Abstract
Cavernous sinus hemangiomas (CSHs) are rare benign vascular tumors. Stereotactic radiosurgery (SRS) has emerged as a principal alternative to microsurgery for small- and medium-sized CSHs. Resection is a reasonable option for large (3–4 cm in diameter) and giant (>4 cm in diameter) CSHs. However, the management of large and giant CSHs remains a challenge for neurosurgeons because of the critical structures in the cavernous sinus and massive hemorrhage during surgery. Multisession CyberKnife radiosurgery provides a good tumor control for large CSHs. Morbidity after SRS or multisession CyberKnife radiosurgery is much less than that after microsurgery for patients with CSHs.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Cavernous sinus hemangiomas are an extremely rare benign tumor that have been reported to account for less than 3% of all benign tumors occurring in the cavernous sinus and 2% of all tumors, both benign and malignant [1,2,3]. Cavernous sinus hemangiomas may present with a variety of neurological features, for example, headache, seizure, hemorrhage, or neurological deficits. The incidence is higher in females in the fifth and sixth decade of their lives. Cavernous sinus hemangiomas can be diagnosed by their characteristic imaging appearance. The optimum treatment strategy is still controversial. Current treatment modalities for symptomatic cavernous sinus hemangiomas include microsurgical resection, embolization, fractionated radiation therapy, and stereotactic radiosurgery. Complete resection of cavernous sinus hemangioma is potentially curative but may be complicated by severe intraoperative hemorrhage and the complicated neurovascular structures. The reported incidences of postoperative complications have varied from 8 to 80%, whereas complete excision can be accomplished in only 30–60% of cases [4,5,6,7,8,9,10,11,12,13,14,15]. Thus, treatment of these lesions remains a therapeutic challenge. Because of the high possibility of profuse bleeding during surgical intervention combined with potential for long-term cranial nerve deficits, some practitioners have focused on radiotherapy and stereotactic radiosurgery for treatment of these lesions. Patients with cavernous sinus hemangiomas often respond well to treatment involving ionizing radiation [16,17,18,19]. In 1999, Iwai et al. reported the first cavernous sinus hemangioma case treated with Gamma Knife radiosurgery. The tumor had previously been partially removed, but removal was complicated by severe bleeding. Radiosurgery was performed as adjuvant therapy and dramatically decreased tumor size [20]. Since then, more reports have been published, all showing good response. In most reports, radiosurgery is used as adjuvant therapy after partial tumor removal. Radiosurgery has also been used as the primary treatment modality with good results for patients who had small- to medium-sized CSHs [21,22,23,24,25,26,27,28,29,30,31,32].
However, the lesion size and location make single-fraction radiosurgery for large CS hemangiomas very challenging. Although the targeting accuracy and dose fall-off of radiosurgery are excellent, single-fraction radiosurgery may not be ideal for large or giant tumors adjacent to optic pathways [33, 34]. As the target lesion size increases, so does the area of normal brain and other critical anatomic structures, such as optical nerves, that is irradiated, thereby increasing the risk of radiation complications [35].
Recently, multisession CyberKnife radiosurgery has been shown to be effective, even in control of large tumors, with excellent functional preservation [36,37,38]. The CyberKnife system (Accuray, Sunnyvale, CA) allows convenient dose fractionation with a frameless delivery that can comfortably treat patients on consecutive days in the outpatient setting without reacquisition of imaging or reapplication of rigid head frame fixation. To achieve favorable local control while maintaining low optic pathway toxicity for large CSHs, we started protocol-based multisession stereotactic radiosurgery with the CyberKnife system for large CSHs in 2007 [36]. This article addresses the safety and efficacy of Gamma Knife radiosurgery and multi-session CyberKnife radiosurgery for CSHs.
2 Presentations
Cavernous sinus hemangiomas have an overwhelming predilection for women. In our center, of the 186 cases, 42 (22.5%) have occurred in men, and 144 (77.5%) have occurred in women. Mean age at diagnosis was 50 years (range, 22–80 years). Clinical signs and symptoms and clinical tumor course cannot distinguish cavernous sinus hemangiomas from other cavernous sinus neoplasms. The most common symptoms in these patients were cranial neuropathy including diplopia, blurred vision, facial numbness, and abducens palsy. The second most common symptoms were headache and dizziness. Less frequent signs or symptoms include endocrinopathy, exophthalmos, trigeminal neuralgia, and nausea, vomiting, and gait instability when the tumor was giant. About 5% of cases were an asymptomatic incidental MRI examination finding. Duration of symptoms was from 1 week to 10 years.
3 Radiographic Appearance
Cavernous sinus hemangiomas characteristically are not calcified and demonstrate bony erosion or remodeling rather than hyperostosis. The MR images obtained in these patients showed well demarcated, low- to iso-signal mass on T1-weighted images, extremely high signal on T2-weighted images (as bright as cerebrospinal fluid signal), high signal on FLAIR-weighted (fluid attenuated inversion recovery) images, and strong homogeneous or heterogeneous enhancement after Gd-DTPA injection. When the CSHs were small to medium-sized tumors, they usually showed sharply delineated and intensely enhanced sellar masses without “dural tail sign.” But when the CSHs were large or giant in size, some tumors showed heterogeneous enhancement and delayed homogeneous enhancement [40,41,42,43,44]. We found the CSHs have no metabolism on 18F- FDG PET CT. That means the SUV value was lower (very low) compared with normal brain tissue. The radiological characteristics of a representative case of CSH are shown in Fig. 46.1.
MR images of a representative case showing radiological characteristics of CSH. A 50-year-old male patient suffered from slight headache and went for an MR scan, which showed a sharply delineated iso- or hypo-signal mass lesion on T1-weighted images (a), extremely high signal on T2-weighted images (b) and FLAIR sequence images (c), and intensely enhanced sellar mass (d). He had an operation for the cavernous sinus hemangioma and CSH was confirmed by histopathology. 18-F FDG PET-CT showed that CSH had no metabolism with lower SUV (e). During the operation only a small piece of tumor was removed because of bleeding. Then the patient was treated with CyberKnife radiosurgery. The CSH reduced in volume at 6 months post CyberKnife (f)
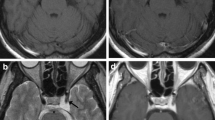
For patients with atypical manifestations, digital subtraction angiography (DSA) was performed to differentiate CSH from meningioma, neurilemmoma, and other cavernous sinus tumors. In patients with CSH, there is no tumor staining on the cerebral angiography with internal carotid artery DSA. But there is little flecked tumor enhancement on external carotid artery DSA. No intracavernous carotid artery stenosis was ever seen despite the complete involvement of the cavernous sinus in these patients (Fig. 46.2).
A 32-year-old female had headache and vomiting and MRI examination was performed. The MRI showed low- to iso-signal giant mass on T1-weighted images (a), extremely high signal on T2-weighted images (as bright as cerebrospinal fluid signal) (b), high signal on FLAIR-weighted (fluid attenuated inversion recovery) images (c) and strong homogeneous or heterogeneous enhancement after Gd-DTPA injection (d). The DSA demonstrates elevation of the left middle cerebral artery and medial deviation of the left internal carotid artery (e). No tumor staining was seen on artery phase angiography although the CSH is a hypervascular lesion by nature (f). The patient had an operation and CSH was confirmed by histopathology
4 Therapeutic Option
Treatment options include the wait-and-see approach with serial images, microsurgery, single-fraction SRS and multisession SRS. If patients have asymptomatic small-sized tumors, a wait-and-see approach with serial images may be a reasonable treatment option, because CSHs are generally slow-growing tumors. Microsurgery was a common treatment before the advent of SRS. Complete resection is an ideal treatment, but it is not so easily achievable without any complications. Although recently skull base surgery has contributed to an increased rate of complete resection, there is no doubt that this is one of the most invasive treatments. SRS is a less invasive treatment option for small- to medium-sized CSHs, and multisession CyberKnife radiosurgery is a treatment option for large or giant CSHs, with good tumor control as well as improving neurological deficits.
5 Clinical Outcomes
Contemporary series of Gamma Knife radiosurgery and multisession CyberKnife radiosurgery for CSHs are shown in Table 46.1.
Our colleague Dr. Wang published a systematic review and meta-analysis of 59 cases of CSHs treated with Gamma Knife radiosurgery (GKR) [46]. The results suggest that GKR not only achieves good tumor control and symptom improvement but also avoids the complications associated with embolization, biopsy, and attempted microsurgical resection. However, most of the patients underwent GKR as an adjuvant treatment after open surgery, and the number of cases using GKR as a primary treatment was limited.
At the author’s institution, 53 patients harboring CSHs were treated using Leksell Gamma Knife radiosurgery. Of the 53 patients, 15 with definitive histopathologic diagnoses after surgery, 38 were diagnosed based on their MR imaging findings. There were 15 male and 38 female patients with a mean age of 52 (range 25–76) years old. The mean volume of the tumors was 13.2 ± 8.2 cm3 (range 1–41 cm3). A mean marginal dose of 13.3 Gy (range 8–15 Gy) was directed to the 49–64% isodose line (mean 53%). The mean radiological and clinical follow-up time of this study was 24 (range 2–67 months) and 34 months (range 2–73 months), respectively. The tumor control rate was 100%. The mean tumor volume reduction was 79.5% (range 16.5–100%) compared with the pre-GKR volume. A typical case of CSH is shown in Fig. 46.3. Neurologically, only two of these patients showed clinical deterioration, and the other 51 patients demonstrated an obvious improvement in symptoms.
Similarly, Lee and colleagues reported the results of GKR in 31 patients with CSHs. The median radiosurgery target volume was 9.3 cm3 and the median marginal dose was 12.6 Gy. At a mean follow-up period of 54 months (range 6–200 months), the tumor control rate was 100%. The average tumor reduction at 12 months was 64%; at 24 months, 73%; at 36 months, 79%; at 48 months, 82%; and at 60 months, 84%. No recurrence was found.
6 Multi-session CyberKnife Radiosurgery
We first reported the effectiveness and safety of multisession CyberKnife radiosurgery for controlling large CSHs. A Phase II study to substantiate the role of CyberKnife radiosurgery was conducted in our department in which we evaluated patients with a large CSH that had a clear “geographic” separation between the tumor and the optic apparatus on MRI. We found that a multi-fraction SRS dose of 21 Gy delivered in three fractions was effective in reducing the tumor volume without causing any new neurological deficits [36].
7 Rationale for Multisession CyberKnife Radiosurgery
Our dose not only was based on our and others’ previous experience with SRS and radiotherapy, but also previous studies concerning the dose tolerance of the optic apparatus and brainstem. Although the exact α/β ratio that corresponds to CSHs has not been established, we assumed a value of 3 Gy and used the linear-quadratic model to estimate the biologically equivalent dose to a single-dose SRS of 10–13 Gy (a dose that yields remarkable tumor shrinkage for CSHs after radiosurgery) for a 3- or 4-fraction course of CyberKnife SRS (listed in Table 46.2). The dose prescription was based on intent to cover the entire tumor with a higher dose while ensuring dose limitation to the visual pathways and brainstem. General guidelines for dose limitations to normal structures included the following. The maximum permissible point dose to the optic nerves and chiasm was 14.1 Gy in 3 fractions (4.7 Gy per fraction) or 15.6 Gy in 4 fractions (3.9 Gy per fraction), similar to a single-fraction SRS of 9 Gy. The maximum permissible point dose to the brainstem was 21 Gy in 3 fractions (7 Gy per fraction) or 23.6 Gy in 4 fractions (5.9 Gy per fraction), similar to a single-session SRS dose of 13 Gy.
Thirty-one patients harboring giant CSHs were treated with multisession CyberKnife radiosurgery at the author’s institution, among whom 27 underwent multisession CyberKnife radiosurgery as the initial treatment. Four patients had a histological diagnosis at the time of a prior resection. In those four patients, three underwent a biopsy and one underwent partial resection. There were 8 male and 23 female patients with a mean age of 54 (range 22–80) years old. The median CSH volume was 64.4 cm3 (range 40.9–145.3 cm3). Three or four sessions of CyberKnife radiosurgery were used with a prescription dose based on the intent to cover the entire tumor with a higher dose while ensuring dose limitation to the visual pathways and brainstem. The median marginal dose to the tumor was 21 Gy (range 19.5–21 Gy) in 3 fractions for 11 patients and 22 Gy (range 18–22 Gy) in 4 fractions for 20 patients. During a median follow-up of 30 months (range 6–78 months), all patients achieved good tumor control. Follow-up MRI scans revealed a median tumor volume reduction of 88.1% (62.3–99.4%) at last examination compared with the pretreatment volume. Ten patients developed new or aggravated temporary headache, and 5 experienced vomiting during the treatment. These acute symptoms were relieved completely after steroid administration. Among the 30 patients with symptoms observed before treatment, 19 achieved complete symptomatic remission, and 11 had partial remission. One patient reported seizures, which were controlled after antiepileptic drug administration. No radiation-induced neurological deficits or delayed complications were reported during the follow-up period [38]. A typical case of giant CSH is shown in Fig. 46.4.
A 43-year old woman had a visual field defect, blurred vision, and ipsilateral abducens nerve palsy. MRI found a cavernous sinus hemangioma on the right side in 1998 (a). The patient underwent resection and experienced severe intraoperative bleeding. Only a piece of tumor was removed during the operation. The tumor progressed slowly (b) and in 2011 the tumor became a giant tumor (c). The pretreatment tumor volume was 76.5 cm3 (d). Three and half years post multisession CyberKnife radiosurgery (21 Gy in 3 fractions), the tumor shrank to 2 cm3 (e, f). Clinical symptoms also improved
Huang and colleagues documented 12 patients with high volume cavernous sinus hemangiomas treated with hypofractionated stereotactic CyberKnife [37]. Initial tumor volumes ranged from 11.8 to 96.6 cm3 with a median of 24.3 cm3. Irradiation doses were 19.5 Gy with 3 fractions in 2 patients, 21 Gy with 3 fractions in 8 patients, 25 Gy with 5 fractions in 1 patient, and 30 Gy with 3 fractions in 1 patient. Follow-up ranged from 3 to 54 months, with a mean follow-up of 16.3 months. All tumor volumes decreased (28.6–94.1%) and symptoms improved (including blurred vision, visual field defects, diplopia, headaches, and facial numbness) after therapy. A patient experienced radiotherapy-related cerebral edema, which resolved after 5 days of mannitol and dexamethasone.
8 Summary
Gamma Knife radiosurgery is a safe and effective treatment for patients with small- to medium-sized cavernous sinus hemangiomas. When the tumor is relatively large (e.g., compressing the optic nerves or chiasm), multisession CyberKnife radiosurgery would be an alternative treatment option. Multisession CyberKnife radiosurgery delivery of 18–22 Gy in 3–4 fractions is effective in reducing tumor volume and improving neurological symptoms without causing any new complications. Until now microsurgery has been a common treatment option for cavernous sinus tumors, but, even with recent refinement of microsurgical techniques, it is unable to avoid the risk of complications such as cranial nerve injury and avoid partial resection of the tumors. Accordingly, single-fraction or multisession stereotactic radiosurgery can be a reasonable alternative to surgical resection not only as adjuvant treatment but also as the initial treatment, with respect to preservation of cranial nerve function.
References
Linskey ME, Sekhar LN. Cavernous sinus hemangiomas: a series, a review, and an hypothesis. Neurosurgery. 1992;30(1):101–8.
Gonzalez LF, Lekovic GP, Eschbacher J, Coons S, Porter RW, Spetzler RF. Are cavernous sinus hemangiomas and cavernous malformations different entities? Neurosurg Focus. 2006;21(1):e6.
Gliemroth J, Missler U, Sepehrnia A. Cavernous angioma as a rare neuroradiologic finding in the cavernous sinus. J Clin Neurosci. 2000;7(6):554–7.
Zhou LF, Mao Y, Chen L. Diagnosis and surgical treatment of cavernous sinus hemangiomas: an experience of 20 cases. Surg Neurol. 2003;60(1):31–6.
Shi J, Hang C, Pan Y, Liu C, Zhang Z. Cavernous hemangiomas in the cavernous sinus. Neurosurgery. 1999;45(6):1308–13; discussion 1313–1314.
Ohata K, El-Naggar A, Takami T, Morino M, El-Adawy Y, El-Sheik K, Inoue Y, Hakuba A. Efficacy of induced hypotension in the surgical treatment of large cavernous sinus cavernomas. J Neurosurg. 1999;90(4):702–8.
Bansal S, Suri A, Singh M, Kale SS, Agarwal D, Sharma MS, Mahapatra AK, Sharma BS. Cavernous sinus hemangioma: a fourteen year single institution experience. J Clin Neurosci. 2014;21(6):968–74.
Cure GC, Mejía JA, Roldan NG. Giant cavernous hemangioma of the cavernous sinus. Case report and review of literature. Rev Chil Neurocirugía. 2010;34:66–71.
Dou Y, Meng Q, Yan Z, Xu J, Che S, Jiao Y, Wu Z. Diagnosis and microsurgical treatment of cavernous sinus hemangioma. Artif Cells Blood Substit Immobil Biotechnol. 2010;38(2):109–12.
Fraser JF, Mass AY, Brown S, Anand VK, Schwartz TH. Transnasal endoscopic resection of a cavernous sinus hemangioma: technical note and review of the literature. Skull Base. 2008;18(5):309–15.
Goel A, Muzumdar D, Sharma P. Extradural approach for cavernous hemangioma of the cavernous sinus: experience with 13 cases. Neurol Med Chir (Tokyo). 2003;43(3):112–8; discussion 119.
Suzuki Y, Shibuya M, Baskaya MK, Takakura S, Yamamoto M, Saito K, Glazier SS, Sugita K. Extracerebral cavernous angiomas of the cavernous sinus in the middle fossa. Surg Neurol. 1996;45(2):123–32.
Suri A, Ahmad FU, Mahapatra AK. Extradural transcavernous approach to cavernous sinus hemangiomas. Neurosurgery. 2007;60(3):483–8; discussion 488–489.
Hashimoto M, Yokota A, Ohta H, Urasaki E. Intratumoral injection of plastic adhesive material for removal of cavernous sinus hemangioma. Technical note. J Neurosurg. 2000;93(6):1078–81.
Kim IM, Yim MB, Lee CY, Son EI, Kim DW, Kim SP, Sohn CH. Merits of intralesional fibrin glue injection in surgery for cavernous sinus cavernous hemangiomas. Technical note. J Neurosurg. 2002;97(3):718–21.
Jamjoom AB. Response of cavernous sinus hemangioma to radiotherapy: a case report. Neurosurg Rev. 1996;19(4):261–4.
Yamamoto Y, Weining Z, Ohashi T. Intracavernous cavernous hemangioma: dynamic CT findings and effectiveness of irradiation—case report. Neurol Med Chir (Tokyo). 1992;32(2):93–5.
Maruishi M, Shima T, Okada Y, Nishida M, Yamane K, Okita S. Cavernous sinus cavernoma treated with radiation therapy—case report. Neurol Med Chir (Tokyo). 1994;34(11):773–7.
Miserocchi G, Vaiani S, Migliore MM, Villani RM. Cavernous hemangioma of the cavernous sinus. Complete disappearance of the neoplasma after subtotal excision and radiation therapy. Case report. J Neurosurg Sci. 1997;41(2):203–7.
Iwai Y, Yamanaka K, Nakajima H, Yasui T. Stereotactic radiosurgery for cavernous sinus cavernous hemangioma: case report. Neurol Med Chir (Tokyo). 1999;39(4):288–90.
Seo Y, Fukuoka S, Sasaki T, Takanashi M, Hojo A, Nakamura H. Cavernous sinus hemangioma treated with gamma knife radiosurgery: usefulness of SPECT for diagnosis: case report. Neurol Med Chir (Tokyo). 2000;40(11):575–80.
Thompson TP, Lunsford LD, Flickinger JC. Radiosurgery for hemangiomas of the cavernous sinus and orbit: technical case report. Neurosurgery. 2000;47(3):778–83.
Kida Y, Kobayashi T, Mori Y. Radiosurgery of cavernous hemangiomas in the cavernous sinus. Surg Neurol. 2001;56(2):117–22.
Nakamura N, Shin M, Tago M, Terahara A, Kurita H, Nakagawa K, Ohtomo K. Gamma knife radiosurgery for cavernous hemangiomas in the cavernous sinus. Report of three cases. J Neurosurg. 2002;7(5):477–80.
Peker S, Kilic T, Sengoz M, Pamir MN. Radiosurgical treatment of cavernous sinus cavernous hemangiomas. Acta Neurochir. 2004;146:337–41.
Ivanov P, Chernov M, Hayashi M, Nakaya K, Izawa M, Murata N, Kubo O, Ujiie H, Muragaki Y, Nakamura R, Iseki H, Hori T, Takakura K. Low-dose gamma knife radiosurgery for cavernous sinus hemangioma: report of 3 cases and literature review. Minim Invasive Neurosurg. 2008;51(3):140–6.
Khan AA, Niranjan A, Kano H, Kondziolka D, Flickinger JC, Lunsford LD. Stereotactic radiosurgery for cavernous sinus or orbital hemangiomas. Neurosurgery. 2009;65(5):914–8.
Chou CW, Wu HM, Huang CI, Chung WY, Guo WY, Shih YH, Lee LS, Pan DH. Gamma knife surgery for cavernous hemangiomas in the cavernous sinus. Neurosurgery. 2010;67(3):611–6.
Yamamoto M, Kida Y, Fukuoka S, Iwai Y, Jokura H, Akabane A, Serizawa T. Gamma knife radiosurgery for hemangiomas of the cavernous sinus: a seven-institute study in Japan. J Neurosurg. 2010;112(4):772–9.
Li P, Ren H, Zhang S, Wang W. Clinical results of Gamma Knife surgery for cavernous sinus hemangiomas. J Neurosurg. 2012;117 Suppl:89–95.
Song SW, Kim DG, Chung HT, Paek SH, Han JH, Kim YH, Kim JW, Kim YH, Jung HW. Stereotactic radiosurgery for cavernous sinus hemangiomas. J Neurooncol. 2014;118(1):163–8.
Lee CC, Sheehan JP, Kano H, Akpinar B, Martinez-Alvarez R, Martinez-Moreno N, Guo WY, Lunsford LD, Liu KD. Gamma knife radiosurgery for hemangioma of the cavernous sinus. J Neurosurg. 2017;126(5):1498–505.
Liu AL, Wang C, Sun S, Wang M, Liu P. Gamma knife radiosurgery for tumors involving the cavernous sinus. Stereotact Funct Neurosurg. 2005;83(1):45–51.
Kuo JS, Chen JC, Yu C, Zelman V, Giannotta SL, Petrovich Z, MacPherson D, Apuzzo ML. Gamma knife radiosurgery for benign cavernous sinus tumors: quantitative analysis of treatment outcomes. Neurosurgery. 2004;54(6):1385–93; discussion 1393–1394.
Mayo C, Martel MK, Marks LB, Flickinger J, Nam J, Kirkpatrick J. Radiation dose-volume effects of optic nerves and chiasm. Int J Radiat Oncol Biol Phys. 2010;76(3):S28–35. J Neurooncol (2012) 107:239–245.
Wang X, Liu X, Mei G, Dai J, Pan L, Wang E. Phase II study to assess the efficacy of hypofractionated stereotactic radiotherapy in patients with large cavernous sinus hemangiomas. Int J Radiat Oncol Biol Phys. 2012;83(2):e223–30.
Huang L, Sun L, Wang W, Cui Z, Zhang Z, Li J, Wang Y, Wang J, Yu X, Ling Z, Qu B. Therapeutic effect of hypofractionated stereotactic radiotherapy using CyberKnife for high volume cavernous sinus cavernous hemangiomas. Technol Cancer Res Treat. 2019;18:1–7.
Wang X, Zhu H, Knisely J, Mei G, Liu X, Dai J, Mao Y, Pan L, Qin Z, Wang E. Hypofractionated stereotactic radiosurgery: a new treatment strategy for giant cavernous sinus hemangiomas. J Neurosurg. 2018;128(1):60–7.
Park CK, Choi SK, Kang IH, Choi MK, Park BJ, Lim YJ. Radiosurgical considerations for cavernous sinus hemangioma: long-term clinical outcomes. Acta Neurochir. 2016;158(2):313–8.
Jinhu Y, Jianping D, Xin L, Yuanli Z. Dynamic enhancement features of cavernous sinus cavernous hemangiomas on conventional contrast-enhanced MR imaging. AJNR Am J Neuroradiol. 2008;29(3):577–81.
Mendonça JL, Viana SL, Matsumine M, Silva RF, Viana MA, Freitas FM. Cavernous angioma of the cavernous sinus: imaging findings. Arq Neuropsiquiatr. 2004;62(4):1004–7.
Salanitri GC, Stuckey SL, Murphy M. Extracerebral cavernous hemangioma of the cavernous sinus: diagnosis with MR imaging and labeled red cell blood pool scintigraphy. AJNR Am J Neuroradiol. 2004;25(2):280–4.
Sohn CH, Kim SP, Kim IM, Lee JH, Lee HK. Characteristic MR imaging findings of cavernous hemangiomas in the cavernous sinus. AJNR Am J Neuroradiol. 2003;24(6):1148–51.
Tannouri F, Divano L, Caucheteur V, Hacourt A, Pirotte B, Salmon I, Balériaux D. Cavernous haemangioma in the cavernous sinus: case report and review of the literature. Neuroradiology. 2001;43(4):317–20.
Xu Q, Shen J, Feng Y, Zhan R. Gamma knife radiosurgery for the treatment of cavernous sinus hemangiomas. Oncol Lett. 2016;11(2):1545–8.
Tang X, Wu H, Wang B, Zhang N, Dong Y, Ding J, Dai J, Yu T, Pan L. A new classification and clinical results of gamma knife radiosurgery for cavernous sinus hemangiomas: a report of 53 cases. Acta Neurochir. 2015;157(6):961–9.
Anqi X, Zhang S, Jiahe X, Chao Y. Cavernous sinus cavernous hemangioma: imaging features and therapeutic effect of gamma knife radiosurgery. Clin Neurol Neurosurg. 2014;127:59–64.
Wang Y, Li P, Zhang XJ, Xu YY, Wang W. Gamma Knife surgery for cavernous sinus hemanginoma: a report of 32 cases. World Neurosurg. 2016;94:18–25.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Wang, E. (2020). Cavernous Sinus Hemangioma. In: Conti, A., Romanelli, P., Pantelis, E., Soltys, S., Cho, Y., Lim, M. (eds) CyberKnife NeuroRadiosurgery . Springer, Cham. https://doi.org/10.1007/978-3-030-50668-1_46
Download citation
DOI: https://doi.org/10.1007/978-3-030-50668-1_46
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-50667-4
Online ISBN: 978-3-030-50668-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)