Abstract
Neurofibromatosis 1 (NF1) is one of the most common inherited disorders and affects one in 3000 people worldwide. It is a multisystem genetic disorder that is primarily associated with cutaneous, neurologic, and orthopedic manifestations. People with NF1 may also develop cardiac disease, however their prevalence, natural history, and pathogenesis have only recently received medical and scientific attention. Solid data demonstrate a higher than expected frequency of congenital heart defects among patients with NF1, warranting early diagnosis in NF1 children to prevent potential long-term hemodynamic consequences. Although no straightforward genotype–phenotype correlations have been clearly described in NF1, a few exceptions have been identified, all of which involve cardiac malformations. NF1 patients also display abnormal cardiac functions, some of which are intrinsic to the disease, and others secondary to an associated hypertension. On the molecularlevel, the role of haploinsufficiency for neurofibromin in heart development and function has been the subject of many studies. All of these studies have shown that complex developmental mechanisms are involved in cardiac development.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Neurofibromatosis 1
- Cardiac defects
- Congenital malformations
- Embryology
- Genotype-phenotype correlation
- NF1 microdeletion
- Noonan syndrome
- Watson syndrome
Neurofibromatosis type 1 (NF1) is one of the most common inherited disorders and affects one in 3000 people worldwide. It is a multisystem genetic disorder that is primarily associated with cutaneous, neurologic, and orthopedic manifestations. NF1 is caused by dominant loss-of-function mutations of the tumor suppressor NF1 (Neurofibromin 1; MIM #613113), which is located at 17q11.2 and contains 60 translated exons over ∼280 kb [1]. It is a completely penetrant Mendelian disease marked by an extremely variable clinical expressivity of the major features as well as the less frequent complications [2,3,4].
The major features of NF1 are multiple neurofibromas, café-au-lait macules, intertriginous freckling, Lisch nodules, tibial pseudarthrosis, and a tendency to develop benign and malignant tumors of the nervous system [5]. NF1 is diagnosed primarily on clinical grounds using the National Institutes of Health (NIH) diagnostic criteria [6]. The diagnosis of NF1 requires at least two out of the following seven NIH criteria:
-
6 or more café au lait macules (>0.5 cm in children or >1.5 cm in adults)
-
2 or more cutaneous/subcutaneous neurofibromas or one plexiform neurofibroma
-
Intertriginous (axillary or groin) freckling
-
Optic pathway glioma
-
2 or more Lisch nodules (iris hamartomas seen on slit lamp examination)
-
Bony dysplasia (sphenoid wing dysplasia, bowing of long bone ± pseudarthrosis)
-
First degree relative with NF1
By using these criteria, the diagnosis of NF1 can be made based on physical examination and by evaluating the patient’s family history. Genetic testing is not necessary if the diagnosis has already been clinically confirmed. These criteria usually appear in the following predictable order: café-au-lait macules (CALMs), axillary freckling, Lisch nodules, and neurofibromas. The characteristic osseous lesions usually develop within the first year of life, and the diagnosis of symptomatic optic glioma appears by 3 years of age [7]. However, these diagnostic criteria emerge in a variable manner, with some patients revealing criteria at a late onset and others never developing certain signs and symptoms. Definitive diagnosis may therefore be delayed by years. Indeed, although the NIH diagnostic criteria are useful for the diagnosis of NF1 in the majority of children at the age of 5 years, they are inadequate for reaching a diagnosis at an earlier age [7, 8]. Ninety-seven percent of NF1 patients meet the NIH criteria by the age of 8 years, and all do so by the age of 20 years [7]. On the other hand, only 50% of children younger than 2 years with sporadic NF1 fulfil a single NIH criterion, leading to delayed diagnosis [7].
Genetic testing is therefore useful for confirming the diagnosis in children who present with an unusual phenotype or those who do not meet the diagnostic criteria. Revising these diagnostic criteria is currently a hot topic in the NF1 community which believes that genetic testing should also be a diagnostic criteria; specially, that a positive genetic test shortens the period of diagnostic uncertainty which entails early appropriate surveillance. Furthermore, the clinical picture may be incomplete in some rare forms in adults, requiring genetic testing to confirm the NF1 diagnosis. Patients with limited and localized clinical features and no family history of NF1 may possibly have segmental NF1 [9].
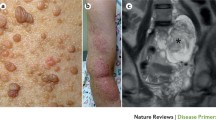
Café-au-lait macules are large, generally oval, well-defined hyperpigmented macules which are usually present at birth and occur in >90% of patients (Figs. 7.1) [7]. CALMs may increase in number and size in early childhood [5]. They have no malignant potential. The majority of patients with NF1 (around 80%) will have more than 5 CALMs by 1 year of age. They become darker with sun exposure and fade with age or become obscured by numerous neurofibromas. Legius syndrome [10, 11], Noonan syndrome and constitutional mismatch repair-deficiency syndrome [12] are other syndromes which exhibit multiple CALMS [13]. Children with multiple CALMs and no other NF1 clinical feature and no family history of NF1 might be referred to genetic testing to confirm a diagnosis of NF1. In a study of 71 patients younger than 20 years of age with six or more CALMs and no non-pigmentary criterion, 66.2% were confirmed to have NF1, 8.5% had Legius syndrome and 25.3% harbored no disease causing variant [14].
Neurofibromas (NF) are benign peripheral nerve sheath tumors and are the cardinal feature of NF1.
Cutaneous (or dermal) NFs are flesh-colored or purplish soft nodules that become pedunculated as they grow (Figs. 7.2 and 7.3). They usually start appearing during late adolescence and are found in the vast majority (>95%) of patients with NF1 [15]. They vary in number from a few lesions to thousands [15]. These tumors are benign and have no risk of malignant transformation, but sometimes lead to significant discomfort and cosmetic disfigurement. Subcutaneous neurofibromas (or peripheral nodular NFs) are firm discrete rubbery nodules bulging under the skin. They affect at least 20% of NF1 patients and usually develop during adolescence [16]. Internal (nodular) NFs are neurofibromas that cannot be felt by physical examination. They are associated with a high-risk phenotype necessitating closer monitoring and management [17]. Plexiform neurofibromas are congenital lesions found in 20 to 26% of individuals with NF1 [18]. They present as a subtle enlargement of soft tissue with a “wrinkled” texture or a patch of hyperpigmentation with or without hypertrichosis. A substantial increase in size occurs during the first decade of life and adolescence [19]. These tumors can invade surrounding structures, including muscle and bone which leads to substantial pain and bone destruction [20].
Malignant peripheral nerve sheath tumors (MPNSTs) are a subtype of sarcoma that arise in preexisting plexiform neurofibromas or subcutaneous NF (Fig. 7.4) [21]. The cumulative lifetime risk of developing MPNST in NF1 patients is 8–13% [21,22,23]. Seventy percent of MPNSTs are high-grade tumors that can metastasize widely and entail a poor prognosis [22]. Symptoms most suggestive of MPNST are a rapid increase in the size of an existing plexiform neurofibroma or alteration in its consistency from soft to hard, persistent or difficult to control pain, or new neurological deficit [22].
Intertriginous freckling is found in >80% of NF1 individuals [5, 15]. They may be found in any area where skinfolds are in apposition, including the axilla, intertriginous area, base of the neck, upper eyelid and under the breasts in women (Figs. 7.5) [24].
Optic pathway gliomas (OPG) are benign tumors seen in 15–20% [25,26,27,28,29,30] of children with NF1. They are slow growing tumors with a low potential of malignancy and they usually occur within the optic pathway including the optic nerve and optic chiasma [26, 31]. They are often indolent; however, due to their space occupying nature they can be locally invasive and become symptomatic [30, 32,33,34,35]. OPG can cause a rapid onset of proptosis leading to moderate-to-severe visual loss in the affected eye [25, 29, 36]. Precocious puberty can occur if the optic pathway tumor impinges on the optic chiasm [29, 37]. Therefore, all children diagnosed with NF1 should have a regular specific pediatric ophthalmological exam.
Lisch nodules are pigmented iris hamartomas that first appear around the age of 3 years and are found in 100% of patients by the age of 30 years [7, 38, 39]. They are asymptomatic 1–2 mm yellow-brown dome shaped papules of the iris; they are best visualized using careful slit-lamp examination of the non-dilated iris.
Congenital dysplasia of the long bones is a classic manifestation of NF1 and occurs in 7.2% of patients [40]. Bowing of long bones leads to a visible deformity and a fragile bone that is susceptible to fracture [41]. Repeated fractures that fail to heal can lead to pseudarthrosis (failure of primary union of the separate bone ends can create a false joint) [40, 42]. Bowing in an NF1 infant necessitates prompt radiographic examination and referral to an orthopedic surgeon.
Sphenoid wing dysplasia is a distinctive feature of NF1 found in a minority of patients (1–7%) [43].
The sphenoid bones comprise multiple ossification centers that fuse to become the important elements of the orbits. Sphenoid wing dysplasia is usually found early in life, is often unilateral and may progress over time. One complication of sphenoid wing dysplasia is a pulsating exophthalmos without visual loss; and an absent sphenoid wing can lead to herniation of the temporal lobe into the orbit [44]. The treatment of sphenoid wing dysplasia should be carried out by a multidisciplinary team including cranio-facial teams familiar with this complex surgery.
People with NF1 may also develop cardiac disease, however their prevalence, natural history, and pathogenesis have only recently received medical and scientific attention. This chapter focuses on the vast array of cardiac manifestations associated with NF1, including congenital and acquired hearing defects, as well as the role of Neurofibromin 1 in cardiac embryogenesis.
Congenital Heart Defects
The incidence and type of congenital heart defects in individuals with NF1 were long undefined and not well-characterized. The previously reported frequencies of congenital heart defects ranged from 0.4 to 8.6% in 8 large series of NF1 patients [45,46,47,48,49,50,51,52]. However, the diagnosis of both NF1 and congenital heart diseases was not clearly established in these patients and not clearly distinguished from Watson and NF1-Noonan syndromes.
It has been suggested that the seemingly conflicting data concerning the prevalence of cardiovascular malformations in NF1 is attributable to the fact that not all NF1 patients are systematically screened by means of echocardiography, especially that the cardiac defect is often mild and asymptomatic. Tedesco et al. were the first to evaluate the prevalence of cardiovascular abnormalities in patients with NF1 using echocardiography with color Doppler scan and found cardiac abnormalities in 13/ 48 young patients (27%) [53]. The same group later described the cardiac abnormalities in 13 out of a total of 69 young patients (18.8%) with NF1 [54]. This was followed by a study in the Turkish population whereby sixty-five NF1 patients were retrospectively studied, looking specifically at their standard electrocardiography and echocardiography. Congenital cardiac abnormalities were found in 11 patients (15.3%) [55]. However, all of these studies are limited by their small sample size.
Two large series have carefully looked at the cardiovascular malformation in patients with NF1. The first one reviewed the cardiovascular abnormalities, in particular the cardiovascular malformations among 2,322 patients with well-established NF1 in the National Neurofibromatosis Foundation International Database (NNFFID) from 1991–98 [56]. A clear diagnosis of cardiovascular disease was found in 97/2,322 (4.2%) of patients with NF1. Cardiovascular malformations were found in 54/2,322 (2.3%) of these NF1 patients, of whom only two had Watson syndrome and only two had NF1-Noonan syndrome. When excluding patients with the latter syndromes, the overall frequency of cardiovascular malformations among NF1 patients was 2%. The second and more recent large series was a retrospective registry-based total population study conducted in Finland that evaluated the congenital anomalies in 465 children with NF1 [57]. NF1 children were found to have a significantly increased risk of congenital cardiovascular anomalies (adjusted OR 3.35), with congenital heart defects occurring in 1.8% of NF1 patients, a figure similar to that reported in the NNFFID study as well as in the studies by Carey et al. [49] and Schorry et al. [50] All of these data demonstrate a higher than expected frequency of congenital heart defects among patients with NF1, warranting early diagnosis in NF1 children to prevent potential long-term hemodynamic consequences.
In the NNFFID study , the predominant (43/54, 80%) cardiovascular malformations were classified as “flow defects,” whereby left or right heart obstruction or simple shunts are thought to be secondary to anomalous embryonic intracardiac hemodynamics [56]. Among these, the majority (25/43) had pulmonary stenosis, usually valvular. When compared to the patients in the Baltimore-Washington Infant Study (BWIS), pulmonary stenosis in NF1 patients constituted a much larger proportion of all cardiovascular malformations than expected. Indeed, the proportion of all cardiovascular malformations that were pulmonary stenosis was 4.3 times greater among NF1 patients. Similarly, individual case reports and smaller series also frequently reported pulmonary stenosis. Fortunately, pulmonary stenosis in patients with NF1 seems to be generally mild and not a source of serious morbidity. It is interesting to note that pulmonary stenosis is also a well-recognized feature of three clinical subtypes of NF1: NF1-Noonan syndrome, Watson syndrome, and individuals with large deletions of the NF1 gene [56, 58]. Pulmonary stenosis should therefore be considered in any NF1 patient with a systolic murmur.
Aortic coarctation was observed in 5/54 of patients in the NNFFID study and was 3.2 times greater among the NF1 patients than in the BWIS population. However, in 3 out of these 5 patients, the coarctation was a fusiform narrowing of the descending thoracic aorta, which is different from the typical juxtaductal shelf usually described [56]; suggesting that aortic coarctation is more of a NF1 vasculopathy rather than a true cardiovascular malformation.
It remains uncertain whether hypertrophic cardiomyopathy (HCM) constitutes a rare manifestation of NF1, or whether their simultaneous occurrence is simply related to chance. None of the reported patients in the NNFFID study had HCM which may have been due to the small sample size and insufficient statistical power to identify this particular association. Lin et al. reviewed the different case reports in the literature: out of the 18 reported patients in the literature at that time, probably 14 had primary HCM (idiopathic hypertrophic subaortic stenosis and left ventricular). None of these patients had molecular analysis for either the NF1 or any of the common HCM mutations [59]. One patient had extrinsic compression by a neurofibroma which resulted in biventricular hypertrophy [60]. In 2 patients, HCM and NF1 were most likely segregating as independent autosomal dominant traits [61]. Histological examination of the myocardium was available in two patients (postmortem examination [62] or biopsy [63]) revealing non-specific changes and fiber disproportion with interstitial fibrosis, respectively. Tedesco et al.’s echocardiography study detected two patients with septal to posterior left ventricular free wall ratio greater than 1.5, suggesting hypertrophic cardiomyopathy [53]. Another case report presented the case of an 18-year-old boy with NF1 with hypertrophic cardiomyopathy diagnosed using echocardiography showing the systolic anteward movement of the anterior leaflet of the mitral valve [64]. Further large series are needed to elucidate whether HCM is truly a rare manifestation of the NF1 mutation or simply a chance incident.
Interestingly, Class I conotruncal defects and Fallot’s tetralogy were rare in the NNFFID study, and there were no atrioventricular canal, anomalous pulmonary venous return, complex single ventricle and laterality defects detected [56]. This is in striking contrast to the Nf1 knockout mouse homozygous mutant embryos discussed below [65].
Table 7.1 describes the different congenital cardiac malformations detected in large series.
Genotype–Phenotype Correlation
The majority of NF1 cases result from truncating mutations or whole gene deletions, causing an expected deterioration or absence of the RNA generated from the mutated allele. This leads to the total amount of protein being translated from a single allele only, creating a dosage effect [66].
For patients with intragenic NF1 mutations which represent over 90% of all NF1 cases, no straightforward genotype–phenotype correlations have been identified except for few exceptions [67].
NF1 and its Related Disorders (NF1-Noonan Syndrome (NFNS) and Watson Syndrome (WS))
Watson syndrome (WS) and NF1–Noonan syndrome (NFNS) were first described clinically and were later shown to be caused by NF1 mutations [68,69,70]. Over 20 years ago, Watson had described three families with an autosomal dominant inheritance of pulmonary stenosis, multiple café-au-lait macules and lower intelligence [71]. Further studies showed that the Watson phenotype is distinct from that of NF1, as adult patients rarely had neurofibromas, if any [72].
Patients with NFNS, have overlapping features of both NF1 and Noonan syndrome. Although at first thought to be a distinct condition, subsequent clinical [51] and molecular studies have proved that the majority of NFNS patients have NF1 gene mutations only, mostly non-truncating mutations consisting of in-frame deletions as compared to typical NF1 [73, 74].
A recent study investigated whether pulmonary stenosis is related to specific types of NF1 gene mutations in NF1, NFNS and WS . The study examined the frequency of different NF1 mutation types in a cohort of published and unpublished cases with NF1/NFNS/WS and pulmonary stenosis [66].
Compared with NF1 in general, NFNS patients had statistically significant higher rates of pulmonary stenosis (9/35 = 26% vs 25/2322 = 1.1%). Further stratification of NFNS group according to the mutation type showed that the higher pulmonary stenosis rate appears to be affected specifically by non-truncating mutations. Eight out of twelve (66.7%) NFNS cases with non-truncating mutations had pulmonary stenosis compared with only 1.1% pulmonary stenosis frequency in NF1 in general. In contrast, only 6.2% (1/16) of NFNS patients with a truncating mutation had pulmonary stenosis (not significantly different from NF1 patients). Looking specifically at individuals with NF1 and pulmonary stenosis, 8/11 (73%) of them had non-truncating mutations, which is largely higher than expected in the NF1 population (19% reported in NF1 cohorts). As for WS, out of the three available published cases with intragenic mutations, two of them had non-truncating mutations. Therefore, non-truncating mutations seem to play a specific role in pulmonary stenosis in NF1 and its related disorders. The authors postulated that unlike in truncating mutations whereby there is decay or absence of the RNA generated from the mutated allele with resulting dosage effect, non-truncating mutations do not result in loss of the NF1 protein. Instead it might result in an abnormally functioning protein with special cardiac effect. This suggests that the pulmonary stenosis is caused by an additional effect of the mutated protein instead of the common loss-of function effect. This effect may be similar to the cardiac effect of Noonan syndrome and other RAS opathies and may characterize a particular role of these mutations in the Ras–MAPK pathway.
NF1 Microdeletion Syndrome (MIM 613675)
In 5–10% of patients, NF1 results from microdeletions that encompass the entire NF1 gene and a variable number of flanking genes [75]. Patients carry a heterozygous deletion of 17q11.2 region usually spanning about 1–1.4 Mb [58, 76]. Typically, they present with a more severe phenotype compared with the one observed in NF1 with intragenic mutation [77].
Numerous studies have described cardiac defects in patients with NF1 microdeletions [52, 58, 77,78,79,80,81,82,83,84,85].
However, the overall frequency of cardiac defects in patients with NF1 microdeletions is still uncertain. Venturin et al. showed a prevalence of cardiovascular malfomations of 18% in the subgroup of patients with NF1 microdeletion [58]. In another study, 8/28 (29%) NF1 microdeletion patients had cardiovascular anomalies [77]. The first thorough investigation of the frequency of heart defects in patients with NF1 microdeletions showed major cardiac abnormalities in 6 of 16 NF1 microdeletion patients whereas none of 16 patients with intragenic NF1 mutations in that study had heart defects [85].
The specific type and frequency of the heart defects described in patients with NF1 microdeletions are quite varied and are shown in Table 7.2.
It is worth mentioning that mitral valve prolapse and aortic dilatation are both associated with connective tissue abnormalities, now generally recognized as a part of NF1 microdeletion syndrome and have been reported in 12% in one series [83].
The higher frequency of cardiovascular malformations in NF1-microdeleted patients is most likely due to the haploinsufficiency of genes lying in the deletion interval, possibly involved in heart morphogenesis. One such gene includes ADAP2 which encodes a protein belonging to the centaurins protein family. Expression analysis studies showed ADAP2 murine ortholog expression in heart during fundamental phases of cardiac morphogenesis [87]. The findings of this study suggested a correlation between ADAP2 haploinsufficiency and the presence of valve defects in NF1-microdeleted patients .
Specific Mutations
A specific mutation consisting of a 3-bp inframe deletion (c.2970–2972 delAAT) in exon 17 of the NF1 gene has recently been found to be associated with a much milder NF1 phenotype with a lack of dermal neurofibromas, and increased rate of pulmonary stenosis [68].
Another genotype-phenotype correlation with cardiac involvement in NF1 are the missense mutations affecting arginine at position 1809. Patients present with pulmonary stenosis, Noonan-like features and developmental delay, but no external plexiform neurofibromas [88, 89]. Affected amino acids reside outside the GAP-related domain (GRD) domain.
A third and more recent genotype-phenotype correlation involves the constitutional missense mutation affecting one of five neighboring NF1 codons—Leu844, Cys845, Ala846, Leu847, and Gly848—located in the cysteine-serine-rich domain (CSRD).
Cardiovascular abnormalities observed in a studied group included hypertension, pulmonic stenosis, mitral valve stenosis, atrial septal defect, ventricular septal defect, Moyamoya disease, pericarditis carcinomatosa, mitral valve insufficiency, mild pulmonary insufficiency, and hypertrophic cardiomyopathy [90].
Acquired Heart Diseases
In addition to congenital heart defects, there is evidence that individuals with NF1 have an increased risk of developing acquired heart diseases. Diastolic function was evaluated in young patients with NF who were free of structural cardiovascular abnormalities using standard Doppler and Pulsed Doppler tissue imaging (DTI), which is a new technique that allows analysis of myocardial velocities and time intervals throughout the cardiac cycle [53]. The main finding of the study are that certain standard and DTI indexes are abnormal in patients with NF as compared to controls, indicating that patients with NF may have abnormal diastolic function. These findings were found in both mild and severe disease, suggesting that the suspected diastolic functional abnormalities may be independent of the clinical severity of NF1. The authors suggested that this may be attributed to cardiac myofibrillar dysplasia or, they may be secondary to abnormal vascular compliance [91].
The same group later evaluated the cardiac function of NF1 patients looking specifically at hypertension. The study revealed the presence of early cardiac morphologic and functional changes in young NF1 patients with hypertension. NF1 hypertensive patients had thicker myocardial walls, increased left ventricular mass index and increased atrial dimension compared with NF1 normotensive patients and healthy subjects [92]. Using standard Doppler, diastolic function was shown to be significantly more impaired in NF1 hypertensives than normotensive NF1 patients and controls. However, using DTI technique (which detects changes induced by hypertension as well as those independent of blood pressure.), myocardial velocities in NF1 patients were found to be significantly higher than controls regardless of blood pressure. Hence the myocardial abnormalities are an early phenotype in patients with NF1 that presents independently of blood pressure and that can be identified using DTI. The authors here postulated that the high DTI values in NF1 normotensives could be explained by the abnormal sympathetic nerve supply to the heart based on the neural crest origin of NF. Furthermore, the presence of a significant decline in myocardial systolic velocity in NF1 hypertensives, when compared with normotensives, could be used as a measure for the long-term follow-up of those patients, since hypertension has been shown to decrease life expectancy in NF1 patients [93].
To sum up, patients with NF1 require a cardiologic assessment at regular intervals. Serial echocardiograms with standard and DTI measurements could provide important information about cardiac damage in NF1 patients diagnosed with hypertension.
Death from Cardiac Disease
Although heart diseases were found to be slightly lower than expected on death certificates of individuals with NF1 [94], sporadic cases of sudden death in adults with NF1 have been reported secondary to coronary artery involvement, vasospasm and myocardial infarction [95,96,97,98,99].
Cardiac diseases related to vascular lesions in patients with NF1 result mainly from an intrinsic pathology of the vessel walls. The NF1 vasculopathy most commonly affects the renal arteries; however, it has also been described in cardiac diseases. It consists of hyperplasia and luminal narrowing, with the proliferating cell type being smooth myocytes. Lesions have been categorized as pure intimal, advanced intimal, intimal-aneurysmal, and nodular [100]. Coronary imaging should therefore be considered in children and adults with NF1 who present with symptoms of angina such as chest pain and syncope.
Table 7.3 describes the different coronary artery anomalies described in NF1 patients in the literature in the form of case reports.
Molecular Basis of Cardiac Malformation and Dysfunction in NF1
The Nf1gene codes for neurofibromin , a large protein of 2800 aa. It includes a Ras-GTPase-activating domain (GAP) capable of accelerating the hydrolysis of GTP-bound Ras, thus down-regulating the activity of Ras proto-oncogenes which are important regulators of cell proliferation, growth, and differentiation [65, 105] Hence, mutations that inactivate Nf1 result in elevated levels of RAS signaling and increased cell proliferation. Further cellular functions for neurofibromin have also been recognized, including modulation of protein kinase A and cyclic adenosine monophosphate pathways [106,107,108]. The full-length of neurofibromin has been shown to bind to the scaffolding domain of Caveolin-1 [109], whereas the C-terminal region of Neurofibromin 1 has been shown to interact with a major class of heparan sulfate proteoglycans [110].
Neurofibromin is a ubiquitous protein [111] and plays a pivotal role in embryogenesis. The role of haploinsufficiency for neurofibromin in heart development and function has been the subject of many studies. Our understanding of the role of Nf1 in embryogenesis stems from animal studies, essentially targeted mouse mutants and more recently zebrafish studies [65, 112,113,114,115,116]. All of these studies have shown that complex developmental mechanisms including neural crest migration are involved in the cardiac development in NF1.
Surprisingly enough, the earliest studies have shown that Nf1 heterozygous mice do not exhibit any in utero cardiovascular malformations. However, Nf1 knockout mouse homozygous mutant embryos have been found to die during midgestation due to outflow tract septation defects (double outlet right ventricle/membranous ventricular septal defect) and concurrent hyperproliferative enlarged endocardial cushions (the precursors of cardiac valves), that could impede forward blood flow [65, 112].
Zebrafish embryos displayed similar cardiovascular abnormalities to those seen in mouse models following transient knockdown of the orthologous Nf1genes (abnormal cardiac valves, presence of pericardial effusions, thinned myocardium) [116].
These cardiovascular malformations were previously thought to be due to abnormal migration of ectomesenchymal cells derived from the cranial neural crest [117]. However, more recent studies demonstrated using conditional gene inactivation that ablating the neurofibromin function specifically in neural crest did not cause cardiac defects. On the other hand, endothelial-specific inactivation of Nf1 recapitulated the cardiovascular phenotype observed in Nf1 knockout embryos: enlarged endocardial cushions, ventricular septal defects, double outlet right ventricle, thinned myocardium and pericardial effusions [113, 114]. These myocardial defects were not reproduced by tissue-specific inactivation of Nf1in myocardial cells indicating that signaling between the endocardium and myocardium is vital for proper myocardial development. Furthermore, loss of Nf1in the embryonic endocardium of these transgenic mice was then shown to be directly associated with an increase in activated GTP-bound Ras with the resulting stimulation of MAPK signaling leading to the abnormal enlargement of the outflow tract septum. Reconstitution of the neurofibromin GRD in endothelial cells is sufficient to rescue cardiovascular development and midgestational lethality in Nf1null embryos; however these mice succumb in the early postnatal period due to overgrowth of neural crest–derived tissues, suggesting that other domains outside the GRD are important for neural crest growth and homeostasis [114].
A more recent study also identified ectopic cardiac blood island formation as a new phenotype that arises in Nf1-deficient murine embryos and that this phenotype is a direct result of dysregulation of the Ras signaling pathway [118].
In a further study, neurofibromin was also shown to function as an important Ras-GAP in the myocardium of adult mice. Tissue-specific inactivation of Nf1 in myocardial cells during mid-gestation led to no distinct phenotype at birth or in young adults, consistent with the aforementioned observations [65, 119]. However, as these cardiac Nf1 deficient mice aged, they developed progressive cardiac hypertrophy, dilatation, fibrosis, and failure with secondary premature mortality. Reconstitution of the Ras-GAP activity in Nf1 deficient myocardial cells led to the rescue of Ras activity and its downstream effectors, along with partial improvement of in the cardiac dysfunction and hypertrophy [119].
The role of Ras activation in cardiac hypertrophy is better highlighted when evaluating patients with Noonan, Costello, and Cardio-facial-cutaneous (CFC) syndromes. Each of these syndromes can be caused by activating mutations in components of the Ras-MAPK pathway and they are all strongly associated with cardiovascular defects including hypertrophic cardiomyopathy [120,121,122].
Parallels have been drawn with Noonan syndrome specifically as the spectrum of heart defects in mouse models of both NF1 and Noonan syndrome is markedly similar, including the enlarged endocardial cushions and double outlet right ventricle [123, 124]. These similarities imply that the in utero cardiovascular manifestations of Nf1 null mice appear to result from endothelially driven hyperproliferation of the endocardial cushions associated with ERK activation.
Conclusion
Although once thought as a rare manifestation of NF1, cardiac malformations are now known to have a higher than expected frequency among patients with NF1 and occur in 2% of these patients. This warrants early diagnosis in NF1 children to prevent potential long-term hemodynamic consequences. Pulmonary stenosis was found to be the most common cardiac defect and related to non-truncating mutations in the Nf1 gene. NF1 patients also present with cardiac function anomalies such as abnormal diastolic function which were found to be independent of the clinical severity of NF1. Furthermore, NF1 patients exhibit early cardiac functional changes independent of blood pressure which can be identified using DTI. Therefore, NF1 patients require cardiology assessment at regular intervals. Serial echocardiograms with standard and DTI measurements could provide important information about cardiac damage in NF1 patients, especially those diagnosed with hypertension. Animal studies have helped tremendously in understanding the role of Nf1 in embryogenesis showing that complex developmental mechanisms are involved in cardiac development. Further experiments are needed to better understand disease pathogenesis of cardiovascular malformations in order to develop potential therapeutic strategies for patients with NF1.
References
Gutmann DH, Ferner RE, Listernick RH, Korf BR, Wolters PL, Johnson KJ. Neurofibromatosis type 1. Nat Rev Dis Primers. 2017;3:17004. https://doi.org/10.1038/nrdp.2017.4.
Easton DF, Ponder MA, Huson SM, Ponder BA. An analysis of variation in expression of neurofibromatosis (NF) type 1 (NF1): evidence for modifying genes. Am J Hum Genet. 1993;53(2):305–13.
Sabbagh A, Pasmant E, Laurendeau I, Parfait B, Barbarot S, Guillot B, et al. Unravelling the genetic basis of variable clinical expression in neurofibromatosis 1. Hum Mol Genet. 2009;18(15):2768–78. https://doi.org/10.1093/hmg/ddp212.
Szudek J, Joe H, Friedman JM. Analysis of intrafamilial phenotypic variation in neurofibromatosis 1 (NF1). Genet Epidemiol. 2002;23(2):150–64. https://doi.org/10.1002/gepi.1129.
Ferner RE, Huson SM, Thomas N, Moss C, Willshaw H, Evans DG, et al. Guidelines for the diagnosis and management of individuals with neurofibromatosis 1. J Med Genet. 2007;44(2):81–8. https://doi.org/10.1136/jmg.2006.045906.
Neurofibromatosis. Conference statement. National Institutes of Health consensus development conference. Arch Neurol. 1988;45(5):575–8.
DeBella K, Szudek J, Friedman JM. Use of the national institutes of health criteria for diagnosis of neurofibromatosis 1 in children. Pediatrics. 2000;105(3 Pt 1):608–14.
Obringer AC, Meadows AT, Zackai EH. The diagnosis of neurofibromatosis-1 in the child under the age of 6 years. Am J Dis Child (1960). 1989;143(6):717–9.
Ruggieri M, Huson SM. The clinical and diagnostic implications of mosaicism in the neurofibromatoses. Neurology. 2001;56(11):1433–43.
Brems H, Chmara M, Sahbatou M, Denayer E, Taniguchi K, Kato R, et al. Germline loss-of-function mutations in SPRED1 cause a neurofibromatosis 1-like phenotype. Nat Genet. 2007;39(9):1120–6. https://doi.org/10.1038/ng2113.
Pasmant E, Sabbagh A, Hanna N, Masliah-Planchon J, Jolly E, Goussard P, et al. SPRED1 germline mutations caused a neurofibromatosis type 1 overlapping phenotype. J Med Genet. 2009;46(7):425–30. https://doi.org/10.1136/jmg.2008.065243.
Wimmer K, Etzler J. Constitutional mismatch repair-deficiency syndrome: have we so far seen only the tip of an iceberg? Hum Genet. 2008;124(2):105–22. https://doi.org/10.1007/s00439-008-0542-4.
Roberts AE, Allanson JE, Tartaglia M, Gelb BD. Noonan syndrome. Lancet (London, England). 2013;381(9863):333–42. https://doi.org/10.1016/s0140-6736(12)61023-x.
Evans DG, Bowers N, Burkitt-Wright E, Miles E, Garg S, Scott-Kitching V, et al. Comprehensive RNA analysis of the NF1 gene in classically affected NF1 affected individuals meeting NIH criteria has high sensitivity and mutation negative testing is reassuring in isolated cases with pigmentary features only. EBioMedicine. 2016;7:212–20. https://doi.org/10.1016/j.ebiom.2016.04.005.
Korf BR. Diagnostic outcome in children with multiple cafe au lait spots. Pediatrics. 1992;90(6):924–7.
Duong TA, Bastuji-Garin S, Valeyrie-Allanore L, Sbidian E, Ferkal S, Wolkenstein P. Evolving pattern with age of cutaneous signs in neurofibromatosis type 1: a cross-sectional study of 728 patients. Dermatology. 2011;222(3):269–73. https://doi.org/10.1159/000327379.
Sbidian E, Bastuji-Garin S, Valeyrie-Allanore L, Ferkal S, Lefaucheur JP, Drouet A, et al. At-risk phenotype of neurofibromatose-1 patients: a multicentre case-control study. Orphanet J Rare Dis. 2011;6:51. https://doi.org/10.1186/1750-1172-6-51.
Darrigo LG Jr, Geller M, Bonalumi Filho A, Azulay DR. Prevalence of plexiform neurofibroma in children and adolescents with type I neurofibromatosis. J Pediatr. 2007;83(6):571–3. https://doi.org/10.2223/JPED.1718.
Tucker T, Friedman JM, Friedrich RE, Wenzel R, Funsterer C, Mautner VF. Longitudinal study of neurofibromatosis 1 associated plexiform neurofibromas. J Med Genet. 2009;46(2):81–5. https://doi.org/10.1136/jmg.2008.061051.
Mautner VF, Hartmann M, Kluwe L, Friedrich RE, Funsterer C. MRI growth patterns of plexiform neurofibromas in patients with neurofibromatosis type 1. Neuroradiology. 2006;48(3):160–5. https://doi.org/10.1007/s00234-005-0033-4.
Evans D, Baser M, McGaughran J, Sharif S, Howard E, Moran A. Malignant peripheral nerve sheath tumours in neurofibromatosis 1. J Med Genet. 2002;39(5):311–4. https://doi.org/10.1136/jmg.39.5.311.
Ferner RE, Gutmann DH. International consensus statement on malignant peripheral nerve sheath tumors in neurofibromatosis. Cancer Res. 2002;62(5):1573–7.
Ducatman BS, Scheithauer BW, Piepgras DG, Reiman HM, Ilstrup DM. Malignant peripheral nerve sheath tumors. A clinicopathologic study of 120 cases. Cancer. 1986;57(10):2006–21.
Friedman JM. Neurofibromatosis 1: clinical manifestations and diagnostic criteria. J Child Neurol. 2002;17(8):548–554. https://doi.org/10.1177/088307380201700802. discussion 71–2, 646–51.
Listernick R, Charrow J, Greenwald MJ, Esterly NB. Optic gliomas in children with neurofibromatosis type 1. J Pediatr. 1989;114(5):788–92.
Lewis RA, Gerson LP, Axelson KA, Riccardi VM, Whitford RP. von Recklinghausen neurofibromatosis. II. Incidence of optic gliomata. Ophthalmology. 1984;91(8):929–35.
Lund AM, Skovby F. Optic gliomas in children with neurofibromatosis type 1. Eur J Pediatr. 1991;150(12):835–8.
Prada CE, Hufnagel RB, Hummel TR, Lovell AM, Hopkin RJ, Saal HM, et al. The use of magnetic resonance imaging screening for optic pathway gliomas in children with neurofibromatosis type 1. J Pediatrics. 2015;167(4):851–6.e1. https://doi.org/10.1016/j.jpeds.2015.07.001.
Blazo MA, Lewis RA, Chintagumpala MM, Frazier M, McCluggage C, Plon SE. Outcomes of systematic screening for optic pathway tumors in children with Neurofibromatosis Type 1. Am J Med Genet Part A. 2004;127A(3):224–9. https://doi.org/10.1002/ajmg.a.20650.
Listernick R, Charrow J, Greenwald M, Mets M. Natural history of optic pathway tumors in children with neurofibromatosis type 1: a longitudinal study. J Pediatr. 1994;125(1):63–6.
Molloy PT, Bilaniuk LT, Vaughan SN, Needle MN, Liu GT, Zackai EH, et al. Brainstem tumors in patients with neurofibromatosis type 1: a distinct clinical entity. Neurology. 1995;45(10):1897–902.
Slamovits TL. Von Recklinghausen neurofibromatosis: II. Incidence of optic gliomata. Ophthalmology. 1985;92(5):714–5.
Kornreich L, Blaser S, Schwarz M, Shuper A, Vishne TH, Cohen IJ, et al. Optic pathway glioma: correlation of imaging findings with the presence of neurofibromatosis. AJNR Am J Neuroradiol. 2001;22(10):1963–9.
Deliganis AV, Geyer JR, Berger MS. Prognostic significance of type 1 neurofibromatosis (von Recklinghausen disease) in childhood optic glioma. Neurosurgery. 1996;38(6):1114–8. discussion 8–9
Listernick R, Darling C, Greenwald M, Strauss L, Charrow J. Optic pathway tumors in children: the effect of neurofibromatosis type 1 on clinical manifestations and natural history. J Pediatr. 1995;127(5):718–22.
Listernick R, Louis DN, Packer RJ, Gutmann DH. Optic pathway gliomas in children with neurofibromatosis 1: consensus statement from the NF1 optic pathway glioma task force. Ann Neurol. 1997;41(2):143–9. https://doi.org/10.1002/ana.410410204.
Laue L, Comite F, Hench K, Loriaux DL, Cutler GB Jr, Pescovitz OH. Precocious puberty associated with neurofibromatosis and optic gliomas. Treatment with luteinizing hormone releasing hormone analogue. Am J Dis Child (1960). 1985;139(11):1097–100.
Lewis RA, Riccardi VM. Von Recklinghausen neurofibromatosis. Incidence of iris hamartomata. Ophthalmology. 1981;88(4):348–54.
Otsuka F, Kawashima T, Imakado S, Usuki Y, Hon-Mura S. Lisch nodules and skin manifestation in neurofibromatosis type 1. Arch Dermatol. 2001;137(2):232–3.
Boulanger JM, Larbrisseau A. Neurofibromatosis type 1 in a pediatric population: Ste-Justine’s experience. Can J Neurol Sci. 2005;32(2):225–31.
Heerva E, Koffert A, Jokinen E, Kuorilehto T, Peltonen S, Aro HT, et al. A controlled register-based study of 460 neurofibromatosis 1 patients: increased fracture risk in children and adults over 41 years of age. J Bone Miner Res Off J Am Soc Bone Miner Res. 2012;27(11):2333–7. https://doi.org/10.1002/jbmr.1685.
Ferner RE. Neurofibromatosis 1. Eur J Hum Genet. 2007;15(2):131–8. https://doi.org/10.1038/sj.ejhg.5201676. [pii]
Pinson S, Creange A, Barbarot S, Stalder JF, Chaix Y, Rodriguez D et al. Recommendations for the treatment of neurofibromatosis type 1. J Fr Ophtalmol. 2002;25(4):423–33. MDOI-JFO-04-2002-25-4-0181-5512-101019-ART13 [pii].
Ferner RE. Neurofibromatosis 1 and neurofibromatosis 2: a twenty first century perspective. The Lancet Neurology. 2007;6(4):340–51. https://doi.org/10.1016/s1474-4422(07)70075-3.
Crowe FW, Schull WJ, Nee JV. A clinical, pathological, and genetic study of multiple neurofibromatosis…. Charles C. Thomas: Springfield, IL; 1956.
Kaufman R, Hartmann A, McAlister WH. Family studies in congenital heart disease IV: congenital heart disease associated with neurofibromatosis. Birth Defects Orig Artic Ser. 1972;7:92–5.
Neiman HL, Mena E, Holt JF, Stern AM, Perry BL. Neurofibromatosis and congenital heart disease. Am J Roentgenol Radium Therapy, Nucl Med. 1974;122(1):146–9.
Holt JF. 1977 Edward B. D. Neuhauser lecture: neurofibromatosis in children. AJR Am J Roentgenol. 1978;130(4):615–39. https://doi.org/10.2214/ajr.130.4.615.
Carey JC, Laub JM, Hall BD. Penetrance and variability in neurofibromatosis: a genetic study of 60 families. Birth Defects Orig Artic Ser. 1979;15(5B):271–81.
Schorry EK, Stowens DW, Crawford AH, Stowens PA, Schwartz WR, Dignan PS. Summary of patient data from a multidisciplinary neurofibromatosis clinic. Neurofibromatosis. 1989;2(2):129–34.
Colley A, Donnai D, Evans DG. Neurofibromatosis/Noonan phenotype: a variable feature of type 1 neurofibromatosis. Clin Genet. 1996;49(2):59–64.
Tonsgard JH, Yelavarthi KK, Cushner S, Short MP, Lindgren V. Do NF1 gene deletions result in a characteristic phenotype? Am J Med Genet. 1997;73(1):80–6.
Tedesco MA, Di Salvo G, Natale F, Caputo S, Calabrese E, Grassia C, et al. Cardiac abnormalities detected by Doppler imaging in patients with neurofibromatosis type 1. Am J Cardiol. 2001;88(10):1198–200.
Tedesco MA, Di Salvo G, Natale F, Pergola V, Calabrese E, Grassia C, et al. The heart in neurofibromatosis type 1: an echocardiographic study. Am Heart J. 2002;143(5):883–8.
Incecik F, Herguner OM, Alinc Erdem S, Altunbasak S. Neurofibromatosis type 1 and cardiac manifestations. Turk Kardiyol Dern Ars. 2015;43(8):714–6. https://doi.org/10.5543/tkda.2015.27557.
Lin AE, Birch PH, Korf BR, Tenconi R, Niimura M, Poyhonen M, et al. Cardiovascular malformations and other cardiovascular abnormalities in neurofibromatosis 1. Am J Med Genet. 2000;95(2):108–17.
Leppavirta J, Kallionpaa RA, Uusitalo E, Vahlberg T, Poyhonen M, Peltonen J, et al. Congenital anomalies in neurofibromatosis 1: a retrospective register-based total population study. Orphanet J Rare Dis. 2018;13(1):5. https://doi.org/10.1186/s13023-017-0756-4.
Venturin M, Guarnieri P, Natacci F, Stabile M, Tenconi R, Clementi M, et al. Mental retardation and cardiovascular malformations in NF1 microdeleted patients point to candidate genes in 17q11.2. J Med Genet. 2004;41(1):35–41.
Coonar AS, McKenna WJ. Molecular genetics of familial cardiomyopathies. Adv Genet. 1997;35:285–324.
Pung S, Hirsch EF. Plexiform neurofibromatosis of the heart and neck. AMA Arch Pathol. 1955;59(3):341–6.
Wille LE, Forre O, Steffensen RW. A familial syndrome with von Recklinghausen’s neurofibromatosis, gammopathy and aorta outflow obstruction. Acta Med Scand. 1980;207(4):297–304.
Sachs RN, Buschauer-Bonnet C, Kemeny JL, Amouroux J, Lanfranchi J. Hypertrophic myocardiopathy and von Recklinghausen’s disease. Rev Med Interne. 1984;5(2):154–6.
Schräder R, Kunkel B, Schneider M, Kaltenbach M. Hypertrophische Kardiomyopathie bei einer Patientin mit Neurofibromatose von Recklinghausen. Med Klin. 1986;81:264–7.
Jurko A Jr, Minarik M, Minarikova E, Nagi AS. Neurofibromatosis associated with hypertrophic cardiomyopathy. Saudi Med J. 2010;31(8):935–6.
Brannan CI, Perkins AS, Vogel KS, Ratner N, Nordlund ML, Reid SW, et al. Targeted disruption of the neurofibromatosis type-1 gene leads to developmental abnormalities in heart and various neural crest-derived tissues. Genes Dev. 1994;8(9):1019–29.
Ben-Shachar S, Constantini S, Hallevi H, Sach EK, Upadhyaya M, Evans GD, et al. Increased rate of missense/in-frame mutations in individuals with NF1-related pulmonary stenosis: a novel genotype-phenotype correlation. Eur J Hum Genet. 2013;21(5):535–9. https://doi.org/10.1038/ejhg.2012.221.
Sabbagh A, Pasmant E, Imbard A, Luscan A, Soares M, Blanche H, et al. NF1 molecular characterization and neurofibromatosis type I genotype-phenotype correlation: the French experience. Hum Mutat. 2013;34(11):1510–8. https://doi.org/10.1002/humu.22392.
Upadhyaya M, Huson SM, Davies M, Thomas N, Chuzhanova N, Giovannini S, et al. An absence of cutaneous neurofibromas associated with a 3-bp inframe deletion in exon 17 of the NF1 gene (c.2970–2972 delAAT): evidence of a clinically significant NF1 genotype-phenotype correlation. Am J Hum Genet. 2007;80(1):140–51. https://doi.org/10.1086/510781.
Tassabehji M, Strachan T, Sharland M, Colley A, Donnai D, Harris R, et al. Tandem duplication within a neurofibromatosis type 1 (NF1) gene exon in a family with features of Watson syndrome and Noonan syndrome. Am J Hum Genet. 1993;53(1):90–5.
Upadhyaya M, Shen M, Cherryson A, Farnham J, Maynard J, Huson SM, et al. Analysis of mutations at the neurofibromatosis 1 (NF1) locus. Hum Mol Genet. 1992;1(9):735–40.
Watson GH. Pulmonary stenosis, cafe-au-lait spots, and dull intelligence. Arch Dis Child. 1967;42(223):303–7.
Allanson JE, Upadhyaya M, Watson GH, Partington M, MacKenzie A, Lahey D, et al. Watson syndrome: is it a subtype of type 1 neurofibromatosis? J Med Genet. 1991;28(11):752–6.
De Luca A, Bottillo I, Sarkozy A, Carta C, Neri C, Bellacchio E, et al. NF1 gene mutations represent the major molecular event underlying neurofibromatosis-Noonan syndrome. Am J Hum Genet. 2005;77(6):1092–101. https://doi.org/10.1086/498454.
Nystrom AM, Ekvall S, Allanson J, Edeby C, Elinder M, Holmstrom G, et al. Noonan syndrome and neurofibromatosis type I in a family with a novel mutation in NF1. Clin Genet. 2009;76(6):524–34. https://doi.org/10.1111/j.1399-0004.2009.01233.x.
Leppig KA, Kaplan P, Viskochil D, Weaver M, Ortenberg J, Stephens K. Familial neurofibromatosis 1 microdeletions: cosegregation with distinct facial phenotype and early onset of cutaneous neurofibromata. Am J Med Genet. 1997;73(2):197–204.
Pasmant E, Sabbagh A, Spurlock G, Laurendeau I, Grillo E, Hamel MJ, et al. NF1 microdeletions in neurofibromatosis type 1: from genotype to phenotype. Hum Mutat. 2010;31(6):E1506–18. https://doi.org/10.1002/humu.21271.
Mautner VF, Kluwe L, Friedrich RE, Roehl AC, Bammert S, Hogel J, et al. Clinical characterisation of 29 neurofibromatosis type-1 patients with molecularly ascertained 1.4 Mb type-1 NF1 deletions. J Med Genet. 2010;47(9):623–30. https://doi.org/10.1136/jmg.2009.075937.
Kayes LM, Burke W, Riccardi VM, Bennett R, Ehrlich P, Rubenstein A, et al. Deletions spanning the neurofibromatosis 1 gene: identification and phenotype of five patients. Am J Hum Genet. 1994;54(3):424–36.
Wu BL, Schneider GH, Korf BR. Deletion of the entire NF1 gene causing distinct manifestations in a family. Am J Med Genet. 1997;69(1):98–101.
Dorschner MO, Sybert VP, Weaver M, Pletcher BA, Stephens K. NF1 microdeletion breakpoints are clustered at flanking repetitive sequences. Hum Mol Genet. 2000;9(1):35–46.
Riva P, Corrado L, Natacci F, Castorina P, Wu BL, Schneider GH, et al. NF1 microdeletion syndrome: refined FISH characterization of sporadic and familial deletions with locus-specific probes. Am J Hum Genet. 2000;66(1):100–9. https://doi.org/10.1086/302709.
Oktenli C, Saglam M, Demirbas S, Thompson P, Upadhyaya M, Consoli C, et al. A large deletion (1.5 Mb) encompassing the neurofibromatosis type 1 (NF1) gene in a patient with sporadic NF1 associated with dysmorphism, mental retardation, and unusual ocular and skeletal features. Clin Dysmorphol. 2003;12(3):199–201. https://doi.org/10.1097/01.mcd.0000077565.66911.43.
Mensink KA, Ketterling RP, Flynn HC, Knudson RA, Lindor NM, Heese BA, et al. Connective tissue dysplasia in five new patients with NF1 microdeletions: further expansion of phenotype and review of the literature. J Med Genet. 2006;43(2):e8. https://doi.org/10.1136/jmg.2005.034256.
De Luca A, Bottillo I, Dasdia MC, Morella A, Lanari V, Bernardini L, et al. Deletions of NF1 gene and exons detected by multiplex ligation-dependent probe amplification. J Med Genet. 2007;44(12):800–8. https://doi.org/10.1136/jmg.2007.053785.
Nguyen R, Mir TS, Kluwe L, Jett K, Kentsch M, Mueller G, et al. Cardiac characterization of 16 patients with large NF1 gene deletions. Clin Genet. 2013;84(4):344–9. https://doi.org/10.1111/cge.12072.
Upadhyaya M, Roberts SH, Maynard J, Sorour E, Thompson PW, Vaughan M, et al. A cytogenetic deletion, del(17)(q11.22q21.1), in a patient with sporadic neurofibromatosis type 1 (NF1) associated with dysmorphism and developmental delay. J Med Genet. 1996;33(2):148–52.
Venturin M, Carra S, Gaudenzi G, Brunelli S, Gallo GR, Moncini S, et al. ADAP2 in heart development: a candidate gene for the occurrence of cardiovascular malformations in NF1 microdeletion syndrome. J Med Genet. 2014;51(7):436–43. https://doi.org/10.1136/jmedgenet-2013-102240.
Rojnueangnit K, Xie J, Gomes A, Sharp A, Callens T, Chen Y, et al. High incidence of Noonan syndrome features including Short stature and pulmonic stenosis in patients carrying NF1 missense mutations affecting p.Arg1809: genotype-phenotype correlation. Hum Mutat. 2015;36(11):1052–63. https://doi.org/10.1002/humu.22832.
Pinna V, Lanari V, Daniele P, Consoli F, Agolini E, Margiotti K, et al. P.Arg1809Cys substitution in neurofibromin is associated with a distinctive NF1 phenotype without neurofibromas. Eur J Hum Genet. 2015;23(8):1068–71. https://doi.org/10.1038/ejhg.2014.243.
Koczkowska M, Chen Y, Callens T, Gomes A, Sharp A, Johnson S, et al. Genotype-phenotype correlation in NF1: evidence for a more severe phenotype associated with missense mutations affecting NF1 codons 844–848. Am J Hum Genet. 2018;102(1):69–87. https://doi.org/10.1016/j.ajhg.2017.12.001.
Tedesco MA, Di Salvo G, Ratti G, Natale F, Calabrese E, Grassia C, et al. Arterial distensibility and ambulatory blood pressure monitoring in young patients with neurofibromatosis type 1. Am J Hypertens. 2001;14(6 Pt 1):559–66.
Tedesco MA, Di Salvo G, Natale F, Graziano L, Grassia C, Calabro R, et al. Early cardiac morphologic and functional changes in neurofibromatosis type 1 hypertensives: an echocardiographic and tissue Doppler study. Int J Cardiol. 2005;101(2):243–7. https://doi.org/10.1016/j.ijcard.2004.03.028.
Zoller M, Rembeck B, Akesson HO, Angervall L. Life expectancy, mortality and prognostic factors in neurofibromatosis type 1. A twelve-year follow-up of an epidemiological study in Goteborg, Sweden. Acta Derm Venereol. 1995;75(2):136–40.
Evans DG, O’Hara C, Wilding A, Ingham SL, Howard E, Dawson J, et al. Mortality in neurofibromatosis 1: in north West England: an assessment of actuarial survival in a region of the UK since 1989. Eur J Hum Genet. 2011;19(11):1187–91. https://doi.org/10.1038/ejhg.2011.113.
Kanter RJ, Graham M, Fairbrother D, Smith SV. Sudden cardiac death in young children with neurofibromatosis type 1. J Pediatr. 2006;149(5):718–20. https://doi.org/10.1016/j.jpeds.2006.07.046.
Daly MP, Rubinstein MN. A case of neurofibromatosis associated with a coronary artery aneurysm and myocardial infarction. Clin Cardiol. 1992;15(8):616–8.
Nogami A, Hiroe M, Marumo F. Regional sympathetic denervation in von Recklinghausen’s disease with coronary spasm and myocarditis. Int J Cardiol. 1991;32(3):397–400.
Hamilton SJ, Allard MF, Friedman JM. Cardiac findings in an individual with neurofibromatosis 1 and sudden death. Am J Med Genet. 2001;100(2):95–9.
Fuchi T, Ishimoto N, Kajinami T, Kajinami M, Ohmichi N, Kinoshita M. A 23-year-old patient with neurofibromatosis associated with acute myocardial infarction, vasospasm and a coronary artery ectasis. Intern Med. 1997;36(9):618–23.
Hamilton SJ, Friedman JM. Insights into the pathogenesis of neurofibromatosis 1 vasculopathy. Clin Genet. 2000;58(5):341–4.
Ruggieri M, D’Arrigo G, Abbate M, Distefano A, Upadhyaya M. Multiple coronary artery aneurysms in a child with neurofibromatosis type 1. Eur J Pediatr. 2000;159(7):477–80.
Kandarpa K, Stoll JF, Reiss C, Rutherford JD, Cohn LM. A case of neurofibromatosis associated with a coronary artery aneurysm and myocardial infarction. Cardiovasc Intervent Radiol. 1988;11(3):143–5.
Evrengul H, Kilic DI, Zungur M, Alihanoglu YI, Tanriverdi H. Myocardial infarction in a 17-year-old patient due to neurofibromatosis-associated coronary aneurysm. Cardiol Young. 2013;23(3):454–6. https://doi.org/10.1017/S1047951112000984.
Halper J, Factor SM. Coronary lesions in neurofibromatosis associated with vasospasm and myocardial infarction. Am Heart J. 1984;108(2):420–2.
Downward J. Cell cycle: routine role for Ras. Curr Biol. 1997;7(4):R258–60.
Brown JA, Gianino SM, Gutmann DH. Defective cAMP generation underlies the sensitivity of CNS neurons to neurofibromatosis-1 heterozygosity. J Neurosci. 2010;30(16):5579–89. https://doi.org/10.1523/JNEUROSCI.3994-09.2010.
The I, Hannigan GE, Cowley GS, Reginald S, Zhong Y, Gusella JF, et al. Rescue of a Drosophila NF1 mutant phenotype by protein kinase a. Science. 1997;276(5313):791–4.
Wolman MA, de Groh ED, McBride SM, Jongens TA, Granato M, Epstein JA. Modulation of cAMP and ras signaling pathways improves distinct behavioral deficits in a zebrafish model of neurofibromatosis type 1. Cell Rep. 2014;8(5):1265–70. https://doi.org/10.1016/j.celrep.2014.07.054.
Boyanapalli M, Lahoud OB, Messiaen L, Kim B, Anderle de Sylor MS, Duckett SJ, et al. Neurofibromin binds to caveolin-1 and regulates ras, FAK, and Akt. Biochem Biophys Res Commun. 2006;340(4):1200–8. https://doi.org/10.1016/j.bbrc.2005.12.129.
Hsueh YP, Roberts AM, Volta M, Sheng M, Roberts RG. Bipartite interaction between neurofibromatosis type I protein (neurofibromin) and syndecan transmembrane heparan sulfate proteoglycans. J Neurosci. 2001;21(11):3764–70.
Viskochil D, Buchberg AM, Xu G, Cawthon RM, Stevens J, Wolff RK, et al. Deletions and a translocation interrupt a cloned gene at the neurofibromatosis type 1 locus. Cell. 1990;62(1):187–92.
Jacks T, Shih TS, Schmitt EM, Bronson RT, Bernards A, Weinberg RA. Tumour predisposition in mice heterozygous for a targeted mutation in Nf1. Nat Genet. 1994;7(3):353–61. https://doi.org/10.1038/ng0794-353.
Gitler AD, Epstein JA. Regulating heart development: the role of Nf1. Cell Cycle. 2003;2(2):96–8.
Ismat FA, Xu J, Lu MM, Epstein JA. The neurofibromin GAP-related domain rescues endothelial but not neural crest development in Nf1 mice. J Clin Invest. 2006;116(9):2378–84. https://doi.org/10.1172/JCI28341.
Xu GF, Lin B, Tanaka K, Dunn D, Wood D, Gesteland R, et al. The catalytic domain of the neurofibromatosis type 1 gene product stimulates ras GTPase and complements ira mutants of S. cerevisiae. Cell. 1990;63(4):835–41.
Padmanabhan A, Lee JS, Ismat FA, Lu MM, Lawson ND, Kanki JP, et al. Cardiac and vascular functions of the zebrafish orthologues of the type I neurofibromatosis gene NFI. Proc Natl Acad Sci U S A. 2009;106(52):22305–10. https://doi.org/10.1073/pnas.0901932106.
Hutson MR, Kirby ML. Neural crest and cardiovascular development: a 20-year perspective. Birth Defects Res C Embryo Today. 2003;69(1):2–13. https://doi.org/10.1002/bdrc.10002.
Yzaguirre AD, Padmanabhan A, de Groh ED, Engleka KA, Li J, Speck NA, et al. Loss of neurofibromin Ras-GAP activity enhances the formation of cardiac blood islands in murine embryos. elife. 2015;4:e07780. https://doi.org/10.7554/eLife.07780.
Xu J, Ismat FA, Wang T, Lu MM, Antonucci N, Epstein JA. Cardiomyocyte-specific loss of neurofibromin promotes cardiac hypertrophy and dysfunction. Circ Res. 2009;105(3):304–11. https://doi.org/10.1161/CIRCRESAHA.109.201509.
Schubbert S, Zenker M, Rowe SL, Boll S, Klein C, Bollag G, et al. Germline KRAS mutations cause Noonan syndrome. Nat Genet. 2006;38(3):331–6. https://doi.org/10.1038/ng1748.
Pandit B, Sarkozy A, Pennacchio LA, Carta C, Oishi K, Martinelli S, et al. Gain-of-function RAF1 mutations cause Noonan and LEOPARD syndromes with hypertrophic cardiomyopathy. Nat Genet. 2007;39(8):1007–12. https://doi.org/10.1038/ng2073.
Quezada E, Gripp KW. Costello syndrome and related disorders. Curr Opin Pediatr. 2007;19(6):636–44. https://doi.org/10.1097/MOP.0b013e3282f161dc.
Araki T, Mohi MG, Ismat FA, Bronson RT, Williams IR, Kutok JL, et al. Mouse model of Noonan syndrome reveals cell type- and gene dosage-dependent effects of Ptpn11 mutation. Nat Med. 2004;10(8):849–57. https://doi.org/10.1038/nm1084.
Arrington DK, Danehy AR, Peleggi A, Proctor MR, Irons MB, Ullrich NJ. Calvarial defects and skeletal dysplasia in patients with neurofibromatosis type 1. J Neurosurg Pediatr. 2013;11(4):410–6. https://doi.org/10.3171/2013.1.peds12409.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Bergqvist, C., Wolkenstein, P. (2021). The Heart in Neurofibromatosis 1. In: Salavastru, C., Murrell, D.F., Otton, J. (eds) Skin and the Heart. Springer, Cham. https://doi.org/10.1007/978-3-030-54779-0_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-54779-0_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-54778-3
Online ISBN: 978-3-030-54779-0
eBook Packages: MedicineMedicine (R0)