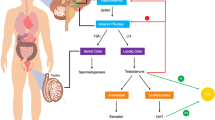
Abstract
Testosterone, the main endogenous active androgen, is used to treat many clinical conditions, such as hypogonadism, infertility, erectile dysfunction, osteoporosis, anemia, and in transgender therapy (female-to-male transsexuals). Androgens are also used by athletes to enhance performance and endurance, and by nonathlete weightlifters or bodybuilders to enhance muscle development and strength. Accordingly, testosterone and other anabolic-androgenic steroids are the main class of appearance and performance enhancing drugs (APEDs), i.e., substances used to improve appearance by building muscle mass or to enhance athletic performance.
Testosterone and other androgens, mainly at supraphysiological levels, affect every single body tissue or system, including the cardiovascular system. Testosterone increases cardiovascular disease risk, causes myocardial infarction, stroke, high blood pressure, blood clots, and heart failure. Among the potential mechanisms whereby testosterone affects the cardiovascular system, both indirect and direct actions have been reported. Indirect actions of testosterone on the cardiovascular system include changes in the lipid profile, insulin sensitivity, and hemostatic mechanisms, modulation of the sympathetic nervous system and renin-angiotensin-aldosterone system. Direct actions of testosterone in the cardiovascular system involves activation of proinflammatory and redox processes, decreased nitric oxide (NO) bioavailability, and stimulation of vasoconstrictor signaling pathways.
This chapter focuses on the effects of androgens, mainly testosterone, on the vascular system. The effects of testosterone on endothelial and vascular smooth muscle cells, as well as mechanisms involved in the effects of testosterone will be reviewed. Effects of testosterone on the perivascular adipose tissue, the immune, sympathetic, and renin-angiotensin systems will also be mentioned.
Similar content being viewed by others
Abbreviations
- AAA:
-
Ascending aortic aneurysms
- ACh:
-
Acetylcholine
- Ang II:
-
Angiotensin II
- APEDs:
-
Appearance and performance enhancing drugs
- AR:
-
Androgen receptor
- ARKO:
-
AR knockout mice
- AT1aR:
-
Angiotensin II type 1A receptor
- AT2R:
-
Angiotensin II type 2 receptor
- Bcl-2:
-
B cell leukemia/lymphoma-2
- BKCa:
-
Large-conductance Ca2+-activated potassium channel
- BSA:
-
Bovine serum albumin
- Ca2+:
-
Calcium ion
- CASMCs:
-
Coronary artery smooth muscle cells
- Cav1.2:
-
L-type voltage-gated Ca2+ channel
- CDP:
-
Collagenase-digestible protein
- CDK:
-
Cyclin-dependent kinase
- CK:
-
Creatine kinase
- COX:
-
Cyclooxygenase
- CSMC:
-
Coronary smooth muscle cell
- DHEA-S:
-
DHEA sulfate
- DHEA:
-
Dehydroepiandrosterone
- DHT:
-
5α-dihydrotestosterone
- ECs:
-
Endothelial cells
- EDCFs:
-
Endothelium-derived contracting factors
- EDHF:
-
Endothelium-derived hyperpolarizing factor
- EDRFs:
-
Endothelium-derived relaxing factors
- EETs:
-
Epoxyeicosatrienoic acids
- eNOS:
-
Endothelial nitric oxide synthase
- ERK1/2:
-
Extracellular signal-regulated kinase 1/2
- Gas6:
-
Growth arrest-specific gene 6
- GPRC6A:
-
GPCR, Class C, group 6, subtype A
- H2O2:
-
Hydrogen peroxide
- HDL:
-
High-density lipoprotein
- HMG-CoA:
-
3-hydroxy 3-methylglutaryl coenzyme A reductase enzyme
- HUVEC:
-
Human umbilical vein endothelial cell
- iNOS:
-
Inducible nitric oxide synthase
- IL-1β:
-
Interleukin-1beta
- IP3:
-
Inositol trisphosphate
- K+:
-
Potassium ion
- Kcnn3:
-
Small conductance calcium-activated potassium channel
- Kv:
-
Voltage-dependent potassium channel
- LDL:
-
Low-density lipoprotein
- LPS:
-
Lipopolysaccharide
- MAPK:
-
Mitogen-activated protein kinase
- NAR:
-
Normal androgen receptor
- NCP:
-
Noncollagen protein
- NF-κB:
-
Nuclear factor-κB
- NLRP3:
-
NOD-, LRR-, and pyrin domain-containing protein 3 inflammasome
- NO:
-
Nitric oxide
- Nox1:
-
Subtype 1 NADPH oxidase
- Nox4:
-
Subtype 4 NADPH oxidase
- ORX:
-
Orchiectomized
- ORXT:
-
ORX treated with testosterone
- Ox-LDL:
-
Oxidized low-density lipoprotein
- PDGF:
-
Platelet-derived growth factor
- PGE2:
-
Prostaglandin E2
- PGF2α:
-
Prostaglandin F2 alpha
- PGI2:
-
Prostacyclin
- PKC:
-
Protein kinase C
- PTOV1:
-
Prostate overexpressed protein 1
- PVAT:
-
Perivascular adipose tissue
- RASMC:
-
Rat aortic smooth muscle cell
- ROS:
-
Reactive oxygen species
- SBP:
-
Systolic blood pressure
- SHR:
-
Spontaneously hypertensive rat
- siRNA:
-
Small interfering RNA
- SK3 channel:
-
Small-conductance calcium-activated potassium channel-3
- SMCs:
-
Smooth muscle cells
- T-BSA:
-
Testosterone-3-carboxymethyl oxime conjugated to bovine serum albumin
- TFM:
-
Testicular feminized male
- THG:
-
Tetrahydrogestrinone
- TNF-α:
-
Tumor necrosis factor-alpha
- TP:
-
Thromboxane-prostanoid
- TxA2:
-
Thromboxane A2
- VCAM-1:
-
Vascular adhesion molecule 1
- VSM:
-
Vascular smooth muscle
- VSMCs:
-
Vascular smooth muscle cells
- WKY:
-
Wistar-Kyoto rat
References
Ajayi AA, Mathur R, Halushka PV (1995) Testosterone increases human platelet thromboxane A2 receptor density and aggregation responses. Circulation 91:2742–2747
Alves MJ, Dos Santos MR, Dias RG et al (2010) Abnormal neurovascular control in anabolic androgenic steroids users. Med Sci Sports Exerc 42:865–871
Alves JV, da Costa RM, Pereira CA et al (2020) Supraphysiological levels of testosterone induce vascular dysfunction via activation of the NLRP3 inflammasome. Front Immunol 11:1647
Baggish AL, Weiner RB, Kanayama G et al (2017) Cardiovascular toxicity of illicit anabolic-androgenic steroid use. Circulation 135:1991–2002
Bahrke MS, Wright JE, Strauss RH et al (1992) Psychological moods and subjectively perceived behavioral and somatic changes accompanying anabolic-androgenic steroid use. Am J Sports Med 20:717–724
Barton M, Prossnitz ER, Meyer MR (2012) Testosterone and secondary hypertension: new pieces to the puzzle. Hypertension 59:1101–1103
Benedit P, Paciucci R, Thomson TM et al (2001) PTOV1, a novel protein overexpressed in prostate cancer containing a new class of protein homology blocks. Oncogene 20:1455–1464
Bhasin S, Woodhouse L, Casaburi R et al (2001) Testosterone dose-response relationships in healthy young men. Am J Physiol Endocrinol Metab 281:E1172–E1181
Bianchi VE (2018) Testosterone, myocardial function, and mortality. Heart Fail Rev 23:773–788
Bowles DK, Maddali KK, Ganjam VK et al (2004) Endogenous testosterone increases L-type Ca2+ channel expression in porcine coronary smooth muscle. Am J Physiol Heart Circ Physiol 287:H2091–H2098
Bowles DK, Maddali KK, Dhulipala VC et al (2007) PKCdelta mediates anti-proliferative, pro-apoptic effects of testosterone on coronary smooth muscle. Am J Physiol Cell Physiol 293:C805–C813
Boydens C, Pauwels B, Van de Voorde J (2016) Effect of resveratrol and orchidectomy on the vasorelaxing influence of perivascular adipose tissue. Heart Vessel 31:608–615
Busse R, Edwards G, Félétou M et al (2002) EDHF: bringing the concepts together. Trends Pharmacol Sci 23:374–380
Bustamante D, Lara H, Belmar J (1989) Changes of norepinephrine levels, tyrosine hydroxylase and dopamine-beta-hydroxylase activities after castration and testosterone treatment in vas deferens of adult rats. Biol Reprod 40:541–548
Campbell WB, Falck JR (2007) Arachidonic acid metabolites as endothelium-derived hyperpolarizing factors. Hypertension 49:590–596
Campelo AE, Cutini PH, Massheimer VL (2012) Cellular actions of testosterone in vascular cells: mechanism independent of aromatization to estradiol. Steroids 77:1033–1040
Carbajal-García A, Reyes-García J, Montaño LM (2020) Androgen effects on the adrenergic system of the vascular, airway, and cardiac myocytes and their relevance in pathological processes. Int J Endocrinol 2020:8849641
Casscells W (1992) Migration of smooth muscle and endothelial cells. Critical events in restenosis. Circulation 86:723–729
Chaytor AT, Martin PE, Edwards DH et al (2001) Gap junctional communication underpins EDHF-type relaxations evoked by ACh in the rat hepatic artery. Am J Physiol Heart Circ Physiol 280:H2441–H2450
Chignalia AZ, Schuldt EZ, Camargo LL et al (2012) Testosterone induces vascular smooth muscle cell migration by NADPH oxidase and c-Src-dependent pathways. Hypertension 59:1263–1271
Chignalia AZ, Oliveira MA, Debbas V et al (2015) Testosterone induces leucocyte migration by NADPH oxidase-driven ROS- and COX2-dependent mechanisms. Clin Sci (Lond) 129:39–48
Chinnathambi V, Balakrishnan M, Ramadoss J et al (2013a) Testosterone alters maternal vascular adaptations: role of the endothelial NO system. Hypertension 61:647–654
Chinnathambi V, Yallampalli C, Sathishkumar K (2013b) Prenatal testosterone induces sex-specific dysfunction in endothelium-dependent relaxation pathways in adult male and female rats. Biol Reprod 89:97
Chinnathambi V, Blesson CS, Vincent KL et al (2014a) Elevated testosterone levels during rat pregnancy cause hypersensitivity to angiotensin II and attenuation of endothelium-dependent vasodilation in uterine arteries. Hypertension 64:405–414
Chinnathambi V, More AS, Hankins GD et al (2014b) Gestational exposure to elevated testosterone levels induces hypertension via heightened vascular angiotensin II type 1 receptor signaling in rats. Biol Reprod 91:6
Crews JK, Khalil RA (1999) Antagonistic effects of 17 beta-estradiol, progesterone, and testosterone on Ca2+ entry mechanisms of coronary vasoconstriction. Arterioscler Thromb Vasc Biol 19:1034–1040
Cruz-Topete D, Dominic P, Stokes KY (2020) Uncovering sex-specific mechanisms of action of testosterone and redox balance. Redox Biol 31:101490
De Smet MA, Lapauw B, De Backer T (2017) Sex steroids in relation to cardiac structure and function in men. Andrologia 49(2). https://doi.org/10.1111/and.12610
Death AK, McGrath KC, Sader MA et al (2004) Dihydrotestosterone promotes vascular cell adhesion molecule-1 expression in male human endothelial cells via a nuclear factor-kappaB-dependent pathway. Endocrinology 145:1889–1897
dos Santos MR, Dias RG, Laterza MC et al (2013) Impaired post exercise heart rate recovery in anabolic steroid users. Int J Sports Med 34:931–935
El Scheich T, Weber A-A, Klee D et al (2013) Adolescent ischemic stroke associated with anabolic steroid and cannabis abuse. J Pediatr Endocrinol Metab 26:161–165
Elagizi A, Köhler TS, Lavie CJ (2018) Testosterone and cardiovascular health. Mayo Clin Proc 93:83–100
Farhat MY, Wolfe R, Vargas R et al (1995) Effect of testosterone treatment on vasoconstrictor response of left anterior descending coronary artery in male and female pigs. J Cardiovasc Pharmacol 25:495–500
Félétou M, Vanhoutte PM (2006) Endothelium-derived hyperpolarizing factor: where are we now? Arterioscler Thromb Vasc Biol 26:1215–1225
Félétou M, Vanhoutte PM (2009) EDHF: an update. Clin Sci (Lond) 117:139–155
Félétou M, Huang Y, Vanhoutte PM (2011) Endothelium-mediated control of vascular tone: COX-1 and COX-2 products. Br J Pharmacol 164:894–912
Fujimoto R, Morimoto I, Morita E et al (1994) Androgen receptors, 5 alpha-reductase activity and androgen-dependent proliferation of vascular smooth muscle cells. J Steroid Biochem Mol Biol 50:169–174
Gonzales RJ, Krause DN, Duckles SP (2004) Testosterone suppresses endothelium-dependent dilation of rat middle cerebral arteries. Am J Physiol Heart Circ Physiol 286:H552–H560
Gonzales RJ, Ghaffari AA, Duckles SP et al (2005) Testosterone treatment increases thromboxane function in rat cerebral arteries. Am J Physiol Heart Circ Physiol 289:H578–H585
Griffith TM (2004) Endothelium-dependent smooth muscle hyperpolarization: do gap junctions provide a unifying hypothesis? Br J Pharmacol 141:881–903
Hashimura K, Sudhir K, Nigro J et al (2005) Androgens stimulate human vascular smooth muscle cell proteoglycan biosynthesis and increase lipoprotein binding. Endocrinology 146:2085–2090
Herman SM, Robinson JT, McCredie RJ et al (1997) Androgen deprivation is associated with enhanced endothelium-dependent dilatation in adult men. Arterioscler Thromb Vasc Biol 17:2004–2009
Herring MJ, Oskui PM, Hale SL et al (2013) Testosterone and the cardiovascular system: a comprehensive review of the basic science literature. J Am Heart Assoc 2:e000271
Higashiura K, Blaney B, Morgan E et al (1996) Inhibition of testosterone 5 alpha-reductase: evidence for tissue-specific regulation of thromboxane A2 receptors. J Pharmacol Exp Ther 279:1386–1391
Higashiura K, Mathur RS, Halushka PV (1997) Gender-related differences in androgen regulation of thromboxane A2 receptors in rat aortic smooth-muscle cells. J Cardiovasc Pharmacol 29:311–315
Hutchison SJ, Sudhir K, Chou TM et al (1997) Testosterone worsens endothelial dysfunction associated with hypercholesterolemia and environmental tobacco smoke exposure in male rabbit aorta. J Am Coll Cardiol 29:800–807
Imig JD (2020) Eicosanoid blood vessel regulation in physiological and pathological states. Clin Sci (Lond) 134:2707–2727
Jones RD, English KM, Jones TH et al (2004) Testosterone-induced coronary vasodilatation occurs via a non-genomic mechanism: evidence of a direct calcium antagonism action. Clin Sci (Lond) 107:149–158
Karanian JW, Ramwell PW (1996) Effect of gender and sex steroids on the contractile response of canine coronary and renal blood vessels. J Cardiovasc Pharmacol 27:312–319
Kumai T, Tanaka M, Watanabe M et al (1994) Possible involvement of androgen in increased norepinephrine synthesis in blood vessels of spontaneously hypertensive rats. Jpn J Pharmacol 66:439–444
Kumar S, Gordon GH, Abbott DH et al (2018) Androgens in maternal vascular and placental function: implications for preeclampsia pathogenesis. Reproduction 156:R155–R167
Lanfranco F, Zitzmann M, Simoni M et al (2004) Serum adiponectin levels in hypogonadal males: influence of testosterone replacement therapy. Clin Endocrinol 60:500–507
Leitman DC, Benson SC, Johnson LK (1984) Glucocorticoids stimulate collagen and noncollagen protein synthesis in cultured vascular smooth muscle cells. J Cell Biol 98:541–549
Ligi I, Grandvuillemin I, Andres V et al (2010) Low birth weight infants and the developmental programming of hypertension: a focus on vascular factors. Semin Perinatol 34:188–192
Ling S, Dai A, Williams MR et al (2002) Testosterone (T) enhances apoptosis-related damage in human vascular endothelial cells. Endocrinology 143:1119–1125
Liu PY, Death AK, Handelsman DJ (2003) Androgens and cardiovascular disease. Endocr Rev 24:313–340
Lopes RA, Neves KB, Pestana CR et al (2014) Testosterone induces apoptosis in vascular smooth muscle cells via extrinsic apoptotic pathway with mitochondria-generated reactive oxygen species involvement. Am J Physiol Heart Circ Physiol 306:H1485–H1494
Maenhaut N, Van de Voorde J (2011) Regulation of vascular tone by adipocytes. BMC Med 9:25
Mäkinen JI, Perheentupa A, Irjala K et al (2011) Endogenous testosterone and brachial artery endothelial function in middle-aged men with symptoms of late-onset hypogonadism. Aging Male 14:237–242
Masuda A, Mathur R, Halushka PV (1991) Testosterone increases thromboxane A2 receptors in cultured rat aortic smooth muscle cells. Circ Res 69:638–643
Matsuda K, Ruff A, Morinelli TA et al (1994) Testosterone increases thromboxane A2 receptor density and responsiveness in rat aortas and platelets. Am J Phys 267:H887–H893
Matsuda K, Mathur RS, Ullian ME et al (1995) Sex steroid regulation of thromboxane A2 receptors in cultured rat aortic smooth muscle cells. Prostaglandins 49:183–196
Matsumoto T, Goulopoulou S, Taguchi K et al (2015) Constrictor prostanoids and uridine adenosine tetraphosphate: vascular mediators and therapeutic targets in hypertension and diabetes. Br J Pharmacol 172:3980–4001
McCredie RJ, McCrohon JA, Turner L et al (1998) Vascular reactivity is impaired in genetic females taking high-dose androgens. J Am Coll Cardiol 32:1331–1335
McCrohon JA, Jessup W, Handelsman DJ et al (1999) Androgen exposure increases human monocyte adhesion to vascular endothelium and endothelial cell expression of vascular cell adhesion molecule-1. Circulation 99:2317–2322
Mishra JS, Hankins GD, Kumar S (2016) Testosterone downregulates angiotensin II type-2 receptor via androgen receptor-mediated ERK1/2 MAP kinase pathway in rat aorta. J Renin-Angiotensin-Aldosterone Syst 17:1470320316674875
Mishra JS, More AS, Hankins GDV et al (2017) Hyperandrogenemia reduces endothelium-derived hyperpolarizing factor-mediated relaxation in mesenteric artery of female rats. Biol Reprod 96:1221–1230
Montezano AC, Touyz RM (2012) Molecular mechanisms of hypertension – reactive oxygen species and antioxidants: a basic science update for the clinician. Can J Cardiol 28:288–295
More AS, Mishra JS, Hankins GD et al (2015) Enalapril normalizes endothelium-derived hyperpolarizing factor-mediated relaxation in mesenteric artery of adult hypertensive rats prenatally exposed to testosterone. Biol Reprod 92:155
Mukherjee TK, Dinh H, Chaudhuri G et al (2002) Testosterone attenuates expression of vascular cell adhesion molecule-1 by conversion to estradiol by aromatase in endothelial cells: implications in atherosclerosis. Proc Natl Acad Sci U S A 99:4055–4060
Müntzing J (1971) Effects of sexual hormones on the adrenergic innervation and the monoamine oxidase activity in the rat ventral prostate. Acta Pharmacol Toxicol (Copenh) 30:208–214
Nakamura Y, Suzuki T, Igarashi K et al (2006) PTOV1: a novel testosterone-induced atherogenic gene in human aorta. J Pathol 209:522–531
Nakao J, Change WC, Murota SI et al (1981) Testosterone inhibits prostacyclin production by rat aortic smooth muscle cells in culture. Atherosclerosis 39:203–209
Naseem SM, Heald FP (1987) Sex mediated lipid metabolism in human aortic smooth muscle cells. Biochem Biophys Res Commun 144:284–291
National Institute on Drug Abuse. http://www.drugabuse.gov
Nheu L, Nazareth L, Xu GY et al (2011) Physiological effects of androgens on human vascular endothelial and smooth muscle cells in culture. Steroids 76:1590–1596
Nishizawa H, Shimomura I, Kishida K et al (2002) Androgens decrease plasma adiponectin, an insulin-sensitizing adipocyte-derived protein. Diabetes 51:2734–2741
Ojeda NB, Royals TP, Black JT et al (2010) Enhanced sensitivity to acute angiotensin II is testosterone dependent in adult male growth-restricted offspring. Am J Physiol Regul Integr Comp Physiol 298:R1421–R1427
Palatini P, Giada F, Garavelli G et al (1996) Cardiovascular effects of anabolic steroids in weight-trained subjects. J Clin Pharmacol 36:1132–1140
Pi M, Parrill AL, Quarles LD (2010) GPRC6A mediates the non-genomic effects of steroids. J Biol Chem 285:39953–39964
Pirompol P, Teekabut V, Weerachatyanukul W et al (2016) Supra-physiological dose of testosterone induces pathological cardiac hypertrophy. J Endocrinol 229:13–23
Pope HG, Kanayama G, Athey A et al (2014) The lifetime prevalence of anabolic-androgenic steroid use and dependence in Americans: current best estimates. Am J Addict Am Acad Psychiatr Alcohol Addict 23:371–377
Porello RA, Dos Santos MR, De Souza FR et al (2018) Neurovascular response during exercise and mental stress in anabolic steroid users. Med Sci Sports Exerc 50:596–602
Quan A, Teoh H, Man RY (1999) Acute exposure to a low level of testosterone impairs relaxation in porcine coronary arteries. Clin Exp Pharmacol Physiol 26:830–832
Rastogi RB, Agarwal RA, Bhargava KP et al (1977) Effect of castration and testosterone treatment on catecholamine metabolism in ventral prostates of normal and chemically sympathectomized rats. Can J Physiol Pharmacol 55:1015–1022
Razmara A, Krause DN, Duckles SP (2005) Testosterone augments endotoxin-mediated cerebrovascular inflammation in male rats. Am J Physiol Heart Circ Physiol 289:H1843–H1850
Ribeiro Júnior RF, Ronconi KS, Jesus ICG et al (2018) Testosterone deficiency prevents left ventricular contractility dysfunction after myocardial infarction. Mol Cell Endocrinol 460:14–23
Ross R (1999) Atherosclerosis – an inflammatory disease. N Engl J Med 340:115–126
Roved J, Westerdahl H, Hasselquist D (2017) Sex differences in immune responses: hormonal effects, antagonistic selection, and evolutionary consequences. Horm Behav 88:95–105
Rowell KO, Hall J, Pugh PJ et al (2009) Testosterone acts as an efficacious vasodilator in isolated human pulmonary arteries and veins: evidence for a biphasic effect at physiological and supra-physiological concentrations. J Endocrinol Investig 32:718–723
Saldanha PA, Cairrão E, Maia CJ et al (2013) Long- and short-term effects of androgens in human umbilical artery smooth muscle. Clin Exp Pharmacol Physiol 40:181–189
Santamarina RD, Besocke AG, Romano LM et al (2008) Ischemic stroke related to anabolic abuse. Clin Neuropharmacol 31:80–85
Schrör K, Morinelli TA, Masuda A et al (1994) Testosterone treatment enhances thromboxane A2 mimetic induced coronary artery vasoconstriction in guinea pigs. Eur J Clin Investig 24(Suppl 1):50–52
Scragg JL, Jones RD, Channer KS et al (2004) Testosterone is a potent inhibitor of L-type Ca(2+) channels. Biochem Biophys Res Commun 318:503–506
Shepherd R, Cheung AS, Pang K et al (2021) Sexual dimorphism in innate immunity: the role of sex hormones and epigenetics. Front Immunol 11:604000
Somjen D, Kohen F, Gayer B et al (2004) Role of putative membrane receptors in the effect of androgens on human vascular cell growth. J Endocrinol 180:97–106
Son BK, Akishita M, Iijima K et al (2010) Androgen receptor-dependent transactivation of growth arrest-specific gene 6 mediates inhibitory effects of testosterone on vascular calcification. J Biol Chem 285:7537–7544
Steroids and Other Appearance and Performance Enhancing Drugs (APEDs) Research Report https://www.drugabuse.gov/publications/research-reports/steroids-other-appearance-performance-enhancing-drugs-apeds/what-are-side-effects-anabolic-steroid-misuse
Tharp DL, Masseau I, Ivey J et al (2009) Endogenous testosterone attenuates neointima formation after moderate coronary balloon injury in male swine. Cardiovasc Res 82:152–160
Toot JD, Reho JJ, Novak J et al (2011) Testosterone increases myogenic reactivity of second-order mesenteric arteries in both defective and normal androgen receptor adult male rats. Gend Med 8:40–52
Tostes RC, Carneiro FS, Carvalho MHC et al (2016) Reactive oxygen species: players in the cardiovascular effects of testosterone. Am J Physiol Regul Integr Comp Physiol 310:R1–R14
Touyz RM, Alves-Lopes R, Rios FJ et al (2018) Vascular smooth muscle contraction in hypertension. Cardiovasc Res 114:529–539
Urhausen A, Albers T, Kindermann W (2004) Are the cardiac effects of anabolic steroid abuse in strength athletes reversible? Heart Br Card Soc 90:496–501
Vanberg P, Atar D (2010) Androgenic anabolic steroid abuse and the cardiovascular system. Handb Exp Pharmacol 195:411–457
Vanhoutte PM, Shimokawa H, Feletou M et al (2017) Endothelial dysfunction and vascular disease – a 30th anniversary update. Acta Physiol (Oxf) 219:22–96
Verdonk K, Danser AH, van Esch JH (2012) Angiotensin II type 2 receptor agonists: where should they be applied? Expert Opin Investig Drugs 21:501–513
Wells KE, Miguel R, Alexander JJ (1996) Sex hormones affect the calcium signaling response of human arterial cells to LDL. J Surg Res 63:64–72
Wilhelmson AS, Fagman JB, Johansson I et al (2016) Increased intimal hyperplasia after vascular injury in male androgen receptor-deficient mice. Endocrinology 157:3915–3923
Williams MR, Ling S, Dawood T et al (2002) Dehydroepiandrosterone inhibits human vascular smooth muscle cell proliferation independent of ARs and ERs. J Clin Endocrinol Metab 87:176–181
Yoo JK, Fu Q (2020) Impact of sex and age on metabolism, sympathetic activity, and hypertension. FASEB J 34:11337–11346
Yue P, Chatterjee K, Beale C et al (1995) Testosterone relaxes rabbit coronary arteries and aorta. Circulation 91:1154–1160
Zhang X, Wang LY, Jiang TY et al (2002) Effects of testosterone and 17-beta-estradiol on TNF-alpha-induced E-selectin and VCAM-1 expression in endothelial cells. Analysis of the underlying receptor pathways. Life Sci 71:15–29
Zhang X, Thatcher SE, Rateri DL et al (2012) Transient exposure of neonatal female mice to testosterone abrogates the sexual dimorphism of abdominal aortic aneurysms. Circ Res 110:e73–e85
Zhang X, Thatcher S, Wu C et al (2015) Castration of male mice prevents the progression of established angiotensin II-induced abdominal aortic aneurysms. J Vasc Surg 61:767–776
Zhou P, Fu L, Pan Z et al (2008) Testosterone deprivation by castration impairs expression of voltage-dependent potassium channels in rat aorta. Eur J Pharmacol 593:87–91
Zhu D, Hadoke PW, Wu J et al (2016) Ablation of the androgen receptor from vascular smooth muscle cells demonstrates a role for testosterone in vascular calcification. Sci Rep 6:24807
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Matsumoto, T., Silva, J.F., Tostes, R.C. (2022). High Testosterone Levels: Impact on the Heart. In: Patel, V.B., Preedy, V.R. (eds) Handbook of Substance Misuse and Addictions. Springer, Cham. https://doi.org/10.1007/978-3-030-67928-6_135-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-67928-6_135-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-67928-6
Online ISBN: 978-3-030-67928-6
eBook Packages: Springer Reference MedicineReference Module Medicine