Abstract
Australia’s separation from other land masses has resulted in the evolution of flora and fauna in relative isolation. The arrival of Europeans some 230 years ago marked the beginning of a mass invasion of the continent by alien plant (and animal) species from across the globe. These mass invasions have had profound effects on the Australian landscape and its native species and have required significant management interventions. In this chapter, we present an overview of the history of alien plant invasions in Australia and the scope of the current situation in terms of the number of species introduced. Seven case studies illustrate the nature of the invasive weed issues and the actions undertaken towards management. Case studies include Australian Weeds of National Significance (WoNS) (Chrysanthemoides monilifera subsp. rotundata, Lantana camara) and environmental and agriculturally important invasive plants (Opuntia inermis, Opuntia stricta, Echium plantagineum, Cucumis myriocarpus, Citrullus lanatus, Andropogon gayanus) as well as recent incursions (Bassia scoparia). Each case study outlines the impacts and risks associated with the invasion and presents the unique management approaches adopted - asset protection, biological control, successful eradication and ecosystem transformation. Several case studies draw comparisons between the establishment and persistence of alien plants in Australia and their native ranges and provide important clues on key traits that contribute to their successful invasion. Results to date have shown that the number of introduced plants to Australia has increased exponentially across most states and territories, particularly in recent years. Targeted control strategies for some WoNS such as Chrysanthemoides monilifera subsp. rotundata have been successful due to significant investment in the study of weed biology and physiology and subsequent development of effective integrated weed management strategies. The management strategies for several WoNS and invasive plants are evolving, through continued involvement of local, state and federal government and nongovernmental agencies and researchers to identify more effective control strategies. Lastly, future management challenges are described, including the expanding alien flora, the potential impact of climate change and risk management associated with weed incursion. Unique Australian insights can provide potential examples for other countries facing similar challenges as alien plants are now a global problem.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Case studies
- Climate change
- Historical invasion patterns
- Impacts
- Invasion biology
- Management
- Weeds of national significance
6.1 Introduction
Australia is known for the unique and diverse native flora and fauna that have originated following a long period of isolation as an island continent. However, with the arrival of European settlers came many non-native plant species including both deliberate (i.e. crops) and accidental introductions. These initial introductions of alien plants from Europe were followed by other introductions from across the globe. Many of the alien plants introduced to Australia have subsequently naturalised and spread over vast areas of the continent, where they pose significant impacts to the native Australian flora and fauna. These plants have invaded arid, tropical, temperate and alpine habitats and encompass all life forms (i.e. herbs, grasses, vines, shrubs and trees), and their management has required the development of a diverse array of approaches and strategies. This chapter presents a brief overview of the history of plant introductions in Australia and describes the factors influencing successful invasion and management responses through a series of case studies highlighting the unique aspects of the Australian context.
While the nature and scope of alien plant invasions have been documented globally (Pimentel 2011), the species, their impacts and effective management responses vary considerably between continents and countries. Multiple factors contribute to this variation including (a) the history of plant invasions in the region investigated (i.e. which species have been introduced, the introduction date and country of origin or donor country (see Pyšek et al. 2021, Chap. 7)), (b) the invasiveness of individual species in each region (including their biology and traits), (c) the resilience of native ecosystems to invasion or individual alien plant species, and (d) the management strategy (including policy and legislation) developed and relative success of implementation. While there are commonalities among successful global invaders, unique aspects of alien plant invasions on each continent are frequently associated with regional variation. This chapter therefore explores plant invasion and management from an Australian perspective.
6.1.1 History of Plant Invasions in Australia: An Overview
6.1.1.1 Pre-1788
Records and evidence of plant invasions prior to the European colonisation of Australia in 1788 are scarce. However, three potential sources of plant invasions during this period of Australia’s history have been described. These include (i) natural invasions, (ii) anthropogenic activity of Aboriginal (and Torres Strait Islander) people, and (iii) other travellers to Australian shores.
-
(i)
Natural invasions (i.e. without human assistance) – Joseph Hooker described the Australian flora [from the essay ‘On the Flora of Australia’ (1859)] as containing three distinct elements: (1) an Australian or autochthonous element (being mainly endemic and near endemic species, many of which are xeromorphic), (2) an Antarctic element (i.e. species like Nothofagus) and (3) an Indo-Malayan element (i.e. tropical and subtropical rainforest assemblages). The characterisation of these three elements led to the idea of ‘mass invasions’ in Australia’s past, although the nature of these invasions was undefined (Beadle 1981). Based on the theory of continental drift, Beadle (1981) provided more conclusive evidence for the origins of the Australian flora based on three main phases: (a) the ancient Gondwanaland flora; (b) the xeromorphic flora, originating from Gondwanan lineages; and (c) the arid zone flora which is derived from Gondwanan lineages, xeromorphic taxa and littoral taxa, although some elements of the Australian flora must have originated outside of Australia to account for the numerous taxa of Southeast Asian origin (Beadle 1981). However, Australia has only been sufficiently close enough for biotic exchange with Asia to have occurred in the past 15 M years, with migratory routes possibly occurring only in the last several million years (Smith 1986). For example, two species of the widespread northern hemisphere genus Rhododendron (R. viriosum and R. lochiae) occur in Australia, both of which are restricted to northern Queensland and are believed to have spread as a result of this biotic exchange (Smith 1986). Analysis of the flora of northern Australia (i.e. above 15oS latitude) revealed that about 28% of the 2220 species also occur outside of Australia (Specht and Mountford 1958). Other natural invasions to Australia include the many ‘cosmopolitan’ coastal species which are widespread across the Indian and Pacific Ocean countries and islands (e.g. Cakile edentula) and are likely to have dispersed through ocean currents (see further discussion by Groves (1986)).
-
(ii)
Aboriginal people – The arrival of Aboriginal people over 50,000 years ago in Australia does not appear to have been associated with any significant plant invasions (Groves 1986). This is possibly not surprising given the Aboriginal people’s close affiliation to the environment, as alien plants are likely to conflict with their cultural connection to the land (Smith 2002). In addition to the Aboriginal people, the Torres Strait Islanders are an indigenous group of people who occupied northern Queensland and at least 38 islands in the 150 km Torres Strait between the northern tip of Australia and the southern coast of Papua New Guinea. While there is no documented evidence that the Torres Strait Islanders introduced plant species into Australia, they regularly travelled between Australia and Papua New Guinea, and thus it is highly likely that some plants could have been dispersed over time. However, certain animals, including dingo (Canis familiaris dingo), have now been dated in terms of their arrival to this continent to be linked with migrating human populations. In 2020, the first whole genome sequencing of the dingo and the New Guinea singing dog was undertaken. It indicated that the ancestors of these two dogs arose in Southeast Asia around 9900 YBP (years before present) and reached Australia 8300 YBP. Given the approximately 150 km of ocean containing numerous islands between Papua New Guinea and Australia, it is assumed that both flora and fauna could have arrived with migrating peoples at that time (Zhang et al. 2020). Phylogenetic studies employing molecular markers such as nuclear, chloroplast and mitochondrial genes as well as whole genome sequencing will certainly assist in determining invasion routes and approximate time of introduction in future population studies (Zhu et al. 2019).
-
(iii)
Other groups of visitors to Australian shores – The Makassans, from the region of Sulawesi in modern-day Indonesia, were regular visitors to northern Australia between 1700 and 1900 collecting trepang (or sea cucumber) . They are believed to have brought tamarind (Tamarindus indica) during these visits, which subsequently naturalised and spread (Macknight 1976). Tamarind is suspected to be the first naturalised plant in Australia as a result of human activities (Groves 1986). While many European [mostly Dutch] explorers mapped significant parts of the Australian coastline following Willem Janszoon’s ‘accidental’ discovery of Australia in 1606 (which he thought at the time was New Guinea) (Peters 2006), there is no formal or written evidence of plant invasions linked to these explorations. However, given the number of expeditions to Australia and the level of European activity in the region just north of Australia, transportation of alien plants during these maritime expeditions undoubtedly occurred (Bean 2007). A number of non-native plant species have been recorded from Timor and Indonesia with origins that pre-date European migration to the region (Bean 2007). Currently, evidence suggests that visitors from southern Asia travelled to the Australian mainland well before 5000 BCE (before the Common Era) based on the presence of archaeological artefacts (see Bowdler 2002), but their role in the dispersal of non-native plants is currently unknown. Bean (2007) argues that it is highly likely there was an alien flora present in Australia prior to European arrival. He hypothesised that some of the plant species collected in 1770 by the European botanists Banks and Solander, combined with specimens from other early botanists, may not be indigenous to Australia based on several key characteristics (i.e. invasive elsewhere, occur beyond natural dispersal limits, form disjunct populations, etc.). Following examination of early collections, Kloot (1984) concluded that about 100 plant species could have naturalised in Australia prior to European arrival. Bean (2007), however, suggests that this number could be much higher.
6.1.1.2 Post-1788
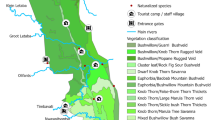
The arrival of the First Fleet in 1788 saw the deliberate [and documented] introduction of many new plant species to Australia. While most were of agricultural importance (i.e. crops and fruit trees), numerous potentially invasive alien species including coffee (Coffea arabica) , guava (Psidium guajava) , bamboo (subfamily Bambusoideae – exact species not documented), prickly pear (Opuntia sp.) and Spanish reed (Arundo donax) were introduced. The first reliable documented account of naturalised plants in Australia comes from Robert Brown who recorded 29 non-native plants growing around Sydney between 1802 and 1804 (Groves 2002), although not all were considered to be ‘weedy’ at the time. Subsequently, nearly all have become widely naturalised (Groves 2002). Following Brown’s account, botanists and naturalists continued to compile lists of naturalised plant species, initially for specific regions and later the individual states and territories (Fig. 6.1). However, it was not until the late 1980s that the first complete national list of naturalised plants was produced (Table 6.1). Since European settlement, the number of alien plant species introduced to Australia has exceeded 29,000 of which approximately 10% (>3000) have become successfully naturalised (Table 6.1). Interestingly, the number of vascular plants introduced into Australia exceeds the number of native vascular plants by 7750 (Table 6.1). Additionally, the recent rate of plant naturalisations has accelerated (see Fig. 6.1). Groves and Hosking (1998) identified 295 taxa that naturalised in Australia over a 25-year period (1971–1995). Dodd et al. (2015) showed the increase in naturalisation rates to be linear over the period 1880–2000, based on herbaria collections of naturalised plants as a proportion of total herbarium collections. It must be noted that the alien flora held in many herbaria across Australia is likely to be an underrepresentation of the actual number, as alien specimens are generally or routinely not collected by trained botanists.
An estimate of the number of naturalised plant species introduced over time for each Australian state/territory. Data collated from published records of naturalised plant species. ACT = Australian Capital Territory, NSW = New South Wales, NT = Northern Territory, Qld = Queensland, SA = South Australia, Tas = Tasmania, Vic = Victoria and WA = Western Australia. The overall trend line is also presented (note: trend line presented is not representative of the mean value across all states territories as they differ in size and invasion history)
The significant increase in the number of naturalised plants reported between 1990 and 1997 may be attributed to the inclusion of species from various compiled data sources rather than an actual time step increase in numbers during this period (Fig. 6.1; Table 6.1). Furthermore, citizen science websites such as Canberra Nature Map (CNM : https://canberra.naturemapr.org/) for the Australian Capital Territory (ACT) have led to a proliferation of reports on new naturalisation rates, which are also not reflective of information supported by historic herbaria collections. For example, analysis of CNM records showed that between 2015 and 2019, over 570 alien plant species (spanning 3800 records/sighting) were reported through CNM, including many species previously not reported in the ACT (Mulvaney unpublished data).
The largest proportion of naturalised flora in Australia originated from Europe (47.4%), followed by the Americas (29.9%), Africa (14.3%), Asia (6.3%) and Oceania (2.1%), with an increasing number of source countries reported over time (Dodd et al. 2015). Initially, plant introductions originated mainly from Europe and Africa, a reflection of the actual trading routes and maritime passages (Groves 1986). The majority of naturalised plant species were imported to Australia originally as ornamental plants (66%) (Groves et al. 2005), with fewer introduced for agricultural purposes (7%); these findings are consistent with similar assessment in other countries like the USA and Canada. In one assessment, Lonsdale (1994) documented 463 grasses and legumes which were introduced into northern Australia between 1947 and 1985 for pasture improvement. Although only 21 were eventually recommended for use, approximately 60 have subsequently become invasive. Given that 94% of all alien plants introduced into Australia (n = 26,242) were introduced through the gardening [ornamental] sector (Virtue et al. 2004), and many of the naturalised ornamental plant species examined were available for sale (Groves et al. 2005), the contribution of this sector to the increase in environmental weeds in recent years is significant (Table 6.2).
In response to the arrival of a large number of invasive alien plants in Australia, the Commonwealth Government released the first National Weeds Strategy in 1997 (ARMCANZ et al. 1997). A key goal of this strategy was ‘to reduce the impact of existing weed problems of national significance’. To further address this goal, the Commonwealth Government took a species-led approach to prioritise 20 Weeds of National Significance (WoNS) in 2000, from a list of 71 major invasive plant species nominated by a range of experts (see Thorp and Lynch 2000). Significant investment and resources were then directed towards these 20 WoNS, including dedicated coordinators, national management groups, creation of national strategies, best practice management manuals and specific funding for a wide range of on-ground management activities and research priorities (e.g. biocontrol and ecological studies). Significant outcomes were achieved in the management of the 20 species selected (see several examples in case studies below). However, management of other nationally significant invasive plant species was put at a distinct disadvantage as a consequence of not being listed as a WoNS. In some cases, these species were not ranked as WoNS as information on their ecology and impacts were either poorly understood or documented.
Given the growing evidence pointing to the need of urgent national management of other species, an expanded list of 32 WoNS species was released in 2012 (the actual number is higher as several listings are groups of weeds, e.g. bitou bush/boneseed, brooms, Opuntioid cacti and asparagus weeds) (Table 6.3). Specifically, invasive vines were highlighted as a rapidly increasing problem following the publication of several Biology of Australian Weeds research articles (e.g. Anredera cordifolia (Vivian-Smith et al. 2007)). This recognition led to invasive vines being listed as a Key Threatening Process under the NSW Threatened Species legislation and subsequently their listing in the second group of WoNS (see Table 6.3).
Despite the creation of the WoNS programme and its successes, there are still many nationally important invasive plant species not encompassed by the WoNS programme. Many of these non-WoNS species were considered to be significant invasive alien species for decades (see non-WoNS case studies in Sect. 6.3). For example, during the first Australian Weeds Conference held in 1954, research papers were presented on nine species - three would go on to become part of the initial 20 WoNS (Rubus fruticosus, Lantana camara and Nassella trichotoma) and a fourth in the expanded list (Lycium ferocissimum). The remaining five species (Phragmites australis, Senecio jacobaea, Chondrilla juncea, Oxalis pes-caprae and Rosa rubiginosa) are still considered to be major invasive alien species today. Raising their profile and highlighting their impacts will require more than just research into their ecology and biology, as it is unlikely that every nationally significant invasive alien plant species in Australia can be accounted for under the single species WoNS approach. However, a focus on individual species can provide models for potential adoption for management of other invasive plant species (as highlighted in the case studies).
The case studies presented below represent a select group of invasive alien plant species chosen to illustrate the state of plant invasions in Australia and likely future directions for invasive plant management. We present examples of invasive alien plants that have seriously impacted the Australian landscape and led to the development of critical management strategies for their reduction or eradication. In some cases, eradication has been successful, while in others the invader continues to be a significant threat. Australians have typically employed various strategies for management, including assessment of the risk to prioritise investment in management, use of biological control agents to control widespread species with significant impacts and the successful integration of chemical, physical and cultural management strategies over time for reduction of propagules and seed banks.
6.2 Case Studies: Weeds of National Significance
6.2.1 Bitou Bush (Chrysanthemoides monilifera subsp. rotundata)
The South African plant Chrysanthemoides monilifera subsp. rotundata was likely accidently introduced into Australia in the early 1900s, where it subsequently established and spread. In the 1950s, however, it was deliberately planted along large sections of the New South Wales (NSW) coast to stabilise and revegetate sand dunes after mining for a range of minerals. By the 1980s there was growing concern about its invasive potential and significant threat to native plants. In 1999 it was listed as a Key Threatening Process (KTP) under the NSW threatened species legislation and as a WoNS in 2000.
In addition to the WoNS programme, the NSW Government released a Threat Abatement Plan (TAP) for the species in 2006 to meet the requirement of the KTP listing. The TAP identified 150+ plant species and 24 ecological communities that were directly threatened. The TAP established a new management approach for invasive plant species that threaten biodiversity, in that the native species threatened by C. monilifera subsp. rotundata were identified along with sites where control would lead to a conservation outcome (see Downey 2010). Over the proceeding 10+ years since the TAP was released, control of C. monilifera subsp. rotundata has occurred at 110+ priority sites outlined in the TAP across coastal NSW providing protection to over 95% of the plant species threatened. The TAP was supported by the broader WoNS programme which included containment zones, a comprehensive biocontrol programme and community education and awareness campaign and control in other priority areas as well as the development of best practice guidelines and management. The WoNS programme also included a range of initiatives and management outcomes for the closely related C. monilifera subsp. monilifera (Boneseed) which is also a major invasive plant species in Australia and jointly a WoNS species. Additionally, the other four subspecies not present in Australia were banned from import based on their risk and invasive potential, a decision that was later supported through detailed modelling (Beaumont et al. 2014). Comparisons of native and alien range data can provide useful insights into management and our general understanding of why a particular species can become invasive as an alien. For example, Beaumont et al. (2014) showed that the climatic envelope for C. monilifera subsp. monilifera in Australia was significantly greater than in its native range, in part because the southern latitudes where it is invasive in Australia and New Zealand are not present in South Africa.
6.2.2 Lantana (Lantana camara)
Lantana camara is a woody shrub originating in South and Central America and was introduced into Australia for ornamental purposes in 1841. Shortly thereafter it escaped cultivation and spread extensively, but it was not until the early 1900s that active management was advocated and initiated. It is now estimated to have invaded over 5 M hectares in Australia, including pastures, croplands and native habitats. Despite 100+ years of active management, these efforts have been largely unsuccessful, as is the case in other parts of the world (Bhagwat et al. 2012).
The first biocontrol agent was released on L. camara in Australia in 1916. Despite the release of 26 other agents over the past 100 years, successful control has not been achieved (Palmer et al. 2010). Genetic analysis of L. camara revealed that the species is a ‘complex’, as no recent records of parental species could be identified. Instead, the current ‘species’ is the outcome of extensive hybridisation combined with polyploidy/polyploidisation within and between wild, cultivated varieties, and naturalised taxa (Goyal and Sharma 2015). Such significant genetic variation may limit successful implementation of a host-specific biocontrol agent as the ‘species’ is unlikely to exist in the native range.
As outlined above, given the area invaded by L. camara and its residence time in Australia and the fact that broad-scale control or eradication is unlikely, one of the major WoNS initiatives was the development of a national plan to protect assets at risk from L. camara invasion (see BQ 2010). This asset-based approach is based on the Australian government’s biosecurity model for all alien species in Australia in which management is based on a theoretical generalised invasion curve of residence time relative to area invaded overlaid with four standard management objectives (prevention, eradication, containment and asset protection) (EWWG 2007). Prior to the development of the national plan, the biological assets threatened by L. camara invasion were thought to be limited to specific species, or areas, despite acknowledgement of broader more significant potential impacts (e.g. Gentle and Duggin, 1997).
Two major approaches were adopted to determine the biodiversity threat. Firstly, a range of rapid assessments was undertaken from invaded and non-invaded sites across southeastern Australia which showed an impact threshold effect of L. camara density on native species. The threshold varied for various plant communities; however, the greater the cover of L. camara, the lower the species richness of native species (Gooden et al. 2009). Secondly, an assessment (using a modified version of the methodology developed for C. monilifera subsp. rotundata) was used to determine the biodiversity at risk (see case study 6.2.1 for details). This assessment revealed that L. camara threatened 1321 native plant species, 158 native animal species and 150+ ecological communities. Of these, 275 native plant and 24 native animal species required immediate protection nationally (Turner and Downey 2010). The identification and prioritisation of specific species at risk from L. camara invasion enabled evaluation of individual sites and targeted conservation efforts on areas of greatest need and likelihood of achieving a successful outcome using a conservation triage approach (Downey et al. 2010a).
Apart from threatening native biodiversity, L. camara has been reported as poisonous to domesticated animals (e.g. sheep, cattle, buffalo and guinea pigs). The liver and kidneys of animals that consume L. camara typically exhibit a characteristic increase in serum glutamic-oxaloacetic transaminase activity and hepatic and renal xanthine oxidase activity, resulting in obstructive jaundice and subsequent photosensitisation (Sharma et al. 1981). Leaves and stems of L. camara have exhibited nematicidal properties that could also potentially assist in its invasion success (Begum et al. 2008) and may potentially provide lead molecules for the development of bionematicides (Udo et al. 2014) and biofungicides (Singh and Srivastava 2012).
Many invasive plant species with successful invasion tendencies have also been noted to possess strong allelopathic potential. Lantana camara shows potential for allelopathic activity resulting in reduced vigour in native plants, thereby reducing native biodiversity and disrupting succession within invaded habitats (Gentle and Duggin 1997; Day et al. 2003). Similar inhibitory effects of L. camara and its residues were also observed on vegetable crops (e.g. Chinese cabbage, chilli and rape seed (Sahid and Sugau, 1993). While the phytotoxic activity of L. camara has been reduced over time through vermicomposting of residues (Hussain et al. 2016), its unique allelochemical composition has proven effective for the control of another invasive alien species, water hyacinth (Eichhornia crassipes) (Saxena 2000). Chemical defences resulting in allelopathic potential have been shown to play a role in plant invasion success (Latif et al. 2017) and in the case of L. camara may also contribute to its persistence.
6.3 Case Studies: Weeds Not of National Significance
6.3.1 Prickly Pear (Opuntia inermis and Opuntia stricta)
The WoNS programme described previously in this chapter was not the first nationally significant species-led programme in Australia. Eighty years earlier the Australian Government initiated the Common wealth Prickly Pear Board in 1920 in response to the enormous scale of the prickly pear (Opuntia inermis and Opuntia stricta ) invasion (Dodd 1940). Prickly pear species were first introduced to Australia in 1788 with the arrival of Europeans, but it was not until ~1900 that the species was reported to be spreading rapidly, and by 1913 it was reported to have infested over 6.3 M hectares. At the peak of invasion in 1925, it was estimated that prickly pear had spread over 24 M hectares, half of which was so infested that it was unable to be grazed by livestock. Many farming properties were subsequently abandoned as the cost of mechanical and chemical control was economically impracticable, exceeding the value of the land by over 20-fold. At its peak, the densities of prickly pear were estimated at 16,000 plants per hectare with an estimated biomass of 250,000 kilograms (kg) per hectare (Dodd 1940).
In the 1920s, the Prickly Pear Board was established with the express purpose of finding an effective biological control agent. The Prickly Pear Board introduced the cactoblastis moth (Cactoblastis cactorum) into Australia in 1925, and it was released after mass rearing in early 1926. By 1930 some 3 billion eggs had been reared and released throughout eastern Australia. The biocontrol agent was so successful that large-scale destruction of prickly pear was observed by 1933. However, this initial success was followed by mass reductions of the moth population due to low prickly pear densities. In subsequent years, prickly pear densities rebounded due to a lack of the biocontrol agent. The moth numbers subsequently recovered, and by 1939 comprehensive control was achieved enabling abandoned land to be reclaimed. The cost of the 19-year programme was AUD£168,600 [equivalent to AUD$12 M in 2019], providing a perspective on the expenses required for the successful management of this invasive plant species (Dodd 1940). Today, prickly pear is still observed across eastern Australia, but at much reduced numbers, and is typically eradicated where possible by spot spraying of herbicides. Although prickly pear infestation has generally been kept under control, a large number of other opuntioid cacti have become significant invasive plant species in Australia in recent years, leading to their inclusion in the second WoNS list. Interestingly, Cactaceae is only one of two major plant families absent from the Australian flora (Beadle 1981).
6.3.2 Paterson’s Curse (Echium plantagineum)
Echium plantagineum is an invasive annual herbaceous plant that was introduced into Australia in the 1840s from Europe. It was initially dispersed as an ornamental plant across southern Australia due to its impressive floral display of deep purple flowers. It readily escaped, and by the 1890s through several additional dispersal pathways (e.g. livestock and contaminated hay), it became widely established across large tracts of southeastern Australia and southern Western Australia (Piggin and Sheppard 1995). Today E. plantagineum has spread throughout every state and territory and invaded over 30 M hectares, forming dense purple monocultures in early to late spring when in bloom, particularly in years with optimal rainfall following a prolonged drought.
Invasion success of this species has been attributed to a range of traits including elevated genetic diversity, tolerance to a range of environmental conditions encountered across Australia (i.e. low rainfall and extreme temperatures to higher elevation and low temperatures), the production of bioactive secondary metabolites supporting improved plant defences (Zhu et al. 2017) and prolific seed set and seed dormancy (Piggin and Sheppard 1995).
Studies comparing plant demography between Australia and the native European range (i.e. Iberian Peninsula in Spain and Portugal) showed that E. plantagineum seedling establishment was two to five times greater in Australia and that seed bank incorporation also was three times higher leading to a greater abundance as an alien in Australia (Grigulis et al. 2001). Echium plantagineum is self-incompatible in the native range, but self-compatible in its invaded territory of Australia (Petanidou et al. 2012), potentially enhancing the ability to colonise and spread. In direct contrast to the less invasive congeneric alien plant species, E. vulgare, E. plantagineum exhibits significantly higher genetic diversity across its genome and possesses a smaller genome size (Zhu et al. 2017). Small genome size has also been associated with enhanced competitive ability and modification of morphological and physiological traits such as larger leaf area and higher photosynthetic rates owing to the reduced allocation of carbon resources for cell cycling (Bennett et al. 1998).
Apart from its physical adaptations for growth in extreme conditions, E. plantagineum has been shown to contain an arsenal of chemical defences that assist in its defence against other plants, pathogens and insect/mammalian herbivores. Aerial tissues of the plant contain high concentrations of pyrrolizidine alkaloids and their N-oxides (Skoneczny et al. 2019). These metabolites deter certain insect herbivores and have also been shown to be associated with livestock toxicity (Molyneux et al. 2011). The alien congener E. vulgare also contains pyrrolizidine alkaloids albeit at relatively lower concentrations (Skoneczny et al. 2017). The roots of E. plantagineum produce and release phytotoxic naphthoquinones, including acetylshikonin and shikonin, which have both been shown to be inhibitory to germinating alien plant seedlings and growth of mammalian cells (Durán et al. 2017). The biosynthesis of phytotoxic naphthoquinones (shikonins) in E. plantagineum is also upregulated in roots exposed to higher temperatures and under short-term drought conditions (Weston et al. 2013; Skoneczny et al. 2019).
Echium plantagineum was ranked 32nd in the assessment of the Australian Weeds of National Significance (WoNS) out of 71 weeds species nominated, despite being ranked as having the second highest cost of control and the third highest current distribution and ninth highest potential distribution and posing a significant threat to grazing livestock and land values (see Thorp and Lynch 2000). It was also not ranked as a WoNS in the second round in 2012, despite being a nationally significant invasive plant species, possibly due to the fact that management can be achieved in many cases with the application of a variety of cost-effective herbicides.
In Australia, the status of E. plantagineum can be contentious, with research showing both negative impacts on crop and pasture lands but positive value for beekeepers and for its use in cosmeceuticals and biomedicinals (Piggin and Sheppard 1995; Durán et al. 2017; Skoneczny et al. 2017). This conflict among diverse stakeholder groups has had a lasting legacy and has contributed to the current legislation and policies for its biological control in Australia.
The initiation of a biological control programme for E. plantagineum began in 1971 with agreement among state agricultural departments with biocontrol research programmes supported for many years. However, in 1978 the programme was challenged by apiarists who believed that the release of biocontrol agents would destroy a ‘valuable’ potential source of nectar for production of honey. The conflict lasted several years and involved multiple reviews/inquires, prolonged and protracted negotiations and finally legal proceedings, and as a result, the biocontrol programme was halted in 1983. The outcome of this conflict and a key resolution was the development and passage of the Commonwealth Biocontrol Act 1984 to ensure that conflicts relating to the release of biocontrol agents in Australia could be administered through a legislative instrument and framework (Cullen and Delfosse 1984). Another key measure to resolving the conflict was a report on the economic status that showed the annual management costs in the state of Victoria alone at AUD$ 3.2 M compared with an annual benefit of AUD$ 900,000 (Field et al. 1986). In 1988 the biocontrol programme was subsequently reinstated, and six agents were released with limited conflict or opposition (see Piggin and Sheppard 1995). Today, several of these agents can be found in high abundance across southern Australia (primarily the leaf beetles and stem/crown weevils) (Weston et al. 2012).
6.3.3 Prickly Paddy Melon (Cucumis myriocarpus) and Camel Melon (Citrullus lanatus)
Cucumis myriocarpus and Citrullus lanatus are annual invasive species in the Cucurbitaceae family and were introduced into Australia in the early to mid-1800s from sub-Saharan Africa. While C. myriocarpus was potentially introduced unintentionally, C. lanatus was introduced deliberately as a feed species for camels that were used at the time to transport construction materials to build roads and railways (Barker 1964). Both species are now considered significant invasive plants in broadacre and mixed cropping agricultural zones. Recently, Llewellyn et al. (2016) described them as major invasive alien species in summer grain crop rotations and fallows in Australia, and Borger et al. (2018) listed C. myriocarpus as a major problem in the Western Australian wheat belt. Both species are also invasive in natural ecosystems across drier inland regions of Australia where they pose a threat to native species (Hallett et al. 2014).
These introduced melons exhibit a similar range of traits that potentially contribute to their invasive ability and impact (Shaik et al. 2017). Such traits include seed dormancy, high seed production, drought tolerance, ability to be pollinated by non-specific pollinators (in the case of C. lanatus) and toxicity to some animal herbivores (i.e. horse, sheep and cattle deaths have been reported). Furthermore, both species have shown potential for allelopathic activity through the production of cucurbitacins and other unknown metabolites and have exhibited antimicrobial and nematocidal activity on soil organisms (Hao et al. 2007; Mafeo and Mashela 2010; Harrison et al. 2012).
Camel and prickly paddy melons can also be prodigious seed producers when irrigated or adequate soil moisture is available. Seeds of each species frequently exhibit dormancy which results in multiple ‘pulses’ of germination from early spring through to mid-summer depending on availability of soil moisture through rainfall or irrigation. The mucilaginous seed coats can adhere to vehicles and grazing animals aiding in seed dispersal, and parrots, particularly galahs, can also impact seed dispersal. The adaptability of C. lanatus to varied climatic conditions also supports its successful invasion across Australia (Ramirez et al. 2014). The ability of both melon species to produce adventitious roots under waterlogged conditions may also support its adaptation to regions experiencing flooding events in arid, subtropical and temperate climates (Shaik et al. 2016a).
Recent studies have shown that Australian populations of C. myriocarpus and C. lanatus exhibit limited genetic diversity, with each species represented by a single genotype in Australia (Shaik et al. 2015; Shaik et al. 2016a; Shaik et al. 2016b). This observed lack of genetic diversity may favour the eradication of these weeds using host-specific biological control agents. However, considerable research efforts are required to ascertain feasibility of introduction of biocontrol agents, given their genetic similarity to economically important melon species (e.g. watermelon). The recent recognition of the ecological and economic impacts of these invasive melons (e.g. Llewellyn et al. 2016) combined with recent studies on their phenology and biology (Shaik et al. 2017) has led to a reassessment of their risk level and status as major invasive plants in drier inland regions of Australia. Prior to this, the melons were considered to be low-priority alien plants of roadsides, railways corridors, stockyards and other disturbed sites (C. lanatus) and cultivated crops (C. myriocarpus). Recognition of their invasive status is likely to lead to improved management outcomes that are aligned to the actual risk and impact posed.
6.3.4 Gamba Grass (Andropogon gayanus)
Andropogon gayanus is an African C4 grass species which was introduced into Australia deliberately as a pasture grass in 1931. It subsequently invaded large tracts of pastureland in northern Australia (Queensland and Northern Territory), and areas with eucalypt open forest, woodland and savannas (Rossiter et al. 2003). Unlike the native grasses which are less than 0.5 m in height, A. gayanus grows to over 4 m, producing over four times the biomass of native grasses (up to 1.7 kg per m2). The increased height and biomass of A. gayanus in invaded sites result in altered fuel loads and increased intensity of bush fires (Rossiter et al. 2003). Rapid changes in ecosystem services illustrate how the grass-fire cycle (D’Antonio and Vitousek 1992) has enabled A. gayanus transformation of invaded ecosystems, particularly with respect to understorey composition across northern Australia.
By modification of grass-fire cycles (i.e. due its high biomass accumulation), A. gayanus has transformed ecosystems across northern Australia. In addition, production of 7 times more shoot N and 2.5 times greater root N compared with native grasses has altered soil N cycle. It also stimulates soil ammonification processes (Rossiter-Rachor et al. 2009) and affects soil moisture availability in invaded sites, with a trebling of water usage and a halving of deep-water drainage compared to uninvaded native grass sites (Rossiter et al. 2002). Andropogon gayanus invasion has also resulted in reduced tree canopy cover leading to mortality of certain native tree species (see Bowman et al. 2014). While such changes have significantly altered the grassland ecosystems of northern inland Australia, invertebrate composition between invaded and uninvaded sites has not yet been reported to be affected (Parr et al. 2010).
Seed biology of A. gayanus may also contribute to the invasion success of this weed in northern Australia. Bebawi et al. (2018) noted that seed persistence is supported by deeper burial of the seed, relative to shallow placement of seed which leads to loss of viability within 1 year of seed shedding in dry tropics of northern Australia. Andropogon gayanus typically germinates and persists well in cooler climates and has strong potential to expand across southern parts of Australia, particularly if predicted changes in Australia’s climatic zones are realised.
At this time, suitable management strategies for the vast areas invaded by A. gayanus include controlled burning, herbicide application and replanting of native trees and understorey vegetation. However, intensive long-term management will be required across vast grazing areas in the northern Australia to limit the spread and manage the grass-fire cycle. This will require additional knowledge about the genetic variation among geographically dispersed populations and their ability to withstand fire and impact establishment of native species. The ability of A. gayanus to transform vast tracts of northern Australia into fire-prone grasslands has led to the species being listed in the second group of WoNS in 2012.
6.3.5 Kochia (Bassia scoparia)
Bassia scoparia was introduced into Australia in 1990 as a forage plant for sodic soils and saline land rehabilitation in southeastern Western Australia. One year later it was sown at 68 sites over an 850 km area and soon naturalised; within 2 years it had dispersed from 60% of infested sites (Dodd and Randall 2002). Prior to initiation of the eradication programme in 1992, 38 of the 52 naturalised sites were < 10 ha in size, with the other 12 ranging in size up to 140 ha, with the total area invaded being 3277 ha in 1993 (Dodd and Randall 2002; Dodd 2004). In spite of the fact that the situation was considered challenging and not ‘ideal’ for achieving eradication (see Rejmánek and Pitcairn 2002), B. scoparia was successfully eradicated in the first nationally funded programme in Australia for a recent or new weed incursion.
Bassica scoparia exhibited several unique characteristics which made it an ideal candidate for successful eradication. Specifically, its distribution was limited and well documented, its seed is short-lived following dispersal (1–3 years with most germinating within 12 months) and although it spreads via wind-blown plants or ‘tumble weeds’, fences established to exclude grazing animals as part of the sowing rehabilitation programme were fortuitously successful in limiting its spread (Dodd and Randall 2002). These attributes supported the successful eradication of B. scoparia and contributed to it being an exceptional case (Panetta and Timmins 2004) as few other examples of plant eradication following establishment over such a significant area have been observed outside of islands.
By 2000, in less than 10 years, B. scoparia was considered to have been eradicated, responding positively to such efforts (herbicide, grazing, burning and mechanical removal) despite the broad scale of infestation, with most sites virtually free from infestation within 2 years (Dodd 2004). While its invasion history was unique, specific management responses aided the successful outcome including rapid response (eradication over a 2-year period) with state and federal funding made available to achieve a positive outcome (Dodd 2004). Both rapid response and multiple control tactics are typically required for the successful eradication of any alien plant (Panetta and Timmins 2004).
6.4 Summary of Case Studies
The case studies presented in this chapter illustrate that adopting a species-led approach in the form of the WoNS programme to manage invasive plant species in Australia has had some proven successes (i.e. the winners), but there have also been numerous failures or ‘losers’ with this approach (i.e. those non-WoNS species). While some of these non-WoNS were listed in the second round (e.g. A. gayanus), it is impractical to manage every invasive plant species in Australia under a species-led approach. However, the Australian WoNS programme has had significant benefits which have flowed on to the management of non-WoNS species. These include well-developed and broad-scale education and awareness campaigns, extensive development of best practice guidelines, the development of approaches to identify those native species at risk due to such threats and the assessment of risk management strategies for prioritisation of management or future investment in control strategies.
It should be noted that significant benefits to invasive plant species management have also occurred outside of the WoNS programme (i.e. the development of the Biocontrol Act; see the E. plantagineum case study). While the control of prickly pear has been a notable success in Australia as previously described, numerous other opuntioid cactus species have become highly invasive in recent decades, leading to this group of plants being listed in the second round of WoNS. In spite of these continuing challenges, the prickly pear management programme has left behind a successful legacy for the implementation of biological control for invasive plant species management in Australia. Research and management programmes for other such non-WoNS Opuntia species have been initiated due to the significant challenges posed by these species and the need to combat such problems.
Management of invasive plants can take several forms, being species-led, site-led and pathways-led (Downey and Sheppard 2006; McGeoch et al. 2016). The species-led approach ‘adopted’ in Australia differs significantly from that formally used in New Zealand in which species-led programmes are focused exclusively on the eradication of a newly established alien plant species (Owen 1998; Downey and Sheppard 2006). In addition to adopting a species-led approach through the WoNS programme, Australia has also adopted a pathways approach in the form of a quarantine and pre-border Weed Risk Assessment (WRA) system (Pheloung et al. 1999) to detect potentially invasive plants species deliberately imported into Australia. Australia initially adopted quarantine measures in 1908 that governed the importation of plant material, but many plant species were still deliberately introduced for crops, pastures and ornamental purposes during the twentieth century (see Hazard 1988; Lonsdale 1994; Cook and Dias 2006), despite widespread advertisement advising of the dangers of plant introductions from the middle of the century (i.e. the 1950s (see https://collections.museumsvictoria.com.au/items/244998)).
In 1965 a comprehensive Quarantine Weeds List was developed, which contained over 130 species, which encompassed potentially dangerous (weedy) species and collated prohibited weeds lists from all Australian states and territories. Within 10 years there were strong arguments to reduce the size of the list by developing a weed seed schedule as it was becoming ‘unworkable’ due to the associated costs to farmers (see Spurrs 1976). In response to the growing concern of the invasive plant problem, the Australian Weed Committee proposed a standard scoring system to be used by the commonwealth, states and territories when assessing the ‘weedy potential’ of a species being imported into Australia, in order to minimise the risk. The work of Hazard (1988) and the Australian Weeds Committee led to the development and adoption of a formal WRA system (Pheloung et al. 1999) to screen deliberate introductions.
Australia held the first international workshop on weed risk assessment in 1999 (see Groves et al. 2001) and a second workshop in 2007 (Downey et al. 2010b). Australia also played a significant role in the development of weed risk assessment approaches globally through both development of a pre-border WRA system (see Pheloung et al. 1999), which has been tested and applied in many countries, and a post-border Weed Risk Management system (see Downey et al. 2010b).
Further pathways-based management approaches have involved detailed assessments of specific ‘importation’ sectors (e.g. those resulting in the sale of ornamental plants or pasture species). For example, Groves et al. (2005) showed that 66% of naturalised and invasive plant species in Australia originated from the gardening sector, and Virtue et al. (2004) revealed that 94% of the 27,000 deliberate plant numbers imported into Australia were associated with the nursery trade. In another assessment of pathways, Lonsdale (1994) showed that 463 exotic grasses and legumes were imported into Australia for pasture improvement purposes of which 13% subsequently became invasive. Many of these species were supported by federally funded research programmes at the time (see Cook and Dias 2006). Such assessment of invasion pathways has been instrumental in the development of a national pre-border WRA system to limit the import of potentially invasive plant species.
Site-led management tends to be poorly defined and is generally not used formally in Australia, in contrast to the approach adopted in New Zealand (Downey and Sheppard 2006) which is based on the protection of specific conservation areas at threat from invasive plant species (Owen, 1998). In fact, site-led management of invasive plant species in Australia has taken on multiple forms, with actions for invasive plant management being included in most Plans of Management for protected areas and a range of sites being identified in state and national plans for WoNS, based on the presence of biodiversity under threat. For example, the Bitou TAP identified approximately 350 sites containing 157 plant species and 24 ecological communities threatened by bitou bush, C. monilifera subsp. rotundata, in NSW (Downey 2010). Such plans that aim to abate the threat of invasive plants to native biodiversity encompass both a species- and site-led approach.
6.5 Future of Plant Invasions in Australia
In the seminal book ‘Australian Weed Management Systems’, Adkins and Walker (2000) outlined three key future challenges for managing plant invasions across Australia. These include (1) the problem of the ‘dynamic and growing’ alien plant flora, which includes the threat from alien plant species already present in Australia that have not yet reached their full potential distribution and abundance, as well as those introduced and not presently recorded as naturalised (i.e. many of the 27,000 species outlined by Virtue et al. (2004)) and those impacted by potential climatic changes (see further discussion below); (2) the need to develop and use environmentally and ecologically sustainable management approaches, driven in part by the increasing use of chemical weed management strategies and the growing issue of herbicide-resistant weed species; and (3) the integration and adoption of weed management approaches and research leading to successful on-the-ground outcomes. Additionally, there is a growing need to ensure that challenges associated with a lack of monitoring following weed management are addressed, as highlighted by Reid et al. (2009) during an evaluation of the 20 WoNS species.
To address the growing alien flora in Australia, particularly arising from the deliberate introduced of ornamental species, a recent initiative called the Plant Sure scheme has been established to enable growers, retail nurseries and purchasers of ornamental plants to grow, sell and buy non-native plants that are unlikely to escape and pose a threat to the environment (see www.gardeningresponsibly.org.au). The Plant Sure scheme has been successfully trialled in NSW, and a second phase is being rolled out including an assessment tool to determine the likely invasiveness of a nominated species, a certification system for non-invasive species and education and awareness material.
A critical future challenge that remains is developing a better understanding of how invasive plants will respond to a rapidly changing Australian climate (Roger et al. 2015). To address this issue, assessments were initially carried out on individual invasive plant species (e.g. Siam weed (Chromolaena odorata)) (Kriticos et al. 2005) and examined the relationship between climate predictions based on native and alien ranges (Beaumont et al. 2009). However, given the number of naturalised plant species in Australia, a single species approach has not generally been practical or useful for managers and policymakers.
In response to this need, Roger et al. (2015) developed a process to assess large numbers of species in a dedicated searchable website for managers (www.weedfutures.net). This website currently has climate change predications for 2035 and 2065 for over 700 non-native naturalised and invasive plant species in Australia. The database behind this website has led to several key publications on the future challenges for alien plants species in Australia in terms of the next generation of invaders (Duursma et al. 2013) and invasion hotspots (O’Donnell et al. 2012) for alien plant species under climate change.
One additional challenge will be to gain an improved understanding of how invasive plants respond to chemical control under elevated levels of atmospheric CO2. For example, Manea et al. (2011) examined the effects of herbicide on four C4 exotic grass species in Australia under ambient and elevated CO2 and found that three of these species showed increase herbicide resistance under an elevated CO2 environment. Given that Adkins and Walker’s (2000) second key challenge was herbicide resistance, which is a growing problem in Australia and elsewhere, any increased resistance due to elevated CO2 could require significant shifts in the management of alien plant species particularly with respect to the use of herbicides.
6.6 Conclusions
The separation of Australia from other land masses has resulted in the evolution of unique native flora and fauna. Colonisation over the past three centuries has resulted in the introduction of various invasive plants to Australia, placing significant selection pressure on their ability to successfully adapt to Australian climate conditions. Incursions of invasive plants have impacted both native vegetation and managed crops, endangering fragile ecosystems and already resource-strained agricultural systems. The management of invasive plants has required significant investment from local, state and federal government agencies, leading to the classification of most impactful invasive plants as Australian Weeds of National Significance (WoNS). Concerted efforts for management have also led to state and national strategies to control and ultimately eradicate some of these species. The coastal WoNS species Chrysanthemoides monilifera subsp. rotundata, for example, has been managed successfully through application of a combination of chemical, physical and biological control strategies. Similarly, the management of most invasive weeds has relied on the use of integrated management strategies, including biological controls, which provide robust control of those species over time. The case studies presented clearly suggest the importance of the study of the biology, physiology and chemistry of weed invaders to target effective control strategies for successful eradication.
Future Australian ecologists, weed scientists, land managers and agriculturalists will need to work together to address the continuing challenge of invasive plants under a changing climate. While Australia has had considerable success in managing the invasion of some key alien plant species of national importance (i.e. WoNS) and has also successfully eradicated several recently introduced species by focused management, the success of invasive alien species management will clearly affect the current status of Australian biodiversity and our ability to preserve fragile native communities, maintain agricultural productivity and protect human health and well-being. Future investments in large-scale management of invasive plants will rely upon multidisciplinary interaction of scientists and landowners and regional, state and national coordination to deliver effective outcomes. We remain hopeful that federal investment in such coordinated efforts will be reinvigorated in coming years, as past programmes have proven highly effective in some cases and also introduced novel strategies for suppression and eradication of invasive plant species down under.
References
Adkins SW, Walker SR (2000) Challenges and future approaches to weed management. In: Sindel BM (ed) Australian weed management systems. R.G. & F.J. Richardson, Melbourne, pp 481–496
ARMCANZ (Agriculture and Resource Management Council of Australia and New Zealand), ANZECC (Australian and New Zealand Environmental and Conservation Council) & Forestry Ministers (1997) The national weeds strategy: a strategic approach to weed problems of national significance. Commonwealth of Australia, Canberra
Barker HM (1964) Camels and the outback. Sir Isaac Pitman & Sons Ltd, Melbourne
Beadle NCW (1981) Vegetation of Australia. Gustav Fisher Verlag, Stuttgart. Cambridge University Press, Cambridge, pp 102–106
Bean AR (2007) A new system for determining which plant species are indigenous in Australia. Syst Bot 20:1–43
Beaumont LJ, Gallagher RV, Thuiller W et al (2009) Different climatic envelopes among invasive populations may lead to underestimations of current and future biological invasions. Divers Distrib 15:409–420
Beaumont LJ, Gallagher RV, Leishman MR et al (2014) How can knowledge of the climate niche inform the weed risk assessment process? A case study of Chrysanthemoides monilifera in Australia. Divers Dist Distrib 20:613–625
Bebawi FF, Campbell SD, Mayer RJ (2018) Gamba grass (Andropogon gayanus Kunth.) seed persistence and germination temperature tolerance. Rangel J 40:463–472
Begum S, Zehra SQ, Siddiqui BS et al (2008) Pentacyclic triterpenoids from the aerial parts of Lantana camara and their nematicidal activity. Chem Biodivers 5:1856–1866
Bennett MD, Leitch IJ, Hanson L (1998) DNA amounts in two samples of angiosperm weeds. Ann Bot 82:121–134
Bhagwat SA, Breman E, Thekaekara T et al (2012) A battle lost? Report on two centuries of invasion and management of Lantana camara L. in Australia, India and South Africa. PLoS One 7:e32407
Borger C, Hashem A, Amjad M et al (2018) Summer weeds within the Western Australian Wheatbelt: a three year GRDC survey. Grains Research and Development Corporation, Canberra
Bowdler S (2002) Hunters and traders in northern Australia. In: Morrison KD, Junker LL (eds) Forager-traders in south and Southeast Asia. Cambridge University Press, Cambridge, pp 167–184
Bowman DMJS, MacDermott HJ, Nichols SC et al (2014) A grass–fire cycle eliminates an obligate-seeding tree in a tropical savanna. Ecol Evol 4:4185–4194
BQ (Biosecurity Queensland on behalf of the National Lantana Management Group) (2010) Plan to protect environmental assets from lantana. Department of Employment, Economic Development and Innovation, Yeerongpilly
Brodie CJ, Reynolds TM (2012) Review of recent plant naturalisations in South Australia and initial screening for weed risk. DENR Technical Report 2012/02, South Australian Department of Environment and Natural Resources, Adelaide
Brown GK, Bostock PD (2019) Introduction to the census of the Queensland
Burbidge NT, Gray M (1972) Flora of the ACT. ANU Press, Canberra
Carr GW (1993) Exotic flora of Victoria and its impact on indigenous biota. In: Foreman DB, Walsh NG (eds) Flora of Victoria: volume 1 – introduction. Melbourne, Inkata Press, pp 256–297
Chapman AD (2009) Numbers of living species in Australia and the world, 2nd edn. A Report for the Australian Biological Resources Study, Toowoomba
Chapman AR (2020) Western Australian flora statistics – 2020 Flora statistics. https://florabase.dpaw.wa.gov.au/statistics/
Chippendale GM (1971) Check list of northern territory plant. Proc Linn Soc NSW 96(4):207–267
Cook GD, Dias L (2006) It was no accident: deliberate plant introductions by Australian government agencies during the 20th century. Aust J Bot 54:601–625
Cullen JM, Delfosse ES (1984) Echium plantagineum: catalyst for conflict and change in Australia. In: Proceedings of the VI international symposium on biological control of weeds. Agriculture Canada, Ottawa, pp 19–25
D’Antonio CM, Vitousek PM (1992) Biological invasions by exotic grasses, the grass/fire cycle, and global change. Annu Rev Ecol Syst 23:63–87
Day MD, Wiley CJ, Playford J et al (2003) Lantana: current management status and future prospects, Monograph 102. Australian Centre for International Agricultural Research, Canberra
de Salas MF, Baker ML (2018) A census of the vascular plants of Tasmania, including Macquarie Island. Tasmanian Herbarium, Tasmanian Museum and Art Gallery, Hobart
Dodd AJ (1940) The biological campaign against prickly-pear. The Commonwealth Prickly Pear Board. Government Printer, Brisbane
Dodd J (2004) Kochia (Bassia scoparia (L.) A.J.Scott) eradication in Western Australia: a review. In: Sindel BM, Johnson SB (eds) Proceedings of the 14th Australian Weeds Conference, 6–9 September 2004, Wagga Wagga. Weeds Society of New South Wales, Sydney, pp 496–499
Dodd J, Randall RP (2002) Eradication of kochia (Bassia scoparia (L.) AJ Scott, Chenopodiaceae) in Western Australia. In: Spafford Jacob H, Dodd J, Moore JH (eds) Proceedings of the 13th Australian weeds conference, 8–13 September 2002, Perth. Plant Protection Society of Western Australia, Perth, pp 300–303
Dodd AJ, Burgman MA, McCarthy M et al (2015) The changing patterns of plant naturalization in Australia. Divers Distrib 21:1038–1050
Downey PO (2010) Managing widespread alien plant species to ensure biodiversity conservation: a case study using an 11-step planning process. Invasive Plant Sci Manag 3:451–461
Downey PO, Sheppard AW (2006) Site- versus species-based approaches to weed management in Australia. In: Preston C, Watts JH, Crossman ND (eds) The Proceedings of the 15th Australian Weeds Conference, Adelaide, 24–28 September 2006. Weed Management Society of South Australia, Adelaide, pp 264–267
Downey PO, Scanlon TJ, Hosking JR (2010) Prioritising alien plant species based on their ability to impact on biodiversity: a case study from New South Wales. Plant Prot Q 25:111–126
Downey PO, Williams MC, Whiffen LK et al (2010a) Managing alien plants for biodiversity outcomes—the need for triage. Invasive Plant Sci Manag 3:1–11
Downey PO, Johnson SB, Virtue JG et al (2010b) Assessing risk across the spectrum of weed management. CAB Rev Persp Agric Veterinary Sci Nutr Nat Resour 5:038
Durán AG, Gutiérrez MT, Rial C et al (2017) Bioactivity and quantitative analysis of isohexenylnaphthazarins in root periderm of two Echium spp.: E. plantagineum and E. gaditanum. Phytochemistry 141:162–170
Duursma DE, Gallagher RV, Roger E et al (2013) Next-generation invaders? Hotspots for naturalised sleeper weeds in Australia under future climates. PLoS One 8:e84222
Everist SL (1960) Strangers within the gates (Plants naturalised in Queensland). Queensland Nat 16:49–60
Ewart AJ (1931) Flora of Victoria. Government Printer, Melbourne
EWWG (Environmental Weeds Working Group) (2007) Guidelines and procedures for managing the environmental impacts of weeds on public land in Victoria 2007. Department of Sustainability and Environment, Melbourne
Field RP, Lane DWA, Bruzzese E et al (1986) Biological control of Paterson’s curse. Submission to the industries assistance commission hearing. Unpublished report. Department of Conservation Forests and Lands and Department of Agriculture and Rural Affairs, Victoria
Forbes SJ, Ross JH (1988) A census of the vascular plants of Victoria. National Herbarium of Victoria, Melbourne
Gentle CB, Duggin JA (1997) Allelopathy as a competitive strategy in persistent thickets of Lantana camara L. in three Australian forest communities. Plant Ecol 132:85–95
Gooden B, French K, Turner PJ et al (2009) Impact threshold for an alien plant invader, Lantana camara L., on native plant communities. Biol Conserv 142:2631–2641
Goyal N, Sharma GP (2015) Lantana camara L. (sensu lato): an enigmatic complex. NeoBiota 25:15–26
Green JW (1985) Census of the vascular plants of Western Australia. Western Australian Herbarium
Grigulis K, Sheppard AW, Ash JE et al (2001) The comparative demography of the pasture weed Echium plantagineum between its native and invaded ranges. J Appl Ecol 38:281–290
Groves RH (1986) Plant invasions of Australia: an overview. In: Groves RH, Burdon JJ (eds) Ecology of biological invasions: an Australian perspective. Australian Academy of Science, Canberra, pp 137–149
Groves RH (2002) Robert Brown and the naturalised flora of Australia. Cunninghamia 7:623–629
Groves RH, Hosking JR (1998) Recent incursions of weeds to Australia 1971–1995, Technical Series 3. CRC for Weed Management Systems, Adelaide
Groves RH, Panetta FD, Virtue JG (eds) (2001) Weed risk assessment. CSIRO Publishing, Melbourne
Groves RH, Hosking JR, Batianoff GN et al (2003) Weed categories for natural and agricultural ecosystem management. Bureau of Rural Sciences, Canberra
Groves RH, Boden R, Lonsdale WM (2005) Jumping the garden fence: invasive garden plants in Australia and their environmental and agricultural impacts. WWF-Australia, Sydney
Hallett LM, Standish RJ, Jonson J et al (2014) Seedling emergence and summer survival after direct seeding for woodland restoration on old fields in South-Western Australia. Ecol Manag Restor 15:140–146
Hao ZP, Wang Q, Christie P et al (2007) Allelopathic potential of watermelon tissues and root exudates. Sci Hortic 112:315–320
Harrison HF, Wechter WP, Kousik CS (2012) Inhibition of bacterial, fungal, and plant growth by testae extracts of Citrullus genotypes. HortScience 47:448–451
Hazard WHL (1988) Introducing crop, pasture and ornamental species into Australia—the risk of introducing new weeds. Aust Plant Intr Rev 19:19–36
Hnatiuk RJ (1990) Census of Australian vascular plants. Australian Government Publishing Service, Canberra
Hooker JD (1859) On the flora of Australia: its origins, affinities and distribution. Lovell Reeve, London
Humphries SE, Groves RH, Mitchell DS (1991) Plant invasions of Australian ecosystems: a status review and management directions. Part 1: In: Walton DW, Reville B, George A et al Plant invasions: the incidence of environmental weeds in Australia. Kowari no. 2. pp. 1–127, Australian Parks and Wildlife Service, Canberra
Hussain N, Abbasi T, Abbasi SA (2016) Transformation of toxic and allelopathic lantana into a benign organic fertilizer through vermicomposting. Spectrochim Acta A Mol Biomol Spectrosc 163:162–169
Johnson RW (1983) The Queensland flora and its collection. Proc Royal Soc Queensland 94:1–18
Keighery G (1999) A checklist of the naturalised vascular plants of Western Australia (including: distribution in IBRA regions and garden escapes). Department of Conservation and Land Management, Perth
Kerrigan RA, Albrecht DE (2007) Checklist of NT Vascular Plant Species www.territorystories.nt.gov.au/jspui/bitstream/10070/213499/1/NT_Checklist_Jan_07.pdf
Kloot PM (1984) The introduced elements of the flora of southern Australia. J Biogeogr 11:63–78
Kloot PM (1986) A review of the naturalised alien Flora of the central areas of South Australia. Technical Paper no. 2, South Australian Department of Agriculture, Adelaide
Kloot PM (1987) The naturalised flora of South Australia. 3. Its origin, introduction, distribution, growth forms and significance. J Adel Bot Gard 10:99–111
Kriticos DJ, Yonow T, McFadyen RE (2005) The potential distribution of Chromolaena odorata (Siam weed) in relation to climate. Weed Res 45:246–254
Latif S, Chiapusio G, Weston LA (2017) Allelopathy and the role of allelochemicals in plant defence. Advan Bot Res 82:19–54
Lazarides M, Cowley K, Hohnen P (1997) CSIRO handbook of Australian weeds. CSIRO Publishing, Melbourne
Lepschi BJ, Cargill DC, Albrecht DE et al (2019) Census of the Flora of the Australian Capital Territory. Version 4.1. Australian National Herbarium Canberra
Llewellyn R, Ronning D, Clarke M et al (2016) Impact of weeds in Australian grain production. Grains Research and Development Corporation, Canberra
Lonsdale WM (1994) Inviting trouble: introduced pasture species in northern Australia. Austral Ecol 19:345–354
Macknight CC (1976) The voyage to Marege’: Macassan trepangers in northern Australia. Melbourne University Press, Melbourne
Mafeo TP, Mashela PW (2010) Allelopathic inhibition of seedling emergence in dicotyledonous crops by Cucumis bio-nematicide. Afr J Biotechnol 9:8349–8354
Manea A, Leishman MR, Downey PO (2011) Exotic C4 grasses have increased tolerance to glyphosate under elevated carbon dioxide. Weed Sci 59:28–36
McGeoch MA, Genovesi P, Bellingham PJ et al (2016) Prioritizing species, pathways, and sites to achieve conservation targets for biological invasion. Biol Invasions 18:299–314
Molyneux RJ, Gardner DL, Colegate SM et al (2011) Pyrrolizidine alkaloid toxicity in livestock: a paradigm for human poisoning? Food Addit Contam Part A 28:293–307
O’Donnell J, Gallagher RV, Wilson PD et al (2012) Invasion hotspots for non-native plants in Australia under current and future climates. Glob Change Biol 18:617–629
Owen SJ (1998) Invasive weed threats: weed-led and site-led programmes identified by the Department of Conservation. Department of Conservation, Wellington, p 87
Palmer WA, Heard TA, Sheppard AW (2010) A review of Australian classical biological control of weeds programs and research activities over the past 12 years. Biol Control 52:271–287
Panetta FD, Timmins SM (2004) Evaluating the feasibility of eradication for terrestrial weed incursions. Plant Prot Q 19:5–11
Parr CL, Ryan BJ, Setterfield SA (2010) Habitat complexity and invasive species: the impacts of Gamba grass (Andropogon gayanus) on invertebrates in an Australian tropical savanna. Biotropica 42:688–696
Petanidou T, Godfree RC, Song DS et al (2012) Self-compatibility and plant invasiveness: comparing species in native and invasive ranges. Perspect Plant Ecol Evol Syst 14:3–12
Peters N (2006) The Dutch down under: 1606–2006. University of Western Australia Press, Perth
Pheloung PC, Williams PA, Halloy SR (1999) A weed risk assessment model for use as a biosecurity tool evaluating plant introductions. J Environ Manag 57:239–251
Piggin CM, Sheppard AW (1995) Echium plantagineum L. In: Groves RH, Shepherd RCH, Richardson RG (eds) The biology of Australian weeds, vol 1. R.G and F.J. Richardson, Melbourne, pp 87–110
Pimentel D (ed) (2011) Biological invasions: economic and environmental costs of alien plant, animal and microbe species, 2nd edn. CRC Press, London
Ramirez AHM, Jhala AJ, Singh M (2014) Factors affecting germination of citronmelon (Citrullus lanatus var. citroides). Weed Sci 62:45–50
Randall RP (2007) The introduced flora of Australia and its weed status. CRC for Australian Weed Management, Adelaide
Randall RP (2017) A global compendium of weeds, 3rd edn, Perth
Reid AM, Morin L, Downey PO et al (2009) Does invasive plant management aid the restoration of natural ecosystems? Biol Conserv 142:2342–2349
Rejmánek M, Pitcairn MJ (2002) When is eradication of exotic pest plants a realistic goal? In: Veitch CR and Clout MN (eds) Turning the tide: the eradication of invasive species – proceedings of the international conference on eradication of island invasive species. pp. 249–253, IUCN Species Survival Commission Occasional Paper no. 27, IUCN – The World Conservation Union, Gland
Roger E, Englert Duursma D, Downey PO et al (2015) A tool to assess potential for alien plant establishment and expansion under climate change. J Environ Manag 159:121–127
Ross JH (1976) An analysis of the flora of Victoria. Muelleria 3:169–176
Rossiter N, Setterfield S, Douglas M et al (2002) Exotic grass invasion in the tropical savanna of northern Australia: ecosystem consequences. In: Bishop AC, Boersma M, Barnes CD (eds) Proceedings of the 12th Australian weeds conference, 12–16 September, Hobart. Tasmanian Weed Society, Devonport, pp 168–171
Rossiter NA, Setterfield SA, Douglas MM (2003) Testing the grass-fire cycle: alien grass invasion in the tropical savannas of northern Australia. Divers Distrib 9:169–176
Rossiter-Rachor NA, Setterfield SA, Douglas MM et al (2009) Invasive Andropogon gayanus (gamba grass) is an ecosystem transformer of nitrogen relations in Australian savanna. Ecol Appl 19:1546–1560
Rozefelds AC, MacKenzie R (2005) A reappraisal of the early history of the weed invasion in Tasmania up to the 1870s. Kanunnah 1:61–90
Rozefelds AC, Cave L, Morris DI et al (1999) The weed invasion in Tasmania since 1970. Aust J Bot 47:23–48
Sahid IB, Sugau JB (1993) Allelopathic effect of lantana (Lantana camara) and Siam weed (Chromolaena odorata) on selected crops. Weed Sci 41:303–308
Saxena MK (2000) Aqueous leachate of Lantana camara kills water hyacinth. J Chem Ecol 26:2435–2447
Shaik RS, Gopurenko D, Urwin NA et al (2015) Population genetics of invasive Citrullus lanatus, Citrullus colocynthis and Cucumis myriocarpus (Cucurbitaceae) in Australia: inferences based on chloroplast and nuclear gene sequencing. Biol Invasions 17:2475–2490
Shaik RS, Lepschi BJ, Gopurenko D et al (2016a) An integrative morphological and molecular approach to identification of three Australian cucurbitaceous invasive weeds: Citrullus colocynthis, C. lanatus and Cucumis myriocarpus. Aust Syst Bot 29:247–264
Shaik RS, Zhu X, Clements DR et al (2016b) Understanding invasion history and predicting invasive niches using genetic sequencing technology in Australia: case studies from Cucurbitaceae and Boraginaceae. Conserv Physiol 4:30
Shaik RS, Burrows GE, Urwin NA et al (2017) The biology, phenology and management of Australian weed-camel melon (Citrullus lanatus (Thunb.) Matsum. and Nakai). J Crop Prot 98:222–235
Sharma OP, Makkar HPS, Dawra RK et al (1981) A review of the toxicity of Lantana camara (Linn) in animals. Clin Toxicol 18:1077–1094
Singh P, Srivastava D (2012) Biofungicidal or biocontrol activity of Lantana camara against phytopathogenic Alternaria alternata. Int J Pharm Sci Res 3:4818–4821
Skoneczny D, Weston P, Zhu X et al (2017) Metabolic profiling and identification of shikonins in root periderm of two invasive Echium spp. weeds in Australia. Molecules:22:330
Skoneczny D, Zhu X, Weston PA et al (2019) Production of pyrrolizidine alkaloids and shikonins in Echium plantagineum L. in response to various plant stressors. Pest Manag Sci 75:2530–2541
Smith JMB (1986) Biogeography of Australian flora and fauna. In: Recher HF, Lunney D, Dunn I (eds) A natural legacy, 2nd edn. Pergamon Press, Sydney, pp 14–30
Smith N (2002) Not from here: plant invasions on aboriginal lands of the top end. Tropical Savannas CRC, Northern Land Council, Centre for Indigenous Natural and Cultural Resource Management, Northern Territory University, Darwin
Souster JE (1947) Some observations on our Alien Flora. The West Aust Nat 1(3):62–64
Specht RL, Mountford CP (1958) The American-Australian scientific expedition to Arnhem Land. 3. In: Botany and plant ecology. Melbourne University Press, Melbourne
Spurrs AB (1976) Proposals to reduce the length of the quarantine prohibited weeds list and to establish a prohibited seed schedule for seed importations. In: Proceedings of the 5th Australian weeds conference, 29 March – 2 April 1976, Melbourne. CSIRO, Melbourne, pp 184–185
Thorp JR, Lynch R (2000) Determination of weeds of national significance. National Weeds Strategy Executive Committee, Launceston
Turner PJ, Downey PO (2010) Ensuring invasive alien plant management delivers biodiversity conservation: insights from a new approach using Lantana camara. Plant Prot Q 25:102–110
Udo IA, Osai EO, Ukeh DA (2014) Management of root-knot disease on tomato with bioformulated Paecilomyces lilacinus and leaf extract of Lantana camara. Braz Arch Biol Tech 57:486–492
Virtue JG, Bennet SJ, Randall RP (2004) Plant introductions in Australia: how can we resolve ‘weedy’ conflicts of interest? In: Sindel BM and Johnson SB (eds) Proceedings of the 14th Australian Weeds Conference, 6–9 September 2004, Wagga Wagga. pp. 42–48, Weeds Society of New South Wales, Sydney
Vivian-Smith G, Lawson BE, Turnbull I et al (2007) The biology of Australian weeds. 46. Anredera cordifolia (Ten.) Steenis. Plant Prot Q 22:2–10
Weston PA, Weston LA, Hildebrand S (2012) Environmental impact on biocontrol agents and secondary chemistry of Paterson’s curse (Echium plantagineum). In Proceedings of the 18th Australasian Weeds Conference, pp. 203–207
Weston PA, Weston LA, Hildebrand S (2013) Metabolic profiling in Echium plantagineum: presence of bioactive pyrrolizidine alkaloids and napthoquinones from accessions across southeastern Australia. Phytochem Rev 12:831–837
White M, Cheal D, Carr GW et al (2018) Advisory list of environmental weeds in Victoria. Arthur Rylah Institute for Environmental Research, Technical Report Series No. 287, Department of Environment, Land, Water and Planning, Melbourne
Woolls W (1867) Notes on introduced plants occurring in the neighbourhood of Sydney. J Proc Linn Soc Bot 10:35–42
Zhang S, Wang GD, Ma P et al (2020) Genomic regions under selection in the feralization of the dingoes. Nat Commun 11:1–10
Zhu X, Weston PA, Skoneczny D et al (2017) Ecology and genetics affect relative invasion success of two Echium species in southern Australia. Sci Rep 7:42792
Zhu X, Gopurenko D, Serrano M et al (2019) Genetic evidence for plural introduction pathways of the invasive weed Paterson’s curse (Echium plantagineum L.) to southern Australia. PLoS One 14(9):e0222696
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Shaik, R.S., Gurusinghe, S., Weston, L.A., Downey, P.O. (2022). A Historical Perspective on Plant Invasion in Australia. In: Clements, D.R., Upadhyaya, M.K., Joshi, S., Shrestha, A. (eds) Global Plant Invasions. Springer, Cham. https://doi.org/10.1007/978-3-030-89684-3_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-89684-3_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-89683-6
Online ISBN: 978-3-030-89684-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)