenomenon of antibiosis. For the antibiosis process, the chemicals involved were l-tomatine, 2-tridecanone, phenolics, and elements including iron zinc (Ferry and Cuthbert Jr 1975; Dimock and Kennedy 1983; Kashyap 1983). The toxicity tested of allelochemical 2-tridecanone was maximum against H. zea, Manduca sexta Linnaeus, and L. decemlineata. Maximum phenolic amounts have been reported to conclude resistance to the H. armigera species (Banerjee and Kalloo 1989), whereas high contents of tomatine are harmful to the greenhouse whitefly, Trialeurodes vaporariorum (Westwood) (Steehius and van Gelder 1985). In tomato, the protease inhibitor and chlorogenic acid were involved in resistance against aphid (Felton et al. 1989). In glandular trichomes of Lycopersicon hirsutum f. typicum accession (LA) 1777, the sesquiterpene carboxylic acids (SCA), (+) E-α-santalen-12-oic, (−)-E-endo-α-bergamoten-12-oic, and (+)-E endo-β-berqamoten-12-ion acids were developed which is more resistant to herbivores generally attacking tomato, L. esculentum, grown commercially. The larvae of tomato fruitworm, H. zea, and the beet armyworm, Spodoptera exigua (Hubner), showed reduction in eating, growth, and survival in the presence of such chemical compounds. In diet found lethal to the larvae, at concentrations as low as 2 mg SCA/g of diet and a concentration of 60 mg SCA/g, the sublethal effects were reported (Frelichowski Jr and Juvik 2001).
Root exudates and extracts from in vitro grown seedlings of sesame showed an inhibitory effect on egg hatch and juvenile penetration by root-knot nematode, Meloidogyne incognita. Analysis of root exudates of sesame showed seven free amino acids, i.e. aspartic acid, glutamic acid, valine, proline, serine, glycine, and leucine, whereas the exudates of okra had ten free amino acids, viz. glycine, serine, leucine, isoleucine, alanine, arginine, glutamic acid, glutamine, lysine, and cystine (Tanda et al. 1989).
1.3.4 Various Plant Defence Mechanisms
Plant defence mechanisms may be categorized into basic, which are found in the host plants disregarding the occurrence of insect or non-insect pests, and incited, which are developed in response to different abiotic and biotic stressors.
1.3.4.1 Basic Plant Defence Mechanisms
Plants have developed an abundance of structural and chemical defence mechanisms that are incorporated into their tissues disregarding the presence or absence of insects. These basic plant defence mechanisms can repel, deter, inebriate, derange, or disrupt the feeding on plant tissues, growth, and development of herbivores (Arora and Dhaliwal 2004; Ram et al. 2004; Mithofer and Boland 2012). These phytotechnological protection systems comprise of the following:
-
(a)
The texture and composition of the plant covers (Johnson 1975)
-
(b)
Existence of anatomical structures, for example, thin veins, thorns, silica, trichomes, or resins (Hanover 1975)
-
(c)
Lack of essential nutrients (House 1961)
-
(d)
Existence of substances similar to hormones which inhibit the growth of insects (Williams 1970)
-
(e)
Inappropriate pH or osmotic pressure (Beck 1965)
-
(f)
Accretion of secondary metabolites (Chapman 1974)
There are a large number of secondary metabolites comprising amino acids to alkaloids, terpenes, phenolics, steroid, cyanogen, and glycosides in mustard oil (Mithofer and Boland 2012). Additionally, plant systems may also transform nitrogen into compounds which are not accessible to herbivores (White 1978). The benefits of similar basic defences to insects are that these are developed during the time of high metabolic activities and can be used over an increased time period. These physiological devices act against a large number of generalist insects; however, regular subjection to such chemicals develops powerful critical pressure on the plant feeders, which may lead in the development of specialist herbivores.
1.3.4.2 Incited Plant Defence Mechanisms
To deter feeding by insect pests and stop colonization, incited plant defence mechanism is operated in the existence of insects and allows the plant (Sadras and Felton 2010). The insect feeding processes switch on many protection signals, resulting in to acceptable defence reactions (Wu and Baldwin 2010; Hogenhout and Bos 2011; Bruce 2015). Various plant species have also been described to acknowledge to insect females for depositing eggs in the same way (Hilker and Meiners 2006). Volatile organic compounds (VOCs) liberated by plants have been reported to allure predators of herbivores (Tamiru et al. 2011; Fatouros et al. 2012) or incite direct protections so that the rate of insect development is decreased on plants nursing eggs (Gieselhardt et al. 2013). Crop plants react to elicitors produced from mouth secretions of herbivores, mechanical injury, and exogenous inducer application. The mouth secretions/regurgitants of herbivores consist of many plant defence extrinsic molecules of elicitors; the crucial ones are conjugates of fatty acids (FACs). FACs are comprised of two related groups or moieties such as a fatty acid or an amino acid. It has been found that the fatty acid and amino acid develop from the plant and the insect , respectively, and are made in the midgut of herbivores. FACs not only act as main elicitors for plants to show the unique insect-plant interplay to discern insect infestation but also are intricated in insect nitrogen metabolism process. The first FAC separated from mouth secretion of the beet armyworm S. exigua larvae was N-(17-hydroxylinolenoyl)-l-glutamine (volicitin), and it excites maize plant tissues to develop volatiles, which allure natural enemies of the insect pest (Alborn et al. 1997). In tobacco plants, regurgitation of the tobacco hornworm, M. sexta, has N-linolenoyl-glu, a potential elicitor of volatile emissions. Additionally, few FACs stimulate mitogen-activated protein kinase (MAPK) pathway, developing many plant defence chemicals playing a part in signalling transduction regarding different stresses such as drought, diseases, and insect infestations.
MAPK signal pathway in eukaryotes and its important contribution in plant gesturing particularly for pathogen stresses are well demonstrated. The chief role of MAPK in governing plant transcriptomes has been described (Wu and Baldwin 2010). In tobacco plants, few FACs stimulate accretion of 7-epi-jasmonic acid, which operates as insect defence genes. Moreover, FACs also encourage nicotine and proteinase inhibitors (PI) in the coyote tobacco, Nicotiana attenuata (Torr. ex S. Watson) (Wu and Baldwin 2010; War and Sharma 2014). The plant plasma membrane is open to the environment and stimulates a large number of events following identification of pest injury. Alterations in cell membrane potential (Vm) actuated by insects are followed by fast electrical cues, which are functioning systematically in nature. Calcium ions (Ca+2) act as a second messenger signal pathways in many crop plants. After the insect attack, the signal may be seen a few seconds as a single transient oscillation or duplicate spikes with particular subcellular localization lag time, amplitude, and frequency. The Ca+2 cues stimulate calmodulin and other calcium-sensing proteins. This encourages a cascade of downstream impacts, like changed protein phosphorylation and gene expression structures (Furstenberg-Hagg et al. 2013). Herbivory results in the gathering of plant hormones; the main are salicylic acid (SA), jasmonic acid (JA), and ethylene. They arbitrate different signal transduction pathways found in plant defence devices against different biotic and abiotic strains. The important transduction pathways associated with plant protection against insects are phenylpropanoid and octadecanoid pathways arbitrated by SA and JA, respectively. All these pathways result in the synthesis and gathering of toxins at the site of feeding or in other plant parts, which are then carried to the location of feeding. Besides, antioxidative enzymes engaged in plant defence get gathered in plant tissues damaging site (Wu and Baldwin 2010). Yan et al. (2015) described the amassing of nonprotein amino acid 5-hydroxynorvaline in leaves of maize inbred line B73 following herbivory by the corn leaf aphid Rhopalosiphum maidis (Fitch) and the beet armyworm S. exigua and in response to application with methyl jasmonate, salicylic acid, and abscisic acid. Basic and actuated defences can either be direct or indirect. Direct defences spot the herbivores, while indirect defences work through the engagement of predators of herbivores in the support of plants. Some volatile organic compounds (VOCs), having terpenoids, fatty acid derivatives, and a few aromatic compounds, act as attractants to the natural enemies of insect pests (Mithofer and Boland 2012).
1.3.5 Insect Defences Encountering Plant Defence Mechanisms
Through an abundance of structural and chemical defence mechanisms, plants avoid themselves from insect attack. These defences may have applied heavy selection pressure on the herbivores leading to the development of adaptations in insects. The insect modifications to plant defences can be physical, behavioural, or biochemical and consist of different devices, for example, penetration barricades, toxic excretions, sequestrations, temporary attaching with carrier proteins and stocking of toxins in adipose plant tissues, enzymatic detoxifications, and feeding site mutations. It is crucial to know about these insect modifications to plant defences to reduce their impacts on the steadiness of resistance in plants to insects. The major counteracting defence insect strategies to plant defences (War and Sharma 2014; Bruce 2015) are briefly presented below.
1.3.5.1 Insect Processes to Plant Defence Mechanisms
Several herbivores have evolved special tools to get the better of the slippery waxy cover which gives a big hurdle to the walking and motion of insects on plant surfaces. For good bonding to the slippery cover, the minute setae on tarsal pulvilli of few chrysomelids expel an adhesive substance (Gorb and Gorb 2002). Leafhoppers of Empoasca species can utilize their tarsal pulvilli as suction cupping device (Lee et al. 1986), though several lepidopteran caterpillars have adhesive silken thread as a rope ladder to the leaf cover to act as a foot clasping instrument (Eigenbrode 2004). To control the complication of trichomes on the plant layers, the aphid Myzocallis schreiberi Hille Ris Lambers and Stroyan possesses a special device in the shape of claws and flexible empodia that function to have a fine grasp on the short woolly trichomes on the host plant surface, the Holm oak, Quercus ilex Linnaeus (Kennedy 1986). The hardness of leaves has been reported to minimize the attack of plant feeders. As a modification to the plant toughness, in caterpillars of Pseudaletia unipuncta Haworth, the head and chewing musculature are double the size when eat on tough grasses as compared to soft artificial nourishment; nevertheless, body mass is alike (Bernays 1986). Water lily beetles Galerucella nymphaeae (Linnaeus) consuming the tough water lily have excessively larger mandibles than conspecifics eating on the great water dock grin, Rumex hydrolapathum Huds., second host with mushy leaves (Pappers et al. 2001).
1.3.5.2 Insect Modifications Against Protease Preclusions
In a few host plants when the insect attack, they make protease inhibitors for protection. Insect attack on N. attenuata immediately produces and gathers trypsin PIs; M. sexta and S. exigua larvae accomplished better on trypsin PI-lacking plants in comparison to alike plants making PIs (Zavala et al. 2004; Steppuhn and Baldwin 2007). Still, several insects have modified to host plant PIs, which enhances the attack to the host crop plants. This defensive response to PIs by herbivores is a key barricade to the exploitation and use of PIs for a firm plant protection permitting the devices by which insects prevent the PI-based plant defence mechanism. In crop insect pests, two kinds of resistance or adaptation strategies to protease inhibitors have been established. One of them is based on the different proteases which are contrary to PIs (Parde et al. 2010). These unfeeling proteases can be found constitutively in the plant system and/or are actuated when the other proteases are forbidden to recompense their damages (Jongsma et al. 1995; Parde et al. 2012). S. exigua has been described to modify to potato proteinase inhibitor II by the gut proteinase actions, which is not developed by the PIs. Additionally, when tested on the soybean proteinase inhibitor (SPI) diet, insensitivity to the inhibitor for larval proteases was observed (Brioschi et al. 2007). Trypsin insensitivity to host plant PIs has been delineated from Agrotis ipsilon (Hufnagel), T. ni, and H. zea (Volpicella et al. 2003). Other resistance mechanisms to PIs in herbivores require the synthesis of particular proteases, which are capable to break down the protease inhibitors so as to decrease their inhibitory action. Proteolytic inactivation modification is a crucial mechanism evolved by herbivores to resist the proteolytic inhibition by PIs. When feeding on artificial diet with soybean PIs, a trypsin-like enzyme is manufactured newly by S. frugiperda (J.E. Smith) larvae (Brioschi et al. 2007). The larvae of diamondback moth, Plutella xylostella Linnaeus, have been observed to be insensitive to mustard trypsin inhibitor 2 (MTI2). Such insensitiveness has been ascribed to the deterioration of MTI2 by the pest , thus preventing the impact of the PI (Yang et al. 2009).
1.3.5.3 Conversions to the Mustard Oil Bomb
The ‘mustard oil bomb’, also called as the glucosinolate-myrosinase system, found in Brassicales (Brassicaceae, Capparidaceae, Tropaeolaceae), composes the most efficient and well-researched plant defence process against herbivores. Glucosinolates are categorized and are secured from thioglucosidase—myrosinase—their hydrolysing enzyme under usual environmental conditions. Although the glucosinolates are found in several plant cell systems, the myrosinase is only centralized in dispersed plant tissue cells. The myrosinase and glucosinolate meet together developing the unstable aglycones on tissue feeding, which automatically adjust into different active substances, mostly nitriles and isothiocyanates (Li et al. 2000; Hopkins et al. 2009). It has been described that more glucosinolate- and myrosinase-rich lines of Brassica juncea (Linnaeus) Czern. are more resistant to larvae of Spodoptera eridania (Cramer) than those with little contents of these inhibiting chemicals (Li et al. 2000). The larvae of T.ni prevented A. thaliana ecotypes that developed isothiocyanates on hydrolysis of glucosinolate and rather attacked on ecotypes that caused nitriles (Lambrix et al. 2001). Additionally, some parasitoids utilize glucosinolates that are produced by feeding herbivores to trace their host insects. In these instances, glucosinolates have a double function for the damaged plant , in direct as well as in indirect protection (Hopkins et al. 2009). Even few insects utilize glucosinolates for their own shield. Myzus persicae (Sulzer), Athalia rosae (Linnaeus), and P. rapae isolate glucosinolates into their haemolymph and body cells (Muller and Brakefield 2003; Kazana et al. 2007; Bridges et al. 2002). Upon the attack of natural enemies, the haemolymph releases glucosinolates that discourage the ants and the predatory wasps (Muller and Brakefield 2003). Few aphids particularly Brevicoryne brassicae (Linnaeus) and Lipaphis erysimi (Kaltenbach) seclude glucosinolates from the sap of phloem (Kazana et al. 2007; Bridges et al. 2002). Moreover, caterpillars of P. rapae clean the glucosinolates from plants by altering them contrary to toxic products to inert metabolites using a process of nitrile-specifier protein (NSP). The NSP procedure in the gut of P. rapae regulates the glucosinolate hydrolysis resulting into nitrile formation rather than toxic isothiocyanates (Wittstock et al. 2004).
1.3.5.4 Modifications Against Tannins
With the protein amino groups, tannins make hydrogen or covalent bonds, which result in the precipitation of proteins and the digestive enzymes of insect pests. Additionally, the chelation process of metal ions in the insect body by tannins minimizes their accessibility to the insects, thus influencing their development and multiplication. Tannins have also been described to stop feeding on plants and develop midgut lesions and pharmacological toxicity in insects (Bernays and Chamberlain 1980). Nevertheless, insects have evolved many adaptations to prevent the tannin poisoning. The important modified procedures insects utilize to keep away from the toxicity of tannins is the pH of the gut in alkaline form, assimilation of tannin using peritrophic membrane, polymerization, and discharging of the polyphenols after concentrating them (War and Sharma 2014). The surfactants created by lipid digestion in the gut lumen avoid protein precipitation (Martin et al. 1987). The degree of oxygen in the foregut also contributes in the tannin toxicity. When the pH is more, oxygen levels are short; it minimizes the tannin autoxidation process, resulting in reduced toxicity. In lowering the toxicity of tannin, the antioxidative structure of insects also contributes significantly. Ascorbate lowers the tannin oxidation and result in reactive oxygen species (ROS) in the insect stomach (Krishnan and Sehnal 2006). Grasshoppers acquire a powerful midgut antioxidative defence mechanism, which empowers them to resist tannins. This antioxidative defence system mostly contains glutathione, α-tocopherol, and ascorbate. In S. gregaria, the resistance to tannins and its relation with peritrophic membrane have been ascribed to the ultrafiltration of tannins. In few insects such as Melanoplus sanguinipes (Fabricius), tannic acid does not tie with the peritrophic membrane. Additionally, peritrophic membrane saves the insect epithelium against lesions and any harm by ROS by assimilating extremely reactive ferrous ions (Barbehenn 2003).
1.3.5.5 Phytochemicals and Their Enzymatic Detoxification
To conquer plant chemical protections, enzymatic detoxification of toxic chemicals arbitrates the modification of herbivores to plant allelochemicals and thus assists the insects. Herbivores respond completely to the toxic allelochemicals, when offered with the natural host plant diet or merged in the artificial diet, by enhancing the metabolic processes that lead to the making of detoxifying enzymes, for example, monooxygenases and glutathione-S-transferases (GST) (Nitao 1989; Wadleigh and Yu 1988). The procedures of detoxification that work in insects rely on the chemistry of plant , and its degrees are mostly affected by the concentration of allelochemicals in the host plant (War and Sharma 2014). Insects use different enzymes for the detoxification of insecticides and allelochemicals of plants, and few strategies are universal (Francis et al. 2005; Scott et al. 2010). The most important is the process of polysubstrate monooxygenases which is also known as mixed-function oxidases. The constituent at the end of this structure is cytochrome P450, which is known as it assimilates maximum light around 450 nm when combined with carbon monoxide. Cytochrome P450 amalgamates even with the toxic substrate and with molecular oxygen, accelerating the oxidation process of the substrate. Cytochrome can merge with various lipophilic substrates and occurs as many isozymes that differ in their substrate explicitness (Feyereisen 2006). The P450s are considered as one of the main operators in insect-plant coexistence, as these are utilized by the host plants to release toxins and by the herbivores for plant chemical detoxification (Schuler 1996). Drosophila mettleri Heed living in desert area feed on cactus having toxic allelochemicals contain adaptable quantities of P450 associated with the metabolism of such toxins (Danielson et al. 1997). The metabolism of isothiocyanates, for example, 2-phyenylethylisothiocyanate, indole-3-carbinol, and indole-3-acetonitrile, in S. frugiperda midgut microsomes is Cyt P450-reliant (Yu 2000). Conversion of lepidopterans to minor metabolites of plants, for example, furanocoumarins, has been ascribed to P450s. Black swallowtail, Papilio polyxenes Fabricius, living on plants having furanocoumarins in diet allows up to 0.1% xanthotoxin (Berenbaum 1991a), which is cleaned by P450 monooxygenases (Bull et al. 1986). An apparent concept of participation of P450 in detoxification of allelochemicals in plant system occurred after CYP6B1 sequencing from P. polyxenes, which instructs for P450s. Coding for P450s, expression of CYP6B161 and CYP6B162 is actuated in cell lines of lepidopterans, showing the participation of P450s in metabolism of linear furanocoumarins, for instance, xanthotoxin and bergapten (Ma et al. 1994). Several P450s found in phytochemical detoxification have been derived from insects, such as from parsnip webworm, Depressaria pastinacella Duponchel (Cianfrogna et al. 2002), M. sexta (Stevens et al. 2000), and Helicoverpa species. Moreover, the transformation of dihydrocamalexic acid to camalexin, which are the main Arabidopsis phytoalexins, is accelerated by cytochrome P450 PAD3 (Schuhegger et al. 2006). Resistance to glucosinolates in aphid is ascribed to the CYP81F2, which is a downriver bit of the indolic glucosinolate pathway mechanism (Pfalz et al. 2009). P450s have also been delineated from several other herbivores where they work to metabolize the phytochemicals. For instance, in Musca domestica Linnaeus, CYP6A1 detoxifies the terpenoids (Andersen et al. 1997). In H. armigera, P450 monooxygenase CYP6AE14 metabolizes gossypol (Mao et al. 2007); in Anopheles gambiae Giles, CYP6Z1 detoxifies xanthotoxin and bergapten, furanochromones, and natural myristicin, safrole, and isosafrole (Chiu et al. 2008). However, CYP6Z2 detoxifies xanthotoxin, lignin, piceatannol, and resveratrol (McLaughlin et al. 2008); and in Diploptera punctata Eschscholtz, CYP4C7 metabolizes sesquiterpenoids (Sutherland et al. 1998). In bark beetles, Ips pini Wood and Bright and Ips paraconfusus Lanier metabolize the monoterpenes, sesquiterpenes, and diterpenoid resin acids by using P450s (Seybold et al. 2006). Another enzyme system, the glutathione-S-transferase (GST) is occurring in insect resistance to host plants by metabolism of xenobiotics and catalyzation of the conjugation of electrophilic molecules using thiol category of lowered glutathione, leading in fast defecation and deterioration (Francis et al. 2005). This enzyme class has been incriminated in detoxification of insecticides that are neurotoxic and influence the development and multiplication of insects. Among them are spinosad, diazinon, DDT, nitenpyram, lufenuron, and dicyclanil (Sintim et al. 2009). Many reports have suggested the significance of GST in insect modification to phyto-glucosinolates, and minor plant metabolites added in the artificial diet of S. frugiperda, S. litura, T. ni, M. persicae, Aulacorthum solani (Kaltenbach), and A. pisum (Enayati et al. 2005). In M. persicae, more production of GST has been assigned to insect modification to glucosinolates and isothiocyanates in Brassicaceae, though there is no straight conflict of isothiocyanates, as aphids prick with their stylets right into the phloem tissues (Francis et al. 2005; Kim et al. 2008).
1.3.5.6 Counter-Defence Mechanisms in Herbivoral Gut Symbiontic Systems
In reply to the insect attack, the evocation of plant defences has been reported to be regulated by chat between jasmonic acid (JA) and salicylic acid (SA) pathways of signalling. Insects have various microbes in their gut area, and such symbionts can adapt to plant-insect interplays (Hogenhout et al. 2009). In tomato, Chung et al. (2013) described that the grubs of Colorado potato beetle, L. decemlineata, utilized digestive track bacteria in their mouth secretions to control anti-herbivore defences. The antibiotic-untreated larvae reduced the development of JA and JA-responsive anti-herbivore defence mechanisms but enhanced SA gathering and SA-reactive gene expression. The reduction at the cellular level or plant defences led in increased larval development. In a study, the gut bacteria in three genera Stenotrophomonas, Pseudomonas, and Enterobacter were incriminated for defence subduing. Hammer and Bowers (2015) suggested the ‘gut microbial facilitation hypothesis’, saying that differences in insects in their ability to feed chemically protected plants can be due to the differences in their related microbial groups. Such reports have worn help from molecular research on gut bacteria. In Japanese common stink bug, Megacopta punctatissima (Montandon), the gut bacteria are competent of decarboxylating oxalate, a minor metabolite common in plants (Nikoh et al. 2011). Bacteria nursing in the gut of the mountain pine beetles is related with the terpene metabolism (Adams et al. 2013) and are efficient in detoxification of terpenes in vitro (Boone et al. 2013). From the midguts of gypsy moth larvae, the Acinetobacter species are able to detoxify the dietary phenolic glycosides (Mason et al. 2014). With the existence of gut bacteria largely in the insect mouth secretions, these may be linked with seizing of plant defence reactions in other instances of insect-plant interplay.
1.4 Insect Responses to Artificially Induced Plant Defence Mechanisms
With a contagious long history in the USA, the Hessian fly (HF), Mayetiola destructor (Say) (Cecidomyiidae: Diptera), is a major pest of wheat and spread in North Africa, Europe, West and Central Asia, North America, and New Zealand (Buntin and Chapin 1990). It has been effectively controlled using insect-resistant varieties bearing HF-specific R-gene(s). Nevertheless, in 6–8 years, virulent biotypes of HF are competent of defeating its resistance (Chen et al. 2009; Stuart et al. 2012). After egg-hatching, the neonate HF larvae creep on the upper surface of leaves and go to the seedling, where it continues feeding in susceptible cultivars but flops to do so in resistant genotypes. HF carrying virulent biotypes on a susceptible variety lead to a suitable interaction obliging pest elite; however, a virulent biotype in 3–5 days on the resistant variety causes incompatible interplay and death of insect (Subramanyam et al. 2015).
About 35 resistance genes (H1–H3, h4, H5–H34, and Hdic) from wheat and associated crop plants have been distinguished and incorporated in wheat varieties grown commercially (Chen et al. 2006; Stuart et al. 2012). For study of gene-for-gene (GNG) interaction, the HF wheat system is regarded as a model process between host and the insect (Hatchett and Gallun 1970; Subramanyam et al. 2015). Plants react to the injury of HF larvae by gathering of reactive oxygen species (Liu et al. 2010) in resistant varieties having R genes and the making of inhibitor enzymes (Wu et al. 2008), lectins (Williams et al. 2002; Subramanyam et al. 2008), and other minor metabolites (Liu et al. 2007). Contrary to this, the adaptable interactions are distinguished by enhanced nutrient accessibility at the place of injury along with the gathering of nitrogen-rich molecules (Liu et al. 2007; Williams et al. 2011). Reports mention that the HF is competent to control resistance via recessive mutations in similar avirulence (HFAvr) genes (Aggrawal et al. 2014). The HFAvr genes code for proteins known as effectors that are inserted with the saliva into the plant tissues during the injury (Hogenhout et al. 2009). Plants bearing R genes can identify such secretions and excite the defensive routes (Chisholm et al. 2006). In virulent HF biotypes, the Avr proteins are adapted to either prevent discovery by the plant or fail to activate the defending mechanism (Chen et al. 2016).
Darwin in his magnum opus On the Origin of Species in 1859 mentioned that the ‘Coadaptations of organic beings to each other…’. Organisms interact with each other with either the similar or another type of animal. Coevolution relates to genetic swap in two interacting animal species. Ehrlich and Raven (1964) reported the first interaction between Monarch butterfly and milkweed (a host plant ). Neither any plant is susceptible to all the herbivores nor is any insect a pest of all plants it experience in environment. Additionally, less than one-third of all insects such as exclusively Lepidoptera, Orthoptera, and Phasmida; predominantly Hemiptera and Thysanoptera; or partially Coleoptera, Diptera, and Hymenoptera are plant pests; however, these belong to half of all herbivores. So mostly all crop plants have evolved having impressive structural and biochemical defensive mechanisms against the insect pests. However, those insects which are competent to break these barricades in one or more plants can reach these plants for feeding (Arora 2012). Insect pests continue evolving adaptations for detoxification or breaching such defensive systems. The results of a coevolutionary system since 400 My are the extant phytophages and their host plants (Labandeira 2013). For discerning their ecosystem, determining the sensory input, and reacting to it appropriately, insects have developed a sensitive biological system (Martin et al. 2011). Lucrative host locating and liking are basically governed by chemical signals. The insect reactions rely on host and environmental conditions (Riffell et al. 2009; Webster et al. 2010). Plants have also developed many structural and chemical defence mechanisms against insects. The insects one after another have developed to prevent these barricades, and many ideas have been put forward.
1.4.1 Coevolution
Theory of coevolution was detailed by Ehrlich and Raven (1964) and backed by Berenbaum (1983) later on. Many plants produce a prototypical phytochemicals according to this theory that is balmily poisonous to phytophages and active in the plant autecologically or physiologically. Few insect species attack on plants with mild phytochemicals, thus reducing plant strength. Due to plant mutations, their recombinants produce novel, more noxious phytochemicals to occur in the plant system. Similar phytochemicals can occur independently in distantly associated plants. Insect attack is decreased because of toxic or repellent characteristics of the novel phytochemicals, so crop plants with higher potent defences are liked by the herbivores. Phyllobrotica species infest monogamously on Scutellaria species as reported by the cladograms (Farrell and Mitter 1990). Evidence is also available at the level of populations. Analysis reports of various populations of wild parsnip, Pastinaca sativa Linnaeus, and its pest the parsnip webworm, Depressaria pastinacella Duponchel, described trait matching between furanocoumarin-based chemical defence mechanisms in plants and cytochrome P450 monooxygenase-based insects’ detoxification profiles (Berenbaum and Zangerl 1998; Zangerl and Berenbaum 2003).
Coevolution is well defined in the brassicaceous plants and the pierid butterflies. The glucosinolate-myrosinase system developed in Brassicales 90 Myr before shows a major finding in anti-herbivore defence devices by plants. However, the Pierinae butterflies which used Fabales as host showed a metabolizing process as nitrile-specifier protein (NSP) and colonized the Brassicales. This resulted in increasing the species diversification rates in Pierinae as compared with that of their sister clade Coliadinae, whose members did not colonize Brassicales, thus lending strong support to the coevolutionary theory (Wheat et al. 2007; Edger et al. 2015).
1.4.2 Sequential Evolution Theory
The evolution of herbivores that comes after the evolution of plants, without affecting plant evolution significantly, is proposed by the theory of sequential evolution (Jermy 1976, 1984). So far reciprocal selective interactions between plants and insects have not been demonstrated. Insects select their hosts generally on the basis of chemical signals. Any alterations in chemical composition of plants or their chemosensory perception by herbivores may result in the development of new associations of insects and host plants. More evidence in support of the theory was presented by Labandeira (1998) and Janz et al. (2006), who showed that species richness in butterfly family Nymphalidae was strongly correlated with diversity of host use.
1.4.3 Diffuse Coevolution Theory
Diffuse coevolution or community coevolution theory suggests that in lieu of the pairwise reciprocal evolutionary interactions, coevolution must be regarded in a community context and not simply as a reciprocal interaction of two different species. Plant may be influenced by herbivore abundance, diseases, competing conspecifics, and plants of different species along with different host plants and insect pests and organisms at higher tropic levels (Fox 1988).
1.4.4 Geographic Mosaic Theory of Coevolution
This theory expresses that the coevolutionary system works at the level of populations rather than at a species level. Thompson (1994, 1999, 2005) stated that interspecific interactions frequently vary in biodiverse system. Additionally, populations vary in the extent to exhibit extreme specialization to one or more species. Gene flow among species, genetic drifting, novel trait selections, and extinction of some species reform the geographic mosaic of coevolution as the adaptations and specialization designs evolved locally, distributed to other population, or are finished. The result is a dynamic geographic pattern of coevolution between any two or more species. Across the Japanese islands, the coevolutionary relationship between the obligate seed predator; the camellia weevil, Camellia japonica Linnaeus; and its host plant , the Japanese camellia, Camellia japonica Linnaeus, serves as an interesting instance of geographic mosaic (Toju and Sota 2006; Toju et al. 2011). The thickness of camellia pericarp through which the female weevils inserted its ovipositors to deposit eggs into seeds linked with the length of the rostrum in female insects. In addition, the pericarp was significantly wider on islands abundant of weevils than on islands lacking weevils, and this characteristic was genetic.
1.5 Applied Aspects of Insect-Plant Correlations
For sustainable agriculture, the complex comprehension of insect-plant correlations has great applied importance. Growers desire to reduce crop damages brought about by insect pests and enhance crop yield. The fundamental strategies of insect-plant interplays are the main to obtain these aims as below:
1.5.1 Reproducing Resistant Cultivars Against Insect Pests
The most eco-friendly are the insect-resistant varieties which deliver economically practicable and ecologically durable choices for insect pest control . For more than a century, the research on breeding of insect-resistant crops has been carried out and bloomed as a new area of studies with the innovative research of Prof. R H Painter at Kansas State University, Manhattan, Kansas, USA, in the first half of the twentieth century (Painter 1951). An excellent early victory in using host plant resistance in pest control was achieved by grafting the European grapevines onto the resistant North American rootstocks (Painter 1951) against the grape phylloxera Daktulosphaira vitifoliae (Fitch) in France. In India, the research of Hussain and Lal (1940) found hairy cotton varieties resistant to jassid and resistant to cultivars, for instance, Punjab 4F, LSS, and 289 F/43, grown on large agriculture areas where jassid was a major pest by 1943. Reproducing stress-resistant plants has obtained great significance over the past 70 years with the participation of national and international agricultural research institutes along with seed producers from private sector. Dozens of insect-resistant varieties have been evolved globally and are cultivated largely for crop production sustainability (Panda and Khush 1995). Economically, insect-resistant genomes used in cropping recently rescue us more than US$2 billion every year (Smith and Clement 2012). Isolation and cloning of genomes for wanted chemical traits have accelerated the breeding of insect-resistant varieties. An advance comprehension of plant resistance reactions to herbivory is also required for further manipulation of generated resistance and plant-emitted volatile substances for breeding of insect pest-resistant cultivars (Sandhu and Arora 2013). Utilization of insect-resistant genomes from unconnected microbes and their chartering into elite germplasm is another fruit-bearing proposal which has established largely in applied research. From Bacillus thuringiensis , 20 Bt genes revealing resistance to lepidopteran and coleopteran pests have been merged into cotton, corn, potato, soybean, and other crops (Shera and Arora 2015).
1.5.2 Insect Pest Management Using Cultural Practices
For suppressing insect pest attack, cropping pattern and insect control measures such as cultural practices play a crucial role. The knowledge of host plant-insect pest association is beneficial to alter the plant ecosystem against the insect pest or in favour of the parasites and predators. Early crop sowing, for example, in Northern India, has been observed to decrease the gall midge and leaf folder attack in rice, shoot fly and headbug losses in sorghum and millets, white grub injury in groundnut, and aphid destruction in crucifers (Dhaliwal and Arora 2006). Enhanced intra-field diversity via interculturing, sowing of trap crops, or hedging rows led to minimum losses by many insect pests. Tomato interculturing in cabbage has been described to decrease the destruction of diamondback moth. Intercropping with sesame resulted in decreased penetration of okra roots by Meloidogyne incognita second-stage juveniles (J2) and delayed nematode maturation; it favoured development of M. incognita males and increased yields of okra and chickpea in field tests (Tanda and Atwal 1988). The largest effect of intercropping sesame with okra was when they were 15–30 cm apart. In pot tests, most J2 penetrated okra roots in sandy loam soil and fewest in clay soil. Trap crop of African marigold decreases the damage of fruit borer H. armigera in tomato (Srinivasan 1994). Napier grass and Napier millet work as trap crops for minimizing the attack of stem borer C. partellus in maize and sorghum (Khan 1999; Dhaliwal and Arora 2006). The natural enemies of herbivores may achieve larger population densities in polycultures as compared to monocultures, because polycultures often provide extra food sources, for example, honeydew, nectar and, pollen, and more refuges where insects can protect in the shade (Coll 1998). Among 130 predators observed in polycultures, more than 50% achieved higher population densities, as compared to monocultures, whereas less than 10% of them attained poor population densities when surveyed (Andow 1991).
1.5.3 Botanical Insecticides
Several crop plants have evolved tracks to diverse groups of chemicals to avoid their utilization by insect pests since many centuries. These biochemicals put forth behavioural, physiological, and biochemical impacts on insects, and some of them may even kill the sensitive insects. Bioinsecticides have been used since ancient times by humans. In various parts of the globe for centuries, many plants and their products such as neem, pyrethrum, Tephrosia, tobacco, derris, Ryania, and sabadilla have been used to save the farm crops, grains, and other commodities from the devastations of insects and other types of pests (Dhaliwal and Arora 2001). Plant chemicals have also acted as prototypes for the evolution and synthesis of Nobel categories of insecticides. Pyrethrum, isolated from the dried flowers of Chrysanthemum cinerariaefolium Linnaeus, has been utilized as an important insecticide since prehistoric times. It is a powerful mephitic against insects and relatively harmless to mammals; however, it is very photolabile (Casida 1973). Consequently, the chemical structure of pyrethrum was explicated to synthesize its analogues with better photostability. During the 1980s, several chemicals such as fenvalerate, deltamethrin, fluvalinate, and cyfluthrin rose as famous insecticides (Dhaliwal and Arora 2006). Likewise, similar synthetic analogues of nicotine, an important bioinsecticide named as neonicotinoids, was developed from tobacco and now largely utilized against a variety of sucking insect pests and mites (Simon-Delso et al. 2015). Accordingly, bioinsecticides have not only demonstrated beneficial directly in pest management but have also worked as a model for the modern groups of synthetic insecticides. As plants possess tens of thousands of similar biochemicals, the compass of their utility in insect pest control is almost unlimited.
1.5.4 Insect Pest Biocontrol
For enhancing the effectiveness of natural biocontrol and integrated pest management technology (IPMT) as we proposed, the significance of research on tritrophic and multitrophic interplays can scarcely be aggravated. Volatile substances released by plants are demonstrated to allure predators of herbivores (Weseloh 1981). Ramachandran et al. (1991) described that the parasitoid Microplitis demolitor Wilkinson was captivated by the volatile 3-octanone liberated by soybean, a host plant of the soybean looper, P. includens. Parasitoid was distinctly more apprehended by the volatile guaiacol, which was located in its hosts’ frass only; however, similar interactions may not benefit the predators. Hare (1992) observed a gamut of interactions between the predators and resistant plants such as synergistic to additive to none apparent and disruptive or antagonistic effects. A meta-analysis of 27 studies on interaction of resistant plant varieties and biocontrol of insect pests was carried out by Dhaliwal et al. (2004). About 29.6% showed antagonistic effect, whereas 25.9 and 33.3% were the cases of synergism and additive relationship, respectively. As understanding of multitrophic interactions enlarges, scientists and IPMT research workers should use it for insect pest control strategies (Verkerk 2004).
1.5.5 Use of Insect Behaviour in Control
In response to olfactory, visual, tactile, acoustic, and gustatory-sensory reaction from the host plant and ecosystem, insect behaviour is educed. For feeding and oviposition on host plants, cues are used by insects which can assist in the exploitation of similar behaviour, resulting in decreased crop losses (Foster and Harris 1997). In pest control, alluring and killing technique is the most famous behavioural manipulation manipulated. The attack of Japanese beetle Popillia japonica Newman is effectively controlled by a mixture of phenethyl propionate, eugenol, and geraniol, a female sex pheromone, and a food lure (Ladd et al. 1981). Foods baits have also been reported beneficial for monitoring and managing of tephritids. In the USA, protein hydrolysate-baited traps having insecticides have been proved effective against the Mediterranean fruit fly, Ceratitis capitata Wiedemann (Chambers 1978). Attraction and annihilation, which are innovative techniques, have resulted successfully against the apple maggot fly, Rhagoletis pomonella (Walsh). Using olfactory and visual stimuli, the female flies search apple plants and acceptable oviposition locations. Wooden spheres in red colour and layered with a sticky material at one trap tree−1 provided a better control of fruits from R. pomonella (Aluja and Prokopy 1993; Foster and Harris 1997).
1.5.6 Insect Control by Push-Pull Mechanism
The push-pull IPMT or stimulo-deterrent proposal in pest control is a new manipulation of the behavioural technology for the use of pulling and pushing constituents in sequence to repel the insect away from the principal host plant and towards the crop used for trapping, from where they may be detached later on (Khan et al. 1997; Cook et al. 2007). This approach has been effectively practicable for the control of stem borers such as C. partellus, Eldana saccharina Walker, Busseola fusca Fuller, and Sesamia inferens Hampson attacking maize and sorghum in Eastern and Southern Africa. The egg-laying female borers are repulsed from the main crop by repellent non-host intercrops, especially molasses grass, silver leaf desmodium, or green leaf desmodium (push), and like to deposit eggs on alluring trap crop plants, basically Napier grass or Sudan grass (pull). Interculturing of molasses grass with maize enhanced parasitization by Cotesia sesamiae Cameron in addition to decreasing the damages of stem borer (Khan et al. 2011). Push-pull plans have also been efficiently used against Helicoverpa in cotton, L. decemlineata in potato, rapeseed pollen beetle Brassicogethes aeneus (Fabricius) in oilseed rape, onion maggot Anthomyia antiqua (Meigen) in onions, striped pea leaf weevil Sitona lineatus (Linnaeus) in beans, western flower thrips, Frankliniella occidentalis (Pergande) in chrysanthemum, and bark beetles (Scolitidae) in conifers, along with many veterinary and medical insect pests (Cook et al. 2007).
1.5.7 Insect Biotype Control
Insect biotypes are insects that can live on and damage cultivars that have resistant genes. The cultivation of insect-resistant varieties regularly applies selection pressure on the aimed pest , which reacts by evolving new physiological and behavioural tools for feeding and multiplication on the resistant varieties. In breeding schemes for cultivar resistance, biotype selection is one of the key pressures experienced. This idea concerns gene-for-gene connection between the host plant-resistant gene and insect pest virulent gene. Aphids have 18 species out of 39, in which 2 or more biotypes have been observed (Sandhu and Arora 2013). Brown plant hopper, Nilaparvata lugens Stal, on rice (Brar et al. 2015) and Hessian fly, M. destructor, on wheat are the key pests in which biotype evolution has resulted in to collapse of resistance in the field (Aggrawal et al. 2014; Subramanyam et al. 2015). Insect resistance can be enhanced by sequential release of varieties, gene pyramiding/stacking, and gene rotation (Sandhu and Arora 2013). A better advance knowledge of insect-plant interactions is important for effective control of insect biotypes for higher resistant genotype stability.
1.5.8 Biocontrol of Weeds
The damages created by weeds are more than those done by insect pests to field crops, and the herbicide utilization surpasses that of insecticides in plant protection programs, so there is an immediate demand to reinforce biological management of weeds. Exotic weeds may be effectively controlled by monophagous or oligophagous insects from the origin of plant place. Prominent successful weed control instances are of shellmound prickly pear, Opuntia stricta (Haworth) Haworth, in Australia using small Argentinian moth, Cactoblastis cactorum Berg (Dodd 1940), and of giant Salvinia, Salvinia molesta D. S. Mitchell, in Papua New Guinea by the release of weevil Cyrtobagous salviniae Calder & Sands imported from Brazil (Room 1990). Extensive schemes on biocontrol of weeds in Hawaii using herbivorous insects and pathogens have been carried out, leading in effective management of 7 out of 21 aimed weeds and significant partial management of another 3 species (Gardner et al. 1995; McFadyen 2003). For the management of native weeds, native insects have also been artificially reared and released. Native coccids, Austrotachardia sp. and Tachardia sp., are released for the suppression of Cassinia sp., native woody shrubs in Australia (Holtkamp and Campbell 1995). For controlling the parasitic weeds, Orobanche spp. in the southern USSR, the stem-boring agromyzid Phytomyza orobanchia Kaltenbach has been conserved and released (Kroschel and Klein 1999).
1.5.9 Insect Pollinator Protection for Crop Production
Crop insect pollinators are crucial for effective pollination and reproduction by a large variety of angiosperms (Tanda 2019a, b, c, 2020, 2021a, b, c, d, e, f). Even self-pollinating cotton crop species may exhibit yield boosting close to an efficient pollinator and its ecosystem (Tanda 1983, 1984, 2020, 2021a, b, c, d, e, f). Coffee shrubs present great yield enhancement in areas with strong native or introduced bee pollinator abundance (Roubik 2002; Tanda 2021a, b). Several investigations on plant-pollinator processes have targeted on a single plant species and mostly one or a few closely related groups. However, new reports have demonstrated that pollinator networks are comparatively vague, due to spatiotemporal differences in foraging by pollinators (Herrera 1996; Waser 1998; Burkle and Alarcon 2011). It is crucial to comprehend the fundamentals of spatial and temporal difference in plant-pollinator interplay to reply questions in group structure and its functioning. It will also be beneficial in designing optimal pollinator’s protection measures (Burkle and Alarcon 2011). Environment and habitat alteration may disorder the coevality between the flower development period of plants and the active time of pollinators. Lack of nectar and pollen in crucial time may result in a decrease in pollinator’s abundance (Hoover et al. 2012; Sharma et al. 2014). An accurate knowledge of the flowering crop plant and insect pollinator interactions may be beneficial in avoiding pollinator reduction and sustainable agricultural production.
1.6 Conclusions
In the agricultural ecosystem, both insects and crop plants are ruling life forms and are engaged in complex interrelationships. For effective reproduction, a large number of flowering plants need the services of pollinators. The flower shape, size, colour, and scent all serve to allure bee pollinators, which generally feed on nectar and pollen developed by these crop plants. Additionally, about 50% of all insect populations are herbivorous and rely on green plants for food, shelter, and egg deposition. Accordingly, the plants have developed a surprising diversity of structural and biochemical barricades to save themselves from insect pests and plant diseases. The insects which are capable to manage these barriers via avoidance, detoxification, and sequestration can obtain sufficient food supplies with very small competition from other insect species. Since 400 million years ago, reciprocal modification and counterconversion between plants and insects have, thus, been the main procedure driving a steady enhancement in biodiversity of both these life forms. The investigations of such interrelationships between insects and flowering plants are of great practical significance for future sustainable agricultural productivity. Genomics, proteomics, and RNAi, advanced bio-techniques of molecular biology, provide exciting breaks for future exploration and accurate comprehension of insect-plant relationships. These biotechnological studies are important for the protection of ecosystem biodiversity and evolving insect-resistant cultivars for sustainable insect pests and weed control system.
References
Abe M, Matsuda K (2000) Feeding responses of four phytophagous lady beetle species (Coleoptera: Coccinellidae) to cucurbitacins and alkaloids. Appl Entomol Zool 35:257–264
Adams AS, Aylward FO, Adams SM et al (2013) Mountain pine beetles colonizing historical and native host trees are associated with a bacterial community highly enriched in genes contributing to terpene metabolism. Appl Environ Micobiol 79:3468–3475
Aggrawal R, Subramanyam S, Zhao C et al (2014) Avirulence effector discovery in a plant galling and plant parasitic arthropod, the Hessian fly (Mayetiola destructor). PLoS One 9(6):e100958
Agrawal AA, Konno K (2009) Latex: a model for understanding mechanisms, ecology, and evolution of plant defence against herbivory. Annu Rev Ecol Evol Syst 40:311–331
Alborn T, Turlings TCH, Jones TH et al (1997) An elicitor of plant volatiles from beet armyworm oral secretion. Science 276:945–949
Aluja M, Prokopy RJ (1993) Host odour and visual stimulation interaction during intratree host finding behaviour of Rhagoletis pomonella flies. J Chem Ecol 19:2671–2696
Andersen JF, Walding JK, Evans PH, Bowers WS, Feyereisen R (1997) Substrate specificity for the epoxidation of terpenoids and active site topology of house fly cytochrome P450 6A1. Chem Res Toxicol 10:156–164
Andow DA (1991) Vegetation diversity and arthropod population response. Annu Rev Entomol 36:561–586
Arabidopsis Genome Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408(6814):796–815
Arora R (2012) Co-evolution of insects and plants. In: Arora R, Singh B, Dhawan AK (eds) Theory and practice of integrated pest management. Scientific Publications, Jodhpur, pp 49–75
Arora R, Dhaliwal GS (2004) Biochemical bases of resistance in plants to insects. In: Dhaliwal GS, Singh R (eds) Host plant resistance to insects: concepts and applications. Panima Publications, New Delhi, pp 84–125
Arora R, Sandhu S (2017) Insect-plant interrelationships. In: Arora R, Sandhu S (eds) Breeding insect resistant crops for sustainable agriculture. Springer, Singapore, pp 1–44
Atwal AS (2000) Essentials of beekeeping and pollination. Kalyani Publications, New Delhi
Auclair JC (1963) Aphid feeding and nutrition. Annu Rev Entomol 8:439–490
Axelrod DI (1960) The evolution of flowering plants. In: Tax S (ed) Evolution after Darwin, vol I. The evolution of life. University of Chicago Press, Chicago, pp 227–305
Ayasse M, Schiesl FP, Paulus HF et al (2003) Pollinator attraction in a sexually deceptive orchid by means of unconventional chemicals. Proc R Soc Lond B 270:517–522
Baker HG, Baker I (1986) The occurrence and significance of amino acids in floral nectars. Plant Syst Evol 151:175–186
Banerjee MK, Kalloo G (1989) Role of phenols in resistance to tomato leaf curl virus, Fusarium wilt and fruit borer in Lycopersicon. Curr Sci 52:575–576
Barbehenn RV (2003) Antioxidants in grasshoppers: higher levels defend the midgut tissues of a polyphagous species than a graminivorous species. J Chem Ecol 29:683–702
Barbosa P, Schulz JC (1987) Insect outbreaks. Academic, San Diego
Beck SD (1965) Resistance of plants to insects. Annu Rev Entomol 10:207–232
Berenbaum MR (1983) Coumarins and caterpillars: a case for co-evolution. Evolution 37:163–179
Berenbaum MR (1991a) Comparative processing of allelochemicals in the papilionidae (Lepidoptera). Arch Insect Biochem Physiol 17:213–221
Berenbaum MR (1991b) Coumarins. In: Rosenthal GA, Berenbaum MR (eds) Herbivores: their interactions with secondary plant metabolites. Academic, London, pp 221–250
Berenbaum MR (1995) Turnabout is fairplay: secondary roles for primary compounds. J Chem Ecol 21:925–940
Berenbaum MR, Zangerl AR (1998) Chemical phenotype matching between a plant and its insect herbivore. Proc Natl Acad Sci U S A 95:13743–13748
Bernays EA (1986) Diet-induced head allometry among foliage chewing insects and its importance for graminivores. Science 231:495–497
Bernays EA, Chamberlain DJ (1980) A study of tolerance of ingested tannin in Schistocerca gregaria. J Insect Physiol 26:415–420
Bodnaryk RP (1992) Leaf epicuticular wax, an antixenotic factor in Brassicaceae that affects the rate and pattern of feeding of flea beetles, Phyllotreta cruciferae Goeze. Can J Plant Sci 72:1295–1303
Boone CK, Keefover-Ring K, Mapes AC et al (2013) Bacteria associated with a tree-killing insect reduce concentrations of plant defence compounds. J Chem Ecol 39:1003–1006
Bottger GT, Sheechan ET, Lukefahr MJ (1964) Relation of gossypol of cotton plants to insect resistance. J Econ Entomol 57:283–285
Brar DS, Sarao PS, Singh KS, Jena KK, Fujita D (2015) Biotechnological approaches for enhancing resistance to planthoppers in rice. In: Singh B, Arora R, Gosal SS (eds) Biological and molecular approaches in pest management. Scientific Publications, Jodhpur, pp 13–38
Bridges M, Jones AME, Bones AM et al (2002) Spatial organization of the glucosinolate myrosinase system in brassica specialist aphids is similar to that of the host plant. Proc R Soc Lond B 269:187–191
Brioschi D, Nadalini LD, Bengtsonb MH et al (2007) General up regulation of Spodoptera frugiperda trypsins and chymotrypsins allows its adaptation to soybean proteinase inhibitor. Insect Biochem Mol Biol 37:1283–1240
Bruce TJA (2015) Interplay between insects and plants: dynamic and complex interactions that have coevolved over millions of years but act in milliseconds. J Exp Bot 66:455–465
Bull DL, Ivie GW, Beier RC et al (1986) In vitro metabolism of a linear furanocoumarin (8-methoxypsoralen, xanthotoxin) by mixed-function oxidases of larvae of black swallowtail butterfly and fall armyworm. J Chem Ecol 12:885–892
Buntin DG, Chapin JW (1990) Biology of Hessian fly (Diptera: Cecidomyiidae) in the Southeastern United States: geographic variation and temperature-dependent phenology. J Econ Entomol 83:1015–1024
Burkle LA, Alarcon R (2011) The future of plant-pollinator diversity: understanding interaction networks across time, space and global change. Am J Bot 98:528–538
Casida JE (ed) (1973) Pyrethrum: the natural insecticide. Academic, New York
Chambers DL (1978) Attractants for fruit fly survey and control. In: Shorey HH, Mckelvey JJ (eds) Chemical control of insect behavior: theory and application. Wiley, New York, pp 327–344
Chapman RF (1974) The chemical inhibition of feeding by phytophagous insects. Bull Entomol Res 64:339–363
Chen MS, Fellers JP, Zhu YC et al (2006) A super-family of genes coding for secreted salivary gland proteins from the Hessian fly, Mayetiola destructor. J Insect Sci 6:12
Chen MS, Echegaray E, Whitworth RJ et al (2009) Virulence analysis of Hessian fly populations from Texas, Oklahoma and Kansas. J Econ Entomol 102:774–780
Chen M-S, Liu S, Wang H et al (2016) Genes expressed differentially in Hussian fly larvae feeding in resistant and susceptible plants. Int J Mol Sci 14(8):1324. https://doi.org/10.3390/ijms17081324
Chhabra KS, Kooner BS, Sharma AK et al (1990) Sources of resistance in chickpea: role of biochemical components on incidence of gram pod borer, Helicoverpa armigera (Hubner). Indian J Entomol 52:423–430
Chisholm ST, Cooker G, Day B et al (2006) Host-microbe interactions: shaping the evolution of the plant immune response. Cell 124:803–814
Chiu TL, Wen Z, Rupasinghe SG et al (2008) Comparative molecular modelling of an Anopheles gambiae CYP6Z1, a mosquito P450 capable of metabolizing DDT. Proc Natl Acad Sci U S A 105:8885–8860
Chow JK, Akhtar Y, Isman MB (2005) The effects of larval experience with a complex plant latex on subsequent feeding and oviposition by the cabbage looper moth: Trichoplusia ni (Lepidoptera: Noctuidae). Chemoecology 15:129–133
Chuang WP, Herde M, Ray S et al (2014) Caterpillar attack triggers accumulation of toxic maize protein RIP2. New Phytol 201:928–939
Chung SH, Rosa C, Scully ED et al (2013) Herbivore exploits orally secreted bacteria to suppress plant defences. Proc Natl Acad Sci U S A 110:15728–15733
Cianfrogna JA, Zangerl AR, Berenbaum MR (2002) Dietary and developmental influences on induced detoxification in an oligophage. J Chem Ecol 28:1349–1364
Coley PD (1983) Herbivory and defensive characteristics of tree species in a lowland tropical forest. Ecol Monogr 53:209–233
Coll M (1998) Parasitoid activity and plant species composition in intercropped systems. In: Pickett CH, Bugg RL (eds) Enhancing biological control: habitat management to promote natural enemies of agricultural pests. Univ California Press, Berkeley, pp 85–119
Cook SM, Khan ZR, Pickett JA (2007) The use of push-pull strategies in integrated pest management. Annu Rev Entomol 52:375–400
Cortes-Cruz M, Snook M, McMullen MD (2003) The genetic basis of C-glycosyl flavone B-ring modification in maize (Zea mays L.) silks. Genome 46:182–194
Cox PA (1991) Abiotic pollination: an evolutionary escape for animal-pollinated angiosperms. Philos Trans R Soc B 333:217–224
Crepet WL, Friis EM, Nixon KC (1991) Fossil evidence for the evolution of biotic pollination. Philos Trans R Soc B 333:187–195
Damle MS, Giri AP, Sainani MN et al (2005) Higher accumulation of proteinase inhibitors in flowers than leaves and fruits as a possible basis for differential feeding preference of Helicoverpa armigera on tomato (Lycopersicon esculentum Mill, Cv. Dhanashree). Phytochemistry 66:2659–2667
Danielson PB, Maclnytre RJ, Fogleman JC (1997) Molecular cloning of a family of xenobiotic inducible drosophilid cytochrome P450s: evidence for involvement in host-plant allelochemical resistance. Proc Natl Acad Sci U S A 94:10797–10802
De Leo F, Volpicella M, Licciulli F et al (2002) Plant-PIs: a database for plant protease inhibitors and their genes. Nucleic Acids Res 30:347–348
Dhaliwal GS, Arora R (2001) Role of phytochemicals in integrated pest management. In: Koul O, Dhaliwal GS (eds) Phytochemical biopesticides. Harwood, Amsterdam, pp 97–118
Dhaliwal GS, Arora R (2006) Integrated pest management: concept and approaches. Kalyani Publications, New Delhi
Dhaliwal GS, Singh R, Jindal V (2004) Host plant resistance and insect pest management: progress and potential. In: Dhaliwal GS, Singh R (eds) Host plant resistance to insects. Panima, New Delhi, pp 517–558
Dimock MH, Kennedy GG (1983) The role of glandular trichomes in the resistance of Lycopersicon hirsutum f. glabratum to Heliothis zea. Entomol Exp Appl 33:263–268
Dixon RA, Strack D (2003) Phytochemistry meets genome analysis, and beyond. Phytochemistry 62:815–816
Dodd AP (1940) The biological campaign against prickly-pear. Commonwealth Prickly Pear Board, Brisbane
Dunaevsky YE, Elpidina EN, Vinokurov KS et al (2005) Protease inhibitors in improvement of plant resistance to pathogens and insects. Mol Biol 39:702–708
Dussourd DE (1995) Entrapment of aphids and whiteflies in lettuce latex. Ann Entomol Soc Am 88(2):163–172
Edger PP, Heidel-Fischer HM, Bekaert M et al (2015) The butterfly plant arms-race by gene and genome duplications. Proc Natl Acad Sci U S A 112:8362–8366
Ehrlich PR, Raven PH (1964) Butterflies and plants: a study in co-evolution. Evolution 18:586–608
Eigenbrode SD (2004) The effects of plant epicuticular waxy blooms on attachment and effectiveness of predatory insects. Arthropod Struct Dev 33:91–102
Eigenbrode SD, Espelie KE (1995) Effects of plant epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194
Eigenbrode SD, Kabalo NN, Stoner KA (1999) Predation, behavior and attachment by Chrysoperla plarabunda larvae on Brassica oleracea with different surface waxblooms. Entomol Exp Appl 90:225–235
Enayati AA, Ranson H, Hemingway J (2005) Insect glutathione transferases and insecticides resistance. Insect Mol Biol 14:3–8
Facchini PJ (2001) Alkaloid biosynthesis in plants: biochemistry, cell biology, molecular regulation, and metabolic engineering applications. Annu Rev Plant Physiol 52:29–66
Faegri K, Pijl LV (1971) The principles of pollination ecology. Pergamon Press, New York
Fahey JW, Zalcmann AT, Talalay P (2001) The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 56:5–51
Fahn A (2000) Structure and function of secretory cells. In: Hallahan DL, Gray JC (eds) Plant trichomes. Academic, New York, p 37
Farrell B, Mitter C (1990) Phylogenesis of insect/plant interactions: have Phyllobrotica leaf beetles (chrysomelidae) and the lamiales diversified in parallel? Evolution 44:1389–1403
Fatouros NE, Broekgaarden C, Bukovinszkine’Kiss G et al (2012) Plant volatiles induced by herbivore egg deposition affect insects of different trophic levels. PLoS One 7(8):e43607. https://doi.org/10.1371/journal.pone.0043607
Felton GW (2005) Indigestion is a plant’s best defence. Proc Natl Acad Sci U S A 102:18771–18772
Felton GW, Broaduray RM, Duffey SS (1989) Inactivation of protease inhibitor activity by plant derived quinones, complications for host-plant resistance against noctoid herbivore. J Insect Physiol 35:981–990
Ferry RL, Cuthbert FP Jr (1975) A tomato fruit worm antibiosis in Lycopersicon. Hortic Sci 10:46
Feyereisen R (2006) Evolution of insect P450. Biochem Soc Trans 34:1252–1255
Foster SP, Harris MO (1997) Behavioral manipulation methods for insect pest-management. Annu Rev Entomol 42:123–146
Fox LR (1988) Diffuse co-evolution within complex communities. Ecology 69:906–907
Fraenkel GS (1959) The raison d’etre of secondary plant substances. Entomol Exp Appl 12:473–486
Francis G, Kerem Z, Makkar HPS et al (2002) The biological action of saponins in animal systems: a review. Br J Nutr 88:587–605
Francis F, Vanhaelen N, Haubruge E (2005) Glutathione S-transferases in the adaptation to plant secondary metabolites in the Myzus persicae aphid. Arch Insect Biochem Physiol 58:166–174
Frelichowski JE Jr, Juvik JA (2001) Sesquiterpene carboxylic acids from a wild tomato species affect larval feeding behavior and survival of Helicoverpa zea and Spodoptera exigua (Lepidoptera: Noctuidae). J Econ Entomol 94:1249–1259
Frey M, Schullehner K, Dick R et al (2009) Benzoxazinoid biosynthesis, a model for evolution of secondary metabolic pathways in plants. Phytochemistry 70:1645–1651
Furstenberg-Hagg J, Zagrobelnby M, Bak S (2013) Plant defence against herbivores. Int J Mol Sci 14:10242–10297
Galai N, Salles J-M, Settele J et al (2009) Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol Econ 68:810–821
Gardner DE, Smith CW, Markin GP (1995) Biological control of alien plants in natural areas of Hawaii. In: Delfosse ES, Scott RR (eds) Proceedings of the 8th international symposium on biological control of weeds. CSIRO, Melbourne, pp 35–40
Gershenzon J, Croteau R (1991) Terpenoids. In: Rosenthal GS, Berenbaum MR (eds) Herbivores: their interaction with secondary plant metabolites. Academic, London, pp 165–220
Geyter ED, Lambert E, Geelen D et al (2007) Novel advances with plant saponins as natural insecticides to control pest insects. Pest Technol 1:96–105
Gieselhardt S, Yoneya K, Blenn B et al (2013) Egg laying of cabbage white butterfly (Pieris brassicae) on Arabidopsis thaliana affects subsequent performance of the larvae. PLoS One. https://doi.org/10.1371/journal.pone.0056991
Glas JJ, Schimmel BCJ, Alba JM et al (2012) Plant glandular trichomes as targets for breeding or engineering of resistance to herbivores. Int J Mol Sci 13:17077–17103
Gorb EV, Gorb SN (2002) Attachment ability of the beetle Chrysolina fastuosa on various plant surfaces. Entomol Exp Appl 105:13–28
Green MB, Hedin PA (1986) Natural resistance of plants to pests: role of allelochemicals. In: ACS Symp Ser 296, American Chemical Society, Washington, DC
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Hammer TJ, Bowers MD (2015) Gut microbes may facilitate insect herbivory of chemically defended plants. Oecologia 179:1–14
Hanover JW (1975) Physiology of tree resistance to insects. Annu Rev Entomol 20:75–95
Harborne JB (1993) Introduction to ecological biochemistry. Academic, London
Harborne JB (1994) Phenolics. In: Mann J, Davidson RS, Hobbs JB, Banthorpe DB, Harborne JB (eds) Natural products: their chemistry and biological significance. Longman, Harlow, pp 362–388
Hare DJ (1992) Effects of plant variation on herbivore-enemy interactions. In: Fritz RS, Simms EL (eds) Plant resistance to herbivores and pathogens. Univ of Chicago Press, Chicago, pp 278–298
Harris P (1974) A possible explanation of plant yield increases following insect damage. Agro Ecosyst 1:219–225
Hatchett JH, Gallun RL (1970) Genetics of the ability of the Hessian fly, Mayetiola destructor to survive on wheat having different genes for resistance. Ann Entomol Soc Am 63:1400–1407
Herrera CM (1996) Floral traits and plant adaptation to insect pollinators: a devil’s advocate approach. In: Lloyd DG, Barrett SCH (eds) Floral biology: studies on floral evolution in animal pollinated plants. Chapman & Hall, New York, pp 65–87
Hilker M, Meiners T (2006) Early herbivore alert: insect eggs induce plant defence. J Chem Ecol 32:1379–1397
Hill DL (1997) The economic importance of insects. Chapman & Hall, London
Hogenhout SA, Bos JIB (2011) Effector proteins that modulate plant-insect interactions. Curr Opin Plant Biol 14:422–428
Hogenhout SA, Vabder Hoorn RAL, Terauchi R et al (2009) Emerging concepts in effector biology of plant-associated organisms. Mol Plant-Microbe Interact 22:115–122
Holtkamp RH, Campbell MH (1995) Biological control, of Cassinia spp. (Asteraceae). In: Delfosse ES, Scott RR (eds) Proceedings of the 8th international symposium on biological control of weeds. CSIRO, Melbourne, pp 447–450
Hoover SER, Ladly JJ, Shchepetkine AR et al (2012) Warming, CO2, and nitrogen deposition interactively affect a plant-pollinator mutualism. Ecol Lett 15:227–234
Hopkins RJ, Van Dam NM, Van Loon JJA (2009) Role of glucosinolates in insect-plant relationships and multitrophic interactions. Annu Rev Entomol 54:57–83
House HL (1961) Insect nutrition. Annu Rev Entomol 6:13–26
Huang T, Jander G, De Vos M (2011) Non-protein amino acids in plant defence against insect herbivores: representative cases and opportunities for further functional analysis. Phytochemistry 72:1531–1537
Huber M, Epping J, Gronover CS et al (2016) A latex metabolite benefits plant fitness under root herbivore attack. PLoS Biol 14(1):e1002332. https://doi.org/10.1371/journal.pbio.1002332
Hussain MA, Lal KB (1940) The bionomics of Empoasca devastens (Distant) on some varieties of cotton in the Punjab. Indian J Entomol 2:123–136
Janz N, Nylin S, Wahlberg N (2006) Diversity begets diversity: host expansions and the diversification of plant-feeding insects. BMC Evol Biol 6:4. https://doi.org/10.1186/1471-2148-6-4
Jeffree CE (1986) The cuticle, epicuticular waxes and trichomes of plants, with reference to their structure, functions and evolution. In: Juniper BE, Southwood TRE (eds) Insects and the plant surface. E. Arnold, London, pp 23–64
Jermy T (1976) Insect-host plant relationship-coevolution or sequential evolution? Symp Biol Hung 16:109–113
Jermy T (1984) Evolution of insect/plant relationships. Am Nat 124:609–630
Johanson B (1953) The injurious effects of the hooked epidermal hairs of the French beans (Phaseolus vulgaris L.) on Aphis craccivora Koch. Bull Entomol Res 44:779–788
Johnson HB (1975) Plant pubescence: an ecological perspective. Bot Rev 41:233–258
Johnson MT (2011) Evolutionary ecology of plant defences against herbivores. Funct Ecol 25:305–311
Jongsma MA, Bakker PL, Peters J et al (1995) Adaptation of Spodoptera exigua larvae to plant proteinase-inhibitors by induction of gut proteinase activity insensitive to inhibition. Proc Natl Acad Sci U S A 92:8041–8045
Kashyap RK (1983) Studies on resistance behavior of tomato genotypes against fruit borer. Dissertation, Haryana Agricultural University, Hisar
Kaur M, Singh K, Rup PJ et al (2006) A tuber lectin from Arisaema helleborifolium Schott with anti-insect activity against melon fruit fly Bactrocera cucurbitae (Coquillett) and anti-cancer effect on human cancer cell lines. Arch Biochem Biophys 445:156–165
Kazana E, Pope TW, Tibbles L et al (2007) The cabbage aphid: a walking mustard oil bomb. Proc R Soc Lond B 274:2271–2277
Kearns CA, Inouye DW, Waser NM (1998) Endangered mutualisms: the conservation of plant-pollinator interactions. Annu Rev Ecol Syst 29:83–112
Kennedy CEJ (1986) Attachment may be a basis for specialization in oak aphids. Ecol Entomol 11:291–300
Khan ZR (1999) Habitat management strategies for control of insect pests in Africa. In: Dhaliwal GS, Arora R, Dhawan AK (eds) Emerging trends in sustainable agriculture. Commonwealth Publications, New Delhi, pp 187–197
Khan ZR, Ampong-Nyarko K, Chiliswa P et al (1997) Inter-cropping increases parasitism of pests. Nature 388:631–632
Khan ZR, Midega C, Pittchar J et al (2011) Push-pull technology: a conservation agriculture approach for integrated management of insect pests, weeds and soil health in Africa. Int J Agric Sustain 9:162–170
Kim JH, Lee BW, Schroeder FC et al (2008) Identification of indole glucosinolate breakdown products with antifeedant effects of Myzus persicae (green peach aphid). Plant J 54:1015–1026
Klein AM, Vaissiere BE, Cane JH et al (2007) Importance of pollinators in changing landscapes for world crops. Proc R Soc Lond B 274:303–313
Krishnan N, Sehnal F (2006) Compartmentalization of oxidative stress and antioxidant defence in the larval gut of Spodoptera littoralis. Arch Insect Biochem Physiol 63:1–10
Kritsky G (2001) Darwin’s Madagascan hawk moth prediction. Am Entomol 37:206–210
Kroschel J, Klein O (1999) Biological control of Orobranche spp. with Phytomyza orobranchia Kalt, a review. In: Kroschel J, Abderabihi M, Betz H (eds) Advances in parasitic weed control at on-farm level, vol 2. Joint action to control Orobranche in the WANA region. Mardarof Verlag, Weikersheim, pp 135–159
Labandeira CC (1998) Early history of arthropod and vascular plant associations. Annu Rev Earth Planet Sci 26:329–377
Labandeira CC (2013) A paleobiologic perspective on plant-insect interactions. Curr Opin Plant Biol 16:414–421
Ladd TL, Klein MG, Tumlison JH (1981) Phenethyl propionate+eugenol+geraniol (3: 7: 3) and Japonilure: a highly effective joint lure for Japanese beetles. J Econ Entomol 74:665–667
Lambrix V, Reichelt M, Mitchell-Olds T et al (2001) The Arabidopsis epithiospecifier protein promotes the hydrolysis of glucosinolates to nitriles and influences Trichoplusia ni herbivory. Plant Cell 13:2793–2807
Lawton JH (1983) Plant architecture and the diversity of phytophagous insects. Annu Rev Entomol 28:23–39
Lee YL, Kogan M, Larsen JR (1986) Attachment of the potato leafhopper to soybean plant surfaces as affected by morphology of pretarsus. Entomol Exp Appl 42:101–108
Li Q, Eigenbrode SD, Stringam GR et al (2000) Feeding and growth of Plutella xylostella and Spodoptera eridania on Brassica juncea with varying glucosinolate concentrations and myrosinase activities. J Chem Ecol 26:401–2419
Liener IE (1991) Lectins. In: Rosenthal GA, Berenbaum MR (eds) Herbivores: their interactions with secondary plant metabolites. Academic, London, pp 327–354
Liu X, Bai J, Li H et al (2007) Gene expression of different wheat genotypes during attack by virulent and avirulent Hessian fly (Mayetiola destructor) larvae. J Chem Ecol 33:2171–2194
Liu X, Williams CE, Nemacheck JA et al (2010) Reactive oxygen species are involved in plant defense against a gall midge. Plant Physiol 152:985–999
Lorenzen JH, Belbyshev NE, Lafta AM et al (2001) Resistant potato selections contain leptine and inhibit development of Colorado potato beetle (Coleoptera: Chrysomelidae). J Econ Entomol 94:1260–1267
Louda S, Mole S (1991) Glucosinolates: chemistry and ecology. In: Rosenthal GA, Berenbaum MR (eds) Herbivores: their interactions with secondary plant metabolites. Academic, London, pp 124–164
Ma R, Cohen MB, Berenbaum MR et al (1994) Black swallowtail (Papilio polyxenes) alleles encode cytochrome P450s that selectively metabolize linear furanocoumarins. Arch Biochem Biophys 310:332–340
Mao YB, Cai WJ, Wang JW et al (2007) Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25:1307–1313
Martin FA, Richard CA, Hensley SD (1975) Host resistance to Diatraea saccharalis (F) relationship of sugarcane internode hardness to larval damage. Environ Entomol 4:687–688
Martin JS, Martin MM, Bernays EA (1987) Failure of tannic acid to inhibit digestion or reduce digestibility of plant protein in gut fluids of insect herbivores: implications for theories of plant defence. J Chem Ecol 13:605–621
Martin JP, Beyerlein A, Dacks AM et al (2011) The neurobiology of insect olfaction: sensory processing in a comparative context. Prog Neurobiol 95:427–447
Mason CJ, Couture JJ, Raffa KF (2014) Plant associated bacteria degrade defence chemicals and reduce their adverse effects on an insect defoliator. Oecologia 175:901–910
McFadyen REC (2003) Biological control of weeds using exotic insects. In: Koul O, Dhaliwal GS (eds) Predators and parasitoids. Taylor & Francis, London, pp 163–183
McLaughlin LA, Niazi U, Bibby J et al (2008) Characterization of inhibitors and substrates of Anopheles gambiae CYP6Z2. Insect Mol Biol 17:125–135
Meisner J, Navon A, Zur M et al (1977) The response of Spodoptera littoralis larvae to gossypol incorporated in artificial diet. Environ Entomol 6:243–244
Mithofer A, Boland W (2012) Plant defence against herbivores: chemical aspects. Annu Rev Plant Biol 63:431–450
Mohan P, Singh R, Narayanan S et al (1994) Relation of gossypol-gland density with bollworm incidence and yield in tree cotton (Gossypium arboreum). Indian J Agric Sci 64:691–696
Muller C, Brakefield PM (2003) Analysis of a chemical defence in sawfly larvae: easy bleeding targets predatory wasps in late summer. J Chem Ecol 29:2683–2694
Nabhan GP, Buchmann SL (1997) Services provided by pollinators. In: Daily GC (ed) Nature’s services: societal dependence on natural ecosystems. Island Press, Washington, DC, pp 133–150
Nepi M, Guarnieri M, Pacini E (2003) ‘Real’ and feed pollen of Lagerstroemia indica: ecophysiological differences. Plant Biol 5:311–314
Nikoh N, Hosokawa T, Oshima K et al (2011) Reductive evolution of bacterial genome in insect gut environment. Genome Biol Evol 3:702–714
Nitao JK (1989) Enzymatic adaptation in a specialist herbivore for feeding on furanocoumarin containing plants. Ecology 70:629–625
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321–326
Owen DF (1980) How plants may benefit from the animals that eat them? Oikos 35:230–235
Painter RH (1951) Insect resistance in crop plants. University of Kansas Press, Lawrence
Panda N, Khush GS (1995) Host plant resistance to insects. CABI, Wallingford
Pappers SM, Van Dommelon H, Van der Velde G et al (2001) Differences in morphology and reproductive traits of Galerucella nymphaeae from four host plant species. Entomol Exp Appl 99:183–191
Parde VD, Sharma HC, Kachole MS (2010) In vivo inhibition of Helicoverpa armigera gut pro-proteinase activation by non host plant protease inhibitors. J Insect Physiol 56:1315–1324
Parde VD, Sharma HC, Kachole MS (2012) Potential of proteinase inhibitors in wild relatives of pigeonpea against cotton bollworm/legume pod borers, Helicoverpa armigera. Am J Plant Sci 3:627–635
Parmar BS, Walia S (2001) Prospects and problems of phytochemical biopesticides. In: Koul O, Dhaliwal GS (eds) Phytochemical biopesticides. Harwood, Amsterdam, pp 133–210
Payne WW (1978) A glossary of plant hair terminology. Brittonia 30:239–255
Pellmyr O, Krenn HW (2002) Origin of a complex key innovation in an obligate insect-plant mutualism. Proc Natl Acad Sci U S A 99:5498–5502
Pfalz M, Vogel H, Kroymann J (2009) The gene controlling the Indole Glucosinolate Modifier 1 quantitative trait locus alters indole glucosinolate structures and aphid resistance in Arabidopsis. Plant Cell 21:985–999
Pillemer EA, Tingey WM (1978) Hooked trichomes and resistance of Phaseolus vulgaris to Empoasca fabae (Harris). Entomol Exp Appl 24:83–94
Platt AW, Farstad CM (1946) The reaction of wheat varieties to wheat stem sawfly attack. Sci Agric 26:231–247
Proctor M, Yeo F, Lack A (1996) The natural history of pollination. Harper Collins, London
Ram P, Singh R, Dhaliwal GS (2004) Biophysical bases of resistance in plants to insects. In: Dhaliwal GS, Singh R (eds) Host plant resistance to insects: concepts and applications. Panima Publications, New Delhi, pp 42–83
Ramachandran R, Norris DM, Phillips JK et al (1991) Volatiles mediating plant-herbivore-natural enemy interactions: soybean looper frass volatiles, 3-octanone and guaiacol, as kairomones for the parasitoid, Microplitis demolitor. J Agric Food Chem 39:2310–2317
Ramirez BW (1970) Host specificity of fig wasps (Agaonidae). Evolution 24:681–691
Rao NV, Reddy AS, Ankaish R et al (1990) Incidence of whitefly (Bemisia tabaci) in relation to leaf characters of upland plant cotton (Gossypium hirsutum). Indian J Agric Sci 60:619–624
Rector BG, Liang GM, Guo Y (2003) Effect of maysin on wild–type, deltamethrin-resistant and Bt-resistant Helicoverpa armigera (Lepidoptera: Noctuidae). J Econ Entomol 96:909–913
Riffell JA, Lei H, Christensen TA et al (2009) Characterization and coding of behaviorally significant odor mixtures. Curr Biol 19:335–340
Room PM (1990) Ecology of a simple plant-herbivore system: biological control of Salvinia. Trends Ecol Evol 5:74–79
Rosenthal GA (1991) Nonprotein amino acids as protective phytochemicals. In: Rosenthal GA, Berenbaum MR (eds) Herbivores: their interactions with secondary plant metabolites. Academic, London, pp 1–34
Rosenthal GA, Berenbaum MR (eds) (1991) Herbivores: their interactions with secondary plant metabolites. Academic, London
Roubik DW (2002) The value of bees to the coffee harvest. Nature 417:708
Roulston TAH, Cane JH, Buckmann SL (2000) What governs protein content of pollen: pollinator preferences, pollen-pistil interactions, or phylogeny? Ecol Monogr 70:617–643
Ruzicka L (1953) Isoprene rule and biogenesis of terpenic compounds. Experientia 9:357–367
Sadras VO, Felton GW (2010) Mechanism of cotton resistance to arthropod herbivory. In: Stewart JM, Oosterhius D, Heitholt JJ et al (eds) Physiology of cotton. Springer, London, pp 213–228
Sahoo BK, Patnaik MP (2003) Effect of biochemicals on the incidence of pigeonpea pod borers. Indian J Plant Prot 31:105–108
Sandhu SK, Arora R (2013) Breeding for insect resistance in crop plants. In: Dhawan AK, Singh B, Bhullar MB, Arora R (eds) Integrated pest management. Scientific Publications, Jodhpur, pp 267–300
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology. Oxford University Press, Oxford
Schuhegger R, Nafisi M, Mansourova M et al (2006) CYP71B15 (PAD3) catalyzes the final step in camalexin biosynthesis. Plant Physiol 141:1248–1254
Schuler M (1996) The role of cytochrome P450 monooxygenases in plant-insect interactions. Plant Physiol 112:1411–1419
Schumutterer H (ed) (1995) The neem tree, Azadirachta indica A. Juss. and other meliaceous plants: source of unique products for integrated pest management, medicine, industry and other purposes. VCH, Weinheim
Scott MI, Thaler SJ, Scott GF (2010) Response of a generalist herbivore Trichoplusia ni to jasmonate-mediated induced defence in tomato. J Chem Ecol 36:490–499
Seybold SJ, Huber DPW, Lee JC et al (2006) Pine monoterpenes and pine bark beetles: a marriage of convenience for defence and chemical communication. Phytochem Rev 5:143–178
Sharma S, Arora R, Singh B (2014) Impact of climate change on agriculturally important insects. J Insect Sci 27:159–188
Shera PS, Arora R (2015) Biointensive integrated pest management for sustainable agriculture. In: Singh B, Arora R, Gosal SS (eds) Biological and molecular approaches in pest management. Scientific Publications, Jodhpur, pp 373–429
Simon-Delso N, Amaral-Rogers X, Belzunces LP et al (2015) Systemic insecticides (neonicotinoids and fipronil): trends, uses, mode of action and metabolites. Environ Sci Pollut Res 22:5–34
Singh R, Agarwal RA (1988) Role of biochemical components of resistant and susceptible cotton and okra in ovipositional preference of cotton leafhopper. Proc Indian Acad Sci (Anim Sci) 97:545–550
Sintim HO, Tashiro T, Motoyama N (2009) Response of the cutworm Spodoptera litura to sesame leaves or crude extracts in diet. J Insect Sci 9:52
Smith CM, Clement SL (2012) Molecular basis of plant resistance to arthropods. Annu Rev Entomol 57:309–328
Sogawa K, Pathak MD (1970) Mechanisms of brown planthopper (Hemiptera: Delphacidae) resistance of Mudgo variety of rice. Appl Entomol Zool 5:145–148
Springer TL, Kindler SD, Sorenson EL (1990) Comparison of pod-wall characteristics with seed damage and resistance to alfalfa seed chalcid (Hymenoptera: Eurytomidae) in Medicago species. Environ Entomol 19:1614–1617
Srinivasan K (1994) Recent trends in insect pest management in vegetable crops. In: Dhaliwal GS, Arora R (eds) Trends in agricultural insect pest management. Commonwealth Publications, New Delhi, pp 345–372
Steehius NM, van Gelder WMJ (1985) Tomato with whitefly resistance is nutritionally safe. Zaasbelangen 39:191–192
Steppuhn A, Baldwin IT (2007) Resistance management in a native plant: nicotine prevents herbivores from compensating for plant protease inhibitors. Ecol Lett 10:499–511
Stevens JL, Snyder MJ, Koener JF et al (2000) Inducible P450s of the CYP9 family from larval Manduca sexta midgut. Insect Biochem Mol Biol 30:559–568
Strong DR, Lawton JH, Southwood TRE (1984) Insects on plants: community patterns and mechanisms. Blackwell, London
Stuart JJ, Chen MS, Shukle R et al (2012) Gall midges (Hessian flies) as plant pathogens. Annu Rev Phytopathol 50:339–357
Subramanyam S, Smith DF, Clemens JC et al (2008) Functional characterization of HFR1, a high mannose N-glycan-specific wheat lectin induced by hessian fly larvae. Plant Physiol 147:412–426
Subramanyam S, Sardesai N, Minocha SC et al (2015) Hessian fly larval feeding triggers enhanced polyamine levels in susceptible but not resistant wheat. BMC Plant Biol 15:3. https://doi.org/10.1186/s12870-014-0396-y
Sutherland TD, Unnithan GC, Anderson JF et al (1998) Cytochrome P450 terpenoid hydroxylase linked to the suppression of insect juvenile hormone synthesis. Proc Natl Acad Sci U S A 95:12884–12889
Talekar NS, Tengkano W (1993) Mechanism of resistance to bean fly (Diptera: Agromyzidae) in soybean. J Econ Entomol 86:981–985
Tallamy DW, Stull J, Ehresman NP et al (1997) Cucurbitacins as feeding and oviposition deterrents to insects. Environ Entomol 26:678–683
Tamiru A, Bruce TJA, Woodcock CM et al (2011) Maize landraces recruit egg and larval parasitoids in response to egg deposition by a herbivore. Ecol Lett 14:1075–1083
Tanda AS (1983) Assessing the role of honey bees in a field of Asiatic cotton (Gossypium arboreum L.). Am Bee J 123:593–594
Tanda AS (1984) Bee pollination increases yield of 2 interplanted varieties of Asiatic cotton (Gossypium arboretum L.). Am Bee J 124(7):539–540
Tanda AS (2019a) Entomophilous crops get better fruit quality and yield: an appraisal. Indian J Entomol 81(2):227–234
Tanda AS (2019b) Floral biology, foraging behavior and efficiency of European honey bee (Apis mellifera) in bitter gourd (Momordica charantia L.) pollination at Sydney Australia. Bee World. Submitted
Tanda AS (2019c) Entomofaunal effect enhances the quality and quantity in okra (Abelmoschus esculenlum L.) plantation. Indian J Entomol 81(1):16–17
Tanda AS (2020) Entpollinatology—a strong relationship between plants and insects for crop improvement. In: 6th Edition of Global conference on plant sciences and molecular biology (GPMB 2020) to be held on September 10-12, 2020, at Paris, France (Accepted, May 26, 2020)
Tanda AS (2021a) Why insect pollinators important in crop improvement?. Indian J Entomol (Accepted)
Tanda AS (2021b) Insect pollinators matter in sustainable world food production. Indian J Entomol (Accepted)
Tanda AS (2021c) Urbanization and its impact on native pollinators. In: The 1st international electronic conference on entomology will be held on 1st–15th July 2021 virtually
Tanda AS (2021d) Native bees are important and need immediate conservation measures: a review. In: The 1st international electronic conference on entomology will be held on 1st–15th July 2021 published in the Proceedings 1 July 2021, 68, x. https://sciforum.net/manuscripts/10523/manuscript.pdf
Tanda AS (2021e) Wild bees and their conservation. Indian J Entomol (Accepted)
Tanda AS (2021f) Biofloral phenology, Foraging Behaviour and entpollinatological effect of honey bees in Pomegranate (Punica granatum) fruit quality and yield. J Hortic 08:2
Tanda AS (2021g) Insect resistance and host plant relations: a milestone in sustainable crop production. Indian J Entomol (Accepted)
Tanda AS, Atwal AS (1988) Effect of sesame intercropping against the root-knot nematode (Meloidogyne Incognita) in okra. Nematologica 34(4):484–492
Tanda AS, Atwal AS, Bajaj YPS (1988) Antagonism of sesame to the root-knot nematode (Meloidogyne Incognita) on okra in tissue culture. Nematologica 34(1):78–87
Tanda AS, Atwal AS, Bajaj YPS (1989) In vitro inhibition of root-knot nematode Meloidogyne incognita by sesame root exudate and its amino acids. Nematologica 35:115–124
Thayumanavan B, Velusamy R, Sadasivam S et al (1990) Phenolic compounds, reducing sugars and free amino acids in rice leaves of varieties resistant to rice thrips. Int Rice Res Newsl 15:14–15
Thien LB, Azuma H, Kawano S (2000) New perspectives on the pollination biology of basal angiosperms. Int J Plant Sci 161:S225–S235
Thompson JN (1994) The co-evolutionary process. Chicago University Press, Chicago
Thompson JN (1999) Specific hypotheses on the geographic mosaic of co-evolution. Am Nat 153:S1–S14
Thompson JN (2005) Co-evolution: the geographic mosaic of co-evolutionary arms race. Curr Biol 15(24):R992–R994
Tingey WM (1984) Glycoalkaloids as pest resistance factors. Am Potato J 61:157–167
Toju H, Sota T (2006) Imbalance of predator and prey armament; Geographic clines in phenotypic interface and natural selection. Am Nat 167:105–117
Toju H, Abe H, Ueno S et al (2011) Climatic gradients of arms race coevolution. Am Nat 177:562–573
Traw MB, Dawson TE (2002) Differential induction of trichomes by three herbivores of black mustard. Oecologia 131:526–532
Uthamasamy S (1996) Biochemical basis of resistance to insects in cotton, Gossypium spp. In: Ananthakrishnan TN (ed) Proceedings of national symposium on biochemical bases of host plant resistance to insects. National Academy of Agricultural Sciences, New Delhi, pp 15–37
Vail SG (1994) Overcompensation, plant-herbivore mutualism, and mutualistic co-evolution—a reply to Mathews. Am Nat 144:534–536
Van Lenteren JC, Hua LZ, Kamerman JW et al (1995) The parasite host relationship between Encarsia Formosa (Hym., Aphelinidae) and Trialeurodes vaporariorum (Hom., Aleyrodidae). XXVI. Leaf hairs reduce the capacity of Encarsia to control greenhouse whitefly on cucumber. J Appl Entomol 119:553–559
Velthius HWW (1992) Pollen digestion and the evolution of sociality in bees. Bee World 127:1383–1389
Verkerk RHJ (2004) Manipulation of tritrophic interactions for IPM. In: Koul O, Dhaliwal GS, Cuperus GW (eds) Integrated pest management: potential, constraints and challenges. CABI, Wallingford, pp 55–72
Vidyachandra B, Roy JK, Bhaskar D (1981) Chemical difference in rice varieties susceptible or resistant to gall midges and stem borers. Int Rice Res Newsl 6(2):7–8
Vilkova NA, Kunzetsova TL, Ismailov AL et al (1988) Effect of cotton cultivars with high content of gossypol on development of cotton bollworm Helicoverpa armigera (Hubner) (Lepidoptera: Noctuidae). Entomol Obozr 4:689–698
Volpicella M, Ceci LR, Cordewener J et al (2003) Properties of purified gut trypsin from Helicoverpa zea adapted to proteinase inhibitors. Eur J Biochem 270:10–19
Wadleigh RW, Yu SJ (1988) Detoxification of isothiocyanate allelochemicals by glutathione-S transferases in three lepidopterous species. J Chem Ecol 14:1279–1288
War AR, Sharma HC (2014) Induced resistance in plants and counter-adaptation by insect pests. In: Chandrasekar R, Tyagi BK, Guri ZZ, Reeck GR (eds) Short views on insect biochemistry and molecular biology, vol 2. International Book Mission, South India, pp 533–547
Waser NM (1998) Pollination, angiosperm speciation and the nature of species boundaries. Oikos 82:198–201
Webster B, Bruce T, Pickett J et al (2010) Volatiles functioning as host cues in a blend become nonhost cues when presented alone to the black bean aphid. Anim Behav 79:451–457
Werker E (2000) Trichome density and development. Adv Bot Res 31:1–36
Weseloh RM (1981) Host location by parasitoids. In: Nordland DA, Jones RJ, Lewis WJ (eds) Semiochemicals: their role in pest control. Wiley, New York, pp 79–95
Wheat CW, Vogel H, Wittstock U et al (2007) The genetic basis of plant-insect coevolutionary key innovation. Proc Natl Acad Sci U S A 104(51):201427–220431
White TCR (1978) The importance of relative food shortage in animal ecology. Oecologia 33:71–86
Wiebes JT (1979) Co-evolution of figs and their insect pollinators. Annu Rev Ecol Syst 10:1–12
Williams CM (1970) Hormonal interactions between plants and insects. In: Sondheimer E, Simeone JB (eds) Chemical ecology. Academic, New York, pp 103–132
Williams CE, Collier CC, Nemcheck JA et al (2002) A lectin-like wheat gene responds systemically to attempted feeding by avirulent first-instar Hessian fly larvae. J Chem Ecol 28:1411–1428
Williams CE, Nemacheck JA, Shukle JT et al (2011) Induced epidermal permeability modulates resistance and susceptibility of wheat seedlings to herbivory be Hessian fly larvae. J Exp Bot 62:4521–4531
Wiseman BR, Snook ME, Isenhour DJ et al (1992) Relationship between growth of corn earworm and fall armyworm larvae (Lepidoptera: Noctuidae) and maysin concentration in corn silks. J Econ Entomol 85:2473–2477
Wittstock U, Agerbirk N, Stauber EJ et al (2004) Successful herbivore attack due to metabolic diversion of a plant chemical defence. Proc Natl Acad Sci U S A 101:4859–4864
Wu JR, Baldwin IT (2010) New insights into plant responses to the attack from insect herbivores. Annu Rev Genet 44:1–24
Wu J, Liu X, Zhang X et al (2008) Differential responses of wheat inhibitor-like genes to Hessian fly, Mayetiola destructor, attacks during compatible and incompatible interactions. J Chem Ecol 34:1005–1012
Xie Y, Arnason JT, Philogene BJR et al (1992) Variation of hydroxamic acid content in maize roots in relation to geographic origin of maize germplasm and resistance to Western corn rootworm (Coleoptera: Chrysomelidae). J Econ Entomol 85:2478–2485
Yan J, Lipka AE, Schmelz EA, Buckler ES, Jander G (2015) Accumulation of 5-hydroxynorvaline in maize (Zea mays) leaves is induced by insect feeding and abiotic stress. J Exp Bot 66:593–602
Yang L, Fang Z, Dicke M et al (2009) The diamondback moth, Plutella xylostella, specifically inactivates Mustard Trypsin Inhibitor 2 (MTI2) to overcome host plant defence. Insect Biochem Mol Biol 33:55–61
Yu SJ (2000) Allelochemical induction of hormone-metabolizing microsomal monoxygenases in the Fall armyworm. Zool Stud 39:243–249
Zangerl AR, Berenbaum MR (2003) Phenotype matching in the wild parsnip and parsnip webworms: causes and consequences. Evolution 57:806–815
Zavala JA, Patankar AG, Gase K et al (2004) Manipulation of endogenous trypsin proteinase inhibitor production in Nicotiana attenuata demonstrates their function as antiherbivore defences. Plant Physiol 134:1181–1190
Zhu-Salzman K, Luthe DS, Felton GW (2008) Arthropod-inducible proteins: broad spectrum defences against multiple herbivores. Plant Physiol 146:852–858
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Tanda, A.S. (2022). Mutualistic Plant Associations Related to Insect Resistance. In: Tanda, A.S. (eds) Molecular Advances in Insect Resistance of Field Crops. Springer, Cham. https://doi.org/10.1007/978-3-030-92152-1_1
Download citation
DOI: https://doi.org/10.1007/978-3-030-92152-1_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92151-4
Online ISBN: 978-3-030-92152-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)