Abstract
Von Hippel–Lindau syndrome (VHL-syndrome) is a rare hereditary tumor syndrome predisposing affected patients to hemangioblastomas in the central nervous system (CNS) and retina, kidney cancer and also to neuroendocrine tumors including pheochromocytoma and pancreatic neuroendocrine tumors.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
FormalPara OpeningVon Hippel–Lindau syndrome (VHL-syndrome) is a rare hereditary tumor syndrome predisposing affected patients to hemangioblastomas in the central nervous system (CNS) and retina, kidney cancer and also to neuroendocrine tumors including pheochromocytoma and pancreatic neuroendocrine tumors.
FormalPara Definition of the DiseaseThe VHL-syndrome is a rare tumor syndrome (prevalence: 1:36,000) inherited as an autosomal dominant trait. The syndrome is caused by germ-line mutations in the tumor suppressor VHL-gene. The major manifestations of the VHL-syndrome include hemangioblastomas in the central nervous system (mostly in the spinal cord and cerebellum) and retina (retinal angioma), renal cysts and clear cell renal carcinoma, endolymphatic sac tumors of the middle ear, cysts, serous cystadenomas and neuroendocrine tumors in the pancreas, and cystadenomas of the epididymis. There are two major subtypes of VHL (◘ Table 52.1): in VHL type 1, there is usually no pheochromocytoma/paraganglioma, whereas in type 2 pheochromocytoma/paraganglioma can develop. The only manifestation in VHL type 2C is pheochromocytoma that is often bilateral. The risk for renal cancer is low in VHL type 2A and high in type 2B. Patients suffering from VHL usually die due to complications related to central nervous system tumors and renal cell cancer. Pheochromocytoma or rarely paraganglioma is observed in 10–30% of VHL-syndrome patients, and as these tumors secrete predominantly norepinephrine, the typical symptoms of pheochromocytoma are lacking. Pheochromocytoma in VHL is rarely metastatic. In sporadic cases, VHL-syndrome is often diagnosed due to central nervous system manifestations.
The pathogenesis of VHL-syndrome is related to the activation of hypoxia-inducible factor 1α that stimulates angiogenesis and the product of VHL-gene is implicated in its degradation. Due to the increased angiogenesis, tumors are usually highly vascularized.
FormalPara TipsThe reader is advised to read the chapter on pheochromocytoma (► Chap. 37), the chapters on intestinal and pancreatic neuroendocrine tumors (► Chaps. 44, 46 and 48), and the previous chapter on MEN2 syndrome (► Chap. 51).
FormalPara Take Home Messages-
Von Hippel–Lindau syndrome (VHL-syndrome) is a rare hereditary tumor syndrome inherited as an autosomal dominant trait.
-
VHL-syndrome predisposes patients to central nervous system hemangioblastomas, retinal angiomas, renal cysts and cancer, pancreatic cysts and cystadenomas, endolymphatic sac tumors, and epididymis cystadenomas.
-
Endocrine manifestations of VHL-syndrome include pheochromocytoma and pancreatic neuroendocrine tumors.
-
VHL-patients usually die of complications associated with central nervous system hemangioblastomas and renal cancer. Pheochromocytoma is rarely metastatic, and pancreatic neuroendocrine tumors are usually indolent.
Cerebellar hemangioblastoma (Lindau tumor) should wake the suspicion for von Hippel–Lindau syndrome. What should be done next?
Hemangioblastomas are uncommon tumors of the central nervous system, and these are important manifestations of the VHL-syndrome. About 40% of VHL-syndrome-associated hemangioblastomas occur in the cerebellum, whereas about 50% in the spinal cord.
A family tree should be taken to search for a potential hereditary case (◘ Fig. 52.1 represents the patient’s family tree), but the syndrome can occur sporadically as well. Genetic testing is mandatory. Careful history taking is important. Ophthalmological examination should be done for the potential retinal angiomas.
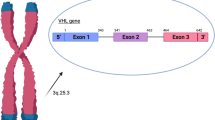
Genetic testing showed a heterozygous Glu94 STOP mutation in the first exon of the VHL-gene that is a known pathogenic mutation of the VHL-syndrome. What is the chance for passing this mutation to the offspring?
Being a heterozygous mutation, there is a 50% chance of passing it to the next generation. As VHL is inherited as an autosomal dominant trait, one mutant allele is enough for disease manifestation, and therefore, half of the offspring can be expected to manifest the disease.
What kind of imaging should be performed?
Abdominal imaging preferably by MRI (or computed tomography (CT)) should be done to investigate the kidneys, pancreas, and adrenals. The spine should be examined for potential spinal cord tumors.
Which hormones should be measured?
Pheochromocytoma/paraganglioma and pancreatic neuroendocrine tumors represent the endocrine manifestations of the VHL-syndrome. Urinary or plasma metanephrines (► Chap. 37) and chromogranin A (CgA) as the general neuroendocrine tumor marker should be taken (► Chap. 44). Pancreatic neuroendocrine tumors in VHL are usually hormonally inactive and indolent, but rarely can metastasize to the liver. All hormones measured were in the normal range.
Should the pancreas tumor be removed?
VHL is associated with neuroendocrine tumors in the pancreas that are usually slowly growing and indolent, and despite having metastatic potential, patients usually do not die of complications related to these. Tumors with a diameter under 3 cm should only be followed up.
Even for kidney tumors that have more serious clinical consequences than pancreatic neuroendocrine tumors in VHL, current clinical practice is rather conservative and aims to preserve as much normal kidney as possible. Kidney-sparing operations are preferred, and small tumors (<3 cm diameter) are usually only followed up.
How should the patient be followed up?
In patients older than 10 years of age, annual physical and ophthalmological examinations, abdominal ultrasound, and plasma/urinary metanephrines can be proposed. Abdominal, brain, and spine MRI should be performed every 2 years along with audiological control (for endolymphatic sac tumors of the middle ear).
Is there a medical treatment option available for VHL-syndrome?
There are reports on the successful use of the tyrosine kinase inhibitor sunitinib having anti-angiogenetic effects in patients suffering from VHL-syndrome. Increased angiogenesis is a major pathogenic feature of VHL-syndrome. Sunitinib is already in use for many years in the treatment of sporadic clear cell renal cancer, and it is also actively used in the treatment of differentiated pancreatic neuroendocrine tumors. Moreover, trials are ongoing for the use of sunitinib in metastatic pheochromocytoma.
Case Presentation
The male patient complained of serious dizziness at the age of 22, and brain magnetic resonance imaging (MRI) revealed a cerebellar tumor. The tumor could be removed, and its histological examination revealed a hemangioblastoma. Four years later, a control MRI revealed a recurrent cerebellar tumor of 4 × 5 cm that was again successfully operated.
Case Continued
Abdominal CT showed multiple cysts in the pancreas and the kidneys (◘ Figs. 52.2a and b), but the adrenals were normal. In the left kidney, the suspicion for renal cancer was raised (solid tumor of 24 × 20 mm). A urological consultation was requested, but due to the small size (<3 cm diameter) of the tumor, only observation was proposed. A small lesion suspicious for tumor was described in the pancreas at the head-corpus boundary (◘ Fig. 52.2a).
Suggested Reading
Bjorklund P, Pacak K, Crona J. Precision medicine in pheochromocytoma and paraganglioma: current and future concepts. J Intern Med. 2016;280(6):559–73. https://doi.org/10.1111/joim.12507.
Crespigio J, Berbel LCL, Dias MA, Berbel RF, Pereira SS, Pignatelli D, Mazzuco TL. Von Hippel-Lindau disease: a single gene, several hereditary tumors. J Endocrinol Investig. 2018;41(1):21–31. https://doi.org/10.1007/s40618-017-0683-1.
Neumann HP, Young WF Jr, Krauss T, Bayley JP, Schiavi F, Opocher G, Boedeker CC, Tirosh A, Castinetti F, Ruf J, Beltsevich D, Walz M, Groeben HT, von Dobschuetz E, Gimm O, Wohllk N, Pfeifer M, Lourenco DM Jr, Peczkowska M, Patocs A, Ngeow J, Makay O, Shah NS, Tischler A, Leijon H, Pennelli G, de Las Heras KVG, Links TP, Bausch B, Eng C. 65 Years of the double helix: genetics informs precision practice in the diagnosis and management of pheochromocytoma. Endoc Relat Cancer. 2018;25(8):T201–t219. https://doi.org/10.1530/erc-18-0085.
Nielsen SM, Rhodes L, Blanco I, Chung WK, Eng C, Maher ER, Richard S, Giles RH. Von Hippel-Lindau disease: genetics and role of genetic counseling in a multiple neoplasia syndrome. J Clin Oncol. 2016;34(18):2172–81. https://doi.org/10.1200/jco.2015.65.6140.
Oudard S, Elaidi R, Brizard M, Le Rest C, Caillet V, Deveaux S, Benoit G, Correas JM, Benoudiba F, David P, Gaudric A, Hammel P, Joly D, Timsit MO, Mejean A, Richard S. Sunitinib for the treatment of benign and malignant neoplasms from von Hippel-Lindau disease: a single-arm, prospective phase II clinical study from the PREDIR group. Oncotarget. 2016;7(51):85306–17. https://doi.org/10.18632/oncotarget.13301.
Sarkadi B, Patocs A. Hereditary diseases predisposing to pheochromocytoma (VHL, NF-1, paraganglioma syndromes, and novel genes). Experientia Suppl. 2019;111:129–47. https://doi.org/10.1007/978-3-030-25905-1_8.
Tsang SH, Sharma T. Von Hippel-Lindau disease. Adv Exp Med Biol. 2018;1085:201–3. https://doi.org/10.1007/978-3-319-95046-4_42.
Varshney N, Kebede AA, Owusu-Dapaah H, Lather J, Kaushik M, Bhullar JS. A review of Von Hippel-Lindau syndrome. J Kidney Cancer VHL. 2017;4(3):20–9. https://doi.org/10.15586/jkcvhl.2017.88.
Yuan G, Liu Q, Tong D, Liu G, Yi Y, Zhang J, Zhang Y, Wang LA, Wang L, Chen R, Guan Y, Yi X, Lan W, Jiang J. A retrospective case study of sunitinib treatment in three patients with Von Hippel-Lindau disease. Cancer Biol Ther. 2018;19(9):766–72. https://doi.org/10.1080/15384047.2018.1470732.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Igaz, P. (2021). Von Hippel–Lindau Syndrome. In: Igaz, P. (eds) Practical Clinical Endocrinology. Springer, Cham. https://doi.org/10.1007/978-3-030-62011-0_52
Download citation
DOI: https://doi.org/10.1007/978-3-030-62011-0_52
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-62010-3
Online ISBN: 978-3-030-62011-0
eBook Packages: MedicineMedicine (R0)