Abstract
In this paper, we develop a compartmental model of the COVID-19 epidemic in Burkina Faso by taking into account the compartments of hospitalized, severely hospitalized patients and dead persons. The model exhibits the traditional threshold behavior. We prove that when the basic reproduction number is less than one, the disease-free equilibrium is locally asymptotically stable. We use real data from Burkina Faso National Health Commission against COVID-19 to predict the dynamic of the disease and also the cumulative number of reported cases. We use public policies in our model in order to reduce the contact rate, and thereby to show how the reduction of daily reported infectious cases evolves with a view to assisting decision makers for a rapid treatment of the reported cases.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- COVID-19
- Statistics
- Data
- Reported and unreported cases
- Mathematical model
- Reproduction number
- Stability
- Public policies
- Basic reproduction number
- Contact function
- Prediction
1 Introduction
Since its appearance in China in December 2019, the coronavirus disease 2019 (COVID-19) has been subject to intensive activities in the field of mathematical modelling [1,2,3,4,5]. Modelling is done to allow a better understanding of the evolution dynamics of the disease. Several models have been proposed, some to describe the dynamics, other to estimate the parameters, but all aim at allowing decision makers to take appropriate measures in dealing with the epidemic. In fact, mathematical models play a very important role in the understanding of the spread of several diseases. The advent of COVID-19 is so another opportunity for mathematical modelers to translate the results of their modelling into clear terms for decision makers. The majority of Western countries have relied on mathematical models to predict the spread of the disease in their countries [1, 5,6,7,8]. This has allowed them to take measures ranging from hand washing to general containment. Similarly, on the African continent, many leaders rely on mathematical models for the management of epidemic such as COVID-19.
In Burkina Faso, a commitee of experts comprising various competences including mathematicians was set up as soon as the first case of coronavirus disease was reported in the country in March 2020. Since this date, the authorities’ concern was to know the dynamics of the pandemic and to see how to act to eliminate it. In doing so, several exchanges took place with public health doctors. It was at the end of these discussions that it was decided to develop a model which highlights the compartments of infected persons reported and not reported. Also it was a question of putting the accent on the high-risk hospitalized and quarantine or low-risk hospitalized. This explains the adoption of such a model study in this paper. The model concerning this paper make it possible to do projections and especially to see the effects of confinements, quarantines and cover fires in the country. Finally, the parameters used in this model for the simulations are parameters specific to the health context of Burkina Faso.
The difference between the former model (see [2]) and this one is that the new model takes into account some concerns of hospital practitioners and health epidemiologists from the Burkina Faso National Health Commission against COVID-19, people, of people hospitalized with serious health situation and death cases. We consider that dead people can contaminate health care workers or their loved ones when handling the bodies.
The paper is organized along 6 Sections. In the Sect. 2, we show the mathematical model, Sect. 3 is devoted to the basic properties. We present real daily data given by Burkina Faso National Health Commission against Coronavirus Disease 2019 (COVID-19) in Sect. 4 and we make simulations according to these data in Sect. 5. In Sect. 6 we end by a conclusion.
2 Mathematical Model
The model built for the coronavirus disease 2019 (COVID-19) in this paper is a SEIRD model, taking into account Susceptibles cases(S), Exposed persons (E), Infected individual (I), Recovered patients (R) and the Dead patients (D). Based on the epidemiological characteristics of COVID-19, the SEIRD model and its variants are more appropriate to study the dynamics of this disease, which is caused by the SARS-Cov-2. The output of the mathematical model gives the following transfer diagram (Fig. 1):
According to Fig. 1, we obtain the following system of ten differential equations describing the dynamics of the disease.
Here, we define the variables as follow:
-
S(t) (Susceptible) represents persons not infected by the disease pathogen.
-
E(t) (Exposed) refers to persons who are in the incubation period after being infected by the disease pathogen, and haves no visible clinical signs. These individuals could infect other people but with a lower probability than people in the infectious compartments. After the incubation period, these persons move to one of the Infectious states. In this model, it can be seen has contact persons
-
I(t) (Infectious) refers the number of persons who are beginning to develop clinical signs, these persons are symptomatic infectious,
-
I d(t) refers to Infectious; that is persons who can infect other people, are developing clinical signs and therefore will be detected and reported by authorities (when arriving in the H R or the H d compartments). After this period, the people in this compartment are taken in charge by sanitary authorities and we classify them as Hospitalized patients. It is the reported infectious cases.
-
I u(t) is the number of unreported symptomatic infectious individuals (i.e., symptomatic infectious with mild symptoms) at time t.
-
Persons hospitalized or quarantined at home (but detected and reported by the authorities) who will recover (denoted by H R): These persons are in hospital (or quarantined at home) and can still infect other people. At the end of this state, these persons move to the Recovered state. It is the low-risk hospitalized cases.
-
Hospitalized people who are going to die (denoted by H d): These persons are hospitalized and can still infect other people. At the end of this state, these persons move to the death state. It is the high-risk hospitalized cases.
-
\(R_{I_u}\) Recovered persons from unreported infectious cases (denoted by \(R_{I_u}\)): These persons have survived the disease, are no longer infectious and have developed a natural immunity to the disease pathogen.
-
\(R_{I_d}\) Recovered persons from reported infectious cases (denoted by \(R_{I_d}\)): These persons have survived the disease, are no longer infectious and have developed a natural immunity to the disease pathogen.
-
D Dead persons (denoted by D): These persons have not survived the disease.
-
N is the number of people within the territory before the start of the pandemic.
The transcription of the transfer diagram gives the following mathematical system:
The initial data of the system is supplemented by
The time t is in day, the asymptomatic infectious individuals I(t) are infectious for an average time period of 1∕α days. We suppose that the population is constant i.e N = S(t) + E(t) + I(t) + R(t).
We notice that 8th, 9th and 10th equations of system (1) are not coupled with the other equations. So we can solve the first seven equations of the system and compute them after the other three equations. Thus, the system taken into account is the following with seven equations:
3 Basic Properties
In this section, we prove the positivity and the boundedness of the solution of the system (1) with initial conditions defined in (2).
For that, let us state the following lemma.
Lemma 3.1 ([9])
Suppose \(\varOmega \subset {\mathbb {R}}\times \mathbb {C}^n\) is open and \(f_i\in C(\varOmega , \mathbb {R}), \;i=1,\,2\,\cdots ,\, n.\) If \(f_i\vert _{x_i(t)=0,\,X_t\in C_{+0}^n}\geq 0\) , X t = (x 1t, x 2t, ⋯ , x nt), then, \(\phi =(\phi _1,\phi _2,\cdots ,\phi _n)\in C([-\tau , 0],\mathbb {R}_{+0}^n)\) is the invariant domain of the following equations.
where \(\mathbb {R}_{+0}^n=\{(x_{1},\,x_{2},\cdots ,\,x_{n}):x_i\geq ,\; i=1,\,2\,\cdots ,\, n.\)
Proposition 3.1
The system (1) is invariant in \(\mathbb {R}_{+}^{10}\).
Proof
Let us write system (1) as
so, note that
From Lemma 3.1, we conclude that system (1) is invariant in \(\mathbb {R}_{+}^{10}\).
Lemma 3.2
The solution of system (1) is bounded in the region
Proof
For system (1), we observe that
\(\dot N=0\), ⇒ N(t) = Cst, ∀t ≥ 0, thus all the solution of system (1) are bounded.
The only equilibrium of the model is the disease free equilibrium (DFE) defined by E DFE = (N, 0, 0, 0, 0, 0, 0, 0, 0, 0) = (S 0, 0, 0, 0, 0, 0, 0, 0, 0, 0).
Proposition 3.2
The basic reproduction number for the model system (1) is defined by
Proof
We use the method of next generation matrix in [10] to compute the reproduction number \(\mathcal {R}_{0}\). So we get:
and
We get the Jacobian matrixes of these two functions \(\mathcal {F}\) and \(\mathcal {V}\) as follow.
and at the point E DFE, we have
and at the point E DFE, we have
Thus, we obtain
where
The basic reproduction number is defined as the dominant eigeinvalue of the matrix − FV −1.
Therefore,

The basic reproduction number \( \mathcal {R}_{0}\) is defined as the number of cases that one infected person generates on average during his infectious period, within an uninfected population and without any special control measures. This number does not change during the spread of the disease.
The effective reproduction number \( \mathcal {R}_{e}(t)\) is defined as the number of cases that one infected person generates during his infectious period. This effective reproduction number depends on time, therefore, on public policies (it changes during the spread of the disease). Furthermore, \(\mathcal {R}_{e}(t)=\mathcal {R}_{e}-\mathcal {R}_{0}\) at t = 0 and the spread of the disease slows when \( \mathcal {R}_{e}(t)<1\).
Without any public policies,
so
and, therefore,

Using Theorem 2 in [10], the following result is established.
Proposition 3.3
The disease free equilibrium E DFE of he model (3) is locally asymptotically stable if \( \mathcal {R}_{0}<1\) and unstable if \( \mathcal {R}_{0}>1\).
4 Data
In this part, we show real data generated by CORUS, the Burkina Faso National Health Commission against the Coronavirus Disease 2019 (COVID-19). Recall that the first cases were reported in Burkina Faso on March 9, 2020.
In Appendix A, Table 1 represents the daily cumulative number of reported infected cases and Table 2 the daily cumulative reported dead cases.
5 Numerical Simulations
For the numerical simulations, we focus on the public policies represented in the model (1) by functions \(m_E,\; m_I,\; m_{I_u},\; m_{I_d}, \;m_{H_R}\) and \(m_{H_d}\). From the beginning date of the public policies, we decrease the contact rate, as a result of the various types of measures taken by the public authorities. The effect of these measures can be seen on the spikes of the different types of infectious cases; the spikes decrease according to the degrees of the measures taken. The curve of the contact rate (contact function) is represented in the Fig. 4.
where γ minq is the minimum contact rate and μ q is the rate at which the contact decreases.
Depending on the public measures taken, μ i increases, so the contact rate γ(t) decreases and it is possible to limit the number of infected persons (see Fig. 4). μ i is chosen in such a way that the simulation for the time interval aligns with the cumulative reported case data. Also, the effective reproduction number \(\mathcal {R}_{e}(t)\) is strongly dependent on the contact function which decreases exponentially by interval as shown in Fig. 5. Thus we are able to predict the future values of the epidemic from the early cumulative reported data. The earlier public policy decisions are made, the better the management of the epidemic. For Burkina Faso, we present the model, first, without any public policy intervention and, secondly with public policies taken by the local government. It is important to observe that the basic reproduction number with the parameters taken for this simulation: \(\mathcal {R}_{0}=\mathcal {R}_{e}(0)=4.5\); this mean that 10 persons infect 45 persons, which is very high. The role of public policies is to reduce this \(\mathcal {R}_{e}\) down to less than one, which will ensure the extinction of the epidemic.
5.1 Situation Without Public Policies
Without any public policies, the situation of the pandemic would be dramatic for the African countries and for Burkina Faso in particular. In fact, as shown in the simulation (see Figs. 2 and 3), 9 millions persons would be infected by the COVID-19 and around 850 thousand would die, that is 9% of infected persons.
5.2 Situation with Public Policies
Fortunately, in Burkina Faso, as in almost all countries in the world, from the first cases of the disease, a succession of barrier measures were taken, ranging from hand washing, to the closing of schools, and, places of worship and markets, and the establishment of curfews. Consequently, in the following simulations, we have chosen our contact function in such a way that the cumulative number of infected persons (model (1)) fits the data produced by the Burkina Faso National Health Commission against COVID-19.
Figure 5 represents the dynamic of the effective reproduction number and Fig. 4 represents the dynamic of the contact function. These two curves clearly show the effects of three measurements according to their implementation time.
Figure 6 presents the histogram of the daily reported infected cases and the infected and the dead given by the model 1.
6 Conclusion
In this paper, we are proposing a model for the transmission of the coronavirus disease 2019. We calculated the \(\mathcal {R}_{0}\) which is very essential in understanding the disease and we have showed the local stability of the disease free equilibrium DFE. We subsequently adjusted the model to the actual data of the National Health Commission against the coronavirus disease 2019. From this adjustment, we have been able to draw a number of consequences for the further management of the pandemic.
Firstly, from Fig. 5, we can say that the peak of the epidemic was reached in Burkina Faso around April 5, 2020 (Fig. 6).
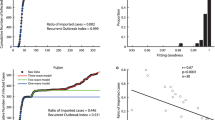
Secondly, the data collected are not sufficiently homogenous, which allow for some reservation on the reliability of the data (Fig. 7). Nevertheless these data constitute a basis to make prescriptions for the dynamics of the disease and especially for removing the barrier measures. We achieved this by fitting the model with the data in Figs. 8, 9 and 10.
Finally, we can notice that the effective reproduction number \(\mathcal {R}_{e}\) would be less than 0.5 at April 15, 2020, naturally according to the data fit with the model (1). We therefore think that one week after this date, certain barrier measures could be reviewed, for instance to allow for the opening of markets but maintaining the rule of distance, the opening of schools under conditions which do not allow the spread of the disease, and authorization for many other sectors of active life to be reopen.
References
B. Ivorra, M. R. Ferrández, M. Vela-Pérez, A. M. Ramos; Mathematical modeling of the spread of the coronavirus disease 2019 (COVID-19) considering its particular characteristics. The case of China; Preprint, March 2020.
A. Guiro, B. Koné and S. Ouaro; Mathematical model of the spread of the coronavirus disease 2019 (COVID-19) in Burkina Faso. Applied Mathematics, 2020, 11, 1204-1218. https://www.scirp.org/journal/am.
Q.Y. Lin, S. Zhao, D. Z. Gao, et coll; (2020) A conceptual model for the coronavirus disease 2019 (COVID-19) outbreak in Wuhan, China with individual reaction and governmental action.Int. J. Infect. Dis. 93 :211–216.
Z. Liu, P. Magal, O. Seydi and G. Webb; Predicting the cumulative number of cases for the COVID-19 epidemic in China from early data, medRxiv preprint, available under a CC-BY-NC-ND 4.0 International license.
Z. Liu, P. Magal, O. Seydi and G. Webb; Understanding Unreported Cases in the COVID-19 Epidemic Outbreak in Wuhan, China, and the Importance of Major Public Health Interventions, Biology 2020, 9, 50; https://doi.org/10.3390/biology9030050.
European Centre for Disease Prevention and Control. Discharge criteria for confirmed COVID-19 cases When is it safe to discharge COVID-19 cases from the hospital or end home isolation, https://www.ecdc.europa.eu/sites/default/files/documents/COVID-19-Discharge-criteria.pdf, March 2020.
Johns Hopkins University (JHU). Coronavirus COVID-19 Global Cases by the Center for Systems Scienceand Engineering (CSSE). https://gisanddata.maps.arcgis.com/apps/opsdashboard/index.html/bda7594740fd40299423467b48e9ecf6, March 2020.
R. Li, S. Pei, B. Chen, Y. Song, T. Zhang, W. Yang, and J. Shaman; Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV2). Science, 2020.
X. Yang, L. Chen, and J. Chen; Permanence and positive periodic solution for the single-species nonautonomous delay diffusive models. Computers & Mathematics with Applications, 32(4):109116, 1996.
P. Van den Driesche and J. Watmough; “Reproduction Numbers and Substhreshold Endemic Equilibria for the Compartmental Models of Disease Transmission”, Mathematical Biosciences, Vol. 180, No. 1–2, 2002, pp. 29–48.
B. Ivorra, A. M. Ramos, and D. Ngom; Be-CoDiS: A mathematical model to predict the risk of human diseases spread between countries. Validation and application to the 2014 Ebola Virus Disease epidemic. Bulletin of Mathematical Biology, 77(9): 1668–1704, 2015.
T. Liu, J. Hu, M. Kang, L. Lin, H. Zhong, J. Xiao, and et al.; Transmission dynamics of 2019 novel coronavirus (2019-ncov). bioRxiv, 2020.
World Health Organization. Report of the WHO-China Joint Mission on Coronavirus Disease 2019. https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf, February 2020.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Appendix A. Tables of Data
Appendix A. Tables of Data
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Guiro, A., Koné, B., Ouaro, S. (2021). Mathematical Modelling of the Evolution Dynamics of the Coronavirus Disease 2019 (COVID-19) in Burkina Faso. In: Toni, B. (eds) The Mathematics of Patterns, Symmetries, and Beauties in Nature. STEAM-H: Science, Technology, Engineering, Agriculture, Mathematics & Health. Springer, Cham. https://doi.org/10.1007/978-3-030-84596-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-84596-4_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-84595-7
Online ISBN: 978-3-030-84596-4
eBook Packages: Mathematics and StatisticsMathematics and Statistics (R0)