Abstract
Preference of egg laying in ripe (or in ripening process), healthy, soft, small fruits (berries and stone fruits) still attached to the plant makes Drosophila suzukii (spotted wing Drosophila, SWD) a severe, harmful pest, capable of producing huge food and economic losses to Asia, Europe, and America. The high degree of polyphagy shown by SWD either in their native region, Eastern and Southeastern Asia, or in the invaded continents, besides its seasonal phenotypic plasticity that allows it adaptations to adverse thermal periods, greatly favors SWD establishment and dispersion into new environments. In view of its high-damaging profile and rapid adaptability to settle in different world regions, several authors have focused on the study of biological and ecological features of this invasive pestiferous insect. In this regard, the chapter provides summarized information on life cycle, oviposition preference, larva feeding effects on fruits, crop and non-crop host plants worldwide range, host preference, continental dispersal, seasonal biology, population dynamic, thermal susceptibility, reproductive behavior, interspecific and intergeneric interactions with other frugivorous dipterans, and relationships with natural enemies in native and introduced regions. A deep knowledge of the relationships between SWD and ecological components of newly invaded landscapes is critical for designing effective, environmental-friendly, SWD management strategies.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
4.1 Introduction
Drosophila suzukii (Matsumura) (Diptera: Drosophilidae), commonly known as the spotted wing Drosophila (SWD), is a major invasive global pest of small, soft, and stone fruits. Originally from Eastern and Southeastern Asia, SWD causes significant economic damage to fruit production in invaded regions of Europe and the American continent (Lee et al. 2019). From the earliest records on a specific invaded continent, SWD was characterized by its rapid range expansion within fruit-growing areas (Asplen et al. 2015). Based on this fact, and taking into account the very damaging profile of SWD, several authors have been giving special attention to biological and ecological features of SWD to understand its adaptation and dispersal strategies to invade and settle in different world regions. Thus, this chapter reviews over a large number of publications and provides summarized information on: (1) life cycle, oviposition preference, and feeding effects; (2) crop and non-crop host plants worldwide range, host preference and continental dispersal; (3) seasonal biology and population dynamics; (4) adult and immature stages thermal susceptibility; (5) mating behavior and sex ratio; (6) interspecific and intergeneric interactions with other frugivorous dipterans; and (7) relationships with natural enemies in native and introduced regions.
4.2 Life Cycle, Oviposition Preference, and Feeding Effects
Two exclusive features of SWD make it an economically dangerous pest: its preference for healthy, ripening fruit and the powerful, sclerosed, and serrated ovipositor of the female, which penetrates the fruit epidermis causing physical damage. Frequently, these oviposition wounds provide access to secondary fruit-feeding organisms, both insects (i.e., other frugivorous drosophilids) and pathogens (fungi and bacteria), which together cause additional losses (Walsh et al. 2011). Eggs develop into larvae within the fruit mesocarp, causing its rapid softening and rotting, bringing as consequence enormous yield losses, with significantly negative economic impact. Nevertheless, SWD may also lay eggs in wounded and/or fermenting fruit (Kienzle et al. 2020). The SWD may thrive well in fallen fruit, when the preferred fruit stages are unavailable or scarce (Bal et al. 2017; Kienzle et al. 2020). As a result, SWD individuals can emerge from eggs laid on berries either intact or injured (Mazzetto et al. 2020; Kienzle et al. 2020).
SWD females lay eggs with an erratic distribution pattern across fruits, which can be explained by random visits of females to fruits and the production of clutches of mostly single eggs (Mitsui et al. 2006; Schlesener et al. 2017). SWD females tend to oviposit at softer parts of the fruit, which imply that they examine fruits very carefully to locate the most suitable parts for egg laying (Kinjo et al. 2014). Tests carried out with different Vaccinium spp. cultivars showed more egg laying in softer fruit blueberry varieties than in firmer fruit varieties (Kinjo et al. 2013). A SWD female may lay 20–419 eggs in a lifetime, depending on the environmental conditions (Hamby et al. 2016). Once the larva hatched, it feeds inside fruit pulp. SWD larvae develop through three instars; the mature larva tends mainly to migrate out of the fruit to pupate in the soil (Woltz and Lee 2017; Lee et al. 2019). A total of 13–14 days is needed for the fly to develop from egg to adult at 22 °C (egg stage duration 1.4 day, larval stage 6 days, and pupal stage 6 days) (Tochen et al. 2014). Adults of both summer and winter morphotypes may live up to 30–179 days in the lab when provided food depending on temperature (Shearer et al. 2016; Rendon et al. 2019; Stockton et al. 2019). Females have a short pre-ovipositional period, during which they mostly feed. After first mate, 1- to 5-day-old females start laying eggs under standard lab conditions (Hamby et al. 2016).
4.3 Host Plants and Continental Dispersal
The SWD has a broad crop and non-crop host plants range, mainly throughout Asia, Europe, and America (Asplen et al. 2015), and with potential for adaptation and establishment in Oceania and Africa (Dos Santos et al. 2017). Soft-skinned and stone fruits such as berries and cherries are highly susceptible to infestation (Mitsui et al. 2010; Cini et al. 2012; Bellamy et al. 2013; Burrack et al. 2013; Lee et al. 2015). In addition to cultivated host species, the SWD can develop in both native and exotic fruit of wild and ornamental non-crop hosts. This occurs in various habitats, such as forests, forest edges, meadows, hedges of agricultural areas, urban gardens and parks, whereby this insect may switch among host plants and environments when ripe fruits are available (Lee et al. 2015; Asplen et al. 2015; Kenis et al. 2016). This feature, besides other biological factors, greatly facilitates its dispersion in invaded fruit-growing regions (Lee et al. 2011a, b, 2012; Cini et al. 2012, 2014; Kenis et al. 2016).
4.3.1 Asia
A total of 58 host plant species, belonging to 11 families, were recorded in Asia (Table 4.1). This complete list includes crop and non-crop fruit species from southeastern Asia, including China, Japan, South Korea (the region where SWD is native), and Turkey. Four species were cited only at the generic level, such as Prunus sp., Rubus sp., Morus sp., and Vaccinium sp. About 16% of the host plant species are cultivated, while the majority are non-crop fruits, upon which SWD multiplies. Most of the SWD host plant species belongs to the Rosaceae (53.4%), in which 55% and 26% belong to Prunus L. and Rubus L., respectively. All of the host plant species cited for Asia had naturally SWD-infested fruits (Table 4.1). Additionally, two of them, Prunus donarium Siebold (Mitsui et al. 2006) and P. avium L. (Kasuya et al. 2013), were infested under lab conditions as well. The SWD has mainly caused economic damage in cherry (Prunus tomentosa Thunb. and P. avium), blueberry (Vaccinium spp.), wax-myrtle (Myrica rubra Sieb. Et Zucc.), and autumn olive (E. umbellate Thunberg) crops (Asplen et al. 2015).
4.3.2 Europe
From the first report of D. suzukii in Europe during 2008 in both Spain (Calabria et al. 2012) and Italy (Cini et al. 2012), the SWD widely spread in 6 years across the continent (Asplen et al. 2015). Severe damage in several small fruit crops, such as sweet cherry (Prunus avium), sour cherry (Prunus cerasus L.), strawberry (Fragaria x ananassa Duch.), raspberry (Rubus idaeus L.), blackberry (Rubus fruticosus aggr.), and blueberry (Vaccinium spp.), was mainly recorded at numerous European locations (Cini et al. 2012; Weydert and Mandrin 2013; Asplen et al. 2015; Weydert et al. 2016; Mazzi et al. 2017). SWD infestations have also been reported in cultivated peach (Prunus persica (L.) Stokes), apricot (Prunus armeniaca L.), plum (Prunus domestica L.), apple (Malus domestica (L.) Borkh.), and fig (Ficus carica L.), although without economically significant damage (Grassi et al. 2011; Weydert and Mandrin 2013; Asplen et al. 2015; Kenis et al. 2016). Similarly, infestations in grape (Vitis vinefera L.) varieties were recorded in both Italy (Grassi et al. 2011; Cini et al. 2012; Kenis et al. 2016) and Germany (Asplen et al. 2015). Although grape cultivars with soft-skinned berries are more susceptible for egg laying (Mazzetto et al. 2020), successful SWD adult development is limited (Asplen et al. 2015). Late-ripening grape varieties were mainly infested by SWD in Italy, probably because of an increase in the number of flies and/or the reduction of alternative hosts (Mazzetto et al. 2020). In fact, surrounding wild vegetation suitable for SWD oviposition increased the capture of flies inside vineyards as well as in other crops (Poyet et al. 2015; Kenis et al. 2016; Mazzetto et al. 2020).
In Europe, 126 species of crop and non-crops fruits belonging to 27 families were recorded as SWD hosts (Table 4.2). From a total of SWD host species cited in Table 4.2, 91 were found producing natural infestations, while the remaining 35 were infested only under lab conditions. However, in 13 of these lab-infested fruit species, D. suzukii laid eggs, but there was no adult emergence (Table 4.2). The highest percentage of host species (41%) belongs to the Rosaceae family, with Prunus as the genus with more host species (24%), followed by Cotoneaster Medik. (14%) and Rubus (12%). Caprifoliaceae, Ericaceae, Solanaceae, Adoxaceae, and Cornaceae also have numerous SWD host species (Table 4.2). Several non-crop host plants such as Rubus spp., Sambucus spp., Prunus spp., Lonicera spp., Arbutus unedo L., and Frangula alnus Mill. (Table 4.2) have been constantly found throughout Europe with high natural infestation rates, and SWD switch between these hosts depending on the fruit seasonality (Arnó et al. 2012, 2016; Asplen et al. 2015; Poyet et al. 2015). Those host plants could be potential resources for SWD population increase in Europe (Asplen et al. 2015; Arnó et al. 2016; Kenis et al. 2016). Green fruits of the wild host Rubus ulmifolius Schott collected at an altitude of 1273 m in Spain were highly infested (Arnó et al. 2016), which shows that SWD can be found within a broad range of altitude.
4.3.3 North America
SWD was first recorded on berry crops in California (USA) in 2008 and from that year it was dispersed through the soft and stone fruits-producing regions of mainland United States and Canada (Table 4.3) (Asplen et al. 2015). The most important economically affected crops in these countries include blueberries, raspberries, blackberries, strawberries, and cherries (Lee et al. 2011a, b; Bellamy et al. 2013). In 2011, SWD was detected in traps in Mexico (García-Cancino et al. 2015), but then it was found infesting Psidium guajava L. (Myrtaceae) crops (Lasa et al. 2017). A total of 85 crop and non-crop fruit species belonging to 21 families were recorded as SWD hosts in North America (Canada, United States, and Mexico) (Table 4.3). Four host plant species were only recorded at the generic level, e.g., Sambucus sp., Lonicera sp., Morus sp., and Rubus sp. The 93% of recorded SWD host plant species was naturally infested, while six fruit species were infested only under lab conditions. Most SWD host species are non-crop fruits, while 23% are cultivated species. The main SWD host plant family is Rosaceae, which includes 27% of the total fruit species recorded as hosts. Both Prunus and Rubus species represented 39% and 31%, respectively, of all Rosaceae species. Other important plant families with numerous SWD host species are Caprifoliaceae, Cornaceae, Adoxaceae, and Ericaceae (Table 4.3).
4.3.4 South America
SWD was first recorded in Brazil in 2013 (Deprá et al. 2014), and then in other South American countries, such as Uruguay (Gonzales et al. 2015), Argentina (Santadino et al. 2015), and Chile (Morales 2020). In 7 years, 31 crop and non-crop fruit species belonging to 10 families were recorded as SWD hosts in South America (Table 4.4). Two host plant species were only recorded at the generic level, such as Butia sp. and Rubus sp. All host fruit species recorded in Table 4.4 were naturally infested by SWD. Most SWD host species (58%) are crop fruits of economic importance. Damage caused by SWD in Rubus idaeus L., R. fruticosus, R. ulmifolius Schott, Vaccinium corymbosum L., V. myrtillus L., and Fragaria x ananassa has been reported for Argentina, Brazil, Chile, and Uruguay (Table 4.4). All of these fruit species are primary SWD hosts (Deprá et al. 2014; Andreazza et al. 2016a; Lauyé 2017; Funes et al. 2018a, b; Bezerra Da Silva et al. 2019; SAG Servicio Agrícola y Ganadero 2019; Wollmann et al. 2016, 2019; Morales 2020).
Other cultivated plant species, such as Malus domestica L., Prunus persica L., Prunus cerasus L., Pyrus communis L., Citrus sinensis (L.) Osbeck, Carica papaya L., Psidium guajava L., Ficus carica L., and Vitis vinifera L. can be used by SWD as secondary hosts or reservoirs (Nunes et al. 2014; Oliveira et al. 2015; Geisler et al. 2015; Andreazza et al. 2015, 2016c, 2017; Borba et al. 2016; Lauyé 2017; Junior et al. 2018; Morales 2020). However, at least for some apple (Oliveira et al. 2015) and peach (Andreazza et al. 2017) cultivars, SWD females showed low oviposition frequencies, without economic damage in the fruit. Interestingly, V. vinifera varietal susceptibility studies carried out under lab conditions in Brazil showed that only some cultivars were vulnerable to SWD attack (Andreazza et al. 2016a). In field studies, the SWD has showed preference for different species within the genus Rubus, being R. idaeus much more attractive to SWD than R. fruticosus (Funes et al. 2018a). Furthermore, preference for determined R. fruticosus varieties has been detected for other frugivorous dipeterans (Funes et al. 2017), which could also be the case for the SWD.
Rosaceae is the plant family with the most SWD host species (45.2%) followed by Myrtaceae (23%) and Ericaceae (10%) (Table 4.4). Wild myrtaceous species (Table 4.4) are particularly alternative SWD hosts in South America (Müller and Nava 2014; Geisler et al. 2015; Alexandre 2016; Andreazza et al. 2015, 2016c; Lauyé 2017; SAG Servicio Agrícola y Ganadero 2019; Morales 2020). SWD pupae were usually recovered from healthy fruit, but there were some exceptions, e.g. SWD pupae were also found from damaged, over-ripen, or decayed Vaccinium ashei Reade fruit (Gonzales et al. 2015) or P. guajava fruit (Escobar et al. 2018), and from Carica papaya rotting fruit on the ground (Junior et al. 2018).
4.3.5 Africa and Oceania
SWD’s potential for further invasions to Africa and Oceania is predicted, due to the environmental suitability of these areas for this species (Dos Santos et al. 2017). In Africa, SWD has been found on berry crops since 2013 in Morocco (North Africa) and on Reunion Island (EPPO European and Mediterranean Plant Protection Organization 2019). Only three crop fruit species were recorded as hosts of the SWD in this continent (Table 4.5). In Oceania, SWD was introduced into Hawaii in the 1980s (Kaneshiro 1983), and recorded in traps in the French Polynesia since 2017 (EPPO European and Mediterranean Plant Protection Organization 2019). Only two crop and two non-crop fruit species were cited as SWD hosts in Hawaii (Table 4.5).
4.4 Seasonal Biology and Population Dynamics
In temperate climate regions , such as Asia, Europe, and North America, adult SWD populations markedly decrease from winter onward (Kinjo et al. 2014; Wang et al. 2016a, b, c; Grassi et al. 2018). In Europe, SWD adult populations frequently reach peaks in spring and autumn, to decline again in the early winter (Weydert and Mandrin 2013; Asplen et al. 2015; Mazzetto et al. 2015; Zerulla et al. 2015; Arnó et al. 2016; Rossi-Stacconi et al. 2016; Zengin and Karaca 2019). This population dynamic is too similar to that reported in California, USA (Harris et al. 2014; Wiman et al. 2014; Wang et al. 2016a, b, c) where SWD populations increase in early spring, decrease during the hottest months of the year but increase again in autumn. Thus, cherries are usually the first commercial fruits available to SWD in Europe (Grassi et al. 2011; Cini et al. 2012; Poyet et al. 2015; Kenis et al. 2016; Mazzi et al. 2017). Damages to cherry crops during spring can be mainly explained by two SWD’s biological abilities. Firstly, SWD adults can adapt to seasonal climate changes and survive harsh conditions through physiological and morphological adaptations (Shearer et al. 2016). These winter adaptations involve: (1) a reproductive diapause in which females have undeveloped ovaries and males produce very few sperm (Zerulla et al. 2015; Rossi-Stacconi et al. 2016; Shearer et al. 2016; Grassi et al. 2018), and (2) a phenotypic plasticity that allows a specific seasonal morphological change, which produces differences between a winter morphotype fly and a summer morphotype fly. The former has higher cold tolerance (10–15°C), darker pigmentation, and a larger body and longer wings than the latter (Asplen et al. 2015; Shearer et al. 2016; Fraimout et al. 2018; Stockton et al. 2018). Seasonal morphologies of SWD adults are unalterable, whereas reproductive dormancy is not; females can develop mature eggs when weather conditions are favorable (Rossi-Stacconi et al. 2016; Wallingford et al. 2016). Therefore, winter morphotype females that mated in autumn and overwintered may have mature eggs in early spring to infest the earliest ripening cherries and any other suitable non-crop fruits occurring at the same time (Panel et al. 2018), which facilitate the development of the summer morphotype SWD first generation, increasing pest population in late spring (Panel et al. 2018). Secondly, winter SWD survivors can use several early spring fruiting non-crop host plants for food and reproduction (Kenis et al. 2016; Grassi et al. 2018). Cases in point as Viscum album L. in Germany (Briem et al. 2016), Hedera helix L. in Italy (Grassi et al. 2018), and Aucuba japonica Thnb. in The Netherlands (Panel et al. 2018) are believed to be the earliest SWD reproductive hosts in early spring. Even though fitness of SWD adults emerging from these non-crop host species is poor, they could potentially attack the first available commercial crops (Grassi et al. 2018; Panel et al. 2018).
In subtropical climate regions like southern Brazil, a fruit-growing area invaded by SWD quite recently, SWD pattern of occurrence and population peaks were similar to those observed in berry-producing areas of the USA in late spring and mid-autumn (Wollmann et al. 2019). Probably, the growth of SWD population in autumn was determined by the occurrence of cooler conditions than in summer, with temperatures within a similar range than that of the fly’s native region (Deprá et al. 2014). The gradual temperature increase in spring favored not only SWD activity but also triggered the onset of the fruiting season for both, commercial berry crops and non-crop fruits, such as strawberry guava (Psidium cattleianum Sabine) and Surinam cherry (Eugenia uniflora L) in southern Brazil (Wollmann et al. 2019). Although some SWD population peaks occurred during the spring-summer period, a significant decrease in the pest activity was detected at temperatures near 30°C, which are considered limitant for SWD oviposition (Wiman et al. 2014). In southern Brazil, SWD can survive at low population levels in alternative hosts such as loquat (Eriobotrya japonica Thunb.) even in winter, with thermal variations between 9 and 10.7°C (Wollmann et al. 2019). Alike in North America and Europe, SWD winter morphotypes were found in subtropical regions of South America, such as southern Brazil, demonstrating that seasonal phenotypic plasticity allows SWD to remain in the same habitat year after year. Similar SWD seasonal activity was found in blueberry crops in northwestern Argentina, another subtropical, South American fruit-growing region. High population peaks were detected in late spring, but the permanence of SWD in blueberry orchards was verified throughout the year, regardless of the presence of blueberry fruit in the area (Funes et al. 2018a). During the fruitless period, from early summer to mid-winter, there were low levels of SWD adult catches in traps (Funes et al. 2018b), which would indicate that SWD females use alternative host fruits, such as P. guajava, in wild vegetation areas between mid-summer and late-autumn (Escobar et al. 2018). SWD winter morphotypes were caught in peach crops in fruit-producing areas of the central-eastern region of Argentina as well (Gonsebatt et al. 2017).
4.5 Thermal Susceptibility
Seasonal temperature variations affect SWD population abundance (Wiman et al. 2014; Zengin and Karaca 2019), as well as daily temperature variations influence SWD adult dispersal between different habitats (Tait et al. 2020). In addition, temperature substantially influences SWD developmental, survival, and reproduction periods (Tochen et al. 2014). High temperatures would be responsible for SWD population summer decline in several temperate and subtropical regions (Arnó et al. 2016; Wollmann et al. 2019). SWD’s activities are null or reduced when temperatures are out of the 10°–31°C range (Kimura 2004; Kinjo et al. 2014; Tochen et al. 2014; Zerulla et al. 2015; Rossi-Stacconi et al. 2016; Grassi et al. 2018). Although SWD adult detection is often difficult in Europe’s and North America’s winter, winter trapping has caught wild SWD individuals despite temperatures below 0°C (Hamby et al. 2016; Rossi-Stacconi et al. 2016; Thistlewood et al. 2018). This is a clear evidence of SWD’s ability to overwinter locally (Rossi-Stacconi et al. 2016; Grassi et al. 2018; Stockton et al. 2018). Thus, these SWD winter morphotype adaptive features allow adult insects to absorb and retain heat throughout the winter (Kimura 1988; Tonina et al. 2016). Furthermore, in contrast with summer morphotypes, winter morphotypes may reduce water loss, improve immune function, and extend their longevity at cold temperatures (Stephens et al. 2015; Shearer et al. 2016; Toxopeus et al. 2016; Wallingford et al. 2016) through physiological changes that prevent ice crystals formation within the hemocoel, and fly death (Lee 1991; Shearer et al. 2016; Toxopeus et al. 2016).
Several studies on SWD thermal tolerance have used static and/or dynamic acclimation protocols in order to learn more about this biological capacity, taking into account closer approximations to natural temperature changes (Kimura 2004; Dalton et al. 2011; Kellermann et al. 2012; Jakobs et al. 2015; Stephens et al. 2015; Shearer et al. 2016; Wallingford et al. 2016; Stockton et al. 2018). In the static acclimation procedure, SWD individuals are kept at a single constant cool temperature for the duration of the acclimation period prior to testing, while a dynamic acclimation process involves gradually subjecting the insect to cold hardening temperatures (Stockton et al. 2018). Static acclimation procedure induces cold tolerance in SWD adults, which survived to temperatures below 0°C for up to 1 h and reached a lower lethal limit at −7.5°C (Jakobs et al. 2015). However, dynamic acclimation significantly improved SWD winter morphotype adults’ survival since the survival rate was 50% after 72 h at −7.5°C (Stockton et al. 2018). Therefore, both acclimation and a suitable overwintering site may allow the survival of small founder populations of SWD winter morphotypes in the spring (Wallingford et al. 2016). Some models of D. suzukii potential distribution relate temperature with pest spread (Dos Santos et al. 2017). According to these models, annual mean temperature, maximum temperature of the warmest month (values >33°C), mean temperature of the coldest quarter (values <−10°C), and annual precipitation are the environmental variables with more influence on SWD distribution. Consequently, SWD potential geographical distribution range includes world subtropical regions with high rainfall throughout the year or during part of it.
There are no records on SWD overwintering as larva or pupa. Both immature stages have lesser cold tolerance compared to adults, and show little survival at temperatures below 5°C (Dalton et al. 2011; Stephens et al. 2015; Aly et al. 2017; Enriquez and Colinet 2017). Pupal survival drops noticeably below 0°C regardless of the dynamic acclimation procedure (Stockton et al. 2018).
Temperature is crucial for SWD development and establishment. According to studies carried out by Schlesener et al. (2020) in Brazil, for egg-to-adult survival, the lowest thermal threshold was 7.8 °C and optimum temperature was 23–25 °C. No SWD emergence occurred at 30–33 °C. The shortest egg-to-adult development time (10 days) was observed at 25–28 °C. The intrinsic rate of population increase was the highest at 23–25 °C. SWD body size is strongly affected by temperature, with 13°C and 28 °C causing the largest and the smallest body sizes, respectively. The annual number of generations ranged from 17.1 to 27.2 in cold and warm regions, respectively.
4.6 Reproduction
A high reproductive potential is one of the strongest reasons for the rapid worldwide spread of the polyphagous SWD . Courtship in drosophilids might involve aerial vibrations, visual displays, substrate-borne vibrations, and sex pheromones (Hamby et al. 2016). SWD sexual behavior is characterized by distinct facts of male courtship leading to female acceptance for mating. Furthermore, it has been reported that time of day and male age modulate D. suzukii mating activity (Revadi et al. 2015). The SWD displays characteristic courtship behavior, but to what extent the different sensory modalities are involved remains unclear. The importance of substrate-borne vibrations produced during male courtship has been demonstrated (Mazzoni et al. 2013), but the role of pheromonal and visual components in courtship remains unknown. Unlike most other drosophilids (e.g., Drosophila melanogaster Meigen), D. suzukii does not produce the male pheromone cis-vaccenyl acetate (Dekker et al. 2015). In this direction, female pheromones such as cuticular hydrocarbons (CHCs) were also studied in SWD since female sexual maturity is accompanied by a quantitative increase in these olfactory signals (Revadi et al. 2015). Despite of having been shown that CHCs were not needed to induce male courtship in D. suzukii, it turned out that these pheromones significantly increased male sexual behavior and that interfering in their naturally occurring ratios disrupts mate recognition (Revadi et al. 2015; Snellings et al. 2018).
Sex ratios are among the most basic of demographic parameters and provide an indication of both the relative survival of females and males and the future breeding potential of a population (Skalski et al. 2005). The production of males and females in a 1:1 ratio is usually the most prevalent evolutionary steady strategy, governed by frequency-dependent natural selection owing to competition for mates among individuals of the same sex (Sapir et al. 2008). SWD sex ratio is subjected to variations, depending on several factors. In SWD, sex ratio fluctuations throughout the year seem to be very conservative regardless the plant environment within the same region, but not between regions. In Italy (Gargani et al. 2015), in a trapping net implemented from January to December, sex ratio fluctuation patterns over time were almost the same in a vineyard than in a botanical garden, in Siena. However, in another location (Gosseto), the sex ratio fluctuation curve was different to that of Sienna, in three orchards surveyed (vineyard, apricot, and blueberry). Nevertheless, sex ratio fluctuation patterns were very similar among the three fruit crops. Besides, the sex ratios (expressed as percentage of females) varied between 0 and 50 in Siena, and from 0 to 100 in Grosseto. The highest sex ratios (biased to females) occurred at different seasons, depending on the location (winter in Siena, autumn in Grosseto). The lowest sex ratios (biased to males) were registered in spring. There was not SWD catches in none of the two locations in summer.
SWD sex ratio (% of females) decreased in parallel with host’s fruit production (berries, and stone and pome fruits) from January to March, in semiarid high-elevation valleys with temperate summers and cold winters, in northwestern Argentina (CF Funes pers. comm.). As food supply slowed down, SWD sex ratios went from female-biased (January, plenty of fruit) to male-biased (March, scarce fruits). SWD sex ratio may vary between undamaged fruit collected from the plant and undamaged fruit lifted from the ground below the tree, as reported by Lasa et al. (2017), who worked in guava (P. guajava) in Mexico. In both cases, sex ratios were female-biased, varying within a range of 58–68%. Drummond et al. (2019) reported that across a 7-year survey, the relative abundances of sexes were slightly male-biased. Sex ratio (% of males) declined linearly between 2012 and 2018. The sex ratio of newly emerging adults remained more or less constant, with a mean of 0.98 males per female (Emiljanowicz et al. 2014).
No effect of egg density was observed on the sex ratio of SWD adults that emerged from attached pupae. Conversely, in detached pupae, sex ratio decreased linearly as egg density increased (Bezerra da Silva et al. 2019). A female-biased sex ratio for D. suzukii cultures probably results from the greater sensitivity of males to dietary ethanol. Without dietary alcohol, the sex ratio for D. suzukii stabilized near that of D. melanogaster, ~1:1 (Sampson et al. 2016).
4.7 Interspecific and Intergeneric Interactions with Other Frugivorous Dipterans
Selective pressure might have facilitated the evolution of Drosophila morphological traits such as a serrated ovipositor in D. suzukii. For instance, interspecific and intraspecific competitors could induce changes in SWD behavior of either female adult oviposition or larvae. The presence of competitors around fallen fruits on the ground probably suppressed D. suzukii oviposition on the fallen fruits and facilitated oviposition on non-fermenting substrates in nature, driving D. suzukii to use ripening fruits on the tree instead (Kidera and Takahashi 2020). Two species co-habiting similar niches would compete to either coexist or exclude the other. Interestingly, D. suzukii and D. melanogaster may have separate ecological niches, but D. melanogaster was observed ovipositing in fruit injuries caused by D. suzukii oviposition (Vilaire et al. 2011).
The presence of D. melanogaster in a substrate significantly reduced SWD emergence and egg laying; conversely, more SWD eggs were laid in blank media, not pre-inoculated with D. melanogaster (Shaw et al. 2018). Chemical cues or signs of previous oviposition by heterospecifics, such as D. melanogaster, can deter D. suzukii female from ovipositing in the same fruit (Shaw et al. 2018; Kidera and Takahashi 2020). This may be due to repellent olfactory cues such as trace amounts of the male D. melanogaster sex pheromone, cis-vaccenyl acetate, transferred to fruit during oviposition. In direct interspecific competition situations, D. melanogaster larvae have greater survival than D. suzukii larvae (Gao et al. 2018). This is due in part to higher tolerance to ethanol produced through decay and fermentation of fruit damaged by larval feeding and that higher levels of ethanol are produced in fruit containing D. melanogaster larvae than fruit containing D. suzukii larvae (Sampson et al. 2016; Gao et al. 2018). Although SWD females prefer to oviposit in ripe fruit, they are able to shift ecological niches and use ripening fruit to avoid competitive pressures and reduce potential ethanol exposure to their larvae (Little et al. 2020).
Zaprionus indianus flies would not be capable of laying eggs in healthy fruit because they lack a serrated ovipositor, but they might take advantage of other dipeterans oviposition punctures for placing their own eggs (Lasa et al. 2017; Shrader et al. 2020). Guavas attached to the tree infested with Z. indianus were also infested with D. suzukii, Anastrepha spp., or both. Therefore, the infestation was possible because of previous injuries caused by the other two frugivorous dipterans (Lasa et al. 2017). Competition from Z. indianus limited D. suzukii numbers in interspecific laboratory studies (Shrader et al. 2020). When reared with grapes, Z. indianus also affected D. suzukii mortality, and increased developmental times to pupation and adult emergence, compared with the intraspecific SWD controls. Pupal volume was scarcely reduced, and it occurred only with the highest interspecific larval densities (Shrader et al. 2020). In laboratory trials with strawberry ripe fruits, there was a significant and positive interaction between damage caused by D. suzukii and infestation by Z. indianus (Bernardi et al. 2017).
4.8 Natural Enemies–Drosophila suzukii Relationships
Reviews on SWD natural enemies were published by Asplen et al. (2015), Haye et al. (2016), Garcia et al. (2017), and Lee et al. (2019), involving several parasitoid, predator, and entomopathogenic species. Parasitoids represent the most numerous and most studied group of SWD’s natural enemies. In total, 30 parasitoid species associated with SWD were recorded throughout the world, either through field collections or through laboratory host specificity tests (Table 4.6). Nine of them are pupa parasitoids and 21 are larva parasitoids. The 40% of the parasitoid species recorded are Asian-native species, the SWD’s origin region. The 75% of Asian parasitoid species belong to the genus Asobara Foerster (Braconidae, Alysiinae), such as Asobara brevicauda van Achterberg & Guerrieri, A. elongata van Achterberg & Guerrieri, A. mesocauda van Achterberg & Guerrieri, A. triangulata van Achterberg & Guerrieri, A. unicolorata van Achterberg & Guerrieri, A. leveri (Nixon), A. japonica Belokobylskij (Guerrieri et al. 2016), A. pleuralis (Ashmead) (Girod et al. 2018a), and A. tabida (Nees) (Mitsui et al. 2007), while 35% are eucoilines (Figitidae: Eucoilinae), such as Ganaspis xanthopoda (Ashmead) (Kasuya et al. 2013), Ganaspis brasiliensis (Ihering), and Leptopilina japonica Novkovic & Kimura (Daane et al. 2016; Girod et al. 2018a; Giorgini et al. 2019). However, the association between G. xanthopoda and D. suzukii is doubtful because G. xanthopoda individuals from Japan, reported by Mitsui et al. (2007) and Kasuya et al. (2013), have been assigned to G. brasiliensis by Bufington and Forshage (2016) and Nomano et al. (2017). In turn, G. xanthopoda individuals tested under lab conditions by Kacsoh and Schlenke (2012) would appear to be G. brasiliensis (Nomano et al. 2017). Interestingly, G. brasiliensis would involve four lineages with different geographic distribution and host ranges (Nomano et al. 2017). These morphologically identical lineages could be a complex of cryptic species (Giorgini et al. 2019).
Among the other parasitoid species , 33.4% are native to America, 10% to Europe, and one of them (3.3%), Asobara citri Fisher, to Africa (Kacsoh and Schlenke 2012). The remaining 13.3% are present worldwide, such as Pachycrepoideus vindemmiae Rondani (Pteromalidae), Trichopria drosophilae Perkins (Diapriidae) (Rossi-Stacconi et al. 2013; Wang et al. 2016a, b, c; Daane et al. 2016), Leptopilina boulardi Barbotin, Carton & Kelner-Pillault, and L. clavipes (Hartig) (Figitidae: Eucoilinae) (Wollmann et al. 2016; Lue et al. 2017). Of the 10 American parasitoid species associated with SWD, six (60%) are undescribed species of eucoilines and diaprines (Table 4.6). The remaining four species are represented by one drosophilid larval parasitoid, Ganaspis hookeri Crawford (Lue et al. 2017), and by three generalist parasitoids that attack cycloraphic dipterous pupae, such as the pteromalines Muscidifurax raptorellus Kogan & Legner (Bonneau et al. 2019) and Spalangia simplex Perkins (García-Cancino et al. 2015) and the diaprine Trichopria anastrephae Lima (Krüger et al. 2019). Of the three European parasitoid species, one is drosophilid larval parasitoid, Leptopilina heterotoma (Thomson) (Figitidae) (Rossi-Stacconi et al. 2015), and two are generalist pupal parasitoids, Spalangia erythromera Förster and Vrestovia fidenas (Walker) (Pteromalidae) (Knoll et al. 2017).
Although more larval parasitoids than pupal parasitoids have been associated with the SWD, few species can successfully overcome the defense response of the SWD larva. Parasitoid eggs or larvae are usually encapsulated (Kacsoh and Schlenke 2012; Poyet et al. 2013). Therefore, Asian-native larval parasitoid species have mainly shown high capacity of developing successfully in SWD. Among them, L. japonica, G. brasiliensis, and A. japonica are the most significant parasitoid species, due to their high natural parasitism rate and greater specificity to SWD (Daane et al. 2016; Girod et al. 2018a; Matsuura et al. 2018; Giorgini et al. 2019). Thus, these three Asian parasitoid species were imported for evaluation as classical biological control agents in North America and/or Europe (Daane et al. 2016; Girod et al. 2018b, c; Wang et al. 2018a, 2019a, b; Giorgini et al. 2019).
Among the generalist parasitoid species , pupal parasitoids P. vindemmiae, M. raptorellus, V. fidenas, T. drosophilae , and T. anastrephae have been mainly evaluated under laboratory conditions, for both their ability to successfully parasitize SWD and their efficiency in reducing the target population (Wang et al. 2016a, b, 2018b; Bonneau et al. 2019; Krüger et al. 2019; Schlesener et al. 2019; Wolf et al. 2019; Yi et al. 2020). Since all of them can attack other dipterous species, their field effectiveness is doubtful. Nevertheless, T. drosophilae has a potential as SWD’s biological control agent (Mazzetto et al. 2016; Kaçar et al. 2017; Rossi-Stacconi et al. 2017; Pfab et al. 2018; Wang et al. 2018b; Yi et al. 2020). This potentiality was verified through T. drosophilae releases in berry fields at Colima and Jalisco, Mexico, where this strategy reduced 50–55% SWD wild populations (Gonzalez-Cabrera et al. 2019). Mass releases of T. drosophilae have been recently accomplished in northern Italy (Bioplanet 2020).
Arthropod predators such as ants, spiders, earwigs, and rove beetles have been found attacking SWD larvae and/or pupae inside field-infested fruit (Woltz and Lee 2017). Similarly, several generalist hemipteran species such as Dicyphus tamaninii Wagner (Hemiptera: Miridae), Orius laevigatus (Fieber), Cardiastethus nazarenus Reuter, and C. fasciventris Garbiglietti (Hemiptera: Anthocoridae) have also been observed predating on SWD eggs, larvae, and/or pupae, inside fallen fruit in berry growing areas of Northern Spain (Arnó et al. 2012; Gabarra et al. 2015). In addition, Labidura riparia Pallas (Dermaptera: Labiduridae) has been recorded as a common and effective SWD larvae and pupae predator, in Spain (Gabarra et al. 2015). Furthermore, several commercially available predator species have been tested under lab conditions for potential use as SWD’s biological control agents. The evaluated predator species were Orius insidiosus Say (Hemiptera: Anthocoridae), Dalotia coriaria Kraatz (Coleoptera: Staphylinidae), Podisus maculiventris Say (Hemiptera: Pentatomidae), Dicyphus hesperus Knight (Hemiptera: Miridae), and Chrysoperla carnea Stephens (Neuroptera: Chrysopidae) (Cuthbertson et al. 2014a; Renkema et al. 2015; Woltz et al. 2015; Renkema and Cuthbertson 2018; Bonneau et al. 2019).
Similarly, several commercial entomopathogenic agents , such as nematodes [Heterorhabditis bacteriophora Poinar (Rhabditida: Heterorhabditidae), Steinernema carpocapsae (Weiser), S. feltiae (Filipjev) and S. kraussei (Steiner) Travassos (Rhabditida: Steinernematidae)], and fungi [Metarhizium brunneum (= M. anisopliae) (Metchnikoff) Sorokin, Isaria fumosorosea (= Paecilomyces fumosoroseus) (Wize) Brown & Smith, Lecanicillium (= Verticillium) lecanii (Zimm.), and Beauveria bassiana (Balsamo-Crivelli) Vuillemin (Ascomycota; Hypocreales)], have been tested against SWD under lab conditions (Cuthbertson et al. 2014b; Woltz et al. 2015; Cossentine et al. 2016; Cuthbertson and Audsley 2016; Renkema and Cuthbertson 2018; Yousef et al. 2018; Lee et al. 2019). Bacteria and viruses have been also reported from laboratory trials or natural infestation (Lee et al. 2019). Commercial Bacillus thuringiensis var. kurstakii or B. thuringiensis var. israeliensis were tested against SWD in lab trials (Biganski et al. 2018; Cahenzli et al. 2018). More details on the use of SWD’s natural enemies as a biological control strategy can be seen in Chap. 8 of this book.
4.9 Concluding Remarks
In brief, egg-laying preference for ripening and ripe healthy small, soft and stone fruits attached to the plant makes SWD a severe and harmful pest for fruit-producing regions of Asia, Europe, and America. Most of the worldwide host fruit species preferred by SWD belong to Rosaceae, a plant family involving several economically important cultivated fruits, such as caneberries, cherries, strawberries, and stone and pome fruits, and a large list of non-crop species. Besides, both non-crop and crop Vaccinium L. plants (Ericaceae) have been recorded as recurrent SWD hosts in all continents. In addition to wild species of Rosaceae and Ericaceae families, several Adoxaceae (Sambucus spp.), Caprifoliaceae (Loricera spp.), and Rhamnaceae (Rhamnus cathartica L. and Frangula spp.) are crucial host plants for SWD population growth and seasonal dynamics in Europe and the USA. In Mexico and South America, Myrtaceae are fruit species highly susceptible to SWD, playing the important role of facilitating SWD multiplication in wild habitats adjacent to host crop fields.
The high degree of polyphagy shown by SWD either in their native region or in the invaded continents, added to the seasonal phenotypic plasticity that allows adaptations to adverse thermal periods, greatly favors SWD establishment and dispersion in the invaded regions. In turn, complex landscape structures such as forests with alternative non-cultivated host plants, shrub vegetation and flowering field margins, and the distance of these structures to host crops in the different invaded areas, facilitate SWD daily spread between different habitats, which can also determine infestation levels.
In both temperate and subtropical climate regions, the SWD seasonal dynamic is essentially related to temperatures more suitable for its movement between habitats, development, survival, and reproduction. The warmest, as well as the coldest, months of the year reduce, with more or less intensity, SWD populations in summer and winter, respectively. Therefore, SWD populations mostly generate two population peaks, in late spring and mid-autumn. However, as SWD can overwinter locally, adults can be captured throughout the year in the invaded areas.
A better understanding of the relationships between SWD and the components of newly invaded landscapes, in terms of reproductive behavior and interspecific and intergeneric interactions, especially with other frugivorous dipterans, and biological controllers would be very important to achieve. This will allow designing efficacious, efficient, and environmental friendly pest management strategies, maximizing the benefits provided by ecosystem services.
A diverse suite of local predators and parasitoids associated with SWD are found in wild vegetation areas surrounding fruit crop fields in the different invaded regions. These natural enemies should be conserved in their natural habitats. Furthermore, it should be noted that the recently found specialist Asian parasitoids could be significant SWD’s biocontrol agents. In addition, several entomopathogens have been laboratory tested, but their suppressive effects on SWD wild populations remains unknown and should be assessed.
References
Agbaba B (2017) A survey of non-crop plants as alternative hosts to raspberries for Drosophila suzukii (spotted wing drosophila). Dissertations, South Dakota State University
Alexandre BG (2016) Avaliação das assembleias de Drosophilidae (Insecta, Diptera) em área de restinga no município de Torres-RS. Dissertation, Universidade Federal do Rio Grande do Sul
Aly MFK, Kraus DA, Burrack HJ (2017) Effects of postharvest cold storage on the development and survival of immature Drosophila suzukii (Diptera: Drosophilidae) in artificial diet and Fruit. J Econ Entomol 110(1):87–93. https://doi.org/10.1093/jee/tow289
Andreazza F, Bernardi D, Botton M, Nava DE (2015) Índice de infestação natural de Drosophila suzukii e Zaprionus indianus (Diptera: Drosophilidae) em frutíferas nativas no município de pelotas. In: Abstracts of the XXIV Congresso de Iniciação Científica e XVII Encontro da Pós-Graduação, Universidade Federal de Pelotas, Pelotas, Rio Grande do Sul, 8–9 May 2018
Andreazza F, Baronio CA, Botton M (2016a) Suscetibilidade de bagas de genótipos de videira pela infestação por Drosophila suzukii (Diptera: Drosophilidae). Pesq agropec bras 51(5):599–606. https://doi.org/10.1590/S0100-204X2016000500021
Andreazza F, Bernardi D, Marangon RB et al (2016b) Técnica de criação de Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae) em dieta artificial. Bol Pesqui Desenvolv 240:1–23
Andreazza F, Haddi K, Oliveira EE (2016c) Drosophila suzukii (Diptera: Drosophilidae) arrives at Minas Gerais State, a main strawberry production region in Brazil. Fla Entomol 99(4):796–798. https://doi.org/10.1653/024.099.0439
Andreazza F, Bernardi D, Dos Santos RSS et al (2017) Drosophila suzukii in southern neotropical region: current status and futures perspectives. Neotrop Entomol 46(6):591–605. https://doi.org/10.1007/s13744-017-0554-7
Arnó J, Riudavets J, Gabarra R (2012) Survey of host plants and natural enemies of Drosophila suzukii in an area of strawberry production in Catalonia (northeast Spain). IOBC/WPRS Bull 80:29–34
Arnó J, Sola M, Riudavets J, Gabarra R (2016) Population dynamics, non-crop hosts, and fruit susceptibility of Drosophila suzukii in Northeast Spain. J Pest Sci 89:713–723. https://doi.org/10.1007/s10340-016-0774-3
Asplen MK, Anfora G, Biondi A et al (2015) Invasion biology of spotted wing Drosophila (Drosophila suzukii): a global perspective and future priorities. J Pest Sci 88(3):469–494. https://doi.org/10.1007/s10340-015-0681-z
Bal HK, Adams C, Grieshop M (2017) Evaluation of off-season potential breeding sources for spotted wing Drosophila (Drosophila suzukii Matsumura) in Michigan. J Econ Entomol 110(6):2466–2470. https://doi.org/10.1093/jee/tox252
Baroffio CA, Richoz P, Fischer S et al (2014) Monitoring Drosophila suzukii in Switzerland in 2012. J Berry Res 4:47–52. https://doi.org/10.3233/JBR-140066
Beers EH, Van Steenwyk RA, Shearer PW, Coates WW, Grant JA (2011) Developing Drosophila suzukii management programs for sweet cherry in the western United States. Pest Manag Sci 67(11):1386–1395. https://doi.org/10.1002/ps.2279
Bellamy DE, Sisterson MS, Walse SS (2013) Quantifying host potentials: indexing postharvest fresh fruits for spotted wing Drosophila, Drosophila suzukii. PLoS One 8:e61227. https://doi.org/10.1371/journal.pone.0061227
Bernardi D, Andreazza F, Botton M et al (2017) Susceptibility and interactions of Drosophila suzukii and Zaprionus indianus (Diptera: Drosophilidae) in damaging strawberry. Neotrop Entomol 46(1):1–7. https://doi.org/10.1007/s13744-016-0423-9
Bezerra Da Silva CS, Park KR, Blood RA et al (2019) Intraspecific competition affects the pupation behavior of spotted-wing Drosophila (Drosophila suzukii). Sci Rep 9:7775. https://doi.org/10.1038/s41598-019-44248-6
Biganski S, Jehle JA, Kleespies RG (2018) Bacillus thuringiensis serovar. israelensis has no effect on Drosophila suzukii Matsumura. J Appl Entomol 142:33–36. https://doi.org/10.1111/jen.12415
Bioplanet (2020) Veneto has started the launch of antagonist insect against Drosophila suzukii. https://www.freshplaza.com/article/9210339/veneto-has-started-the-launch-of-antagonist-insect-against-drosophila-suzukii. Accessed 21 Apr 2020
Bolda M (2008) New fruit fly pest in strawberries and caneberries. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=821. Accessed 14 May 2020
Bonneau P, Renkema J, Fournier V et al (2019) Ability of Muscidifurax raptorellus and other parasitoids and predators to control Drosophila suzukii populations in raspberries in the laboratory. Insects 10(3):68. https://doi.org/10.3390/insects10030068
Borba RS, Foppa F, Frare JB (2016) Ocorrência de Drosophila suzukii (Matsumura, 1931) (Diptera, Drosophilidae) em pêssego. In: Abstracts of the XXVI Congresso Brasileiro de Entomologia, Brazilia, 13–17 March 2016
Briem F, Eben A, Gross J (2016) An invader supported by a parasite: mistletoe berries as a host for food and reproduction of spotted wing drosophila in early spring. J Pest Sci 89(3):749–759. https://doi.org/10.1007/s10340-016-0739-6
Bufington ML, Forshage M (2016) Redescription of Ganaspis brasiliensis (Ihering, 1905), new combination, (Hymenoptera: Figitidae) a natural enemy of the invasive Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae). Proc Entomol Soc Wash 118(1):1–13. https://doi.org/10.4289/0013-8797.118.1.1
Burrack HJ, Fernandez GE, Spivey T et al (2013) Variation in selection and utilization of host crops in the field and laboratory by Drosophila suzukii Matsumura (Diptera: Drosophilidae), an invasive frugivore. Pest Manag Sci 69:1173–1180. https://doi.org/10.1002/ps.3489
CABI EPPO Centre for Agricultural Bioscience International, European and Mediterranean Plant Protection Organization (2016) Distribution maps of plant pests, Map 766 (1st revision). https://www.cabi.org/isc/abstract/20163203811. Accessed 19 Apr 2020
Cahenzli F, Strack T, Daniel C (2018) Screening of 25 different natural crop protection products against Drosophila suzukii. J Appl Entomol 142(6):563–577. https://doi.org/10.1111/jen.12510
Calabria G, Máca J, Bachli G et al (2012) First records of the potential pest species Drosophila suzukii (Diptera: Drosophilidae) in Europe. J Appl Entomol 136:139–147. https://doi.org/10.1111/j.1439-0418.2010.01583.x
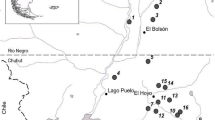
Cichón LI, Lago JD, Garrido SA (2015) Primera detección de Drosophila suzukii (Matsumura, 1939) (Diptera: Drosophilidae) en frambuesas del Valle de Río Negro, Argentina. In: Abstracts of the IX Congreso Argentino de Entomología, Universidad Nacional de Misiones, Posadas, 10–12 September 2018
Cini A, Ioratti C, Anfora G (2012) A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull Insectol 65:149–160
Cini A, Anfora G, Escudero-Colomar LA et al (2014) Tracking the invasion of the alien fruit pest Drosophila suzukii in Europe. J Pest Sci 87:559–566. https://doi.org/10.1007/s10340-014-0617-z
Chabert S, Allemand R, Poyet M, Eslin P, Gibert P (2012) Ability of European parasitoids (Hymenoptera) to control a new invasive Asiatic pest, Drosophila suzukii. Biol Control 63:40–47. https://doi.org/10.1016/j.biocontrol.2012.05.005
Cossentine J, Robertson M, Buitenhuis R (2016) Impact of acquired entomopathogenic fungi on adult Drosophila suzukii survival and fecundity. Biol Control 103:129–137. https://doi.org/10.1016/j.biocontrol.2016.09.002
Cuthbertson AGS, Audsley N (2016) Further screening of entomopathogenic fungi and nematodes as control agents for Drosophila suzukii. Insects 7(2):24. https://doi.org/10.3390/insects7020024
Cuthbertson AG, Blackburn LF, Audsley N (2014a) Efficacy of commercially available invertebrate predators against Drosophila suzukii. Insects 5(4):952–960. https://doi.org/10.3390/insects5040952
Cuthbertson AGS, Collins DA, Blackburn LF et al (2014b) Preliminary screening of potential control products against Drosophila suzukii. Insects 5(2):488–498. https://doi.org/10.3390/insects5020488
Daane KM, Wang X, Biondi A et al (2016) First exploration of parasitoids of Drosophila suzukii in South Korea as potential classical biological agents. J Pest Sci 89:823–835. https://doi.org/10.1007/s10340-016-0740-0
Dagatti CV, Marcucci B, Herrera ME et al (2018) Primera detección de Drosophila suzukii (Diptera: Drosophilidae) en frutos de zarzamora en Mendoza, Argentina. Rev Soc Entomol Arge 77(3):26–29. https://doi.org/10.25085/rsea.770304
Dalton DT, Walton VM, Shearer PW et al (2011) Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Manag Sci 67(11):1368–1374. https://doi.org/10.1002/ps.2280
Dekker T, Revadi S, Mansourian S et al (2015) Loss of Drosophila pheromone reverses its role in sexual communication in Drosophila suzukii. Proc R Soc B 282:20143018. https://doi.org/10.1098/rspb.2014.3018
Deprá M, Poppe JL, Schmitz HJ et al (2014) The first records of the invasive pest Drosophila suzukii in south American continent. J Pest Sci 87:379–383. https://doi.org/10.1007/s10340-014-0591-5
Diepenbrock LM, McPhie DR (2018) Use of a novel host plant family, Basallaceae, by Drosophila suzukii (Diptera: Drosophilidae). Proc Entomol Soc Wash 120(2):255–259. https://doi.org/10.4289/0013-8797.120.2.255
Dos Santos LA, Mendes MF, Krüger AP et al (2017) Global potential distribution of Drosophila suzukii (Diptera, Drosophilidae). PLoS One 12(3):e0174318. https://doi.org/10.1371/journal.pone.0174318
Dreves AJ (2008) Fruit fly, “Spotted Wing Drosophila,” identified in wine grapes. https://today.oregonstate.edu/archives/2009/oct/fruit-fly-“spotted-wing-drosophila”-identified-wine-grapes. Accessed 20 May 2020
Drummond F, Ballman E, Collins J (2019) Population dynamics of spotted wing Drosophila (Drosophila suzukii (Matsumura)) in maine wild blueberry (Vaccinium angustifolium Aiton). Insects 10(7):205. https://doi.org/10.3390/insects10070205
Efil L (2018) Çanakkale ili çilek alanlarında yeni bir zararlı Drosophila suzukii (Matsumura) (Diptera: Drosophilidae)’nin yayılış alanları ve bulaşıklılığı. Türk Tarım ve Doğa Bilimleri Dergisi 5(3):280–284. https://doi.org/10.30910/turkjans.448354
Elsensohn JE, Loeb GM (2018) Non-crop host sampling yields insights into small-scale population dynamics of Drosophila suzukii (Matsumura). Insects 9(1):5. https://doi.org/10.3390/insects9010005
Emiljanowicz LM, Ryan GD, Langille A et al (2014) Development, reproductive output and population growth of the fruit fly pest Drosophila suzukii (Diptera: Drosophilidae) on artificial diet. J Econ Entomol 107(4):1392–1398. https://doi.org/10.1603/ec13504
Enriquez T, Colinet H (2017) Basal tolerance to heat and cold exposure of the spotted wing drosophila, Drosophila suzukii. PeerJ 5:e3112. https://doi.org/10.7717/peerj.3112
EPPO European and Mediterranean Plant Protection Organization (2014) EPPO gobal database. https://gd.eppo.int/taxon/DROSSU/distribution. Accessed 13 Apr 2020
EPPO European and Mediterranean Plant Protection Organization (2018) EPPO gobal database. https://gd.eppo.int/taxon/DROSSU/hosts. Accessed 13 Apr 2020
EPPO European and Mediterranean Plant Protection Organization (2019) EPPO gobal database. https://gd.eppo.int/taxon/DROSSU/hosts. Accessed 15 Apr 2020
Escobar LI, Ovruski SM, Kirschbaum DS (2018) Foreign invasive pests Drosophila suzukii (Matsumura) and Zaprionus indianus Gupta (Diptera: Drosophilidae) threaten fruit production in northwestern Argentina. Dros Info Serv 101:9–14
Fraimout A, Loiseau A, Price DK et al (2015) New set of microsatellite markers for the spotted-wing Drosophila suzukii (Diptera: Drosophilidae): a promising molecular tool for inferring the invasion history of this major insect pest. Eur J Entomol 112(4):855–859. https://doi.org/10.14411/eje.2015.079
Fraimout A, Jacquemart P, Villarroel B et al (2018) Phenotypic plasticity of Drosophila suzukii wing to developmental temperature: implications for flight. J Exp Biol 221(Pt 13):jeb166868. https://doi.org/10.1242/jeb.166868
Funes CF, Escobar LI, Meneguzzi NG et al (2017) Occurrence of Anastrepha fraterculus and Ceratitis capitata (Diptera: Tephritidae) in organically grown Rubus spp. (Rosales: Rosaceae), in two very contrasting environments of northwestern Argentina. Fla Entomol 100:672–674. https://doi.org/10.1653/024.100.0330
Funes CF, Escobar LI, Heredia AM et al (2018a) Primera detección e incidencia de Drosophila suzukii (Diptera: Drosophilidae) en cultivos de berries de Tucumán, Argentina. In: Abstracts of the X Congreso Argentino de Entomología, Mendoza, 21–24 May 2018
Funes CF, Escobar LI, Palavecino BE et al (2018b) Detección de Drosophila suzukii (Diptera: Drosophilidae) en cultivos de arándano de Tucumán, Argentina. In: Abstracts of the X Congreso Argentino de Entomología, Mendoza, 21–24 May 2018
Funes CF, Gallardo FE, Reche VA et al (2019) Parasitoides de Sudamérica asociados a las plagas invasoras Drosophila suzukii y Zapronius indianus (Diptera: Drosophilidae) y su potencial como agentes de control biológico. Semiárida Rev Fac Agr UNLPam 29(supl):25–27
Gabarra R, Arnó J, Riudavets J (2012) Primeros resultados sobre Drosophila suzukii: huéspedes, susceptibilidad de los frutos y enemigos naturales. Phytoma España 240:46–52
Gabarra R, Riudavets J, Rodríguez GA et al (2015) Prospects for the biological control of Drosophila suzukii. Biocontrol 60:331–339. https://doi.org/10.1007/s10526-014-9646-z
Garcia FRM, Wollmann J, Krüger AP et al (2017) Biological control of Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae): state of the art and prospects. In: Davenport L (ed) Biological control: methods, applications and challenges. Nova Science Publishers, Inc., New York, pp 1–28
García-Cancino MD, Hernández AG, Cabrera JG et al (2015) Parasitoids of Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) in Colima, Mexico. Southwest Entomol 40(4):855–858. https://doi.org/10.3958/059.040.0418
Gargani E, Bagnoli B, Simoni S (2015) Drosophila suzukii in Tuscany (Italy), from cherry crops to vineyards and beyond. International Centre for Advanced Mediterranean Agronomic Studies (CIHEAM). Watch Letter n°33. https://www.iamm.ciheam.org/publications/177/019_Gargani.pdf. Accessed 3 Jun 2020
Garrido SA, Cichón LI, Lago JD, Gallardo FE, Navarro MD (2018) Primer registro de Leptopilina boulardi (Hymenoptera: Figitidae) asociada a Drosophila suzukii (Diptera: Drosophilidae) en el Alto Valle de Rio Negro y Neuquén, Patagonia Argentina. Rev Soc Entomol Arge 77:22–27
Gao HH, Zhai YF, Chen H, Wang YM, Liu Q, Hu QL, Ren FS, Yu Y (2018) Ecological niche difference associated with varied ethanol tolerance between Drosophila suzukii and Drosophila melanogaster (Diptera: Drosophilidae). Fla Entomol 101:498–504. https://doi.org/10.1653/024.101.0308
Geisler FCS, Santos J, Holdefer DR et al (2015) Primeiro registro de Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae) para o estado do Paraná, Brasil e de novos hospedeiros. Rev Css Amb 9:125–129. https://doi.org/10.18316/1981-8858.15
Giorgini M, Wang X, Wang Y et al (2019) Exploration for native parasitoids of Drosophila suzukii in China reveals a diversity of parasitoid species and narrow host range of the dominant parasitoid. J Pest Sci 92:509–522. https://doi.org/10.1007/s10340-018-01068-3
Girod P, Borowiec N, Buffington M et al (2018a) The parasitoid complex of D. suzukii and other fruit feeding Drosophila species in Asia. Sci Rep 8:11839. https://doi.org/10.1038/s41598-018-29555-8
Girod P, Lierhmann O, Urvois T et al (2018b) Host specifcity of Asian parasitoids for potential classical biological control of Drosophila suzukii. J Pest Sci 91:1241–1250. https://doi.org/10.1007/s10340-018-10033-z
Girod P, Rossignaud L, Haye T et al (2018c) Development of Asian parasitoids in larvae of Drosophila suzukii feeding on blueberry and artifcial diet. J Appl Entomol 142(5):483–494. https://doi.org/10.1111/jen.12496
Gong X, Bräcker L, Bölke N et al (2016) Strawberry accessions with reduced Drosophila suzukii emergence from fruits. Front Plant Sci 7:1880. https://doi.org/10.3389/fpls.2016.01880
Gonsebatt G, San Pedro P, Fanara JJ et al (2017) Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) en durazneros (Prunus persica) en el sur de la provincia de Santa Fé: primer registro. In: Abstracts of the XIX Congreso y XXXVII Reunión Anual de Biología, Santa Fé, 28 November 2017
Gonzales G, Mary A, Goñi B (2015) Drosophila suzukii (Matsumura) found in Uruguay. Dros Info Serv 98:103–107
Gonzalez-Cabrera J, Moreno-Carrillo G, Sanchez-Gonzalez JA (2019) Single and combined release of Trichopria drosophilae (Hymenoptera: Diapriidae) to control Drosophila suzukii (Diptera: Drosophilidae). Neotrop Entomol 48:949–956. https://doi.org/10.1007/s13744-019-00707-3
Grassi A, Palmieri L, Giongo L (2009) Nuovo fitofago per i piccolo frutti in Trentino. Terra Trent. 55:19–23
Grassi A, Giongo L, Palmieri L (2011) Drosophila (Sophophora) suzukii (Matsumura), new pest of soft fruits in Trentino (North Italy) and in Europe. IOBC/WPRS Bull 70:121–128
Grassi A, Gottardello A, Dalton DT et al (2018) Seasonal reproductive biology of Drosophila suzukii (Diptera: Drosophilidae) in temperate climates. Environ Entomol 47(1):166–174. https://doi.org/10.1093/ee/nvx195
Guerrieri E, Giorgini M, Cascone P et al (2016) Species diversity in the parasitoid genus Asobara (Hymenoptera: Braconidae) from the native area of the fruit fly pest Drosophila suzukii (Diptera: Drosophilidae). PLoS One 11(2):e0147382. https://doi.org/10.1371/journal.pone.0147382
Hamby KA, Bolda MP, Sheehan ME et al (2014) Seasonal monitoring for Drosophila suzukii (Diptera: Drosophilidae) in California commercial raspberries. Environ Entomol 43:1008–1018. https://doi.org/10.1603/EN13245
Hamby KA, Bellamy DE, Chiu JC et al (2016) Biotic and abiotic factors impacting development, behavior, phenology, and reproductive biology of Drosophila suzukii. J Pest Sci 89(3):605–619. https://doi.org/10.1007/s10340-016-0756-5
Harris DW, Hamby KA, Wilson HE et al (2014) Seasonal monitoring of Drosophila suzukii (Diptera: Drosophilidae) in a mixed fruit production system. J Asia Pac Entomol 17(4):857–864. https://doi.org/10.1016/j.aspen.2014.08.006
Hauser M (2011) A historic account of the invasion of Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) in the continental United States, with remarks on their identification. Pest Manag Sci 67(11):1352–1357. https://doi.org/10.1002/ps.2265
Haye T, Girod P, Cuthbertson AGS et al (2016) Current SWD IPM tactics and their practical implementation in fruit crops across different regions around the world. J Pest Sci 89(3):643–651. https://doi.org/10.1007/s10340-016-0737-8
Ideo S, Watada M, Mitsui H, Kimura MT (2008) Host range of Asobara japonica (Hym.: Braconidae), a larval parasitoid of drosophilid flies. Entomol Sci 11(1):1–6. https://doi.org/10.1111/j.1479-8298.2007.00244.x
Jakobs R, Gariepy TD, Sinclair BJ (2015) Adult plasticity of cold tolerance in a continental-temperate population of Drosophila suzukii. J Insect Physiol 79:1–9. https://doi.org/10.1016/j.jinsphys.2015.05.003
Junior Z, Fornazier MJ, Andreazza F et al (2018) Spread of two invasive flies (Diptera: Drosophilidae) infesting commercial fruits in Southeastern Brazil. Fla Entomol 101(3):522–525. https://doi.org/10.1653/024.101.0328
Kaçar G, Wang XG, Biondi A, Daane KM (2017) Linear functional response by two pupal Drosophila parasitoids foraging within single or multiple patch environments. PLoS One 12(8):e0183525. https://doi.org/10.1371/journal.pone.0183525
Kacsoh BZ, Schlenke TA (2012) High hemocyte load is associated with increased resistance against parasitoids in Drosophila suzukii, a relative of D. melanogaster. PLoS One 7(4):e34721. https://doi.org/10.1371/journal.pone.0034721
Kaneshiro KY (1983) Drosophila (Sophophora) suzukii (Matsumura). Proc Hawaii Entomol Soc 24:179
Kasuya N, Mitsui H, Ideo S et al (2013) Ecological, morphological and molecular studies on Ganaspis individuals (Hymenoptera: Figitidae) attacking Drosophila suzukii (Diptera: Drosophilidae). Appl Entomol Zool 48:87–92. https://doi.org/10.1007/s13355-012-0156-0
Kellermann V, Loeschcke V, Hoffmann AA et al (2012) Phylogenetic constraints in key functional traits behind species’ climate niches: patterns of desiccation and cold resistance across 95 Drosophila species. Evol 66(11):3377–3389. https://doi.org/10.1111/j.1558-5646.2012.01685.x
Kenis M, Tonina L, Eschen R et al (2016) Non-crop plants used as hosts by Drosophila suzukii in Europe. J Pest Sci 89:735–748. https://doi.org/10.1007/s10340-016-0755-6
Kidera H, Takahashi KH (2020) Chemical cues from competitors change the oviposition preference of Drosophila suzukii. Entomol Exp Appl 168(4):304–310. https://doi.org/10.1111/eea.12889
Kienzle R, Groß LB, Caughman S et al (2020) Resource use by individual Drosophila suzukii reveals a fexible preference for oviposition into healthy fruits. Sci Rep 10:3132. https://doi.org/10.1038/s41598-020-59595-y
Kimura MT (1988) Adaptations to temperate climates and evolution of overwintering strategies in the Drosophila melanogaster species group. Evol 42:1288–1297. https://doi.org/10.1111/j.1558-5646.1988.tb04188.x
Kimura MT (2004) Cold and heat tolerance of drosophilid flies with reference to their latitudinal distributions. Oecologia 140:442–449. https://doi.org/10.1007/s00442-004-1605-4
Kinjo H, Kunimi Y, Ban T et al (2013) Oviposition efficacy of Drosophila suzukii (Diptera: Drosophilidae) on different cultivars of blueberry. J Econ Ent 106:1767–1771. https://doi.org/10.1603/EC12505
Kinjo H, Kunimi Y, Nakai M (2014) Effects of temperature on the reproduction and development of Drosophila suzukii (Diptera: Drosophilidae). Appl Entomol Zool 49(2):297–304. https://doi.org/10.1007/s13355-014-0249-z
Knoll V, Ellenbroek T, Romeis J et al (2017) Seasonal and regional presence of hymenopteran parasitoids of Drosophilain Switzerland and their ability to parasitize the invasive Drosophila suzukii. Sci Rep 7:40697. https://doi.org/10.1038/srep40697
Koch JB, Dupuis JR, Jardeleza MK et al (2020) Population genomic and phenotype diversity of invasive Drosophila suzukii in Hawai‘i. Biol Invasions 22:1753–1770. https://doi.org/10.1007/s10530-020-02217-5
Krüger AP, Schlesener DCH, Martins LN et al (2018a) Effects of irradiation dose on sterility induction and quality parameters of Drosophila suzukii (Diptera: Drosophilidae). J Econ Entomol 111(2):741–746. https://doi.org/10.1093/jee/tox349
Krüger AP, Schlesener DC, Martins LN et al (2018b) Radiation effects on Drosophila suzukii (Diptera: Drosophilidae) reproductive behaviour. J Appl Entomol 143:88–94. https://doi.org/10.1111/jen.12563
Krüger AP, Scheunemann T, Vieira JGA et al (2019) Effects of extrinsic, intraspecific competition and host deprivation on the biology of Trichopria anastrephae (Hymenoptera: Diapriidae) reared on Drosophila suzukii (Diptera: Drosophilidae). Neotrop Entomol 48(6):957–965. https://doi.org/10.1007/s13744-019-00705-5
Lasa R, Tadeo E, Dinor LA et al (2017) Fruit firmness, superficial damage and location modulate infestation by Drosophila suzukii and Zaprionus indianus (Diptera: Drosophilidae): The case of guava Psidium guajava L., in Veracruz, Mexico. Entomol Exp Appl 162:4–12. https://doi.org/10.1111/eea.12519
Lauyé MA (2017) Diversidad de Drosophilidae (Diptera) en frutos de interés productivo y agronómico, en localidades de la región sur de Uruguay, con especial atención a la especie invasora Drosophila suzukii (Matsumura, 1931). Dissertation, Universidad de la República (Uruguay)
Lavagnino NJ, Diaz BM, Cichón LI et al (2018) New records of the invasive pest Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) in the South American continent. Rev Soc Entomol Argent 77(1):27–31. https://doi.org/10.25085/rsea.770105
Lee RE (1991) Principles of insect low temperature tolerance. In: Insects at low temperature. Springer, Boston, pp 17–46
Lee JC, Bruck DJ, Curry H et al (2011a) The susceptibility of small fruits and cherries to the spotted-wing drosophila, Drosophila suzukii. Pest Manag Sci 67(11):1358–1367. https://doi.org/10.1002/ps.2225
Lee JC, Bruck DJ, Dreves AJ et al (2011b) In focus: spotted wing drosophila, Drosophila suzukii, across perspectives. Pest Manag Sci 67(11):1349–1351. https://doi.org/10.1002/ps.2271
Lee JC, Burrack HJ, Barrantes LD et al (2012) Evaluation of monitoring traps for Drosophila suzukii (Diptera: Drosophilidae) in North America. J Econ Entomol 105(4):1350–1357. https://doi.org/10.1603/EC12132
Lee JC, Dreves AJ, Cave AM et al (2015) Infestation of wild and ornamental noncrop fruits by Drosophila suzukii (Diptera: Drosophilidae). Ann Entomol Soc Am 108(2):117–129. https://doi.org/10.1093/aesa/sau014
Lee JC, Wang XG, Daane KM et al (2019) Biological control of spotted-wing Drosophila (Diptera: Drosophilidae)-current and pending tactics. J Integr Pest Manag 10(1):22. https://doi.org/10.1093/jipm/pmz021
Little CM, Chapman TW, Moreau DL et al (2017) Susceptibility of selected boreal fruits and berries to the invasive pest Drosophila suzukii (Diptera: Drosophilidae). Pest Manag Sci 73(1):160–166. https://doi.org/10.1002/ps.4366
Little CM, Chapman TW, Hillier NK (2020) Plasticity is key to success of Drosophila suzukii (Diptera: Drosophilidae) invasion. J Insect Sci 20(3):5. https://doi.org/10.1093/jisesa/ieaa034
Lochbaum T (2017) Drosophila suzukii (Diptera: Drosophilidae): Distribución estacional, incidencia y estrategias preliminares de manejo en el departamento Confluencia, Neuquén. Primer Encuentro Nacional Técnico-Científico sobre la plaga Drosophila suzukii. SENASA, Buenos Aires
Lue CH, Mottern JL, Walsh GC et al (2017) New record for the invasive spotted wing Drosophila, Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae) in Anillaco, Western Argentina. Proc Entomol Soc Wash 119(1):146–150. https://doi.org/10.4289/0013-8797.119.1.146
Magnacca KN, Foote D, O’Grady PM (2008) A review of the endemic Hawaiian Drosophilidae and their host plants. Zootaxa 1728:1–58
Matsuura A, Mitsui H, Kimura MT (2018) A preliminary study on distributions and oviposition sites of Drosophila suzukii (Diptera: Drosophilidae) and its parasitoids on wild cherry tree in Tokyo, central Japan. Appl Entomol Zool 53(1):47–53. https://doi.org/10.1007/s13355-017-0527-7
Mazzetto F, Pansa MG, Ingegno BL et al (2015) Monitoring of the exotic fly Drosophila suzukii in stone, pome and soft fruit orchards in NW Italy. J Asia Pac Entomol 18(2):321–329. https://doi.org/10.1016/j.aspen.2015.04.001
Mazzetto F, Marchetti E, Amiresmaeili N et al (2016) Drosophila parasitoids in northern Italy and their potential to attack the exotic pest Drosophila suzukii. J Pest Sci 89(1):837–850. https://doi.org/10.1007/s10340-016-0746-7
Mazzetto F, Lessio F, Giacosa S et al (2020) Relationships between Drosophila suzukii and grapevine in North-western Italy: seasonal presence and cultivar susceptibility. Bull Insectol 73(1):29–38
Mazzi D, Bravin E, Meraner M et al (2017) Economic impact of the introduction and establishment of Drosophila suzukii on Sweet cherry production in Switzerland. Insects 8:18. https://doi.org/10.3390/insects8010018
Mazzoni V, Anfora G, Virant-Doberlet M (2013) Substrate vibrations during courtship in three Drosophila species. PLOS One 8(11):e80708. https://doi.org/10.1371/journal.pone.0080708
Miller B, Anfora G, Bufngton M et al (2015) Seasonal occurrence of resident parasitoids associated with Drosophila suzukii in two small fruit production regions of Italy and the USA. B Insectol 68(2):255–263
Mitsui H, Kimura MT (2010) Distribution, abundance and host association of two parasitoid species attacking frugivorous drosophilid larvae in central Japan. Eur J Entomol 107(4):535–540. https://doi.org/10.14411/eje.2010.061
Mitsui H, Takahashi KH, Kimura MT (2006) Spatial distributions and clutch sizes of Drosophila species ovipositing on cherry fruits of different stages. Popul Ecol 48(3):233–237. https://doi.org/10.1007/s10144-006-0260-5
Mitsui H, Van Achterberg K, Nordlander G et al (2007) Geographical distributions and host associations of larval parasitoids of frugivorous Drosophilidae in Japan. J Nat Hist 41:1731–1738. https://doi.org/10.1080/00222930701504797
Mitsui H, Beppu K, Kimura MT (2010) Seasonal life cycles and resource uses of flower- and fruit-feeding Drosophilid flies (Diptera: Drosophilidae) in central Japan. Entomol Sci 13:60–67. https://doi.org/10.1111/j.1479-8298.2010.00372.x
Morales K (2020) The spotted wing Drosophila in the south of the world: Chilean case and its first productive impacts. In: Invasive species—introduction pathways, economic impact, and possible management options. https://www.intechopen.com/online-first/the-spotted-wing-drosophila-in-the-south-of-the-world-chilean-case-and-its-first-productive-impacts. Accessed 15 Apr 2020
Müller FA, Nava DE (2014) Primeiro relato de Drosophila suzukii (Diptera: Drosophilidae) em frutos de araçá e pitanga em municípios da região sul do Rio Grande do Sul, Brasil. In: Abstracts of the XXV Congresso Brasileiro de Entomologia, Universidade Federal de Goiás, 4–18 September 2014
Nava DE, Botton M, Bernardi D et al (2015) Bioecologia, monitoramento e controle de Drosophila suzukii na cultura do morangueiro. Série documentos 398:1–28
Nomano FY, Mitsui H, Kimura MT (2015) Capacity of Japanese Asobara species (Hymenoptera: Braconidae) to parasitize a fruit pest Drosophila suzukii (Diptera: Drosophilidae). J Appl Entomol 139:105–113. https://doi.org/10.1111/jen.1214
Nomano FY, Kasuya N, Matsuura A et al (2017) Genetic differentiation of Ganaspis brasiliensis (Hymenoptera: Figitidae) from East and Southeast Asia. Appl Entomol Zool 52:429–437. https://doi.org/10.1111/jen.12141
Novkovic B, Mitsui H, Suwito A, Kimura MT (2011) Taxonomy and phylogeny of Leptopilina species (Hymenoptera: Cynipoidea: Figitidae) attacking frugivorous drosophilid flies in Japan, with description of three new species. Entomol Sci 14:333–346. https://doi.org/10.1111/j.1479-8298.2011.00459.x
Nunes AM, Schlesener DCH, Souza DS et al (2014) Primeiros registros de Drosophila suzukii (Diptera: Drosophilidae) em agroecossistemas na metade sul do Rio Grande do Sul. In: Abstracts of the XXV Congresso Brasileiro de Entomologia, Universidade Federal de Goiás, 4–18 September 2014
O’Grady PM, Beardsley JW, Perreira WD (2002) New records for introduced Drosophilidae (Diptera) in Hawaii. Bishop Mus Occ Pap 69:34–35
Oliveira AS, Amaral Neto JAB, Santos RSS (2015) Primeiro registro de Drosophila suzukii (Diptera: Drosophilidae) em pomar de macieira em Vacaria- Rio Grande do Sul. In: Abstracts of the XIII Encontro de Iniciação Científica e IX Encontro de Pós-Graduandos da Embrapa Uva e Vinho, Bento Gonçalves, 16–17 July 2015
Orhan A, Aslantaş R, Önder BŞ et al (2016) First record of the invasive vinegar fly Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) from eastern Turkey. Turk J Zool 40:290–293. https://doi.org/10.3906/zoo-1412-25
Panel ADC, Zeeman L, van der Sluis BJ et al (2018) Overwintered Drosophila suzukii are the main source for infestations of the first fruit crops of the season. Insects 9:145. https://doi.org/10.3390/insects9040145
Pfab F, Stacconi MVR, Anfora G et al (2018) Optimized timing of parasitoid release: a mathematical model for biological control of Drosophila suzukii. Theor Ecol 11:489–450. https://doi.org/10.1007/s12080-018-0382-3
Poyet M, Havard S, Prévost G et al (2013) Resistance of Drosophila suzukii to the larval parasitoids Leptopilina heterotoma and Asobara japonica is related to haemocyte load. Physiol Entomol 38:45–53. https://doi.org/10.1111/phen.12002
Poyet M, Le Roux V, Gibert P et al (2015) The wide potential trophic niche of the Asiatic fruit fly Drosophila suzukii: The key of Its invasion success in temperate Europe? PLoS One 10(11):e0142785. https://doi.org/10.1371/journal.pone.0142785
Rendon D, Walton V, Tait G et al (2019) Interactions among morphotype, nutritions, and temperature impact fitness of an invasive fly. Ecol Evol 9(5):2615–2252. https://doi.org/10.1002/ece3.4928
Renkema JM, Cuthbertson AGS (2018) Impact of multiple natural enemies on immature Drosophila suzukii in strawberries and blueberries. Biocontrol 63:719–728. https://doi.org/10.1007/s10526-018-9874-8
Renkema JM, Telfer Z, Gariepy T et al (2015) Dalotia coriaria as a predator of Drosophila suzukii: functional responses, reduced fruit infestation and molecular diagnostics. Biol Control 89:1–10. https://doi.org/10.1016/j.biocontrol.2015.04.024
Revadi S, Lebreton S, Witzgall P et al (2015) Sexual behavior of Drosophila suzukii. Insects 6(1):183–196. https://doi.org/10.3390/insects6010183
Rossi-Stacconi M, Grassi A, Dalton D et al (2013) First field records of Pachycrepoideus vindemiae as a parasitoid of Drosophila suzukii in European and Oregon small fruit production areas. Entomol 1(1):11–16. https://doi.org/10.4081/entomologia.2013.e3
Rossi-Stacconi MV, Bufington M, Daane KM et al (2015) Host stage preference, efficacy and fecundity of parasitoids attacking Drosophila suzukii in newly invaded areas. Biol Control 84:28–35. https://doi.org/10.1016/j.biocontrol.2015.02.003
Rossi-Stacconi MV, Kaur R, Mazzoni V et al (2016) Multiple lines of evidence for reproductive winter diapause in the invasive pest Drosophila suzukii. J Pest Sci 89(3):689–700. https://doi.org/10.1007/s10340-016-0753-8
Rossi-Stacconi MV, Panel A, Baser N et al (2017) Comparative life history traits of indigenous Italian parasitoids of Drosophila suzukii and their effectiveness at different temperatures. Biol Control 112:20–27. https://doi.org/10.1016/j.biocontrol.2017.06.003
SAG Servicio Agrícola y Ganadero (2019) Agrícola/Plagas y Enfermedades/Plagas Relevantes Presentes/Drosophila suzukii. https://www.sag.gob.cl/ambitos-de-accion/drosophila-suzukii. Accessed 16 May 2020
Sampson BJ, Mallette T, Addesso KM et al (2016) Novel aspects of Drosophila suzukii (Diptera: Drosophilidae) biology and an improved method for culturing this invasive species with a modified D. melanogaster diet. Fla Entomol 99(4):774–780. https://doi.org/10.1653/024.099.0433
Santadino MV, Riquelme Virgala MB, Ansa MA et al (2015) First record of Drosophila suzukii (Diptera: Drosophilidae) in blueberry (Vaccinium spp.) from Argentina. Rev Soc Entomol Argen 74(3-4):183–185
Santos RSS (2014) Ocorrência de Drosophila suzukii (Matsumura, 1931) (Diptera: Drosophilidae) atacando frutos de morango no Brasil. http://ainfo.cnptia.embrapa.br/digital/bitstream/item/106368/1/Comunicado-Tecnico-159.pdf. Accessed 20 Apr 2020
Sapir Y, Mazer SJ, Holzapfel C (2008) Sex ratio. In: Erik JS, Brian F (eds) Encyclopedia of ecology. Academic, Oxford, pp 3243–3248
Sarto V, Sorribas R (2011) Drosophila suzukii (Matsumura 1931) nueva amenaza para las producciones agrícolas. Phytoma España 234:1–6
Sasaki M, Sato R (1995) Bionomics of the cherry Drosophila, Drosophila suzukii Matsumura (Diptera: Drosophilidae) in Fukushima Prefecture. Ann Rep Plant Prot North Jpn 46:170–172
Schlesener DCH, Wollmann J, Pazini JB et al (2017) Effects of insecticides on adults and eggs of Drosophila suzukii (Diptera, Drosophilidae). Rev Colomb Entomol 43(2):208–214. https://doi.org/10.25100/socolen.v43i2.5945
Schlesener DCH, Wollmann J, Kruger AP et al (2018) Biology and fertility life table of Drosophila suzukii on artificial diets. Entomol Exp Appl 166(11–12):932–936. https://doi.org/10.1111/eea.12736
Schlesener DCH, Wollmann J, Pazini JB et al (2019) Insecticide toxicity to Drosophila suzukii (Diptera: Drosophilidae) parasitoids: Trichopria anastrephae (Hymenoptera: Diapriidae) and Pachycrepoideus vindemmiae (Hymenoptera: Pteromalidae). J Econ Entomol 112(3):1197–1206. https://doi.org/10.1093/jee/toz033
Schlesener DCH, Wollmann J, Kruger AP et al (2020) Effect of temperature on reproduction, development, and phenotypic plasticity of Drosophila suzukii in Brazil. Entomol Exp Appl. https://doi.org/10.1111/eea.12954
Shaw B, Brain P, Wijnen H et al (2018) Reducing Drosophila suzukii emergence through inter-species competition. Pest Manag Sci 74(6):1466–1471. https://doi.org/10.1002/ps.4836
Shearer PW, West JD, Walton VM et al (2016) Seasonal cues induce phenotypic plasticity of Drosophila suzukii to enhance winter survival. BMC Ecol 16:11. https://doi.org/10.1186/s12898-016-0070-3
Shrader ME (2017) Drosophila suzukii (Matsumura) (Diptera: Drosphilidae): risk assessment for an invasive vinegar fly in Virginia vineyards. Dissertation, Virginia Polytechnic Institute and State University
Shrader ME, Burrack HJ, Pfeiffer DG (2020) Effects of interspecific larval competition on developmental parameters in nutrient sources between Drosophila suzukii (Diptera: Drosophilidae) and Zaprionus indianus. J Econ Entomol 113(1):230–238. https://doi.org/10.1093/jee/toz297
Skalski JR, Ryding KE, Millspaugh JJ (2005) Wildlife demography: analysis of sex, age, and count data. Academic, San Diego
Snellings Y, Herrera B, Wildemann B et al (2018) The role of cuticular hydrocarbons in mate recognition in Drosophila suzukii. Sci Rep 8:4996. https://doi.org/10.1038/s41598-018-23189-6
Steffan SA, Lee JC, Singleton ME et al (2013) Susceptibility of cranberries to Drosophila suzukii (Diptera: Drosophilidae). J Econ Entomol 106(6):2424–2427. https://doi.org/10.1603/EC13331
Stephens AR, Asplen MK, Hutchison WD et al (2015) Cold hardiness of winter-acclimated Drosophila suzukii (Diptera: Drosophilidae) adults. Environ Entomol 44(6):1619–1626. https://doi.org/10.1093/ee/nvv134
Stewart TJ (2015) Agricultural landscape ecology of spotted wing drosophila in the central valley of California. Dissertation, Jordan College of Agricultural Sciences and Technology California State University
Stockton D, Wallingford A, Loeb G (2018) Phenotypic plasticity promotes overwintering survival in a globally invasive crop pest, Drosophila suzukii. Insects 9:105. https://doi.org/10.3390/insects9030105
Stockton D, Wallingford A, Rendon D et al (2019) Interactions between biotic and abiotic factors affect survival in overwintering Drosophila suzukii (Diptera: Drosophilidae). Environ Entomol 48(2):454–464. https://doi.org/10.1093/ee/nvy192
Sward GFH (2017) Evaluating host plant use by spotted wing Drosophila, Drosophila suzukii, in Minnesota. Dissertation, University of Minnesota
Swoboda-Bhattarai KA, Burrack HJ (2016) Drosophila suzukii infestation in ripe and ripening caneberries. Acta Hortic 1133(65):419–430. https://doi.org/10.17660/ActaHortic.2016.1133.65
Tait G, Cabianca A, Grassi A et al (2020) Drosophila suzukii daily dispersal between distinctly different habitats. Entomol Gen 40(1):25–37. https://doi.org/10.1127/entomologia/2019/0876
Thistlewood HMA, Gill P, Beers EH et al (2018) Spatial analysis of seasonal dynamics and overwintering of Drosophila suzukii (Diptera: Drosophilidae) in the Okanagan-Columbia Basin, 2010-2014. Environ Entomol 47(2):221–232. https://doi.org/10.1093/ee/nvx178
Tochen S, Dalton DT, Wiman N et al (2014) Temperature related development and population parameters for Drosophila suzukii (Diptera: Drosophilidae) on cherry and blueberry. Environ Entomol 43(2):501–510. https://doi.org/10.1603/EN13200
Tonina L, Mori N, Giomi F, Battisti A (2016) Development of Drosophila suzukii at low temperatures in mountain areas. J Pest Sci 89:667–678. https://doi.org/10.1007/s10340-016-0730-2
Toxopeus J, Jakobs R, Ferguson LV et al (2016) Reproductive arrest and stress resistance in winter-acclimated Drosophila suzukii. J Insect Physiol 89:37–51. https://doi.org/10.1016/j.jinsphys.2016.03.006
Vilaire A, Lavine LC, Walsh DB (2011) The interspecific competition between invasive Drosophila suzukii and endemic Drosophila melanogaster on fruit hosts. In: Abstracts of the Conference: Entomological Society of America Annual Meeting, Reno, NV, 13–16 November 2011
Vilela CR, Mori L (2014) The invasive spotted-wing Drosophila (Diptera, Drosophilidae) has been found in the city of São Paulo (Brazil). Rev Bras Entomol 58(4):371–375. https://doi.org/10.1590/S0085-56262014000400004
Wallingford AK, Lee JC, Loeb GM (2016) The influence of temperature and photoperiod on the reproductive diapause and cold tolerance of spotted-wing drosophila, Drosophila suzukii. Entomol Exp Appl 159(3):327–337. https://doi.org/10.1111/eea.12443
Walsh DB, Bolda MP, Goodhue RE et al (2011) Drosophila suzukii (Diptera: Drosophilidae): invasive pest of ripening soft fruit expanding its geographic range and damage potential. J Integr Pest Manag 2(1):G1–G7. https://doi.org/10.1603/IPM10010
Wang XG, Kaçar G, Biondi A et al (2016a) Life-history and host preference of Trichopria drosophilae, a pupal parasitoid of spotted wing drosophila. Biocontrol 61:387–397. https://doi.org/10.1007/s10526-016-9720-9
Wang XG, Kaçar G, Biondi A et al (2016b) Foraging efficiency and outcomes of interactions of two pupal parasitoids attacking the invasive spotted wing Drosophila. Biol Control 96:64–71. https://doi.org/10.1016/j.biocontrol.2016.02.004
Wang XG, Stewart TJ, Biondi A et al (2016c) Population dynamics and ecology of Drosophila suzukii in Central California. J Pest Sci 89(3):701–712. https://doi.org/10.1007/s10340-016-0747-6
Wang XG, Nance AH, Jones JML et al (2018a) Aspects of the biology and reproductive strategy of two Asian larval parasitoids evaluated for classical biological control of Drosophila suzukii. Biol Control 121:58–65. https://doi.org/10.1016/j.biocontrol.2018.02.010
Wang XG, Serrato MA, Son Y et al (2018b) Thermal performance of two indigenous pupal parasitoids attacking the invasive Drosophila suzukii (Diptera: Drosophilidae). Environ Entomol 47(3):764–772. https://doi.org/10.1093/ee/nvy053
Wang XG, Kaçar G, Daane KM (2019a) Temporal dynamics of host use by Drosophila suzukii in California’s San Joaquin Valley: implications for area-wide pest management. Insects 10(7):206. https://doi.org/10.3390/insects10070206
Wang XG, Hogg BN, Hougardy E et al (2019b) Potential competitive outcomes among three solitary larval endoparasitoids as candidate agents for classical biological control of Drosophila suzukii. Biol Control 130:18–26. https://doi.org/10.1016/j.biocontrol.2018.12.003
Wang XG, Biondi A, Daane KM (2020) Functional responses of three candidate Asian larval parasitoids evaluated for classical biological control of Drosophila suzukii (Diptera: Drosophilidae). J Econ Entomol 113(1):73–80. https://doi.org/10.1093/jee/toz265
Weydert C, Mandrin JF (2013) Le ravageur émergent Drosophila suzukii: situation en France et connaissances acquises en verger (2eme partie). Infos CTIFL 292:32–40
Weydert C, Trottin Y, Mandrin JF et al (2016) Drosophila suzukii connaissance du ravageur, moyens de protection. Infos CTIFL:1–16
Wilson HE, Hamby KA, Zalom FG (2013) Host susceptibility of “French Prune” Prunus domestica to Drosophila suzukii (Diptera: Drosophilidae). Acta Hortic (985):249–254. https://doi.org/10.17660/ActaHortic.2013.985.32
Wiman NG, Walton VM, Dalton DT et al (2014) Integrating temperature-dependent life table data into a matrix projection model for Drosophila suzukii population estimation. PLoS One 9(9):e106909. https://doi.org/10.1371/journal.pone.0106909
Wolf S, Baur H, Collatz J (2019) Life history of Vrestovia fidenas, a potential control agent of Drosophila suzukii. Biocontrol 64(3):263–275. https://doi.org/10.1007/s10526-019-09933-5
Wollmann J, Schlesener DCH, Ferreira MS et al (2016) Parasitoids of Drosophilidae with potential for parasitism on Drosophila suzukii in Brazil. Dros Info Serv 99:38–42
Wollmann J, Schlesener DCH, Ferreira MS et al (2019) Population dynamics of Drosophila suzukii (Diptera: Drosophilidae) in berry crops in southern Brazil. Neotrop Entomol 48(4):699–705. https://doi.org/10.1007/s13744-019-00686
Woltering SB, Romeis J, Collatz J (2019) Influence of the rearing host on biological parameters of Trichopria drosophilae, a potential biological control agent of Drosophila suzukii. Insects 10(6):183. https://doi.org/10.3390/insects10060183
Woltz JM, Lee JC (2017) Pupation behavior and larval and pupal biocontrol of Drosophila suzukii in the field. Biol Control 110:62–69. https://doi.org/10.1016/j.biocontrol.2017.04.007
Woltz JM, Donahue KM, Bruck DJ et al (2015) Efficacy of commercially available predators, nematodes and fungal entomopathogens for augmentative control of Drosophila suzukii. J Appl Entomol 139:759–770. https://doi.org/10.1111/jen.12200
Yi C, Cai P, Lin J et al (2020) Life history and host preference of Trichopria drosophilae from Southern China, one of the effective pupal parasitoids on the Drosophila Species. Insects 11(2):103. https://doi.org/10.3390/insects11020103
Yousef M, Aranda-Valera E, Quesada-Moraga E (2018) Lure-and-infect and lure-and-kill devices based on Metarhizium brunneum for spotted wing drosophila control. J Pest Sci 91:227–235. https://doi.org/10.1007/s10340-017-0874-8
Yu D, Zalom FG, Hamby KA (2013) Host status and fruit odor response of Drosophila suzukii (Diptera: Drosophilidae) to figs and mulberries. J Econ Entomol 106(4):1932–1937. https://doi.org/10.1603/EC12480
Zengin E, Karaca I (2019) Dynamics of trapped adult populations of Drosophila suzukii Matsumura (Diptera: Drosophilidae) and its parasitoids in Uşak Province, Turkey. Egypt J Biol Pest Co 29:43. https://doi.org/10.1186/s41938-019-0147-3
Zerulla FN, Schmidt S, Streitberger M et al (2015) On the overwintering ability of Drosophila suzukii in South Tyrol. J Berry Res 5:41–48. https://doi.org/10.3233/JBR-150089
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kirschbaum, D.S., Funes, C.F., Buonocore-Biancheri, M.J., Suárez, L., Ovruski, S.M. (2020). The Biology and Ecology of Drosophila suzukii (Diptera: Drosophilidae). In: Garcia, F.R.M. (eds) Drosophila suzukii Management. Springer, Cham. https://doi.org/10.1007/978-3-030-62692-1_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-62692-1_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-62691-4
Online ISBN: 978-3-030-62692-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)