Abstract
Persistent infection with hepatitis C virus (HCV) is a major risk factor for hepatocellular carcinoma (HCC). Accumulating evidence suggests that not only inflammation and subsequent fibrosis but also HCV itself are associated with hepatocarcinogenesis. To date, studies using transgenic mouse and cell-culture models, in which HCV proteins are expressed, indicate the direct pathogenicity of HCV, including oncogenic activity. In particular, the core protein of HCV induces excessive oxidative stress by impairing the mitochondrial electron transfer system by disrupting the function of the molecular chaperone, prohibitin. HCV also modulates intracellular signaling pathways, including mitogen-activated protein kinase, promoting the proliferation of hepatocytes. In addition, HCV induces disorders in lipid and glucose metabolism, thereby accelerating the progression of liver fibrosis and the development of HCC. Due to the development of direct-acting antivirals, which was made possible by basic research, HCV can be eradicated from almost all infected patients. However, such patients can develop HCC long after eradication of HCV, suggesting the genetic and/or epigenetic changes induced by HCV may be persistent. These results enhance our understanding of the role of HCV in hepatocarcinogenesis and will facilitate the development of therapeutic and preventive strategies for HCV-induced HCC.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
1 Introduction
Worldwide, about 170 million people are persistently infected with hepatitis C virus (HCV), which induces chronic hepatitis, cirrhosis, and eventually hepatocellular carcinoma (HCC) (Saito et al. 1990). Owing to the recent advances in HCV research, particularly the identification of the JFH-1 strain (Wakita et al. 2005), the HCV lifecycle has been elucidated. Accordingly, direct-acting antivirals (DAAs) against HCV were developed. DAAs can eliminate HCV efficiently and safely, and almost all HCV-infected patients achieve a sustained viral response (SVR). However, HCC can develop even in patients who achieved an SVR, albeit at a lower frequency than in untreated patients. This post-SVR HCC is an important problem in clinical practice.
As mechanisms of hepatocarcinogenesis by HCV, DNA damage induced by cytokines and oxidative stress by chronic inflammation, or mutations of genomic DNA induced by repeated cellular destruction and regeneration, have been considered. In fact, an elevated level of serum alanine aminotransferase (reflecting hepatitis activity) and a lower platelet count (reflecting progression of fibrosis) are predictive of HCC development. However, accumulating in vitro data suggest that the core protein, which constitutes the HCV particle, affects cell proliferation, transcription, and apoptosis, suggesting that HCV itself may be carcinogenic (Koike 2007). Furthermore, we and other groups have reported that transgenic mice harboring the HCV core protein gene developed HCC in the absence of hepatic inflammation (Moriya et al. 1998; Machida et al. 2006; Lerat et al. 2002; Naas et al. 2005). These data indicate that HCV, and particularly its core protein, promote hepatocarcinogenesis by modulating the gene expression and functions of host cells involved in processes necessary for malignant transformation, i.e., HCV itself has oncogenic activity. Also, hepatic steatosis, accumulation of oxidative stress, and insulin resistance, which are frequent in HCV-infected patients, also occur in HCV core-transgenic mice, and mitochondrial dysfunction might contribute to these effects. Therefore, knowledge of the mechanism underlying HCC development in persistent HCV infection is needed.
2 Hepatitis C Virus and Viral Proteins
HCV is an enveloped RNA virus belonging to the family Flaviviridae, and contains a positive-sense, single-stranded RNA genome of approximately 9600 nucleotides (nt) within the nucleocapsid (Houghton et al. 1991). The genome consists of a large open reading frame (ORF) encoding a polyprotein of approximately 3010 amino acids (aa) (Fig. 1). The ORF is contiguous to highly conserved untranslated regions (UTR) at the 5′- and 3′-termini. The complete 5′–UTR consists of 341 nt and acts as an internal ribosomal entry site. This promotes translation of the RNA genome using a cap-independent mechanism rather than ribosome scanning from the 5′–end of a capped molecule.
Structure of the HCV genome. The HCV genome encodes a polyprotein of 3,010 aa, which is processed to structural and nonstructural proteins by cellular or viral proteases. One of the structural proteins, the core protein, has shown a variety of characteristics in vitro and in vivo. ISDR, interferon sensitivity-determining region
The polyprotein is processed by cellular and viral proteases to generate the viral structural and non-structural proteins. The structural proteins, which are encoded by the NH2-terminal quarter of the genome, include the core protein and the envelope proteins, E1 and E2. E2 has an alternative form, E2-p7, which is reportedly associated with virion assembly and release (Joyce et al. 2009, Atoom et al. 2014). NS2, NS3, NS4A, NS4B, NS5A, and NS5B are the non-structural proteins of HCV coded in the remaining portion of the genome. These include serine protease (NS3/4A), NTPase/helicase (NS3), and RNA-dependent RNA polymerase (NS5B).
The core protein of HCV occupies residues 1–191 of the precursor polyprotein and is cleaved between the core and E1 proteins by a host signal peptidase. The C-terminal membrane anchor of the core protein is further processed by a host signal-peptide peptidase (Moradpour et al. 2007). The mature core protein is estimated to consist of 177–179 aa and has a high level of homology among HCV genotypes. The HCV core protein possesses a hydrophilic N-terminal region (domain 1; residues 1–117) followed by a hydrophobic region (domain 2) from residues 118 to 170. Domain 1 is rich in basic residues and is implicated in RNA binding and homo-oligomerization. The amphipathic helices I and II (residues 119–136 and 148–164, respectively) in domain 2 are involved in the association of HCV core protein with lipids (Boulant et al. 2006). In addition, the region spanning residues 112–152 is associated with the membranes of the endoplasmic reticulum (ER) and mitochondria (Suzuki et al. 2005). The core protein is also localized to the nucleus (Miyamoto et al. 2007; Shirakura et al. 2007) and binds to the nuclear proteasome activator 28γ (PA28γ)/REGγ, resulting in its PA28γ-dependent degradation (Moriishi et al. 2003). Autophagy is involved in the degradation of organelles and elimination of microorganisms; disruption of autophagy leads to disorders involving protein deposition. Replication of HCV RNA induces autophagy in a strain-dependent manner, suggesting that HCV harnesses autophagy to prevent cell death and dysfunction of autophagy is implicated in the genotype-specific pathogenesis of HCV (Taguwa et al. 2011).
3 Possible Role of HCV in Hepatocarcinogenesis
The mechanism underlying hepatocarcinogenesis in HCV infection is unclear, despite the fact that nearly 80% of patients with HCC in Japan and 30% of those worldwide (Perz et al. 2006) are persistently infected with HCV (Kiyosawa et al. 1990; Saito et al. 1990; Yotsuyanagi et al. 2000). These lines of evidence prompted us to evaluate the role of HCV in hepatocarcinogenesis. HCV–induced inflammation leads to necrosis of hepatocytes; their subsequent regeneration enhances genetic aberrations in host cells, the accumulation of which leads to HCC. This hypothesis presupposes indirect involvement of HCV in HCC via hepatic inflammation. This poses the question: can inflammation alone explain the high incidence (90% over 15 years) or multicentric nature of HCC in HCV-infected patients?
The putative indirect role of HCV must be weighed against the rarity of HCC in patients with autoimmune hepatitis in which severe inflammation in the liver persists, even after the development of cirrhosis. Therefore, viral proteins may induce neoplasia. This possibility was evaluated by introducing HCV genes into hepatocytes, but the result was negative. This is likely because of the weak carcinogenic activity of HCV, which takes a long time to manifest. Indeed, HCC development in HCV-infected individuals requires 30–40 years. Humanized immunocompromised mice harboring human hepatocytes support HCV replication, but this does not induce HCC. Therefore, investigations of the carcinogenetic activity of HCV in vivo have used transgenic mice.
4 In Vivo Oncogenic Activity of HCV Core Protein in Mice
A major issue regarding the pathogenesis of HCV-associated liver lesions is the direct pathological effects of HCV proteins. Although several strategies have been applied, the relationship between HCV proteins and disease phenotype remains unclear. For this purpose, several lines of mice transgenic for HCV cDNA have been established (Table 1); some carry the entire coding region of the HCV genome (Lerat et al. 2002), the core region only (Machida et al. 2006; Moriya et al. 1997), the envelope region only (Koike et al. 1995; Pasquinelli et al. 1997), the core and envelope regions (Lerat et al. 2002; Naas et al. 2005), and the core to NS2 regions (Wakita et al. 1998). Although mRNA from the NS region of HCV cDNA has been detected in the liver of such transgenic mice (Honda et al. 1999; Lerat et al. 2002), HCV NS proteins have not. The reason for this is unclear but may be because the HCV NS proteins are harmful to mouse development. If so, establishment of mouse strains that produce the HCV proteins at low levels may be feasible.
We have engineered transgenic mouse lines carrying the HCV genome by introducing cDNA of HCV genotype 1b (Moriya et al. 1997; Moriya et al. 1998). The four transgenic mouse lines carry the core gene, envelope genes, NS genes, or only NS5A gene under the same transcriptional regulatory element. Among them, only transgenic mice carrying the core gene developed HCC in two independent lineages (Moriya et al. 1998). The envelope gene-transgenic mice did not develop HCC, despite high levels of the E1 and E2 proteins (Koike et al. 1995; Koike et al. 1997). The transgenic mice carrying the NS genes or NS5A gene also did not develop HCC.
The core gene-transgenic mice, early in life, develop hepatic steatosis, a histologic characteristic of chronic hepatitis C, along with lymphoid follicle formation and bile duct damage (Bach et al. 1992). Thus, the core gene-transgenic mice recapitulate chronic hepatitis C. Notably, significant hepatic inflammation is not observed in these mice. Late in life, the core gene-transgenic mice develop HCC. The development of steatosis and HCC is reproduced in other HCV-transgenic mouse lines, which harbor the entire HCV genome or its structural genes, including the core gene (Lerat et al. 2002; Machida et al. 2006; Naas et al. 2005). Therefore, the HCV core protein per se has oncogenic potential in vivo. In fact, the core protein modulates intracellular signaling pathways, including mitogen-activated protein kinase in vivo (Tsutsumi et al. 2002b; Tsutsumi et al. 2003), which promotes the proliferation of hepatocytes. Further investigation of the core-transgenic mice revealed that the HCV core protein exerts several effects (see below) and may play a role in hepatocarcinogenesis.
5 Induction of Oxidative Stress via Mitochondria by HCV
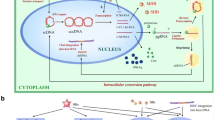
Augmentation of oxidative stress is implicated in the pathogenesis of liver disease in HCV–infected patients (Farinati et al. 1995). Reactive oxygen species (ROS) are endogenous oxygen-containing molecules formed as normal products during aerobic metabolism. ROS can induce genetic mutations as well as chromosomal alterations and thus contribute to carcinogenesis (Fujita et al. 2008; Kato et al. 2001). While the HCV core protein is localized predominantly to the cytoplasm in association with lipid droplets, it is also present in the nucleus and mitochondria (Moriya et al. 1998). Mitochondria are a major source of ROS and HCV induces oxidative stress in vivo as well as in vitro by localizing to mitochondria and disrupting their function. Hepatic ROS production is increased in the HCV core-transgenic mice at an early age, which is compensated for by upregulation of catalase and reduced synthesis of glutathione. However, in older mice, the compensatory effect is inadequate, leading to ROS accumulation in hepatocytes (Moriya et al. 2001). NS5A also induces ER stress and increases Ca efflux, leading to enhanced Ca influx into mitochondria and an increased ROS level (Tardif et al. 2002). Induction of oxidative stress is also observed in vitro in HCV-replicating cells such as subgenomic-replicon cells and JFH-1 cells (Boudreau et al. 2009). In addition, oxidative stress is enhanced in the liver of chimeric mice harboring HCV-infected human hepatocytes (Joyce et al. 2009). Furthermore, patients with chronic hepatitis C have increased oxidative DNA damage in hepatocytes and peripheral leukocytes (Fujita et al. 2008; Yen et al. 2012). These data suggest that HCV directly contributes to hepatocarcinogenesis by inducing oxidative stress, which triggers DNA damage. Also, the main site of HCV-induced ROS production is mitochondria. Indeed, proteomic profiling of biopsy specimens from HCV-infected human livers with advanced fibrosis revealed impairment of both key mitochondrial processes, including fatty acid oxidation, and the response to oxidative stress (Diamond et al. 2007). The mechanism underlying the HCV-induced increased ROS production by mitochondria has been investigated.
HCV core protein is localized to, and induces morphological changes of, mitochondria. In addition, HCV core protein suppresses the activity of complex I in the mitochondrial respiratory chain (Korenaga et al. 2005; Piccoli et al. 2007). This suppression is mediated in part by the direct interaction of HCV core protein with a mitochondrial protein, prohibitin. Prohibitin is a ubiquitously expressed and highly conserved protein that plays the predominant role in inhibiting cell-cycle progression and cellular proliferation by attenuating DNA synthesis (Mishra et al. 2005). It is localized to the nucleus and interacts with transcription factors vital for cell-cycle progression. Mitochondrial prohibitin acts as a chaperone by stabilizing newly synthesized mitochondrial proteins (Nijtmans et al. 2000). By two-dimensional polyacrylamide gel electrophoresis (2D-PAGE) of mitochondria isolated from HepG2 cells stably expressing the HCV core protein, prohibitin was found to be upregulated. We found that the interaction between prohibitin and mitochondria-encoded subunit II of COX is suppressed in core-expressing cells (Tsutsumi et al. 2009). This suggests that HCV core protein disrupts the formation and function of the mitochondrial respiratory chain by interacting with prohibitin and suppressing its function as a chaperone, leading to increased oxidative stress. Indeed, suppression of prohibitin function results in increased ROS production and mice lacking intrahepatic expression of prohibitin exhibits ROS accumulation and HCC development (Theiss et al. 2007; Ko et al. 2010). HCV core protein is also associated with mitophagy, mitochondrion-specific autophagy. Under normal conditions, mitochondria with morphological or functional abnormalities are rapidly removed by mitophagy, but abnormal mitochondria accumulate in the presence of the HCV core protein. This suppressive effect may be due to interaction of the HCV core protein with Parkin, a promoter of mitophagy. Parkin is an E3 ubiquitin ligase predominantly localized to the cytoplasm, but translocates to mitochondria in response to their depolarization owing to activation of PTEN-induced putative kinase 1 (PINK1). In mitochondria, PINK1 ubiquitinates itself and outer mitochondrial proteins, thereby priming mitophagy. However, in the presence of HCV core protein, Parkin is retained in the cytoplasm, leading to suppression of mitophagy (Hara et al. 2014). Other factors associated with mitophagy, such as the ‘mitophagy receptors’ Bnip3 and Nix, may also be targeted by HCV core protein. In any case, the accumulation of abnormal mitochondria caused by the suppression of mitophagy further increases oxidative stress, which contributes to DNA damage, finally leading to the development of HCC.
6 Effect of HCV on Iron Metabolism
As discussed above, chronic hepatitis C is characterized by increased oxidative stress. Iron accumulation in the liver aggravates oxidative stress, as indicated by increased levels of DNA adducts in the liver (Farinati et al. 1995). In addition, iron accumulates in the liver of HCV core-transgenic mice (Moriya et al. 2010). Therefore, the contribution of abnormal iron metabolism by HCV to hepatocarcinogenesis is focused, and in fact, the risk of HCC development is higher in HCV-infected patients with elevated hepatic iron accumulation. As a possible molecular mechanism, HCV core protein modulates the expression of heme-oxygenase-1 (HO-1), a key factor in iron metabolism. HO-1 catalyzes the initial and rate-limiting reaction in heme catabolism and cleaves pro-oxidant heme to biliverdin, which in mammals is converted to bilirubin; both biliverdin and bilirubin have antioxidant activity (Stocker et al. 1987). HO-1 has been suggested to be an important antioxidant in the presence of glutathione depletion (Oguro et al. 1998). Thus, HO-1 is an endogenous protective mechanism against oxidative stress, and particularly iron overload. Also, in cultured cells and transgenic mice, HCV decreases the expression of hepcidin, a protein that suppresses iron absorption from the gastrointestinal tract, leading to increased absorption and subsequent accumulation of iron (Nishina et al. 2008; Miura et al. 2008). Therefore, HCV-induced abnormalities of iron metabolism contribute to ROS accumulation. This notion is supported by the fact that phlebotomy decreases the incidence of HCC in HCV-infected patients.
7 Induction of Hepatic Steatosis and Insulin Resistance by HCV
In patients with chronic hepatitis C, hepatic steatosis and diabetes mellitus are more frequent comorbidities, as compared to patients with other chronic liver diseases. The grade of hepatic steatosis correlates with the intrahepatic HCV load, and hepatic steatosis and insulin resistance are improved by elimination of HCV. Furthermore, hepatic steatosis and insulin resistance occur in HCV core-transgenic mice at an early age in the absence of inflammation, suggesting direct involvement of HCV. Several mechanisms of intrahepatic lipid accumulation by HCV have been proposed (Fig. 2)—upregulation of intrahepatic fatty acid synthesis via the activation of sterol regulatory element binding protein-1 (SREBP1), decreased consumption of fatty acids due to disruption of mitochondrial function, increased import of fatty acids due to insulin resistance, and reduced export of very low-density lipoprotein (VLDL) due to downregulation of the activity of microsomal triglyceride protein (MTP) (Perlemuter et al. 2002). Hepatic steatosis is beneficial to HCV because lipid droplets in hepatocytes are indispensable for viral replication (Miyanari et al. 2007). Insulin resistance is caused by functional suppression of insulin receptor substrate-1, a key factor in the intracellular insulin signaling pathway, due to upregulation of tumor necrosis factor-α and suppressor of cytokine signaling (Tsutsumi et al. 2002b; Shintani et al. 2004; Miyoshi et al. 2005).
Molecular mechanisms of HCV-induced intrahepatic lipid accumulation. HCV, and particularly the core protein, affects several pathways associated with lipid metabolism and induces hepatic steatosis. Underlining indicates cellular proteins or processes affected by HCV. First, the core protein induces insulin resistance, promoting the peripheral release and hepatic uptake of fatty acids. Second, the core protein suppresses the activity of MTP, inhibiting the secretion of VLDL from the liver, resulting in an increased hepatic triglyceride level. Third, the transcription factor, SREBP-1c, is upregulated by the core protein, resulting in increased production of triglycerides. Finally, impaired β-oxidation of fatty acids due to mitochondrial dysfunction induced by the core protein leads to the accumulation of fatty acids. PA28g, proteasome activator 28g; RXRα, retinoid X receptor; LXRα, liver X receptor; SREBP1, sterol regulatory element binding protein-1; FAS, fatty acid synthase; PPARα peroxisome proliferator-activated receptor-α; MTP, microsomal triglyceride transfer protein; VLDL, very low–density lipoprotein
In patients with chronic hepatitis C, the presence of hepatic steatosis or insulin resistance is independently associated with a poor response to interferon-based therapy and progression to fibrosis, and with HCC development in patients with liver cirrhosis. DNA damage accompanied by lipid-induced chronic inflammation and ROS production, and insulin-induced cell proliferation, are possible mechanisms of hepatocarcinogenesis. The HCV core protein activates the nuclear receptors retinoid X receptor-α (RXRα) and peroxisome proliferator-activated receptor-α (PPARα) (Tsutsumi et al. 2002a). PPARα forms a heterodimer with RXRα to modulate the expression of genes related to lipid metabolism. The fact that HCV core-transgenic mice lacking the PPARα gene (core-transgenic/PPARα-knockout mice) do not develop hepatic steatosis and HCC suggests that these nuclear receptors play important roles in HCV-induced hepatocarcinogenesis (Tanaka et al. 2008).
8 Interaction of HCV Core Protein with Host Proteins
HCV proteins, particularly the core protein, are associated with intracellular signaling, transcription, transformation, apoptosis, and autophagy. However, most of the data are from in vitro cell-culture studies, and the results differed according to the cell line or expression system used. Therefore, whether the data reflect the situation in HCV-infected patients is unclear, so demonstrating the effect of HCV in vivo is vital, for which transgenic mice are useful.
Proteasome activator 28γ (PA28γ) interacts with the HCV core protein in vitro in normal hepatocytes of HCV core-transgenic mice (Moriishi et al. 2003). PA28γ is a well-conserved, proteasome-associated protein that mediates the degradation of host proteins by binding to, and regulating the activity of, the 20S proteasome, although the mechanism is unclear. Overexpression of PA28γ promotes degradation of the HCV core protein. In contrast, nuclear accumulation of the HCV core protein occurs in PA28γ-knockout hepatocytes, suggesting degradation by the PA28γ-proteasome system. PA28γ-knockout mice have an almost normal phenotype without pathological changes, but core-transgenic/PA28γ-knockout mice do not develop hepatic steatosis, unlike young core-transgenic mice (Moriishi et al. 2007). The HCV core protein promotes the binding of heterodimers of liver X receptor-α (LXRα) and RXRα to LXR-responsive element, which activates SREBP1c expression, but this effect is downregulated in the absence of PA28γ. Furthermore, the increased accumulation of ROS and the insulin resistance in core-transgenic mice are absent in core-transgenic/PA28γ-knockout mice and, surprisingly, core-transgenic/PA28γ-knockout mice do not develop HCC. These findings suggest that PA28γ plays an important role in the induction of HCC by the HCV core protein. Also, functional activation of PA28γ by interaction with the core protein may induce hepatocarcinogenesis because PA28γ expression is upregulated in several cancers (Chen et al. 2013; Okamura et al. 2003). Given that the development of HCC is prevented in its absence, PA28γ may be a novel therapeutic target.
9 Conclusion
The results of HCV mouse studies indicate that the HCV core protein has carcinogenic activity in vivo; thus, HCV has hepatic oncogenic potential. In transgenic mice, HCV proteins, particularly the core protein, induce hepatic steatosis, mitochondrial dysfunction, insulin resistance, and ROS accumulation, and the interactions of these mechanisms result in HCC development (Fig. 3).
Accumulation of a complete set of cellular genetic aberrations is required for the development of neoplasia, such as colorectal cancer (Kinzler and Vogelstein 1996). Mutations in the APC gene (inactivation), in the K-ras gene (activation), and in the p53 gene (inactivation) accumulate, resulting in the development of colorectal cancer. This theory, Vogelstein-type carcinogenesis, has been extended to other cancers. The induction of HCC by HCV core protein suggests an alternative mechanism of hepatocarcinogenesis. The HCV core protein may enable some of the steps in hepatocarcinogenesis to be skipped, leading to the development of HCC even in the absence of the set of genetic aberrations required for carcinogenesis (Fig. 4). Such non–Vogelstein induction of HCC may explain the unusual events in HCV carriers (Koike 2005).
The role of HCV in hepatocarcinogenesis. Multiple steps are required to induce cancer. Hepatocarcinogenesis requires the accumulation of genetic mutations in hepatocytes. The HCV core protein may enable some of the required steps to be skipped. The effect of the core protein would be one-step up in the stairway to HCC, even in the absence of a set of genetic aberrations necessary for carcinogenesis. Such a non-Vogelstein mechanism may explain the atypical modes of hepatocarcinogenesis in the presence of HCV, such as the very high incidence and multicentric nature of HCC. CRC, colorectal cancer; HCC, hepatocellular carcinoma; APC, adenomatous polyposis coli
Due to the remarkable progress in therapies for HCV infection, almost all HCV-infected patients achieve an SVR, but some cannot eliminate HCV due to the progression of liver fibrosis or drug-resistance mutations. Furthermore, HCC can develop in patients who have achieved an SVR. Therefore, it is important to develop therapeutic strategies to prevent and cure HCC. For this purpose, the above-mentioned mechanisms of HCV-induced hepatocarcinogenesis are useful, and compounds targeting mitochondria, nuclear receptors, or PA28γ may be promising candidate anti–hepatocarcinogenic agents.
References
Alonzi T, Agrati C, Costabile B, Cicchini C, Amicone L, Cavallari C, Rocca CD, Folgori A, Fipaldini C, Poccia F, Monica NL, Tripodi M (2004) Steatosis and intrahepatic lymphocyte recruitment in hepatitis C virus transgenic mice. J Gen Virol 85:1509–1520
Atoom AM, Taylor NG, Russell RS (2014) The elusive function of the hepatitis C virus p7 protein. Virology 462–463:377–387
Bach N, Thung SN, Schaffner F (1992) The histological features of chronic hepatitis C and autoimmune chronic hepatitis: a comparative analysis. Hepatology 15:572–577
Boudreau HE, Emerson SU, Korzeniowska A, Jendrysik MA, Leto TL (2009) Hepatitis C virus (HCV) proteins induce NADPH oxidase 4 expression in a transforming growth factor beta-dependent manner: a new contributor to HCV-induced oxidative stress. J Virol 83(24):12934–12946
Boulant S, Montserret R, Hope RG, Ratinier M, Targett-Adams P, Lavergne JP et al (2006) Structural determinants that target the hepatitis C virus core protein to lipid droplets. J Biol Chem 281:22236–22247
Chen D, Yang X, Huang L, Chi P (2013) The expression and clinical significance of PA28 γ in colorectal cancer. J Investig Med 61:1192–1196
Diamond DL, Jacobs JM, Paeper B, Proll SC, Gritsenko MA, Carithers RL Jr et al (2007) Proteomic profiling of human liver biopsies: hepatitis C virus-induced fibrosis and mitochondrial dysfunction. Hepatology 46:649–657
Farinati F, Cardin R, De Maria N, Della Libera G, Marafin C, Lecis E, Burra P, Floreani A, Cecchetto A, Naccarato R (1995) Iron storage, lipid peroxidation and glutathione turnover in chronic anti-HCV positive hepatitis. J Hepatol 22:449–456
Frelin L, Brenndörfer ED, Ahlén G, Weiland M, Hultgren C, Alheim M, Glaumann H, Rozell B, Milich DR, Bode JG, Sällberg M (2006) The hepatitis C virus and immune evasion: non-structural 3/4A transgenic mice are resistant to lethal tumour necrosis factor alpha mediated liver disease. Gut 55:1475–1483
Fujita N, Sugimoto R, Ma N, Tanaka H, Iwasa M, Kobayashi Y, Kawanishi S, Watanabe S, Kaito M, Takei Y (2008) Comparison of hepatic oxidative DNA damage in patients with chronic hepatitis B and C. J Viral Hepat 15:498–507
Hara Y, Yanatori I, Ikeda M, Kiyokage E, Nishina S, Tomiyama Y, Toida K, Kishi F, Kato N, Imamura M, Chayama K, Hino K (2014) Hepatitis C virus core protein suppresses mitophagy by interacting with parkin in the context of mitochondrial depolarization. Am J Pathol 184:3026–3039
Honda A, Arai Y, Hirota N, Sato T, Ikegaki J, Koizumi T, Hatano M, Kohara M, Moriyama T, Imawari M, Shimotohno K, Tokuhisa T (1999) Hepatitis C virus structural proteins induce liver cell injury in transgenic mice. J Med Virol 59:281–289
Houghton M, Weiner A, Han J, Kuo G, Choo QL (1991) Molecular biology of hepatitis C viruses. Implications for diagnosis, development and control of viral diseases. Hepatology 14:381–388
Joyce MA, Walters KA, Lamb SE, Yeh MM, Zhu LF, Kneteman N, Doyle JS, Katze MG, Tyrrell DL (2009) HCV induces oxidative and ER stress, and sensitizes infected cells to apoptosis in SCID/Alb-uPA mice. PLoS Pathog 5:e1000291
Kato J, Kobune M, Nakamura T, Kuroiwa G, Takada K, Takimoto R, Sato Y, Fujikawa K, Takahashi M, Takayama T, Ikeda T, Niitsu Y (2001) Normalization of elevated hepatic 8-hydroxy-2′-deoxyguanosine levels in chronic hepatitis C patients by phlebotomy and low iron diet. Cancer Res 61:8697–8702
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87:159–170
Kiyosawa K, Sodeyama T, Tanaka E, Gibo Y, Yoshizawa K, Nakano Y et al (1990) Interrelationship of blood transfusion, non-A, non-B hepatitis and hepatocellular carcinoma: analysis by detection of antibody to hepatitis C virus. Hepatology 12:671–675
Ko KS, Tomasi ML, Iglesias-Ara A, French BA, French SW, Ramani K, Lozano JJ, Oh P, He L, Stiles BL, Li TW, Yang H, Martínez-Chantar ML, Mato JM, Lu SC (2010) Liver-specific deletion of prohibitin 1 results in spontaneous liver injury, fibrosis, and hepatocellular carcinoma in mice. Hepatology 52:2096–2108
Koike K (2005) Molecular basis of hepatitis C virus-associated hepatocarcinogenesis: lessons from animal model studies. Clin Gastroenterol Hepatol 3:S132–S135
Koike K (2007) Hepatitis C virus contributes to hepatocarcinogenesis by modulating metabolic and intracellular signaling pathways. J Gastroenterol Hepatol 22(Suppl 1):S108–S111
Koike K, Moriya K, Ishibashi K, Matsuura Y, Suzuki T, Saito I et al (1995) Expression of hepatitis C virus envelope proteins in transgenic mice. J Gen Virol 76:3031–3038
Koike K, Moriya K, Yotsuyanagi H, Shintani Y, Fujie H, Ishibashi K et al (1997) Sialadenitis resembling Sjögren’s syndrome in mice transgenic for hepatitis C virus envelope genes. Proc Natl Acad Sci USA 94:233–236
Korenaga M, Wang T, Li Y, Showalter LA, Chan T, Sun J, Weinman SA (2005) Hepatitis C virus core protein inhibits mitochondrial electron transport and increases reactive oxygen species (ROS) production. J Biol Chem 280:37481–37488
Lerat H, Honda M, Beard MR, Loesch K, Sun J, Yang Y et al (2002) Steatosis and liver cancer in transgenic mice expressing the structural and nonstructural proteins of hepatitis C virus. Gastroenterology 122:352–365
Machida K, Cheng KT, Lai CK, Jeng KS, Sung VM, Lai MM (2006) Hepatitis C virus triggers mitochondrial permeability transition with production of reactive oxygen species, leading to DNA damage and STAT3 activation. J Virol 80:7199–7207
Majumder M, Ghosh AK, Steele R, Zhou XY, Phillips NJ, Ray R, Ray RB (2002) Hepatitis C virus NS5A protein impairs TNF-mediated hepatic apoptosis, but not by an anti-FAS antibody, in transgenic mice. Virology 294:94–105
Mishra S, Murphy LC, Nyomba BL, Murphy LJ (2005) Prohibitin: a potential target for new therapeutics. Trends Mol Med 11:192–197
Miura K, Taura K, Kodama Y, Schnabl B, Brenner DA (2008) Hepatitis C virus-induced oxidative stress suppresses hepcidin expression through increased histone deacetylase activity. Hepatology 48:1420–1429
Miyamoto H, Moriishi K, Moriya K, Murata S, Tanaka K, Suzuki T et al (2007) Hepatitis C virus core protein induces insulin resistance through a PA28γ-dependent pathway. J Virol 81:1727–1735
Miyanari Y, Atsuzawa K, Usuda N, Watashi K, Hishiki T, Zayas M, Bartenschlager R, Wakita T, Hijikata M, Shimotohno K (2007) The lipid droplet is an important organelle for hepatitis C virus production. Nat Cell Biol 9:1089–1097
Miyoshi H, Fujie H, Shintani Y, Tsutsumi T, Shinzawa S, Makuuchi M, Kokudo N, Matsuura Y, Suzuki T, Miyamura T, Moriya K, Koike K (2005) Hepatitis C virus core protein exerts an inhibitory effect on suppressor of cytokine signaling (SOCS)-1 gene expression. J Hepatol 43:757–763
Moradpour D, Penin F, Rice CM (2007) Replication of hepatitis C virus. Nat Rev Microbiol 5:453–463
Moriishi K, Okabayashi T, Nakai K, Moriya K, Koike K, Murata S et al (2003) Proteasome activator PA28γ-dependent nuclear retention and degradation of hepatitis C virus core protein. J Virol 77:10237–10249
Moriishi K, Mochizuki R, Moriya K, Miyamoto H, Mori Y, Abe T et al (2007) Critical role of PA28g in hepatitis C virus-associated steatogenesis and hepatocarcinogenesis. Proc Natl Acad Sci USA 104:1661–1666
Moriya K, Yotsuyanagi H, Shintani Y, Fujie H, Ishibashi K, Matsuura Y et al (1997) Hepatitis C virus core protein induces hepatic steatosis in transgenic mice. J Gen Virol 78:1527–1531
Moriya K, Fujie H, Shintani Y, Yotsuyanagi H, Tsutsumi T, Matsuura Y et al (1998) Hepatitis C virus core protein induces hepatocellular carcinoma in transgenic mice. Nat Med 4:1065–1068
Moriya K, Nakagawa K, Santa T, Shintani Y, Fujie H, Miyoshi H et al (2001) Oxidative stress in the absence of inflammation in a mouse model for hepatitis C virus-associated hepatocarcinogenesis. Cancer Res 61:4365–4370
Moriya K, Miyoshi H, Shinzawa S, Tsutsumi T, Fujie H, Goto K, Shintani Y, Yotsuyanagi H, Koike K (2010) Hepatitis C virus core protein compromises iron-induced activation of antioxidants in mice and HepG2 cells. J Med Virol 82:776–792
Naas T, Ghorbani M, Alvarez-Maya I, Lapner M, Kothary R, De Repentigny Y et al (2005) Characterization of liver histopathology in a transgenic mouse model expressing genotype 1a hepatitis C virus core and envelope proteins 1 and 2. J Gen Virol 86:2185–2196
Nijtmans LG, de Jong L, Artal Sanz M, Coates PJ, Berden JA, Back JW et al (2000) Prohibitins act as a membrane-bound chaperone for the stabilization of mitochondrial proteins. EMBO J 19:2444–2451
Nishina S, Hino K, Korenaga M, Vecchi C, Pietrangelo A, Mizukami Y, Furutani T, Sakai A, Okuda M, Hidaka I, Okita K, Sakaida I (2008) Hepatitis C virus-induced reactive oxygen species raise hepatic iron level in mice by reducing hepcidin transcription. Gastroenterology 134:226–238
Oguro T, Hayashi M, Nakajo S, Numazawa S, Yoshida T (1998) The expression of heme oxygenase-1 gene responded to oxidative stress produced by phorone, a glutathione depletor, in the rat liver; the relevance to activation of c-jun n-terminal kinase. J Pharmacol Exp Ther 287:773–778
Okamura T, Taniguchi S, Ohkura T, Yoshida A, Shimizu H, Sakai M, Maeta H, Fukui H, Ueta Y, Hisatome I, Shigemasa C (2003) Abnormally high expression of proteasome activator-gamma in thyroid neoplasm. J Clin Endocrinol Metab 88:1374–1383
Pasquinelli C, Shoenberger JM, Chung J et al (1997) Hepatitis C virus core and E2 protein expression in transgenic mice. Hepatology 25:719–727
Perlemuter G, Sabile A, Letteron P, Vona G, Topilco A, Koike K et al (2002) Hepatitis C virus core protein inhibits microsomal triglyceride transfer protein activity and very low density lipoprotein secretion: a model of viral-related steatosis. FASEB J 16:185–194
Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP (2006) The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol 45:529–538
Piccoli C, Scrima R, Quarato G, D’Aprile A, Ripoli M, Lecce L et al (2007) Hepatitis C virus protein expression causes calcium-mediated mitochondrial bioenergetic dysfunction and nitro-oxidative stress. Hepatology 46:58–65
Saito I, Miyamura T, Ohbayashi A, Harada H, Katayama T, Kikuchi S et al (1990) Hepatitis C virus infection is associated with the development of hepatocellular carcinoma. Proc Natl Acad Sci USA 87:6547–6549
Shintani Y, Fujie H, Miyoshi H, Tsutsumi T, Kimura S, Moriya K et al (2004) Hepatitis C virus and diabetes: direct involvement of the virus in the development of insulin resistance. Gastroenterology 126:840–848
Shirakura M, Murakami K, Ichimura T, Suzuki R, Shimoji T, Fukuda K et al (2007) E6AP ubiquitin ligase mediates ubiquitylation and degradation of hepatitis C virus core protein. J Virol 81:1174–1185
Suzuki R, Sakamoto S, Tsutsumi T, Rikimaru A, Tanaka K, Shimoike T et al (2005) Molecular determinants for subcellular localization of hepatitis C virus core protein. J Virol 79:1271–1281
Stocker R, Yamamoto Y, McDonagh AF, Glazer AN, Ames BN (1987) Bilirubin is an antioxidant of possible physiological importance. Science 235:1043–1046
Taguwa S, Kambara H, Fujita N, Noda T, Yoshimori T, Koike K, Moriishi K, Matsuura Y (2011) Dysfunction of autophagy participates in vacuole formation and cell death in cells replicating hepatitis C virus. J Virol 85:13185–13194
Tanaka N, Moriya K, Kiyosawa K, Koike K, Gonzalez FJ, Aoyama T (2008) PPARalpha activation is essential for HCV core protein-induced hepatic steatosis and hepatocellular carcinoma in mice. J Clin Invest 118:683–694
Tardif KD, Mori K, Siddiqui A (2002) Hepatitis C virus subgenomic replicons induce endoplasmic reticulum stress activating an intracellular signaling pathway. J Virol 76:7453–7459
Theiss AL, Idell RD, Srinivasan S, Klapproth JM, Jones DP, Merlin D et al (2007) Prohibitin protects against oxidative stress in intestinal epithelial cells. FASEB J 21:197–206
Tsutsumi T, Suzuki T, Shimoike T, Suzuki R, Moriya K, Shintani Y et al (2002a) Interaction of hepatitis C virus core protein with retinoid X receptor alpha modulates its transcriptional activity. Hepatology 35:937–946
Tsutsumi T, Suzuki T, Moriya K, Yotsuyanagi H, Shintani Y, Fujie H et al (2002b) Intrahepatic cytokine expression and AP-1 activation in mice transgenic for hepatitis C virus core protein. Virology 304:415–424
Tsutsumi T, Suzuki T, Moriya K, Shintani Y, Fujie H, Miyoshi H et al (2003) Hepatitis C virus core protein activates ERK and p38 MAPK in cooperation with ethanol in transgenic mice. Hepatology 38:820–828
Tsutsumi T, Matsuda M, Aizaki H, Moriya K, Miyoshi H, Fujie H, Shintani Y, Yotsuyanagi H, Miyamura T, Suzuki T, Koike K (2009) Proteomics analysis of mitochondrial proteins reveals overexpression of a mitochondrial protein chaperone, prohibitin, in cells expressing hepatitis C virus core protein. Hepatology 50:378–386
Wakita T, Taya C, Katsume A et al (1998) Efficient conditional transgene expression in hepatitis C virus cDNA transgenic mice mediated by the Cre/loxP system. J Biol Chem 273:9001–9006
Wakita T, Pietschmann T, Kato T, Date T, Miyamoto M, Zhao Z, Murthy K, Habermann A, Kräusslich HG, Mizokami M, Bartenschlager R, Liang TJ (2005) Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat Med 11:791–796
Yen HH, Shih KL, Lin TT, Su WW, Soon MS, Liu CS (2012) Decreased mitochondrial deoxyribonucleic acid and increased oxidative damage in chronic hepatitis C. World J Gastroenterol 18:5084–5089
Yotsuyanagi H, Shintani Y, Moriya K, Fujie H, Tsutsumi T, Kato T et al (2000) Virological analysis of non-B, non-C hepatocellular carcinoma in Japan: frequent involvement of hepatitis B virus. J Infect Dis 181:1920–1928
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Koike, K., Tsutsumi, T. (2021). The Oncogenic Role of Hepatitis C Virus. In: Wu, TC., Chang, MH., Jeang, KT. (eds) Viruses and Human Cancer. Recent Results in Cancer Research, vol 217. Springer, Cham. https://doi.org/10.1007/978-3-030-57362-1_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-57362-1_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-57361-4
Online ISBN: 978-3-030-57362-1
eBook Packages: MedicineMedicine (R0)