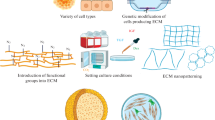
Abstract
Decellularized tissue-derived materials – i.e., biomaterials derived from organs or tissues deprived of their potentially immunogenic cellular components – offer an attractive option for the generation of realistic in vitro tissue and disease models and regenerative therapies. A plethora of decellularization protocols and processing methods make such materials available from numerous tissues and in various formats. This chapter provides an overview of the different forms of tissue-derived decellularized extracellular matrix (dECM) materials for biomedical applications, focusing on innovative dECM-based hydrogels. The chapter concludes with a discussion of the challenges and perspectives of dECM materials concerning standardization and control of physicochemical properties for directing tissue repair and function.
Similar content being viewed by others
References
Abaci A, Guvendiren M. Designing decellularized extracellular matrix-based bioinks for 3D bioprinting. Adv Healthc Mater. 2020;9(24):1–18. https://doi.org/10.1002/adhm.202000734.
Ahn G, et al. Precise stacking of decellularized extracellular matrix based 3D cell-laden constructs by a 3D cell printing system equipped with heating modules. Sci Rep. Springer US. 2017;7(1):1–11. https://doi.org/10.1038/s41598-017-09201-5.
Ali M, et al. A photo-crosslinkable kidney ECM-derived bioink accelerates renal tissue formation. Adv Healthc Mater. 2019;8(7):1–10. https://doi.org/10.1002/adhm.201800992.
Anderson BD, et al. Functional characterization of a bioengineered liver after heterotopic implantation in pigs. Commun Biol. Springer US. 2021;4(1). https://doi.org/10.1038/s42003-021-02665-2.
Asnaghi MA, et al. Thymus extracellular matrix-derived scaffolds support graft-resident thymopoiesis and long-term in vitro culture of adult thymic epithelial cells. Adv Funct Mater. 2021; 31(20). https://doi.org/10.1002/adfm.202010747.
Auger FA, Gibot L, Lacroix D. The pivotal role of vascularization in tissue engineering. Annu Rev Biomed Eng. 2013;15:177–200. https://doi.org/10.1146/annurev-bioeng-071812-152428.
Badylak SF, Gilbert TW. Immune response to biologic scaffold materials. Semin Immunol. 2008;20(2):109–16. https://doi.org/10.1016/j.smim.2007.11.003.
Badylak SF, Taylor D, Uygun K. Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix scaffolds. Ann Rev Biomed Eng. 2011;13(1):27–53. https://doi.org/10.1146/annurev-bioeng-071910-124743.
Bhaskar B, et al. Biomaterials in tissue engineering and regenerative medicine. 2021. https://doi.org/10.1007/978-981-16-0002-9.
Biehl A, et al. Towards a standardized multi-tissue decellularization protocol for the derivation of extracellular matrix materials. Biomater Sci. Royal Society of Chemistry. 2022. https://doi.org/10.1039/d2bm01012g.
Bosworth LA, Downes S. Electrospinning for tissue regeneration. 2011, p. 3–33. https://doi.org/10.1533/9780857092915.1.3.
Brightman AO, et al. Time-lapse confocal reflection microscopy of collagen fibrillogenesis and extracellular matrix assembly in vitro. Biopolymers. 2000;54(3):222–34. https://doi.org/10.1002/1097-0282(200009)54:3<222::AID-BIP80>3.0.CO;2-K.
Brown BN, et al. Inductive, scaffold-based, regenerative medicine approach to reconstruction of the temporomandibular joint disk. J. Oral Maxillofac. Surg. Elsevier Inc. 2012;70(11):2656–68. https://doi.org/10.1016/j.joms.2011.12.030.
Brown M et al. Decellularized extracellular matrix: new promising and challenging biomaterials for regenerative medicine. Biomaterials. Elsevier Ltd. 2022;289(August):121786. https://doi.org/10.1016/j.biomaterials.2022.121786.
Cheung HK, et al. Composite hydrogel scaffolds incorporating decellularized adipose tissue for soft tissue engineering with adipose-derived stem cells. Biomaterials. Elsevier Ltd. 2014;35(6):1914–23. https://doi.org/10.1016/j.biomaterials.2013.11.067.
Cho AN et al. Microfluidic device with brain extracellular matrix promotes structural and functional maturation of human brain organoids. Nat Commun. Springer US. 2021;12(1). https://doi.org/10.1038/s41467-021-24775-5.
Cramer MC, Badylak SF. Extracellular matrix-based biomaterials and their influence upon cell behavior. Ann Biomed Eng. 2020;48(7):2132–53. https://doi.org/10.1007/s10439-019-02408-9.
Crapo PM, Gilbert TW, Badylak SF. An overview of tissue and whole organ decellularization processes. Biomaterials. 2011;32(12):3233–43. https://doi.org/10.1016/j.biomaterials.2011.01.057.
De Waele J, et al. 3D culture of murine neural stem cells on decellularized mouse brain sections. Biomaterials. Elsevier Ltd. 2015;41:122–31. https://doi.org/10.1016/j.biomaterials.2014.11.025.
Dewavrin J-Y et al. Tuning the architecture of three-dimensional collagen hydrogels by physiological macromolecular crowding. Acta Biomater. Acta Materialia Inc. 2014;10(10):4351–59. https://doi.org/10.1016/j.actbio.2014.06.006.
Duan Y, et al. Hybrid gel composed of native heart matrix and collagen induces cardiac differentiation of human embryonic stem cells without supplemental growth factors. J Cardiovasc Transl Res. 2011;4(5):605–15. https://doi.org/10.1007/s12265-011-9304-0.
Edgar L, et al. Utility of extracellular matrix powders in tissue engineering. Organogenesis. Taylor & Francis. 2018;14(4):172–86. https://doi.org/10.1080/15476278.2018.1503771.
Ferreira LP, Gaspar VM, Mano JF. Decellularized extracellular matrix for bioengineering physiomimetic 3D in vitro tumor models. Trends Biotechnol. Elsevier Ltd. 2020;38(12):1397–1414. https://doi.org/10.1016/j.tibtech.2020.04.006.
Freytes DO, et al. Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials. 2008;29(11):1630–7. https://doi.org/10.1016/j.biomaterials.2007.12.014.
Gao G, et al. Tissue engineered bio-blood-vessels constructed using a tissue-specific bioink and 3D coaxial cell printing technique: a novel therapy for ischemic disease. Adv Funct Mater. 2017;27(33). https://doi.org/10.1002/adfm.201700798.
Gao M et al. Comparative evaluation of decellularized porcine liver matrices crosslinked with different chemical and natural cross-linking agents. Xenotransplantation. 2019;26(1). https://doi.org/10.1111/xen.12470.
Ghosh P, et al. Microspheres containing decellularized cartilage induce chondrogenesis in vitro and remain functional after incorporation within a poly(caprolactone) filament useful for fabricating a 3D scaffold. Biofabrication. IOP Publishing. 2018;10(2). https://doi.org/10.1088/1758-5090/aaa637.
Gilbert TW, et al. Production and characterization of ECM powder: implications for tissue engineering applications. Biomaterials. 2005;26(12):1431–5. https://doi.org/10.1016/j.biomaterials.2004.04.042.
Giobbe GG, et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat Commun. Nature Publishing Group. 2019;10(1):5658. https://doi.org/10.1038/s41467-019-13605-4.
Godier-furnémont AFG, et al. Composite scaffold provides a cell delivery platform for cardiovascular repair. Proc Natl Acad Sci USA. 2011;108:7974–9. https://doi.org/10.1073/pnas.1104619108.
Grover GN, Rao N, Christman KL. Myocardial matrix–polyethylene glycol hybrid hydrogels for tissue engineering. Nanotechnology. 2014;25(1):014011. https://doi.org/10.1088/0957-4484/25/1/014011.
Guyette JP, et al. Perfusion decellularization of whole organs. Nat Protoc. 2014;9(6):1451–68. https://doi.org/10.1038/nprot.2014.097.
Hamidi H, Ivaska J. Every step of the way: integrins in cancer progression and metastasis. Nat Rev Cancer. Springer US. 2018;18(9):533–48. https://doi.org/10.1038/s41568-018-0038-z.
Hussey GS, Dziki JL, Badylak SF. Extracellular matrix-based materials for regenerative medicine. Nat Rev Mater. Springer US. 2018;3(7):159–73. https://doi.org/10.1038/s41578-018-0023-x.
Jang J, et al. Tailoring mechanical properties of decellularized extracellular matrix bioink by vitamin B2-induced photo-cross-linking. Acta Biomater. Acta Materialia Inc. 2016. https://doi.org/10.1016/j.actbio.2016.01.013.
Johnson TD, Lin SY, Christman KL. Tailoring material properties of a nanofibrous extracellular matrix derived hydrogel. Nanotechnology. 2011;22(49):494015. https://doi.org/10.1088/0957-4484/22/49/494015.
Keane TJ, Swinehart IT, Badylak SF. Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods. Elsevier Inc. 2015;84(March):25–34. https://doi.org/10.1016/j.ymeth.2015.03.005.
Kim H, et al. Light-activated decellularized extracellular matrix-based bioinks for volumetric tissue analogs at the centimeter scale. Adv Funct Mater. 2021;31(32). https://doi.org/10.1002/adfm.202011252.
Kim JW, et al. Kidney decellularized extracellular matrix enhanced the vascularization and maturation of human kidney organoids. Adv Sci. 2022a;2103526:2103526. https://doi.org/10.1002/advs.202103526.
Kim JW, et al. Kidney decellularized extracellular matrix enhanced the vascularization and maturation of human kidney organoids – SI. Adv Sci. 2022b;2(1):2103526. https://doi.org/10.1002/advs.202103526.
Koh I, et al. The mode and dynamics of glioblastoma cell invasion into a decellularized tissue-derived extracellular matrix-based three-dimensional tumor model. Sci Rep. Springer US. 2018;8(1):1–12. https://doi.org/10.1038/s41598-018-22681-3.
Kühn S, et al. Cell-instructive multiphasic gel-in-gel materials. Adv Funct Mater. 2020:1908857. https://doi.org/10.1002/adfm.201908857.
LeCheminant J, Field C. Porcine urinary bladder matrix: a retrospective study and establishment of protocol. J Wound Care. 2012;21(10):476–82. https://doi.org/10.12968/jowc.2012.21.10.476.
Lee H, et al. Development of liver decellularized extracellular matrix bioink for three-dimensional cell printing-based liver tissue engineering. Biomacromolecules. 2017;18(4):1229–37. https://doi.org/10.1021/acs.biomac.6b01908.
Lewis PL, et al. Directing the growth and alignment of biliary epithelium within extracellular matrix hydrogels. Acta Biomat. Acta Materialia Inc. 2019;85:84–93. https://doi.org/10.1016/j.actbio.2018.12.039.
Li X, Xie H. Decellularized materials, decellularized materials, Edited by Li X, Xie H. Singapore: Springer Singapore; 2021. https://doi.org/10.1007/978-981-33-6962-7.
Li C, et al. Design of biodegradable, implantable devices towards clinical translation. Nat Rev Mater. Springer US. 2020;5(1):61–81. https://doi.org/10.1038/s41578-019-0150-z.
Lin Z, et al. Bioactive decellularized extracellular matrix hydrogel microspheres fabricated using a temperature-controlling microfluidic system. ACS Biomater Sci Eng. 2022;8(4):1644–55. https://doi.org/10.1021/acsbiomaterials.1c01474.
Ma X, et al. Rapid 3D bioprinting of decellularized extracellular matrix with regionally varied mechanical properties and biomimetic microarchitecture. Biomaterials. Elsevier Ltd. 2018;185:310–21. https://doi.org/10.1016/j.biomaterials.2018.09.026.
Ma S, et al. Functional extracellular matrix hydrogel modified with MSC-derived small extracellular vesicles for chronic wound healing. Cell Prolif. 2022;55(4):1–14. https://doi.org/10.1111/cpr.13196.
Magno V, et al. Macromolecular crowding for tailoring tissue-derived fibrillated matrices. Acta Biomater. 2017;55:109–19. https://doi.org/10.1016/j.actbio.2017.04.018.
Mao Q, et al. Fabrication of liver microtissue with liver decellularized extracellular matrix (dECM) bioink by digital light processing (DLP) bioprinting. Mater Sci Eng C. Elsevier. 2020;109(2019):110625. https://doi.org/10.1016/j.msec.2020.110625.
McDade JK, et al. Interactions of U937 macrophage-like cells with decellularized pericardial matrix materials: influence of cross-linking treatment. Acta Biomater. Acta Materialia Inc. 2013;9(7):7191–9. https://doi.org/10.1016/j.actbio.2013.02.021.
Medberry CJ, et al. Hydrogels derived from central nervous system extracellular matrix. Biomaterials. Elsevier Ltd. 2013;34(4):1033–40. https://doi.org/10.1016/j.biomaterials.2012.10.062.
Mora-Navarro C, et al. Monitoring decellularization via absorbance spectroscopy during the derivation of extracellular matrix scaffolds. Biomed Mater (Bristol). 2022;17(1). https://doi.org/10.1088/1748-605X/ac361f.
Moura BS, et al. Advancing tissue decellularized hydrogels for engineering human organoids. Adv Funct Mater. 2022. https://doi.org/10.1002/adfm.202202825.
Ng WL, et al. Applying macromolecular crowding to 3D bioprinting: fabrication of 3D hierarchical porous collagen-based hydrogel constructs. Biomater Sci. 2018;6(3):562–74. https://doi.org/10.1039/C7BM01015J.
O’Neill JD, et al. The regulation of growth and metabolism of kidney stem cells with regional specificity using extracellular matrix derived from kidney. Biomaterials. Elsevier Ltd. 2013;34(38):9830–41. https://doi.org/10.1016/j.biomaterials.2013.09.022.
Pati F, et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun. Nature Publishing Group. 2014;5(1):3935. https://doi.org/10.1038/ncomms4935.
Petrou CL, et al. Clickable decellularized extracellular matrix as a new tool for building hybrid-hydrogels to model chronic fibrotic diseases: in vitro. J Mater Chem B. 2020;8(31):6814–26. https://doi.org/10.1039/d0tb00613k.
Pullen LC. Bioengineered organs: not a matter of “If”. Am J Transplant. 2022;22(1):1–2. https://doi.org/10.1111/ajt.16639.
Rijal G, Li W. A versatile 3D tissue matrix scaffold system for tumor modeling and drug screening. Sci Adv. 2017;3(9):1–17. https://doi.org/10.1126/sciadv.1700764.
Romero-López M, et al. Recapitulating the human tumor microenvironment: colon tumor-derived extracellular matrix promotes angiogenesis and tumor cell growth. Biomaterials. Elsevier Ltd. 2017;116:118–29. https://doi.org/10.1016/j.biomaterials.2016.11.034.
Sasse KC, et al. Accelerated healing of complex open pilonidal wounds using MatriStem extracellular matrix xenograft: nine cases. J Surg Case Rep. 2013;2013(4):rjt025. https://doi.org/10.1093/jscr/rjt025.
Sawkins MJ, et al. Hydrogels derived from demineralized and decellularized bone extracellular matrix. Acta Biomater. Acta Materialia Inc. 2013;9(8):7865–73. https://doi.org/10.1016/j.actbio.2013.04.029.
Scarritt ME, Pashos NC, Bunnell BA. A review of cellularization strategies for tissue engineering of whole organs. Front Bioeng Biotechnol. 2015;3(Mar):1–17. https://doi.org/10.3389/fbioe.2015.00043.
Seif-Naraghi SB, et al. Injectable extracellular matrix derived hydrogel provides a platform for enhanced retention and delivery of a heparin-binding growth factor. Acta Biomater. Acta Materialia Inc. 2012;8(10):3695–703. https://doi.org/10.1016/j.actbio.2012.06.030.
Singelyn JM, Christman KL. Modulation of material properties of a decellularized myocardial matrix scaffold. Macromol Biosci. 2011;11(6):731–8. https://doi.org/10.1002/mabi.201000423.
Sivandzade F, Mashayekhan S. Design and fabrication of injectable microcarriers composed of acellular cartilage matrix and chitosan. J Biomater Sci Polym Ed. Taylor & Francis. 2018;29(6):683–700. https://doi.org/10.1080/09205063.2018.1433422.
Skardal A, et al. A hydrogel bioink toolkit for mimicking native tissue biochemical and mechanical properties in bioprinted tissue constructs. Acta Biomater. Acta Materialia Inc. 2015;25:24–34. https://doi.org/10.1016/j.actbio.2015.07.030.
Smoak MM, et al. Bioinspired electrospun dECM scaffolds guide cell growth and control the formation of myotubes. Sci Adv. 2021;7(20). https://doi.org/10.1126/sciadv.abg4123.
Sonnenberg SB, et al. Delivery of an engineered HGF fragment in an extracellular matrix-derived hydrogel prevents negative LV remodeling post-myocardial infarction. Biomaterials. 2015;45:56–63. https://doi.org/10.1016/j.biomaterials.2014.12.021.
Soucy KG, et al. Feasibility Study of Particulate Extracellular Matrix (P-ECM) and Left Ventricular Assist Device (HVAD) therapy in chronic ischemic heart failure bovine model. ASAIO J. 2015;61(2):161–9. https://doi.org/10.1097/MAT.0000000000000178.
Spang MT, Christman KL. Extracellular matrix hydrogel therapies: in vivo applications and development. Acta Biomater. 2018;68(December):1–14. https://doi.org/10.1016/j.actbio.2017.12.019.
Sullivan KE, et al. Extracellular matrix remodeling following myocardial infarction influences the therapeutic potential of mesenchymal stem cells. Stem Cell Res Ther. 2014;5(1). https://doi.org/10.1186/scrt403.
Sun D, et al. Novel decellularized liver matrix-alginate hybrid gel beads for the 3D culture of hepatocellular carcinoma cells. Int J Biol Macromol. Elsevier B.V. 2018;109:1154–63. https://doi.org/10.1016/j.ijbiomac.2017.11.103.
Sun W, et al. Utilization of an acellular cartilage matrix-based photocross-linking hydrogel for tracheal cartilage regeneration and circumferential tracheal repair. Adv Funct Mater. 2022. https://doi.org/10.1002/adfm.202201257.
Traverse JH, et al. First-in-man study of a cardiac extracellular matrix hydrogel in early and late myocardial infarction patients. JACC Basic Transl Sci. Elsevier. 2019;4(6):659–69. https://doi.org/10.1016/J.JACBTS.2019.07.012.
Tremmel DM, et al. A human pancreatic ECM hydrogel optimized for 3-D modeling of the islet microenvironment. Sci Rep. Nature Publishing Group UK. 2022;12(1):1–14. https://doi.org/10.1038/s41598-022-11085-z.
Tsiapalis D, Zeugolis DI. It is time to crowd your cell culture media – physicochemical considerations with biological consequences. Biomaterials. Elsevier Ltd. 2021;120943. https://doi.org/10.1016/j.biomaterials.2021.120943.
Turner AEB, Flynn LE. Design and characterization of tissue-specific extracellular matrix-derived microcarriers. Tissue Eng C Methods. 2012;18(3):186–97. https://doi.org/10.1089/ten.tec.2011.0246.
Uriel S, et al. The role of adipose protein derived hydrogels in adipogenesis. Biomaterials. 2008;29(27):3712–9. https://doi.org/10.1016/j.biomaterials.2008.05.028.
Visser J, et al. Crosslinkable hydrogels derived from cartilage, meniscus, and tendon tissue. Tissue Eng A. 2015;21(7–8):1195–206. https://doi.org/10.1089/ten.tea.2014.0362.
Wang X, et al. Injectable extracellular matrix microparticles promote heart regeneration in mice with post-ischemic heart injury. Adv Healthc Mater. 2022;11(8):1–16. https://doi.org/10.1002/adhm.202102265.
Wen X, et al. Cauda Equina-derived extracellular matrix for fabrication of nanostructured hybrid scaffolds. Tissue Eng A. 2015;21:1095–105. https://doi.org/10.1089/ten.tea.2014.0173.
Williams C, et al. Cardiac extracellular matrix-fibrin hybrid scaffolds with tunable properties for cardiovascular tissue engineering. Acta Biomater. Acta Materialia Inc. 2015;14(December):84–95. https://doi.org/10.1016/j.actbio.2014.11.035.
Wu J, et al. An injectable extracellular matrix derived hydrogel for meniscus repair and regeneration. Acta Biomater. Acta Materialia Inc. 2015;16:49–59. https://doi.org/10.1016/j.actbio.2015.01.027.
Xu Z, et al. Kidney extracellular matrix hydrogel enhances therapeutic potential of adipose-derived mesenchymal stem cells for renal ischemia reperfusion injury. Acta Biomater. Elsevier Ltd. 2020. https://doi.org/10.1016/j.actbio.2020.07.056.
Yazdanpanah G, et al. A light-curable and tunable extracellular matrix hydrogel for in situ suture-free corneal repair. Adv Funct Mater. 2022;32(24). https://doi.org/10.1002/adfm.202113383.
Yi HG, et al. A bioprinted human-glioblastoma-on-a-chip for the identification of patient-specific responses to chemoradiotherapy. Nat Biomed Eng. Springer US. 2019;3(7):509–19. https://doi.org/10.1038/s41551-019-0363-x.
Young DA, et al. Stimulation of adipogenesis of adult adipose-derived stem cells using substrates that mimic the stiffness of adipose tissue. Biomaterials. Elsevier Ltd. 2013;34(34):8581–8. https://doi.org/10.1016/j.biomaterials.2013.07.103.
Yu C, et al. Decellularized adipose tissue microcarriers as a dynamic culture platform for human adipose-derived stem/stromal cell expansion. Biomaterials. Elsevier Ltd. 2017;120:66–80. https://doi.org/10.1016/j.biomaterials.2016.12.017.
Yu C, et al. Scanningless and continuous 3D bioprinting of human tissues with decellularized extracellular matrix. Biomaterials. Elsevier Ltd. 2019;194:1–13. https://doi.org/10.1016/j.biomaterials.2018.12.009.
Zhu Y, et al. Injectable, porous, biohybrid hydrogels incorporating decellularized tissue components for soft tissue applications. Acta Biomater. Acta Materialia Inc. 2018;73:112–26. https://doi.org/10.1016/j.actbio.2018.04.003.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this entry
Cite this entry
Magno, V., Werner, C. (2023). Tissue-Derived Decellularized Materials for Biomedical Applications. In: Maia, F.R.A., Oliveira, J.M., Reis, R.L. (eds) Handbook of the Extracellular Matrix . Springer, Cham. https://doi.org/10.1007/978-3-030-92090-6_42-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-92090-6_42-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92090-6
Online ISBN: 978-3-030-92090-6
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences