Abstract
Magnetic resonance imaging (MRI) is an established imaging modality integral to many new cancer management pathways. Amongst the many strengths of MRI are its superior soft tissue contrast resolution, multi-planar capability and the lack of ionising radiation. MRI has been shown to be of high accuracy in primary disease assessment in a multitude of cancer types and locations. It is important to note that many MRI techniques are now feasible and available for response evaluation. MRI has established roles in a multitude of tumour response evaluation setting. However, tumour and MRI sequence-specific parameters and criteria may be necessary for optimal assessment of different clinical scenarios. Besides, with its versatility, advances in hardware and sequence design, a vast number of functional MRI parameters are available, which are at different stages of validation. Standardisation and validation remain a challenge to implementation. The chapter will cover the advantages, limitations and pitfalls of using MRI for treatment response assessment in oncology patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Magnetic resonance imaging (MRI) is an established imaging modality integral to many modern cancer management pathways. Amongst the many strengths of MRI are its superior soft tissue contrast resolution, multi-planar capability and the lack of ionising radiation. MRI is also widely available in many hospitals.
With MRI, the anatomical arrangement of normal and pathological structures, and their tissue properties, are probed by echoes returned from radiofrequency pulse sequences targeted at regions of interest in the body, within magnetic fields in the scanner [1]. Exogenous contrast agents are often used in conjunction [2]. Current clinical MRI examinations utilise multiple pulse sequences to generate images with different weighting to tissue contrast. These images can provide important details of oncological disease status, such as nature and structure of tumours, tumour size, tissue planes, adjacent neuromuscular bundle or organ invasion.
Anatomical coverage of clinical MRI examinations is traditionally limited to individual body parts (such as brain, liver, pelvis). This is as a trade-off for meticulous spatial and contrast resolution and for scan times to be reasonably tolerable for patients. Clinical MRI examinations are, therefore, adept to loco-regional disease assessment and response evaluation.
Functional MRI parameters have recently been enabled by advances in MRI hardware and sequence developments. Diffusion-weighted imaging (DWI) sequences are now widely adopted. Tumour vascularity and perfusion can be assessed with different methods, such as with dynamic contrast enhanced sequences using exogenous contrast agents [3], or arterial spin labelling [4], with MR tagging of blood as endogenous tracer. Tissue hypoxia can be probed with sequences such as blood oxygen level-dependent (BOLD) MRI [5]. Chemical composition within tumours and its microenvironment may be glimpsed by techniques such as MRI spectroscopy or chemical exchange saturation transfer [6, 7]. Functional MRI sequence development continues to be an expanding field of research.
Another area of development has been the expansion to whole body coverage. While this may not be a novel concept, improvement in hardware, such as improved gradient systems, interconnected phase array coils and protocol and sequence designs, such as parallel imaging strategies, have enabled adequate quality images with full body coverage to be attainable within an acceptable scan duration. These have paved ways for the technique to be entering routine clinical use in malignancy with tendency for disseminated lesions, such as in metastatic prostate, breast cancer and in multiple myeloma [8,9,10].
MRI has been shown to be of high accuracy in primary disease assessment in a multitude of cancer type and location, for example, in the brain, head and neck, hepatobiliary, rectum, gynaecological tract and in the prostate, bones and soft tissues [11,12,13,14,15,16,17]. It is intuitive then that MRI would be the natural candidate of choice for response evaluation for these tumours. Depending on the treatment options for the particular clinical context, this may be after the use of ablation, chemotherapy or radiation for loco-regional disease (in neoadjuvant, primary treatment or adjuvant setting), or after systemic treatment in more widespread disease.
It is important to note that while many MRI techniques are now feasible and available for response evaluation, there is much variation in the extent to which these have been validated or adopted into clinical practice. For many indications (e.g. in post-neoadjuvant rectal tumour), use of MRI has been well studied [12, 18, 19]. In other areas (e.g. whole body MRI in multiple myeloma [8]), robust validation of MRI evaluation of treatment response remains work in progress. Some advanced techniques may show potential, but clinical translation may be further away on the horizon.
The rest of the chapter would focus on some of the general concepts underpinning response evaluation with MRI, with particular emphasis on established criteria and DWI.
2 Response Evaluation by Tumour Burden or Anatomical Parameters
Change in tumour burden, as visualised on radiological images, is widely thought of as useful for response evaluation in cancer management. Its acceptance is built on the premise that changes in tumour burden (such as size and number of lesions) can act as surrogate predictors for patients’ survival and quality of life, the ultimate end points of cancer therapeutics.
2.1 WHO and RECIST Criteria
In 1981, the World Health Organization (WHO) first published tumour response criteria [20]. Over the next decades, Response Evaluation Criteria in Solid Tumours (RECIST) was published in 2000 [21], attempting to further standardise measurement of tumour burden on anatomical imaging. This was further validated in cohorts of patients with metastatic cancer undergoing cytotoxic therapy treatment, resulting in the refined criteria published as RECIST 1.1 in 2009 [22].
By providing internationally agreed principles and common languages, they provide standardised frameworks for reporting tumour response, thereby increasing robustness and comparability of clinical trial results from different research centres. This is important for when an ‘objective response’ may be an important end point in determining the success of a phase II clinical trial, or for when a clear definition of disease progression is needed for trials using progression-free survival as an end point, such as in a phase III trial [21].
While the main use of these criteria may be for clinical trials reporting, especially in the metastatic disease context, it is undoubted that many key features have influence on day-to-day clinical oncological imaging practice. Common to the evolving WHO, RECIST 1.0 and 1.1 criteria are:
-
1.
Definitions of measurable lesions
-
2.
Specification of how dimensions should be measured and
-
3.
How changes of these measurements over time should be computed to generate response categories
In RECIST 1.1, multiple strategies are adopted to increase practicality, precision, reproducibility and robustness of response evaluation. For example, defining a minimum size for individual measurable lesion (at least 10 mm for longest dimension of a tumour, 15 mm for short axis measurement of nodes) and summing together of the diameters of target lesions as a single parameter (SPD) to define response category may help to limit impact of individual measurement errors. Limiting the number of lesions, which needs to be longitudinally measured to up to five ‘target lesions’ (selected based on size, perceived reproducibility on repeated measurements and representativeness of all involved organs), reduces the burden to reporters. Clarity of the different response categories with appropriate thresholds increases robustness and reproducibility at the response category level (e.g. Partial response is defined when there has been at least 30% in the SPD of target lesions. Progressive disease can be defined when there is new lesion, or at least 20% increase (and at least 5 mm absolute increase) in the SPD of target lesions compared to baseline, or the smallest SPD, achieved on the study).
MRI may be used under RECIST1.1. It is acknowledged that there are many acquisition variables in MRI, which may impact greatly on image quality, lesion conspicuity and measurement. Rather than specifying exact sequences to be used in any particular MRI study, RECIST1.1 stipulated that scanning sequences should be optimised, and the same scanner and protocol should be matched as closely as possible across imaging time points. Unless for isotropic acquisition, maximal diameter of each target lesion should always be measured on the planes in which a particular MRI sequence is acquired in (not re-formatted).
2.2 iRECIST
With increasing use of immune modulators as anticancer therapy, it has become recognised that some patients may demonstrate unusual pattern of response as per anatomical parameters. These may include a delayed response or even apparent initial increases in tumour burden, which is followed by late, deep and durable responses (pseudoprogression). In light of this, investigators began to apply different modifications to the conventional RECIST, culminating in the RIECIST steering group to propose an additional response evaluation algorithm for use in studies evaluating efficacy of immunotherapies in 2017 (iRECIST) [23, 24].
Many key features, particularly surrounding lesion measurements and summations remain similar to RECIST1.1. A main new change is the requirement of a confirmatory imaging study in 4–8 weeks if there are features of progressive disease on initial response evaluation studies. Termed iUPD (unconfirmed PD) at this initial time point, if there are features reaching threshold of further growth on the following confirmatory study, then disease progression is confirmed (confirmed PD, iCPD). Assuming the patient remains clinically stable, it has been recommended that treatment be continued after iUPD. It is acknowledged that it is challenging to differentiate between pseudo and true progression. The proposed strategy hoped to strike the balance between the undesirable early discontinuation of an effective treatment and equally undesirable continuation of a non-effective therapy. Robust validation of this approach is an on-going effort.
2.3 Other Response Evaluation by Anatomical MRI in Specific Disease Contexts
Comprehensive coverage and detail critique of all the individual scenarios, where MRI can be used for response evaluation, are beyond the scope of this chapter, but some of the more established areas in use in clinical practice are described.
2.3.1 Brain Tumour
Specific challenges in measuring brain tumour burden for response evaluation had led to the community adopting specific response criteria (e.g. that proposed by the Response Assessment in Neuro-Oncology (RANO) working group) [25], such as for high-grade and low-grade gliomas, leptomeningeal metastases and brain metastasis. MRI is an indispensable tool in these.
With irregular shapes of gliomas, bi-dimensional measures are used for measurements of gliomas in RANO criteria, unlike single-dimension measures used in RECIST. Furthermore, how response evaluation of gliomas may be described on different specific MRI sequences has been specified. This is because tumoural changes in different MR sequences may carry different biological significance.
For example, anti-angiogenic agent, such as bevacizumab, is believed to reduce vascular permeability, even if a glioma is biologically progressing. Definition of PD has, therefore, been expanded to include changes such as significant increase of T2/FLAIR signal areas and emergence of new lesion (beyond the more intuitive increased of enhancing tumour area). These are in order to capture patients with non-enhancing but progressing disease. In addition, post-contrast images are not used in response criteria for low-grade glioma, as these tumours do not often show contrast enhancement.
Other key features of the RANO criteria are the incorporation of clinical status and the patient’s reliance on corticosteroid use as part of overall response evaluation criteria (Fig. 3.1). Pseudoprogression can also confound response evaluation of brain tumours, leading to some authors to propose additional immune-related response criteria (irRC) for immunotherapy trials. These adopt similar concepts of iRECIST by specifying need for a confirmatory scan at a later time point to confirm PD.
Glioblastoma multiforme response evaluation. Top row T2 weighted axial images of the brain; bottom row corresponding T1 post-contrast axial images; from right, (c, f)—baseline post debulking surgery; (b, e) 2 months post-radiotherapy and commencement of steroid and temozolomide; (a, d) 5 months post-radiotherapy and commencement of steroid and temozolomide. (c, f) show residual tumour manifesting as enhancing cavitating tumour with intermediate to high surrounding T2 tumour signal. (b, e) show increase in size of enhancing cavitating lesion with new adjacent nodules, and some increase in T2 signal change, in keeping with radiological progression; however, the patient remained stable and well at this stage. (a, d) similar further progressive radiological change, and by this time point, there was parallel clinical deterioration, further supporting progressive disease despite treatment
2.3.2 Hepatocellular Carcinoma (HCC)
HCC is the third most common cause of cancer related death. Liver-directed, loco-regional therapies are commonly used as a stand-alone palliative modality or as a bridge to selected patients for whom liver transplant may be a curative option. These may include thermal ablations, or transarterial embolization with varying agents to include chemotherapy, drug-eluting beads or radioactive spheres. These treatments induce local disease control through tumour necrosis, and it is recognised that there can be poor correlation between the clinical benefit and conventional size-based methods of response assessment. For instance, following adequate radiofrequency ablation, it would be anticipated that the focal area of the ablation zone would be greater than the pre-treatment tumour size.
Modified RECIST (mRECIST) was proposed as an improved tool, which has also been adopted by the European Association for the Study of the Liver (EASL) [26, 27]. Hepatic carcinogenesis is associated with a peculiar vascular derangement, manifesting as arterial enhancement, followed by washout on portal venous and/or delayed phases on imaging including MRI. For a lesion >1 cm and in a high pre-test population cohort (cirrhotic patient), this typical imaging appearance is considered diagnostic of HCC, which in turn also provides a handle for MRI evaluation of response evaluation. One of the key modifications in mRECIST is of taking measurements of only the viable (arterial enhancing) component of tumour for assessment of tumour volume and for summation to compute overall response at a patient level. Necrotic areas are ignored. mRECIST has been validated by studies showing that responders had better survival compared to non-responders to these loco-regional treatments.
Furthermore, the Liver Imaging Reporting and Data System (LI-RADS) lexicon [28], originally proposed by the American College of Radiology, has recently expanded with addition of a treatment response algorithm. This has some similarities and overlaps with mRECIST, including placing emphasis on the arterially enhancing tumour residuum. On the other hand, LI-RADS provides a more detail lexicon of MRI features and is a lesion-by-lesion response assessment tool.
2.3.3 Rectal Cancer
Neoadjuvant chemoradiation (CRT) has become standard treatment for patients with locally advanced rectal cancer. Neoadjuvant treatment aims to downsize and downstage tumours, with an aim to increasing resectability and long-term local disease control. In a minority of patients showing complete response, it is becoming apparent that they may be spared the morbid surgical resection without significantly comprising long-term outcome. Standardised and accurate MRI response evaluation is thus of paramount importance [29].
Change in tumour length is advocated as a useful measure for response evaluation, as a compromise between the measurements on any one imaging plane, which may not be reproducible, given the often complex non-orthogonal planes on which tumour grows, and ultimate 3D volumetry [29] (Fig. 3.2). Treatment effect can also be inferred by reduction of often intermediate T2 signal representing viable tumour and emergence of low-signal fibrotic tissues. This had been used in a semi-quantitative scoring system (MRI tumour regression grade), with a five-point scale of relative proportion of tumour signal and fibrotic signal, which has shown moderate success when validated with pathological tumour regression [30]. In addition, some primary non-mucinous tumour undergo mucinous change with CRT, manifesting as increasing high T2 signal, which is also a form of treatment response [31].
Rectal tumour response evaluation post-neoadjuvant treatment. Top row: post-neoadjuvant treatment MRI; bottom row: pre-treatment scan for comparison; from left to right: sagittal T2 TSE, coronal oblique T2 TSE, b-1000 DWI images, ADC maps. (a, b, e, f) show the primary rectal tumour in different planes pre- and post-treatment, highlighting complex orientation of the tumour. These also show the potential limitation of using single measurement of tumour length as a parameter for response evaluation (significant reduction in tumour bulk on both imaging planes: red arrows in (b, f) and orange arrow in (a, e), but only comparatively modest change in tumour length demonstrated by the orange arrows in (a, e)). Images (g, h) show high DWI signal on b-1000 images and corresponding low ADC value (yellow arrow in h), inferring restricted water diffusion due to increased cellularity pre-treatment. These has improved post treatment (c, d). A persistent low ADC band in (d) may represent a residual fibrotic band when correlated to the same region in image (b)
Furthermore, specific to the neoadjuvant, pre-surgical setting is the need to precisely describe residual tumour extent for assessment of resectability and planning surgical approach. MRI has been shown to be able to predict feasibility of successful sphincter preserving low-rectal cancer surgery, and with lesser success also at predicting outcome of clearance of the mesorectal fascia, an important surgical landmark [30].
3 Response Evaluation with Diffusion-Weighted Imaging (DWI)
Diffusion of water could be probed in vivo by DWI MRI. A basic DWI sequence involves a pair of de-phasing and refocusing pulses. Water molecules, which had moved away of its voxel between these pulses, would lead to incomplete rephasing, and hence signal loss. The degree of signal loss is related to the degree of water motion [32].
The sensitivity of the diffusion sequence to water motion can be varied by changing the parameter known as the b-value (a higher b value may be more sensitive, but at the expense of signal to noise ratio and/or acquisition time) [32]. In clinical practice, DWI MRI is usually performed with at least two b-values (e.g. b = 50 or 0 s/mm2 and other b-values from 0 to 1000 s/mm2), which enable calculation of an apparent diffusion coefficient (ADC), latter often presented in the form of a parametric map.
In biological tissues, diffusion of water molecules is thought to be ‘restricted’ by increased cell membranes and macromolecules [33]. DWI-derived parameter of ‘diffusivity’ can, therefore, act as a surrogate imaging marker of cellularity. This may be exploited for assessment of tumour response to treatment.
Some authors had proposed that DWI may act as a predictor for response to treatment, postulating that hypoxic tumours, which may have areas of necrosis (manifesting as tumours with higher ADC on pre-treatment MRI) would have a poorer response. This has been observed in some studies showing an inverse relationship between pre-treatment ADC values and eventual treatment outcome, for example, in primary colorectal tumour and liver metastases. However, such relationship is not observed consistently across tumour types and contexts [32, 34].
Use of DWI MRI in early and interim response evaluation had shown success in some studies (e.g. in breast cancer, melanoma, head and neck), although more widespread validation remains lacking.
In conventional post-treatment response evaluation settings, many studies have confirmed that responded tumours have an increased ADC, supporting the general notion that cellularity decreases with treatment response [35,36,37,38]. There are, however, important caveats. Some tumour response may be accompanied by reduced ADC, reflecting treatment response with fibrosis and scarring (Fig. 3.2). Response evaluation in lymph nodes is difficult with inherent ‘physiological’ low ADC (high cellularity) (Figs. 3.3, 3.4, and 3.5). Increases in T2 signal of tumour as a result of treatment may result in apparent increase in DWI signal (T2 shine through) though this should be readily appreciable by reviewing the ADC parametric map. Interpretation of bone marrow lesion DWI response may sometimes be difficult because of potential for development of reactive marrow hyperplasia, fatty replacement or fibrosis as a result of post-treatment change, which also influence DWI signal return [39]. Standardisation of techniques remains a challenge, from variations of acquisition techniques to models for ADC calculations. Quantitative measures of DWI (changes in absolute ADC values, volumetry) had been studied with success, but qualitative assessment of change remains the routine mainstay assessment method. Despite advances, it remains the case that DWI is an adjunctive MRI response evaluation tool, and it should be assessed in conjunction with other MRI, clinical and laboratory findings (Fig. 3.3).
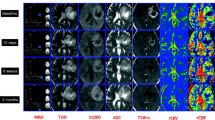
Paediatric lymphoma response evaluation. Whole body MRI with DWI. 3D maximal intensity projection (MIP) of b-800 images. (a) Interim study post two cycles of treatment, (b) baseline image. See also Figs. 3.4 and 3.5. At baseline, there are extensive thoracic lymphadenopathy showing enlargement and increased DWI signal, in keeping with known diagnosis of classical Hodgkin lymphoma (stage 2). Blue arrows on both images show prominent right inguinal lymph nodes, which have high DWI signal but with normal morphology (see Fig. 3.5). These are confirmed benign on FDG PET and ultrasound (not shown). Orange arrows show ‘false-positive’ areas of high DWI signal at the sternum and anterior ends of ribs on the interim study and left anterior ilium of the baseline study; these are thought to be as a result of combination of field inhomogeneity and imperfect fat suppression, as well as respiratory motion on the interim study. This is confirmed by absence of signal abnormalities on other structural sequences (not shown). Dotted green and blue lines correspond to approximate levels of axial images in Figs. 3.4 and 3.5
Paediatric lymphoma response evaluation. Bottom row showing baseline appearances with intermediate T2 signal (middle column) enlarged anterior mediastinal nodes; these demonstrate high DWI signal on b-800 images (left hand column) and corresponding low ADC value (right hand column). There is reduction in size and DWI signal on the interim study (top row)
Paediatric lymphoma response evaluation. Bottom row showing baseline appearances of the inguinal level. Small right inguinal lymph nodes (red arrows) have normal morphology including retained fatty hilum, corroborated to be benign on FDG PET and ultrasound (not shown). These show inherent ‘physiological’ high DWI signal. These remain unchanged on the interim response scan (top row). Green arrows show retracted right testis in the right inguinal canal as an incidental finding
More sophisticated methods of DWI modelling may allow extraction of additional biologically relevant imaging markers. For instance, diffusion tensor imaging (DTI) exploits the directionality of water molecular diffusion and could be used to study tumour microstructural changes with treatment [40]. Intravoxel incoherent motion (IVIM) explores the bulk motion of water flow within tumour microvasculature (as supposed to water diffusion), which may be dominant in low b-values images and may be modelled through bi-exponential decay of DWI signal loss [41, 42]. These are more resource-intensive with need for longer acquisition and post-processing time. Tumour response evaluation with these parameters had been shown to be feasible, but these remain exploratory.
4 Conclusion
With superior soft tissue contrast resolution and widespread availability, MRI has established roles in a multitude of tumour response evaluation setting. Evaluation of anatomical tumour burden change remains the main parameter for response evaluation. Tumour and MRI sequence specific parameters and criteria may be necessary in different clinical scenarios for optimal assessment.
In addition, with its versatility, advances in hardware and sequence design, a vast number of functional MRI parameters are available, which are at different stages of validation. DWI had been described briefly in this chapter, but many exciting developments are beyond the scope of this chapter. Standardisation and validation remain a challenge to implementation.
References
Pooley RA. Fundamental physics of MR imaging. Radiographics. 2005;25(4):1087–99.
American College of Radiology, Committee on Drugs and Contrast Media. ACR manual on contrast media [Internet]. 2015 [cited 2020 July 16]. Available from: http://www.acr.org/~/link.aspx?_id=29C40D1FE0EC4E5EAB6861BD213793E5&_z=z.
Jackson A, O’Connor JPB, Parker GJM, Jayson GC. Imaging tumor vascular heterogeneity and angiogenesis using dynamic contrast-enhanced magnetic resonance imaging. Clin Cancer Res. 2007;13(12):3449–59.
Grade M, Hernandez Tamames JA, Pizzini FB, Achten E, Golay X, Smits M. A neuroradiologist’s guide to arterial spin labeling MRI in clinical practice. Neuroradiology. 2015;57(12):1181–202.
O’Connor JPB, Robinson SP, Waterton JC. Imaging tumour hypoxia with oxygen-enhanced MRI and BOLD MRI. Br J Radiol. 2019;92(1096):20180642.
Horská A, Barker PB. Imaging of brain tumors: MR spectroscopy and metabolic imaging. Neuroimaging Clin N Am. 2010;20(3):293–310.
Wu B, Warnock G, Zaiss M, Lin C, Chen M, Zhou Z, et al. An overview of CEST MRI for non-MR physicists. EJNMMI Phys. 2016;3(1):19.
Messiou C, Hillengass J, Delorme S, Lecouvet FE, Moulopoulos LA, Collins DJ, et al. Guidelines for acquisition, interpretation, and reporting of whole-body MRI in myeloma: myeloma response assessment and diagnosis system (MY-RADS). Radiology. 2019;291(1):5–13.
Padhani AR, Lecouvet FE, Tunariu N, Koh D-M, De Keyzer F, Collins DJ, et al. METastasis reporting and data system for prostate cancer: practical guidelines for acquisition, interpretation, and reporting of whole-body magnetic resonance imaging-based evaluations of multiorgan involvement in advanced prostate cancer. Eur Urol. 2017;71(1):81–92.
Kosmin M, Makris A, Joshi PV, Ah-See M-L, Woolf D, Padhani AR. The addition of whole-body magnetic resonance imaging to body computerised tomography alters treatment decisions in patients with metastatic breast cancer. Eur J Cancer. 2017;77:109–16.
Villanueva-Meyer JE, Mabray MC, Cha S. Current clinical brain tumor imaging. Neurosurgery. 2017;81(3):397–415.
Taylor FGM, Swift RI, Blomqvist L, Brown G. A systematic approach to the interpretation of preoperative staging MRI for rectal cancer. Am J Roentgenol. 2008;191(6):1827–35.
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, et al. PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol. 2016;69(1):16–40.
Kaur H, Hindman NM, Al-Refaie WB, Arif-Tiwari H, Cash BD, Chernyak V, et al. ACR appropriateness criteria® suspected liver metastases. J Am Coll Radiol. 2017;14(5):S314–25.
Becker M, Burkhardt K, Dulguerov P, Allal A. Imaging of the larynx and hypopharynx. Eur J Radiol. 2008;66(3):460–79.
Crombé A, Marcellin P-J, Buy X, Stoeckle E, Brouste V, Italiano A, et al. Soft-tissue sarcomas: assessment of mri features correlating with histologic grade and patient outcome. Radiology. 2019;291(3):710–21.
Balcacer P, Shergill A, Litkouhi B. MRI of cervical cancer with a surgical perspective: staging, prognostic implications and pitfalls. Abdom Radiol N Y. 2019;44(7):2557–71.
Brown G, Kirkham A, Williams GT, Bourne M, Radcliffe AG, Sayman J, et al. High-resolution MRI of the anatomy important in total mesorectal excision of the rectum. Am J Roentgenol. 2004;182(2):431–9.
Lambregts DMJ, Maas M, Stokkel MPM, Beets-Tan RGH. Magnetic resonance imaging and other imaging modalities in diagnostic and tumor response evaluation. Semin Radiat Oncol. 2016;26(3):193–8.
Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981;47(1):207–14.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst. 2000;92(3):12.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017;18(3):e143–52.
Persigehl T, Lennartz S, Schwartz LH. iRECIST: how to do it. Cancer Imaging. 2020;20(1):2.
Chukwueke UN, Wen PY. Use of the response assessment in neuro-oncology (RANO) criteria in clinical trials and clinical practice. CNS Oncol. 2019;8(1):CNS28.
Lencioni R, Llovet J. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010;30(01):052–60.
Galle PR, Forner A, Llovet JM, Mazzaferro V, Piscaglia F, Raoul J-L, et al. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182–236.
Chernyak V, Fowler KJ, Kamaya A, Kielar AZ, Elsayes KM, Bashir MR, et al. Liver imaging reporting and data system (LI-RADS) version 2018: imaging of hepatocellular carcinoma in at-risk patients. Radiology. 2018;289(3):816–30.
Beets-Tan RGH, Lambregts DMJ, Maas M, Bipat S, Barbaro B, Curvo-Semedo L, et al. Magnetic resonance imaging for clinical management of rectal cancer: updated recommendations from the 2016 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol. 2018;28(4):1465–75.
Lambregts DMJ, Boellaard TN, Beets-Tan RGH. Response evaluation after neoadjuvant treatment for rectal cancer using modern MR imaging: a pictorial review. Insights Imaging. 2019;10(1):15.
Kalisz KR, Enzerra MD, Paspulati RM. MRI evaluation of the response of rectal cancer to neoadjuvant chemoradiation therapy. Radiographics. 2019;39(2):538–56.
Heijmen L, Verstappen MCHM, ter Voert EEGW, Punt CJA, Oyen WJG, de Geus-Oei L-F, et al. Tumour response prediction by diffusion-weighted MR imaging: ready for clinical use? Crit Rev Oncol Hematol. 2012;83(2):194–207.
Koh D-M, Collins DJ. Diffusion-weighted MRI in the body: applications and challenges in oncology. Am J Roentgenol. 2007;188(6):1622–35.
Bains LJ, Zweifel M, Thoeny HC. Therapy response with diffusion MRI: an update. Cancer Imaging. 2012;12(2):395–402.
Afaq A. Diffusion-weighted magnetic resonance imaging for tumour response assessment: why, when and how? Cancer Imaging. 2010;10(1A):S179–88.
Li SP, Padhani AR. Tumor response assessments with diffusion and perfusion MRI. J Magn Reson Imaging. 2012;35(4):745–63.
Bonekamp S, Corona-Villalobos CP, Kamel IR. Oncologic applications of diffusion-weighted MRI in the body. J Magn Reson Imaging. 2012;35(2):257–79.
Chavhan GB, Caro-Dominguez P. Diffusion-weighted imaging in pediatric body magnetic resonance imaging. Pediatr Radiol. 2016;46(6):847–57.
Koh D-M, Blackledge M, Padhani AR, Takahara T, Kwee TC, Leach MO, et al. Whole-body diffusion-weighted MRI: tips, tricks, and pitfalls. Am J Roentgenol. 2012;199(2):252–62.
Furman-Haran E, Nissan N, Ricart-Selma V, Martinez-Rubio C, Degani H, Camps-Herrero J. Quantitative evaluation of breast cancer response to neoadjuvant chemotherapy by diffusion tensor imaging: initial results. J Magn Reson Imag JMRI. 2018;47(4):1080–90.
Kato H, Esaki K, Yamaguchi T, Tanaka H, Kajita K, Furui T, et al. Predicting early response to chemoradiotherapy for uterine cervical cancer using intravoxel incoherent motion MR imaging. Magn Reson Med Sci. 2019;18(4):293–8.
Marzi S, Piludu F, Forina C, Sanguineti G, Covello R, Spriano G, et al. Correlation study between intravoxel incoherent motion MRI and dynamic contrast-enhanced MRI in head and neck squamous cell carcinoma: evaluation in primary tumors and metastatic nodes. Magn Reson Imaging. 2017;37:1–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Wan, S. (2021). MRI and Diffusion-Weighted MRI in Treatment Response Evaluation Overview. In: Fanti, S., Gnanasegaran, G., Carrió, I. (eds) Atlas of Clinical PET-CT in Treatment Response Evaluation in Oncology. Springer, Cham. https://doi.org/10.1007/978-3-030-68858-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-68858-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-68857-8
Online ISBN: 978-3-030-68858-5
eBook Packages: MedicineMedicine (R0)