Abstract
Immune and inflammatory myopathies (IIM) are characterized by weakness with muscle inflammation and are frequently accompanied by extra-muscular manifestations. Previously, patients were diagnosed with dermatomyositis if a characteristic rash accompanied muscle involvement or polymyositis if no rash was present. The discovery of unique clinical features, autoantibodies, and histopathological patterns that associate with specific clinical phenotypes has been instrumental in understanding IIM and predicting treatment response and prognosis. While numerous classification systems exist, the most commonly agreed-upon criteria recognize four main categories: dermatomyositis, sporadic inclusion body myositis, antisynthetase syndrome, and immune-mediated necrotizing myopathy. Treatment still largely relies on expert opinion and empirical use of corticosteroids and steroid-sparing agents. A deeper understanding of the molecular pathways that drive pathogenesis and accurate disease classification will aid clinical research and the development of more precision therapies. This chapter discusses current knowledge of the epidemiology, clinical characteristics, diagnostic evaluation, classification, pathogenesis, treatment, and prognosis of IIM.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Myositis
- Immune and inflammatory myopathy
- Dermatomyositis
- Polymyositis
- Inclusion body myositis
- Antisynthetase syndrome
- Immune-mediated necrotizing myopathy
- Myositis-specific antibodies
-
1.
Immune and inflammatory myopathies (IIM) are characterized by weakness with immune or inflammatory changes on muscle biopsy and commonly have extra-muscular manifestations.
-
2.
Unique clinical features, autoantibodies, and histopathological patterns are used to phenotypically categorize patients and predict treatment response and prognosis.
-
3.
The most commonly agreed-upon criteria recognize four main categories: dermatomyositis, sporadic inclusion body myositis, antisynthetase syndrome, and immune-mediated necrotizing myopathy.
-
4.
Excluding sporadic inclusion body myositis, IIM treatment still largely relies on empirical use of corticosteroids and steroid-sparing agents.
Introduction
Immune and inflammatory myopathies (IIM), collectively known as myositis, are characterized by progressive weakness and inflammatory cellular infiltrates within skeletal muscle. Damage to specific tissues within skeletal muscle, such as connective tissue or blood vessels, may cause syndromes involving multiple organ systems other than muscle, including skin, lungs, and joints. IIM subtypes have historically been defined by clinical and histopathological differences and traditionally were classified as polymyositis (PM) or dermatomyositis (DM) . Progress has been made in revising Bohan and Peter’s original diagnostic criteria from 1975 in order to more accurately align clinical, autoantibody, and histopathological data with prognosis and response to treatment [1, 2]. PM has been overvalued [3, 4], and pathologic criteria isolated two new subgroups, previously referred to as PM, including sporadic inclusion body myositis (sIBM) [1, 2] and immune-mediated necrotizing myopathy (IMNM) [2]. These classification approaches, however, define overlapping entities. For example, antisynthetase syndrome is often classified as DM, as PM, or as an overlap syndrome [5,6,7]. Myopathology [8] and autoantibodies can help define subgroups of patients in terms of clinical or pathologic phenotypes, prognosis, and response to treatment [9,10,11,12,13]. The most up-to-date, and commonly accepted, classification criteria will be used in this chapter and eliminates PM as a distinct entity recognizing only DM, IMNM, sIBM, and antisynthetase syndrome [14]. PM does not represent a subgroup of patients, and use of this term should probably be discontinued [14]. It is important to recognize that all classification criteria have their drawbacks. A major issue with the criteria used for this chapter is that it fails to account for other forms of IIM, such as brachio-cervical inflammatory myopathy, focal myositis, eosinophilic myositis, and granulomatous myopathies. This chapter describes current knowledge of the epidemiology, clinical characteristics, diagnostic evaluation, classification, pathogenesis, treatment, and prognosis of IIM.
Epidemiology
Most epidemiologic studies have used criteria that fail to distinguish sIBM, IMNM, and antisynthetase syndrome (discussed previously), causing inaccuracies in incidence and prevalence studies and making it easier to analyze data for IIM as a collective group. Incidence rates for IIM range between 4.27 and 7.89 per 100,000 person years, and prevalence ranges from 9.54 to 32.74 cases per 100,000 individuals [15,16,17]. sIBM prevalence has been reported as 9.3 per million [18]. Using recent classifications, of all IIMs, sIBM accounts for 29.6%, IMNM 35%, DM 20%, and antisynthetase syndrome 15.4% [14]. DM, IMNM, and antisynthetase syndromes occur more frequently in females [19]. DM may affect children and adults [19], whereas sIBM is seen more commonly in male patients over the age of 50 [20]. Mean annual medical costs and number of ambulatory visits, specialty visits, and inpatient hospital stays are significantly higher among subjects with IIM compared to matched controls [21].
Clinical Features
Dermatomyositis
Dermatomyositis may present with subacute onset progressive proximal weakness, cutaneous manifestations, or both [22]. The deltoids tend to be more severely affected [14]. Some patients may present with only skin changes and are considered to have hypomyopathic or amyopathic forms of disease [23]. Others may present with isolated muscle weakness and never develop rash or only develop rash months later [24]. Juvenile patients may present initially with a febrile illness [24]. Pathognomonic skin features include violaceous periorbital edema (heliotrope rash) and papular lesions on the extensor surfaces of metacarpophalangeal and interphalangeal joints, Gottron’s papules (Fig. 20.1a). Other findings may include an erythematous rash over extensor surfaces of limbs (Gottron’s sign), over the neck and chest (V sign), and over the back of the neck and shoulders (shawl sign), limb edema, alopecia, skin ulcers, calcinosis, and panniculitis (Fig. 20.1b–f) [14]. Lesions may be photosensitive and pruritic [25]. Juvenile patients more commonly develop cutaneous calcinosis (30–70% of juvenile cases and 10% of adult cases) over pressure points [25, 26]. Myalgias may also be present [27].
Immune-Mediated Necrotizing Myopathy
IMNM is typically characterized by rapid progression of severe proximal weakness, with prominent involvement of the psoas muscles and exceptionally high creatine kinase (CK). Toxic or drug-induced etiologies, as well as some hereditary myopathies (e.g., limb-girdle muscular dystrophy), may appear similar to IMNM and should be ruled out [28,29,30]. Patients may have mild myalgias or have no muscle pain whatsoever [31]. Extra-muscular manifestations are generally mild if they occur [32,33,34].
Antisynthetase Syndromes
Overlap myositis occurs when a patient has an autoimmune myopathy associated with other autoimmune diseases such as systemic lupus erythematosus, rheumatoid arthritis, Sjögren’s syndrome, or systemic sclerosis [35]. Antisynthetase syndrome, with autoantibodies targeting aminoacyl tRNA synthetases, is the most representative form of overlap myositis [36]. Patients may present with a combination of inflammatory myopathy, interstitial lung disease (ILD), arthritis, Raynaud syndrome, fever, or hyperkeratotic finger lesions called mechanic’s hands (Fig. 20.1g) [37]. Antisynthetase syndrome may also cause skin rashes similar to dermatomyositis [37]. Myopathic features include proximal weakness similar to DM, although some patients may have no weakness at all and clinical manifestations of muscle disease may be limited to myalgias in isolation.
sIBM
sIBM often presents slowly with progression over 5–8 years before affected patients come to medical attention [18, 38]. Characteristic findings include asymmetric wasting and weakness of the wrist flexors, deep finger flexors, and quadriceps muscles (Fig. 20.2a, b) [24]. Tibialis anterior weakness, dysphagia, and mild facial weakness may also be present [39,40,41,42]. In a study of 57 patients with sIBM, the initial presenting symptoms were quadriceps weakness (79%), finger weakness (12%), foot drop (7%), and dysphagia (1.8%) [43]. Asymmetric involvement was very common (82%), with the patient’s non-dominant side commonly being more severely affected [43]. There may be evidence of a generalized sensory peripheral neuropathy on clinical exam [44]. Up to 15% of sIBM patients have a coexisting autoimmune disorder or condition with altered immune function [45]. Sporadic IBM is not associated with heart disease [39] or an increased risk of malignancy [46]. Primary respiratory failure is rare; however, progressive dysphagia may occur and may lead to aspiration [40, 41].
Diagnostic Evaluation
Elevated Muscle Enzymes
Serum CK levels are a sensitive measure of muscle disease activity in IIM [24]. They do not correlate well with disease activity when comparing different patients, but they can reflect changes in disease activity within an individual patient. Levels are typically highest in IMNM (2300 U/L–7000 U/L) and lowest in sIBM (160 U/L–793 U/L) [14]. Aldolase levels may also be prominently elevated, presumably from intramuscular connective tissue damage. For example, antisynthetase syndromes with perimysial pathology may have isolated aldolase elevation [42]. Other muscle enzymes, including myoglobin, lactate dehydrogenase, aspartate aminotransferase, and alanine aminotransferase, may also be elevated. Patients taking hepatotoxic steroid-sparing agents, such as azathioprine (AZA) or methotrexate, may develop elevated transaminases. The liver enzyme gamma-glutamyl transpeptidase (GGT) can aid in differentiating liver damage in IIM patients, as it is not released by damaged muscle [47].
Electrodiagnostics
Electromyography (EMG) typically reveals an irritable myopathic pattern characterized by increased insertional and spontaneous activity (fibrillation potentials, positive sharp waves, and occasionally complex repetitive discharges), polyphasic motor unit action potentials (MUAPs) with small duration and low amplitude, and early MUAP recruitment. sIBM patients may have evidence of neuropathy on nerve conduction studies and mixed myopathic and neurogenic changes on EMG [44].
Muscle Imaging
Muscle MRI in sIBM patients demonstrates severe involvement of the anterior compartment of the thigh and forearm [48]. DM, IMNM, and antisynthetase syndrome patients often have a nonspecific pattern with hyperintensities on intramuscular T2-weighted magnetic resonance imaging (MRI) scans [48, 49]. Some recommend using muscle MRI to select the site of muscle biopsy [50]. Caution should be used with this approach as neurogenic changes from denervation appear similar to changes related to myositis on MRI. MRI also cannot distinguish between IIM and hereditary myopathies [51].
Antibodies
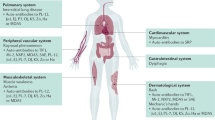
A screen for autoantibodies is common in the evaluation of patients with IIM or suspected IIM. However, their role in the pathophysiology of IIM is unclear. Some may be directly involved in pathophysiology and others simply an epiphenomenon. Antibodies are categorized as myositis-associated autoantibodies (MAAs) or myositis-specific autoantibodies (MSAs). MSAs are found predominantly in the serum of patients with IIM, but are not 100% specific for IIM [52, 53]. MAAs are primarily encountered in other connective tissue diseases and occasionally found in patients with IIM [52, 53]. MSAs can help classify homogenous phenotypic subsets of patients and help predict the degree of muscle, skin, and lung involvement, as well as risk of an associated malignancy (Table 20.1) [14, 29, 32, 33, 41, 52, 54,55,56,57,58,59,60,61,62,63]. Recent classification schemes suggest MSAs are crucial for accurate categorization of IIM [14].
Dermatomyositis
Approximately 70% of patients with DM have a dermatomyositis-specific autoantibody [52], many associated with a unique clinical phenotype (Table 20.1) [56,57,58,59,60,61]. Autoantibodies against Mi2, a nuclear antigen, are associated with classic DM characteristics, severe skin manifestations, proximal weakness, and a lower risk of associated malignancy relative to DM associated with other MSAs [56]. DM patients with autoantibodies against nuclear matrix protein NXP2 are more likely to present with both proximal and distal muscle weakness, subcutaneous edema, and dysphagia and are more prone to develop calcinosis [58]. Patients with anti-NXP2 or anti-transcription intermediary factor (TIF)-1 autoantibodies are associated with increased risk of malignancy within 3 years of diagnosis. Accordingly, comprehensive cancer screening or positron emission tomography-computed tomography (PET-CT) scans are particularly important for these patients [57, 58, 64, 65]. DM patients with antibodies against small ubiquitin-like modifier activating enzyme (SAE) or melanoma differentiation-associated gene 5 (MDA5) typically have more cutaneous than muscle involvement [59,60,61, 66]. MDA5 patients are commonly hypomyopathic or amyopathic and may develop ulcers on the palmar surface of their hands and a rapidly progressive form of ILD [59, 60, 66]. IIM patients suspected to have interstitial lung disease should initially be evaluated and monitored using pulmonary function tests (carbon monoxide diffusion and inspiratory and expiratory pressures) and high-resolution CT scans.
IMNM
Several autoantibodies associated with IMNM have been identified, each with specific characteristics and clinical outcomes (Table 20.1) [29, 32, 33, 62, 63]. These include anti-signal recognition particle (SRP) and anti-3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR) autoantibodies. Patients with SRP or HMGCR antibodies often share several features, including high CK, and an aggressive refractory disease course in some patients [62]. The IMNM classification does not perfectly overlap with all patients with SRP or HMGCR antibodies. Only two-thirds of IMNM patients are reported to have antibodies to SRP or HMGCR, and around 20% of patients with SRP or HMGCR antibodies do not have key histopathology characteristics of IMNM [33, 63, 67]. Only two-thirds of HMGCR patients have necrosis or regeneration, and one-third have lymphocytic infiltrates [68]. Approximately 60% of these patients will have prominent perimysial pathology, and as high as 37% will have systemic features such as ILD and skin rash, features more commonly seen with overlap or antisynthetase syndromes [68]. The association between statin usage and increased risk of developing IMNM associated with HMGCR antibodies is a subject of ongoing debate [68]. However, it is clear that some patients do have a form of disease triggered by exposure to statins, likely from feedback mechanisms that lead to increased HMGCR expression in muscle tissue [29]. Patients with HMGCR antibodies might have an increased risk of malignancy [69]. Some patients may even present with a slowly progressive disease course and be misdiagnosed with limb-girdle muscular dystrophy [29]. SRP patients tend to have more severe weakness than HMGCR patients [32, 62]. In addition to necrosis and regeneration, SRP muscle pathology demonstrates prominent endomysial fibrosis and capillary pathology [70]. SRP patients may be at greater risk for developing interstitial lung disease and possibly cardiac involvement when compared to patients with HMGCR antibodies [62, 71]. If cardiac involvement is suspected, an electrocardiogram (ECG) and echocardiogram should be performed. Seronegative IMNM is thought to be associated with increased risk of malignancy, female predominance, frequent occurrence of associated connective tissue disorders, and increased risk of extra-muscular disease activity [69, 72].
Antisynthetase Syndrome
Autoantibodies against histidyl (anti-Jo-1)-, threonyl (anti-PL7)-, and alanyl (anti-PL12)-tRNA synthetases are the most common [36, 73]. About 90% of patients with anti-Jo-1 autoantibodies have an inflammatory myopathy, while approximately 50% of patients with anti-PL12 autoantibodies present with interstitial lung disease but no muscle involvement [36]. Muscle weakness tends to be more severe in patients with anti-Jo-1 autoantibodies, while lung involvement is more severe in patients with anti-PL7 and anti-PL12 autoantibodies [36, 73].
Sporadic IBM
Autoantibodies against cytosolic 5′-nucleotidase 1A (NT5C1A) are present in 30–60% of patients with sIBM. NT5C1A autoantibodies are not specific for sIBM, as they are found in 15–20% of patients with DM, 10% of patients with systemic lupus erythematosus, and 12% of patients with Sjögren’s syndrome [74,75,76,77]. SIBM patients with NT5C1A antibodies are more commonly female, have greater motor and functional disability, and have more prominent bulbar, facial, and respiratory involvement [78].
Histopathology
Muscle biopsy is a valuable diagnostic tool in patients suspected to have an IIM. The key pathological characteristics of IIM initially recognized by Bohan and Peter criteria were degeneration, regeneration, necrosis, and interstitial mononuclear infiltrates. Inflammatory cell infiltrates are not specific to IIM as they can be seen in muscular dystrophies such as dysferlinopathy, calpainopathy, facioscapulohumeral muscular dystrophy, metabolic myopathies following rhabdomyolysis, granulomatous disorders, myasthenia gravis, vasculitis, and lymphoma, among other disorders [24, 79,80,81,82,83,84]. Muscle biopsies from patients with DM, IMNM, sIBM, and antisynthetase syndrome are known to have many unique pathological features, suggesting different pathophysiological mechanisms exist for each [8, 82,83,84]. Each IIM affects specific regions or tissues within skeletal muscle, including connective tissue, blood vessels, and muscle fibers (Fig. 20.3). Pathology can also predict lung involvement, risk of malignancy, and response to immunomodulatory treatment.
Muscle anatomy. Individual muscle fibers are surrounded by the endomysium, which contains capillaries. Muscle fibers are grouped into fascicles, which are separated by the perimysium. Perimysial connective tissue may contain vasculature. The epimysial connective tissue envelops all fascicles within the muscle
Dermatomyositis
“Perifascicular atrophy” is the classic feature described in DM (Fig. 20.4a). Some claim this finding is very specific for DM; however, there are several inconsistences [14, 41, 85]. For example, some patients will have prominent perifascicular necrosis instead of atrophy, and others may have minimal inflammatory infiltrates and prominent necrosis similar to IMNM [41, 86]. The classic dermatomyositis clinicopathological picture may therefore be more accurately characterized as dermatomyositis with vascular pathology (DM-VP) [87]. DM-VP biopsies demonstrate a perifascicular myopathy with muscle fiber atrophy, reduced cytochrome oxidase staining, and increased MHC class 1 expression (Fig. 20.4a, b) [8]. The vascular pathology is characterized by abnormal, damaged endomysial capillaries with alkaline phosphatase staining, C5b-9 deposition, and lymphocytic foci surrounding larger vessels in vascular perimysium (Fig. 20.4c, d) [8].
Dermatomyositis with vascular pathology histopathology. H&E demonstrating perifascicular atrophy. Note absence of atrophy adjacent to vascular perimysium (arrow) (a). Perifascicular pattern of reduced cytochrome oxidase staining (arrows) (b). Alkaline phosphatase highlights enlarged, abnormal endomysial capillaries (c). Perivascular lymphocytic infiltrates around intermediate-sized vessels (arrow), distant from muscle fiber atrophy (d)
A clinically different subset of patients that are often included under the umbrella categorization of DM have damage to perimysial connective tissue and perifascicular muscle fiber pathology that is often mistaken for DM-VP [8, 88]. Biopsies demonstrate perimysial connective tissue pathology including fragmentation, acid phosphatase-positive histiocytic cells, and alkaline phosphatase staining of the perimysium (Fig. 20.5a–c). Muscle fiber pathology includes necrosis and regeneration, more prominent in regions neighboring the perimysium (Fig. 20.5d) [8, 88]. These disorders have been termed immune myopathies with perimysial pathology (IMPP) [89]. When compared to DM-VP, IMPP is associated with the clinical picture of antisynthetase syndrome with increased risk of ILD, Raynaud phenomenon, mechanic’s hands, inflammatory arthritis, and a higher CK level. IMPP also predicts a sustained response to immunomodulatory therapy and is less frequently associated with a concurrent malignancy [88]. Because of this, IMPP patients require regular screening for ILD. While IMPP patients may have MSAs such as anti-Jo-1 or HMGCR, the large percentage of patients without MSAs emphasizes the important role of myopathology in identifying patients at higher risk of severe comorbid conditions such as ILD.
Immune myopathy with perimysial pathology. Perifascicular pattern of necrosis and regenerating fibers (white arrows), with widened, pale, cellular perimysium (dark arrows) (a). Acid phosphatase-positive histiocytic cells occupying the perimysium (b). Alkaline phosphatase stains the perimysium (c). C5b9 stains the perimysium and cytoplasm of necrotic fibers (d)
Regional ischemic immune myopathy (RIIM) is another distinctive pathologic group observed in dermatomyopathy patients and is likely caused by ischemia in border zones between damaged intermediate-sized perimysial blood vessels [90]. Histopathology reveals an unusual pattern of regional muscle fiber necrosis and regeneration with capillary loss in border zones between intermediate-sized perimysial vessels, vascular pathology with damaged walls of intermediate-sized perimysial veins, and connective tissue with expression of the ischemia marker carbonic anhydrase IX but no mononuclear inflammatory foci [90].
IMNM
The term “necrotizing” may be misleading and imply the whole muscle is necrotic. Immune myopathy “with myofiber necrosis” may be more accurate indicating single myofibers are undergoing necrosis. Regardless, IMNM biopsies typically demonstrate scattered necrotic muscle fibers, although these may be rare or completely absent. Different stages of necrosis/myophagocytosis and regeneration should also be identified [91]. Lymphocytic infiltrates are minimal, if present at all [91]. Sarcolemmal MHC class 1 expression may be seen on non-necrotic and non-regenerating fibers but is often less robust than that seen in other IIMs (Fig. 20.6a–d) [62, 91]. Patchy C5b-9 deposition may be seen. Anti-SRP myopathies more commonly have prominent endomysial fibrosis and enlarged capillaries (Fig. 20.6d–f) [62, 70]. Anti-HMGCR myopathies frequently have perimysial pathology and nuclear abnormalities (Fig. 20.6g–j) [62, 88]. It should be noted that muscle fiber necrosis by itself is not useful for subclassifying IIM [91]. Many different myopathic disorders have prominent muscle fiber necrosis with variable patterns. For example, IMPPs have prominent necrotic fibers near the perimysium [88]. Brachio-cervical inflammatory myopathy has randomly scattered necrotic fibers [92]. Regional ischemic immune myopathy (RIIM) has necrosis of muscle fibers in border zones between vessels [90]. Hereditary and other types of acquired myopathies may also have abundant scattered necrosis.
Anti-signal recognition particle (SRP) myopathy and anti-3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR) myopathy. Varying degrees of pathology seen in anti-SRP myopathy (a-d). Early pathology with scattered necrotic (dark arrow) and regenerating fibers (white arrow) (a). Later in disease with mild pathology (b), intermediate pathology with moderately increased endomysial connective tissue (arrows) (c), and severe pathology with prominently increased connective tissue (d). Ulex staining highlights enlarged capillaries (arrow) (e). C5b-9 stains the sarcoplasm of necrotic fibers (arrow) (f). Anti-HMGCR myopathy more commonly demonstrates immune myopathies with perimysial pathology (IMPP) pathology associated with necrosis. H&E with widened, fragmented, and cellular perimysium with fatty replacement (arrows) (g). Alkaline phosphatase highlights the perimysium (dark arrow) and sarcoplasm of immature fibers (white arrows) (h). Acid phosphatase highlights histiocytic cells within the perimysium (arrow) (i). Congo red staining illustrating nuclear pathology with irregular shapes and clear centers (arrow) (j)
Antisynthetase Syndrome
Muscle biopsies most commonly demonstrate an IMPP pattern with damaged, fragmented perimysium with adjacent perifascicular myofiber necrosis (Fig. 20.5) [8, 88, 93]. The key clinical difference between DM and antisynthetase syndrome patients with IMPP is simply the presence or absence of antisynthetase autoantibodies [88, 93]. Some antisynthetase syndrome patients may have more widespread necrosis and regeneration [93]. On electron microscopy, nuclear actin aggregation may be seen [94].
Sporadic IBM
Muscle biopsies from patients with sIBM demonstrate a coexistence of mononuclear inflammatory cells and protein aggregation. Specifically pathology reveals an IIM with vacuoles, aggregates, and mitochondrial pathology (Fig. 20.7a–i) [8]. This combination of findings has been abbreviated as IM-VAMP [8]. Atrophic fibers are often grouped and may have a neurogenic appearance [95]. The inflammatory infiltrate is located within the endomysium and composed of CD8 T cells that surround and invade non-necrotic fibers [1, 96]. MHC class 1 is often expressed on the sarcolemma. Vacuoles contain granular basophilic debris and are immuno-reactive for markers of autophagy, amyloid, and aggregation-prone proteins such as TAR DNA-binding protein 43 and phosphorylated neurofilament [1, 8, 97,98,99,100,101]. Aggregates are visualized on H&E as eosinophilic inclusions and may also be highlighted using AMPDA or SMI-31. These tubulo-filamentous inclusions may be seen on electron microscopy and gave rise to the name inclusion body myositis [102]. Mitochondrial pathology manifests as scattered cytochrome oxidase-negative fibers. Abnormal myonuclei are also seen [8, 103]. Muscle from individual sIBM patients may show one or all of these features. Individual muscle fibers typically show only one of these features [8].
Sporadic inclusion body myositis (sIBM). H&E demonstrating several key features of sIBM including fiber size variability and endomysial inflammatory cell infiltrates invading non-necrotic fibers (arrows) (a). Focal invasion of non-necrotic fiber by CD8-positive cells (arrow) (b). Aggregates demonstrated by AMPDA (arrows) (c), LC-3 (d), and desmin staining (arrow) (e). Congo red staining illustrating nuclear pathology with clear centers and irregular borders (black arrows) (f). Rimmed vacuoles on Congo red staining (white arrow) (f) and Gomori trichrome (arrow) (g). Cytochrome oxidase-negative fibers (arrow) (h). MHC-1 upregulation on sarcolemma (arrows) (i)
Others
Many other IIM exist and are not included or well categorized by current classification schemes. These disorders are best understood based on their histopathological patterns.
Routine muscle pathology in brachio-cervical inflammatory myopathy (BCIM , also referred to as B-cell inflammatory myopathy) can be similar to that seen in sIBM with focal invasion by inflammatory cells that are commonly endomysial and perivascular [92]. Perimysial connective tissue staining for alkaline phosphatase; foci of B-cell inflammatory infiltrates (CD20 positive), often associated with ectopic lymphoid structures (ELS); or prominent endomysial C5b-9 complement deposition aid in distinguishing BCIM from other forms of IIM. BCIM syndromes frequently overlap with other immune disorders including myasthenia gravis and rheumatoid arthritis and preferentially involve the proximal arms and posterior neck [92].
Histiocytic inflammatory myopathies have focal collections of cells located in the endomysium or perimysium. Acid phosphatase, esterase, and CD68 stains label cells in the centers of these histiocytic inflammatory foci [8]. In contrast, these stains label only 10–30% of cells in focal mononuclear cell collections. Muscle fiber damage appears as replacement of fibers by histiocytic cells and endomysial connective tissue. The best described histiocytic syndromes are granulomatous myopathies, some of which are associated with sarcoidosis [8, 104]. Histiocytic foci and granulomas in muscle can occur without myopathy in systemic sarcoidosis or vasculitic lesions [8, 104]. Collections of histiocytic cells are also found in macrophagic myofasciitis (MMF) and inflammatory myopathy with abundant macrophages (IMAMs) [8, 105,106,107]. MMF and IMAM may be related to immunizations and be clinically silent [108]. Therefore, identifying IMAM histiocytic cell collections should not preclude further search for alternative causes of weakness.
Pathogenesis
The clinical and histopathological distinctions between IIMs suggest different pathogenic processes underlie each, but the precise mechanisms leading to tissue injury are poorly defined.
Dermatomyositis
The precise mechanisms responsible for DM are unknown. Several different DM models have been proposed. One model focuses on a central role for type 1 interferons (IFN) causing capillary, myofiber, and connective tissue injury [109,110,111]. Alternatively, DM myofiber injury may result from an antibody and complement-mediated microangiopathy [8, 112, 113], and the resulting hypoxia triggers IFN production [114]. The pathogenic role of myositis-specific autoantibodies in DM is uncertain [11, 41]. DM in some patients is a paraneoplastic syndrome associated with cancer through unknown mechanisms. Future studies of DM pathogenesis should avoid lumping together distinct clinicopathological groups (DM-VP, IMPP, RIIM), which likely have different pathomechanisms.
A combination of genetic risk factors and exposure to environmental factors may be required to trigger DM. Certain class 2 HLA alleles have been implicated in dermatomyositis pathogenesis [115]. Exposure to ultraviolet light is also a known risk factor for developing dermatomyositis [116]. However, the majority of people with known genetic risk factors and high ultraviolet light exposure never develop DM. Mutations in TIF1 genes in tumors from patients with DM positive for anti-TIF1 autoantibodies have been reported [117]. Once a patient has developed DM, it is unclear what mechanisms maintain muscle damage and weakness.
IMNM
The mechanisms underlying this condition are unknown. Despite the lack of substantial immune cell invasion of muscle, this condition can respond to immunosuppressive therapies, suggesting it is immune-mediated. While statins are known to cause rhabdomyolysis, their association with anti-HMGCR myopathy is not clear, and the condition should not be called a statin myopathy [29, 68]. That stated, there is evidence that HMGCR is expressed by muscle fibers, particularly regenerating fibers, and that antigen expression is increased by statin exposure (via feedback mechanisms similar to those present in hepatocytes) [29]. Accordingly, in patients with anti-HMGCR antibodies, exposure to statins could lead to increased antigen expression and further, immune-mediated, muscle damage. Class 2 HLA-allele DRB1*08:03 is associated with anti-SRP myopathy, and DRB1*11:01 is an immunogenic risk factor for anti-HMGCR myopathy [118]. Some have proposed anti-SRP and anti-HMGCR antibodies are directly pathogenic [67]; however, these antibodies were unable to induce necrosis in vitro [119], indicating further studies are required.
Antisynthetase Syndrome
Little is known about what triggers and maintains autoimmunity in antisynthetase syndrome. A pathogenic role for these antibodies remains unproven. Mouse models of myositis induced by immunization with histidyl-tRNA synthetase are not dependent on the development of antibody responses [120]. Instead, they are thought to be mediated by innate immune mechanisms or by the action of histidyl-tRNA synthetase as a chemokine [120].
Sporadic IBM
Pathologic features of sIBM can be divided into two categories: inflammatory changes and myodegenerative pathologies [8]. These two pathologies have led to pathomechanistic speculation as to whether sIBM is a primary inflammatory, or a primary degenerative, myopathy.
Several lines of evidence suggest that unlike other IIM, sIBM is a primary degenerative myopathy. Rimmed vacuoles are immuno-reactive for autophagic markers such as LC3 suggesting they are autophagic in origin [97,98,99,100]. Inclusions are also immuno-reactive for aggregate-prone proteins including amyloid precursor protein, phosphorylated neurofilament, and TDP-43 [99, 121,122,123,124]. Rimmed vacuoles may also be found in hereditary inclusion body myopathies or protein aggregate myopathies. Dominantly inherited mutations in the ubiquitin adaptor valosin containing protein (VCP) cause a multisystem degenerative syndrome manifesting with IBM, Paget’s disease of bone (PDB), motor neuron disease, and fronto-temporal dementia [125]. Rare variants in SQSTM1 have also been identified in patients with a similar phenotype [126]. Both SQSTM1 and VCP accumulate in sIBM patient muscle, often within or adjacent to rimmed vacuoles [100, 125]. FYCO1, similar to SQSTM1, is an autophagic adaptor protein that binds autophagosomes and facilitates their maturation to acidic lysosomes along microtubules [127]. FYCO1 is a strong marker of rimmed vacuoles, and disease-associated variants impair autophagosome binding in skeletal muscle suggesting they may disrupt autophagic degradation [128]. FYCO1 variants are statistically overrepresented in sIBM patients compared to controls and may serve as risk alleles [128]. These studies support that the degeneration in sIBM patient muscle may be due to a more global disruption in protein degradation pathways, and future treatment strategies aimed at improving protein degradation or protein aggregates may be therapeutic for sIBM. As proof of concept, mice expressing pathogenic VCP mutations were treated with a small molecule, arimoclomol, that enhances the heat shock response. This causes a coordinated upregulation of protein chaperones to facilitate proper folding or degradation of misfolded proteins [129]. Arimoclomol reduced both ubiquitin and TDP-43 pathology and increased forelimb grip strength. These data were supported by a phase II clinical trial in 16 sIBM patients [129].
A number of observations have also strongly implicated autoimmunity as a central pathologic mechanism in sIBM. For example, the invasion of myofibers by cytotoxic CD8+ T cells is a prominent feature in muscle biopsies from sIBM patients [96, 130, 131]. Both oligoclonal and polyclonal expansions of T cells exist within muscle from sIBM patients and support the idea that there is a continuous antigen-driven inflammatory process in sIBM [132]. Many sIBM patients have abnormal clonal expansions of circulating granular lymphocytes that express CD57, a marker of persistent antigenic stimulation that defines a population of T cells with increased cytotoxic potential and resistance to apoptosis [133]. In fact, most sIBM patients meet criteria for T-cell large granular lymphocytic leukemia (T-LGL) [133]. In sIBM, muscle is invaded by the CD8+ CD57+ lymphocytes, which contain cytotoxic granules, analogous to T-LGL where these same cells invade the bone marrow, spleen, and liver. These findings suggest persistent antigenic stimulation of T cells precipitates a neoplastic-like disorder, with cytotoxic T cells invading muscle and circulating in the blood [134].
Dense inflammatory collections consistent with ELS have also been identified in sIBM muscle [135]. Clonally related B cells and plasma cells within these intramuscular lymphoid structures suggest antigen-stimulated maturation of antibody-producing plasma cells occurs locally within sIBM muscle. These findings led to the discovery of autoantibodies targeting cytosolic 5′-nucleotidase 1A (NT5C1A) [136, 137], an enzyme that catalyzes the hydrolysis of adenosine monophosphate to adenosine and inorganic phosphate. NT5C1A is aberrantly localized to perinuclear regions and vacuole rims in sIBM skeletal muscle cells [138]. Whether the abnormal distribution of NT5C1A plays a role in triggering an autoimmune response in sIBM has not been determined.
Another interesting point regarding sIBM pathogenesis is the sIBM-like syndrome that develops in human immunodeficiency virus (HIV)-positive patients. Initially these patients may present, at a younger age of onset, with very high CK levels and proximal weakness that may improve with treatment. However, all patients eventually develop features most consistent with inclusion body myositis, including finger and wrist flexor weakness, rimmed vacuoles, or anti-NT5C1A autoantibodies [139].
Classification
There have been many attempts to establish classification and diagnostic criteria for IIMs. Bohan and Peter proposed their system to establish clear guidelines for diagnosis and classification of PM and DM [140]. These criteria are too inclusive, allowing patients with various muscular dystrophies to be diagnosed with IIM [79], and they are unable to distinguish sIBM, IMNM, antisynthetase syndrome, and DM. Many other classification schemes have been proposed, all attempting to improve the homogeneity of diagnostic categories, so treatment and prognosis may be evaluated accurately. No universally accepted classification system currently exists. IIMs such as BCIM, focal myositis, and others are distinct and well characterized clinically and pathologically, yet are not recognized by current classification schemes [92, 141, 142].
Clinical-serologic associations have helped to more accurately categorize patients and predict risk of malignancy or ILD; however, the utility of classification schemes based on MSAs is limited [10]. Many MSAs lack specificity for a distinct syndrome [14, 52, 53]. MSAs also lack sensitivity as many IIM patients are seronegative [14, 52, 53].
Other classification schemes have placed more emphasis on muscle pathology and facilitated the initial distinction between sIBM and PM [22, 143]. The importance of histopathologic criteria was demonstrated by a retrospective follow-up study of 165 IIM patients that suggested the diagnosis of PM is rare and actually includes a heterogeneous group of disorders [4].
In 2003, two new distinct pathologic entities were proposed at a consensus conference of the European Neuromuscular Centre (ENMC), IMNM and nonspecific myositis, which included patients with nonspecific perimysial/perivascular infiltrates, but without biopsy features diagnostic of DM or PM [144]. In 2011, another classification system was proposed based solely on myopathology that avoided inconsistencies of other clinical classification systems [8]. It utilizes pathologic characteristics, types of muscle fiber damage, and tissues involved to subclassify IIMs. It defined six new pathologic classes: IMPP (seen in antisynthetase syndrome or “DM” cases with ILD), myovasculopathies (seen in dermatomyopathies such as DM-VP and RIIM), immune polymyopathies (such as anti-SRP and HMGCR myopathies), immune myopathies with endomysial pathology (seen in BCIM), histiocytic inflammatory myopathy (seen in granulomatous disorders, MMF, and IMAM), and IM-VAMP (seen in sIBM) [8, 68, 70, 87,88,89,90, 92, 99, 104].
While this system provides consistent and inclusive classification, such specialized myopathological techniques are not widely available. In addition, accurate interpretation of specimens is also problematic [14]. This is evident in the most recent classification scheme, which proposes using only clinical findings and MSAs while excluding histopathology [14]. They note this system may be used to determine what type of IIM a patient has, not if a patient has IIM. Based on phenotypic, biological, and immunologic data, four clusters (DM, IBM, IMNM, antisynthetase syndrome) were identified. They developed a simplified decisional tree with 78.4% correct estimation of their self-defined clusters using three variables: DM rash, antisynthetase syndrome antibodies, and finger flexor scores of 3 or less on the Medical Research Council (MRC) scale [14]. Many problems result from this oversimplification. By ignoring histopathology, many antisynthetase antibody-negative patients are miscategorized. This includes seronegative IMPP patients who are still at increased risk of ILD [88]. This scheme also improperly classifies 35% of sIBM patients as IMNM and 8.7% of IMNM patients as sIBM [14]. This leads to a very problematic situation of incorrectly initiating or withholding immunosuppression in the setting of not having a biopsy to guide further management. While many aspects of these criteria are not ideal, they have been useful in eliminating polymyositis as a diagnostic entity.
Treatment
Treatment for IIMs remains challenging. The absence of standardized treatment guidelines is reflective of their low prevalence, phenotypic heterogeneity, and suboptimal classification systems. Currently, treatment requires a multidisciplinary approach managed by experienced clinicians.
IIMs Other Than sIBM
The shortage of adequate randomized trials has resulted in treatment strategies relying on historical clinical practice, case series, and expert opinion.
Glucocorticoids are first-line treatment, but side effects (weight gain, osteoporosis, hypertension, diabetes) limit their use as a monotherapy. At initial presentation, intravenous methylprednisolone (IVMP) is typically given at 1 gram daily for 3–5 days depending on severity. More conservative approaches will initiate prednisone at starting doses of 0.5–1 mg/kg/day at a maximum of 100 mg/day. Some will maintain daily prednisone for 4–6 weeks and then taper. We utilize pulse dose steroids to minimize side effects [145,146,147], starting at 1 gram/week for 1–2 months, followed by 1 gram every 2 weeks for another 1–2 months, at which time patients are reassessed. Further tapering is facilitated by slowly increasing time between doses or reducing total dose and guided by repeat clinical examinations.
Other immunosuppressive and immunomodulatory drugs commonly used for IIMs include methotrexate, azathioprine, mycophenolate mofetil, cyclosporine, tacrolimus, intravenous immunoglobulin (IVIg), rituximab, and cyclophosphamide (Table 20.2). Certain clinical settings guide the selection of different drugs. Methotrexate is useful as a steroid-sparing agent for muscle and joint disease when relatively quick onset (months) is desired, but may cause lung toxicity and should be avoided in patients with ILD [148]. Azathioprine is useful in patients with normal thiopurine methyltransferase activity for long-term immunosuppression when rapid onset is not necessary [149]. Mycophenolate mofetil, cyclosporine, and tacrolimus may be useful for ILD refractory to corticosteroids [150,151,152]. Cyclosporine and tacrolimus have been used for skin manifestations in DM [150, 151]. Cyclophosphamide may be used in patients with more severe ILD who do not respond to steroids; however, it is associated with more adverse events including infertility [153]. IVIg has shown efficacy in a randomized controlled trial and in a retrospective study for the management of dermatomyositis [154, 155]. IVIg and methotrexate are also effective for anti-HMGCR myopathy [91, 156]. Subcutaneous immunoglobulins (SCIg) may be an alternative to intravenous administration, but reports of SCIg use in IIM are quite limited. Rituximab, a monoclonal antibody targeting CD20 on B lymphocytes, was assessed in refractory DM and polymyositis [157]. While the rituximab arm of this study failed to meet the investigator defined primary endpoint, there were clear benefits to rituximab use in this patient population in that 83% of subjects receiving rituximab met the trial definition of improvement, a criteria generated from measures including muscle strength, muscle enzyme testing, and qualitative disease severity scales [157]. It also appears to be efficacious in patients with antisynthetase syndrome, with or without ILD, and in patients with anti-Mi2, anti-SRP, and anti-HMGCR antibodies [32, 91, 158].
The treatment strategy for juvenile DM is similar to adults [159]. The initial prednisone dose is 2 mg/kg, and methotrexate is the main steroid-sparing agent, although azathioprine, cyclosporine, and tacrolimus have been used. IVIg is the preferred agent for refractory cases. Rituximab is increasingly utilized, and cyclophosphamide is used for severe or life-threatening cases [159].
Evidence is conflicting regarding the use of anti-tumor necrosis factor agents in IIM [160,161,162]. In fact, exposure to anti-TNF drugs has been reported as a precipitant for IIMs in the literature. Abatacept, a fusion protein that inhibits T-cell co-stimulation, showed benefit by reducing disease activity in a pilot study of 20 IIM patients [163]. Case reports have noted efficacy in IIM for tofacitinib [164] and ruxolitinib (Janus kinase inhibitors) [165], tocilizumab (IL-6 antagonist) [166], anakinra (IL-1 antagonist) [167], and alemtuzumab (anti-CD52) [168]; however, confirmatory studies are required.
Sporadic IBM
In contrast to other IIMs, no pharmacological therapy has been shown to be effective for sIBM. Treatment of this form of myositis remains largely supportive. Immunosuppressive drugs, such as corticosteroids, azathioprine, methotrexate, or etanercept, have not shown efficacy in sIBM [134, 169]. Alemtuzumab showed a trend toward a reduction of biomarkers in a pilot study that was not confirmed in a subsequent study [170]. Bimagrumab [171] (a monoclonal antibody that blocks the myostatin pathway) and follistatin [172] (myostatin inhibitor locally delivered using an adeno-associated virus) improved thigh muscle volume and performance on the 6-minute walk test but did not significantly improve muscle strength. Rapamycin, also known as sirolimus, improved performance on the 6-minute walk test but did not improve quadriceps strength [173]. Oxandrolone and simvastatin were also not effective [174, 175]. A randomized controlled trial (NCT02483845) investigating natalizumab, an FDA-approved therapy for multiple sclerosis that prevents T-cell egression out of vasculature, is ongoing. A large randomized controlled trial of arimoclomol is ongoing (NCT02753530).
Management
Physical Exercise
Physical exercise and rehabilitation programs under the supervision of a physical therapist are safe in all types of IIM and are generally recommended to increase strength and reduce disability [176, 177].
Skin Disease
Patients with skin manifestations should use sunscreen and avoid UV rays. Topical steroids and tacrolimus have been used [178]. Hydroxychloroquine, an antimalarial drug, also is also commonly used for cutaneous manifestations.
Calcinosis
Calcinosis commonly fails to respond to immunosuppressive and immunomodulatory therapies. Diltiazem may help [179]. Abatacept and sodium thiosulfate, a calcium chelator, improved calcinosis in a case report [180]. Surgical excision is an option [159].
Dysphagia
Dysphagia may occur in all subtypes of IIM and is particularly common in sIBM. IVIg may improve swallowing in sIBM and other forms of IIM [181,182,183]. Cricopharyngeomyotomy, pharyngoesophageal dilation, and injection of botulinum toxin may be used when dysphagia results from failure of upper esophageal sphincter relaxation [184,185,186].
Treatment of Associated ILD
Patients with even mild ILD should be intensively treated from onset with glucocorticoids and a second-line immunosuppressant agent (tacrolimus or mycophenolate mofetil). When ILD progression is detected, immediate, intensive treatment should be initiated. This includes methylprednisolone pulses along with a second-line immunosuppressant (tacrolimus, cyclophosphamide, or rituximab). Other treatments to consider include two courses of polymyxin in 24 hours, daily plasmapheresis for 3 days followed by every other day for a total of seven sessions, and IVIg after each plasmapheresis session [41, 187, 188]. Lung transplantation may be considered as a last-resort treatment [188].
Conclusions and Future Directions
Currently, four main types of inflammatory myopathies are recognized: dermatomyositis, immune-mediated necrotizing myopathy, sporadic inclusion body myositis, and antisynthetase syndrome. The ongoing controversy regarding classification of IIMs will likely only be resolved through a deeper understanding of pathogenesis. Improved alignment of clinical, laboratory, and histopathologic data will facilitate the development of more efficacious treatments.
References
Griggs RC, Askanas V, DiMauro S, Engel A, Karpati G, Mendell JR, et al. Inclusion body myositis and myopathies. Ann Neurol. 1995;38(5):705–13.
Hoogendijk JE, Amato AA, Neuromuscular BL. 119th ENMC international workshop: trial design in adult idiopathic inflammatory myopathies, with the exception of inclusion body myositis. Neuromuscul Disord. 2004;14(5):337–45.
Amato AA, Griggs RC. Unicorns, dragons, polymyositis, and other mythological beasts. Neurology. 2003;61(3):288–9.
van der Meulen MFG, Bronner IM, Hoogendijk JE, Burger H, van Venrooij WJ, Voskuyl AE, et al. Polymyositis: an overdiagnosed entity. Neurology. 2003;61(3):316–21.
Matsushita T, Hasegawa M, Fujimoto M, Hamaguchi Y, Komura K, Hirano T, et al. Clinical evaluation of anti-aminoacyl tRNA synthetase antibodies in Japanese patients with dermatomyositis. J Rheumatol. 2007;34(5):1012–8.
Hamaguchi Y, Fujimoto M, Matsushita T, Kaji K, Komura K, Hasegawa M, et al. Common and distinct clinical features in adult patients with anti-aminoacyl-tRNA synthetase antibodies: heterogeneity within the syndrome. PLoS One. 2013;8(4):e60442.
Marguerie C, Bunn CC, Beynon HL, Bernstein RM, Hughes JM, So AK, et al. Polymyositis, pulmonary fibrosis and autoantibodies to aminoacyl-tRNA synthetase enzymes. Q J Med. 1990;77(282):1019–38.
Pestronk A. Acquired immune and inflammatory myopathies: pathologic classification. Curr Opin Rheumatol. 2011;23(6):595–604.
Troyanov Y, Targoff IN, Tremblay J-L, Goulet J-R, Raymond Y, Senécal J-L. Novel classification of idiopathic inflammatory myopathies based on overlap syndrome features and autoantibodies: analysis of 100 French Canadian patients. Medicine. 2005;84(4):231–49.
Love LA, Leff RL, Fraser DD, Targoff IN, Dalakas M, Plotz PH, et al. A new approach to the classification of idiopathic inflammatory myopathy: myositis-specific autoantibodies define useful homogeneous patient groups. Medicine. 1991;70(6):360–74.
Mammen AL. Autoimmune myopathies: autoantibodies, phenotypes and pathogenesis. Nat Rev Neurol. 2011;7(6):343–54.
Allenbach Y, Benveniste O. Apport des auto-anticorps au cours des myopathies auto-immunes. Rev Neurol. 2013;169(8–9):656–62.
Benveniste O, Stenzel W, Allenbach Y. Advances in serological diagnostics of inflammatory myopathies. Curr Opin Neurol. 2016;29(5):662–73.
Mariampillai K, Granger B, Amelin D, Guiguet M, Hachulla E, Maurier F, et al. Development of a new classification system for idiopathic inflammatory myopathies based on clinical manifestations and myositis-specific autoantibodies. JAMA Neurol. 2018;75(12):1528–37.
Smoyer-Tomic KE, Amato AA, Fernandes AW. Incidence and prevalence of idiopathic inflammatory myopathies among commercially insured, Medicare supplemental insured, and Medicaid enrolled populations: an administrative claims analysis. BMC Musculoskelet Disord. 2012;13(1):103.
Bernatsky S, Joseph L, Pineau CA, Bélisle P, Boivin JF, Banerjee D, et al. Estimating the prevalence of polymyositis and dermatomyositis from administrative data: age, sex and regional differences. Ann Rheum Dis. 2009;68(7):1192–6.
Furst DE, Amato AA, Iorga ŞR, Gajria K, Fernandes AW. Epidemiology of adult idiopathic inflammatory myopathies in a U.S. managed care plan. Muscle Nerve. 2012;45(5):676–83.
Needham M, Corbett A, Day T, Christiansen F, Fabian V, Mastaglia FL. Prevalence of sporadic inclusion body myositis and factors contributing to delayed diagnosis. J Clin Neurosci. 2008;15(12):1350–3.
Prieto S, Grau JM. The geoepidemiology of autoimmune muscle disease. Autoimmun Rev. 2010;9(5):A330–4.
Mastaglia FL, Phillips BA. Idiopathic inflammatory myopathies: epidemiology, classification, and diagnostic criteria. Rheum Dis Clin N Am. 2002;28(4):723–41.
Furst DE, Amato AA, Iorga ŞR, Bancroft T, Fernandes AW. Medical costs and health-care resource use in patients with inflammatory myopathies in an insured population. Muscle Nerve. 2012;46(4):496–505.
Dalakas MC. Polymyositis, dermatomyositis, and inclusion-body myositis. N Engl J Med. 1991;325(21):1487–98.
Lundberg IE, Tjärnlund A, Bottai M, Werth VP, Pilkington C, de Visser M, et al. 2017 European League Against Rheumatism/American College of Rheumatology Classification Criteria for adult and juvenile idiopathic inflammatory myopathies and their major subgroups. Arthritis Rheumatol. 2017;69(12):2271–82.
Amato AA, Barohn RJ. Evaluation and treatment of inflammatory myopathies. J Neurol Neurosurg Psychiatry. 2009;80(10):1060–8.
Khan S, Christopher-Stine L. Polymyositis, dermatomyositis, and autoimmune necrotizing myopathy: clinical features. Rheum Dis Clin N Am. 2011;37(2):143–58.
Orlow SJ, Watsky KL. Skin and bones. II. J Am Acad Dermatol. 1991;25(3):447–62.
Shirani Z, Kucenic MJ, Carroll CL, Fleischer AB, Feldman SR, Yosipovitch G, et al. Pruritus in adult dermatomyositis. Clin Exp Dermatol. 2004;29(3):273–6.
Schneider I, Stoltenburg G, Deschauer M, Winterholler M, Hanisch F. Limb girdle muscular dystrophy type 2L presenting as necrotizing myopathy. Acta Myol. 2014;33(1):19–21.
Mohassel P, Mammen AL. Anti-HMGCR myopathy. J Neuromuscul Dis. 2018;5(1):11–20.
Mohassel P, Landon-Cardinal O, Foley AR, Donkervoort S, Pak KS, Wahl C, et al. Anti-HMGCR myopathy may resemble limb-girdle muscular dystrophy. Neurol Neuroimmunol Neuroinflamm. 2019;6(1):e523.
Rider LG, Miller FW. Deciphering the clinical presentations, pathogenesis, and treatment of the idiopathic inflammatory myopathies. JAMA. 2011;305(2):183–90.
Fernandez IP, Parks C, Werner JL, Albayda J, Paik JJ, Danoff SK, et al. Longitudinal course of disease in a large cohort of myositis patients with autoantibodies recognizing the signal recognition particle. Arthritis Care Res (Hoboken). 2017;69(2):263–70.
Suzuki S, Nishikawa A, Kuwana M, Nishimura H, Watanabe Y, Nakahara J, et al. Inflammatory myopathy with anti-signal recognition particle antibodies: case series of 100 patients. Orphanet J Rare Dis. 2015;10(1):61.
Tiniakou E, Pinal-Fernandez I, Lloyd TE, Albayda J, Paik J, Werner JL, et al. More severe disease and slower recovery in younger patients with anti-3-hydroxy-3-methylglutaryl-coenzyme a reductase-associated autoimmune myopathy. Rheumatology (Oxford). 2017;56(5):787–94.
Clements PJ, Furst DE, Campion DS, Bohan A, Harris R, Levy J, et al. Muscle disease in progressive systemic sclerosis: diagnostic and therapeutic considerations. Arthritis Rheum. 1978;21(1):62–71.
Pinal-Fernandez I, Casal-Dominguez M, Huapaya JA, Albayda J, Paik JJ, Johnson C, et al. A longitudinal cohort study of the anti-synthetase syndrome: increased severity of interstitial lung disease in black patients and patients with anti-PL7 and anti-PL12 autoantibodies. Rheumatology (Oxford). 2017;56(6):999–1007.
Dalakas MC. Inflammatory muscle diseases. N Engl J Med. 2015;373(4):393–4.
Needham M, Mastaglia FL. Inclusion body myositis: current pathogenetic concepts and diagnostic and therapeutic approaches. Lancet Neurol. 2007;6(7):620–31.
Cox FM, Delgado V, Verschuuren JJ, Ballieux BE, Bax JJ, Wintzen AR, et al. The heart in sporadic inclusion body myositis: a study in 51 patients. J Neurol. 2009;257(3):447–51.
Voermans NC, Vaneker M, Hengstman GJD, Laak ter HJ, Zimmerman C, Schelhaas HJ, et al. Primary respiratory failure in inclusion body myositis. Neurology. 2004;63(11):2191–2.
Selva-O’Callaghan A, Pinal-Fernandez I, Trallero-Araguás E, Milisenda JC, Grau-Junyent JM, Mammen AL. Classification and management of adult inflammatory myopathies. Lancet Neurol. 2018;17(9):816–28.
Nozaki K, Pestronk A. High aldolase with normal creatine kinase in serum predicts a myopathy with perimysial pathology. J Neurol Neurosurg Psychiatry. 2009;80(8):904–8.
Needham M, James I, Corbett A, Day T, Christiansen F, Phillips B, et al. Sporadic inclusion body myositis: phenotypic variability and influence of HLA-DR3 in a cohort of 57 Australian cases. J Neurol Neurosurg Psychiatry. 2008;79(9):1056–60.
Amato AA, Gronseth GS, Jackson CE, Wolfe GI, Katz JS, Bryan WW, et al. Inclusion body myositis: clinical and pathological boundaries. Ann Neurol. 1996;40(4):581–6.
Koffman BM, Rugiero M, Dalakas MC. Immune-mediated conditions and antibodies associated with sporadic inclusion body myositis. Muscle Nerve. 1998;21(1):115–7.
Lotz BP, Engel AG, Nishino H, Stevens JC, Litchy WJ. Inclusion body myositis observations in 40 patients. Brain. 1989;112(3):727–47.
Rosales XQ, Chu M-L, Shilling C, Wall C, Pastores GM, Mendell JR. Fidelity of gamma-glutamyl transferase (GGT) in differentiating skeletal muscle from liver damage. J Child Neurol. 2008;23(7):748–51.
Tasca G, Monforte M, De Fino C, Kley RA, Ricci E, Mirabella M. Magnetic resonance imaging pattern recognition in sporadic inclusion-body myositis. Muscle Nerve. 2015;52(6):956–62.
Pinal-Fernandez I, Casal-Dominguez M, Carrino JA, Lahouti AH, Basharat P, Albayda J, et al. Thigh muscle MRI in immune-mediated necrotising myopathy: extensive oedema, early muscle damage and role of anti-SRP autoantibodies as a marker of severity. Ann Rheum Dis. 2017;76(4):681–7.
Van De Vlekkert J, Maas M, Hoogendijk JE, de Visser M, Van Schaik IN. Combining MRI and muscle biopsy improves diagnostic accuracy in subacute-onset idiopathic inflammatory myopathy. Muscle Nerve. 2015;51(2):253–8.
Theodorou DJ, Theodorou SJ, Kakitsubata Y. Skeletal muscle disease: patterns of MRI appearances. Br J Radiol. 2014;85(1020):e1298–308.
Betteridge Z, McHugh N. Myositis-specific autoantibodies: an important tool to support diagnosis of myositis. J Intern Med. 2016;280(1):8–23.
Targoff IN. Myositis specific autoantibodies. Curr Rheumatol Rep. 2006;8(3):196–203.
Kalluri M, Sahn SA, Oddis CV, Gharib SL, Christopher-Stine L, Danoff SK, et al. Clinical profile of anti-PL-12 autoantibody: cohort study and review of the literature. Chest. 2009;135(6):1550–6.
Hervier B, Devilliers H, Stanciu R, Meyer A, Uzunhan Y, Masseau A, et al. Hierarchical cluster and survival analyses of antisynthetase syndrome: phenotype and outcome are correlated with anti-tRNA synthetase antibody specificity. Autoimmun Rev. 2012;12(2):210–7.
Ghirardello A, Zampieri S, Iaccarino L, Tarricone E, Bendo R, Gambari PF, et al. Anti-Mi-2 antibodies. Autoimmunity. 2005;38(1):79–83.
Fiorentino DF, Chung LS, Christopher-Stine L, Zaba L, Li S, Mammen AL, et al. Most patients with cancer-associated dermatomyositis have antibodies to nuclear matrix protein NXP-2 or transcription intermediary factor 1γ. Arthritis Rheum. 2013;65(11):2954–62.
Albayda J, Pinal-Fernandez I, Huang W, Parks C, Paik J, Casciola-Rosen L, et al. Antinuclear matrix protein 2 autoantibodies and edema, muscle disease, and malignancy risk in dermatomyositis patients. Arthritis Care Res (Hoboken). 2017;69(11):1771–6.
Labrador-Horrillo M, Martinez MA, Selva-O’Callaghan A, Trallero-Araguás E, Balada E, Vilardell-Tarrés M, et al. Anti-MDA5 antibodies in a large Mediterranean population of adults with Dermatomyositis. J Immunol Res. 2014;2014(5):1–8.
Narang NS, Casciola-Rosen L, Li S, Chung L, Fiorentino DF. Cutaneous ulceration in dermatomyositis: association with anti-melanoma differentiation-associated gene 5 antibodies and interstitial lung disease. Arthritis Care Res (Hoboken). 2015;67(5):667–72.
Ge Y, Lu X, Shu X, Peng Q, Wang G. Clinical characteristics of anti-SAE antibodies in Chinese patients with dermatomyositis in comparison with different patient cohorts. Sci Rep. 2016;7(1):188.
Watanabe Y, Uruha A, Suzuki S, Nakahara J, Hamanaka K, Takayama K, et al. Clinical features and prognosis in anti-SRP and anti-HMGCR necrotising myopathy. J Neurol Neurosurg Psychiatry. 2016;87(10):1038–44.
Mammen AL, Chung T, Christopher-Stine L, Rosen P, Rosen A, Doering KR, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum. 2011;63(3):713–21.
Trallero-Araguás E, Rodrigo-Pendás JÁ, Selva-O’Callaghan A, Martínez-Gómez X, Bosch X, Labrador-Horrillo M, et al. Usefulness of anti-p155 autoantibody for diagnosing cancer-associated dermatomyositis: a systematic review and meta-analysis. Arthritis Rheum. 2012;64(2):523–32.
Selva-O’Callaghan A, Grau JM, Gámez-Cenzano C, Vidaller-Palacín A, Martínez-Gómez X, Trallero-Araguás E, et al. Conventional cancer screening versus PET/CT in dermatomyositis/polymyositis. Am J Med. 2010;123(6):558–62.
Sato S, Hoshino K, Satoh T, Fujita T, Kawakami Y, Fujita T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum. 2009;60(7):2193–200.
Allenbach Y, Arouche-Delaperche L, Preusse C, Radbruch H, Butler-Browne G, Champtiaux N, et al. Necrosis in anti-SRP+ and anti-HMGCR+myopathies: role of autoantibodies and complement. Neurology. 2018;90(6):e507–17.
Alshehri A, Choksi R, Bucelli R, Pestronk A. Myopathy with anti-HMGCR antibodies: perimysium and myofiber pathology. Neurol Neuroimmunol Neuroinflamm. 2015;2(4):e124.
Allenbach Y, Keraen J, Bouvier A-M, Jooste V, Champtiaux N, Hervier B, et al. High risk of cancer in autoimmune necrotizing myopathies: usefulness of myositis specific antibody. Brain. 2016;139(Pt 8):2131–5.
Miller T, Al-Lozi MT, Lopate G, Pestronk A. Myopathy with antibodies to the signal recognition particle: clinical and pathological features. J Neurol Neurosurg Psychiatry. 2002;73(4):420–8.
Targoff IN, Johnson AE, Miller FW. Antibody to signal recognition particle in polymyositis. Arthritis Rheum. 1990;33(9):1361–70.
Lim J, Rietveld A, De Bleecker JL, Badrising UA, Saris CGJ, van der Kooi AJ, et al. Seronegative patients form a distinctive subgroup of immune-mediated necrotizing myopathy. Neurol Neuroimmunol Neuroinflamm. 2019;6(1):e513.
Trallero-Araguás E, Grau-Junyent JM, Labirua-Iturburu A, García-Hernández FJ, Monteagudo-Jiménez M, Fraile-Rodriguez G, et al. Clinical manifestations and long-term outcome of anti-Jo1 antisynthetase patients in a large cohort of Spanish patients from the GEAS-IIM group. Semin Arthritis Rheum. 2016;46(2):225–31.
Lloyd TE, Christopher-Stine L, Pinal-Fernandez I, Tiniakou E, Petri M, Baer A, et al. Cytosolic 5′-Nucleotidase 1A as a target of circulating autoantibodies in autoimmune diseases. Arthritis Care Res (Hoboken). 2015;68(1):66–71.
Herbert MK, Stammen-Vogelzangs J, Verbeek MM, Rietveld A, Lundberg IE, Chinoy H, et al. Disease specificity of autoantibodies to cytosolic 5′-nucleotidase 1A in sporadic inclusion body myositis versus known autoimmune diseases. Ann Rheum Dis. 2016;75(4):696–701.
Muro Y, Nakanishi H, Katsuno M, Kono M, Akiyama M. Prevalence of anti-NT5C1A antibodies in Japanese patients with autoimmune rheumatic diseases in comparison with other patient cohorts. Clin Chim Acta. 2017;472:1–4.
Lilleker JB, Rietveld A, Pye SR, Mariampillai K, Benveniste O, Peeters MTJ, et al. Cytosolic 5′-nucleotidase 1A autoantibody profile and clinical characteristics in inclusion body myositis. Ann Rheum Dis. 2017;76(5):862–8.
Goyal NA, Cash TM, Alam U, Enam S, Tierney P, Araujo N, et al. Seropositivity for NT5c1A antibody in sporadic inclusion body myositis predicts more severe motor, bulbar and respiratory involvement. J Neurol Neurosurg Psychiatry. 2016;87(4):373–8.
Gallardo E, García RR, De Luna N, Pou A, Brown RH, Illa I. Inflammation in dysferlin myopathy: Immunohistochemical characterization of 13 patients. Neurology. 2001;57(11):2136–8.
Krahn M, De Munain AL, Streichenberger N, Bernard R, Pécheux C, Testard H, et al. CAPN3 mutations in patients with idiopathic eosinophilic myositis. Ann Neurol. 2006;59(6):905–11.
Arahata K, Ishihara T, Fukunaga H, Orimo S, Lee JH, Goto K, et al. Inflammatory response in facioscapulohumeral muscular dystrophy (FSHD): Immunocytochemical and genetic analyses. Muscle Nerve. 1995;18(S13):S56–66.
Maeshima S, Koike H, Noda S, Noda T, Nakanishi H, Iijima M, et al. Clinicopathological features of sarcoidosis manifesting as generalized chronic myopathy. J Neurol. 2015;262(4):1035–45.
Fenichel GM, Shy GM. Muscle biopsy experience in myasthenia gravis. Arch Neurol. 1963;9(3):237–43.
Tahsili-Fahadan P, Rashidi A, Cimino PJ, Bucelli RC, Keyrouz SG. Neurologic manifestations of intravascular large B-cell lymphoma. Neurol Clin Pract. 2016;6(1):55–60.
Suárez-Calvet X, Gallardo E, Pinal-Fernandez I, De Luna N, Lleixà C, Díaz-Manera J, et al. RIG-I expression in perifascicular myofibers is a reliable biomarker of dermatomyositis. Arthritis Res Ther. 2017;19(1):174.
Pinal-Fernandez I, Casciola-Rosen LA, Christopher-Stine L, Corse AM, Mammen AL. The prevalence of individual histopathologic features varies according to autoantibody status in muscle biopsies from patients with dermatomyositis. J Rheumatol. 2015;42(8):1448–54.
Pestronk A, Schmidt RE, Choksi R. Vascular pathology in dermatomyositis and anatomic relations to myopathology. Muscle Nerve. 2010;42(1):53–61.
Bucelli RC, Pestronk A. Immune myopathies with perimysial pathology: clinical and laboratory features. Neurol Neuroimmunol Neuroinflamm. 2018;5(2):e434.
Mozaffar T, Pestronk A. Myopathy with anti-Jo-1 antibodies: pathology in perimysium and neighbouring muscle fibres. J Neurol Neurosurg Psychiatry. 2000;68(4):472–8.
Cai C, Alshehri A, Choksi R, Pestronk A. Regional ischemic immune myopathy: a paraneoplastic dermatomyopathy. J Neuropathol Exp Neurol. 2014;73(12):1126–33.
Allenbach Y, Mammen AL, Benveniste O, Stenzel W. Immune-Mediated Necrotizing Myopathies Working Group. 224th ENMC International Workshop: Clinico-sero-pathological classification of immune-mediated necrotizing myopathies Zandvoort, The Netherlands, 14–16 October 2016. Neuromuscul Disord. 2018;28(1):87–99.
Pestronk A, Kos K, Lopate G, Al-Lozi MT. Brachio-cervical inflammatory myopathies: clinical, immune, and myopathologic features. Arthritis Rheum. 2006;54(5):1687–96.
Noguchi E, Uruha A, Suzuki S, Hamanaka K, Ohnuki Y, Tsugawa J, et al. Skeletal muscle involvement in antisynthetase syndrome. JAMA Neurol. 2017;74(8):992–9.
Stenzel W, Preuße C, Allenbach Y, Pehl D, Junckerstorff R, Heppner FL, et al. Nuclear actin aggregation is a hallmark of anti-synthetase syndrome-induced dysimmune myopathy. Neurology. 2015;84(13):1346–54.
Mitsui T, Kunishige M, Kuroda Y, Kaji R. Heterogeneous muscular involvement in inclusion body myositis. Lab Med. 2010;41(7):393–6.
Arahata K, Engel AG. Monoclonal antibody analysis of mononuclear cells in myopathies. I: quantitation of subsets according to diagnosis and sites of accumulation and demonstration and counts of muscle fibers invaded by T cells. Ann Neurol. 1984;16(2):193–208.
Hiniker A, Daniels BH, Lee HS, Margeta M. Comparative utility of LC3, p62 and TDP-43 immunohistochemistry in differentiation of inclusion body myositis from polymyositis and related inflammatory myopathies. Acta Neuropathol Commun. 2013;1(1):29.
Lünemann JD, Schmidt J, Schmid D, Barthel K, Wrede A, Dalakas MC, et al. β-Amyloid is a substrate of autophagy in sporadic inclusion body myositis. Ann Neurol. 2007;61(5):476–83.
Temiz P, Weihl CC, Pestronk A. Inflammatory myopathies with mitochondrial pathology and protein aggregates. J Neurol Sci. 2009;278(1–2):25–9.
Nogalska A, Terracciano C, D’Agostino C, Engel WK, Askanas V. p62/SQSTM1 is overexpressed and prominently accumulated in inclusions of sporadic inclusion-body myositis muscle fibers, and can help differentiating it from polymyositis and dermatomyositis. Acta Neuropathol. 2009;118(3):407–13.
Mendell JR, Sahenk Z, Gales T, Paul L. Amyloid filaments in inclusion body myositis: novel findings provide insight into nature of filaments. arch neurol. 1991;48(12):1229–34.
Dalakas M. Muscle biopsy findings in inflammatory myopathies. Rheum Dis Clin N Am. 2002;28(4):779–98, vi.
Greenberg S, Pinkus J, Amato A. Nuclear membrane proteins are present within rimmed vacuoles in inclusion-body myositis. Muscle Nerve. 2006;34(4):406–16.
Mozaffar T, Lopate G, Pestronk A. Clinical correlates of granulomas in muscle. J Neurol. 1998;245(8):519–24.
Israeli E, Agmon-levin N, Blank M, Shoenfeld Y. Macrophagic myofasciitis a vaccine (alum) autoimmune-related disease. Clin Rev Allergy Immunol. 2011;41(2):163–8.
Hara S, Henmi T, Kawakami A, Fujikawa K, Mukae H, Ishimatsu Y, et al. Clinical, serologic and magnetic resonance imaging of 3 cases of inflammatory myopathy with abundant macrophages in the japanese population. Rheumatol Int. 2013;33(4):1059–64.
Bassez G, Authier F-J, Lechapt-Zalcman E, Delfau-Larue M, Plonquet A, Coquet M, et al. Inflammatory myopathy with abundant macrophages (imam): a condition sharing similarities with cytophagic histiocytic panniculitis and distinct from macrophagic myofasciitis. J Neuropathol Exp Neurol. 2003;62(5):464–74.
Iach B, Cupler E. Macrophagic myofasciitis in children is a localized reaction to vaccination. J Child Neurol. 2007;23(6):614–9.
Greenberg SA. Dermatomyositis and type 1 interferons. Curr Rheumatol Rep. 2010;12(3):198–203.
Greenberg SA. Proposed immunologic models of the inflammatory myopathies and potential therapeutic implications. Neurology. 2007;69(21):2008–19.
Greenberg SA. Type 1 interferons and myositis. Arthritis Res Ther. 2010;12 Suppl 1(Suppl 1):S4.
Dalakas MC. Inflammatory disorders of muscle: progress in polymyositis, dermatomyositis and inclusion body myositis. Curr Opin Neurol. 2004;17(5):561–7.
Dalakas MC, Hohlfeld R. Polymyositis and dermatomyositis. Lancet. 2003;362(9388):971–82.
De Luna N, Suárez-Calvet X, Lleixà C, Díaz-Manera J, Olivé M, Illa I, et al. Hypoxia triggers IFN-I production in muscle: implications in dermatomyositis. Sci Rep. 2017;7(1):8595.
Miller FW, Chen W, O’Hanlon TP, Cooper RG, Vencovsky J, Rider LG, et al. Genome-wide association study identifies HLA 8.1 ancestral haplotype alleles as major genetic risk factors for myositis phenotypes. Genes Immun. 2015;16(7):470–80.
Mamyrova G, Rider LG, Ehrlich A, Jones O, Pachman LM, Nickeson R, et al. Environmental factors associated with disease flare in juvenile and adult dermatomyositis. Rheumatology (Oxford). 2017;56(8):1342–7.
Pinal-Fernandez I, Ferrer-Fabregas B, Trallero-Araguás E, Balada E, Martinez MA, Milisenda JC, et al. Tumour TIF1 mutations and loss of heterozygosity related to cancer-associated myositis. Rheumatology (Oxford). 2018;57(2):388–96.
Ohnuki Y, Suzuki S, Shiina T, Uruha A, Watanabe Y, Suzuki S, et al. HLA-DRB1 alleles in immune-mediated necrotizing myopathy. Neurology. 2016;87(18):1954–5.
Arouche-Delaperche L, Allenbach Y, Amelin D, Preusse C, Mouly V, Mauhin W, et al. Pathogenic role of anti-signal recognition protein and anti-3-Hydroxy-3-methylglutaryl-CoA reductase antibodies in necrotizing myopathies: Myofiber atrophy and impairment of muscle regeneration in necrotizing autoimmune myopathies. Ann Neurol. 2017;81(4):538–48.
Soejima M, Kang EH, Gu X, Katsumata Y, Clemens PR, Ascherman DP. Role of innate immunity in a murine model of histidyl–transfer RNA synthetase (Jo-1)–mediated myositis. Arthritis Rheum. 2011;63(2):479–87.
Askanas V, Engel WK, Alvarez RB. Light and electron microscopic localization of beta-amyloid protein in muscle biopsies of patients with inclusion-body myositis. Am J Pathol. 1992;141(1):31–6.
Mirabella M, Alvarez RB, Bilak M, Engel WK, Askanas V. Difference in expression of phosphorylated tau epitopes between sporadic inclusion-body myositis and hereditary inclusion-body myopathies. J Neuropathol Exp Neurol. 1996;55(7):774–86.
Salajegheh M, Pinkus JL, Taylor JP, Amato AA, Nazareno R, Baloh RH, et al. Sarcoplasmic redistribution of nuclear TDP-43 in inclusion body myositis. Muscle Nerve. 2009;40(1):19–31.
Weihl CC, Temiz P, Miller SE, Watts G, Smith C, Forman M, et al. TDP-43 accumulation in inclusion body myopathy muscle suggests a common pathogenic mechanism with frontotemporal dementia. J Neurol Neurosurg Psychiatry. 2008;79(10):1186–9.
Watts GDJ, Wymer J, Kovach MJ, Mehta SG, Mumm S, Darvish D, et al. Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nat Genet. 2004;36(4):377–81.
Bucelli RC, Arhzaouy K, Pestronk A, Pittman SK, Rojas L, Sue CM, et al. SQSTM1 splice site mutation in distal myopathy with rimmed vacuoles. Neurology. 2015;85(8):665–74.
Pankiv S, Johansen T. FYCO1: linking autophagosomes to microtubule plus end-directing molecular motors. Autophagy. 2010;6(4):550–2.
Güttsches AK, Brady S, Krause K, Maerkens A, Uszkoreit J, Eisenacher M, et al. Proteomics of rimmed vacuoles define new risk allele in inclusion body myositis. Ann Neurol. 2017;81(2):227–39.
Ahmed M, Machado PM, Miller A, Spicer C, Herbelin L, He J, et al. Targeting protein homeostasis in sporadic inclusion body myositis. Sci Transl Med. 2016;8(331):331ra41–1.
Arahata K, Engel AG. Monoclonal antibody analysis of mononuclear cells in myopathies. IV: cell-mediated cytotoxicity and muscle fiber necrosis. Ann Neurol. 1988;23(2):168–73.
Arahata K, Engel AG. Monoclonal antibody analysis of mononuclear cells in myopathies. V: identification and quantitation of T8+ cytotoxic and T8+ suppressor cells. Ann Neurol. 1988;23(5):493–9.
Fyhr I-M, Moslemi A-R, Lindberg C, Oldfors A. T cell receptor β-chain repertoire in inclusion body myositis. J Neuroimmunol. 1998;91(1–2):129–34.
Greenberg SA, Pinkus JL, Amato AA, Kristensen T, Dorfman DM. Association of inclusion body myositis with T cell large granular lymphocytic leukaemia. Brain. 2016;139(5):1348–60.
Weihl CC, Mammen AL. Sporadic inclusion body myositis – a myodegenerative disease or an inflammatory myopathy. Neuropathol Appl Neurobiol. 2017;43(1):82–91.
Salajegheh M, Pinkus JL, Amato AA, Morehouse C, Jallal B, Yao Y, et al. Permissive environment for B-cell maturation in myositis muscle in the absence of B-cell follicles. Muscle Nerve. 2010;42(4):576–83.
Salajegheh M, Lam T, Greenberg SA. Autoantibodies against a 43 KDa muscle protein in inclusion body myositis. PLoS One. 2011;6(5):e20266.
Pluk H, van Hoeve BJA, van Dooren SHJ, Stammen-Vogelzangs J, van der Heijden A, Schelhaas HJ, et al. Autoantibodies to cytosolic 5′-nucleotidase 1A in inclusion body myositis. Ann Neurol. 2013;73(3):397–407.
Larman HB, Salajegheh M, Nazareno R, Lam T, Sauld J, Steen H, et al. Cytosolic 5′-nucleotidase 1A autoimmunity in sporadic inclusion body myositis. Ann Neurol. 2013;73(3):408–18.
Lloyd TE, Pinal-Fernandez I, Michelle EH, Christopher-Stine L, Pak K, Sacktor N, et al. Overlapping features of polymyositis and inclusion body myositis in HIV-infected patients. Neurology. 2017;88(15):1454–60.
Bohan A, Peter JB. Polymyositis and dermatomyositis. N Engl J Med. 1975;292(7):344–7.
Smith AG, Urbanits S, Blaivas M, Grisold W, Russell JW. Clinical and pathologic features of focal myositis. Muscle Nerve. 2000;23(10):1569–75.
Rojana-Udomsart A, Fabian V, Hollingsworth PN, Walters SE, Zilko PJ, Mastaglia FL. Paraspinal and scapular myopathy associated with scleroderma. J Clin Neuromuscul Dis. 2010;11(4):213–22.
Carpenter S, Karpati G, Heller I, Eisen A. Inclusion body myositis: a distinct variety of idiopathic inflammatory myopathy. Neurology. 1978;28(1):8–17.
Hoogendijk JE, Amato AA, Lecky BR, Choy EH, Lundberg IE, Rose MR, et al. 119th ENMC international workshop: trial design in adult idiopathic inflammatory myopathies, with the exception of inclusion body myositis, 10-12 October 2003, Naarden, The Netherlands. Neuromuscul Disord. 2004;14(5):337–45.
Connolly AM, Schierbecker J, Renna R, Florence J. High dose weekly oral prednisone improves strength in boys with Duchenne muscular dystrophy. Neuromuscul Disord. 2002;12(10):917–25.
Escolar DM, Hache LP, Clemens PR, Cnaan A, McDonald CM, Viswanathan V, et al. Randomized, blinded trial of weekend vs daily prednisone in Duchenne muscular dystrophy. Neurology. 2011;77(5):444–52.
Matsubara S, Sawa Y, Takamori M, Yokoyama H, Kida H. Pulsed intravenous methylprednisolone combined with oral steroids as the initial treatment of inflammatory myopathies. J Neurol Neurosurg Psychiatry. 1994;57(8):1008.
Vencovsky J, Jarosová K, Machácek S, Studýnková J, Kafková J, Bartůnková J, et al. Cyclosporine A versus methotrexate in the treatment of polymyositis and dermatomyositis. Scand J Rheumatol. 2000;29(2):95–102.
Dalakas MC. Inflammatory myopathies: management of steroid resistance. Curr Opin Neurol. 2011;24(5):457–62.
Go DJ, Park JK, Kang EH, Kwon HM, Lee YJ, Song Y-W, et al. Survival benefit associated with early cyclosporine treatment for dermatomyositis-associated interstitial lung disease. Rheumatol Int. 2016;36(1):125–31.
Labirua-Iturburu A, Selva-O’Callaghan A, Martínez-Gómez X, Trallero-Araguás E, Labrador-Horrillo M, Vilardell-Tarrés M. Calcineurin inhibitors in a cohort of patients with antisynthetase-associated interstitial lung disease. Clin Exp Rheumatol. 2013;31(3):436–9.
Hanaoka H, Iida H, Kiyokawa T, Takakuwa Y, Kawahata K. Mycophenolate mofetil treatment with or without a calcineurin inhibitor in resistant inflammatory myopathy. Clin Rheumatol. 2018;372:1734–6.
Barnes H, Holland AE, Westall GP, Goh NS, Glaspole IN. Cyclophosphamide for connective tissue disease-associated interstitial lung disease. Cochrane Database Syst Rev. 2018;1(12):CD010908.
Kampylafka EI, Kosmidis ML, Panagiotakos DB, Dalakas M, Moutsopoulos HM, Tzioufas AG. The effect of intravenous immunoglobulin (IVIG) treatment on patients with dermatomyositis: a 4-year follow-up study. Clin Exp Rheumatol. 2012;30(3):397–401.
Dalakas MC, Illa I, Dambrosia JM, Soueidan SA, Stein DP, Otero C, et al. A controlled trial of high-dose intravenous immune globulin infusions as treatment for dermatomyositis. N Engl J Med. 1993;329(27):1993–2000.
Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med. 2015;373(17):1680–2.
Oddis CV, Reed AM, Aggarwal R, Rider LG, Ascherman DP, Levesque MC, et al. Rituximab in the treatment of refractory adult and juvenile dermatomyositis and adult polymyositis: a randomized, placebo-phase trial. Arthritis Rheum. 2013;65(2):314–24.
Aggarwal R, Bandos A, Reed AM, Ascherman DP, Barohn RJ, Feldman BM, et al. Predictors of clinical improvement in rituximab-treated refractory adult and juvenile dermatomyositis and adult polymyositis. Arthritis Rheumatol. 2014;66(3):740–9.
Malik A, Hayat G, Kalia JS, Guzman MA. Idiopathic inflammatory myopathies: clinical approach and management. Front Neurol. 2016;7(21):1487.
Chen D, Wang X-B, Zhou Y, Zhu X-C. Efficacy of infliximab in the treatment for dermatomyositis with acute interstitial pneumonia: a study of fourteen cases and literature review. Rheumatol Int. 2013;33(10):2455–8.
Schiffenbauer A, Garg M, Castro C, Pokrovnichka A, Joe G, Shrader J, et al. A randomized, double-blind, placebo-controlled trial of infliximab in refractory polymyositis and dermatomyositis. Semin Arthritis Rheum. 2018;47(6):858–64.
Muscle Study Group. A randomized, pilot trial of etanercept in dermatomyositis. Ann Neurol. 2011;70(3):427–36.
Kerola AM, Kauppi MJ. Abatacept as a successful therapy for myositis—a case-based review. Clin Rheumatol. 2014;34(3):609–12.
Kurtzman DJB, Wright NA, Lin J, Femia AN, Merola JF, Patel M, et al. Tofacitinib citrate for refractory cutaneous dermatomyositis: an alternative treatment. JAMA Dermatol. 2016;152(8):944–5.
Hornung T, Janzen V, Heidgen F-J, Wolf D, Bieber T, Wenzel J. Remission of recalcitrant dermatomyositis treated with ruxolitinib. N Engl J Med. 2014;371(26):2537–8.
Narazaki M, Hagihara K, Shima Y, Ogata A, Kishimoto T, Tanaka T. Therapeutic effect of tocilizumab on two patients with polymyositis. Rheumatology (Oxford). 2011;50(7):1344–6.
Zong M, Dorph C, Dastmalchi M, Alexanderson H, Pieper J, Amoudruz P, et al. Anakinra treatment in patients with refractory inflammatory myopathies and possible predictive response biomarkers: a mechanistic study with 12 months follow-up. Ann Rheum Dis. 2014;73(5):913–20.
Ruck T, Bittner S, Kuhlmann T, Wiendl H, Meuth SG. Long-term efficacy of alemtuzumab in polymyositis. Rheumatology (Oxford). 2015;54(3):560–2.
Benveniste O, Guiguet M, Freebody J, Dubourg O, Squier W, Maisonobe T, et al. Long-term observational study of sporadic inclusion body myositis. Brain. 2011;134(Pt 11):3176–84.
Schmidt K, Kleinschnitz K, Rakocevic G, Dalakas MC, Schmidt J. Molecular treatment effects of alemtuzumab in skeletal muscles of patients with IBM. BMC Neurol. 2016;16(1):48.
Amato AA, Sivakumar K, Goyal N, David WS, Salajegheh M, Praestgaard J, et al. Treatment of sporadic inclusion body myositis with bimagrumab. Neurology. 2014;83(24):2239–46.
Mendell JR, Sahenk Z, Al-Zaidy S, Rodino-Klapac LR, Lowes LP, Alfano LN, et al. Follistatin gene therapy for sporadic inclusion body myositis improves functional outcomes. Mol Ther. 2017;25(4):870–9.
Lilleker JB, Bukhari M, Chinoy H. Rapamycin for inclusion body myositis: targeting non-inflammatory mechanisms. Rheumatology (Oxford). 2018;129:611.
Rutkove SB, Parker RA, Nardin RA, Connolly CE, Felice KJ, Raynor EM. A pilot randomized trial of oxandrolone in inclusion body myositis. Neurology. 2002;58(7):1081–7.
Sancricca C, Mora M, Ricci E, Tonali PA, Mantegazza R, Mirabella M. Pilot trial of simvastatin in the treatment of sporadic inclusion-body myositis. Neurol Sci. 2011;32(5):841–7.
Munters LA, Dastmalchi M, Andgren V, Emilson C, Bergegård J, Regardt M, et al. Improvement in health and possible reduction in disease activity using endurance exercise in patients with established polymyositis and dermatomyositis: a multicenter randomized controlled trial with a 1-year open extension follow up. Arthritis Care Res (Hoboken). 2013;65(12):1959–68.
Alexanderson H, Munters LA, Dastmalchi M, Loell I, Heimburger M, Opava CH, et al. Resistive home exercise in patients with recent-onset polymyositis and dermatomyositis – a randomized controlled single-blinded study with a 2-year follow up. J Rheumatol. 2014;41(6):1124–32.
Yoshimasu T, Ohtani T, Sakamoto T, Oshima A, Furukawa F. Topical FK506 (tacrolimus) therapy for facial erythematous lesions of cutaneous lupus erythematosus and dermatomyositis. Eur J Dermatol. 2002;12(1):50–2.
Balin SJ, Wetter DA, Andersen LK, Davis MDP. Calcinosis cutis occurring in association with autoimmune connective tissue disease: the Mayo Clinic experience with 78 patients, 1996–2009. Arch Dermatol. 2012;148(4):455–62.
Arabshahi B, Silverman RA, Jones OY, Rider LG. Abatacept and sodium thiosulfate for treatment of recalcitrant juvenile dermatomyositis complicated by ulceration and calcinosis. J Pediatr. 2012;160(3):520–2.
Walter MC, Lochmüller H, Toepfer M, Schlotter B, Reilich P, Schröder M, et al. High-dose immunoglobulin therapy in sporadic inclusion body myositis: a double-blind, placebo-controlled study. J Neurol. 2000;247(1):22–8.
Dalakas MC, Sonies B, Dambrosia J, Sekul E, Cupler E, Sivakumar K. Treatment of inclusion-body myositis with IVIg: a double-blind, placebo-controlled study. Neurology. 1997;48(3):712–6.
Marie I, Menard JF, Hatron PY, Hachulla E, Mouthon L, Tiev K, et al. Intravenous immunoglobulins for steroid-refractory esophageal involvement related to polymyositis and dermatomyositis: a series of 73 patients. Arthritis Care Res (Hoboken). 2010;62(12):1748–55.
Oh TH, Brumfield KA, Hoskin TL, Stolp KA, Murray JA, Bassford JR. Dysphagia in inflammatory myopathy: clinical characteristics, treatment strategies, and outcome in 62 patients. Mayo Clin Proc. 2007;82(4):441–7.
Langdon PC, Mulcahy K, Shepherd KL, Low VH, Mastaglia FL. Pharyngeal dysphagia in inflammatory muscle diseases resulting from impaired suprahyoid musculature. Dysphagia. 2012;27(3):408–17.
Liu LW, Tarnopolsky M, Armstrong D. Injection of botulinum toxin a to upper esophageal sphincter for oropharyngeal dysphagia in two patients with inclusion body myositis. Can J Gastroenterol. 2004;18(6):397–9.
Teruya A, Kawamura K, Ichikado K, Sato S, Yasuda Y, Yoshioka M. Successful polymyxin B Hemoperfusion treatment associated with serial reduction of serum anti-CADM-140/MDA5 antibody levels in rapidly progressive interstitial lung disease with amyopathic dermatomyositis. Chest. 2013;144(6):1934–6.
Morisset J, Johnson C, Rich E, Collard HR, Lee JS. Management of myositis-related interstitial lung disease. Chest. 2016;150(5):1118–28.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Findlay, A.R., Bucelli, R.C. (2021). Immune and Inflammatory Myopathies. In: Piquet, A.L., Alvarez, E. (eds) Neuroimmunology. Springer, Cham. https://doi.org/10.1007/978-3-030-61883-4_20
Download citation
DOI: https://doi.org/10.1007/978-3-030-61883-4_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-61882-7
Online ISBN: 978-3-030-61883-4
eBook Packages: MedicineMedicine (R0)