Abstract
Kruger National Park (KNP), South Africa, covers an area of 20,000 km2 and is one of the world’s most widely known protected areas. Like many protected areas, KNP is facing an increasing problem with the introduction and spread of alien plants. However, species prioritization using poor baseline data remains a key challenge for managers globally. Publications, expert opinion, and field observations indicate that 407 alien plant taxa have been recorded in the KNP; this list also included hybrids, ornamentals, records that could only be identified to the genus level, and extralimitals (i.e., species native elsewhere in South Africa; 22 species in total); in addition, two species are considered eradicated, and three potentially eradicated. Such extensive lists of poorly defined species’ statuses accumulated over long periods of time poses challenges to current decision-making processes. This is especially important for the management of naturalized (maintaining self-sustaining populations) and invasive species (subset of naturalized species that have spreading populations), because management needs to identify and target high-priority species and vulnerable sites. Here, we provide an up-to-date inventory of alien plant species that occur in natural areas in KNP (i.e., beyond tourist camps and other infrastructure) and thus represent a potential threat to the native species diversity in the park. We identified 146 such alien taxa, of which 30 are casuals, 58 are naturalized, 21 have become invasive, and for 37 species, the status remains to be determined. Twelve of the invasive species in KNP are globally widespread, occurring in more than 100 regions, and five (i.e., Pontederia crassipes, Lantana camara, Opuntia stricta, Chromolaena odorata and Mimosa pigra) are listed among 100 of the world’s worst invasive alien species. The alien flora in KNP comprises 41 families. Solanaceae (45.5%) and Asteraceae (26.1%) are over-represented among invasive species compared to non-invasive species. The alien flora of KNP mostly originates from North America and South America, and largely consists of perennials and herbaceous species. We found no significant results regarding the effect of origin and life span on invasion status. Despite a steady increase in the numbers of alien and naturalized plants since the 1980s, species we classified as invasive generally represent earlier introductions and have not increased substantially since the beginning of this century. Our paper highlights that carefully revised naturalized and invasive species lists reflecting the current situation in protected areas present a strong knowledge base for effective management strategies. In addition to addressing data gaps related to the distribution of alien species on a global scale, knowledge from large protected areas such as KNP contributes to understanding invasions in landscapes with varying and unique habitat types.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Protected areas (PAs), demarcated as important for biodiversity conservation and provision of ecosystem services and resources for human well-being, require carefully designed management programs to maintain or restore natural ecosystems (Watson et al. 2014). Due to the risks and impacts that invasive alien species pose to these areas, management is a high priority (Moodley et al. 2020; Pyšek et al. 2020b). To facilitate proactive management and prioritize resources toward species that are most likely to become problematic, reliable information on the status of the alien flora in an area is required. Of the total number of alien species introduced into an area, only a small proportion become naturalized, and of these, only a small proportion become invasive and cause ecological, economic, or social impacts (Williamson and Fitter 1996; Richardson and Pyšek 2006; Pyšek et al. 2012, 2020b; Essl et al. 2019). When faced with many species, the challenge that arises during decision-making is to identify which subset of species have the potential to naturalize or have already become naturalized. Risk assessment and priority-setting tools are available and have been used in a wide range of situations (Blackburn et al. 2011; Leung et al. 2012; Nentwig et al. 2016; Vanderhoeven et al. 2017; Roy et al. 2018; Foxcroft et al. 2019; Kumschick et al. 2020, 2022). Applying these tools to species deemed as representing a high risk due to their potential for invasion is necessary for effective decision-making (e.g., early detection, rapid response, long-term monitoring, and informing legislation). Therefore, by identifying and isolating those species that have a higher potential for invasion, attention can be focused on the most important problems.
Kruger National Park (KNP), South Africa, has listed numerous alien species; by 2017, ~ 360 plants and 30 animals were documented, although the proportion of naturalized or invasive species had not been comprehensively assessed (Foxcroft et al. 2017; with several corrections increasing the list since). While protected as a conservation area for a century, various pathways of introduction have allowed species to penetrate the park. Previous work has shown the importance of ornamental horticulture as a primary pathway of introduction into the park for landscaping in tourist camps and staff gardens (Foxcroft et al. 2008, 2019; Keet et al. 2022). Rivers are also a major source of invasion (Foxcroft et al. 2019; Pyšek et al. 2020a) since all the major rivers originate outside KNP and flow through a variety of different land use types that are invaded to various extents. Despite the influx of species into the KNP, most are unlikely to become highly invasive. A risk analysis of riparian invasive alien plants (Foxcroft et al. 2007) reported 153 species in the upper water catchment areas, with 88 species occurring inside the park. The high human population density in areas surrounding PAs is also an important predictor of alien species richness in a park (Spear et al. 2013), as well as roads (outside and inside the park; Foxcroft et al. 2011, 2019; Jarošík et al. 2011), and the movement of contaminated equipment, materials or soils (Foxcroft et al. 2019).
Identifying target species for management is highly important and has substantial implications. An assessment of the management costs of invasive alien species between 1997 and 2016 in KNP indicated that ZAR 350 million (~ US$ 27 million, 2017 values) was spent on control programs (van Wilgen et al. 2017). However, a large proportion of this funding, approximately 40%, was spent on species that were subsequently considered of lower priority. In a global context, the economic costs of invasive alien species in PAs amounted to at least US$ 930.6 million between 1975 and 2020, the majority of which comprise management costs (Moodley et al. 2022). However, it is probable that most of the management costs incurred did not include the high-priority species—for example, management costs for the 100 of the world’s worst invasive species reflect relatively low expenditures between 1960 and 2020, while a greater concern is the fact that costs were only reported for 60% of these species (Cuthbert et al. 2022). By drawing on published alien species lists in PAs, important insights can be gained on which species to prioritize. These lists can serve as a knowledge base for effective management strategies and support the appropriate allocation of limited resources (Pyšek et al. 2013a, 2020b; Hulme et al. 2014). Such lists can, however, serve such a purpose only if they are regularly updated and contain reliable information based on the current state of knowledge, which in many cases can only be gained by ongoing refinements through expert opinion and improved field experience for a given area.
Here, we present an assessment of the alien flora in the Kruger National Park, South Africa. From the alien species inventories collated over time in this area we selected species that are known to occur in the wild in the park and are, or have the potential to become, naturalized or invasive in the park’s landscape and in particular, riparian habitats. Besides being important for informing management actions, such data also provide information on alien species’ ability to naturalize in a unique environmental setting with a minimum anthropogenic disturbance outside the very small human footprint in the KNP (tourist camps/staff villages, roads, and associated infrastructure). Therefore, the aim of this paper is to (i) provide a comprehensive account of the current alien plant species present in KNP, and (ii) review the status of naturalized and invasive species.
Methods
Study area
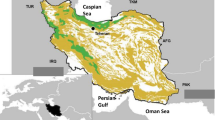
Kruger National Park is in the north-eastern region of South Africa, bordering Mozambique and Zimbabwe (Fig. 1), and covers an area of about 20,000 km2, which is equivalent in size to Slovenia or Israel. It extends about 360 km north–south and averages 60 km west–east (du Toit et al. 2003). The KNP falls within a semi-arid savanna (du Toit et al. 2003), with the Tropic of Capricorn crossing through the northern half of the park. Six major rivers flow through the park (excluding the Limpopo River forming the KNP’s northern border with Zimbabwe), from the western interior of South Africa, through the KNP and into Mozambique. These rivers have a combined drainage area of 90,923 km2.
The area of KNP is environmentally heterogeneous, comprised of a mosaic of geological units (granitoid bedrock in the western vs. basalt in the eastern part, with gabbro intrusions), altitude (140–780 m a.s.l.), climate (450–750 mm of annual precipitation) and vegetation (mixture of woody and grassland savanna; du Toit et al. 2003; MacFadyen et al. 2016). There are 19 vegetation types in KNP, dominated by various types of savanna classified as mopaneveld, lowveld, bushveld and shrubland (Mucina and Rutherford 2006). These factors are reflected in the floristic composition of savanna communities at a finer scale, from the major rivers, through seasonal rivers and crest habitats (Hejda et al. 2022).
Data collation
The first alien flora in KNP was published in 1937 (Obermeijer 1937) and updated at intervals (Codd 1951; Van der Schijff 1957, 1969; Gertenbach 1985; Macdonald and Gertenbach 1988; Foxcroft et al. 2003). The most recent updates were within inventories of alien species across the South African National Parks (SANParks) estate by Spear et al. (2011) and Foxcroft et al. (2017). We excluded the list published by Keet et al. (2022) as their study focused on ornamental and cultivated alien plant species surrounding tourist facilities and staff accommodation (i.e., it does not align with the scope of this study). A revised version of the SANParks catalogue of alien species from 2017 was deposited in the Global Register of Introduced and Invasive Species (GRISS) (Foxcroft et al. 2020). Pyšek et al. (2020a) later reported several additional species not yet recorded in KNP. Species observed and verified by KNP staff since these inventories were published have also been added. Some updates to the previous lists were due to taxonomic changes, as well as species being added due to new information indicating that records were incorrectly aggregated under the same name. For example, two separate entries were created for Azolla filiculoides. The initial entry in KNP still bears the name A. filiculoides, while subsequent entries are labelled as A. cristata. In addition, Opuntia cespitosa, which was previously published in SANParks as O. humifusa (Foxcroft et al. 2017), was originally misidentified as O. stricta. Supplementary Material 1 contains revisions and updates to the previous checklist.
We used the above-mentioned articles and databases as a basis for compiling our updated alien plant species list. To do so, we filtered the full list of 407 taxa by selecting those that could be reliably classified as alien to South Africa (Fig. 2). We excluded ‘extralimital’ species (i.e., those that are native to South Africa but alien in KNP: 22 in total), records only identified to the genus level, as well as hybrids and varieties (i.e., mostly ornamental species with none being listed as potentially naturalized), cultivated fruit trees and ornamental species (i.e., species which have not yet escaped from cultivation and require ongoing nurturing to survive). Species names and authorities were verified using World Flora Online (WFO, www.worldfloraonline.org) and Plants of the World Online (www.powo.science.kew.org) for the currently accepted version.
Procedure for determining the naturalized and invasive flora of the Kruger National Park (KNP). (i) All information sources were collated to generate an overall list, (ii) data were corrected for species name changes and thereafter classified according to predefined criteria, (iii) the native range was assigned as well as pathways into KNP, and (iv) extralimital and cultivated species were excluded from developing the list of 146 species occurring in natural areas
In this study, we focus on alien species that occur in natural areas, i.e., species found outside cultivation, tourist facilities, and staff villages. This was because we aimed to provide information on the current state of naturalized and invasive plants in KNP. We evaluated each alien species’ status in KNP, using the following categories: casual (occurring in the wild outside cultivation but incapable of surviving in the long-term and maintaining self-sustaining populations), naturalized (forming self-sustaining populations in the wild and representing a permanent component of the flora) and invasive (subset of naturalized species that establish populations at significant distances from the point of introduction; Richardson et al. 2000). The species were categorized using the published lists and aforementioned publications on alien species in KNP, expert opinion via personal communications with KNP staff, external scientists, and field observations (mainly by LCF and GRN). Combining all these sources ensured a more objective classification of the status of most alien species. Yet, for some species, there was insufficient information to assign a category; such species were marked as ‘status unknown’, and we suggest that as new information arises, these species be revisited.
For each species, we recorded the family and region of origin using the classification of Brummit (2001). Where known, we included the date of the first introduction/observation, and where unknown, we included the date of publication in which the taxon was first reported (Supplementary Table 1). In addition, we classified species according to life form (i.e., aquatic, climber, geophyte, grass, herb, shrub, succulent, tree), life span (i.e., perennial or annual, biennial). Using the above data, we aimed to assess whether any patterns may characterize the presence of naturalized or invasive species. The main/first pathway of introduction (i.e., cultivated as ornamental, not cultivated, cultivated for fruit) was also included to provide information that can be used in developing preventative measures.
Data analyses
To explore plant trait-based differences among alien species in KNP, we analysed the species’ status as invasive or non-invasive (with non-invasive species including casual, naturalized, and those with unknown status). To test if the continent of origin, life form, and life span were differently represented among the categories of species status (invasive vs. non-invasive), we used univariate Pearson’s chi-square tests. All analyses were performed in the R software package (version 3.6.2; R Core Team 2017).
Results
Numbers of alien taxa in KNP by invasion status categories
The alien flora of KNP that occurs in natural areas (i.e., in the wild) currently comprises 146 species that are alien to South Africa. Of these, 30 are considered casual, 58 naturalized and 21 invasive in KNP. For the remaining 37 species, the status remains unknown as currently there is insufficient information to enable accurate classification (Fig. 3, Supplementary Table 1).
Categories of alien species recognized in our study. Only alien taxa occurring in the Kruger National Park (KNP) outside camps (i.e., highlighted in green) were considered for analysis using predefined criteria to filter the taxa. We excluded extralimital species that are not native to KNP but occur as native in other parts of South Africa, and species only occurring in cultivation inside camps, or within camp boundaries but not known to spread beyond camp limits. See text for exact definitions
The list of invasive species is provided in Table 1 and the complete list of alien flora is provided in Supplementary Table 1. Some of the invasive species in KNP are globally widespread, with 12 of them reported as naturalized from more than 100 regions (out of 843 as defined by van Kleunen et al. 2015; Pyšek et al. 2017); Ricinus communis (recorded in 44.2% of GloNAF regions), Datura stramonium (32.3%) and Melia azedarach (24.2%) are the three most widely distributed species globally. Twelve of the 19 invasive species recorded in KNP are included in the Global Invasive Species Database (Invasive Species Specialist Group 2021), while Pontederia crassipes, Lantana camara, Opuntia stricta, Chromolaena odorata and Mimosa pigra are listed among 100 of the world’s worst alien invasive species (Invasive Species Specialist Group 2013).
Determining which species have been eradicated across a PA covering 20,000 km2 is difficult. For example, plants removed from the southern part of KNP have later been recorded elsewhere, such as in the north of the park, which is approximately 350 km apart. Only two species are now considered highly likely eradicated (Acacia dealbata and Schinus terebinthifolia) and three species potentially eradicated. This contrasts with the estimate by Macdonald and Gertenbach (1988), who suggested that 10 species were eradicated, including the potential eradication of Salvinia molesta. Of these, seven are currently casual, three are naturalized, and one is invasive. The reinvasion of S. terebinthifolia from outside the KNP is possible due to its presence in the town of Phalaborwa along the KNP western border. Cylindropuntia leptocaulis was listed as eradicated in Foxcroft et al. (2017) following control of the first observations in 1991 and in 2011 (C. Madadzhe, pers. comm.) in Shingwedzi, however, a new population was reported in November 2022 (R. Nethengwe and L.C. Foxcroft, pers. obs.). Opuntia microdasys has potentially been eradicated from the southern KNP, but it may be too soon to determine conclusively. Cylindropuntia imbricata and Opuntia aurantiaca, previously known from the southern KNP, were considered eradicated (Foxcroft et al. 2017), but have since been found in various locations in the Pafuri (northern) and Malelane (southern) regions of KNP (T. Thwala and I. Paterson, pers. comm.). A new naturalized population of Pontederia cordata, previously considered probably eradicated, was recorded in Skukuza in 2021 and control measures initiated, but it is too soon to determine its eradication status. A naturalized population of Colocasia esculenta was considered eradicated, but ornamental plants are occasionally reported. Hylocereus undatus was previously considered eradicated; however, ongoing control of regrowth is still required in Skukuza. Harrisia martinii was eradicated from within the KNP boundary (N. van Wyk, pers. comm.), but as the population was located along the boundary fence, reinvasion is possible.
Dynamics of invasions from the 1920s until the present
Tagetes minuta (presently invasive), Cocculus hirsutus (naturalized) and Dysphania ambrosioides (status currently unknown) were the first three aliens recorded in KNP in 1930, followed by Gomphrena serrata and Argemone mexicana (both now naturalized) and Boerhavia diffusa (status unknown) in 1932. These species were followed by two presently invasive species, Lantana camara and Melia azedarach, in 1940 and 1948, respectively. A relatively steep increase followed in the next decade when the total number of all aliens increased from 20 in 1949 to 60 in 1958. Over a period of 28 years, between 1959 and 1999, additional 65 species were reported. Overall, since 1930, there has been an average detection rate (as a proxy of introduction) of 3.3 species per year, declining to 1.8 since 2000 (Fig. 4). This contrasts with invasive species, which have an overall average of 1.5 species per year; however, there has only been one new invasive species since 2000, Mimosa pudica (Fig. 4). Similar to invasive species, naturalized species have increased with an average of 1.9 overall, but 12 new naturalized species have been recorded since 2000. Six new species were recorded since the last published species lists.
Characteristics of the alien flora of KNP: taxonomy, origin, and life history
Alien species in KNP represent 41 families, of which 21 contain naturalized species and nine contain invasive species. The most represented families are Asteraceae (23 species, i.e., 15.8% of all aliens), Leguminosae (12.3%), Cactaceae (7.5%), and Solanaceae (7.5%) (Table 2). The families with the greatest representation of invasive species are Solanaceae (45.5% of its alien members) and Asteraceae (26.1%), while Cactaceae (18.2%) and Leguminosae (11.1%) have the lowest proportions of invasive species. Sixteen families are represented by a single species, and nine families include species of unknown status (Table 2).
The alien species in KNP originate from eight (of the nine) botanical continents (Fig. 5). The majority of species are native to Southern America (79 non-invasive vs. 16 invasives), closely followed by Northern America (63 vs. 15). Intercontinental introductions within Africa distantly follow (27 vs. 3) along with species from Temperate Asia (28 vs. 1), Tropical Asia (26 vs. 2), Australasia (11 vs. 2) and Europe (10 vs. 1). Species from the Pacific (7) have not yet become invasive. In addition, species status did not differ significantly by the species’ continent of origin: χ² (7, N = 291) = 8.072, p = 0.326.
Herbaceous plants are the prevailing life form amongst the alien flora of KNP, with 46% already naturalized and 13% invasive, while shrubs (52% naturalized and 24% invasive), climbers (26% naturalized and 9% invasive), and trees (36% naturalized and 11% invasive) are also distinct among the alien flora (Fig. 6A). Among specific life history groups, only two of the 15 succulents are invasive (Cereus jamacaru, Opuntia stricta). In comparison, out of the seven aquatic plants; two are naturalized (Azolla cristata, Salvinia molesta) and three are invasive (Pistia stratiotes, Pontederia crassipes, Pontederia cordata); S. molesta and A. cristata are likely to become invasive and widespread in the absence of biological control. In addition, although most of the alien flora comprise perennials, a slightly higher proportion of annual/biennial plants than perennials are invasive, with 16.7% and 12.3% of all taxa in a given life-span category (Fig. 6B). However, invasion status did not differ significantly by species’ life form χ² (7, N = 155) = 8.017, p = 0.331 or life span χ² (1, N = 154) = 0.544, p = 0. 461.
Frequency distribution of life histories A and life span B in the alien flora of the Kruger National Park (KNP). Note that the total number of taxa in the given categories exceed the total number present in KNPs alien flora as some taxa were assigned to more than one category. Non-invasive comprises casual, naturalized, and unknown species
Discussion
Our study provides an updated species inventory with a focus on species that are currently occurring in natural areas of KNP; this sets it apart from the majority of regional inventories of alien floras that rely on cumulative data over periods of time (Pyšek et al. 2017). Compared with previous lists, especially the most recent one, which included ~ 360 alien plant taxa (including extralimitals; Foxcroft et al. 2017), the complete list that we used to identify the currently present aliens included 407 taxa. We also list six new species: one detected by external/visiting scientists (Richardia scabra) and four recorded by the MOSAIK project (Bidens bipinnata, Boerhavia repens, Verbesina encelioides and Gomphrena celosioides; Pyšek et al. 2020a). The species most recently recorded by KNP staff members in 2022 is Pueraria montana var. lobata, a naturalized species that has the potential to quickly become invasive if not managed.
As PAs aim to conserve key elements of biological diversity, the impacts of biological invasions may be considered potentially more damaging in PAs than elsewhere but addressing this issue requires re-evaluating general PA policies, as well as overall priorities (Hulme et al. 2014). The importance of surveillance cannot be understated and should be a high priority. For example, although already naturalized, the detection of P. montana var. lobata before being considered widely invasive is providing managers with an opportunity to find and remove individual plants.
Complete inventories, which contain up-to-date taxonomic information, form a minimum requirement on which to base management actions. While there is a growing body of literature on the distribution of alien species in other regions globally (e.g., GloNaF database; van Kleunen et al. 2015, 2019; Pyšek et al. 2017) and their impacts (EICAT classification; Blackburn et al. 2014), local expert opinion and field experience is important to remain informed on the current distribution and invasion status of a species. However, one challenge for the KNP list was that approximately a quarter of the taxa could not be classified and are listed with their invasion status as unknown, resulting in missing information that may have unforeseen consequences. This is not surprising in the KNP where access is limited to a small road network (relative to the surface area of the KNP), hence the information coming from systematic surveys in open landscapes is comparably scarce (Pyšek et al. 2020a). Access to riparian areas is even more difficult, as roads are not always close to rivers, and working in riparian areas carries risks from dangerous animals. There is also a relative lack of highly skilled botanists that can identify alien species, especially uncommon or newly introduced species. This is not unexpected as Pyšek et al. (2013b) argued that the lack of taxonomists globally can hamper advances in science and management of alien species. This could potentially explain the limited number of casual records. In addition to biological factors limiting their survival, such as relatively arid conditions outside of riparian areas, their presence is also much less likely to be recorded.
The potential threat from naturalized and invasive species that are present in KNP is suggested by their global naturalization success as derived from the GloNAF and can be used to highlight the most dangerous invaders. Similarly, 12 species currently present in KNP are highlighted in the Global Invasive Species Database, which lists invasive alien species that threaten native biodiversity and natural areas, and five species are among the 100 of the world’s worst species.
The small number of invasive species compared to naturalized and casual species does not lessen the substantial threat these species pose. A few species alone have demonstrated the potential for widespread invasion, exemplified by species such as Opuntia stricta (a cactus mainly occurring in drier savanna areas), Parthenium hysterophorus (a herb of riparian and disturbed moist habitats), Lantana camara (a widespread shrub especially in riparian areas), and Pontederia crassipes (free-floating aquatic macrophyte on many rivers). Pontederia crassipes has been present in the Crocodile River since 1977 and later in the Letaba River and Olifants River (below the confluence with the Letaba River). The first population of P. crassipes was reported from the Sabie River in October 2020, although the source and precise date of introduction remain unknown.
The role of ornamental species in driving plant invasions in national parks, including KNP, was demonstrated by two studies (Foxcroft et al. 2008, 2019). A recent study comparing ornamental species checklists from 2003 and 2020 found that the total number of ornamental species has increased significantly, but species listed in national legislation or KNP regulations have reduced (Keet et al. 2022). Keet et al. (2022) suggest that one of the reasons for the increase was the greater sampling effort, however, the effect was less likely to have influenced legislated species which are more well known. Management actions targeting the removal of ornamental species may be contributing to the park's slow rate of new invasive alien plants, as some of the most invasive species are included in the regulations. A study that included 139 potential transformer species (subset of invasive plants that change the character, condition, form, or nature of ecosystems over a substantial area relative to the extent of that ecosystem; Richardson et al. 2000) across SANParks based on pathways and potential impacts of invasion listed 63 species for KNP (Foxcroft et al. 2019). While that list also included potential transformer species in camps or other infrastructure, our results were in agreement with 42 species that are already in the wild.
Mimosa pudica is the only invasive species reported to have been introduced since 2000, however, its potential for causing severe impacts in KNP is unknown. Parthenium hysterophorus is becoming one of the most invasive species in the park, and its distribution highlights the importance of systematic surveillance. It was first reported in 1991 along the Sand River (by Matthysen, G. Zambatis, Skukuza Herbarium Records, pers. comm.), and was either completely removed or disappeared. However, it was later recorded in the Lower Sabie–Crocodile Bridge region of the park in 2001, with rapidly increasing populations from its southern and western borders and along the main waterways in the southern region of the park. By 2016, ZAR 11.8 million had been spent on the manual and chemical control of P. hysterophorus (van Wilgen et al. 2017), but this was considered ineffective because the plants continued to spread.
The 2017 alien plant taxa list for SANParks (Foxcroft et al. 2017) included 118 taxa in KNP that appear in the South African National Environmental Management: Biodiversity Act (Act No. 10 of 2004, hereafter NEM:BA), Alien and Invasive Species Regulations (2016). The regulations mandate the management of listed alien and invasive species, including one Category 1a species (i.e., invasive species which must be eradicated from the environment), 93 Category 1b (i.e., invasive species requiring compulsory control as part of an invasive species control program), seven Category 2 (i.e., invasive species regulated by area) and 17 Category 3 species (i.e., invasive species regulated by activity). By focusing on the updated list of 146 alien species occurring in natural areas in KNP, managers can prioritize 83 of the NEM:BA legislated species (Alien and Invasive species Lists 2020), comprising one Category 1a species, 71 Category 1b species, seven Category 2 species, and three Category 3 species.
Given that we did not find any significant relationship between species status, origin, and life history (i.e., growth form and duration), factors other than the general species traits we examined here are responsible for invasion success. Invasion success in KNP is strongly associated with habitat affinities; the conditions in savanna ecosystems may facilitate (e.g., nutrients and soil moisture) or inhibit (e.g., fire, grazing) invasion and given the potentially stressful conditions such as drought and high temperatures, which generally do not promote plant invasions (Pyšek et al. 2017), the invasions occur mainly along rivers. For example, an intensive survey across 60 sites in KNP recorded 20 naturalized species (Pyšek et al. 2020a), with 60% occurring along perennial rivers.
The relatively low naturalization rate of alien species in KNP over the last 20–30 years allows some speculation on the collective outcome of a range of influences. Initially, the higher detection rate between 1959 and 1999, with 65 species added by the latter date, reflected increasing awareness of the alien species problem, and resulted in increased efforts to record new species. Additionally, a number of general botanical surveys (e.g., Van der Schijff 1957, 1969), followed by surveys aimed specifically at recording alien plants (e.g., Gertenbach 1985; Macdonald and Gertenbach 1988) were conducted during this period. However, over the last two decades the risks posed by alien species have become well known and methods of early detection and increased active management were implemented. Therefore, the prevention and management of intentional introductions into KNP (e.g., Foxcroft et al. 2008; Keet et al. 2022) have also likely contributed to the current suppressed rates of naturalization.
One problem associated with classifying data on alien species is dealing with uncertainty. Brock and Daehler (2020) presented a framework, based on a unified framework for biological invasions (Blackburn et al. 2011), for tracking alien plants in Hawai’i, which raised similar challenges we faced when assigning some species’ statuses in KNP. These included our “casual” and “unknown” categories, which correspond to the “questionably naturalized” category used by Brock and Daehler (2020). However, one drawback from applying the framework in the context of KNP is that compared to Hawai’i, or many islands, the accessibility for landscape-wide botanical monitoring is severely restricted in Kruger due to accessibility and presence of wild animals. This makes obtaining precise information about population dynamics and abundances of alien plants, which is needed for assigning invasion status, difficult, and represents a challenge for botanical research (Hejda et al. 2022).
Conclusions
Our aim was to filter the KNP list containing all alien plant taxa that accumulated through long-term research according to predefined criteria to highlight those that are invasive or naturalized and are likely to be amongst the most important species to consider for management. Our study identified 146 species that managers, and surveillance and mapping teams, should target as priority species, improving monitoring methods, and updating the status of species currently listed as unknown. These actions will contribute to building an up-to-date, useful, and accurate spatial database that can be used by managers to develop a comprehensive strategy for the control of naturalized and invasive species in the KNP. Information on the naturalization of alien species also contributes to the global knowledge base with special insights into the invasion processes driven by alien species following their introduction into areas with minimal anthropogenic disturbances. Collecting, storing, and sharing baseline information on the status and distribution of alien species is important for developing effective management measures in protected areas. Protected areas cover more than 22 million km2, representing approximately 16% of the worlds terrestrial and inland water ecosystems. Consequently, large areas that prioritize the management of alien species as a core function, have the potential to significantly contribute to global data collection and conservation efforts.
Data availability
All data generated or analysed during this study are included in this article and Supplementary Information files.
References
Blackburn TM, Pyšek P, Bacher S, Carlton JT, Duncan RP, Jarošík V, Wilson JRU, Richardson DM (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339. https://doi.org/10.1016/j.tree.2011.03.023
Blackburn TM, Essl F, Evans T, Hulme PE, Jeschke JM, Kühn I, Kumschick S, Marková Z, Mrugała A, Nentwig W, Pergl J, Pyšek P, Rabitsch W, Ricciardi A, Richardson DM, Sendek A, Vilà M, Wilson JRU, Winter M, Genovesi P, Bacher S (2014) A unified classification of alien species based on the magnitude of their environmental impacts. PLoS Biol 12:e1001850. https://doi.org/10.1371/journal.pbio.1001850
Brock KC, Daehler CC (2020) Applying an invasion and risk framework to track non-native island floras: a case study of challenges and solutions in Hawai‘i. NeoBiota 62:55–79. https://doi.org/10.3897/neobiota.62.52764
Brummit RK (2001) World geographical scheme for recording plant distributions, Ed. 2. Hunt Institute for Botanical Documentation, Carnegie Mellon University, Pittsburgh. http://rs.tdwg.org/wgsrpd/doc/data/
Codd LEW (1951) Trees and shrubs of the Kruger National Park. Mem Bot Surv S Afr 26:1–192
Cuthbert RN, Diagne C, Haubrock PJ, Turbelin AJ, Courchamp F (2022) Are the “100 of the world’s worst” invasive species also the costliest? Biol Invasions 24:1895–2004. https://doi.org/10.1007/s10530-021-02568-7
du Toit J, Rogers KH, Biggs HC (eds) (2003) The Kruger experience: ecology and management of savanna heterogeneity. Island Press, Washington, D.C.
Essl F, Dawson W, Kreft H, Pergl J, Pyšek P, van Kleunen M, Weigelt P, Mang T, Dullinger S, Lenzner B, Moser D, Maurel N, Seebens H, Stein A, Weber E, Chatelain C, Inderjit, Kartesz J, Morozova O, Nishino M, Novak PM, Pagad S, Shu W, Winter M (2019) Drivers of the relative richness of naturalized and invasive plant species on the earth. AoB Plants 11:plz051. https://doi.org/10.1093/aobpla/plz051
Foxcroft LC, Henderson L, Nichols GR, Martin BW (2003) A revised list of alien plants for the Kruger National Park. Koedoe 46:21–44. https://doi.org/10.4102/koedoe.v46i2.54
Foxcroft LC, Rouget M, Richardson DM (2007) Risk assessment of riparian alien plant invasion into protected areas—a landscape approach. Conserv Biol 21:412–421. https://doi.org/10.1111/j.1523-1739.2007.00673.x
Foxcroft LC, Richardson DM, Wilson JRU (2008) Ornamental plants as invasive aliens: problems and solutions in Kruger National Park, South Africa. Environ Manag 41:32–51. https://doi.org/10.1007/s00267-007-9027-9
Foxcroft LC, Jarošík V, Pyšek P, Richardson DM, Rouget M (2011) Protected area boundaries as a natural filter of plant invasions from surrounding landscapes. Conserv Biol 25:400–405. https://doi.org/10.1111/j.1523-1739.2010.01617.x
Foxcroft LC, Van Wilgen NJ, Baard JA, Cole NS (2017) Biological invasions in South African National Parks. Bothalia 47:a2158. https://doi.org/10.4102/abc.v47i2.2158
Foxcroft LC, Spear D, van Wilgen NJ, McGeoch MA (2019) Assessing the association between pathways of alien plant invaders and their impacts in protected areas. NeoBiota 43:1–25. https://doi.org/10.3897/neobiota.43.29644
Foxcroft LC, Baard JA, Bredenkamp NJ, Pagad S (2020) Protected Areas—Global Register of Introduced and Invasive Species—Kruger National Park, South Africa. Version 1.1. Invasive Species Specialist Group ISSG. Checklist dataset https://doi.org/10.15468/kgu2nt accessed via GBIF.org on 2020–05–28
Gertenbach WPD (1985) Alien plant section species lists. kruger national park, scientific services department, skukuza. Unpublished records, South African National Parks
Hejda M, Čuda J, Pyšková K, Zambatis G, Foxcroft LC, MacFadyen S, Storch D, Tropek R, Pyšek P (2022) Water availability, bedrock, disturbance by herbivores, and climate determine plant diversity in South-African savanna. Sci Rep 12:338. https://doi.org/10.1038/s41598-021-02870-3
Hulme PE, Pyšek P, Pergl J, Jarošík V, Schaffner U, Vilà M (2014) Greater focus needed on plant invasion impacts in protected areas. Conserv Lett 7:459–466. https://doi.org/10.1111/conl.12061
Invasive Species Specialist Group (ISSG) (2013) 100 of the world’s worst invasive alien species. IUCN, Gland, http://www.iucngisd.org/gisd/100_worst.php
Invasive Species Specialist Group (ISSG) (2021) Global Invasive Species Database. IUCN, Gland. http://www.iucngisd.org/gisd/about.php
Jarošík V, Pyšek P, Foxcroft LC, Richardson DM, Rouget M, MacFadyen S (2011) Predicting incursion of plant invaders into Kruger National Park, South Africa: the interplay of general drivers and species-specific factors. PLoS ONE 6:e28711. https://doi.org/10.1371/journal.pone.0028711
Keet J-H, Datta A, Foxcroft LC, Kumschick S, Nichols GR, Richardson DM (2022) Assessing the level of compliance with alien plant regulations in a large African protected area. Biol Invasions 24:3831–3844. https://doi.org/10.1007/s10530-022-02883-7
Kumschick S, Wilson JRU, Foxcroft LC (2020) A framework to support alien species regulation: the Risk Analysis for Alien Taxa (RAAT). NeoBiota 62:213–239. https://doi.org/10.3897/neobiota.62.51031
Kumschick S, Foxcroft LC, Wilson JR, Nkuna KV (2022) A framework for conducting risk analyses for alien species in South African National Parks. Scientific Report 02/2022. South African National Parks, Skukuza. https://doi.org/10.5281/zenodo.6519982
Leung B, Roura-Pascual N, Bacher S, Heikkilä J, Brotons L, Burgman MA, Dehnen-Schmutz K, Essl F, Hulme PE, Richardson DM, Sol D, Vilà M (2012) TEASIng apart alien species risk assessments: a framework for best practices. Ecol Lett 15:1475–1493. https://doi.org/10.1111/ele.12003
Macdonald IAW, Gertenbach WPD (1988) A list of alien plants in the Kruger National Park. Koedoe 31:137–150. https://doi.org/10.4102/koedoe.v31i1.491
MacFadyen S, Hui C, Verburg PH, Van Teeffelen AJA (2016) Quantifying spatiotemporal drivers of environmental heterogeneity in Kruger National Park, South Africa. Landsc Ecol 31:2013–2029. https://doi.org/10.1007/s10980-016-0378-6
Moodley D, Foxcroft LC, Novoa A, Pyšková K, Pergl J, Pyšek P (2020) Invasive alien species add to the uncertain future of protected areas. NeoBiota 57:1–5. https://doi.org/10.3897/neobiota.57.52188
Moodley D, Angulo E, Cuthbert RN, Leung B, Turbelin A, Novoa A, Kourantidou M, Heringer G, Haubrock PJ, Renault D, Robuchon M, Fantle-Lepczyk J, Courchamp F, Diagne C (2022) Surprisingly high economic costs of biological invasions in protected areas. Biol Invasions 24:1995–2016. https://doi.org/10.1007/s10530-022-02732-7
Mucina L, Rutherford MC (2006) The vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria
Nentwig W, Bacher S, Pyšek P, Vilà M, Kumschick S (2016) The generic impact scoring system (GISS): a standardized tool to quantify the impacts of alien species. Environ Monit Assess 188:315. https://doi.org/10.1007/s10661-016-5321-4
Obermeijer AA (1937) A preliminary list of the plants found in the Kruger National Park. Ann Transvaal Mus 17:185–227
Pyšek P, Danihelka J, Sádlo J, Chrtek J Jr, Chytrý M, Jarošík V, Kaplan Z, Krahulec F, Moravcová L, Pergl J, Štajerová K, Tichý L (2012) Catalogue of alien plants of the Czech Republic (2nd edition): checklist update, taxonomic diversity and invasion patterns. Preslia 84:155–255. https://www.preslia.cz/P122Pysek.pdf
Pyšek P, Hulme PE, Meyerson LA, Smith GF, Boatwright JS, Crouch NR, Figueiredo E, Foxcroft LC, Jarošík V, Richardson DM, Suda J, Wilson JRU (2013) Hitting the right target: taxonomic challenges for, and of, plant invasions. AoB PLANTS 5:plt042. https://doi.org/10.1093/aobpla/plt042
Pyšek P, Genovesi P, Pergl J, Monaco A, Wild J (2013) Plant invasions of protected areas in Europe: an old continent facing new problems. In: Foxcroft LC, Pyšek P, Richardson DM, Genovesi P (eds) Plant invasions in protected areas: patterns, problems and challenges. Springer, Dordrecht, pp 209–240
Pyšek P, Pergl J, Essl F, Lenzner B, Dawson W, Kreft H, Weigelt P, Winter M, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Dullinger S, Ebel AL, Figueiredo E, Fuentes N, Genovesi P, Groom QJ, Henderson L, Inderjit, Masciadri S, Maurel N, Meerman J, Morozova O, Moser D, Nickrent D, Nowak PM, Pagad S, Patzelt A, Pelser PB, Seebens H, Shu W, Thomas J, Velayos M, Weber E, Wieringa JJ, Baptiste MP, van Kleunen M (2017) Naturalized alien flora of the world: species diversity, taxonomic and phylogenetic patterns, geographic distribution and global hotspots of plant invasion. Preslia 89:203–274. https://doi.org/10.23855/preslia.2017.203
Pyšek P, Hejda M, Čuda J, Zambatis G, Pyšková K, MacFadyen S, Storch D, Tropek R, Foxcroft LC (2020a) Into the great wide open: do alien plants spread from rivers to dry savanna in the Kruger National Park? NeoBiota 60:61–77. https://doi.org/10.3897/neobiota.60.54608
Pyšek P, Hulme PE, Simberloff D, Bacher S, Blackburn TM, Carlton JT, Dawson W, Essl F, Foxcroft LC, Genovesi P, Jeschke JM, Kühn I, Liebhold AM, Mandrak NE, Meyerson LA, Pauchard A, Pergl J, Roy HE, Seebens H, van Kleunen M, Vilà M, Wingfield MJ, Richardson DM (2020b) Scientists’ warning on invasive alien species. Biol Rev 95:1511–1534. https://doi.org/10.1111/brv.12627
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing
Richardson DM, Pyšek P (2006) Plant invasions: merging the concepts of species invasiveness and community invasibility. Prog Phys Geogr 30:409–431. https://doi.org/10.1191/0309133306pp490pr
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107. https://doi.org/10.1046/j.1472-4642.2000.00083.x
Roy HE, Rabitsch W, Scalera R, Stewart A, Gallardo B, Genovesi P, Essl F, Adriaens T, Bacher S, Booy O, Branquart E, Brunel S, Gordon H, Dean H, D’hondt B, Josefsson M, Kenis M, Kettunen M, Linnamagi M, Lucy F, Martinou A, Moore N, Nentwig W, Nieto A, Pergl J, Peyton J, Roques A, Schindler S, Schönrogge K, Solarz W, Stebbing PD, Trichkova T, Vanderhoeven S, van Valkenburg J, Zenetos A (2018) Developing a framework of minimum standards for the risk assessment of alien species. J Appl Ecol 55:526–538. https://doi.org/10.1111/1365-2664.13025
Spear D, McGeoch MA, Foxcroft LC, Bezuidenhout H (2011) Alien species in South Africa’s National Parks (SANParks). Koedoe 53:1032. https://doi.org/10.4102/koedoe.v53i1.1032
Spear D, Foxcroft LC, Bezuidenhout H, McGeoch MA (2013) Human population density explains alien species richness in protected areas. Biol Conserv 159:137–147. https://doi.org/10.1016/j.biocon.2012.11.022
Vanderhoeven S, Branquart E, Casaer J, D’hondt B, Hulme PE, Shwartz A, Strubbe D, Turbe A, Verreycken H, Adriaens T (2017) Beyond protocols: improving the reliability of expert-based risk analysis underpinning invasive species policies. Biol Invasions 19:2507–2517. https://doi.org/10.1007/s10530-017-1434-0
Van der Schijff HP (1957) Ekologiese studie van die flora van die Nasionale Krugerwildtuin [Ecological study of the flora of Kruger National Park]. DSc thesis, Potchefstroom University for Christian Higher Education, Potchefstroom
Van der Schijff HP (1969) A checklist of the plants of the Kruger National Park. Publikasies van die Universiteit van Pretoria, Nuwe reeks 53: 1–100
van Kleunen M, Dawson W, Essl F, Pergl J, Winter M, Weber E, Kreft H, Weigelt P, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Ebel AL, Figueiredo E, Fuentes N, Groom QJ, Henderson L, Inderjit, Masciadri S, Meerman J, Morozova O, Moser D, Nickrent DL, Patzelt A, Pelser PB, Baptiste MP, Poopath M, Schulze M, Seebens H, Shu W, Thomas J, Velayos M, Wieringa JJ, Pyšek P (2015) Global exchange and accumulation of non-native plants. Nature 525:100–103. https://doi.org/10.1038/nature14910
van Kleunen M, Pyšek P, Dawson W, Essl F, Kreft H, Pergl J, Weigelt P, Stein A, Dullinger S, König C, Lenzner B, Maurel N, Moser D, Seebens H, Kartesz J, Nishino M, Aleksanyan A, Ansong M, Antonova LA, Barcelona JF, Breckle SW, Brundu G, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Conn B, de Sá DM, Dufour-Dror J-M, Ebel A-L, Figueiredo E, Fragman-Sapir O, Fuentes N, Groom QJ, Henderson L, Inderjit, Krestov P, Kupriyanov A, Masciadri S, Meerman J, Morozova O, Nickrent D, Nowak A, Patzelt A, Pelser PB, Shu W-S, Thomas J, Uludag A, Velayos M, Verkhosina A, Villaseñor JL, Weber E, Wieringa J, Yazlık A, Zeddam A, Zykova E, Winter M (2019) The global naturalized alien flora (GloNAF) database. Ecology 100:e02542. https://doi.org/10.1002/ecy.2542
van Wilgen BW, Fill JM, Govender N, Foxcroft LC (2017) An assessment of the evolution, costs and effectiveness of alien plant control operations in Kruger National Park, South Africa. NeoBiota 35:35–59. https://doi.org/10.3897/neobiota.35.12391
Watson JEM, Dudley N, Segan DB, Hockings M (2014) The performance and potential of protected areas. Nature 515:67–73. https://doi.org/10.1038/nature13947
Williamson M, Fitter A (1996) The varying success of invaders. Ecology 77:1661–1666
Acknowledgements
LCF thanks South African National Parks, the DSI-NRF Centre of Excellence for Invasion Biology (CIB), Department of Botany and Zoology, Stellenbosch University. This work is based on research supported in part by the National Research Foundation of South Africa (Grant Numbers 146068) to LCF; LCF was supported by grant no. 19-28807X (Czech Science Foundation) for his research stay at the Department of Invasion Ecology, Institute of Botany, Czech Academy of Sciences, to work on this publication. PP and DM were supported by grant no. 22-23532S (Czech Science Foundation) and by long-term research development project RVO 67985939 (Czech Academy of Sciences). We thank Khensani Nkuna for producing the map.
Funding
Open access funding provided by Stellenbosch University. DSI-NRF Centre of Excellence for Invasion Biology (CIB), Stellenbosch University, National Research Foundation of South Africa, Czech Science Foundation, Czech Academy of Sciences.
Author information
Authors and Affiliations
Contributions
LCF was the project leader, conceived the idea, and maintained the KNP data. PP was involved in conceiving the idea, developing methodology and structuring the paper. LCF and DM collated and prepared the data. GNR collaborated on KNP data collection and data preparation. DM conducted the statistical analysis. LCF wrote the first draft and all authors contributed.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Foxcroft, L.C., Moodley, D., Nichols, G.R. et al. Naturalized and invasive alien plants in the Kruger National Park, South Africa. Biol Invasions 25, 3049–3064 (2023). https://doi.org/10.1007/s10530-023-03098-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-023-03098-0