Abstract
The disease caused by COVID-19 was declared a pandemic by the World Health Organization on March 11, 2020. Identified for the first time in the city of Wuhan, the Chinese authorities have mandated a quarantine strategy in the city of Wuhan in order to limit the spread of COVID-19 in a wider spectrum. Inspired by this prevention protocol against this new virus, the majority of the countries in the world have also adopted strict quarantine measures to fight against this new coronavirus pneumonia. In this article, we will establish a six-compartment SLIR model taking into account quarantine strategy, in which the dynamics of the COVID-19 epidemic is modeled by a system of six nonlinear differential equations, describing the interactions between susceptible, exposed, infected, and recovered. The basic reproduction number R 0 depending on the quarantine strategy efficacy is calculated. We give the equilibrium points of the system, then we discuss, according to the value of R 0, the global stability of the equilibrium solutions. Numerical simulations are presented in order to validate our theoretical results and we discuss the effectiveness of quarantine measures in the fight against the spread of the pandemic caused by COVID-19.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Mathematical models play an essential role to describe the dynamics of many infectious diseases. The first models usually use three main populations that are the susceptible S(t), the infectious I(t), and the removed individuals R(t) at a specific time t. The basic SIR formulation is introduced in the pioneer work [1]; but, when an individual is incubated but still not yet infectious, another class should be added; this class is called latent compartment noted by L(t). A mutation process was observed in many infections such as tuberculosis [2], human immunodeficiency virus [3], dengue fever [4], influenza [5], and other sexually transmitted diseases. This phenomenon can result in the observation on two or more strains of the studied pathogen. Hence, multi-strain model can better describe different type of diseases.
Recently, two-strain SLIR epidemic model has been tackled [6], the authors consider two incidence rates, the first is bilinear while the second is non-monotonic. More recently, the same problem with two strains is treated by choosing both the incidences as non-monotonic [7]. The generalization of a multi-strain SLIR epidemiological model with general incidence rates is studied in [8]; the authors compare the numerical simulations with COVID-19 clinical data. In this work, we continue the investigation of this last kind of problems by taking into consideration the effect of quarantine measures on SLIR model with two non-monotonic incidence rates. The two-strains SLIR epidemiological model that we consider is formulated as follows:
with
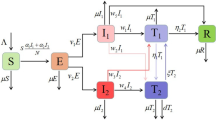
This model contains six variables, that are, susceptible individuals (S), two categories of latent individuals: (L 1) and (L 2), two categories of infectious individuals: (I 1) and (I 2), and removed individuals (R). The parameters of the model (1) are described in Table 1 and the two-strain SLIR diagram is illustrated in Fig. 1; the parameters are given in Table 1. The last two new parameters to the model u 1 and u 2 represent the efficiency of quarantine in reducing the first strain infection and the second strain infection, respectively.
The present work is organized as follows. In the next section, we will prove the positivity and the boundedness results. In Sect. 3, we fulfilled the global analysis of our model. In Sect. 4, we will give some results of the numerical simulations. Short conclusion is given in the last section.
2 Positivity and Boundedness of Solutions
Since our problem is related to the population dynamics, we will prove that all model variables are positive and bounded. First, we will assume that all the parameters in our model are positive.
Proposition 2.1
For any positive initial conditions S(0), L 1(0), L 2(0), I 1(0), I 2(0), R(0), the variables of the model (1) S(t), L 1(t), L 2(t), I 1(t), I 2(t), and R(t) will remain positive for all t > 0.
Proof
First, let
Let us now demonstrate that T = +∞.
Assume that 0 < T < +∞; by continuity, we will have S(T) = 0 or L 1(T) = 0 or L 2(T) = 0 or I 1(T) = 0 or I 2(T) = 0 or R(T) = 0. If S(T) = 0 before the other variables L 1, L 2, I 1, I 2, R, become zero. Therefore
From the first equation of the system (1), we have
If L 1(T) = 0 before S, L 2, I 1, I 2, and R. Then
From the second equation of the system (1) with the fact L 1(T) = 0, which gives
Since u 1 and u 2 reflect the effectiveness of quarantine, we have u i ∈ [0, 1], i = 1, 2. Therefore, α(1 − u 1) and m are positive, and we have
Also, if I 1 = 0 before S, L 1, L 2, I 2, R become zero, then
But from the fourth equation of the system (1) with I 1(T) = 0, we will have
Since γ 1 > 0, we have
Similar proofs for L 2(t), I 2(t), and R(t).
We conclude that T could not be finite; this completes the proof.
Proposition 2.2
The biologically feasible region is represented by
is positively invariant.
Proof
Let the total acting population
By adding all equations in system (1), we will have
therefore,
when t = 0, we will have
Therefore
hence,
Consequently, we conclude that \(\mathcal {H}\) is positively invariant which completes the proof.
3 Analysis of the Model
This section is devoted to the equilibria global stability by using some suitable Lyapunov functionals [9, 10]. Since the first five equations of the system (1) are not dependent of R and since the total population verifies Eq. (2), thus we can omit the sixth equation and the system (1) can be reduced to
with
3.1 The Basic Reproduction Number
It is well known that the basic reproduction number can be defined as the average number of new cases of an infection caused by one infected individual, in a population consisting of susceptible individuals only. We use the next generation matrix FV −1 to calculate the basic reproduction number R 0. Indeed, the formula that gives us the basic reproduction number is R 0 = ρ(FV −1), where ρ stands for the spectral radius, F is the nonnegative matrix of new infection cases, and V is the matrix of the transition infections associated with model (3).

Then,

This implies that
with
and
Denoting
then,
and
3.2 Steady States
The model (3) has four equilibrium points, one called disease-free equilibrium and the others called endemic equilibria given as follows:
-
The disease-free equilibrium \(\mathcal {E}_{f}=\left (\displaystyle {\frac {\Lambda }{\delta }},0,0,0,0\right ).\)
-
The strain 1 endemic equilibrium \(\displaystyle {\mathcal {E}_{s_{1}}=\left (S^{*}_{s_{1}},L^{*}_{1,s_{1}},L^{*}_{2,s_{1}},I^{*}_{1,s_{1}},I^{*}_{2,s_{1}}\right )},\)
where
$$\displaystyle \begin{aligned} &S^{*}_{s_{1}}\; =\displaystyle{\frac{ac}{\alpha (1-u_1)\gamma_{1}}(R^{1}_{0}-\frac{\alpha (1-u_1)}{\delta}I^{*}_{1,s_{1}})}\; , \; L^{*}_{1,s_{1}}=\displaystyle{\frac{c}{\gamma_{1}}I^{*}_{1,s_{1}}} \;,\\ &I^{*}_{1,s_{1}}=\displaystyle{\frac{2 \delta (R^{1}_{0}-1)}{\sqrt{\alpha (1-u_1)^{2}+4m\delta^{2}(R^{1}_{0}-1)}+\alpha (1-u_1)}} \;,\\ &I^{*}_{2,s_{1}}=0 \; ,\; L^{*}_{2,s_{1}}=0 \;. \end{aligned} $$ -
The strain 2 endemic equilibrium \(\displaystyle {\mathcal {E}_{s_{2}}=\left (S^{*}_{s_{2}},L^{*}_{1,s_{2}},L^{*}_{2,s_{2}},I^{*}_{1,s_{2}},I^{*}_{2,s_{2}}\right )},\)
where
$$\displaystyle \begin{aligned} &S^{*}_{s_{2}} \; =\displaystyle{\frac{be}{\beta (1-u_2) \gamma_{2}}(R^{2}_{0}-\frac{\beta (1-u_2)}{\delta}I^{*}_{2,s_{2}} )} \; ,\; L^{*}_{2,s_{2}}=\displaystyle{\frac{e}{\gamma_{2}}I^{*}_{2,s_{2}}}\; , \\ &I^{*}_{2,s_{2}}=\displaystyle{\frac{2 \delta (R^{2}_{0}-1)}{\sqrt{\beta (1-u_2)^{2}+4k\delta^{2}(R^{2}_{0}-1)}+\beta (1-u_2)}} \;, \\ &I^{*}_{1,s_{2}}= 0 \; , \; L^{*}_{1,s_{2}}=0 \;. \end{aligned} $$ -
The endemic equilibrium \(\displaystyle {\mathcal {E}_{t}=\left (S^{*}_{t},L^{*}_{1,t},L^{*}_{2,t},I^{*}_{1,t},I^{*}_{2,t}\right )},\) where
$$\displaystyle \begin{aligned} \qquad \qquad &S^{*}_{t} \;=\displaystyle{\frac{\Lambda}{\delta}(1-\frac{\alpha (1-u_1) I^{*}_{1,t} }{\delta R^{1}_{0}}- \frac{\beta (1-u_2) I^{*}_{2,t}}{\delta R^{2}_{0}})} \;,\\ \qquad \qquad &L^{*}_{1,t}= \displaystyle{\frac{c}{\gamma_{1}}I^{*}_{1,t}} \; , \; L^{*}_{2,t} = \displaystyle{\frac{e}{\gamma_{2}}I^{*}_{2,t}} \; ,\\ \qquad \qquad &I^{*}_{1,t}=\displaystyle{ \sqrt{\frac{1}{m}(R^{1}_{0} \;S^{*}_{t} \frac{\delta}{\Lambda}-1)}} \; , \; I^{*}_{2,t}=\displaystyle{ \sqrt{\frac{1}{k}(R^{2}_{0}\; S^{*}_{t} \frac{\delta}{\Lambda}-1)}} \;. \end{aligned} $$
Remark 3.1
-
(1)
From the components of the equilibrium point \(\mathcal {E}_{s_{1}}\) (respectively, \(\mathcal {E}_{s_{2}}\)), we conclude that this strain 1 endemic equilibrium (respectively strain 2 endemic equilibrium) exists when \(R^{1}_{0}>1\) (respectively, \(R^{2}_{0}>1\)).
-
(2)
From the last equilibrium point \(\mathcal {E}_{t}\) components, we can conclude that this endemic equilibrium exists when \(R^{1}_{0}>1\) and \(R^{2}_{0}>1\).
3.3 Global Stability
Theorem 1
If \(R^{1}_0\leq 1\) and \(R^{2}_0 \leq 1\) . Then the disease-free equilibrium point \(\mathcal {E}_{f}\) is globally asymptotically stable.
Proof
First, we consider the following Lyapunov function in \(\mathbb {R}^{5}_{+}\):
The time derivative is given by
since the arithmetic mean is greater than or equal to the geometric mean, it follows
Therefore when \(R^{1}_{0}\leq 1\) and \( R^{2}_{0}\leq 1\), we will have \(\dot {\mathcal {L}}_f \leq 0,\) then the disease-free equilibrium point \(\mathcal {E}_{f}\) is globally asymptotically stable. In order to establish the global stability of the endemic steady state \(\mathcal {E}_{s_{1}}\), \(\mathcal {E}_{s_{2}}\), and \(\mathcal {E}_{s_{t}}\), we will need the following numbers:
We call R m (respectively R k) the strain 1 inhibitory effect reproduction number (respectively the strain 2 inhibitory effect reproduction number).
Theorem 2
If \( R^{2}_0 \leq 1< R^{1}_0\) and R m ≤ 1. Then the strain 1 endemic equilibrium point \(\mathcal {E}_{s_{1}}\) is globally asymptotically stable.
Proof
First, we consider the Lyapunov function \(\mathcal {L}_1\) defined by
The time derivative is given by
We have
Therefore
Then,
Therefore,
By the relation between arithmetic and geometric means we have
and
If \(R^{2}_0\leq 1\). Then
Since R m ≤ 1, we have that \(m (\frac {\Lambda }{\delta })^{2} \leq 1\)
which implies, \(1-m I_{1}I^{*}_{1,s_{1}}\geq 0\)
Consequently,
We conclude that the point \(\mathcal {E}_{s_{1}}\) is globally asymptotically stable when \(R^{2}_0\leq 1\), \(1<R^{1}_0 \), and R m ≤ 1.
Theorem 3
If \( R^{1}_0 \leq 1< R^{2}_0\) and R k ≤ 1. Then the strain 2 endemic equilibrium point \(\mathcal {E}_{s_{2}}\) is globally asymptotically stable.
Proof
Let us consider the following Lyapunov function:
It easy to verify that
It is easy to see that
We have
Then,
Therefore
The hypothesis (H 2) implies that
Then,
hence,
If \(R^{1}_0\leq 1\). Then
By the relation between arithmetic and geometric means we have
and
Then
We conclude that the point \(\mathcal {E}_{s_{2}}\) is globally asymptotically stable when \( R^{1}_0\leq 1<R^{2}_0 \).
Theorem 4
If \(R^{1}_0 > 1, \ R^{2}_0 > 1\) , R m ≤ 1, and R k ≤ 1. Then the endemic equilibrium point \(\mathcal {E}_{s_{t}}\) is globally asymptotically stable.
Proof
For the proof of this last result it will be enough to consider the following Lyapunov function \(\mathcal {L}_3\):
4 Numerical Simulations
In this section, we will perform some numerical simulations in order to check the impact of quarantine measures in reducing the spread of COVID-19. Indeed, Fig. 2 shows the evolution of infection for λ = 1, α = 0.9, β = 1.45, γ 1 = 0.5, γ 2 = 0.3, μ 1 = 0.15, μ 2 = 0.15, δ = 0.2, m = 1.75, k = 2.85 and different values of u 1 and u 2.
In the case when no quarantine strategy is undertaken, i.e. u 1 = u 2 = 0, we observe that the disease persists and the infected cases stay at very high level. When the effectiveness of the quarantine measures is increased, u 1 = u 2 = 0.25 or u 1 = u 2 = 0.75, a significant reduce of the infection cases is observed; we can also observe a considerable reduce of the latent individuals. Finally, when the quarantine measures are fully established, i.e. u 1 = u 2 = 1, an interesting result is observed. In this last case, the disease dies out, which is represented by the vanishing of all strains infected individuals and also the latent ones. The susceptible individuals will reach in this situation their maximal level. We conclude that the quarantine measures reduce significantly the spread of COVID-19.
5 Conclusion
Modeling epidemiological phenomena makes it possible to better understand several mechanisms that influence the spread of many diseases. In this work, we have studied the effectiveness of quarantine against the spread of COVID-19. Indeed, we have established the problem via six-compartment SLIR model, in which the dynamics of the COVID-19 epidemic is modeled by a system of six nonlinear differential equations, describing the interactions between susceptible, exposed, infected, and healed. First, we have calculated the basic reproduction number depending on the quarantine efficacy. Next, we have given the disease-free equilibrium and three other endemic equilibria, and then we have discussed, according to the value of the basic reproduction number, the global stability of each equilibrium. Numerical simulations are presented in order to discuss the effectiveness of quarantine measures in reducing the spread of COVID-19 pandemic.
References
Kermack WO, McKendrick AG. A contribution to the mathematical theory of epidemics. Proc R Soc London A 1927; 115(772):700–721.
Golub JE, Bur S, Cronin W, Gange S, Baruch N, Comstock G, Chaisson RE. Delayed tuberculosis diagnosis and tuberculosis transmission. Inter J Tuber 2006; 10(1):24–30.
Brenchley JM, Price DA, Schacker TW, Asher TE, Silvestri G, Rao S, Kazzaz Z, Bornstein E, Lambotte O, Altmann D, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nature Medicine 2006; 12(12):1365.
Gubler DJ. Dengue and dengue hemorrhagic fever. Clin Microb Reviews, 1998; 11(3):480–496.
Nuno M, Castillo-Chavez C, Feng Z, Martcheva M. Mathematical models of influenza: the role of cross-immunity, quarantine and age-structure. Math Epi Berlin: Springer; 2008.
Bentaleb D, Amine S. Lyapunov function and global stability for a two-strain SEIR model with bilinear and non-monotone. Inter J Biomath 2019; 12(02):1950021.
Meskaf, A., Khyar, O., Danane, J., Allali, K.: Global stability analysis of a two-strain epidemic model with non-monotone incidence rates. Chaos Sol. Frac. 133, 109647 (2020)
Khyar, O., Allali, K. Global dynamics of a multi-strain SEIR epidemic model with general incidence rates: application to COVID-19 pandemic. Nonlinear Dyn (2020). https://doi.org/10.1007/s11071-020-05929-4.
Korobeinikov A. Global properties of basic virus dynamics models. Bull Math Biol 2004; 66(4):879–883.
Souza MO, Zubelli JP. Global stability for a class of virus models with cytotoxic t lymphocyte immune response and antigenic variation. Bull Math Biol 2011; 73(3):609–625.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Khyar, O., Allali, K. (2021). Dynamic Analysis of SLIR Model Describing the Effectiveness of Quarantine Against the Spread of COVID-19. In: Mondaini, R.P. (eds) Trends in Biomathematics: Chaos and Control in Epidemics, Ecosystems, and Cells. BIOMAT 2020. Springer, Cham. https://doi.org/10.1007/978-3-030-73241-7_15
Download citation
DOI: https://doi.org/10.1007/978-3-030-73241-7_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-73240-0
Online ISBN: 978-3-030-73241-7
eBook Packages: Mathematics and StatisticsMathematics and Statistics (R0)