Abstract
Positron emission tomography/computed tomography with 18F-fluorodeoxyglucose (18F-FDG PET/CT) has been increasingly used in the evaluation of breast cancer patients. Although not routinely performed for diagnosis of primary cancer or initial axillary staging, PET/CT has a high sensitivity for distant metastases and can help staging patients with advanced disease when other conventional methods are inconclusive. It is also indicated to detect recurrence that may be suspected based on clinical, radiological, or laboratory/tumor marker findings. The introduction of new tracers, such as FES, and the availability of breast dedicated and PET/MRI equipment may increase the future role of PET imaging in breast cancer patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Breast neoplasms
- Neoplasm staging
- Positron emission tomography
- Positron emission tomography/computed tomography with fluorodeoxyglucose F18
- Radioactive tracers
1 Introduction
1.1 Basic Aspects of 18F-FDG-PET
Positron emission tomography (PET) allows the assessment of different metabolic parameters based on the detection of in vivo biodistribution of intravenously administered compounds, labeled with positron-emitting isotopes. Short-lived positron emitters are used to label several organic molecules, without interfering in their biological properties. During image acquisition, the positron emitted by the radiopharmaceutical interacts with an electron, and both undergo annihilation, emitting two gamma rays in opposite directions. In a typical PET equipment, these rays are detected simultaneously by scintillation crystals arranged like a ring around the patient in the detector system (Fig. 9.1). PET/CT cameras integrate PET imaging to a computed tomography (CT), adding the anatomical and morphological information of the CT to the functional information of PET. More recently, PET/MRI scan has also become available, allowing the integration of PET and magnetic resonance imaging (MRI) in a single study. This chapter will mainly review the application of PET/CT in breast cancer but will also discuss some aspects of PET/MR.
Fluorodeoxyglucose (FGD) is the most studied radiopharmaceutical in the evaluation of breast cancer. The FDG molecule is a glucose analog, in which a hydroxyl group has been replaced by fluorine-18, obtained in a cyclotron. The uptake of 18F-FDG by tumor cells occurs through non-insulin-dependent glucose transport systems (GLUTs 1 and 4) and, like glucose, undergoes phosphorylation due to hexokinase. Phosphorylated FDG does not progress in the metabolic pathway beyond this step and remains trapped in the tumor cell (Fig. 9.2). Most breast cancers present increased metabolic activity, although there are variations according to tumor characteristics and histology, to be discussed below.
PET/CT study is usually acquired 60 min after administration of 18F-FDG to the fasting patient, who needs to remain at rest during the interval. Visual analysis is the basis for interpreting a PET/CT study. Semiquantitative measures can help to compare or monitor the degree of metabolic uptake, the most used being the SUV – standardized uptake value. SUV refers to the activity in a given volume of interest in relation to the total activity administered, corrected by the patient’s weight.
\( SUV=\frac{\mathrm{Tissue}\kern0.17em \mathrm{activity}\kern0.17em \mathrm{concentration}\left(\frac{\mathrm{MBq}}{\mathrm{mL}}\right)}{\mathrm{Injected}\kern0.17em \mathrm{dose}\;\left(\mathrm{MBq}\right)}\times \mathrm{body}\kern0.17em \mathrm{weight}\;\left(\mathrm{g}\right) \)
Other tissues also show physiological uptake of FDG. Among the organs with intense physiological uptake of 18F-FDG is the brain, impairing the ability to detect metastases at this site. Activity in the kidneys, ureters, and bladder resulting from the excretion of the radiopharmaceutical generally does not interfere with the interpretation of the study. It should also be remembered that inflammatory changes with infiltration by macrophages or granulation tissue (e.g., postoperative) have high metabolic activity and may lead to a false-positive study. Other causes of false-positive studies in the breast include benign conditions such as breast changes in pregnancy and lactation (Fig. 9.3), gynecomastia, mastitis, fat necrosis (Fig. 9.4), fibroadenoma, intraductal papilloma, and atypical ductal hyperplasia [1].
2 18F-FDG-PET/CT Indications in Breast Cancer
The National Comprehensive Cancer Network (NCCN) Guidelines do not indicate PET/CT in the staging of clinical stages I and II or operable stage III breast cancer. FDG PET/CT is suggested to be most helpful in situations where standard staging studies are equivocal or suspicious, especially in the setting of locally advanced or metastatic disease [2]. Regarding surveillance, the American Society of Clinical Oncology and NCCN guidelines recommend only regular history, physical examination, and mammography for breast cancer routine follow-up. Systematic 18F-FDG PET/CT is not indicated [3].
Although not appropriate for all patients with breast cancer, the use of 18F-FDG PET/CT can have an impact on patient care in multiple settings, including initial staging, treatment response assessment, and evaluation of suspected recurrence [4].
Influence of Histologic Subtypes and Receptor Status
Breast cancer is considered as a group of diseases with different molecular characteristics that originate in breast epithelial tissue but have different prognosis, patterns of recurrence, and dissemination after primary multidisciplinary treatments, leading to significant changes in diagnostic and therapeutic approaches [5].
18F-FDG uptake depends on the histologic and biologic characteristics of the breast tumor and is influenced by its receptor status, grade, and histologic type [3, 4]. Invasive carcinoma of no special type (NST) exhibits higher uptake than invasive lobular carcinoma (ILC) [3, 6]. Lobular breast cancers may be occult at FDG PET even with large dimensions [4]. This is probably due to the lower density of tumor cells in lobular carcinomas, lower expression of GLUT1, lower proliferation rates, and diffuse infiltrative tumor growth patterns into surrounding tissue, which may lead to false-negative scans [3]. Untreated osseous metastases from ILC are more likely sclerotic and missed by FDG PET than IDC metastases, showing a lower avidity and uptake similar to the background. In addition, ILC differs from NST in its patterns of metastatic spread, with a greater propensity to metastasize to the gastrointestinal tract and retroperitoneum, which are areas often difficult to assess with FDG PET as they are common sites of physiologic and variable FDG avidity [4].
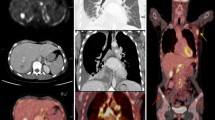
Estrogen receptor (ER)-negative tumors and grade 3 cancers have significantly higher FDG avidity than ER-positive tumors and lower-grade malignancies [4]. Also, triple negative breast cancers demonstrate particularly elevated uptake of fluorodeoxyglucose (FDG) on PET. They are also known to result in early metastatic disease and have a propensity for extra skeletal metastases, increasing the importance of imaging for systemic staging [6] (Fig. 9.5).
Restaging with PET/CT in a patient with triple negative breast cancer presenting with reduction of strength in the left hemibody. MIP (a), sagittal PET/CT fusion (b), and axial PET/CT fusion (c–e) images show multiple metastasis with intense FDG uptake on the central nervous system, lungs, liver, and bones
Human epidermal growth factor receptor 2 (HER2)- positive breast cancers are characterized by a high expression of HER2 gene, which promotes tumor growth and progression and therefore tends to be more aggressive and FDG avid. Poorly differentiated tumors are more aggressive tumors and are more FDG avid [7]. There is also a positive correlation between the tumor proliferation index (Ki-67 expression) and the intensity of 18F-FDG uptake [3].
Question
Is the sensitivity of FDG PET similar for lobular and invasive carcinoma of no special type breast cancer? Why?
2.1 Primary Breast Tumor Detection and Staging (T)
FDG PET/CT is not indicated for screening in early-stage cancers [8]. Although it offers the opportunity to provide an overview of disease in a single procedure, the recommendation against the use of PET scanning is supported by many factors [3, 8]: it has low sensitivity for the primary breast tumor, and neither FDG PET nor CT is sensitive enough to detect breast cancers smaller than 1 cm [4]; the SUV in small lesions is susceptible to the partial volume averaging effect, which may lead to a lower value. This is further hampered by potential breathing motion artifact, as the PET acquisition is performed at tidal volume breathing [8]; 18F-FDG imaging has lower sensitivity than the sentinel node technique in assessing axillary lymph node involvement, and the risk of distant metastases in early-stage cases is also low [3]. In addition to that, there is a high rate of false-positive scans in early-stage cancers, leading to unwarranted patient anxiety and delay of care in those patients [3].
However, in high-risk patients like those with inflammatory (T4d) or locally advanced breast cancer (LABC), the role of 18F-FDG imaging in detecting local and distant metastasis has been highlighted [3]. FDG PET is helpful in local staging and particularly helpful in evaluation of internal mammary nodes and distant nodal metastasis [7]. It also helps to evaluate the equivocal findings on standard imaging and, in some cases, can detect unknown sites of distant metastasis even though the standard imaging is negative for lesions [7].
Any FDG-avid breast focus found during staging or surveillance of an extra-mammary malignancy should be thoroughly investigated in patients with reasonable life expectancy [8] (Fig. 9.6). These lesions have a 30–40% chance of being malignant, including previously unsuspected primary breast malignancies, metastases to the breast, and breast lymphoma [4]. The likelihood that the lesion is a primary breast cancer is approximately 6%, and more than half of these will be early-stage disease, with potential for cure [8].
Question
An intense area of FDG uptake was detected in the breast of a patient during the investigation of a solitary pulmonary nodule. Considering that FDG PET is not indicated for detecting a primary breast tumor, do you think this finding should be further evaluated?
2.2 Nodal Staging (N)
For the evaluation of locoregional nodal metastases, it is useful to make a distinction between axillary and regional extra axillary nodes [4]. Axillary lymph node status is one of the main prognostic factors in breast cancer. If there are no palpable lymph nodes on clinical examination, the currently accepted approach for axillary staging is sentinel lymph node biopsy. This technique has the advantage of detecting even micrometastases (<2 mm) or isolated tumor cells [9]. The sentinel lymph node biopsy predicts the state of axillary disease with an accuracy greater than 95%. FDG PET/CT has poor sensitivity for axillary nodal metastases compared with sentinel lymph node biopsy, because clinically relevant axillary nodal metastases are often subcentimeter in size. But the specificity of FDG PET/CT for axillary nodes has been shown to be far better than its sensitivity. Thus, the presence of an FDG-avid axillary node is likely to represent nodal malignancy [4].
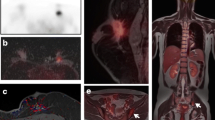
Locoregional extra axillary nodes, including internal mammary, infraclavicular, and supraclavicular nodes, may be clinically occult and less commonly identified by sentinel node evaluation (Fig. 9.7). It is in this group of nodes where FDG PET/CT evaluation begins to show value in patient staging through the detection of unsuspected extra axillary nodal metastases [4]. Axillary node clearance by axillary dissection is usually limited to levels I and II. 18F-FDG uptake suggesting involvement at level III (infraclavicular) or in extra axillary locoregional nodes (supraclavicular or internal mammary) may have important implications in surgical management and in the design of radiation therapy fields [3, 9].
Question
A patient with a palpable axillary node had an FDG PET study acquired prior to the surgery. Intense FDG uptake was noted in multiple axillary nodes and also in the internal mammary node. What is the practical implication of this finding?
2.3 Metastatic Staging (M)
The most common sites of distant metastasis in breast cancer are bones, lungs, liver, and brain, and the conventional imaging studies for detecting distant metastasis include contrast-enhanced CT, bone scintigraphy, and MRI [10]. FDG PET/CT has higher sensitivity than conventional modalities for detection of unsuspected distant metastasis in patients with locally advanced breast cancer, changing the patient’s stage and converting patient’s management from curative-intent therapy by surgery with or without neoadjuvant therapy to palliative systemic therapies [4].
Groheux et al. compared a conventional staging approach including bone scanning, chest radiography or dedicated CT, and liver ultrasound or contrast-enhanced CT for abdomen–pelvis with a single session of staging with 18F-FDG PET/CT. 18F-FDG PET/CT outperformed conventional imaging for bone, distant lymph nodes, and liver metastases, whereas CT was more sensitive for lung metastases [3]. PET lacks sensitivity for detection of infracentimetric pulmonary nodules because of the partial-volume effect and respiratory movements. Careful scrutiny of CT images from PET/CT can reveal small nodules without 18F-FDG uptake. However, CT performed during free breathing is less efficient than standard diagnostic thoracic CT [3].
For evaluation of bone metastasis in breast cancer patients, both bone scintigraphy and FDG PET/CT are convenient whole-body imaging tools [11]. FDG PET acts as a tumor-specific tracer and reflects the glucose usage by tumor cells in viable metastatic lesions, while bone scintigraphy mainly reflects the response of surrounding bone to cancer. Bone scan may fail to show early response to effective therapy and may even show a “flare” related to bone healing. Similar findings may occur with other modalities including CT [11, 12]. Metabolic flare may be seen at FDG PET, with temporarily increasing FDG avidity after successful therapy; however, it occurs in the first 1–2 weeks and, thus, is not a confounding issue on scans that are normally performed months after initiating therapy. Rather, FDG metabolic flare may be an indicator of future response to therapy [4].
PET is generally considered to be superior to CT and bone scintigraphy in detecting lytic or mixed bone metastases and bone marrow metastases (Fig. 9.8), but a multimodality approach is recommended for the investigation of bone metastases due to the low sensitivity of PET in detecting sclerotic bone metastases in some cases. In addition, sclerotic lesions without FDG accumulation can be detected on CT images of a PET/CT study [13].
FDG PET is not as sensitive as MRI in the evaluation of brain metastases. Cerebral cortex is highly FDG avid, and metastases often appear as focal areas of hypometabolism, which may also be seen in non-neoplastic entities. Some lesions do manifest as focal areas of hypermetabolism, although this can be difficult to detect in the setting of normal physiologic gray matter metabolism [14]. Furthermore, inflammatory tissue can also exhibit high FDG tracer uptake, diminishing diagnostic specificity [15].
Question
Why, despite the high global sensitivity for breast cancer metastases, PET FDG is not indicated for the detection of brain lesions?
2.4 Recurrence
Breast cancer recurrence can be suggested by clinical symptoms, radiologic findings, or rising levels of tumor markers (carcinoma antigen 15-3, carcinoembryonic antigen or cancer antigen 125) [3]. 18F-FDG PET/CT has a high diagnostic accuracy in detecting breast cancer recurrence in case of elevated levels of serum tumor markers, and it can be used in addition to conventional imaging techniques [16] (Figs. 9.9 and 9.10). The European Society for Medical Oncology (ESMO) guidelines and NCCN suggest that FDG PET/CT can be useful for identifying the site of relapse when traditional imaging methods are equivocal or conflicting, because it allows better discrimination between posttreatment scar or fibrosis and viable tumor tissue [17]. Moreover, this imaging modality can improve the identification of isolated locoregional relapse as well as isolated metastatic lesions, that is, a situation where patients may benefit from a more aggressive multidisciplinary approach [17]. PET/CT is also efficient in patients with suspected recurrence but with negative tumor marker results [3].
2.5 Response Assessment to Neoadjuvant Chemotherapy (NAC)
Breast cancer response to NAC has traditionally been assessed by conventional imaging modalities, which sometimes have difficulties in differentiating fibrosis from residual tumors. 18F-FDG PET/CT and enhanced MRI are used in this clinical setting [18]. Studies using 18F-FDG PET/CT to monitor early tumor responses to NAC showed a moderate accuracy to identify pathological responses in breast cancer patients, with better results than mammography, sonography, and MRI in predicting pathologic complete response (pCR) during NAC for locally advanced breast cancer [19].
18F-FDG PET/CT can differentiate changes in tumor glucose metabolism before morphologic changes. The decrease in 18F-FDG uptakes in tumors after chemotherapy is an indicator to assess the treatment response in triple-negative breast cancer (TNBC) and HER2-positive subtypes [18, 20]. Enhanced MRI can provide information on lesion microvasculature and depict changes in the physiologic characteristics of tumors [18]. PET/MRI holds the promise to improve therapy-response evaluation because it has the high sensitivity of PET and the high specificity of the MRI component [18].
2.6 Response Assessment in Metastatic Disease
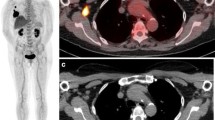
Accurate assessment of treatment response is vital to provide the most effective therapy as well as to avoid unnecessary treatment escalation [21]. The current standard of measuring treatment response in metastatic breast cancer relies on size measurements of tumors, usually on CT [4]. Nonetheless, there are some inherent limitations in the size criteria. Distinguishing viable from nonviable residual tumor tissue is often difficult, and osseous metastases are in general nonmeasurable [22]. Metabolic changes measured by FDG PET may better predict treatment response than anatomic changes because PET/CT can differentiate active tumors from post-therapeutic changes and assess metabolic activity in osseous metastases [4, 22]. 18F-FDG PET/CT seems to be accurate in directing treatment of metastatic bone disease as it reflects tumor activity, which is structurally difficult to be assessed by CT scan alone or by bone scintigraphy (Fig. 9.11). The latter reflects bone reaction against metastatic tumor that increases as the disease responds to treatment [21].
Response assessment in a patient with metastatic breast cancer after chemotherapy. Pretreatment FDG PET/CT images (a and b) show intense bone lesion FDG uptake in the sternum. (b) Follow-up FDG PET/CT images after chemotherapy (c and d) show significant decrease in the intensity and extent of FDG uptake in the sternum
FDG PET was able to distinguish responders from nonresponders after distinct and varied courses of hormonal and chemotherapies of breast cancer metastases [4]. Riedl et al. showed that metabolic assessment by FDG PET/CT was a better predictor of both progression-free and disease-specific survival than Response Evaluation Criteria in Solid Tumors (RECIST) evaluation on CT, and this difference would alter overall patient management in 25% of the patients, reducing the morbidity and costs of ineffective therapies in clinical practice [22].
Question
Is FDG PET a good method to assess whether a persistent residual mass after therapy corresponds to a viable tumor or fibrosis?
3 Prognostic Value
FDG PET has been found to have also a role in predicting the prognosis of breast cancer [23], indicating those patients more likely to progress, also with a correlation between SUV measurement and histologic grade, proliferation index, and triple-negative status [3]. High uptake reflects aggressive tumors and has poor prognosis [7]. Although SUVmax is one of the most widely used parameters in clinical settings, it does not show the uptake of the entire tumor mass and may not reflect the intratumor heterogeneity sufficiently [23]. Therefore, there has been an increasing interest in volumetric parameters such as metabolic tumor volume (MTV) and total lesion glycolysis (TLG), which are becoming easier to measure due to commercially available softwares [23]. The metabolic parameters SUVmax, MTV, and TLG of the lesion before treatment are related to the recurrence rate. The higher the metabolic parameters of the primary lesion, the greater the possibility of recurrence and distant metastasis [24].
4 Other PET Technologies and Tracers
4.1 PET/MR and Positron Emission Mammography (PEM)
After the rapid incorporation of PET/CT into clinical practice for staging and prognostic evaluation of breast cancer patients, hybrid PET/MRI equipment was developed. MRI provides not only high resolution but also high contrast images and the possibility of better tissue characterization with the combination of different pulse sequences. MRI sensitivity in the detection of breast lesions is well established, especially for patients with limitations in conventional mammographic/ultrasound evaluation, such as young women, dense breasts, and multifocal/multicentric lesions. Incorporation of metabolic information by FDG PET can increase the specificity of the method, but care must be taken with false-negative results in cases of small tumors (<1–2 cm) and histologic variations such as tubular carcinoma, well-differentiated, or in situ ductal carcinomas.
PET/MR for breast imaging includes specific position and pulse sequences. PET/MR mammography is acquired in the prone position with the breast hanging and allows better identification of lesions in the breast and regional lymph nodes [25]. MR imaging sequences usually include a T2 fat-suppressed sequence, a T1 non-fat-suppressed sequence, and post-contrast T1 sequences. Even though there is no increase in radiation dose, the additional time spent on a PET/MRI exam should be considered in obtaining additional images. Among the additional MR sequences that may be indicated for FDG PET/MR mammography, diffusion-weighted imaging (DWI) is probably the most useful. Restricted diffusion and a low apparent diffusion coefficient (ADC) are observed in tissues with high cellularity, and this finding may provide prognostic information on breast cancer patients, although false-negative results may occur due to the association of necrotic tissues with aggressive tumors. Prognostic information obtained from DWI and FDG uptake may be incremental, as they reflect different biological properties of the tumor and are not directly correlated, even though when considered as isolated parameters, FDG uptake has a higher prognostic value than ADC values [26, 27].
Whole-body PET/MR is limited in the detection of breast lesions. A PET/MR mammography, with the acquisition of prone breast imaging using a breast coil, has better results in local assessment for determining the local extent of malignant lesions and for surgery planning [28, 29]. Another option is the fusion of breast PET and MR based on software. Post-acquisition fusion of PET/CT and MRI, guided by landmarks, has shown significant increases in specificity and positive predictive value (PPV) for breast lesions, as compared to MR alone [30].
MRI is generally considered to be of low effectiveness in preoperative lymph node evaluation. The same limitations of the PET component described in PET/CT apply to PET/MR. A PET/MR mammography allows better discrimination of lymph nodes than a whole-body study; however, the method is not able to detect minimal infiltrations and therefore does not replace a sentinel lymph node biopsy. Despite the limited sensitivity, there are situations in which a patient with locally advanced disease or during follow-up can benefit from the PET/MR study by detecting the metabolic alteration in lymph nodes that are not enlarged or by detecting previously unsuspected locoregional extra-axillary lymph node metastases [31]. Local staging may be well addressed by hybrid whole body PET/MR, combining the high sensitivity of MR for multifocal disease with the high sensitivity of PET for axillary nodal disease [32].
Regarding distant metastases detection, whole-body PET/MRI combination of structural and functional information in a single study may be valuable [33]. However, it is not yet clear whether there are advantages over performing sequential PET/CT and MRI studies. In the detection of distant metastasis, the MR component of a whole-body PET/MR will improve detection of lesions in the brain, liver, and bones and decrease the sensitivity for small lung lesions as compared to CT.
Low sensitivity of whole-body PET for breast cancer is due to the limited resolution of the method (5–6 mm). Resolution and lesion detectability can be improved using positron emission mammography (PEM). However, limitations arising from histological types with low metabolic activity are only partially resolved with these devices. PEM equipment consists of detectors arranged as plates, which compress the breast and allow the acquisition of images with greater spatial resolution and better sensitivity. There is also the configuration with the breast hanging through an opening in the examination table, with the PET detector ring positioned below. Both systems have limitations for lesions close to the thoracic wall.
PET mammography reports are preferentially described with a standardized interpretation lexicon similar to BI-RADS. Sensitivity and specificity of the method are higher than MR in a direct comparison and can contribute to a reduction in unnecessary biopsies [34]. Even without the CT exposure, administration of FDG implies in a whole-body radiation exposure more than ten times higher than that from a two-view screen film mammography [34]. Also, the technical complexity of the method, including those related to maintaining a nuclear medicine facility, makes it difficult to include PET mammography as a tool for screening or in primary patient evaluation.
Question
Will PET/MR be a suitable equipment for breast cancer screening in the foreseeable future?
4.2 Non-FDG Tracers for Breast Cancer
While 18F-FDG remains the most widely used radiopharmaceutical in PET studies, other tracers available or under development have a great prospect of clinical application in breast cancer imaging. New radiotracers allow a noninvasive method not only for staging but also for assessment of receptor status, metabolic activity, and proliferation [35]. The clinical introduction of these radiopharmaceuticals depends on factors such as local production and availability, clinical validation, and national regulatory agencies approval.
18F-NaF (sodium fluoride) presents high affinity for areas of bone remodeling, determined by the fluoride ion exchange in hydroxyapatite crystals. Its higher and faster uptake, together with the improved resolution of PET in relation to scintigraphy, allows a higher sensitivity than a bone scan, especially for osteoblastic metastasis (Fig. 9.12). However, there is still a need for further cost-benefit analysis before the recommendation to replace the methods [36].
18FMISO and 18FAZA are hypoxia markers that have prognostic value. Hypoxia, in addition to radioresistance, is associated with greater tumor aggressiveness and worse response to treatment, and a boost in hypoxemic tumors may allow optimization of radiotherapy results [37].
18F-FLT (fluorothymidine) is a labeled nucleotide that traces DNA synthesis and correlates with Ki-67 expression, used as an imaging proliferation marker. FLT PET may be used in the assessment of early response to chemotherapy and endocrine therapy [38].
18F-FES (fluoroestradiol) and other receptor tracers
18F-FES is currently used to evaluate the estrogen receptor status in breast cancer patients, with the advantage of a simultaneous evaluation of multiple sites and of sites not accessible to a biopsy. Visual and semiquantitative measures (SUV) in a FES PET/CT can identify patients that will most likely benefit from endocrine therapy [39, 40]. Clinical trials are currently under way to assess the value of FES PET/CT as predictive marker of response to endocrine therapy.
18F-Fluoro Furanyl Norprogesterone (18F-FFNP) is a progesterone receptor tracer, also under evaluation to determine its value as a predictor of response to hormone therapy [41].
89Zr-trastuzumab is one of many different tracers developed to study human epidermal growth factor type 2 (HER2) receptor status. HER2 PET is a possible method to predict response to trastuzumab-based therapy [41, 42].
Question
FES PET can be used to evaluate hormone receptor status. Which lesions should be presumed to respond to hormonal therapy, those with high or with low radiopharmaceutical uptake?
5 Conclusion
18F-FDG PET/CT is indicated for distant metastases detection in patients with advanced breast cancer, particularly when other methods are inconclusive. It is also indicated to detect suspected recurrence. New tracers and equipment may increase the future role of PET imaging in breast cancer patients.
References
Dong A, Wang Y, Lu J, Zuo C. Spectrum of the breast lesions with increased 18F-FDG uptake on PET/CT. Clin Nucl Med. 2016;41:543–57. https://doi.org/10.1097/rlu.0000000000001203.
Rashmi Kumar JB. NCCN Guidelines Version 6.2020 Breast Cancer. Version 6.2020 — September 8, 2020. NCCN Guidelines Version 6.2020 Breast Cancer. Version 6.2020 — September 8, 2020. [cited 2020 Sep 27]. Available at: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf
Groheux D, Cochet A, Humbert O, Alberini J-L, Hindie E, Mankoff D. 18F-FDG PET/CT for staging and restaging of breast cancer. J Nucl Med. 2016;57:17S–26S. https://doi.org/10.2967/jnumed.115.157859.
Ulaner GA. PET/CT for patients with breast cancer: where is the clinical impact? Am J Roentgenol. 2019;213:254–65. https://doi.org/10.2214/ajr.19.21177.
Hortobagyi GN, Connolly JL, D’Orsi CJ, Edge SB, Mittendorf EA, Rugo HS, et al. AJCC cancer staging manual. Breast. 2017;2017:589–636. https://doi.org/10.1007/978-3-319-40618-3_48.
Provenzano E, Ulaner GA, Chin S-F. Molecular classification of breast cancer. PET Clin. 2018;13(3):325–38.
Lebron L, Greenspan D, Pandit-Taskar N. PET imaging of breast cancer. PET Clin. 2015;10:159–95. https://doi.org/10.1016/j.cpet.2014.12.004.
Benveniste AP, Marom EM, Benveniste MF, Mawlawi O, Fox PS, Yang W. Incidental primary breast cancer detected on PET–CT. Breast Cancer Res Treat. 2015;151:261–8. https://doi.org/10.1007/s10549-015-3402-7.
Yararbas U, Avci NC, Yeniay L, Argon AM. The value of 18F-FDG PET/CT imaging in breast cancer staging. Bosn J Basic Med Sci. 2018;18(1):72–9.
Paydary K, Seraj SM, Zadeh MZ, Emamzadehfard S, Shamchi SP, Gholami S, et al. The evolving role of FDG-PET/CT in the diagnosis, staging, and treatment of breast cancer. Mol Imaging Biol. 2019;21:1–10. https://doi.org/10.1007/s11307-018-1181-3.
Park S, Yoon J-K, Lee SJ, Kang SY, Yim H, An Y-S. Prognostic utility of FDG PET/CT and bone scintigraphy in breast cancer patients with bone-only metastasis. Medicine. 2017;96:e8985. https://doi.org/10.1097/md.0000000000008985.
Peterson LM, O’Sullivan J, Wu Q, et al. Prospective study of serial 18F-FDG PET and 18F-fluoride PET to predict time to skeletal-related events, time to progression, and survival in patients with bone-dominant metastatic breast cancer. J Nucl Med. 2018;59:1823–30. https://doi.org/10.2967/jnumed.118.211102.
Aliyev A, Aksoy SY, Özhan M, Ekmekçi̇oğlu Ö, Vatankulu B, Kocael PÇ, et al. The role of FDG PET/CT in detection of distant metastasis in theinitial staging of breast cancer. Turk J Med Sci. 2016;46:349–60. https://doi.org/10.3906/sag-1409-1.
Fink KR, Fink JR. Imaging of brain metastases. Surg Neurol Int. 2013;4(Suppl 4):S209–19.
Galldiks N, Langen K-J, Albert NL, Chamberlain M, Soffietti R, Kim MM, et al. PET imaging in patients with brain metastasis—report of the RANO/PET group. Neuro-Oncology. 2019;21:585–95. https://doi.org/10.1093/neuonc/noz003.
Göktaş İ, Cayvarlı H. The role of F-FDG PET/CT in evaluating elevated levels of tumor markers in breast cancer. Mol Imaging Radionucl Ther. 2018;27(1):3–9.
Piva R, Ticconi F, Ceriani V, Scalorbi F, Fiz F, Capitanio S, et al. Comparative diagnostic accuracy of 18F-FDG PET/CT for breast cancer recurrence. Breast Cancer. 2017;9:461–71. https://doi.org/10.2147/bctt.s111098.
Liu Q, Wang C, Li P, Liu J, Huang G, Song S. The role of18F-FDG PET/CT and MRI in assessing pathological complete response to neoadjuvant chemotherapy in patients with breast cancer: a systematic review and Meta-analysis. Biomed Res Int. 2016;2016:1–10. https://doi.org/10.1155/2016/3746232.
Tian F, Shen G, Deng Y, Diao W, Jia Z. The accuracy of 18F-FDG PET/CT in predicting the pathological response to neoadjuvant chemotherapy in patients with breast cancer: a meta-analysis and systematic review. Eur Radiol. 2017;27:4786–96. https://doi.org/10.1007/s00330-017-4831-y.
Grapin M, Coutant C, Riedinger J-M, Ladoire S, Brunotte F, Cochet A, et al. Combination of breast imaging parameters obtained from 18F-FDG PET and CT scan can improve the prediction of breast-conserving surgery after neoadjuvant chemotherapy in luminal/HER2-negative breast cancer. Eur J Radiol. 2019;113:81–8. https://doi.org/10.1016/j.ejrad.2019.02.005.
Al-Muqbel KM, Yaghan RJ. Effectiveness of 18F-FDG-PET/CT vs bone scintigraphy in treatment response assessment of bone metastases in breast cancer. Medicine. 2016;95:e3753. https://doi.org/10.1097/md.0000000000003753.
Riedl CC, Pinker K, Ulaner GA, Ong LT, Baltzer P, Jochelson MS, et al. Comparison of FDG-PET/CT and contrast-enhanced CT for monitoring therapy response in patients with metastatic breast cancer. Eur J Nucl Med Mol Imaging. 2017;44:1428–37. https://doi.org/10.1007/s00259-017-3703-7.
Pak K, Seok JW, Kim HY, Nguyen TL, Kim K, Kim SJ, et al. Prognostic value of metabolic tumor volume and total lesion glycolysis in breast cancer: a meta-analysis. Nucl Med Commun. 2020;41:824–9. https://doi.org/10.1097/mnm.0000000000001227.
Qu Y-H, Long N, Ran C, Sun J. The correlation of F-FDG PET/CT metabolic parameters, clinicopathological factors, and prognosis in breast cancer. Clin Transl Oncol. 2020;23(3):620–7. https://doi.org/10.1007/s12094-020-02457-w.
Heusner TA, Kuemmel S, Umutlu L, Koeninger A, Freudenberg LS, et al. Breast cancer staging in a single session: whole-body PET/CT mammography. J Nucl Med. 2008;49:1215–22. https://doi.org/10.2967/jnumed.108.052050.
Razek AAKA, Khalek AA, Gaballa G, Denewer A, Nada N. Invasive ductal carcinoma: correlation of apparent diffusion coefficient value with pathological prognostic factors. NMR Biomed. 2010;23:619–23. https://doi.org/10.1002/nbm.1503.
Kitajima K, Yamano T, Fukushima K, Miyoshi Y, Hirota S, Kawanaka Y, et al. Correlation of the SUVmax of FDG-PET and ADC values of diffusion-weighted MR imaging with pathologic prognostic factors in breast carcinoma. Eur J Radiol. 2016;85(5):943–9.
Grueneisen J, Nagarajah J, Buchbender C, Hoffmann O, Schaarschmidt BM, Poeppel T, et al. Positron emission tomography/magnetic resonance imaging for local tumor staging in patients with primary breast cancer. Investig Radiol. 2015;50:505–13. https://doi.org/10.1097/rli.0000000000000197.
Sasaki M, Tozaki M, Kubota K, Murakami W, Yotsumoto D, Sagara Y, et al. Simultaneous whole-body and breast 18F-FDG PET/MRI examinations in patients with breast cancer: a comparison of apparent diffusion coefficients and maximum standardized uptake values. Jpn J Radiol. 2018;36:122–33. https://doi.org/10.1007/s11604-017-0707-y.
Moy L, Noz ME, Maguire GQ Jr, Melsaether A, Deans AE, Murphy-Walcott AD, et al. Role of fusion of prone FDG-PET and magnetic resonance imaging of the breasts in the evaluation of breast cancer. Breast J. 2010;16(4):369–76. https://doi.org/10.1111/j.1524-4741.2010.00927.x.
Moon E-H, Lim ST, Han Y-H, Jeong YJ, Kang Y-H, Jeong H-J, et al. The usefulness of F-18 FDG PET/CT-mammography for preoperative staging of breast cancer: comparison with conventional PET/CT and MR-mammography. Radiol Oncol. 2013;47(4):390–7.
Melsaether A, Moy L. Breast PET/MR imaging. Radiol Clin N Am. 2017;55:579–89. https://doi.org/10.1016/j.rcl.2016.12.011.
Cho I-H, Kong E-J. Potential clinical applications of 18F-Fluorodeoxyglucose positron emission tomography/magnetic resonance mammography in breast cancer. Nucl Med Mol Imaging. 2017;51:217–26. https://doi.org/10.1007/s13139-016-0446-5.
Narayanan D, Berg WA. Use of breast-specific PET scanners and comparison with MR imaging. Magn Reson Imaging Clin N Am. 2018;26(2):265–72.
Ulaner GA, Riedl CC, Dickler MN, Jhaveri K, Pandit-Taskar N, Weber W. Molecular imaging of biomarkers in breast cancer. J Nucl Med. 2016;57:53S–9S. https://doi.org/10.2967/jnumed.115.157909.
Arvola S, Jambor I, Kuisma A, Kemppainen J, Kajander S, Seppänen M, et al. Comparison of standardized uptake values between 99mTc-HDP SPECT/CT and 18F-NaF PET/CT in bone metastases of breast and prostate cancer. EJNMMI Res. 2019;9(1):6. https://doi.org/10.1186/s13550-019-0475-z.
Daimiel I. Insights into hypoxia: non-invasive assessment through imaging modalities and its application in breast cancer. J Breast Cancer. 2019;22:155. https://doi.org/10.4048/jbc.2019.22.e26.
Kostakoglu L, Duan F, Idowu MO, Jolles PR, Bear HD, Muzi M, et al. A phase II study of 3’-Deoxy-3’-18F-Fluorothymidine PET in the assessment of early response of breast cancer to neoadjuvant chemotherapy: results from ACRIN 6688. J Nucl Med. 2015;56:1681–9. https://doi.org/10.2967/jnumed.115.160663.
Linden HM, Stekhova SA, Link JM, Gralow JR, Livingston RB, Ellis GK, et al. Quantitative fluoroestradiol positron emission tomography imaging predicts response to endocrine treatment in breast cancer. J Clin Oncol. 2006;24:2793–9. https://doi.org/10.1200/jco.2005.04.3810.
van Kruchten M, de Vries EG, Brown M, de Vries EF, Andor WJ, Rudi AJ, et al. PET imaging of oestrogen receptors in patients with breast cancer. Lancet Oncol. 2013;14:e465–75. https://doi.org/10.1016/s1470-2045(13)70292-4.
Website. [cited 2020 Aug 15]. Available from: (NCT02398773 and NCT 02455453, available at https://clinicaltrials.gov)
Gebhart G, Lamberts LE, Wimana Z, Garcia C, Emonts P, Ameye L, et al. Molecular imaging as a tool to investigate heterogeneity of advanced HER2-positive breast cancer and to predict patient outcome under trastuzumab emtansine (T-DM1): the ZEPHIR trial. Ann Oncol. 2016;27:619–24. https://doi.org/10.1093/annonc/mdv577.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Sapienza, M.T., Zampieri, P.F. (2022). Nuclear Medicine Based Methods: PET FDG and Other Tracers. In: Kim Hsieh, S.J., Morris, E.A. (eds) Modern Breast Cancer Imaging. Springer, Cham. https://doi.org/10.1007/978-3-030-84546-9_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-84546-9_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-84545-2
Online ISBN: 978-3-030-84546-9
eBook Packages: MedicineMedicine (R0)