Abstract
Aquaculture plays an important role in providing foods and healthy diets. According to the Food and Agriculture Organization (FAO), aquaculture is defined as the farming of aquatic organisms. There are many kinds of aquaculture, such as fish farming, shrimp farming, mariculture, and seaweed culture. Aquaculture has tended to increase its contribution to global seafood production every year. The rapidly increased production of fed aquaculture has led to an effect on the coastal environment. Recently, the use of seaweed in aquaculture has been increasingly recognized due to its benefits and numerous feedstocks. Several types of seaweed have been used for the feeding and treatment of wastewater from aquaculture practices. Techniques in seaweed aquaculture have developed dramatically in Asia, but there are still numerous challenges to overcome. In this article, we intend to address the roles and applications of seaweed in aquaculture and integrated multitrophic aquaculture systems, as well as perspectives for the future.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Aquaculture refers to the farming of aquatic organisms, and it is one of the fastest-growing food-producing industries in the world. Farming (fish, mollusks, crustaceans, and aquatic plants) may be carried out in various aquatic environments, e.g., ponds, lagoons, oceans, and closed man-made farming systems on land. Over the past decade, aquaculture has played an important role due to the global decline in aquatic resources from overexploitation. Aquaculture is used as a tool to supplement the seafood supply, which is widely recognized as an effective means of meeting the seafood needs of the population. Aquaculture is also a more sustainable option for consumers compared to other farm proteins. In addition, aquaculture emits less greenhouse gas compared to other farming practices. In recent decades, the increased global demand for seafood has been fulfilled by increasing aquaculture, which provided 178.5% of the world fisheries and aquaculture production in 2018 (FAO 2020). With the rapid increase in production, aquaculture activities have affected the environment in a variety of ways. In particular, fed aquaculture includes fish and shrimp, which need to be supplemented with food as an exogenous source of energy (Beveridge 1996; Troell et al. 2004). Unlike unfed aquaculture, this type of system includes seaweed and shellfish (mussels and oysters). Seaweed requires sunlight for use as an energy source to turn nutrient-rich effluents into profitable resources, and shellfish feed on microscopic plankton.
According to the Food and Agriculture Organization (FAO 2014), the global aquaculture production was 90.4 million tons (live weight equivalent) in 2012, with a value of US$ 144 billion. This included 23.8 million tons of aquatic algae and 66.6 million tons of food fish and was estimated at 26.1 million and 70.5 million tons in 2013, respectively. Production increased in 2016 and included 30.1 million tons of aquatic plants and 80.0 million tons of food fish (FAO 2018). Aquaculture has tended to increase its contribution to global seafood production every year. A production volume of 90% was produced in Asia, and China alone produced 13.5 million tons of aquatic algae and 43.5 million tons of food fish, or a 1:3 aquatic algae and food fish ratio. The farmed aquatic plants included mostly seaweeds, and in 2016, China and Indonesia were by far the major producers of aquatic plants (FAO 2018).
Several techniques of seaweed cultivation in aquaculture systems have been continuously developed, but there are still numerous challenges to overcome. In this article, we address the role and application of seaweed in aquaculture and integrated multitrophic aquaculture systems (IMTAs) and their prospects in the future.
2 Role of Seaweed Utilization in Aquaculture
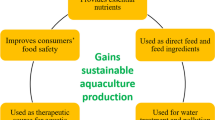
The use of seaweeds in aquaculture has been increasingly recognized due to its positive benefits and numerous feedstocks (Shama et al. 2019). In recent years, seaweed aquaculture has received increased attention for promotion and monitoring climate and environmentally friendly bioeconomic development (Chung et al. 2017). Over the decades, seaweed has been utilized in aquaculture in a variety of ways. It has been used for the feeding and treatment of wastewater discharged from shrimp and fish farming. Since seaweed is rich in minerals, vitamins and fatty acids (Ratana-arporn and Chirapart 2006), it has preferentially been used in the farming of aquatic animals, especially as an ingredient in animal diets. In addition, the use of seaweed in aquaculture systems can improve the water quality, and farmers expect seaweed to help increase the rate of growth, survival rate, and disease tolerance to help them make a profit from aquaculture crops (Buschmann 1996).
In the feed industry , seaweed and its colloidal substances (agar, carrageenan, and alginate) are utilized as mineral carbohydrate sources and binders. Seaweed colloids are also used as emulsifiers, thickeners, and sticky substances. Additionally, seaweed is a source of many important nutrients of amino acids, vitamins such as B6, K, A, and C and minerals such as iodine, iron, magnesium, sodium, and manganese. The seaweed mineral content is the most important because minerals are essential for animal cells; thus, seaweed can be used as a substitute for a proportion of raw materials used in food production (Takeshi et al. 2005). However, seaweed is viewed as a complete feed and/or supplementary feed for supplementation to improve color and stimulate immunity. Also, polysaccharides are considered dietary fibers and binders of feed ingredients.
In farming aquatic plants, seaweed is a major dominant practice in over 50 countries. Seaweed farming has expanded by 8% per year in the past, increasing from 6.2% in the previous decade (FAO 2016) and more than doubling in recent years. The major production of seaweed in the world is the farming of Kappaphycus alvarezii and Eucheuma spp. as raw material for carrageenan extraction, especially in Indonesia (FAO 2018, 2020). According to the FAO (2020), seaweed production in Indonesia increased 15.2-fold, from 1.9% in 2000 to 28.8% in 2018, while the world aquaculture production of aquatic algae increased approximately 205.7%, from 10.6 billion tons in 2000 to 32.4 billion tons in 2018.
For a long time, several cultivation methods have been used to integrate seaweed with aquaculture systems. Seaweed polyculture is one of the known methods that involves the production of two or more cultured species in the same physical space at the same time. The objective is to produce multiple products (e.g., seaweed, fish, and shrimp) that have economic value. Seaweed farming through polyculture systems is prevalent in Southeast Asian countries due to its feasibility of providing additional income to fish farmers. Polyculture systems integrating seaweed farming with other marine fauna, such as clams, abalone, and fish, has been rapidly developing in the region.
In recent years, in Indonesia, the stratified double floating net cage (SDFNC) technique has been developed for seaweed polyculture with fish and shrimp to increase productivity and maintain a healthy water ecosystem for sustainable aquaculture (Putro et al. 2016). This culture system is a combination of red seaweed (Eucheuma cottonii), rabbitfish (Siganus sp.), black tiger shrimp (Penaeus monodon), and Pacific white shrimp (Litopenaeus vannamei). E. cottonii (3 g) had the highest absolute mean weight (282.35 g) when cocultured with Gracilaria verrucosa (3 g), shrimp Penaeus vannamei (100 juveniles, ~2.1 g), and milkfish Chanos chanos (25 juveniles, ~3.1 g) (Yala et al. 2017). Polyculture conditions could enhance the growth of E. cottonii but not G. verrucosa due to competition between seaweed species. However, both shrimp and fish species had good growth and were not influenced by the culture system.
Also, integrated intensive aquaculture has been developed from traditional extensive polyculture over the past two decades by integrating the culturing of fish or shrimp with shellfish and/or seaweed (Neori et al. 2004). Integrating seaweed into the farming of fish and shrimp can balance the nutrient input and metabolize dissolved oxygen, acidity and carbon dioxide levels, in one step, in the culture systems. It has been proven that using seaweed in integrated aquaculture provides an excellent system for sustainable environments (Neori et al. 2004; Hernández et al. 2005). Several works have revealed different species of seaweed that are effective in removing nutrients from water bodies. Some species show high potential for use in IMTA in recirculating aquaculture systems (RAS) (Hayashi et al. 2008; Neori 2008; Marinho et al. 2013). In recent years, Kang et al. (2013) developed a seaweed species-selection index for selecting suitable species in seaweed-based integrated aquaculture systems. The available literature-based information, reference data, and physiological seaweed experiments were used to synthesize the index and to identify and prioritize the desired species. The species with the highest scores are considered in the integrated aquaculture suitability index.
2.1 Seaweed in Open-Sea and Land-Based Aquaculture
The seaweed types chosen for open-sea aquaculture are typically those with value either as a foodstuff or in industrial applications. In general, open-sea culture systems lack control over the dilution of waste caused by natural seawater movement. In contrast, land-based systems are mostly closed-circulation systems, thus allowing control of nutrient-rich waste. The use of seaweed integrated with fish cultures in open-water and land-based aquaculture systems has been well documented in several regions (Troell et al. 1997; Marinho-Soriano et al. 2002; Chirapart and Lewmanomont 2004; Hernández et al. 2005; Pellizzari and Reis 2011). The red seaweed Gracilaria chilensis was cocultured in salmon cage farms in open-sea systems in southern Chilean waters (Troell et al. 1997), and Grote (2019) reviewed the open-sea and land-based cultivation of Palmaria palmata in Europe. Seaweed produces cheap nutritious biomass at an average market value of $30–40 per ton -fresh weight basis (Noeri and Guttman 2017). Production mostly takes place in coastal waters in several Southeast Asian countries.
Land-based aquaculture covers the farming systems of aquatic organisms on land, which usually occurs in pond cultures, rather than in the ocean. Several works involving integrated land-based culture have been performed using an integrated design (Neori et al. 2000; Marinho-Soriano et al. 2002; Chirapart and Lewmanomont 2004; Hernández et al. 2005). The intensive land-based culture of abalone (Haliotis discus hannai), seaweed (Ulva lactuca or Gracilaria conferta) and fish (Sparus aurata) could achieve nutrient recycling, reduced water use, reduced nutrient discharge and high yields (Neori et al. 2000). Likewise, the cultivation of Ulva rotundata and Gracilariopsis longissima in effluents from intensive marine culturing (grow-out phase) of gilthead seabream Sparus aurata revealed that the dissolved nutrient content was reduced in effluents from the fish tank (Hernández et al. 2005).
2.2 Seaweed in Integrated Multitrophic Aquaculture (IMTA)
Currently, seaweed has been widely discussed for its benefits in cultivation in IMTA systems. IMTA is a type of culture system that incorporates marine plants and aquatic animals, and it has the potential to decrease costs and improve efficiency and productivity for a number of species and systems. IMTA is an idea that has been developing over the last 20 years and is becoming increasingly well known. Barrington et al. (2009) described IMTA as a cultivation practice that combines in appropriate proportions of fed aquaculture species (e.g., finfish/shrimp) with organic extractive aquaculture species (e.g., shellfish/herbivorous fish) and inorganic extractive aquaculture species (e.g., seaweed); the purpose is to create balanced systems for environmental sustainability (biomitigation), economic stability (product diversification and risk reduction) and social acceptability (better management practices). According to Troell (2009), IMTA is defined as fed aquaculture (e.g., fish) combined with inorganic extractive species (e.g., seaweed) and organic extractive species (e.g., shellfish). It also refers to more intensive cultivation of the different species in proximity to each other, connected by nutrient and energy transfers through water. Recently, the production of biomass of Eucheuma cottonii has been observed in the Multi Trophic Sea Farming system in the marine aquaculture area of Gerupuk Bay, Central Lombok, Indonesia. The highest biomass production of seaweed was obtained when cocultured with abalone grouper compared to when cocultured with lobster abalone, abalone red carp, and abalone pomfret fish (Sukiman et al. 2014). A meta-analysis of integrated multitrophic aquaculture has been reported, which showed the most successful growth of commercial extractive species within close proximity to open-water fish farms (Kerrigan and Suckling 2016).
3 Seaweed Use for Sustainable Aquaculture
According to the FAO report, seaweed in aquaculture systems is mainly produced in the Asian region. Several seaweed species, e.g., Caulerpa lentillifera, Gracilaria fisheri, G. tenuistipitata, Ulva intestinalis, and Ulva rigida, have been used for the treatment of effluent from farms (Chirapart and Lewmanomont 2004; Chaitanawisuti et al. 2011; Aníbal et al. 2014; Kunawongdet 2020). These seaweed species are raised in effluent ponds due to their ability to effectively absorb and reduce wastewater from brackish aquaculture. Seaweed species commonly grow profusely throughout the year in tropical water ponds and irrigation canals, such in Thailand. They have been used in intensive prawn culture systems for nutrient removal from culture systems before recycling the water or discharging it into the environment (Baliao and Tookwinas 2002; Chaitanawisuti et al. 2011).
3.1 Seaweed in Shrimp Culture
Seaweed has been used in shrimp aquaculture as an ingredient in the diet, as a biofilter, and in nursing areas. Using seaweed in white shrimp farming in several countries has led to the highest annual yields and value of aquaculture compared to other marine species.
3.1.1 Seaweed in the Shrimp Diet
According to FAO reports, the production of aquaculture worldwide has increased annually, constituting an important human protein source. Thus, to sustain a high rate of growth, it is imperative to have viable production and development of economical aquaculture systems, low costs, and higher-quality feeds because feed is the major operational cost for most aquaculture enterprises (Rana et al. 2009). The rising cost of commercial aquafeed is therefore inducing some farmers to opt for alternative feeds.
In 2013, the shrimp farming business in Thailand collapsed due to the disease outbreak caused by Vibrio bacteria (Vibrio sp.) and viruses (Sitthimong 2020), resulting in a decline in white shrimp production. Several researchers have attempted to use seaweed species to increase resistance to pathogenesis in white shrimp and have provided associated guidelines (Sirirustananun et al. 2011; Kanjana et al. 2011; Wongprasert et al. 2014). Seaweed has been applied for the prevention of disease through enhancing immune function (Takeshi et al. 2005). It has been used as a dietary supplement, providing nutrients such as vitamin C and polysaccharide components. Among the seaweed species, Gracilaria fisheri is mainly applied for diet supplementation in Thailand (Rudtanatip et al. 2014). Following green seaweed, Ulva rigida has also been used, as potent protein supplementation in the feed of the white shrimp Litopeneaus vannamei, and feed meal is a food source that provides minerals and vitamins for shrimp (Tamtin et al. 2016). This algal species was used as a supplemental protein source at proportions ranging from 6% to 12% in the shrimp diet with a reduction in fishmeal. Generally, the reduction of fishmeal and its replacement with alternative protein sources mostly involve soybeans. Many experiments have reported that seaweed stimulates the growth, immunity, and pathogen resistance of the white shrimp L. vannamei (Wongprasert et al. 2014; Zahra et al. 2017; Niu et al. 2019), as well as the taste and color of shrimp meat when compared to the use of soybean meal instead of fishmeal. The white shrimp L. vannamei is both omnivorous and herbivorous (Martínez-Córdova and Peña-Messina 2005), and it can use plant nutrients, which are starchy protein-based ingredients. The use of seaweed in the diet could provide effective protein digestion in the diet, which is not different from using fishmeal or soy meal as the main protein source. The protein level found in sea lettuce varied from 10.37% to 11.60%, and the corresponding protein digestibility efficiency of the shrimp was approximately 84.01–84.32% (Tamtin et al. 2016). Furthermore, other work reported that protein in the white prawn diet was reduced by 25–30% (Cuzon et al. 2004). Nevertheless, there is a need for more research to clarify the seaweed state, such as its role in nutrition and disease prevention throughout all the production phases of the feed industry.
3.1.2 Seaweed as a Biofilter in Shrimp Culture
Several seaweed species, e.g., Caulerpa lentillifera, Gracilaria fisheri, Solieria robusta, Halymenia sp. Acanthophora spicifera and Grateloupia indica, have been evaluated for their potential use as biofilters, especially Gracilaria fisheri, due to its ability to adapt and tolerate nutrient-enriched seawater (Teekayu and Khidprasert 1996; Chokwiwattanawanit 2000). Seaweed has been used as a biofilter in shrimp aquaculture systems for nutrient absorption, especially that of NH4+-N, NO2-N, NO3-N, and PO43--P (Table 20.1). Chokwiwattanawanit (2000) described the process of water treatment in which shrimp farm effluent circulated to oyster (Crassostrea lugubris) or green mussel (Perna viridis) ponds to precipitate suspended sediment for 1–2 weeks, after which clear water flowed into the seaweed ponds (Gracilaria fisheri), while nutrient-enriched seawater was absorbed to yield improved water quality. The duration of the treatment process was about 2 weeks to allow nitrogen and phosphorous absorption. Cultivation of the Gracilaria sp. has been developed in shrimp pond effluents in Brazil, and the cultivation success is related to the techniques used and the environmental conditions of the effluent (Marinho-Soriano et al. 2002). It has been proven that the use of Gracilaria verrucosa as a biofilter in traditional aquaculture ponds could increase the production of Vannamei shrimp from 1 ton per ha to 3.5 ton per ha while gaining a seaweed production of 5 ton per ha after 3 months of culture (Andayani et al. 2016). Also, the algal species Ulva clathrata cultured in outdoor tanks showed efficiency in removing the main inorganic nutrients from the effluent water of shrimp (Litopenaeus vannamei) aquaculture, with uptake rates of 70–82% of the total ammonium nitrogen and 50% of the phosphate within 15 h (Copertino et al. 2009).
Caulerpa lentillifera has shown the potential ability to remove nutrients from aquaculture effluents, especially NO3-N (Guo et al. 2015; Manori Bambaranda et al. 2019a, b). The efficiency of nutrient uptake has shown a relationship between the nutrient concentration and density of C. lentillifera. The highest reduction in the concentration of the nutrient components (NO2−, NO3−, and PO43−) occurred at 30 g/L algal density, while the highest concentration reduction for NH3-N occurred at 40 g/L algal density (Manori Bambaranda et al. 2019a). In recirculating aquaculture systems, utilizing C. lentillifera for biofiltration has shown potential for use in the effective treatment of aquaculture effluent integrating fish and the production of seaweed (Manori Bambaranda et al. 2019b).
In the case of a water treatment system with separate units, the use of shellfish causes sludge to be suspended well, but there is a disadvantage in that sediment filtering causes shellfish to excrete waste in the organic sediment form of nitrogen and phosphorus compounds, while in another unit, seaweed is suitable for the treatment of nitrogen and phosphorus solutions (Tookwinas et al. 2001; Pariyawatee et al. 2003). Therefore, a system that combines the hallmarks of the treatment system in each subsystem is an ideal form for effectively treating wastewater from shrimp culture. In a closed system, the shrimp farm effluent will be drained from the culture pond into a treatment unit consisting of biological systems (shellfish and algae) and physical systems (filtration) by pumping water from the shrimp pond to the biological treatment pond containing mollusks and subsequently pumping it into a treatment pond containing seaweed (Gracilaria fisheri). After 3 days of treatment, the treated water will be recycled back through the filter and into the pond (Tookwinas et al. 2001). It has been reported that the removal efficiency of nitrate-N and phosphate-P was decreased by approximately 60% after treatment with this type of system (Pariyawatee et al. 2003). Some species of seaweed, e.g., Caulerpa lentillifera, Ulva sp. and Gracilaria fisheri, had high rates of removing ammonium, with a 92–97% absorption efficiency, and Caulerpa lentillifera showed the lowest removal rates, at 90% overall (Kaewmesri 2000; Pariyawatee et al. 2003).
Seaweed is preferably used in treatment systems for wastewater from shrimp farming in Thailand (Fig. 20.1), which can help reduce the amount of hardness and suspended solids in the water. The shrimp farm effluent circulates to the seaweed pond to absorb nutrients in the water, after which the water in the pond is filled with oxygen, and microbes are provided in another unit and recycled to the culture units. The seaweed will absorb the total ammonia–nitrogen, nitrite–nitrogen, nitrate–nitrogen and orthophosphate throughout the biofilter recirculating systems and improve the seawater quality, which is within the safety conditions required for circulation and periodicity.
3.1.3 Seaweed for Balancing the Food Chain in Shrimp Culture
Seaweed has been used for cocultivation with tiger prawns (Penaeus monodon) in tanks or earthen ponds, which is prevalent in Southeast Asian countries (Songsangjinda and Inek 2003; Tsutsui et al. 2010, 2015; Anh et al. 2018). Another green seaweed, the gut weed Ulva intestinalis, has been used in tiger prawn farming in Thailand. Algal species are used as sources for gathering zooplankton, and algal spores are used for feeding zooplankton to increase post larval production, during which Ulva thalli are harvested from shrimp ponds as frequently as possible to balance the food chain in shrimp culture ponds (Anonymous 2010). Coculturing tiger prawns with the filamentous green seaweed Chaetomorpha ligustica has a dietary advantage, especially in early-age juveniles (Tsutsui et al. 2010), and it reduces feed costs in intensive prawn aquaculture (Tsutsui et al. 2015). Likewise, the coculture of tiger prawns and G. tenuistipitata with a reduction in the feeding ratio of up to 50% satiation resulted in improved growth, feed cost, and shrimp color, as well as better water quality in the culture tanks (Anh et al. 2018).
3.2 Seaweed in Fish Culture
3.2.1 Seaweed in Fish Feed
According to the high cost, fluctuating quality, and uncertain availability of fish meal in fish diets, several sources of conventional plant oilseed meals are used, such as soybean (Voorhees et al. 2019; Abdel-Warith et al. 2020) and cottonseed (El-Sayed 1999; Bu et al. 2017) meal. There have been a few species of seaweed used to feed fish directly. The species of Ulva green seaweed are important for biofiltering fish pond effluents. This alga has been harvested in ponds as a feed additive for red tilapia in the coastal areas of Thailand. The alga has also been used as a replacement raw material for food production or optimization to improve color. It improves the growth rate, survival rate, efficiency, and feed conversion ratio (FCR.), in addition to enhancing immunity (Prud’homme van Reine and Trono 2001).
Several studies have reported the use of seaweed as an ingredient in tilapia fish feed (Azaza et al. 2008; Siddik and Anh 2015; Putri et al. 2017; Yangthong and Ruensirikul 2020). The green seaweed Ulva rigida was used as a replacement for soybean meal in the practical tilapia fish diet formulated to contain 28% crude protein, 7.5% lipids and 15 kJ gross energy (Azaza et al. 2008). They stated that replacement with seaweed species decreased the apparent protein digestibility (APD) with increasing inclusion levels of ulva meal. The use of seaweed as an ingredient causes a reduction in protein and fat contents because of the replacement of feedstock with vegetables in the diet ingredients. In addition, saponins, which are found in several seaweed species (Feroz 2018), interrupt the absorption of dietary fat and inhibit the breakdown of fat in the gallbladder, so the fish cannot fully use fat from the diet. The increased level of algae replacement leads to a reduction in body fat storage (Tacon 1997; Guillaume and Choubert 2001; Azaza et al. 2008).
3.2.2 Seaweed as a Biofilter in Fish Culture
Similar to the wastewater treatment system in shrimp culture, seaweed is also used as a biofilter in fish culture. The red seaweed Gracilaria and the green seaweed Caulerpa and Ulva are commonly applied for water treatment in aquaculture systems. Gracilaria fisheri is the most widely applied for nutrient absorption in fish farming in Thailand. These seaweed species have high efficiencies in the removal of the total ammonia from fish farm effluent. G. fisheri had the best efficiency in the removal of ammonia, nitrite and phosphate (99.6%, 70.9% and 98.4%, respectively), while C. lentillifera showed the best efficiency in the removal of nitrate (92.0%) (Thongcanarak and Predalumpaburt 2008). However, in the case of Ulva intestinalis, this alga is not suitable for water treatment in standing-water conditions, since the seaweed cannot grow and it dies under nutrient depletion – conditions. Inorganic nitrogen and phosphate are released again into water when seaweed dies and decomposes. Although seaweed species should be used for water treatment, their efficiency and ability to adapt to survive under the specific conditions of effluent from aquaculture ponds should be considered. Fish effluent was treated with seaweed according to the removal periodicity of the system. The total periodicity took approximately 1 month to compensate for inorganic carbon depletion and to reach low levels of nitrate and phosphate. Throughout the farming period, the ammonia, nitrite, nitrate and BOD values in the fish ponds increased, while in the treatment unit, seaweed decreased the ammonia, nitrite, nitrate and BOD (Thongcanarak and Predalumpaburt 2008).
The process of wastewater treatment in fish farming was divided into 4 units, as in the shrimp culture treatment. The effluent from the fish culture unit flowed to the settling unit and then to the seaweed pond for nutrient absorption from the effluent seawater. Likewise, in the removal timeline of the process, the total periodicity took a month to complete the treatment. The schematic diagram of the system is shown in Fig. 20.2.
3.3 Seaweed in Abalone Culture
Several species of seaweed have been used in abalone aquaculture on several continents, mainly in Asian countries, e.g., China, Japan, Taiwan and Thailand. Abalone farming has been attempted for at least two decades in Thailand; however, farmed abalone has been categorized as a prime cultured species of aquatic animals due to its high value. This was an incentive that made private entrepreneurs raising black tiger prawns or white shrimp initially become interested in abalone instead of sea shrimp. Due to pricing problems and shrimp disease outbreaks in Thailand, the Department of Fisheries has turned to developing abalone farming and has been successful in producing young abalone. For feeding abalone, red seaweed was used as a supplementary feed in parallel with complete feed in pellet form. However, to increase immunity, abalone feed is mixed with seaweed at a proportion of 5%, and the most popular species are Gracilaria tenuistipitata and G. fisheri (Prud’homme van Reine and Trono 2001).
4 Conclusion and Perspective
There are many critical aspects of sustainability aquaculture that need to be addressed. Aquaculture practices have shown negative ecological impacts. The discharge of organic and inorganic waste from fed aquaculture into the environment causes eutrophication and pollution, introduces diseases and affects biodiversity in the ecosystem. The use of seaweed in aquaculture has been increasingly recognized due to its positive benefits and numerous supplies. Seaweed is used for diet supplementation and balancing the food chain in aquaculture (shrimp, fish, and abalone), and it can prevent disease by enhancing immune function. However, more research is needed to clarify its role in nutrition and disease prevention. In particular, the saponins in some seaweeds may affect dietary fat absorption and inhibit the breakdown of fat in the gallbladder. Increasing the level of algae replacement leads to a reduction in body fat storage in fish.
Seaweed plays an important role in wastewater treatment in aquaculture systems. The utilization of seaweed in shrimp culture is similar to its use in fish culture—it is used as a supplementary feed or mixed into the feed ingredients, in addition to its use in the treatment of effluent from aquaculture units. The nutrient removal system consists of the combination of treatment unit using mollusks, such as oysters (Crassostrea lugubris) or mussels (Perna viridis), with treatment unit using seaweed. This combination effectively improves the effluent quality from shrimp ponds. Treatment using mollusks is a disadvantage that causes organic sludge, but nitrogen and phosphorus are increased and can be absorbed by seaweed in the next treatment unit.
However, seaweed can also be used for many purposes other than absorbing nutrients in seawater. There may be other harmful substances, including heavy metals, that algae take up at the same time as nitrogen and phosphorus adsorption. Therefore, the objectives of its use should be considered. In the case of using seaweed for treating or reducing heavy metals or negative elements, the harvesting or utilization of seaweed after treatment should be considered environmentally friendly. In addition, towards the end of the treatment period, a problem of phosphate release from dead seaweed would occur during the life stage of the algae and in the period of depleted nutrient concentration. Therefore, the ability of seaweed to adapt and the effect of the aquaculture effluent must also be considered. During the treatment period, algae should be left in the system for at least 1 week to treat nitrate nitrogen and 2–3 weeks to treat phosphate phosphorous (Suthiniam et al. 2009). Furthermore, the efficiency of seaweed can provide successful treatment of wastewater in the early stages of the system. However, the nitrogen and phosphate compounds released from the dead seaweed may be troublesome in the maintenance of the seaweed unit, since it may take a period of time to regrow the seaweed in the system. The use of seaweed also has to take into account the adaptability of seaweed in aquaculture. Therefore, monocultivation of seaweed is needed to support the water treatment unit, which requires a support area.
References
Abdel-Warith AA, Younis EM, Al-Asgah NA, Mahboob S (2020) Effect of replacing fish meal by full fat soybean meal on growth performance, feed utilization and gastrointestinal enzymes in diets for African catfish Clarias gariepinus. Braz J Biol 80(3):535–543
Andayani S, Yuwanita R, Izzah N (2016) Biofilter application using seaweed (Gracilaria verrucosa) to increase production of Vannamei shrimp in traditional pond district Bangil-Pasuruan. Res J Life Sci 3(1):16–22
Anh NTN, Ngan LTH, Vinh NH, Hai TN (2018) Co-culture of red seaweed (Gracilaria tenuistipitata) and black tiger shrimp (Penaeus monodon) with different feeding rations. Int J Sci Res Publ 8(9):269–277
Aníbal J, Madeira HT, Carvalho LF, Esteves E, Veiga-Pires C, Rocha C (2014) Macroalgae mitigation potential for fish aquaculture effluents: an approach coupling nitrogen uptake and metabolic pathways using Ulva rigida and Enteromorpha clathrata. Environ Sci Pollut Res 21:13324–13334
Anonymous (2010) Role of gut weeds (Ulva intestinalis Linnaeus) on improving water and sediment qualities and producing natural foods for commercial culture of banana shrimp (Penaeus merguiensis de Man, 1988). Ministry of Agriculture and Cooperatives, Department of Fisheries, Freshwater Aquaculture Research and Development Division. (in Thai)
Azaza MS, Mensi F, Ksouri J, Dhraief MN, Brini B, Abdelmouleh A, Kraïem MM (2008) Growth of Nile tilapia (Oreochromis niloticus L.) fed with diets containing graded levels of green algae Ulva meal (Ulva rigida) reared in geothermal waters of southern Tunisia. J Appl Ichthyol 24:202–207
Baliao DD, Tookwinas S (2002) Best management practices for a mangrove-friendly shrimp farming. SEAFDEC Aquaculture Department/Association of Southeast Asian Nations, Bangkok/Tigbauan
Barrington K, Chopin T, Robinson S (2009) Integrated multi-trophic aquaculture (IMTA) in marine temperature waters. In: Soto D (ed) Integrated mariculture: a global review. FAO Fisheries and Aquaculture Technical Paper, vol 529, pp 7–46
Beveridge MCM (1996) Cage aquaculture, 2nd edn. Fishing News Books Ltd., Cambridge, p 346
Bu X, Chen A, Lian X, Chen F, Zhang Y, Muhammad I, Ge X, Yang Y (2017) An evaluation of replacing fish meal with cottonseed meal in the diet of juvenile Ussuri catfish Pseudobagrus ussuriensis: growth, antioxidant capacity, nonspecific immunity and resistance to Aeromonas hydrophila. Aquaculture 479:829–837
Buschmann AH (1996) An introduction to integrated farming and the use of seaweeds as biofilters. Hydrobiologia 326(327):59–60
Chaitanawisuti N, Santhaweesuk W, Kritsanapuntu S (2011) Performance of the seaweeds Gracilaria salicornia and Caulerpa lentillifera as biofilters in a hatchery scale recirculating aquaculture system for juvenile spotted babylons (Babylonia areolata). Aquac Int 19:1139–1150
Chirapart A, Lewmanomont K (2004) Growth and production of Thai agarophyte cultured in natural pond using the effluent seawater from shrimp culture. Hydrobiologia 512:117–126
Chokwiwattanawanit A (2000) Efficiency of the macroalgae Caulerpa lentillifera and Acanthophora spicifera for the treatment of nitrogen compound from shrimp pond effluent. M Sc Thesis, Environmental Science, Graduate School, Chulalongkorn University. (in Thai)
Chung IK, Sondak CFA, Beardall J (2017) The future of seaweed aquaculture in a rapidly changing world. Eur J Phycol 52(4):495–505. https://doi.org/10.1080/09670262.2017.1359678
Copertino MS, Tormena T, Seeliger U (2009) Biofiltering efficiency, uptake and assimilation rates of Ulva clathrata (Roth) J. Agardh (Chlorophyceae) cultivated in shrimp aquaculture waste water. J Appl Phycol 21:31–45
Cuzon G, Brito A, Jiménez-Yan L, Brito R, Tomás G, Taboada G, Gaxiola G (2004) The effects of animal or plant-based diets on energy partitioning in selected ontogenetic stages of the shrimp Litopenaeus vannamei. In: Cruz Suárez LE, Ricque Marie D, Nieto Lopez MG, Villarreal D, Scholz UY, Gonzalez M (eds) Avances en nutricion acuicola VII. Memorias del VII Simposium internacional de nutricion acuicola, Hermosillo, Sonora, Mexico. 16–19 Nov 2004
El-Saidy DMSD (1999) Evaluation of cottonseed meal as partial and complete replacement of fishmeal in practical diets of Nile tilapia, Oreochromis niloticus (L.) fingerlings. Egypt J Aquat Biol Fish 3:1–18
FAO (2014) The state of world fisheries and aquaculture. Opportunities and challenges. Rome, 233 p
FAO (2016) The state of world fisheries and aquaculture 2016. Contributing to food security and nutrition for all. Rome, 200 p
FAO (2018) The state of world fisheries and aquaculture 2018 – meeting the sustainable development goals. Rome, 227 p
FAO (2020) The state of world fisheries and aquaculture 2020 – sustainability in action Rome, 224 p
Feroz B (2018) Saponins from marine macroalgae: a review. J Mar Sci Res Dev 8:255. https://doi.org/10.4172/2155-9910.1000255
Grote B (2019) Recent developments in aquaculture of Palmaria palmata (Linnaeus) (Weber & Mohr 1805): cultivation and uses. Rev Aquac 11:25–41
Guillaume J, Choubert G (2001) Digestive physiology and nutrient digestibility in fishes. In: Guillaume J, Kaushik S, Bergot P, Métailler R (eds) Nutrition and feeding of fish and crustaceans. Springer-Praxis Publishing, Chichester, pp 27–56
Guo H, Yao JT, Sun ZM, Duan DL (2015) Effects of salinity and nutrients on the growth and chlorophyll fluorescence of Caulerpa lentillifera. Chin J Oceanol Limn 33:410–418
Hayashi L, Yokoya NS, Ostini S, Pereira RTL, Braga ES, Oliveira EC (2008) Nutrients removed by Kappaphycus alvarezii (Rhodophyta, Solieriaceae) in integrated cultivation with fishes in recirculating water. Aquaculture 277(3–4):185–191
Hernández I, Fernández-Engo MA, Pérez-Lloréns JL, Vergara JJ (2005) Integrated outdoor culture of two estuarine macroalgae as biofilters for dissolved nutrients from Sparus aurata waste waters. J Appl Phycol 17:557–567
Kaewmesri A (2000) The treatment of wastewater from Penaeus monodon farming by the agarophyte, Gracilaria fisheri (Xia & Abbott) Abbott, Zhang & Xia. M Sc Thesis, Graduate School, Mahidol University, Bangkok (in Thai)
Kang YH, Hwang JR, Chung IK, Park SR (2013) Development of a seaweed species-selection index for successful culture in a seaweed-based integrated aquaculture system. J Ocean Univ China (Ocean Coast Sea Res) 12(1):125–133
Kanjana K, Radtanatip T, Asuvapongpatana S, Withyachumnarnkul B, Wongprasert K (2011) Solvent extracts of the red seaweed Gracilaria fisheri prevent Vibrio harveyi infections in the black tiger shrimp Penaeus monodon. Fish Shellfish Immunol 30:389–396
Kerrigan D, Suckling CC (2016) A meta-analysis of integrated multitrophic aquaculture: extractive species growth is most successful within close proximity to open-water fish farms. Rev Aquac 0:1–13
Khidprasert S (1995) Absorption of nitrogen compounds from shrimp pond effluence by marine algae. M Sc Thesis, Graduate School, Kasetsart University, Bangkok (in Thai)
Kunawongdet P (2020) Biological water treatment in recirculating aquaculture system by using Caulerpa lentillifera J Agardh. Burapha Sci J 25(2):509–523. (in Thai)
Limhang K, Saeheng L, Khunpol K (2014) Efficiency for treatment of nitrogen compound of the water from rearing pacific white shrimp (Litopenaeus vannamei) using sea grapes (Caulerpa macrophysa). Proceedings of 52nd Kasetsart University annual conference: fisheries. Agricultural Extension and Home Economics, Bangkok, pp 226–233. (in Thai)
Manori Bambaranda BVAS, Sasaki N, Chirapart A, Salin KR, Tsusaka TW (2019a) Optimization of macroalgal density and salinity for nutrient removal by Caulerpa lentillifera from aquaculture effluent. PRO 7(303):1–16. https://doi.org/10.3390/pr7050303
Manori Bambaranda BVAS, Tsusaka TW, Chirapart A, Salin KR, Sasaki N (2019b) Capacity of Caulerpa lentillifera in the removal of fish culture effluent in a recirculating aquaculture system. PRO 7(440):1–15. https://doi.org/10.3390/pr7070440
Marinho G, Nunes C, Sousa-Pinto I, Pereira R, Rema P, Valente LMP (2013) The IMTA-cultivated Chlorophyta Ulva spp. as a sustainable ingredient in Nile tilapia (Oreochromis niloticus) diets. J Appl Phycol 25(5):1359–1367
Marinho-Soriano E, Morales C, Moreira WSC (2002) Cultivation of Gracilaria (Rhodophyta) in shrimp pond effluents in Brazil. Aquac Res 33:1081–1086
Martínez-Córdova LR, Peña-Messina E (2005) Biotic communities and feeding habits of Litopenaeus vannamei (Boone 1931) and Litopenaeus stylirostris (Stimpson 1974) in monoculture and polyculture semi-intensive ponds. Aquac Res 36:1075–1084
Neori A (2008) Essential role of seaweed cultivation in integrated multi-trophic aquaculture farms for global expansion of mariculture: an analysis. J Appl Phycol 20:567–570
Neori A, Guttman L (2017) Thoughts on algae cultivation toward an expansion of aquaculture to the scale of agriculture. In: Proceedings of 7th international conference on innovation in chemical, agricultural, biological and environmental sciences (ICABES-2017), London (UK). 4–6 Dec 2017. https://doi.org/10.15242/HEAIG.H1217234. Accessed 21 Nov 2020
Neori A, Shpigel M, Ben-Ezra D (2000) A sustainable integrated system for culture of fish, seaweed and abalone. Aquaculture 186:279–291
Neori A, Chopin T, Troell M, Buschmann AH, Kraemer GP, Halling C, Shpigel M, Yarish C (2004) Integrated aquaculture: rationale, evolution and state of the art emphasizing seaweed biofiltration in modern mariculture. Aquaculture 231:361–391
Niu J, Xie J-J, Guo T-Y, Fang H-H, Zhang Y-M, Liao S-Y et al (2019) Comparison and evaluation of four species of macro-algae as dietary ingredients in Litopenaeus vannamei under normal rearing and WSSV challenge conditions: effect on growth, immune response, and intestinal microbiota. Front Physiol 9(1880):1–15
Pariyawathee S, Songsangjinda P, Deraxbudsarakom S, Tuntichodok P, Chiayvareesajja S (2003) Optimum condition of environmental factors for growth of sea grape (Caulerpa lentillifera J. Agardh). Thai Fish Gaz 56:443–448. (in Thai)
Pellizzari F, Reis RP (2011) Seaweed cultivation on the southern and southeastern Brazilian coast. Rev Bras Farmacogn Braz J Pharmacogn 21(2):305–312
Prud’homme van Reine WF, Trono Jr GC (2001) Plant resources of South-East Asia 15(1), cryptogams: algae. Backhuys Publishers, Leiden
Putri NT, Jusadi D, Setiawati M, Sunarno MTD (2017) Potential use of green algae Caulerpa lentillifera as feed ingredient in the diet of Nile tilapia Oreochromis niloticus. Jurnal Akuakultur Indonesia 16(2):195–203
Putro SP, Widowati W, Rahmansyah R, Suminto S (2016) Application of polyculture using stratified double net cage: a case study at Awerange Gulf, Barru, South Sulawesi, Indonesia. AACL Bioflux 9(4):815–822
Rana KJ, Siriwardena S, Hasan MR (2009) Impact of rising feed ingredient prices on aquafeeds and aquaculture production. FAO Fisheries and Aquaculture Technical Paper. No. 541. Rome, 63 p
Ratana-arporn P, Chirapart A (2006) Nutritional evaluation of tropical green seaweeds Caulerpa lentillifera and Ulva reticulata. Kasetsart J (Nat Sci) 40(Suppl):75–83
Rudtanatip T, Asuvapongpatana S, Withyachumnarnkul B, Wongprasert K (2014) Sulfated galactans isolated from the red seaweed Gracilaria fisheri target the envelope proteins of white spot syndrome virus and protect against viral infection in shrimp haemocytes. J Gen Virol 95:1126–1134
Shama A, Joyce SG, Mari FD, Ranga Rao A, Ravishankar GA, Hudaa N (2019) Macroalgae and microalgae: novel sources of functional food and feed. In: Ravishankar GA, Ranga Rao A (eds) Handbook of algal technologies and phytochemicals. Food, health and nutraceutical applications, vol-I. CRC Press, USA, pp 207–219
Siddik MAB, Anh NTN (2015) Preliminary assessment of the gut weed Ulva intestinalis as food for herbivorous fish. Int Aquat Res 7:41–46
Sirirustananun N, Chen JC, Lin YC, Yeh ST, Liou CH, Chen LL, Sim SS, Chiew SL (2011) Dietary administration of a Gracilaria tenuistipitata extract enhances the immune response and resistance against Vibrio alginolyticus and white spot syndrome virus in the white shrimp Litopenaeus vannamei. Fish Shellfish Immunol 31:848–855
Sitthimong C (2020) The situation of white shrimp farming. https://www.fisheries.go.th/extension/whitesh.htm. Accessed 2 Nov 2020
Songsangjinda P, Inek S (2003) Efficiency of oyster (Crassostrea lugubris) green mussel (Perna viridis) and seaweed (Gracilaria fisheri) on quality improvement of effluent from shrimp pond. Technical Paper No. 2/2003, Coastal Fisheries Research and Development Bureau, Department of Fisheries. Ministry of Agriculture and Cooperatives. 15 p. (in Thai)
Sriveerachi T, Pankdee S (2005) Culture on seaweed Gracilaria edulis (Gmelin) Silva and Acanthophora spicifera (Vahl) Børgesen in biologically-treated water pond. Technical Paper No. 17/2005, Coastal Fisheries Research and Development Bureau, Department of Fisheries. Ministry of Agriculture and Cooperatives. (in Thai)
Sukiman F, Rohyani IS, Ahyadi H (2014) Growth of seaweed Eucheuma cottonii in multi trophic sea farming systems at Gerupuk Bay, Central Lombok, Indonesia. Nus Biosci 6:82–85
Suthiniam S, Chiewwarisajja S, Chiewwarisajja J, Silapaachan K (2009) Efficiency of nitrogen and phosphorus treatment of feather seaweed in effluents from white shrimp farming. Thai Fish Gaz 62(3):215–220. (in Thai)
Tacon AGJ (1997) Fishmeal replacers: review of antinutrients within oilseeds and pulses – a limiting factor for the aquafeed green revolution? In: AGJ T, Basurco B (eds) Feeding tomorrow’s fish. CIHEAM, Zaragoza, pp 153–182. (Cahiers Options Méditerranéennes; n. 22)
Takeshi S, Yumiko Y, Joko S (2005) Mineral components and antioxidant activities of tropical seaweeds. J Ocean Univ China 4:205–208
Tamtin M, Chaisri C, Kosawadpat P, Kaukaew J, Lia-Thongkhom N (2016) Nutritional value of sea lettuce (Ulva rigida) and application as feed ingredients in Pacific white shrimp (Litopenaeus vannamei Boone, 1931) diets. Technical Paper No. 12/2016, Coastal Fisheries Research and Development Bureau, Department of Fisheries. Ministry of Agriculture and Cooperatives. (in Thai)
Teekayu K, Khidprasert S (1996) Wastewater treatment from shrimp farming using seaweed, Gracilaria. Reduce the amount of nitrogen compounds. Faculty of Agriculture, Bang Phra Rajamangala Institute of Technology (in Thai)
Thongcanarak P, Predalumpaburt Y (2008) Comparison the efficiency of 4 seaweeds in the removal of nitrogen and phosphate in effluent from orange spotted grouper (Epinephelus coioides Hamilton, 1822) culture. Technical Paper No. 40/2008, Coastal Fisheries Research and Development Bureau, Department of Fisheries. Ministry of Agriculture and Cooperatives. (in Thai)
Tookwinas S, Songsangjinda P, Matsuda O, Na-anan P, Ekpanithanpong U (2001) Experiment on integrated physical and biological treatment processes for treatment of intensive marine shrimp farming’s effluent. In: Proceedings of the JSPS-NRCT seminar on sustainable shrimp culture and health management diseases and environment. Tokyo University of Fisheries, Tokyo
Troell M (2009) Integrated marine and brackish water aquaculture in tropical regions: research, implementation and prospects. In: Soto D (ed) A global review of integrated marine aquaculture. FAO Fisheries and Aquaculture Technical Paper. No. 529, Rome
Troell M, Halling C, Nilsson A, Buschmann AH, Kautsky N, Kautsky L (1997) Integrated marine cultivation of Gracilaria chilensis (Gracilariales, Rhodophyta) and salmon cages for reduced environmental impact and increased economic output. Aquaculture 156:45–61
Troell M, Tyedmers P, Kautsky N, Rönnbäck P (2004) Aquaculture and energy use. In: Encyclopedia of energy, vol 1, pp 97–108. Amsterdam, Boston. Elsevier Academic Press.
Tsutsui I, Kanjanaworakul P, Srisapoome P, Aue-umneoy D, Hamano K (2010) Growth of giant tiger prawn, Penaeus monodon Fabricius, under co-culture with a discarded filamentous seaweed, Chaetomorpha ligustica (Kützing) Kützing, at an aquarium-scale. Aquacult Int 18:545–553
Tsutsui I, Songphatkaew J, Meeanan C, Aue-umneoy D, Sukchai H, Pinphoo P et al (2015) Co-culture with Chaetomorpha sp. enhanced growth performance and reduced feed conversion ratio of the giant tiger prawn, Penaeus monodon. Int Aquat Res 7:193–199
Voorhees JM, Barnes ME, Chipps SR, Brown ML (2019) Bioprocessed soybean meal replacement of fish meal in rainbow trout (Oncorhynchus mykiss) diets. Cogent Food Agric 5(1):1579482. https://doi.org/10.1080/23311932.2019.1579482. Accessed 9 Dec 2020
Wongprasert K, Rudtanatip T, Praiboon J (2014) Immunostimulatory activity of sulfated galactans isolated from the red seaweed Gracilaria fisheri and development of resistance against white spot syndrome virus (WSSV) in shrimp. Fish Shellfish Immunol 36:52–60
Yala ZR, Sulistiawati D, Masyahoro N (2017) Seaweed (Eucheuma cottonii) growth in polyculture application. AACL Bioflux 10(5):1064–1073
Yangthong M, Ruensirikul J (2020) Feed intake stimulation of juvenile spotted scat (Scatophagus argus Linnaeus, 1766) using dietary seaweed supplementation (Ulva sp.). Aquaculture 529:735626. https://doi.org/10.1016/j.aquaculture.2020.735626. Accessed 8 Dec 2020
Zahra A, Sukenda S, Wahjuningrum D (2017) Extract of seaweed Gracilaria verrucosa as immunostimulant to controlling white spot disease in Pacific white shrimp Litopenaeus vannamei. Jurnal Akuakultur Indonesia 16(2):174–183
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Chirapart, A., Ruangchuay, R. (2022). Seaweed in Aquaculture: An Overview. In: Ranga Rao, A., Ravishankar, G.A. (eds) Sustainable Global Resources Of Seaweeds Volume 1. Springer, Cham. https://doi.org/10.1007/978-3-030-91955-9_20
Download citation
DOI: https://doi.org/10.1007/978-3-030-91955-9_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-91954-2
Online ISBN: 978-3-030-91955-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)